Abstract
Consistent individual differences in behavioural types may not only cause variation in life-history decisions, but may also affect the choice of social partners and sociality in general. Here, we tested whether and how behavioural type influences the establishment of social ties using the cooperatively breeding cichlid, Neolamprologus pulcher. In a habitat saturation experiment with individuals pre-tested for behavioural type, we first analysed whether behavioural type affected the likelihood of settlement (i.e. social status), group sizes, and the types of dominant and subordinate individuals accepted as group members. Corrected for effects of body size and sex, the behavioural type did not affect settlement. However, bold dominant males only accepted smaller females, and grouped with bold subordinates, while shy dominant males accepted larger females than themselves, and grouped with shy subordinates. Second, we analysed the relationships between behavioural type and the aggressiveness or affiliation social network. Behavioural type significantly affected the number and quality of connections within the two networks. We show that behavioural types affect group composition, social networks and status achieved, in interaction with body size. Thus, the interactions within groups may depend not only on age, size and sex, but also on the behavioural type of the individuals involved.
Keywords: cooperation, social network, body size, status, Cichlidae
1. Introduction
It has been recognized that individual ‘states’ (sensu McNamara & Houston 1996), like dominance status, sex and body size, may influence the likelihood of acquiring and maintaining dominant positions in the future (e.g. van de Pol et al. 2007). However, individuals of the same species may not only differ in their states, but they may also consistently differ in the expression of their behavioural traits across context and/or time, resulting in behavioural types (alternatively known as temperament, coping styles or animal personalities; e.g. Koolhaas et al. 1999; Sih et al. 2004; Réale et al. 2007). Typically, individuals differ consistently in the so-called bold–shy continuum (reviewed in Sih et al. 2004), where shy individuals are both non-aggressive and hardly explore novel environments, and where bold individuals are both aggressive and explorative. The consistency or repeatability of behavioural traits averages at 0.37 (Bell et al. 2009). We refer to these individual differences in the expression of behavioural traits as ‘behavioural types’ (sensu Sih et al. 2004).
Behavioural types might affect social connections with the neighbourhood and eventually may affect future social positions similar to states (e.g. site-related dominance, Heg et al. 2000; also McDonald 2007; Ryder et al. 2008; Krause et al. 2010). Recently, social network theory has provided a comprehensive and detailed tool to describe social connections and how they may affect settlement decisions by individuals (McDonald 2007) or group stability (e.g. Flack et al. 2006) and thus might potentially affect access to reproductive opportunities (e.g. van Oers et al. 2008).
Whether behavioural types may affect social connections and settlement has been little explored (but see Fraser et al. 2001; Pike et al. 2008; Harcourt et al. 2009; Webster et al. 2009; reviewed also in Cote et al. 2010), and even fewer studies have explored effects in a cooperatively breeding species (but see Raihani et al. 2008; Schürch & Heg 2010a). This is unfortunate, because in cooperative breeders one can expect strong effects of the behavioural types on life-history decisions (Wolf et al. 2007), which is likely to coevolve with how they behave in a social context (Schürch & Heg 2010b). For instance, shy, non-explorative and non-aggressive subordinates may benefit from a high likelihood to survive to old age and inherit the breeding position, because they do not engage in risky exploration behaviour (to locate vacancies). They may also avoid eviction from the group because they do not aggressively challenge the dominants inside their group, and by providing help (Bergmüller & Taborsky 2007). On the other hand, bold, explorative and aggressive subordinates who try to disperse early in life may acquire those benefits from independent breeding, but such dispersal might be involuntary owing to aggressive interactions with the dominants and may fail due to predation risk. They might increase their likelihood of early successful dispersal by investing less in costly helpful behaviour and shunt the extra energy into growth and reproductive competition.
Differences in behavioural types in cooperative breeders may immediately affect sociality as well, because certain behavioural types will remain in the natal group, whereas others disperse and form new groups, leading to groups consisting of certain behavioural types (e.g. shy dominants and subordinates, or bold dominants and subordinates), with at present unknown consequences for the degree of sociality in such groups, and eventually with unknown fitness consequences for all group members. For instance, an aggressive and bold disperser forming a new group may experience a spill-over effect at adulthood (sensu Arnqvist & Henriksson 1997) on his likelihood of accepting additional immigrants as subordinate helpers (Schürch & Heg 2010a). Either they are too aggressive to accept any immigrants as helpers or they are more likely to tolerate them because they are able to aggressively dominate immigrants (Schürch & Heg 2010a) and skew reproduction in their own favour (Heg 2008; Heg & Hamilton 2008; Heg et al. 2008a,b). Thereby, through their behaviour subordinate and dominant behavioural types may affect group size (Heg et al. 2005a), group structure (Werner et al. 2003), helper effects (Brouwer et al. 2005), within-group interactions (Hamilton et al. 2005; Mitchell et al. 2009a) and the partitioning of reproduction among the group members (‘reproductive skew’, e.g. Heg 2008; Heg & Hamilton 2008).
The purpose of this paper is to analyse how individual states and behavioural types may interact to determine settlement patterns and the formation of social ties in a highly social cichlid, Neolamprologus pulcher. This species lives in extended family groups (Balshine et al. 2001), where usually the younger subordinate group members are related to the older dominant breeder pair (Dierkes et al. 2005). Subordinates engage in various helping behaviours, such as digging out breeding and hiding shelters, predator defence and caring for eggs (Taborsky & Limberger 1981; Taborsky 1984). Subordinates of both sexes may engage in reproduction as well (e.g. Heg et al. 2006, 2008a,b; Heg 2008; Stiver et al. 2009). Individuals differ consistently in their aggressiveness–boldness–explorativeness axis and helping behaviour (Bergmüller & Taborsky 2007; Heg et al. 2009; Schürch & Heg 2010b). Social status and body sizes of group members (and their differences) profoundly affect the behaviour, survival and reproduction of these cichlids (see references above), which need to be accounted for in any experiment addressing the potential effects of behavioural types.
