Abstract
Background
Variations of porB1A and porB1B genes and their serotypes exist in Neisseria gonorrhoeae isolates from different geographical areas, and some site mutations in the porB1B gene correlate with drug resistance.
Methods
The β-lactamase production of N. gonorrhoeae isolates was determined by paper acidometric test and nitrocefin discs. The porB1A and porB1B genes of 315 non-penicillinase-producting N. gonorrhoeae (non-PPNG) strains were amplified by PCR for sequencing to determine serotypes and site mutations. A duplex PCR was designed to simultaneously detect both porB1A and porB1B genes. Penicillin and tetracycline resistance was assessed by an in vitro drug sensitivity test.
Results
Of the N. gonorrhoeae isolates, 31.1% tested positive for porB1A and 68.9% for porB1B genes. All the 98 porB1A+ isolates belonging to IA6 serotype with either no mutation at the 120 and 121 sites (88.8%) or a D120G (11.2%) mutation and were no resistance to both penicillin and tetracycline. Among the 217 porB1B+ isolates, 26.7%, 22.6% and 11.5% belonged to IB3, IB3/6 and IB4 serotypes, respectively. Particularly, two novel chimeric serotypes, IB3/6-IB2 and IB2-IB4-IB2, were found in 77 and 8 porB1B+ isolates. Two hundred and twelve (97.7%) of the porB1B+ isolates were presented G120 and/or A121 mutations with 163 (76.9%) at both sites. Interestingly, within the 77 porB1B+ isolates belonging to IB3/6-IB2 serotype, 15 were discovered to possess novel deletions at both A121 and N122 sites. All the replacement mutations at these sites in PorB1B were correlated with resistance and the deletion mutation showed the highest resistance.
Conclusion
N. gonorrhoeae isolates circulating in Eastern China include a sole PorB1A serotype (IA6) and five PorB1B serotypes. Multiple mutations in porB1B genes, including novel A121 and N122 deletions, are correlated with high levels of penicillin and tetracycline resistance.
Background
Gonorrhoea caused by infection with Neisseria gonorrhoeae is a global sexually transmitted disease. In Chinese populations, gonorrhoea is the most common sexually transmitted disease which causes a serious public health problem [1-5].
The outer membrane of N. gonorrhoeae bears many proteins such as porins that have been studied in considerable detail [6-10]. Gonococcal porins are a group of outer membrane proteins that occur in large amounts on the surface [11-17]. PorB1A and PorB1B porins, which have 65-80% amino acid identity, share 60% of the total gonococcal porin proteins [12,18]. PorB1A and PorB1B are encoded by the same allele and any particular N. gonorrhoeae strain expresses either PorB1A or PorB1B [12,19,20]. PorB1A is present in 10-30% of N. gonorrhoeae isolates, while PorB1B occurs in 70-90% [21,22]. PorB1A and PorB1B are the serotyping basis of N. gonorrhoeae and mutations are more common in porB1B gene than in porB1A gene [19,22,23]. Many investigations revealed geographical diversity of the predominant porB1A and porB1B genotypes isolates in different areas [22,24-27]. Therefore, determination of porB1A and porB1B genotype distribution in N. gonorrhoeae isolates in different areas is very important for providing a high index of discrimination of different gonococcal strains, identifying the circulating strains and predominant serotypes, and tracking strain transmission in sexual contacts [28-32].
N. gonorrhoeae easily develops resistance to many antibiotics. Previous studies demonstrated that the replacement mutations at the 120 and 121 sites in PorB1B protein enable these strains to increase their resistance to penicillin and tetracycline [33,34]. However, replacement mutations at the 120 and 121 sites in PorB1A make little contribution to resistance against the two antibiotics [9,19]. Thus, identification of the two resistance-related site mutations in PorB1B sequences of N. gonorrhoeae isolates, and determination of the correlation between the site mutations and drug resistance are important for chemotherapy of gonorrhoea in the clinical setting. In the present study, we established a duplex polymerase chain reaction (PCR) system to rapidly confirm the porB1A and porB1B genes in N. gonorrhoeae isolates from Chinese patients. And the predominant serotypes based on porB1A and porB1B genotyping of the isolates circulating in Eastern China were determined by sequence analysis. Particularly, mutation at the 120 and 121 sites in PorB1B sequence from the non-penicillinase-producting N. gonorrhoeae (non-PPNG) isolates, as well as the correlation between the mutation patterns and the resistance to penicillin and tetracycline, were investigated.
Methods
Ethics statement
This research was conducted in accordance with the Declaration of Helsinki, and informed consent was obtained from all patients in this study for collection of clinical specimens to isolate N. gonorrhoeae strains, according to a protocol approved by the Ethics Committee of Zhejiang University.
Bacterial strains and growth
The clinical N. gonorrhoeae strains were isolated from gonorrhea patients from 2005 to 2008 by the clinical laboratories of several hospitals in Zhejiang and Jiangsu provinces, China. All the gonococcal isolates were first identified by the hospital laboratories and subsequently rechecked by our laboratory using microscopy after Gram staining plus oxidase, catalase, and carbohydrate degradation tests [35]. The gonococci were cultured on GCB blood agar plates (bioMérieux, Co., Ltd, Shanghai, China) at 37°C in the presence of 5% CO2 for 24 h. Staphylococcus aureus ATCC25923, Staphylococcus epidermidis ATCC12228, Streptococcus pyogenes ATCC29212, Escherichia coli ATCC25922, Pseudomonas aeruginosa ATCC27543, Klebsiella pneumoniae ATCC700603, Serratia marcescens ATCC14041, Enterobacter cloacae ATCC13047 and Proteus mirabilis ATCC25933 were used to determine the specificity of duplex PCR, and were provided by the Chinese National Institute for Control of Pharmaceutical and Biological Products, and cultured in MH broth (bioMrieux) at 37°C for 24 h.
