Abstract
Background
Intra-abdominal adhesions arise after more than 50% of all abdominal operations and are an important source of postoperative complications. They attach normally separated organs to each other and can cause major problems for the affected patients by giving rise to small bowel obstruction, chronic pelvic pain, dyspareunia, infertility, and higher complication rates in subsequent operations. They are also a frequent source of medicolegal conflict. Thus, every physician should be familiar with their mechanism of origin, their consequences, and the methods by which they can be prevented.
Methods
A selective PubMed/Medline search from 1960 onward as well as articles to which these publications referred. The expert consensus position of the European Society for Gynaecological Surgery is also taken into consideration.
Results
Adhesions arise through aberrant wound healing after peritoneal injury with further influence from a variety of other factors. Preventive measures include minimizing peritoneal injury intraoperatively through the meticulous observance of basic surgical principles, moistening the mesothelium to keep it from drying out, irrigating the peritoneal cavity to remove blood and clot, and keeping the use of intra-abdominal foreign material to a minimum.
Conclusion
Adhesions are an inevitable consequence of intra-abdominal surgery. They can be prevented to some extent with meticulous surgical technique and certain other measures. For operations carrying a high risk of postoperative adhesions, e.g., surgery on the adnexa or bowel, commercially available peritoneal instillates or barrier methods can be used to limit adhesion formation.
Intra-abdominal adhesions following surgery represent a major unsolved problem (1). They occur after 50% to 100% of all surgical interventions in the abdomen and can complicate the work of the surgeon considerably (2). Dembrowski published the first data on induction of adhesions in an animal model in 1889 (3), and the intervening 120 years have seen extensive studies in vitro and in vivo. Nevertheless, the literature contains neither an official definition of adhesions nor a recognized standardized classification for objective assessment of their extent and severity. Accordingly, study findings are often imprecise and do not lend themselves to adequate interpretation. By the same token, there is a lack of clinically oriented guidelines for the diagnosis, treatment and options for reduction of adhesions.
The severe consequences of intra-abdominal adhesions for patients, physicians, and healthcare systems stand in stark contrast to the low level of awareness and knowledge—due not least to the lack of standardization and the patchy data—among doctors. Against that backdrop, this article sets out to:
Increase clinicians’ awareness of adhesions and their consequences
Offer an overview of the pathogenesis of adhesions
Describe universally applicable and readily implemented strategies to reduce the occurrence of adhesions
Introduce commercial products for reduction of adhesions.
Material and methods
We performed the literature search for this review with the aid of our working group’s existing database. This database, comprising articles published in PubMed/Medline since 1960, is updated monthly by addition of all articles found using the search terms “adhesions”, “intraperitoneal adhesions”, “intraabdominal adhesions”, “adhesion reduction”, “adhesion prophylaxis”, and “adhesion formation”. It also contains relevant publications found in the reference lists of the articles identified. The expert consensus position of the European Society for Gynaecological Endoscopy was taken into consideration.
Types of adhesions
Intra-abdominal adhesions may be congenital or acquired. Congenital adhesions arise during physiological organogenesis—like the frequently observed attachment of the sigmoid colon to the left pelvic wall—or can be traced back to abnormal embryonal development of the abdominal cavity. They are usually asymptomatic and are diagnosed incidentally (4).
Postmortem examination of patients who had not undergone surgery identified postinflammatory adhesions in 28% of cases (5). These are caused by intra-abdominal inflammation or can be attributed to endometriosis, peritonitis, radiotherapy, or long-term peritoneal dialysis (4, 6, 7).
Postoperative adhesions form after 50% to 100% of all abdominopelvic interventions (2). They develop as a result of wound healing and are influenced by various factors (7) (box 1).
Box 1. Overview of factors that influence the formation of adhesions*1.
Complexity of operation (e1)
Previous illness (e.g., diabetes) (4)
Poor nutritional status (4)
Intra-abdominal placement of foreign bodies (e.g. meshes) (4)
Excessive coagulation with tissue necrosis (e4)
Accompanying bacterial infection (4)
-
Laparoscopy
-
Laparoscopy
Dehydration owing to dry gas (e7)
-
Laparoscopy
Mesothelial hypoxia owing to use of CO2 (e8)
-
Laparotomy
Dehydration owing to light and heat (e4)
-
Laparotomy
-
Laparotomy
*1The numbers in parentheses are reference citations
The greater omentum is involved in 80% of cases of postoperative intra-abdominal adhesions, the bowel in only around 50% (8). Ovarian adhesions can be demonstrated in over 90% of patients after gynecological adnexal surgery (9); this is explained by the high sensitivity of the ovarian epithelium and its proximity to other peritoneal surfaces (10). Patients at high risk of already having or developing adhesions are those with previous or planned adnexal interventions, ablation of endometriosis, or bowel surgery involving large peritoneal defects, together with all those who have undergone previous abdominal surgery with pronounced formation of adhesions.
Diagnosis
Intra-abdominal adhesions are predominantly diagnosed intraoperatively. Careful history taking can substantiate the suspicion of adhesions; no other clinical investigations or imaging procedures enable a confident diagnosis. Evidence pointing to adhesions may be yielded by high-resolution ultrasonography and functional cine MRI, both of which detect limited movement relative to one another of organs joined by adhesions (e11, e12). However, neither of these modalities is established in routine clinical practice.