We performed a habitat saturation experiment (Heg et al. in press), where individuals of different states (body size and sex) and behavioural types (pre-tested for exploration propensity, boldness and aggressiveness) competed for group membership in a large octagonal tank containing 24 breeding patches. Then we analysed the data using two approaches. In the first, more traditional approach, we analysed data on an individual basis and established whether and how the behavioural types of the individuals determined settlement patterns, correcting for effects of state (body size and sex, which are both known to affect sociality in these cichlids, e.g. Mitchell et al. 2009a). Since the effects of behavioural type on sociality in a cooperative breeder have hardly been explored, we did not have any a priori expectations concerning the direction and the magnitude of the effects.
This first approach neglects the fact that individuals differ in the extent and magnitude of social connections they make both within their group and to outsiders. For instance, some dominant males defend multiple dominant females in separate breeding patches both in the field and in the laboratory (Limberger 1983), or engage in reproduction with subordinate females together with dominant females (Heg 2008) or in a separate patch from dominant females (Heg & Hamilton 2008). Some subordinates live more peripheral or central inside their groups (Werner et al. 2003), or regularly make visits to other groups (Bergmüller et al. 2005).
To better account for this complexity, we also used a second approach, using social network theory. Social network analyses allowed us to summarize the social connections made by individuals in a succinct way (e.g. Croft et al. 2008; Whitehead 2008). We derived the network metrics weighted in-degree centrality (number and strength of incoming ties), weighted out-degree centrality (number and strength of outgoing ties) and eigenvector centrality (ties to individuals with many ties add more to eigenvector centrality), from two matrices of directed and weighted dyadic interactions for each trial separately (following Newman 2004): one for aggression and the other for affiliation (see §2). Independent from the effects of state, we expected bold, aggressive and more explorative individuals to obtain a more central and connected position in the aggressiveness networks (i.e. higher levels of degree and eigenvector centrality) compared with shy, non-aggressive and less-explorative individuals. In contrast, we expected the reverse for the affiliation networks. Eventually, the position of individuals within a social network might influence the amount and quality of opportunities to help (e.g. alloparental care). Identifying the factors shaping the pattern of ties within a network might thus shed some light on whether and how behavioural types and the propensity to engage in costly cooperation are related. We predicted that well-connected individuals in the affiliation network (i.e. with higher levels of degree and eigenvector centrality) have more access to breeding shelters, and thus were more likely to engage in cooperative tasks. Hence, we also measured cooperation in the individuals' performance in (allo)parental brood care.
2. Material and methods
(a). Assessment of behavioural type
All behavioural tests were conducted by S.R., who was blind to the sex, social status and treatment of the individuals tested. Exploration propensity was assessed inside a 400 l tank (130 × 65 × 65 cm, 5 cm sand layer). At one short side, the tank contained a home compartment of 30 × 65 × 65 cm with a pot half, separated from the exploration compartment by an opaque partition. The fish were transferred to this home compartment and allowed to acclimatize for 10 min. The opaque partition was removed and the fish were allowed to explore the exploration compartment containing 10 pot halves for 5 min (the distribution of these pots was varied, so individuals never experienced the same constellation when tested again). The following parameters were assessed: (i) latency until entering the exploration compartment (seconds), (ii) latency until entering the first exploration pot (seconds), (iii) time spent in locomotion (seconds), (iv) frequency of entering any exploration pot, (v) frequency of entering a 5 cm radius around any exploration pot, and (vi) number of different exploration pots entered (1–10).
Boldness was assessed inside the 40 l home tank of each individual (50 × 30 × 30 cm), containing a pot half 20 cm from the rear end. A novel object was placed at the front side and this induced the fish to hide inside their pot half. Novel objects used were: red and black plastic beetle, mixed coloured stone bird, a white funnel, a dark-blue globe, a white cross or a blue plastic device. All novel objects were about equally sized and individuals never experienced the same novel object twice. Recording for 5 min started immediately and the following two parameters were recorded: (i) the latency to leave the pot half and approaching the object (seconds) and (ii) the estimated shortest distance to approach the object (0–30 cm, with 0 indicating touching the object).
Aggressiveness was assessed inside the 40 l home tank of each individual (see above). A mirror (46 × 15 cm) was placed at the long side of the tank, which induced the individuals to hide inside their pot half. Recording for 5 min started immediately and the following parameters were recorded: (i) time spent hiding inside their pot (seconds), (ii) frequency of restrained aggression (slow approach to the mirror, fin-spreading display, head-down display, s-bend display), and (iii) frequency of overt aggression (fast approach to the mirror and all contacts with their mirror image—includes biting, ramming, mouth-fighting, tail beating).
The three behavioural tests were conducted in randomized order for each individual within 1 day, and repeated the next day (again randomized). In total, 272 individuals were tested, of which 216 individuals entered the habitat saturation experiment. These 216 individuals were tested on days t-2 and t-1 before release into the ring-tank (electronic supplementary material, figure S1a, first and second test series) and again on two consecutive days after the habitat saturation experiment (on days 40–52, third and fourth test series). Note that three individuals died before we could conduct the third and fourth series.
(b). Experimental set-up
The habitat saturation experiment was conducted inside a large 8000 l octagonal tank (‘ring-tank’) and monitored by D.H. and R.S., who were blind to the behavioural test results of the individual fish. This tank contained 24 breeding patches (e.g. electronic supplementary material, figure S1b, each patch with 12 pot halves and a shelter between two stone slabs), where 72 fish could build cooperative groups as follows (three trials were conducted, giving 3 × 72 = 216 individuals tested in total, rotating the location of the 24 patches between trials). During each trial, a large male (mean ± s.d.: standard length (SL) mm 55.4 ± 3.7, and range: 48.2–64.1, n = 39) and large female (53.9 ± 4.6, 48.0–66.2, n = 33), a medium male (40.8 ± 3.1, 32.8–46.4, n = 33) and medium female (40.4 ± 3.6, 33.2–46.0, n = 39), and a small male (28.8 ± 3.9, 18.4–35.7, n = 36) and small female (30.6 ± 4.9, 20.3–42.0, n = 36) were released inside the ring-tank at randomly chosen different sides of the tank (day 0, electronic supplementary material, figure S1b). Every third day this procedure was repeated (electronic supplementary material, figure S1a), until 72 fish were released (electronic supplementary material, figure S1c, last release on day 33, giving 12 sets of six fish released). Cichlids were individually marked with fin-clips taken from the dorsal and anal fin, individual body measurements taken (body size SL in 0.1 mm and body mass in 1 mg accuracy) and sexed by inspection of the gonadal papilla. See the electronic supplementary material and Heg et al. (in press) for more details.
