Abstract
Human melanocytes are not simply pigment-producing cells. It may be part of the inflammatory response, during which the pigmentary system may produce more melanin or suppress melanization. Toll-like receptors (TLRs) have been implicated in both innate host defense against pathogens and inflammatory response. Therefore, it may be possible that activation of TLRs in melanocytes may play a role in the modulation of melanogenesis. In this study, we investigated whether normal human melanocytes expressed TLRs and analyzed pigmentation changes upon TLR stimulation. The expression of TLR1~10 mRNA in cultured human melanocyte was analyzed using RT-PCR, Western blotting and immunocytochemistry. Human melanocytes constitutively express mRNA and protein for TLR2, 3, 4, 5, 7, 9 and 10. Stimulation of TLR1/2 and 4 with Pam3CSK4 and lipopolysaccharide induced pigmentation of melanocytes. Activation of TLR5 and 7 with flagellin and imiquimod treatments reduced pigmentation of melanocytes and zebrafish. In summary, the results provided evidence for TLRs expression in normal human melanocytes. It is speculated that a response of melanocyte to TLR ligands may play a role in the pigmentary change in the skin.
Keywords: Melanocytes, Pigmentation, Toll-like receptors
Human melanocytes are not merely pigment-producing cells. They also act as phagocytic cells that contribute to inflammatory responses1. Indeed, there exists evidence that human melanocytes and melanin inhibit proliferation of bacterial, fungal, and parasitic infections in the epidermis and dermis. For example, radicals and other compounds produced during melanogenesis are believed to exert strong antimicrobial activity2.
Toll-like receptors (TLRs) are part of the innate immune system involved in the response to microbial infections. At least 10 members of the TLR family have been identified in mammals3. TLRs recognize a wide range of microbial ligands, including lipopolysaccharides (LPS), bacterial lipoproteins, bacterial heat shock proteins, viral single- and double-stranded RNA, and bacterial flagellin. The innate immune system governs the interconnecting pathways of microbial recognition, inflammation, microbial clearance, and cell death3. The pigmentary system may produce more melanin, or suppress melanization, in response to inflammatory events. It is therefore possible that TLR activation in melanocytes may play a role in pigmentation modulation. The expression of TLR4 and TLR7, and the effects of their ligands, LPS or imiquimod in melanocyte biology have been shown previously4,5. It has also been reported that TLR expression in human melanocytes could play a role in the local innate inflammatory response in the epidermis6.
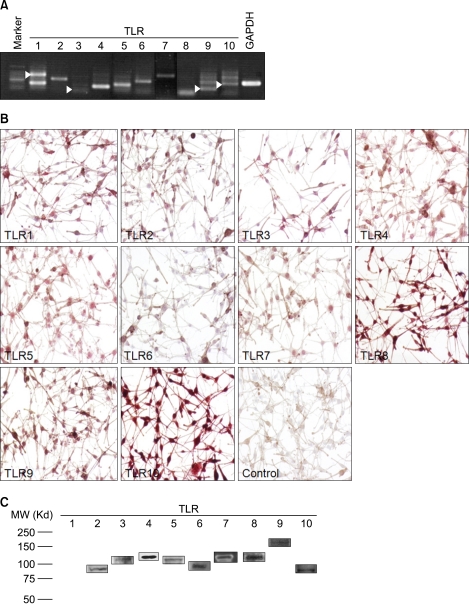
The objective of this study was to investigate whether normal human melanocytes express all TLR members, and analyze pigmentation changes upon TLR stimulation. To investigate whether TLRs 1~10 are constitutively expressed in cultured normal human melanocytes, we performed RT-PCR, immunocytochemistry and Western blot analysis. The primers sequences used were as follows: TLR1 sense oligonucleotide, 5'-AAAAGAAGACCCTGAGGGCC-3' and anti-sense oligonucleotide, 5'-TCTGAAGTCCAGCTGACCCT-3'; TLR2 sense