β-lactamase detection
Paper acidometric test was applied by the clinical laboratories of hospitals to primarily detect the β-lactamase production of gonococcal isolates [5], and subsequently BBL(tm) Cefinase(tm) Paper Disc (BD, USA) based on nitrocefin coloration was used by our laboratory to further determine the production of gonococcal β-lactamase [36]. Among all the N. gonorrhoeae isolates, 315 strains were identified as non-PPNG that were used in this study.
DNA preparation
The gonococci and the other bacteria were collected by centrifugation and washed twice with phosphate buffered saline (PBS). Genomic DNA of each precipitated strain was extracted with a bacterial genomic DNA extraction kit (BioColor Inc., Shanghai, China) and then dissolved in TE buffer for detecting the DNA concentration with ultraviolet (UV) spectrophotometry [37].
Amplification of entire porB1A and porB1B genes
One pair of forward (porB1A/1B-F) and reverse (porB1A/1B-R) primers was used to amplify both the entire porB1A and porB1B genes in genomic DNA of the gonococcal isolates because they have the same nucleotide sequences at the 5' and 3' terminals (Table 1) [19,38,39]. A High Fidelity PCR Kit (TaKaRa Co., Ltd, Dalian, China), in which a Taq-Pfu mixture was used as the DNA polymerase, was used to amplify the two target genes. PCR was initiated by incubation at 94°C for 5 min, followed by 30 cycles at 94°C for 30 s, 54°C for 30 s and 72°C for 90 s to amplify each the two genes, and then incubation at 72°C for 10 min. The products in 1.5% ethidium bromide pre-stained agarose gel after electrophoresis were observed under UV light. The target products are predicted to be 981 bp (entire porB1A gene) and 1044 bp (entire porB1B gene).
Table 1.
Primer sequences used in PCR for amplifying porB1A and porB1B genes
| Primers | Sequences (5' to 3')* | Products and sizes |
|---|---|---|
| porB1A/1B | F: ATGAAAAAATCCCTGATTGCC | 981 bp for entire porB1A gene, and 1044 bp for entire porB1B gene |
| R: TTAGAATTTGTGGCGCAGAAC | ||
| porB1A/1B-D | F1: GCCATTTGGCAGTTGGAACA | 520 bp for partial sequence of porB1A gene, and 201 bp and 583 bp for partial sequence of porB1B gene |
| F2: GATACGGCGAAGGCACTAAA | ||
| R: CTTCGGTTTGAGAGTTGTGC |
* F: forward primer, R: reverse primer
Sequencing and genotyping
The PCR products of the entire porB1A and porB1B genes from the N. gonorrhoeae isolates were purified with a PCR products purification kit (BioColor) and then ligated into plasmid pMD-18-T to form recombinant pMD-18-TporB1A and pMD-18-TporB1B with a T-A cloning kit (TaKaRa). The inserted target segments in recombinant plasmids were sequenced using the double-stranded dideoxy chain termination method by Invitrogen Co., Ltd, Shanghai, China. The obtained sequencing data were analyzed as well as compared to the sequences of PorB1A and PorB1B serotypes published in GenBank using Clustalx software.
Duplex PCR
The sequencing data of porB1A and porB1B genes from the N. gonorrhoeae isolates showed that these genes have over 80% identity at the 5' terminal sequences (1-240 bp segments in both genes) and approximately 90% identity at the 3' terminal sequences (714-981 bp segment in porB1A gene and 777-1044 bp segment in porB1B gene). However, a 63 bp segment in the middle region of porB1B gene that was absent in porB1A gene enabled us to design a specific forward primer only for detecting the porB1B gene. Thus, by using two different forward primers (20 pmol porB1A/1B-D-F1 and 20 pmol porB1A/1B-D-F2) and one common reverse primer (20 pmol porB1A/1B-D-R) (Table 1), a duplex PCR system was established to simultaneously detect the porB1A and porB1B genes in the gonococcal isolates. The total volume per duplex PCR was a 50 μl mixture in which 100 ng DNA template was used. The PCR was initiated by incubation at 94°C for 3 min, followed by 35 cycles at 94°C for 30 s, 54°C for 30 s and 72°C for 60 s, and then incubation at 72°C for 7 min. In agarose gel, the porB1A gene product presented a 520 bp fragment and the porB1B gene product showed two fragments of 201 bp and 583 bp.
Drug sensitivity test
Susceptibility of the N. gonorrhoeae isolates to penicillin (bioMérieux) and tetracycline (bioMérieux) was assessed on GC agar plates (bioMérieux) by the standard proportion method [33,34]. Briefly, each freshly cultured isolate was suspended in GC broth to a density of 104/μl, and 5 μl of gonococcal suspension was spotted onto GC agar plates containing increasing concentrations of penicillin or tetracycline (0.03, 0.06, 0.12, 0.25, 0.5, 1, 2, 4, 8, 16, 32, 64 and 128 μg/ml). After inoculation, the plates were incubated at 37°C in 5% CO2 for 24 h. The minimal inhibitory concentration (MIC) was defined as the minimum concentration of antibiotic at which no more than 5 colonies were observed after incubation. A MIC value ≥2 mg/L was considered as resistance to the antibiotics [40,41]. This test was repeated as three independent experiments.
Results
Sensitivity and specificity of duplex PCR
In our duplex PCR, porB1A and porB1B gene segments were effectually amplified when using a DNA concentration of N. gonorrhoeae as low as 10 ng, and negative results were shown when using 10 to 500 ng DNA templates of S.aureus ATCC25923, S. epidermidis ATCC12228, S. pyogenes ATCC29212, E. coli ATCC25922, P. aeruginosa ATCC27543, K. pneumoniae ATCC700603, S. marcescens ATCC14041, E. cloacae ATCC13047 and P. mirabilis ATCC25933.
Distribution of porB1A and porB1B genes in N. gonorrhoeae isolates
Of the 315 N. gonorrhoeae isolates tested, 31.1% (98/315) and 68.9% (217/315) were found by separate PCR to possess porB1A and porB1B genes, respectively. The duplex PCR established in this study accurately distinguished the porB1A and porB1B genes (Figure 1), and presented the same positive rates for porB1A (31.1%) and porB1B (68.9%) genes in the same gonococcal isolates.