(c). Behavioural recording during the habitat saturation experiment
The location (patches and pots visited), status (group members: dominant or subordinate; or non-group members: fish who failed to settle inside a group and patch, and remained near the water surface: so-called ‘aggregation’ fish) and group membership (the identity of the dominants assisted by the subordinates) was determined for each individual daily from day 0 (first set released) to day 39 (6 days after the last, 12th, set was released).
Ten-minute focal behavioural observations of each individual present in the ring-tank were made at the end of each trial on days 35–37 (n = 216 individuals in total); the individual order of observation was randomized. We recorded the actor and recipient of every aggressive (fast approach, restrained and overt aggression; see Hamilton et al. 2005) and affiliative (slow approach, joining, bumping, as well as submissive behaviour: tail quivering and zig-zag swimming; see Hamilton et al. 2005) encounter, and these were used to construct the directed and weighted adjacency matrices of interactions for the social network analyses. For details of the behavioural recorded variables see the electronic supplementary material.
We also made 15 min parental and alloparental brood care observations (fanning and egg cleaning (each mouthing movement at the eggs summed)) on the day the clutch was produced (n = 275 carers of 127 different individuals in 69 clutches in total, see the electronic supplementary material for details). We used these data to test whether alloparental brood care (expressed as the proportion of total brood care) showed a relationship with the social network parameters of subordinates (data of n = 64 subordinates averaged per individual).
(d). Social network analyses
The interactions recorded during the focal observations were summed up for each dyad in each trial to determine the strengths of ties between individuals, for aggression and affiliation separately (see Hamilton et al. 2005). We used the igraph package for R2.9.1 to construct the network from the observational data and to calculate centrality measures (Csárdi & Nepusz 2006; R Development Core Team 2009). We then calculated for the aggression and affiliation networks separately (e.g. Newman 2004): (i) the weighted in- and out-degree: number and weight of links an individual has (which was highly correlated with betweenness and closeness centrality, see the electronic supplementary material) and (ii) the eigenvector centrality (a proportional measure ranging between 0 and 1): the centrality of each individual is proportional to the sum of the centralities of those individuals to whom he or she is connected (determined iteratively).
(e). Statistical analyses
Statistical analyses were conducted using SPSS v. 17.0 (Norusis 2007) and R2.9.1 (R Development Core Team 2009). We started with a total of 272 individuals, who were tested twice for exploration propensity, boldness and aggressiveness (first and second test series). From these 272 individuals, 216 individuals entered the habitat saturation experiment. To summarize the many variables of the three behavioural tests (exploration propensity, boldness, aggressiveness), we extracted one principal component using a categorical principal component analysis (CatPCA; Norusis 2007, see electronic supplementary material). We refer to this principal component as the individual ‘behavioural type’ (as in Sih et al. 2004).
All effects of behavioural type on the settlement and groups formed were corrected for trial (1–3), set (1–12, 1 = 1st released on day 0, 12th = last released on day 36), body size (SL mm) and sex effects. The effect of focal behavioural type on the focal status achieved in the experiment was analysed using a multinomial regression (Norusis 2007). In the remainder of the multi-factorial analyses, we used generalized estimating equations (GEEs) to account for repeated measures per individual (Norusis 2007, see the electronic supplementary material for more details).
The effects of the behavioural type on the weighted in-degree and the eigenvector centrality were analysed using generalized linear models (GLMs) with quasi-Poisson distribution and log-link for degree, and with a gamma distribution and log-link for the eigenvector centrality, square-root transformed to control for many values near zero (using R2.9.1). The dispersion (scaling) parameters were adjusted using the deviance method (Faraway 2006). See the electronic supplementary material for more details. However, since the individual network parameters (the dependent variables weighted in-degree and eigenvector centrality) cannot be independently assessed for any two individuals measured within the same trial (i.e. they are derived from the same single network analysis and may show clusters of individuals of high weighted in-degree and eigenvector centrality), the robustness of the above GLMs was checked by running each model 1000 times and drawing 100 individuals at random for each model separately. We then report the percentage of models (out of 1000) showing a significant effect for each independent parameter (p-value smaller than 0.05, later called bootstrap % p < 0.05).
3. Results
We extracted one categorical principal component from the three behavioural tests (exploration, boldness and aggression) which consistently distinguished the individuals in the different behavioural types (see electronic supplementary material, table S1, we refer to the extracted principal component as ‘behavioural type’). Low scores denote low explorative, shy and non-aggressive individuals, whereas high scores denote explorative, bold and aggressive individuals (averaged for the two pre-release behavioural tests).
(a). Settlement and body size
At the end of the trials, dominant males defended on average 3.1 ± 1.9 patches (±s.d.: n = 26, range 1–9) inside the ring-tank, containing an average of 1.9 ± 1.3 different dominant females (±s.d.: n = 26, range: 0–5, three males did no longer have a dominant female in their defended patch, although they had one earlier in the experiment). Dominant females defended significantly less patches on average (1.8 ± 1.0, range: 1–5) inside the ring-tank than did the dominant males (Mann–Whitney U-test: z = −3.6, p < 0.001).