oligonucleotide, 5'-AACCCTAGGGGAAACATCTCT-3', and antisense oligonucleotide, 5'-GGAATATGCAGCCTCCGGAT-3'; TLR3 sense oligonucleotide, 5'-AAATTGGGCAAGAACTCACAGG-3'; and antisense oligonucleotide, 5'-GTGTTTCCAGAGCCGTGCTAA-3'; TLR4 sense oligonucleotide, 5'-TACAAAATCCCCGACAACCTC-3'; and antisense oligonucleotide, 5'-AGCCACCAGCTTCTGTAAACT-3'; TLR5 sense oligonucleotide, 5'-GCATTAAGGGGACTAAGCCTC-3', and antisense oligonucleotide, 5'-AAAAGGGAGAACTTTAGGGACT-3'; TLR6 sense oligonucleotide, 5'-TTGACAGTTTTGAGACTTTCCC-3'; and antisense oligonucleotide, 5'-TGGACCTCTGGTGAGTCCTG-3'; TLR7 sense oligonucleotide, 5'-TCCAGTGTCTAAAGAACCTGG-3'; and antisense oligonucleotide, 5'-TGGTAAATATACCACACATCCC-3'; TLR8 sense oligonucleotide, 5'-TAATAGGCTGAAGCACATCCC-3'; and antisense oligonucleotide, 5'-TCCCAGTAAAACAAATGGTGAG-3'; TLR9 sense oligonucleotide, 5'-GTGCCCCACTTCTCCATG-3'; and antisense oligonucleotide, 5'-GGCACAGTCATGATGTTGTTG-3'; TLR10 sense oligonucleotide, 5'-TGACCACAATTCATTTGACTACTC-3'; and antisense oligonucleotide, 5'-TTGAATACTTTTGGGCAAGCACC-3'. The RT-PCR studies indicated that human melanocytes constitutively expressed mRNA for TLR1, 2, 3, 4, 5, 6, 7, 9, and 10, but not for TLR8 (Fig. 1A). Immunocytochemical staining showed immunoreactivity for TLR1 (1:50 dilution; Santa Cruze Biotechnology, Santa Cruz , CA, USA), 2 (1:50; Santa Cruz Biotechnology), 3 (1:50; Santa Cruz Biotechnology), 4 (1:30; Abcam, Cambridge, MA, USA), 5 (1:50; Santa Cruz Biotechnology), 7 (1:30; Abcam), 9 (1:50; Santa Cruz Biotechnology), and 10 (1:50; Santa Cruz Biotechnology), but not for TLR6 (1:50; Santa Cruz Biotechnology) in human melanocytes (Fig. 1B). The expression of TLRs 2~10 protein was further confirmed by Western blot analysis (Fig. 1C). Cultured human melanocytes constitutively express mRNA and protein for TLR2, 3, 4, 5, 7, 9, and 10.
Fig. 1.
Toll-like receptors (TLRs) are expressed in human melanocytes. (A) Expression of TLR1-10 was assessed by RT-PCR. The protein expression was detected by immunocytochemistry (B: ×200) and Western blotting (C). Negative controls were made by applying normal rabbit serum before treatment with primary antibody.
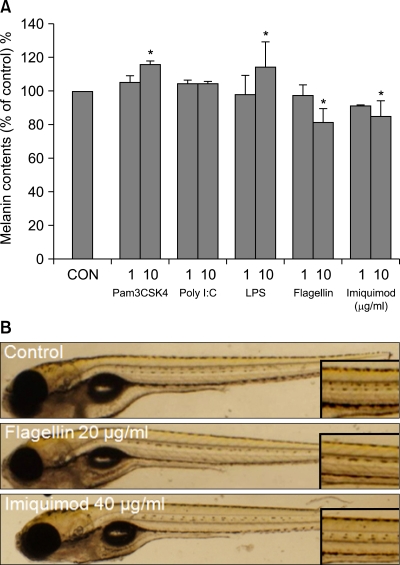
To investigate the functional role of TLRs in pigmentation, melanocytes were treated with Pam3CSK4 (InvivoGen, San Diego, CA, USA), polyinosinic-polycytidylic acid (PolyI:C; InvivoGen), LPS (Sigma Chemical Co., St. Louis, MO, USA), flagellin (InvivoGen) or imiquimod (InvivoGen), a ligand of TLR1/2, 3, 4, 5, or 7 for 5 days (Fig. 2A). Addition of 10 µg/ml Pam3CSK4 or LPS increased melanin contents to 114 or 112% (mean±SD of control, n=3, p<0.05) of the control cells (100%), respectively. Treatment of 10 µg/ml flagellin or imiquimod reduced melanin contents to 91 or 85% (mean±SD of control, n=3, p<0.05) of the control cells (100%), respectively. There was no change with PolyI:C treatment. The effects of TLR ligands was further investigated using the zebrafish model system7. As shown in Fig. 2B, flagellin (20 µg/ml) and imiquimod (40 µg/ml) inhibit zebrafish body pigmentation. However, there was no remarkable change with treatment of Pam3CSK4 or LPS (data not shown). Taken together, these results suggest that activation of TLR2, 4, 5 and 7 modulate melanocyte pigmentation.