Complications of adhesions
The intra-abdominal adhesions that arise from the beginning of the surgical procedure can cause complications decades later (8, 11). The patients’ symptoms include meteorism, irregular bowel movementsoder ist eher “variable stool consistency” gemeint?, chronic abdominal pain, digestive disorders, unwanted childlessness, and intestinal occlusions, and often fail to be associated with their cause (12). In contrast to congenital or postinflammatory adhesions, which are mostly asymptomatic, postoperative adhesions cause 40% of all cases of intestinal obstruction. Stenoses of the large intestine are produced principally by malignancies and only rarely by adhesions, but adhesions cause 65% to 75% of small bowel occlusions—the most serious of all adhesion-induced complications (8). Particularly colectomy, involving a large peritoneal incision, carries an 11% cumulative risk of ileus within the first year after operation (13).
Adhesions are responsible for 15% to 20% of all cases of female secondary infertility (14). Paraovarian, peritubal adhesions can lead to follicular entrapment and reduced mobility and mechanical blockade of the fallopian tubes. This may limit oocyte transport, increasing the risk of ectopic pregnancy (14, 15).
Chronic lower abdominal pain severely impairs the quality of life of those affected and forms the indication for 30% to 50% of all laparoscopies and 5% of hysterectomies (16). In his review of 11 studies, DiZerega showed that adhesions had been responsible for the chronic lower abdominal pain in only 40% of the women who had undergone surgery (17). In 25% of cases the cause remained unclear. Accordingly, it is difficult to advise those suffering from such pain whether an operation will reveal the cause and whether laparotomic or laparoscopic adhesiolysis may relieve their symptoms. In a prospective study, Keltz et al. observed a significant reduction in chronic abdominal pain after right-sided paracolic adhesiolysis (18). In contrast, Swank et al. found no amelioration of pain after laparoscopic release of adhesions not constricting the bowel (19).
Patients who have undergone surgery previously should be fully informed about adhesiolysis and its potential complications and required to give their written consent [Originalwortlaut: “schriftliche Aufklärung”]. Discussion points should include extension of the operation and anesthesia time, the increased blood loss and the significantly higher risk of injury to the omentum, bladder, ureters and vessels (20). Reoperations have a 20% rate of enterotomy—often associated with poorer patient outcome and longer hospital stay (20). Particularly in the case of known extensive intra-abdominal adhesions, the indication for any further operation should be considered very carefully because of the up to 85% likelihood of regrowth or de novo formation of adhesions (21). If this occurs, future minimally invasive surgery may be difficult or even impossible (20, e2). Adhesion-related changes in pelvic anatomy can also complicate or prevent:
Pathogenesis
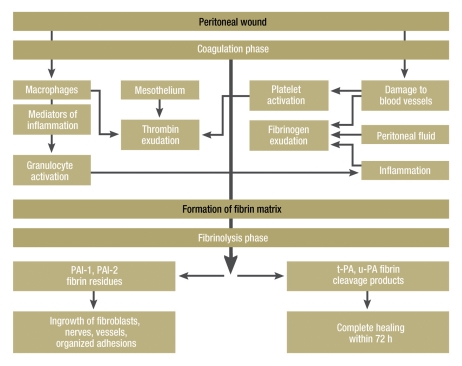
Since intra-abdominal adhesions arise from aberrant peritoneal wound healing processes, any mesothelial damage by surgical trauma or bacterial inflammation can lead to their formation (22). Damage to the peritoneum is followed by capillary bleeding and increased vascular permeability with consequent exudation of fibrinogen (6, 22, e2). After cleavage of fibrinogen to fibrin and its bonding with fibronectin the defect is closed and a temporary wound bed forms (22, e13). Within the ensuing 72 h endogenous fibrinolytic activity of the mesothelial cells leads to breakdown of these fibrin deposits and thus to complete regeneration (e15).
A key role in the origin of adhesions is attributed to a pathological reduction in peritoneal fibrinolysis capacity (e16). This may result from destruction of mesothelia, from their insufficient supply with blood, from increased synthesis of fibrinolysis antagonists following trauma, from hypoxia, from radical formation, or from bacterial infection (22, e14, e16– e18). In the course of the subsequent organization processes the persisting fibrin matrix gives rise to a mesothelialized tissue structure that is stabilized by connective tissue and may contain arterioles, venules, capillaries, and nerve fibers (e14). An overview of the identified pathophysiological associations and the factors thought to be involved in the origin of adhesions is provided by the Figure, Table 1 and the eBox.
Figure.
Overview of pathophysiological interrelationships and factors thought to be involved in the origin of adhesions (modified from e16)
Table. Overview of the principal factors affecting the fibrinolytic capacity of the mesothelium.
| Factor | Fibrinolytic activity | Reference |
| Urokinase-like plasminogen activator (u-PA) | ↑ | (e15) |
| Tissue plasminogen activator (t-PA) | ↑ | (e15) |
| Matrix metalloproteinases (MMP) | ↑ | (6) |
| Tissue-derived inhibitors (TIMP) | ↓ | (6) |
| Plasminogen activation ‧inhibitors (PAI 1/ 2) | ↓ | (e14, e19) |
| Mechanical destruction of ‧mesothelium | ↓ | (e16) |
| Mesothelial ischemia | ↓ | (e16) |
| Hypoxy | ↓ | (e18, e20) |
| Radical formation | ↓ | (e18) |
| Bacterial lipopolysaccharide | ↓ | (e18, e21) |
| Interleukins (e.g., IL-1, IL-6) | ↓ | (18) |
| Neurokinin-1 receptor (NK-1) | ↓ | (e20) |
| Substance P (SP) | ↓ | (e16, e20) |
| Tumor necrosis factor α (TNFα) | ↓ | (e17, e22) |
| Transforming growth factor β (TGFβ) | ↓ | (e17, e23) |
| Intracellular adhesion molecule (ICAM 1) | ↓ | (4, e17) |
| Vascular cell adhesion molecule (VCAM) | ↓ | (4, e17) |
ebox. Supplementary information on pathogenesis of adhesions .