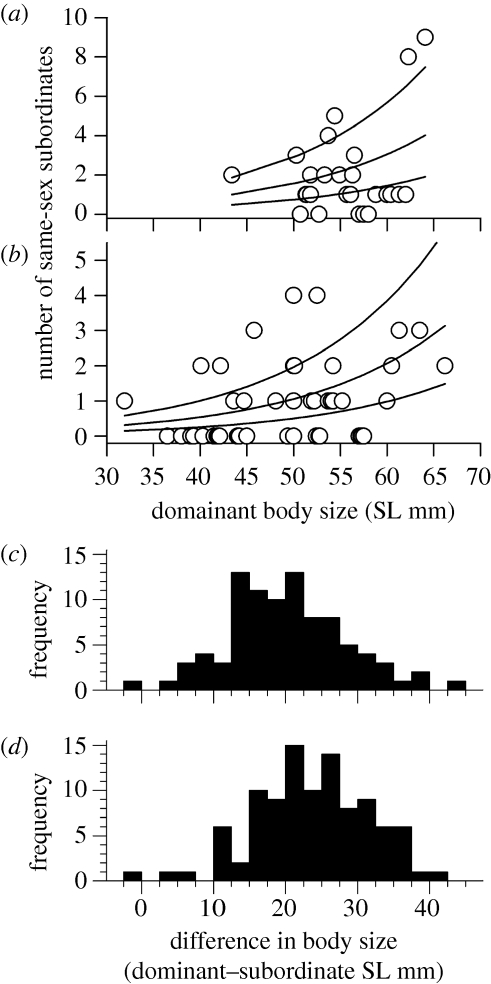
Subordinates defended on average 1.5 ± 0.7 patches (±s.d.: n = 100, range: 1–4) inside the ring-tank, and the sexes did not differ in this respect (males: 1.5 ± 0.7, n = 54; females: 1.4 ± 0.7, n = 46; Mann–Whitney U-test: z = −0.9, p = 0.34). Behavioural types of the dominants did not influence the total number of subordinates accepted as group members (GLM: n = 74, p = 0.25). Also, behavioural type did not influence how many same-sex subordinates dominants accepted as group members (GLM, n = 74, p = 0.33 and removed from the model). In contrast, the number of same-sex subordinates increased with the dominant's body size, both for males (figure 1a) and females (figure 1b; p = 0.002, coefficient ± s.e.: 0.067 ± 0.021) and decreased for individuals who became dominant but were released later into the ring-tank (p = 0.003, −0.124 ± 0.042, corrected for trial p = 0.86 and dominant sex p = 0.13 effects).
Figure 1.
Dominant body size and the number of same-sex subordinates accepted for (a) males (n = 26) and (b) females (n = 48). Upper, middle and lower lines (from GLM in main text): released on day 0, 15 and 33 from the start of the saturation experiment, respectively. Difference in body size (SL mm) between (c) dominant females and subordinates (n = 91), (d) dominant males and subordinates (n = 100).
As expected, dominant males (figure 1c) and females (figure 1d) accepted, with two exceptions, only subordinates substantially smaller in body size than themselves. Note that nine subordinates were not clearly associated with a dominant female at the end of the trial, although they made occasional visits to neighbouring patches containing a dominant female. The difference in body size between dominant males and their subordinates was significantly larger than between the dominant females and their subordinates (figure 1c,d; paired t-test, t90 = 4.5, p < 0.001).
(b). Behavioural type and social status
Overall, the behavioural type of the individuals did not predict the status achieved in the ring-tank (dominant, subordinate or aggregation fish, n = 216: multinomial regression, effects of type: likelihood ratio (LR) χ2 = 0.1, d.f. = 2, p = 0.93). However, as expected, status achieved depended on body size SL mm (LR χ2 = 181.7, d.f. = 2, p < 0.001), sex (LR χ2 = 35.6, d.f. = 2, p < 0.001) and the day of release (LR χ2 = 33.3, d.f. = 2, p < 0.001; effect of trial: LR χ2 = 7.2, d.f. = 4, p = 0.12).
(c). Dominant male and dominant female behavioural types
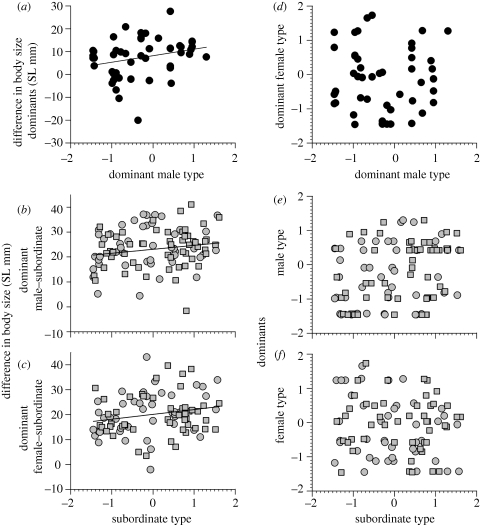
Dominant males and females did not mate size-assortatively inside the ring-tank (Pearson r: all females r = 0.12, n = 48, p = 0.43; only largest female per male r = −0.07, n = 23, p = 0.74). However, aggressive–bold–explorative dominant males only paired with dominant females smaller than themselves, whereas opposite males also paired with dominant females larger than themselves (figure 2a; GEE with individual identifier as subject, dependent variable male SL − female SL mm: effect of male type Wald χ2 = 7.1, d.f. = 1, p = 0.008; effect of female type Wald χ2 = 1.2, d.f. = 1, p = 0.27 non-significant and removed from the model). Dominant males and females did not mate type-assortatively inside the ring-tank (figure 2d; male type versus female type: all females r = −0.001, n = 48, p = 0.99; only largest female per male r = 0.003, n = 23, p = 0.99).
Figure 2.