Figure 1.
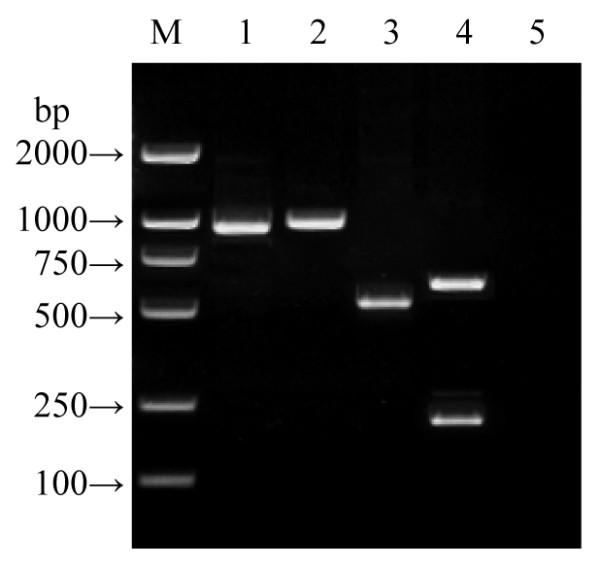
Amplicons of entire or partial porB1A and porB1B genes. Note: Lane M: DNA marker (BioColor); Lanes 1 and 2: amplicons of entire porB1A gene (981 bp) and porB1B gene (1044 bp), respectively; Lanes 3 and 4: amplicons of partial porB1A gene (520 bp) and partial porB1B gene (583 bp and 201 bp) by duplex PCR, respectively; Lane 5: blank control.
porB1A serotype in N. gonorrhoeae isolates
Compared to the porB1A sequences belonging to different PorB1A serotypes in GenBank, all the 98 porB1A+ isolates had 99.47% to 100% nucleotide sequence identity and 99.04% to 100% amino acid sequence identity to the IA-6 serotype (GenBank accession No.: L19962, IA-6 serotype) (Figure 2) [19].
Figure 2.
Amino acid sequences from porB1A genes of N. gonorrhoeae isolates. Note: (IA6): reported amino acid sequence of IA6 serotype (GenBank accession No: L19962); (1) and (2): PorB1A sequences from 56 and 23 gonococcal isolates, respectively; (3) to (5): PorB1A sequences from 8 gonococcal isolates; (6) PorB1A sequences from 11 gonococcal isolates. Underlined areas indicate positions of primers.
porB1B serotypes in N. gonorrhoeae isolates
Compared to the reported porB1B sequences belonging to different serotypes in GenBank [31,32], 26.7% (58/217), 22.6% (49/217) and 11.5% (25/217) of the 217 porB1B+ gonococcal isolates belonged to serotypes IB3, IB3/6, and IB4, respectively. Some (35.5%; 77/217) of the porB1B+ isolates were chimeras of the IB3/6 and IB2 serotypes (IB3/6-IB2 serotype) in which about half the sequences from the 5' terminal were like the IB3/6 serotype and the rest were like the IB2 serotype. Also, 3.7% (8/217) of the porB1B+ isolates were chimeras of the IB2 and IB4 serotypes (IB2-IB4-IB2 serotype) in which the sequence in the middle was like the IB4 serotype but those at the two terminals were like the IB2 serotype (Figure 3).
Figure 3.
Amino acid sequences from porB1B genes of N. gonorrhoeae strains. Note: (IB3), (IB3/6), (IB2) and (IB4): reported PorB1B sequences belonging to serotypes IB3 (GenBank accession No: U75639), IB3/6 (GenBank accession No: U75641), IB2 (GenBank No: U75640) and IB4 (GenBank No: AF090797), respectively; (1): PorB1B sequence from 58 gonococcal isolates referring to IB3 serotype; (2): PorB1B sequence from 49 gonococcal isolates referring to IB3/6 serotype (D means the 27 isolates with A121D mutation, the 19 isolates with A121G mutation and the remaining 3 isolates with no mutation at the A121 site); (3): PorB1B sequence from 62 gonococcal isolates referring to IB3/6-IB2 chimeric serotype without A121 and N122 deletion (K means the 53 strains with G120K mutation and the remaining 9 strains with G120N mutation); (4): PorB1B sequence from 15 gonococcal isolates referring to IB3/6-IB2 chimeric serotype with A121 and N122 deletions; (5): PorB1B sequence from 25 gonococcal isolates referring to IB4 serotype; (6): PorB1B sequence from 8 gonococcal isolates referring to IB2-IB4-IB2 chimeric serotype. Underlined areas indicate the positions of primers. The signal "/" means lack of the amino acid residue.
Mutations at the 120 and 121 sites in PorB1A and PorB1B
Mutations at the 120 and 121 sites in PorB1A and PorB1B sequences from all 315 N. gonorrhoeae isolates are listed in Table 2. In the 98 porB1A+ isolates, 88.8% (87/98) had D120 and G121 in the PorB1A sequences which is identical to those in the reported PorB1A sequence belonging to the IA-6 serotype (GenBank accession No.: L19962) [19], and the remaining 11 porB1A+ isolates had a D120G mutation alone (Figure 2). In the 217 porB1B+ gonococcal isolates, only 2.3% (5/217, three belonging to the IB3/6 serotype and two belonging to the IB3 serotype) had no mutation at the 120 and 121 sites (G120 and A121), while 97.7% (212/217) showed various mutations at the sites. Particularly, in the 212 porB1B+ isolates with the site mutations, 15 isolates (6.9%, 15/217) belonging to the IB3/6-IB2 serotype had a deletion of the amino acid residues at both 121 and 122 sites (Figure 3).
Table 2.