Surgical trauma, i.e., the combined impact of cutting, coagulation, and pressure-induced ischemia – particularly from excessively tight knots – may bring about peritoneal damage (22, e2). Equally, mesothelial injury results from bacterial inflammation processes, from contact, from bright surgical lights, or from use of dry drapes (22). Capillaries at the trauma site leak blood containing complement and coagulation factors. Local peritoneal macrophages and mesothelial cells start to secrete proinflammatory cytokines, histamine, prostaglandins, and kinins, leading to potentiated influx of further inflammation-related cellsgut so? Deutsch: Entzündungszellen, increased vascular permeability, and subsequent fibrinogen exudation (6, 22, e2). Thrombin is formed by activated complement and coagulation cascades and breaks fibrinogen down to fibrin, which then combines with fibronectin from the peritoneal connective tissue to form a temporary wound bed, into which migrate peritoneal cells and fibroblasts (22, e13, e14). Within the next 72 h local mesothelial fibrinolysis begins. This physiological fibrinolytic activity is based on synthesis of urokinase-like plasminogen activator (u-PA) and tissue plasminogen activator (t-PA), which release plasmin, a local protease with broad substrate specificity, from plasminogen (e15, e16). Plasmin degrades fibrin polymers, components of the extracellular matrix and basal membrane, and activates other proteases, e.g., matrix metalloproteinases (6). This depletion of fibrin deposits then results in complete healing (e15).
A key part in the origin of adhesions is played by pathological reduction of peritoneal fibrinolysis capacity (e16). This results from:
Reduced release of plasminogen activators following loss of or insufficient supply of blood to mesothelia (e16)
Reduction in the activity of plasminogen activators by a local and systemic increase in protease antagonists – plasminogen activator inhibitors PAI 1 and 2 – after surgical trauma (e14).
As shown by in-vitro and in-vivo studies at molecular level, this disequilibrium between plasminogen activators and protease antagonists is based on increased expression of inflammation mediators (e.g., substance P) – particularly of cytokines (e.g., tumor necrosis factor), growth factors (e.g., transforming growth factor), and adhesion molecules (intercellular adhesion molecule-1 and vascular adhesion molecule-1). (25, e4, e5).
Prevention of postoperative adhesions
Strategies for reduction of adhesions are based on their pathophysiological mechanisms of origin (box 2).
Box 2. Practical tips: general strategies for reduction of adhesions*1.
Preference for tissue-sparing and microinvasive surgical techniques
Minimization of operating time and of heat and light
Avoidance of peritoneal trauma by superfluous contact and coagulation
Limited placement of intra-abdominal foreign bodies such as patches, meshes, and suture material
Use of moistened abdominal drapes and swabs and occasional application of saline solution to minimize dehydration of mesothelial surfaces
Irrigation of the abdominal cavity to remove residual intra-abdominal blood depots
Reduction of infection risk by ensuring sterile working conditions and giving antibiotics as required
Laparotomy: preferential use of latex- and powder-free gloves
Laparoscopy: use of humidified gases at appropriately low insufflation pressure
High-risk patients: use of barrier techniques or peritoneal instillates after appropriate explanation
Damage to the serosa and the use of intra-abdominal foreign bodies should be kept to a minimum (4). Blood and clot in association with a peritoneal wound constitute a potentiating factor, because additional fibrin has to be degraded by the fibrinolytic activity of the peritoneum (e24). Before closure of the abdominal wall, therefore, it is advisable to perform careful—though not excessive, to avoid necrosis—hemostasis and irrigate repeatedly with saline and Ringer solution. There is no consensus in the literature as to whether laparoscopy is associated with fewer de novo and recurring adhesions than laparotomy (8, e26). A lower rate of adhesion development in laparoscopic interventions could be related to reduction of peritoneal trauma as a result of more exact preparation under magnification (e3). Moreover, contamination of the abdominal cavity and adhesion-potentiating foreign-body reactions are reduced (e9). Further advantages include a minimized incidence of postoperative infections and a tamponade effect of the pneumoperitoneum in the event of hemorrhage. A disadvantage of laparoscopy, related to the longer operating time and the high insufflation pressure, is the risk of mesothelial injury; this can be reduced by using humidified and warmed gases (e25). With regard to development of adhesions, minimally invasive access via natural orifices (Natural Orifice Transluminal Endoscopic Surgery, NOTES) seems to be superior to both laparoscopy and laparotomy. In an animal study, Dubcenco found the lowest number and severity of adhesions in the group in which endoscopy was carried out by the orogastric route (e27).
In those at high risk the use of adhesion-reducing adjuvants can be considered independent of the extent and location of the mesothelial defect. The widely used, commercially available adjuvants licensed for use in Germany include:
Humidified and warmed insufflation gases for laparoscopy
Medicinal agents
Colloid and crystalloid solutions
Separators: fluids for peritoneal instillation or site-specific mechanical barriers.