(a) Dominant male type and the difference in body size to his dominant females (n = 23 males with 48 females, line with coefficient ± s.e.: 2.87 ± 1.08 from GEE described in the text). (b,c) Subordinate female type (grey circles) or male type (grey squares) and their difference in body size with either the (b) dominant male (n = 46 subordinate females and 54 males) or (c) dominant female (n = 43 subordinate females and 48 males). Regression lines in (b,c) are from the two GEE models described in the text and note that the effects of subordinate sex were non-significant. (d–f) Correlations between behavioural types of group members: (d) there was no correlation between the dominant male's behavioural type and his dominant female partners' behavioural types (n = 48). There was a significant, but weak, correlation between subordinate type (grey circles: females; grey squares: males) and (e) dominant male type (n = 46; 54, respectively), but not versus (f) dominant female type (n = 43; 48, respectively). Note that subordinate sexes were similar.
(d). Dominants and subordinates behavioural types
To explore the difference in body size between dominants and subordinates further, we constructed two GEE models. Aggressive–bold–explorative subordinates of both sexes grouped with relatively large dominant males (figure 2b; model 1: GEE with dominant male identifier as subjects: effect of subordinate type, Wald χ2 = 5.0, d.f. = 1, p = 0.026, coefficient ± s.e.: 1.59 ± 0.71 and intercept 23.61 ± 1.30) and also with relatively large dominant females (figure 2c; model 2: GEE with dominant female identifier as subjects: effect of subordinate type, Wald χ2 = 5.4, d.f. = 1, p = 0.02, coefficient ± s.e.: 1.94 ± 0.83 and intercept 20.08 ± 1.20). In model 1, the effect of subordinate sex and the interaction between subordinate sex × behavioural type were non-significant and removed (p = 0.46 and 0.77, respectively), but note that in model 2, the interaction approached significance (sex p = 0.55 and interaction p = 0.053: the intercept and slope were larger for subordinate females when compared with subordinate males). Note the similarities in the behavioural type effect on size-assortative group building in figure 2a–c.
There was a significant, but weak correlation between the subordinate behavioural type and the dominant male behavioural type (figure 2e; Pearson r = 0.298, n = 100, p = 0.003), but not between the subordinate behavioural type and dominant female behavioural type (figure 2f; Pearson r = −0.118, n = 91, p = 0.27, subordinate sexes were similar in these respects). However, since there were also correlations between subordinate behavioural type and the body size difference with the dominant male (Pearson r = 0.181, n = 100, p = 0.07) and dominant female (Pearson r = 0.206, n = 91, p = 0.051), we also computed a partial correlation, which was again significant for subordinate behavioural type versus dominant male behavioural type (partial r = 0.295, p = 0.003), but not versus dominant female behavioural type (partial r = −0.095, p = 0.38).
(e). Social networks
We constructed six social networks: an aggression and an affiliation network for each of the three trials separately (electronic supplementary material, figure S2). Note that one individual did not have any aggressive ties during the observation, reducing the sample size to 215 individuals for the aggression network.
(i). Aggression network
Individuals varied widely in both their weighted in-degree (mean ± s.d.: 10.6 ± 7.3, range: 0–49), and eigenvector centrality (0.11 ± 0.19, range: 0.00002–1, n = 215). The individual's behavioural type did not affect its weighted in-degree. Rather, the weighted in-degree significantly increased with body size and depended also on social status and the interaction between social status and body size (figure 3a–c; see electronic supplementary material, table S2): in smaller fish, individuals from the aggregation had the highest weighted in-degree, followed by dominants and then subordinates; in larger fish, subordinates had the highest weighted in-degree, followed by aggregation fish and then dominants. These results were also robust in the bootstrap estimates (see electronic supplementary material, table S2).
Figure 3.
Social network parameters and behavioural type for (a,d) dominants, (b,e) subordinates and (c,f) aggregation fish. (a–c) Behavioural type did not affect the aggression-weighted in-degree, which instead depended on social status, body size and their interaction. (d–f) Behavioural type affected the affiliation-weighted in-degree depending on social status, body size and the interaction between behavioural type and body size. GLM curve fits are shown in each panel from the models given in electronic supplementary material, table S2.
In contrast, behavioural type did significantly affect the eigenvector centrality in conjunction with body size, social status and their interactions with behavioural type (see electronic supplementary material, table S2). Aggressive–bold–explorative and large dominants and aggregation fish had the highest eigenvector centralities when compared with small-sized fish of the same behavioural type and status. Subordinate fish had small eigenvector centralities overall, and the eigenvector centralities declined towards smaller and more aggressive–bold–explorative fish. However, the effects of behavioural type were not robust in the bootstrap estimates, whereas the effects of body size, status and their interaction remained robust. Averaged alloparental brood care performed by each individual subordinate (expressed as proportion of total brood care) did not affect their weighted in-degree and eigenvector centrality (n = 63, proportion care: p = 0.32 and 0.73, interaction with sex: p = 0.93 and 0.062, respectively, GLMs corrected for effects of trial, set, body size and sex).
(ii). Affiliation network
Again, individuals varied widely in both their weighted in-degree (mean ± s.d.: 20.1 ± 21.1, range: 0–111) and eigenvector centrality (mean ± s.d.: 0.11 ± 0.22, range: 0.00003–1, n = 216). Behavioural type in interaction with body size, as well as social status by itself significantly affected the weighted in-degree of the focal fish (figure 3d–f; see electronic supplementary material, table S2). Peaceful–shy–non-explorative and large fish had the highest weighted in-degree and weighted in-degree declined towards smaller fish, but this decline was less strong in aggressive–bold–explorative fish. The decline was more pronounced in dominants (figure 3d), followed by subordinates (figure 3e) and almost absent in aggregation fish (figure 3f). However, only the effects of body size and status remained robust in the bootstrap estimates (electronic supplementary material, table S2).
Status and behavioural type also affected the eigenvector centrality, but the latter only in interaction with the focal's sex. The eigenvector centrality significantly declined from dominants to subordinates to aggregation fish, and was lower for males when compared with females (electronic supplementary material, table S2). These results were also robust in the bootstrap estimates (see electronic supplementary material, table S2). Averaged alloparental brood care performed by each individual subordinate (expressed as proportion of total brood care) did not affect their weighted in-degree and eigenvector centrality (n = 64, proportion care: p = 0.45 and 0.77, interaction with sex: p = 0.47 and 0.55, respectively, GLMs corrected for effects of trial, set, body size and sex).