G120 and A121 mutations in PorB1A and PorB1B of N. gonorrhoeae isolates*
| Serotypes | Cases (n) | Site mutation patterns (n) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| G120K/A121D | G120K/A121G | G120N/A121D | G120D/A121G | G120D | G120K | G120N | A121G | ||
| IB3 | 56 | 56 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| IB3/6 | 46 | 27 | 19 | 0 | 0 | 0 | 0 | 0 | 0 |
| IB4 | 25 | 0 | 0 | 0 | 0 | 25 | 0 | 0 | 0 |
| IB3/6-IB2 | 62 | 44 | 0 | 9 | 0 | 0 | 9 | 0 | 0 |
| IB3/6-IB2# | 15 | 0 | 0 | 0 | 0 | 0 | 0 | 15 | 0 |
| IB2-IB4-IB2 | 8 | 8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| IA6 | 98 | 0 | 0 | 0 | 87 | 0 | 0 | 0 | 11 |
| Total | 310 | 135 | 19 | 9 | 87 | 25 | 9 | 15 | 11 |
*: 5 isolates (3 for IB3/6 and 2 for IB3 serotypes) without G120 and A121 mutations not included. G, K, A, D and N indicate glycine, lysine, aspartic acid and asparagine, respectively. #: isolates with deletions of both A121 and N122.
Correlation between resistance and mutations at 120 and 121 sites in PorB1A and PorB1B
The MICs of penicillin and tetracycline for all the 98 porB1A+ gonococcal isolates were 0.06-1 mg/L (<2 mg/L), while the MICs for both antibiotics in the 5 porB1B+ gonococcal isolates with no mutations at the 120 and 121 sites in PorB1B were 0.12-0.5 mg/L (<2 mg/L). However, the MICs of penicillin and tetracycline in the 197 porB1B+ isolates with replacement mutations at the 120 and/or 121 sites were 2-8 mg/L and 2-16 mg/L, respectively, and the 15 porB1B+ isolates with deletion mutations at both the 121 and 122 sites had the highest resistance to penicillin (MICs = 4-8 mg/L) and tetracycline (both MICs = 4-16 mg/L) (Table 3). According to the MIC value (≥2 mg/L) that defines resistance to penicillin and tetracycline, all the porB1A+ isolates and the 5 porB1B+ isolates without site mutations were no resistance to both antibiotics, while all the remaining 212 porB1B+ isolates with different site mutations were penicillin and tetracycline resistant.
Table 3.
MICs of penicillin and tetracycline for N. gonorrhoeae isolates
| Site mutations* | Serotypes | Cases (n) | MICs (mg/L) | |
|---|---|---|---|---|
| Penicillin | Tetracycline | |||
| G120K/A121D | IB3 | 56 | 2-4 | 2-8 |
| IB3/6 | 27 | 2-4 | 2-8 | |
| IB3/6-IB2 | 44 | 2-4 | 2-8 | |
| IB2-IB4-IB2 | 8 | 2-4 | 2-4 | |
| G120K/A121G | IB3/6 | 19 | 2-4 | 2-8 |
| G120N/A121D | IB3/6-IB2 | 9 | 2 | 2-4 |
| G120D | IB4 | 25 | 2-4 | 2-8 |
| G120K | IB3/6-IB2 | 9 | 2-4 | 2-8 |
| G120N/A121 and N122 deletion | IB3/6-IB2 | 15 | 4-8 | 4-16 |
| G120G/A121A | IB3/6 | 3 | 0.12 | 0.25 |
| IB3 | 2 | 0.25 | 0.25 | |
| D120D/G121G | IA6 | 87 | 0.06-0.5 | 0.06-0.5 |
| D120G/G121G | IA6 | 11 | 0.12-0.25 | 0.12-1 |
*: G, K, A, D and N indicate glycine, lysine, aspartic acid and asparagine, respectively.
Discussion
The porins of N. gonorrhoea stimulate the host immune system to produce specific antibodies which activate the complement system and promote phagocytosis to eliminate the invading gonococci [9,42]. Furthermore, gonococcal PorB1A and PorB1B are closely associated with transmission of gonorrhea [38,42], and mutations at the 120 and 121 sites in PorB1B are related to drug resistance [9,19,33,34]. Thus, determination of the predominance of PorB1A and PorB1B serotypes and drug resistance-associated mutations in the porB1B gene in different areas is important for serological diagnosis and chemotherapy of gonorrhoea.
For rapid and convenient discrimination of porB1A and porB1B genes in N. gonorrhoeae isolates, we designed a duplex PCR system by which both genes were simultaneously detectable. The results indicated that the duplex PCR accurately recognizes the porB1A and porB1B genes in all the 315 tested isolates with high sensitivity and specificity. By using the duplex PCR, we found 31.1% of the 315 isolates tested were positive for the porB1A gene and the porB1B gene was detectable in 68.9% of the isolates. This porB1A+ and porB1B+ proportion in N. gonorrhoeae isolates is close to that of previous reports [21,22].
The gonococcal porB1B gene differs from the porB1A gene in having many more mutations in its sequence [19,38,39]. Among the 217 porB1B+ isolates in this study, 77 (35.5%), 58 (26.7%), and 49 (22.6%) isolates belonged to the IB3/6-IB2, IB3 and IB3/6 serotypes, and only 25 (11.5%) and 8 (3.7%) referred to the IB4 and IB2-IB4-IB2 serotypes, respectively. These data indicate that the predominant serotypes of porB1B+ N. gonorrhoeae in Eastern China are IB3/6-IB2, IB3 and IB3/6 (84.8%, 184/217), which quite differs from the IB2, IB4 and IB3 reported as the predominant serotypes in nations in Europe and North America [19,22-24]. Bash and colleagues (2005) showed that the porB1B gene has a high recombinant mutation frequency compared to the porB1A gene [43]. In our study, two novel recombinant mutations formed IB3/6-IB2 and IB2-IB4-IB2 chimeric serotypes, which also indicates a high frequency of recombination among different gonococcal porB1B genes. There have been reports of N. gonorrhoeae strains that react to both PorB1A and PorB1B antibodies, raising the possibility of mosaicism between porB1A and porB1B alleles [44,45]. However, no such mosaicism was found in all the N. gonorrhoeae isolates tested in this study.