Attempted drug treatment can involve local and systemic anti-inflammatory agents, fibrinolytics, or antibiotic solutions. Moreover, colloids (dextran) and crystalloid solutions (Ringer lactate or saline) have been used, alone or with corticosteroids or heparin, to separate peritoneal surfaces. No clinical study has yet demonstrated a clear adhesion-reducing benefit of these substances (25).
The 4% glucose polymer icodextrin is an adhesion-inhibiting peritoneal instillate. Besides its application for intraoperative moistening of peritoneal surfaces it is instilled into the abdominal cavity (e28). By virtue of its osmotic activity it is thought to retain fluid in the peritoneal cavity for 3 to 4 days and keep organs and injured peritoneal surfaces separated from each other until it is eliminated via the kidneys. Randomized, double-blind multicenter studies have confirmed the adhesion-reducing properties of icodextrin after surgery. Comparison of icodextrin and Ringer lactate revealed an advantage for the former with regard to the reduction of incidence (52% vs. 32%), extent (52% vs. 47%), and severity (65% vs. 37%) of adhesions. Clinical improvement was observed in 49% of patients following treatment with icodextrin, against 38% after Ringer lactate (e28– e30). Data from the European registry on the use of icodextrin (adeptTM Registry for Clinical Evaluation, ARIEL) demonstrate high user-friendliness and high patient safety. Complications described after icodextrin instillation are septic and inflammatory states, anastomotic insufficiency, and labial swelling (e31).
Cross-linked esters of hyaluronic acid form a viscous gel that is applied to traumatized peritoneal surfaces after abdominopelvic surgery to help keep them separate during the healing process. Few studies have been conducted on the efficacy of hyaluronic acid esters in preventing adhesions. In a group of 52 patients in a randomized multicenter study, application of hyaluronic acid gel was shown to reduce formation of adhesions after laparoscopic enucleation of myoma. Following treatment 62% of these patients were free of adhesions, compared to 41% of those who did not receive the gel. Application of the gel significantly lowered the difference in severity of intra-abdominalDeutsch: intraperitoneal adhesions between first and subsequent operations (0.3 ± 0.9 vs. 0.8 ± 1.0, p<0.05) (e32). Furthermore, Pellicano et al. documented an increase in the rate of pregnancy from 38.8% to 77.8% in previously infertile women 12 months after laparoscopic enucleation of myoma with application of gel (e33).
Carboxymethylcellulose (CMC) and polyethylene oxide (PEO) form a gel-like resorbable barrier to adhesions. In a randomized study, 37 high-risk patients received a CMC/PEO barrier in the course of laparoscopic ablation of endometriosis. Follow-up laparoscopy documented a significant adhesion-reducing effect of this measure as assessed using the American Fertility Society score, with a decrease from 8.4±3 points to 6.2 ± 2 points. In the non-treated control group there was increased growth of adhesions and thus a rise in the score from 10 ± 2.5 points to 14 ± 3 points (e34).
A barrier membrane consisting of hyaluronic acid and CMC can separate peritoneal surfaces for around 7 days (10). Because of its high fragility this membrane is predominantly used in laparotomies (e35). The efficacy of such membranes in reducing intra-abdominal adhesions after enucleation of myoma and colectomy has been investigated in a number of randomized studies. With regard to the gynecological data, the Cochrane analysis by Ahmad et al. notes that the positive findings reported by Diamond et al. (e35) have to be interpreted with caution owing to statistical deficiencies (24). Follow-up laparoscopy 8 to 12 weeks after use of the barrier membrane on abdominal wall closure in patients undergoing colectomy and creation of an ileal pouch showed that 51% of treated patients were free of adhesions, against 6% in the control group (e36, e37). [worauf bezieht sich der nächste Satz? Auf die Studie e38 oder auf das zuvor gesagte?: Die Membran wurde als einziges Agens hinsichtlich Prävention des adhäsionsbedingten Dünndarmverschlusses nach Darmresektion untersucht] In a multicenter study, Fazio et al. (e38) investigated use of the barrier membrane as sole preventive measure against adhesion-related occlusion of the small intestine after bowel resection. The membrane resulted in a 1.6% absolute and 47% relative reduction in the occurrence of this complication. It should be pointed out, however, that application of the membrane to the anastomosis sutures increased the risk of anastomotic insufficiency (e38).
Another type of adhesion barrier, applied as a spray, comprises a pair of polyethyleneglycols in a two-component system. The barrier is sprayed onto injured serosal surfaces and seals them for 7 to 14 days. Early clinical pilot studies showed an adhesion-preventing benefit of the spray, but this effect was not confirmed in subsequent, more extensive trials (e39, 23). Evaluation of the next-generation product in a porcine model showed a reduction in number (ca. 46%) and extent (ca. 83%) of the adhesions formed (e40).
Oxidized regenerated cellulose can be applied to injured surfaces as a resorbable membrane, following careful hemostasis. Moistening of the membrane stops it slipping and provides a physical barrier between tissues until the membrane is resorbed after 4 weeks. In their Cochrane analysis, Ahmad et al. conclude that oxidized regenerated cellulose leads to a reduction in the occurrence of pelvic adhesions after gynecological laparotomy and laparoscopy (24). It is advised, however, that this finding be interpreted with caution.