4. Discussion
Previous work has shown that groups in cooperatively breeding cichlids are body size-structured, meaning that the largest individuals dominate all others (e.g. Balshine-Earn et al. 1998; Hamilton et al. 2005; Heg & Hamilton 2008; Mitchell et al. 2009a,b), dominant pairs only accept small-sized same-sex subordinates (e.g. Heg et al. 2005a,b) and dominant females usually only accept large-sized immigrant dominant males (e.g. Balshine-Earn et al. 1998; Fitzpatrick et al. 2008). This body size-dependent hierarchy inside the group also affects reproductive participation (and suppression) by the group members (e.g. Fitzpatrick et al. 2006; Heg et al. 2006, 2008a,b; Heg 2008; Heg & Hamilton 2008; Stiver et al. 2009).
We have now shown in a habitat saturation experiment, where individuals were free to settle and build groups of dominants and subordinates inside a large ring-tank, that the behavioural type of individuals may also affect sociality in various ways. Behavioural type did not affect the likelihood of obtaining a dominant or subordinate position (instead of remaining inside the aggregation), nor did it affect the number and sexes of subordinate dominants accepted as group members (which were all affected by dominant body size and the difference between dominant and subordinate body size, as in natural groups: Balshine et al. 2001; Heg et al. 2005a). However, behavioural type did affect the size difference within the dominant pairs and between dominants and their subordinates (larger for aggressive–bold–explorative individuals). Both effects could be interpreted as a spill-over effect of being aggressive in dominant males: although aggressive dominant males would benefit from accepting larger females as mates (since female size correlates with clutch size: e.g. Heg 2008), and larger subordinates as group members (since large group members may deter predators, e.g. Taborsky 1984), they actually only accepted smaller fish as both mates and subordinates. Alternatively, aggressive dominant males might be avoided by relatively large dominant females and subordinates, who prefer to settle elsewhere. Moreover, male-dominant behavioural type correlated positively with subordinate behavioural type (in both sexes), but no such correlation was found between female-dominant behavioural type and her subordinates' behavioural types. There was also no correlation between the types of the dominants.
Non-random association of individuals of group-living animals is widely known for morphological traits such as size, and also sex (Krause & Ruxton 2002). However, assortative mixing based on the behavioural phenotype has rarely been tested. Notable exceptions include the work on guppies and sticklebacks, both in the laboratory (Budaev 1997; Dyer et al. 2008) and in the wild (Croft et al. 2009). As our experimental approach in the laboratory provided a uniform habitat, we can exclude an effect of behavioural type on habitat choice as a mechanism leading to behavioural type-assortative groups. Also, our analysis accounted for effects of size, so we can rule out at least this option as well. The fact that behavioural type had an effect on the size difference between dominants, and also between dominants and subordinates further suggests that the choice is actively made by the individuals involved.
We then commenced with social network analyses, to explore the potentially more subtle effects of behavioural types on sociality. Again, effects of body size, social status and sometimes sex were striking. For instance, large individuals had higher levels of aggression-weighted in-degree (number of aggressive ties). Nevertheless, corrected for these effects of body size, sex and status, we also found smaller effects of behavioural type. However, effects of behavioural type were always present in interaction with body size or social status or both. Large aggressive–bold–exploratory dominants and aggregation fish had very high levels of aggression eigenvector centrality, whereas all other individuals had low levels. This suggests that large aggressive, bold and explorative individuals are centres of aggressive activity inside colonies of cooperatively breeding cichlids, mainly interacting among themselves (for natural colonies see Heg et al. 2008a,b; Flack et al. 2006). In subordinates, eigenvector centrality was generally low, but decreased even further towards bold and small individuals. No effects of behavioural type on aggression-weighted in-degree were detected. In contrast, large and peaceful–shy–non-explorative individuals had high levels of affiliation-weighted in-degree when compared with smaller individuals; however, the decline of weighted in-degree for smaller fish was less pronounced for bold individuals and it also declined from dominants, to subordinates, to individuals remaining in the aggregation. The bootstrap analyses indicate that further testing will be needed to assert these results, since the effects of behavioural type were often not robust, whereas effects of body size and status were always robust (see electronic supplementary material, table S2). As it is now, the results suggest that shy, less aggressive and less explorative individuals maintain many peaceful connections within the colony, even though their position in the network is rather determined by aggressive interactions. Keeping such affiliative ties might help to counteract any negative effects of low exploration activity on finding alternative groups (e.g. Bergmüller et al. 2005; Heg et al. 2008b), for example, to improve their social position (‘queuing’, Stiver et al. 2006), because peaceful interactions with neighbouring groups might facilitate dispersal to these groups (see also Stiver et al. 2004, 2006, 2007; Bergmüller et al. 2005; Fitzpatrick et al. 2008).
Finally, we did not find any effect of the individual's propensity to cooperate (expressed as proportion investment in brood care) on this individual's social network parameters, suggesting that cooperative individuals do not maintain more central and well-connected positions within their neighbourhood. However, there are some indications that shy individuals engaged more in (allo)parental brood care than bold individuals, corrected for effects of body size (S. Rothenberger 2007, unpublished data).
5. Conclusions and future directions
We have demonstrated weak but significant effects of behavioural type on the initial group composition of a cooperatively breeding fish. These settlement patterns will affect which types of group members interact or cooperate with each other, and thus shape the selective processes maintaining variation in animal temperaments. Non-random assortment of behavioural types into groups may also increase the speed of evolution towards sociality, on top of kin-selected benefits. Furthermore, social network analyses suggest that the behavioural type may affect the strength and type of interactions individuals have with group members and non-group members, where bold individuals are centres of aggression within the colony and shy individuals maintain many friendly connections. We suggest that this may affect the likelihood of between-group dispersal and group take-overs depending on the behavioural type(s) of the individuals involved.