Olesky et al. (2002) used the site mutation technique to generate mutants at the 120 and 121 sites in PorB1B, and found stronger resistance to penicillin and tetracycline in the G120K, G120D/A121D, G120K/A121R and G120P/A121P mutants [46]. Among the 217 porB1B+ isolates in this study, only five had no mutations at the 120 and 121 sites, while the other 212 had various mutations at these sites, in which 76.9% (163/212) had double-site mutations (G120K/A120D, G120K/A121G and G120N/A121D in 135, 19 and 9 isolates). All the 212 porB1B+ isolates had a mutation at the G120 site replaced with lysine (K) (76.9%, 163/212), aspartate (D) (11.8%, 25/212) or asparagine (N) (11.3%, 24/212). On the other hand, 76.9% of the 212 porB1B+ strains had a mutation at the A121 site replaced with aspartate (D) (88.3%, 144/163) or glycine (G) (11.7%, 19/163). However, the G120R, G120P, A121H and A121P mutations described by Olesky et al were not found, suggesting differences in the amino acids replacement by natural and artificial mutations. Particularly, in this study we found that 15 gonococcal isolates had A121 and N122 deletion mutations in PorB1B that had not previously been reported.
Many previous data revealed that plasmid-mediated resistance of N. gonorrhoeae confer the very high MICs of penicillinand tetracycline [47-49], while chromosomally mediated resistance such as the site mutations at G120 or A121 in gonococcal PorB1B contributes to the moderate higher MICs of the two antibiotics [33,34]. According to our drug sensitivity tests for the non-PPNG isolates, the G120K/A121D, G120K/A121G, G120N/A121D, G120D and G120K natural porB1B mutants also had stronger resistance to penicillin and tetracycline than porB1B+ isolates with no G120 and A121 mutations (Table 3). Interestingly, the 15 porB1B+ isolates with A121 and N122 deletions had the highest MICs, which hints at a greater influence of the A121 deletion on resistance than any of the replacement mutations. This resistance relating to chromosomally-mediated site mutations in PorB1B has been shown in many previous reports [33,46,50-52]. However, in these reports the PorB1B mutation-associated resistance only noted MICs of penicillin and tetracycline in the range of 2-8 μg/ml. Previous data revealed that site mutations in the penA, mtrR, ponA, and rpsJ genes also participate in chromosomally-mediated gonococcal resistance to penicillin and tetracycline [47,53-55]. Thus, synergistic action of site mutations in multiple chromosomal genes associated with resistance in N. gonorrhoeae isolates is an important subject for investigation. Such action may be responsible for the high resistance to both antibiotics found in this study.
We found that all the tested porB1A+ gonococcal isolates were no resistance to both penicillin and tetracycline, whereas 97.7% (212/217) of the porB1B+ isolates had site mutation-based resistance in the PorB1B sequences. The high proportion (100%) of resistance-associated mutations at the 120 and 121 sites in PorB1B of N. gonorrhoeae isolates from other Chinese areas has also been reported [51,52,56]. Thus, the duplex PCR established in this study for rapid identification of porB1A+ and porB1B+ gonococci provides a useful tool for selecting antibiotics to treat gonorrhea in China.
Conclusions
The duplex PCR system we designed simultaneously recognizes porB1A and porB1B genes in N. gonorrhoeae isolates with high sensitivity and specificity. porB1B+ N. gonorrhoeae is the predominant genotype in Eastern China. All the porB1A+ isolates had the conserved porB1A gene sequence belonging to the IA6 serotype alone. However, the porB1B+ isolates had high site mutations in the porB1B sequence that resulted in five PorB1B serotypes. All the porB1A+ isolates were no resistance to both penicillin and tetracycline. However, multiple penicillin and tetracycline resistance-related mutations such as G120K/A121D, G120K/A121G and G120N/A121D occurred in PorB1B isolates. A novel mutation due to deletion of both A121 and N122 in PorB1B is correlated with high resistance to penicillin and tetracycline.
List of Abbreviations
STD: sexual transmitted disease; G: glycine; D: aspartic acid; K: lysine; R: arginine; P: proline; A: alanine; H: histidine; N: asparagine; PCR: polymerase chain reaction; DNA: deoxyribonucleic acid; PBS: phosphate buffered saline; TE: Tris-EDTA; Tris: trihydroxymethyl aminomethane; EDTA: ethylenediamine tetraacetic acid; UV: ultraviolet rays; MIC: minimal inhibitory concentration
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
AS participated in the experimental design, obtained funding for the study, performed PCR and drafted the manuscript. XF supervised the collection of gonococcal isolates, and helped to draft the manuscript. PD analyzed the sequencing data. YG helped to draft the manuscript. RT helped to collect the gonococcal isolates. YM helped to perform PCR. XL carried out the drug sensitivity tests. JY participated in the experimental design and obtained funding for the study. All authors have read and approved the final manuscript.
Pre-publication history
The pre-publication history for this paper can be accessed here:
Contributor Information
Aihua Sun, Email: sunah123@126.com.
Xingli Fan, Email: enterjoyfan@hotmail.com.
Ye Gu, Email: 13588178859@139.com.
Peng Du, Email: pdu1145@163.com.
Renxian Tang, Email: tangrenxian-t@163.com.
Yafei Mao, Email: myf@zju.edu.cn.
Xuai Lin, Email: lxai122@163.com.
Jie Yan, Email: Med_bp@zju.edu.cn.
Acknowledgements
This work was supported by grants from the Science and Technical Research Project of the Health Bureau of Zhejiang Province, China (2004A018), and the State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, the First Affiliated Hospital, Zhejiang University, China (2008ZZ06). We thank the hospitals in Zhejiang Province and Jiangsu Province, China, for isolating and providing the strains of N. gonorrhoeae from patients. We also thank Dr. IC Bruce, Zhejiang University School of Medicine, for reading the manuscript.