Perspective
Since surgical treatment of adhesions is highly likely to be associated with the induction of new adhesions, reduction or prevention of adhesions should be every surgeon’s primary goal. In this regard, the Clinical Adhesion Research and Evaluation Group (CARE Group) has been founded at Giessen University Hospital. This interdisciplinary group aims to optimize patient care by integrating existing strategies into routine clinical practice and conducting research into new techniques for reducing adhesions. The general measures described in this review can readily be put into practice and comprise minimization of peritoneal injury by the following means: meticulous observance of established surgical principles, moistening of the mesothelium to keep it from drying out, reduction of the use of intra-abdominal foreign materials to a minimum, and irrigation of the abdominal cavity to remove blood and clot. Adhesion-reducing agents differ, sometimes considerably, in their indications and area of surgery. Their use is particularly advisable in high-risk patients.
Conclusive interpretation of the partially controversial study findings on adhesion-reducing adjuvants is hampered by the limited number of studies, the small numbers of patients, the large variety of factors influencing adhesion development, and the lack of a standardized classification of adhesions. The result is skepsis among clinicians and low acceptance of adhesion-reducing products. Moreover, it is often difficult to arrange for these products to be used because there is no provision for their reimbursement under the diagnosis-related groups system. Further high-quality studies are therefore required.
Key Messages.
Adhesions result from peritoneal trauma and aberrant wound healing processes and can therefore develop after any intra-abdominal operation.
Intra-abdominal adhesions occur in 50 to 100% of patients with previous surgery.
Particularly in patients with previous surgery, adhesion-related complications can occur at any time.
The possibility of adhesions and the associated risk must always be documented in writing in the course of preoperative explanation of the planned procedure for purposes of consent.
General strategies for preventing adhesions should be integrated into routine clinical practice. The use of commercially available peritoneal instillates or barrier techniques is particularly advisable in patients at high risk of developing adhesions.
Acknowledgments
Translated from the original German by David Roseveare
We thank Prof. Rudy Leon DeWilde of Pius Hospital Oldenburg, member of the Expert Adhesions Working Party of the European Society for Gynaecological Endoscopy (ESGE), for his help in writing and revising the manuscript.
Footnotes
Conflict of interest statement
Prof. Tinneberg has received reimbursement of travel costs and lecture fees from Baxter. Dr. Hackethal has received reimbursement of travel costs from Baxter und has consultancy contracts with NordicPharma und Fischer & Paykel. Dr. Tchartchian has a consultancy contract with and has received reimbursement of travel costs from Covidien.
Dr. Brüggmann, Dr. Wallwiener und Prof. Münstedt declare that no conflict of interest exists according to the guidelines of the International Committee of Medical Journal Editors.
References
- 1.Diamond MP, El-Hammady E, Wang R, Kruger M, Saed G. Regulation of expression of tissue plasminogen activator and plasminogen activator inhibitor-1 by dichloroacetic acid in human fibroblasts from normal peritoneum and adhesions. Am J Obstet Gynecol. 2004;190:926–934. doi: 10.1016/j.ajog.2004.02.009. [DOI] [PubMed] [Google Scholar]
- 2.DiZerega GS. Contemporary adhesion prevention. Fertil Steril. 1994;61:219–235. doi: 10.1016/s0015-0282(16)56507-8. [DOI] [PubMed] [Google Scholar]
- 3.von Dembowski T. Über die Ursachen der peritonealen Adhäsionen nach chirurgischen Eingriffen mit Rücksicht auf die Frage des Ileus nach Laparotomien. Langenbecks Arch Chir. 1889;37 [Google Scholar]
- 4.Liakakos T, Thomakos N, Fine PM, Dervenis C, Young RL. Peritoneal adhesions: etiology, pathophysiology, and clinical significance. Dig Surg. 2001;18:260–273. doi: 10.1159/000050149. [DOI] [PubMed] [Google Scholar]
- 5.Weibel MA, Mayno G. Peritoneal adhesions and their relationship to abdominal surgery. A postmortem study. Am J Surg. 1973;126:345–353. doi: 10.1016/s0002-9610(73)80123-0. [DOI] [PubMed] [Google Scholar]
- 6.Cheong YC, Laird SM, Li TC, Shelton JB, Ledger WL, Cooke ID. Peritoneal healing and adhesion formation/reformation. Human Reprod Update. 2001;7:556–566. doi: 10.1093/humupd/7.6.556. [DOI] [PubMed] [Google Scholar]