Fig. 2.
Effects of toll-like receptor (TLR) ligands on pigmentation. Melanocytes were treated with Pam3CSK4, PolyI:C, LPS, flagellin or imiquimod, a ligand (1~10µg/ml) of TLR1/2, 3, 4, 5, or 7 for 5 days. (A) Melanin content was determined by measuring the absorbance at 490 nm. The values indicate the mean of three independent experiments±SD. *p<0.05. (B) Zebrafish was treated with ligands (20 or 40 µg/ml) for 24 hrs and was visualized under microscope. The results were reproducible in three independent experiments. Magnified view (Inset).
The present study demonstrated the in vitro expression of a panel of TLRs in normal human melanocytes. Bacterial cell wall components are recognized by TLR1, 2, 3, 5, and 6, known as extracellular TLRs. We showed that treatment of TLRs (1/2, 4 and 5) with synthetic bacterial lipoprotein, PolyI:C, LPS and bacterial flagellin modulated melanocyte pigmentation. TLR3, 7, 8, and 9 are located in the cytoplasm and recognize mainly viral components. In our study, stimulation with imiquimod and synthetic TLR7/8 ligand reduced melanocyte pigmentation. It was shown that flagellin and imiquimod treatment also reduced zebrafish body pigmentation. It appears that zebrafish body pigmentation was too intense to show the subtle changes of increased pigmentation. Taken together, these results suggest that TLRs in melanocytes may play a role in microbial- or inflammation-related pigmentary changes. However, further investigations are needed to confirm the functional role of TLRs in pigmentation and validate the outcomes of this study, since it remains unclear whether pigmentary changes by TLRs ligands in this study were related to TLR activation.
In summary, we have demonstrated TLRs expression in normal human melanocytes. Further investigations are warranted to determine whether melanocyte response to TLR ligands may play a role in skin pigmentary changes.
Footnotes
This work was supported by the Korea Research Foundation Grant funded by the Korean Government (MOEHRD, Basic Research Promotion Fund) (KRF-2007-1-E00049).
References
- 1.Plonka PM, Passeron T, Brenner M, Tobin DJ, Shibahara S, Thomas A, et al. What are melanocytes really doing all day long...? Exp Dermatol. 2009;18:799–819. doi: 10.1111/j.1600-0625.2009.00912.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Terhorst D, Kalali BN, Ollert M, Ring J, Mempel M. The role of toll-like receptors in host defenses and their relevance to dermatologic diseases. Am J Clin Dermatol. 2010;11:1–10. doi: 10.2165/11311110-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 3.Mackintosh JA. The antimicrobial properties of melanocytes, melanosomes and melanin and the evolution of black skin. J Theor Biol. 2001;211:101–113. doi: 10.1006/jtbi.2001.2331. [DOI] [PubMed] [Google Scholar]
- 4.Yu N, Zhang S, Zuo F, Kang K, Guan M, Xiang L. Cultured human melanocytes express functional toll-like receptors 2-4, 7 and 9. J Dermatol Sci. 2009;56:113–120. doi: 10.1016/j.jdermsci.2009.08.003. [DOI] [PubMed] [Google Scholar]
- 5.Ahn JH, Park TJ, Jin SH, Kang HY. Human melanocytes express functional Toll-like receptor 4. Exp Dermatol. 2008;17:412–417. doi: 10.1111/j.1600-0625.2008.00701.x. [DOI] [PubMed] [Google Scholar]
- 6.Kang HY, Park TJ, Jin SH. Imiquimod, a Toll-like receptor 7 agonist, inhibits melanogenesis and proliferation of human melanocytes. J Invest Dermatol. 2009;129:243–246. doi: 10.1038/jid.2008.184. [DOI] [PubMed] [Google Scholar]
- 7.Choi TY, Kim JH, Ko DH, Kim CH, Hwang JS, Ahn S, et al. Zebrafish as a new model for phenotype-based screening of melanogenic regulatory compounds. Pigment Cell Res. 2007;20:120–127. doi: 10.1111/j.1600-0749.2007.00365.x. [DOI] [PubMed] [Google Scholar]