Acknowledgements
We thank Denis Réale, Niels Dingemanse, Jonathan Wright and Anahita Kazem for their invitation to contribute to this issue, and these editors and the anonymous referees for comments on the manuscript. This work was funded by the Swiss National Science Foundation grant SNSF 3100A-108473 to D.H. and approved by LANAT of the Kanton Bern, and thus complied with the legal requirements of Switzerland.
Footnotes
One contribution of 16 to a Theme Issue ‘Evolutionary and ecological approaches to the study of personality’.
References
- Arnqvist G., Henriksson S.1997Sexual cannibalism in the fishing spider and a model for the evolution of sexual cannibalism based on genetic constraints. Evol. Ecol. 11, 255–273 10.1023/A:1018412302621 (doi:10.1023/A:1018412302621) [DOI] [Google Scholar]
- Balshine S., Leach B., Neat F., Reid H., Taborsky M., Werner N.2001Correlates of group size in a cooperatively breeding cichlid fish (Neolamprologus pulcher). Behav. Ecol. Sociobiol. 50, 134–140 10.1007/s002650100343 (doi:10.1007/s002650100343) [DOI] [Google Scholar]
- Balshine-Earn S., Neat F. C., Reid H., Taborsky M.1998Paying to stay or paying to breed? Field evidence for direct benefits of helping behavior in a cooperatively breeding fish. Behav. Ecol. 9, 432–438 10.1093/beheco/9.5.432 (doi:10.1093/beheco/9.5.432) [DOI] [Google Scholar]
- Bell A. M., Hankison S. J., Laskowski K. L.2009The repeatability of behaviour: a meta-analysis. Anim. Behav. 77, 771–783 10.1016/j.anbehav.2008.12.022 (doi:10.1016/j.anbehav.2008.12.022) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmüller R., Taborsky M.2007Adaptive behavioural syndromes due to strategic niche specialization. BMC Ecol. 7 10.1186/1472-6785-7-12 (doi:10.1186/1472-6785-7-12) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmüller R., Heg D., Peer K., Taborsky M.2005Extended safe havens and between-group dispersal of helpers in a cooperatively breeding cichlid. Behaviour 142, 1643–1667 10.1163/156853905774831800 (doi:10.1163/156853905774831800) [DOI] [Google Scholar]
- Brouwer L., Heg D., Taborsky M.2005Experimental evidence for helper effects in a cooperatively breeding cichlid. Behav. Ecol. 16, 667–673 10.1093/beheco/ari042 (doi:10.1093/beheco/ari042) [DOI] [Google Scholar]
- Budaev S. V.1997‘Personality’ in the guppy (Poecilia reticulata): a correlational study of exploratory behavior and social tendency. J. Comp. Psychol. 111, 399–411 10.1037/0735-7036.111.4.399 (doi:10.1037/0735-7036.111.4.399) [DOI] [Google Scholar]
- Cote J., Clobert J., Brodin T., Fogarty S., Sih A.2010Personality-dependent dispersal: characterization, ontogeny and consequences for spatially structured populations. Phil. Trans. R. Soc. B 365, 4065–4076 10.1098/rstb.2010.0176 (doi:10.1098/rstb.2010.0176) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croft D. P., James R., Krause J.2008Exploring animal social networks. Princeton, NJ: Princeton University Press [Google Scholar]
- Croft D., Krause J., Darden S., Ramnarine I., Faria J., James R.2009Behavioural trait assortment in a social network: patterns and implications. Behav. Ecol. Sociobiol. 63, 1495–1503 10.1007/s00265-009-0802-x (doi:10.1007/s00265-009-0802-x) [DOI] [Google Scholar]
- Csárdi G., Nepusz T.2006The igraph software package for complex network research. InterJournal Complex Syst. 1695 [Google Scholar]
- Dierkes P., Heg D., Taborsky M., Skubic E., Achmann R.2005Genetic relatedness in groups is sex-specific and declines with age of helpers in a cooperatively breeding cichlid. Ecol. Lett. 8, 968–975 10.1111/j.1461-0248.2005.0801.x (doi:10.1111/j.1461-0248.2005.0801.x) [DOI] [PubMed] [Google Scholar]
- Dyer J. R. G., Croft D. P., Morrell L. J., Krause J.2008Shoal composition determines foraging success in the guppy. Behav. Ecol. 20, 165–171 10.1093/beheco/arn129 (doi:10.1093/beheco/arn129) [DOI] [Google Scholar]
- Faraway J. J.2006Extending the linear model with R: generalized linear, mixed effects and nonparametric regression models. London, UK: Chapman & Hall [Google Scholar]
- Fitzpatrick J., Desjardins J., Stiver K., Montgomerie R., Balshine S.2006Male reproductive suppression in the cooperatively breeding fish. Neolamprologus pulcher. Behav. Ecol. 17, 25–33 10.1093/beheco/ari090 (doi:10.1093/beheco/ari090) [DOI] [Google Scholar]
- Fitzpatrick J. L., Desjardins J. K., Milligan N., Stiver K. A., Montgomerie R., Balshine S.2008Female-mediated causes and consequences of status change in a social fish. Proc. R. Soc. B 275, 929–936 10.1098/rspb.2007.1449 (doi:10.1098/rspb.2007.1449) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flack J. C., Girvan M., de Waal F. B. M., Krakauer D. C.2006Policing stabilizes construction of social niches in primates. Nature 439, 426–429 10.1038/nature04326 (doi:10.1038/nature04326) [DOI] [PubMed] [Google Scholar]
- Fraser D. F., Gilliam J. F., Daley M. J., Le A. N., Skalski G. T.2001Explaining leptokurtic movement distributions: intrapopulation variation in boldness and exploration. Am. Nat. 158, 124–135 10.1086/321307 (doi:10.1086/321307) [DOI] [PubMed] [Google Scholar]