- 7.Monk BJ, Berman ML, Montz FJ. Adhesions after extensive gynecologic surgery: clinical significance, etiology and prevention. Am J Obstet Gynecol. 1994;170:1396–1403. doi: 10.1016/s0002-9378(94)70170-9. [DOI] [PubMed] [Google Scholar]
- 8.Menzies D, Ellis H. Intestinal obstruction from adhesions: How big is the problem? Ann R Coll Surg Engl. 1990;72:60–63. [PMC free article] [PubMed] [Google Scholar]
- 9.Pittaway DE, Daniell JF, Maxson WS. Ovarian surgery in an infertility patient as an indication for a short-interval second-look laparoscopy: a preliminary study. Fertil Steril. 1985;44:611–614. doi: 10.1016/s0015-0282(16)48975-2. [DOI] [PubMed] [Google Scholar]
- 10.Diamond MP, Pellicer A, Boyers SP, DeCherney AH. The effect of periovarian adhesions on follicular development in patients undergoing ovarian stimulation for in vitro fertilization-embryo transfer. Fertil Steril. 1988;49:100–103. doi: 10.1016/s0015-0282(16)59657-5. [DOI] [PubMed] [Google Scholar]
- 11.Ellis H, Moran BJ, Thompson JN, et al. Adhesion-related readmissions after abdominal pelvic surgery: a retrospective cohort study. Lancet. 1999;353:1476–1480. doi: 10.1016/S0140-6736(98)09337-4. [DOI] [PubMed] [Google Scholar]
- 12.Attard JA, MacLean AR. Adhesive small bowel obstruction: epidemiology, biology and prevention. Can J Surg. 2007;50:291–300. [PMC free article] [PubMed] [Google Scholar]
- 13.Nieuwenhuijzen M, Reijnen MM, Kuijpers JH, van Goor H. Small bowel obstruction after total or subtotal colectomy: a 10-year retrospective review. Br J Surg. 1998;85:1242–1245. doi: 10.1046/j.1365-2168.1998.00841.x. [DOI] [PubMed] [Google Scholar]
- 14.Stovall TG, Elder RF, Ling FW. Predictors of pelvic adhesions. J Reprod Med. 1989;34:345–348. [PubMed] [Google Scholar]
- 15.Molloy D, Martin M, Speirs A, et al. Performance of patients with a „frozen pelvis“ in an in vitro fertilization program. Fertil Steril. 1987;47:450–455. doi: 10.1016/s0015-0282(16)59054-2. [DOI] [PubMed] [Google Scholar]
- 16.Howard FM. The role of laparoscopy as a diagnostic tool in chronic pelvic pain. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14:467–494. doi: 10.1053/beog.1999.0086. [DOI] [PubMed] [Google Scholar]
- 17.DiZerega GS. Biochemical events in peritoneal tissue repair. Eur J Surg Suppl. 1997;577:10–16. [PubMed] [Google Scholar]
- 18.Keltz MD, Gera PS, Olive DL. Prospective randomized trial of right-sided paracolic adhesiolysis for chronic pelvic pain. JSLS. 2006;10:443–446. [PMC free article] [PubMed] [Google Scholar]
- 19.Swank DJ, Swank-Bordewijk SC, Hop WC, et al. Laparoscopic adhesiolysis in patients with chronic abdominal pain: a blinded randomized controlled multi-center trial. Lancet. 2003;361:1247–1251. doi: 10.1016/s0140-6736(03)12979-0. [DOI] [PubMed] [Google Scholar]
- 20.van der Krabben AA, Dijkstra FR, Nieuwenhuijzen M, et al. Morbidity and mortality of inadvertent enterotomy during adhesiotomy. Br J Surg. 2000;87:467–471. doi: 10.1046/j.1365-2168.2000.01394.x. [DOI] [PubMed] [Google Scholar]
- 21.Diamond MP, Freeman ML. Clinical implications of postsurgical adhesions. Hum Reprod Update. 2001;7:567–576. doi: 10.1093/humupd/7.6.567. [DOI] [PubMed] [Google Scholar]
- 22.Reed KL, Fruin AB, Bishop-Bartolomei KK, et al. Neurokinin-1 receptor and substance P messenger RNA levels increase during intraabdominal adhesion formation. J Surg Res. 2002;108:165–172. doi: 10.1006/jsre.2002.6533. [DOI] [PubMed] [Google Scholar]
- 23.DeWilde RL, Trew G. on behalf of the Expert Adhesions Working Party of the European Society of Gynaecological Endoscopy (ESGE): Postoperative abdominal adhesions and their prevention in gynaecological surgery. Expert consensus position. Part 2 - steps to reduce adhesions. Gynecol Surg. 2007;4:243–253. [Google Scholar]
- 24.Ahmad G, Duffy JM, Farquhar C, et al. Barrier agents for adhesion prevention after gynaecological surgery. Cochrane Database Syst Rev. 2008;(2) doi: 10.1002/14651858.CD000475.pub2. CD000475. [DOI] [PubMed] [Google Scholar]
- 25.Metwally M, Watson A, Lilford R, Vandekerckhove P. Fluid and pharmacological agents for adhesion prevention after gynaecological surgery. Cochrane Database Syst Rev. 2006;19 doi: 10.1002/14651858.CD001298.pub3. CD001298. [DOI] [PubMed] [Google Scholar]
- e1.DeCherney AH, DiZerega GS. Clinical problem of intraperitoneal postsurgical adhesion formation following general surgery and the use of adhesion prevention barriers. Surg Clin North Am. 1997;77:671–688. doi: 10.1016/s0039-6109(05)70574-0. [DOI] [PubMed] [Google Scholar]
- e2.Dijkstra FR, Nieuwenhuijzen M, Reijnen MMPJ, van Goor H. Recent clinical developments in pathophysiology, diagnosis and treatment of intra-abdomainal adhesions. Scand J Gastroenterol. 2000;(Suppl 232):52–59. [PubMed] [Google Scholar]