References
- WHO. Global prevalence and incidence of selected curable sexually transmitted infections: overview and estimates. Vol. 2001. Geneva, WHO; pp. 14–19. [PubMed] [Google Scholar]
- Tazil L, Losada MP, Gu WM, Yang Y, Xue L, Crandall KA, Viscidi RP. Population dynamics of Neisseria gonorrhoeae in Shanghai, China: a comparative study. BMC Infectious Diseases. 2010;10:13. doi: 10.1186/1471-2334-10-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Workowski KA, Berman SM. Sexually transmitted diseases treatment guidelines. MMWR Recomm Rep. 2006;55:1–94. [PubMed] [Google Scholar]
- Low N, Broutet N, Sarkodie YA, Barton P, Hossain M, Hawkes S. Global control of sexually transmitted infections. Lancet. 2006;368:2001–2016. doi: 10.1016/S0140-6736(06)69482-8. [DOI] [PubMed] [Google Scholar]
- Su X, Jing F, Qimuge, Dia X, Sun H, Ye S. Surveillance of antimicrobial susceptibilities in Neisseria gonorrhoeae in Nanjing, china, 1999-2006. Sex Transm Dis. 2007;34:995–999. doi: 10.1097/OLQ.0b013e3180ca8f24. [DOI] [PubMed] [Google Scholar]
- Swanson J. Surface-exposed protein antigens of the gonococcal outer membrane. Infect Immun. 1981;34:804–816. doi: 10.1128/iai.34.3.804-816.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotschlich EC, Seiff ME, Blake MS, Koomey M. Porin protein of Neisseria gonorrhoeae: cloning and gene structure. Proc Natl Acad Sci USA. 1987;84:8135–8139. doi: 10.1073/pnas.84.22.8135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ley VDP, Heckels JE, Virji M, Hoogerhout P, Poolman JT. Topology of outer membrane porins in pathogenic Neisseria spp. Infect Immun. 1991;59:2963–2971. doi: 10.1128/iai.59.9.2963-2971.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pérez-Losada M, Viscidi RP, Demma JC, Zenilman J, Crandall KA. Population genetics of Neisseria gonorrhoeae in a high-prevalence community using a hypervariable outer membrane porB and 13 slowly evolving housekeeping genes. Mol Biol Evol. 2005;22:1887–1902. doi: 10.1093/molbev/msi184. [DOI] [PubMed] [Google Scholar]
- Zhu WY, Thomas CE, Chen CJ, Van Dam CN, Johnston RE, Davis NL, Sparling PF. Comparison of immune responses to gonococcal PorB delivered as outer membrane vesicles, recombinant protein, or Venezuelan equine encephalitis virus replicon particles. Infect Immun. 2005;73:7558–7568. doi: 10.1128/IAI.73.11.7558-7568.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virji M, Zak K, Heckels JE. Monoclonal antibodies to gonococcal outer membrane protein IB: use in investigation of the potential protective effect of antibodies directed against conserved and type-specific epitopes. J Gen Microbiol. 1986;132:1621–1629. doi: 10.1099/00221287-132-6-1621. [DOI] [PubMed] [Google Scholar]
- Carbonetti NH, Sparling PF. Molecular cloning and characterization of the structural gene for protein I: the major outer membrane protein of Neisseria gonorrhoeae. Genetics. 1987;84:9084–9088. doi: 10.1073/pnas.84.24.9084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbonetti NH, Simnad VI, Seifert HS, So M, Sparling PF. Genetics protein I of Neisseria gonorrhoeae: construction of hybrid porins. Genetics. 1988;85:6841–6845. doi: 10.1073/pnas.85.18.6841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckels JE, Fletcher JN, Virji M. The potential protective effect of immunization with outer-membrane protein I from Neisseria gonorrhoeae. J Gen Microbiol. 1989;135:2269–2276. doi: 10.1099/00221287-135-8-2269. [DOI] [PubMed] [Google Scholar]
- Elkins C, Carbonetti NH, Varela VA, Stirewalt D, Klapper DG, Sparling PF. Antibodies to N-terminal peptides of gonococcal porin are bactericidal when gonococcal lipopolysaccharide is not sialylated. Mol Microbiol. 1992;6:2617–2628. doi: 10.1111/j.1365-2958.1992.tb01439.x. [DOI] [PubMed] [Google Scholar]
- Elkins C, Barkley KB, Carbonetti NH, Stirewalt D, Sparling PF. Immunobiology of purified recombinant outer membrane porin protein I of Neisseria gonorrhoeae. Mol Microbiol. 1994;14:1059–1075. doi: 10.1111/j.1365-2958.1994.tb01339.x. [DOI] [PubMed] [Google Scholar]
- Swanson J, Dorward D, Lubke L, Kao D. Porin polypeptide contributes to surface charge of gonococci. J Bacteriol. 1997;179:3541–3548. doi: 10.1128/jb.179.11.3541-3548.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kühlewein C, Rechner C, Meyer TF, Rudel T. Low-phosphate-dependent invasion resembles a general way for Neisseria gonorrhoeae to enter host cells. Infect Immun. 2006;74:4266–4273. doi: 10.1128/IAI.00215-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mee BJ, Thomas H, Cooke SJ, Lambden PR, Heckels JE. Structural comparison and epitope analysis of outer-membrane protein PIA from strains of Neisseria gonorrhoeae with differing serovar specificities. J Gen Microbiol. 1993;139:2613–2620. doi: 10.1099/00221287-139-11-2613. [DOI] [PubMed] [Google Scholar]
- Unemo M, Olcén P, Jonasson J, Fredlund H. Molecular typing of Neisseria gonorrhoeae isolates by pyrosequencing of highly polymorphic segments of the porB gene. J Clin Microbiol. 2004;42:2926–2934. doi: 10.1128/JCM.42.7.2926-2934.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooke SJ, Jolley K, Ison CA, Young H, Heckels JE. Naturally occurring isolates of Neisseria gonorrhoeae, which display anomalous serovar properties, express PIA/PIB hybrid porins, deletions in PIB or novel PIA molecules. FEMS Microbiol Lett. 1998;162:75–82. doi: 10.1111/j.1574-6968.1998.tb12981.x. [DOI] [PubMed] [Google Scholar]