- e3.Menzies D. Prospective adhesions: their treatment and relevance in clinical practice. Ann R Coll Surg Engl. 1993;75:147–153. [PMC free article] [PubMed] [Google Scholar]
- e4.Drollette CM, Badaway SZA. Pathophysiology of pelvic adhesions. J Reprod Med. 1992;37:107–121. [PubMed] [Google Scholar]
- e5.Yesildaglar N, Koninckx PR. Adhesion formation in intubated rabbits increases with high insufflation pressure during endoscopic surgery. Hum Reprod. 2000;15:687–691. doi: 10.1093/humrep/15.3.687. [DOI] [PubMed] [Google Scholar]
- e6.Molinas CR, Koninckx PR. Hypoxaemia induced by CO2 or helium pneumoperitoneum is a co-factor in adhesion formation in rabbits. Hum Reprod. 2000;15:1758–1763. doi: 10.1093/humrep/15.8.1758. [DOI] [PubMed] [Google Scholar]
- e7.Ott DE. Laparoscopy and adhesion formation, adhesions and laparoscopy. Semin Reprod Med. 2008;26:322–330. doi: 10.1055/s-0028-1082390. [DOI] [PubMed] [Google Scholar]
- e8.Molinas CR, Mynbaev O, Pauwels A, Novak P, Koninckx PR. Peritoneal mesothelial hypoxia during pneumoperitoneum is a cofactor in adhesion formation in a laparoscopic mouse model. Fertil Steril. 2001;76:560–567. doi: 10.1016/s0015-0282(01)01964-1. [DOI] [PubMed] [Google Scholar]
- e9.Torre M, Favre A, Pini Prato A, Brizzolara A, Martucciello G. Histologic study of peritoneal adhesions in children and rat model. Pediatr Surg Int. 2002;18:673–676. doi: 10.1007/s00383-002-0872-6. [DOI] [PubMed] [Google Scholar]
- e10.Luijendijk RW, de Lange DC, Wauters CC, et al. Foreign material in postoperative adhesions. Ann Surg. 1996;223:242–248. doi: 10.1097/00000658-199603000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e11.Mussack T, Fischer T, Ladurner R, et al. Cine MR imaging vs. high-resolution ultrasonography for detection of adhesions after laparoscopic and open incisional hernia repair. Surg Endosc. 2005;19:1538–1543. doi: 10.1007/s00464-005-0092-y. [DOI] [PubMed] [Google Scholar]
- e12.Buhmann-Kirchhoff S, Lang R, Kirchhoff C, et al. Functional cine MR imaging for the detection and mapping of intraabdominal adhesions: method and surgical correlation. Eur Radiol. 2008;18:1215–1223. doi: 10.1007/s00330-008-0881-5. [DOI] [PubMed] [Google Scholar]
- e13.Reijnen MM, Bleichrodt RP, van Goor H. Pathophysiology of intra-abdominal adhesion and abscess formation, and the effect of hyaluronan. Br J Surg. 2003;90:533–541. doi: 10.1002/bjs.4141. [DOI] [PubMed] [Google Scholar]
- e14.diZerega GS, Campeau JD. Peritoneal repair and post-surgical adhesion formation. Hum Reprod Update. 2001;7:547–555. doi: 10.1093/humupd/7.6.547. [DOI] [PubMed] [Google Scholar]
- e15.Sulaiman H, Dawson L, Laurent GJ, Bellingan GJ, Herrick SE. Role of plasminogen activators in peritoneal adhesion formation. Biochem Soc Trans. 2002;30:126–131. doi: 10.1042/. [DOI] [PubMed] [Google Scholar]
- e16.Holmdahl L, Erikkson E, Risberg B. Fibrinolysis in the human peritoneum during operation. Surgery. 1996;119:701–705. doi: 10.1016/s0039-6060(96)80196-6. [DOI] [PubMed] [Google Scholar]
- e17.Halme J. Release of tumor necrosis factor-alpha by human peritoneal macrophages in vivo and in vitro. Am J Obstet Gynecol. 1989;161:1718–1725. doi: 10.1016/0002-9378(89)90957-5. [DOI] [PubMed] [Google Scholar]
- e18.Fletcher NM, Jiang ZL, Diamond MP, Abu-Soud HM, Saed GM. Hypoxia-generated superoxide induces the development of the adhesion phenotype. Free Radic Biol Med. 2008;45:530–536. doi: 10.1016/j.freeradbiomed.2008.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e19.Raftery AT. Method of measuring fibrinolytic activity in a single layer of cells. J Clin Pathol. 1981;34:625–629. doi: 10.1136/jcp.34.6.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e20.Reed KL, Heydrick SJ, Aarons CB, et al. A neurokinin-1 receptor antagonist that reduces intra-abdominal adhesion formation decreases oxidative stress in the peritoneum. Am J Physiol Gastrointest Liver Physiol. 2007;293G:544–551. doi: 10.1152/ajpgi.00226.2007. [DOI] [PubMed] [Google Scholar]
- e21.van Hinsbergh VW, Kooistra T, Scheffer MA, Hajo van Bockel J, van Muijen GN. Characterization and fibrinolytic properties of human omental tissue mesothelial cells. Comparison with endothelial cells. Blood. 1990;75:1490–1497. [PubMed] [Google Scholar]
- e22.Whawell SA, Scott-Coombed DN, Vipond MN, et al. Tumour necrosis factor mediated release of plasminogen activator inhibitor-1 by human peritoneal mesothelial cells. Br J Surg. 1994;81:214–216. doi: 10.1002/bjs.1800810218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e23.Tietze L, Elbrecht A, Schauerte C, et al. Modulation of pro- and antifibrinolytic properties of human peritoneal mesothelial cells by transforming growth factor-b1 (TGF-b1), tumor necrosis factor-a (TNF-a) and interleukin-1b (IL-1b) Thromb Haemost. 1998;79:362–370. [PubMed] [Google Scholar]
- e24.Golan A, Winston RML. Blood and intraperitoneal adhesion formation in the rat. J Obstet Gynaecol. 1989;9:248–252. [Google Scholar]
- e25.Peng Y, Zheng M, Ye Q, Chen X, Yu B, Liu B. Heated and humidified CO2 prevents hypothermia, peritoneal injury, and intra-abdominal adhesions during prolonged laparoscopic insufflations. J Surg Res. 2009;151:40–47. doi: 10.1016/j.jss.2008.03.039. [DOI] [PubMed] [Google Scholar]
- e26.Gutt CN, Oniu T, Schemmer P, Mehrabi A, Buchler MW. Fewer adhesions induced by laparoscopic surgery? Surg Endosc. 2004;18:898–906. doi: 10.1007/s00464-003-9233-3. [DOI] [PubMed] [Google Scholar]
- e27.Dubcenco E, Assumpcao L, Dray X, et al. Adhesion formation after peritoneoscopy with liver biopsy in a survival porcine model: comparison of laparotomy, laparoscopy, and transgastric natural orifice transluminal endoscopic surgery (NOTES) Endoscopy. 2009;41:971–978. doi: 10.1055/s-0029-1215229. [DOI] [PubMed] [Google Scholar]
- e28.diZerega GS, Verco SJS, Young P, et al. A randomized, controlled pilot study of the safety and efficacy of 4% icodextrin solution in the reduction of adhesions following laparoscopic gynaecological surgery. Hum Reprod. 2002;17:1031–1038. doi: 10.1093/humrep/17.4.1031. [DOI] [PubMed] [Google Scholar]
- e29.Brown CB, Luciano AA, Martin D, Peers E, Scrimgeour A, diZerega GS. on behalf of the Adept Adhesion Reduction Study Group: Adept (4% icodextrin solution) reduces adhesions after laparoscopic surgery for adhesiolysis: a doubleblind, randomized, controlled study. Fertil Steril. 2007;88:1413–1426. doi: 10.1016/j.fertnstert.2006.12.084. [DOI] [PubMed] [Google Scholar]
- e30.Kossi J, Gronlund S, Uotila-Nieminen M, Crowe A, Knight A, Keranen U. The effect of 4% icodextrin solution on adhesiolysis surgery time at the Hartmann’s reversal: a pilot, multicentre, randomized control trial vs lactated Ringer’s solution. Colorectal Dis. 2009;11:168–172. doi: 10.1111/j.1463-1318.2008.01562.x. [DOI] [PubMed] [Google Scholar]
- e31.Menzies D, Pascual MH, Walz MK, et al. Use of Icodextrin 4% solution in the prevention of adhesion formation following general surgery: from the multicentre ARIEL Registry. Ann R Coll Surg Engl. 2006;88:375–382. doi: 10.1308/003588406X114730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e32.Mais V, Bracco GL, Litta P, Gargiulo T, Melis GB. Reduction of postoperative adhesions with an auto-crosslinked hyaluronan gel in gynaecological laparoscopic surgery: a blinded, controlled, randomized, multicentre study. Hum Reprod. 2006;21:1248–1254. doi: 10.1093/humrep/dei488. [DOI] [PubMed] [Google Scholar]
- e33.Pellicano M, Guida M, Bramante S, et al. Reproductive outcome after autocross-linked hyaloronic acid gel application in infertile patients who underwent after laparoscopic myomectomy. Fertil Steril. 2005;83:498–500. doi: 10.1016/j.fertnstert.2004.09.019. [DOI] [PubMed] [Google Scholar]
- e34.DiZerega GS, Coad J, Donnez J. Clinical evaluation of endometriosis and differential response to surgical therapy with and without application of Oxiplex/AP* adhesion barrier gel. Fertil Steril. 2007;87:485–489. doi: 10.1016/j.fertnstert.2006.07.1505. [DOI] [PubMed] [Google Scholar]
- e35.Diamond MP. The Seprafilm Adhesion Study Group. Reduction of adhesions after uterine myomectomy by Seprafilm membrane (HAL-F): a blinded, prospective, randomized, multicenter clinical study. Fertil Steril. 1996;66:904–910. [PubMed] [Google Scholar]
- e36.Becker JM, Dayton MT, Fazio VW, et al. Prevention of postoperative abdominal adhesions by a sodium hyaluronate-based bioresorbable membrane: a prospective, randomized, double-blind multicenter study. J Am Coll Surg. 1996;183:297–306. [PubMed] [Google Scholar]
- e37.Vrijland WW, Tseng LN, Eijkman HJ, et al. Fewer intraperitoneal adhesions with use of hyaluronic acid-carboxymethylcellulose membrane: a randomized clinical trial. Ann Surg. 2002;235:193–199. doi: 10.1097/00000658-200202000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e38.Fazio VW, Cohen Z, Fleshman JW, et al. Reduction in adhesive small-bowel obstruction by Seprafilm adhesion barrier after intestinal resection. Dis Colon Rectum. 2006;49:1–11. doi: 10.1007/s10350-005-0268-5. [DOI] [PubMed] [Google Scholar]
- e39.Mettler L, Audebert A, Lehmann-Willenbrock E, Schieve K, Jacobs VR. Prospective clinical trial of SpayGel as a barrier to adhesion formation: an interim analysis. J Am Assoc Gynecol Laparosc. 2003;10:339–344. doi: 10.1016/s1074-3804(05)60258-7. [DOI] [PubMed] [Google Scholar]
- e40.Ferland R, Campbell PK. Pre-clinical evaluation of a next-generation spray adhesion barrier for multiple site adhesion protection. Surg Technol Int. 2009;18:137–143. [PubMed] [Google Scholar]