- IIina EN, Malakhova MV, Vereshochagin VA, Govorun VM, Sergienko VI, Zubkov MM, Vasilev MM, Kubanova AA. Molecular typing of N. gonorrhoeae strains prevalent in the Russian Federation. Bull Exp Biol Med. 2003;136:179–182. doi: 10.1023/A:1026379209810. [DOI] [PubMed] [Google Scholar]
- Mcknew DL, Lyun F, Zenilman JM, Bash MC. Porin variation among clinical isolates of Neisseria gonorrhoeae over a 10-year period, as determined by Por variable region typing. J Infect Dis. 2003;187:1213–1222. doi: 10.1086/374563. [DOI] [PubMed] [Google Scholar]
- Knapp JS, Tam MR, Nowinski RC, Holmes KK, Sandström EG. Serological classification of Neisseria gonorrhoeae with use of monoclonal antibodies to gonococcal outer membrane protein I. J Infect Dis. 1984;150:44–48. doi: 10.1093/infdis/150.1.44. [DOI] [PubMed] [Google Scholar]
- Ison CA. Genotyping of Neisseria gonorrhoeae. Curr Opin Infect Dis. 1998;11:43–46. doi: 10.1097/00001432-199802000-00010. [DOI] [PubMed] [Google Scholar]
- Thompson DK, Deal CD, Ison CA, Zenilman JM, Bash MC. A typing system for Neisseria gonorrhoeae based on biotinylated oligonucleotide probes to PIB gene variable regions. J Infect Dis. 2000;181:1652–1660. doi: 10.1086/315464. [DOI] [PubMed] [Google Scholar]
- Unemo M, Olcn P, Albert J, Fredlund H. Comparison of serologic and genetic porB-based typing of Neisseria gonorrhoeae: consequences for future characterization. J Clin Microbiol. 2003;41:4141–4147. doi: 10.1128/JCM.41.9.4141-4147.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unemo M, Vorobieva V, Firsova N, Ababkova T, Leniv I, Haldorsen BC, Rfedlund H, Skogen V. Neisseria gonorrhoeae population in Arkhangelsk, Russia: phenotypic and genotypic heterogeneity. Clin Microbiol Infect. 2007;13:873–878. doi: 10.1111/j.1469-0691.2007.01764.x. [DOI] [PubMed] [Google Scholar]
- Viscidi RP, Demma JC, Gu J, Zenilman J. Comparison of sequencing of the por gene and typing of the opa gene for discrimination of Neisseria gonorrhoeae strains from sexual contacts. J Clin Microbiol. 2000;38:4430–4438. doi: 10.1128/jcm.38.12.4430-4438.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unemo M, Olcen P, Berglund T, Albert J, Fredlund H. Molecular epidemiology of Neisseria gonorrhoeae: sequence analysis of the porB gene confirms presence of two circulating strains. J Clin Microbiol. 2002;40:3741–3749. doi: 10.1128/JCM.40.10.3741-3749.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hobbs MM, Alcom TM, Davis RH, Fischer W, Thomas JC, Martin I, Ison C, Sparling PF, Cohen MS. Molecular typing of Neisseria gonorrhoeae causing repeated infections: evolution of porin during passage within a community. J Infect Dis. 1999;179:371–381. doi: 10.1086/314608. [DOI] [PubMed] [Google Scholar]
- Lynn F, Hobbs MM, Zenilman JM, Behets FM, Van Damme K, Rasamindrakotroka A, Bash MC. Genetic typing of the porin protein of Neisseria gonorrhoeae from clinical noncultured samples for strain characterization and identification of mixed gonococcal infections. J Clin Microbiol. 2005;43:368–375. doi: 10.1128/JCM.43.1.368-375.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin IM, Ison CA, Aanensen DM, Fenton KA, Spratt BG. rapid sequence-based indentification of gonococcal transmission clusters in a large metropolitan area. J Infect Dis. 2004;189:1497–505. doi: 10.1086/383047. [DOI] [PubMed] [Google Scholar]
- Gill MJ, Simjee S, Al-Hattawi K, Robertson BD, Easmon CS, Ison CA. Gonococcal resistance to β-lactams and tetracycline involves mutation in loop 3 of the porin encoded at the penB locus. Antimicrob Agents Chemother. 1998;42:2799–2803. doi: 10.1128/aac.42.11.2799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olesky M, Zhao S, Rosenberg RL, Nicholas RA. Porin-mediated antibiotic resistance in Neisseria gonorrhoeae: ion, solute, and antibiotic permeation through PIB proteins with penB mutations. J Bacteriol. 2006;188:2300–2308. doi: 10.1128/JB.188.7.2300-2308.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosa J, Ramirez-Arcos S, Ruben M, Li H, Llanes R, Llop A, Dillon JA. High percentages of resistance to tetracycline and penicillin and reduced susceptibility to azithromycin characterize the majority of strain types of Neisseria gonorrhoeae isolates in Cuba, 1995-1998. Sex Transm Dis. 2003;30:443–448. doi: 10.1097/00007435-200305000-00012. [DOI] [PubMed] [Google Scholar]
- Montgomery K, Raymundo L Jr, Drew WL. Chromogenic cephalosporin spot test to detect betalactamase in clinically significant bacteria. J Clin Microbiol. 1979;9:205–207. doi: 10.1128/jcm.9.2.205-207.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambrook J, Russell DW. Molecular cloning, a laboratory manual. 2. Vol. 7. New York, Gold Spring Harbor Laboratory Press; 2001. pp. 30–7. 35. [Google Scholar]
- Cooke SJ, de la Paz H, La Poh C, Ison CA, Heckels JE. Variation within serovars of Neisseria gonorrhoeae detected by structural analysis of outer-membrane protein PIB and by pulsed-field gel electrophoresis. Microbiology. 1997;143:1415–1422. doi: 10.1099/00221287-143-4-1415. [DOI] [PubMed] [Google Scholar]
- Fudyk TC, Maclean IW, Simonsen JN, Njagi EN, Kimani J, Brunham RC, Plummer FA. Genetic diversity and mosaicism at the por locus of Neisseria gonorrhoeae. J Bacteriol. 1999;181:5591–5599. doi: 10.1128/jb.181.18.5591-5599.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arreaza L, Salcedo C, Alcalá B, Berrón S, Martín E, Vázquez JA. Antibiotic resistance of Neisseria gonorrhoeae in Spain: trends over the last two decades. J Antimicrob Chemother. 2003;51:153–156. doi: 10.1093/jac/dkg034. [DOI] [PubMed] [Google Scholar]
- Ng LK, Martin I, Lau A. the National Gonococcal Surveillance Program Members. Trends of chromosomally mediated antimicrobial resistance in Neisseria gonorrhoeae in Canada: 1994-1999. Sex Transm Dis. 2003;30:896–900. doi: 10.1097/01.OLQ.0000099560.11350.1A. [DOI] [PubMed] [Google Scholar]
- Zhu W, Thomas CE, Sparling PF. DNA immunization of mice with a plasmid encoding Neisseria gonorrhea PorB protein by intramuscular injection and epidermal particle bombardment. Vaccine. 2004;22:660–669. doi: 10.1016/j.vaccine.2003.08.036. [DOI] [PubMed] [Google Scholar]
- Bash MC, Zhu P, Gulati S, McKnew D, Rice PA, Lynn F. por Variable-region typing by DNA probe hybridization is broadly applicable to epidemiologic studies of Neisseria gonorrhoeae. J Clin Microbiol. 2005;43:1522–1530. doi: 10.1128/JCM.43.4.1522-1530.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De La Fuente L, Va'zquez JA. Multilocus enzyme analysis of African type penicillinase-producing Neisseria gonorrhoeae (PPNG) strains isolated in Spain. Sex Transm Dis. 1991;18:150–152. doi: 10.1097/00007435-199107000-00005. [DOI] [PubMed] [Google Scholar]
- Gill MJ, Jayamohan J, Lessing MP, Ison CA. Naturally occurring PIA/PIB hybrids of Neisseria gonorrhoeae. FEMS Microbiol Lett. 1994;119:161–166. doi: 10.1111/j.1574-6968.1994.tb06883.x. [DOI] [PubMed] [Google Scholar]
- Olesky M, Hobbs M, Nicholas RA. Identification and analysis of amino acid mutations in porin IB that mediate intermediate-level resistance to penicillin and tetracycline in Neisseria gonorrhoeae. Antimicrob Agents Chemother. 2002;46:2811–2820. doi: 10.1128/AAC.46.9.2811-2820.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagman KE, Pan W, Spratt BG, Balthazar JT, Judd RC, Shafer WM. Resistance of Neisseria gonorrhoeae to antimicrobial hydrophobic agents is modulated by the mtrRCDE efflux system. Microbiology. 1995;141:611–622. doi: 10.1099/13500872-141-3-611. [DOI] [PubMed] [Google Scholar]
- Lindberg R, Fredlund H, Nicholas R, Unemo M. Neisseria gonorrhoeae isolates with reduced susceptibility to cefixime and ceftriaxone: association with genetic polymorphisms in penA, mtrR, porB1b, and ponA. Antimicrob Agents Chemother. 2007;51:2117–2122. doi: 10.1128/AAC.01604-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao S, Duncan M, Tomberg J, Davies C, Unemo M, Nicholas RA. Genetics of chromosomally mediated intermediate resistance to ceftriaxone and cefixime in Neisseria gonorrhoeae. Antimicrob Agents Chemother. 2009;53:3744–3751. doi: 10.1128/AAC.00304-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodford N, Bindayna KM, Easmon CS. et al. Associations between serotype and susceptibility to antibiotics of Neisseria gonorrhoeae. Genitourin Med. 1989;65:86–91. doi: 10.1136/sti.65.2.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Liao M, Gu W, Bell K, Wu L, Eng NF, Zhang CG, Chen Y, Jolly AM, Dillon JAR. Antimicrobial susceptibility and molecular determinants of quinolone resistance in Neisseria gonorrhoeae isolates from Shanghai. J Antimicrob Chemother. 2006;58:868–872. doi: 10.1093/jac/dkl301. [DOI] [PubMed] [Google Scholar]
- Liao MM, Bell K, Gu WM, Yang Y, Eng NF, Fu WK, Wu L, Zhang CG, Chen Y, Jolly AM, Dillon JAR. Clusters of circulating Neisseria gonorrhoeae strains and association with antimicrobial resistance in Shanghai. J Antimicrobial Chemother. 2008;61:478–87. doi: 10.1093/jac/dkm544. [DOI] [PubMed] [Google Scholar]
- Chalkley LJ, van Vuuren S, Ballard RC, Botha PL. Characterisation of penA and tetM resistance genes of Neisseria gonorrhoeae isolated in southern Africa--epidemiological monitoring and resistance development. S Afr Med J. 1995;85:775–780. [PubMed] [Google Scholar]
- Ropp PA, Hu M, Olesky M, Nicholas RA. Mutations in ponA, the gene encoding penicillin-binding protein 1, and a novel locus, penC, are required for high-level chromosomally mediated penicillin resistance in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2002;46:769–777. doi: 10.1128/AAC.46.3.769-777.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu M, Nandi S, Davies C, Nicholas RA. High-level chromosomally mediated tetracycline resistance in Neisseria gonorrhoeae results from a point mutation in the rpsJ gene encoding ribosomal protein S10 in combination with the mtrR and penB resistance determinants. Antimicrob Agents Chemother. 2005;49:4327–4334. doi: 10.1128/AAC.49.10.4327-4334.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao MM, Helgeson S, Gu WM, Yang Y, Jolly AM, Dillon JAR. Comparison of Neisseria gonorrhoeae multiantigen sequence typing and porB sequence analysis for identification of clusters of N. gonorrhoeae isolates. J Clin Microbiol. 2009;47:489–491. doi: 10.1128/JCM.01612-08. [DOI] [PMC free article] [PubMed] [Google Scholar]