- Hamilton I. M., Heg D., Bender N.2005Size differences within a dominance hierarchy influence conflict and help in a cooperatively breeding cichlid. Behaviour 142, 1591–1613 10.1163/156853905774831846 (doi:10.1163/156853905774831846) [DOI] [Google Scholar]
- Harcourt J. L., Sweetman G., Johnstone R. A., Manica A.2009Personality counts: the effect of boldness on shoal choice in three-spined sticklebacks. Anim. Behav. 77, 1501–1505 10.1016/j.anbehav.2009.03.004 (doi:10.1016/j.anbehav.2009.03.004) [DOI] [Google Scholar]
- Heg D.2008Reproductive suppression in female cooperatively breeding cichlids. Biol. Lett. 4, 606–609 10.1098/rsbl.2008.0365 (doi:10.1098/rsbl.2008.0365) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heg D., Hamilton I.2008Tug-of-war over reproduction in a cooperatively breeding cichlid. Behav. Ecol. Sociobiol. 62, 1249–1257 10.1007/s00265-008-0553-0 (doi:10.1007/s00265-008-0553-0) [DOI] [Google Scholar]
- Heg D., Ens B. J., Jeugd H. P. V. D., Bruinzeel L. W.2000Local dominance and territorial settlement of nonbreeding oystercatchers. Behaviour 137, 473–530 10.1163/156853900502187 (doi:10.1163/156853900502187) [DOI] [Google Scholar]
- Heg D., Brouwer L., Bachar Z., Taborsky M.2005aLarge group size yields group stability in the cooperatively breeding cichlid Neolamprologus pulcher. Behaviour 142, 1615–1641 10.1163/156853905774831891 (doi:10.1163/156853905774831891) [DOI] [Google Scholar]
- Heg D., Bachar Z., Taborsky M.2005bCooperative breeding and group structure in the Lake Tanganyika cichlid. Neolamprologus savoryi. Ethology 111, 1017–1043 [Google Scholar]
- Heg D., Bergmüller R., Bonfils D., Otti O., Bachar Z., Burri R., Heckel G., Taborsky M.2006Cichlids do not adjust reproductive skew to the availability of independent breeding options. Behav. Ecol. 17, 419–429 10.1093/beheco/arj056 (doi:10.1093/beheco/arj056) [DOI] [Google Scholar]
- Heg D., Heg-Bachar Z., Brouwer L., Taborsky M.2008aExperimentally induced helper dispersal in colonially breeding cooperative cichlids. Environ. Biol. Fish. 83, 191–206 10.1007/s10641-007-9317-3 (doi:10.1007/s10641-007-9317-3) [DOI] [Google Scholar]
- Heg D., Jutzeler E., Bonfils D., Mitchell J. S.2008bGroup composition affects male reproductive partitioning in a cooperatively breeding cichlid. Mol. Ecol. 17, 4359–4370 10.1111/j.1365-294X.2008.03920.x (doi:10.1111/j.1365-294X.2008.03920.x) [DOI] [PubMed] [Google Scholar]
- Heg D., Jutzeler E., Mitchell J. S., Hamilton I. M.2009Helpful female subordinate cichlids are more likely to reproduce. PLoS ONE 4, e5458. 10.1371/journal.pone.0005458 (doi:10.1371/journal.pone.0005458) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heg D., Rothenberger S., Schürch R.In press Habitat saturation, benefits of philopatry, relatedness and cooperative breeding in a cichlid. Behav. Ecol. [Google Scholar]
- Koolhaas J., Korte S., De Boer S., Van Der Vegt B., Van Reenen C., Hopster H., De Jong I., Ruis M., Blokhuis H.1999Coping styles in animals: current status in behavior and stress-physiology. Neurosci. Biobehav. Rev. 23, 925–935 10.1016/S0149-7634(99)00026-3 (doi:10.1016/S0149-7634(99)00026-3) [DOI] [PubMed] [Google Scholar]
- Krause J., Ruxton G.2002Living in groups. Oxford, UK: Oxford University Press [Google Scholar]
- Krause J., James R., Croft D. P.2010Personality in the context of social networks. Phil. Trans. R. Soc. B 365, 4099–4106 10.1098/rstb.2010.0216 (doi:10.1098/rstb.2010.0216) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Limberger D.1983Pairs and harems in a cichlid fish, Lamprologus brichardi. Z. Tierpsychol. 62, 115–144 10.1111/j.1439-0310.1983.tb02146.x (doi:10.1111/j.1439-0310.1983.tb02146.x) [DOI] [Google Scholar]
- McDonald D. B.2007Predicting fate from early connectivity in a social network. Proc. Natl Acad. Sci. USA 104, 10 910–10 914 10.1073/pnas.0701159104 (doi:10.1073/pnas.0701159104) [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNamara J. M., Houston A. I.1996State-dependent life histories. Nature 380, 215–221 10.1038/380215a0 (doi:10.1038/380215a0) [DOI] [PubMed] [Google Scholar]
- Mitchell J. S., Jutzeler E., Heg D., Taborsky M.2009aDominant members of cooperatively-breeding groups adjust their behaviour in response to the sexes of their subordinates. Behaviour 146, 1665–1686 10.1163/000579509X12459328580026 (doi:10.1163/000579509X12459328580026) [DOI] [Google Scholar]
- Mitchell J. S., Jutzeler E., Heg D., Taborsky M.2009bGender differences in the costs that subordinate group members impose on dominant males in a cooperative breeder. Ethology 115, 1162–1174 10.1111/j.1439-0310.2009.01705.x (doi:10.1111/j.1439-0310.2009.01705.x) [DOI] [Google Scholar]
- Newman M.2004Analysis of weighted networks. Phys. Rev. E 70, 056131. 10.1103/PhysRevE.70.056131 (doi:10.1103/PhysRevE.70.056131) [DOI] [PubMed] [Google Scholar]
- Norusis M. J.2007SPSS 15.0 Advanced statistical procedures companion. Englewood Cliffs, NJ: Prentice-Hall [Google Scholar]
- Pike T. W., Samanta M., Lindström J., Royle N. J.2008Behavioural phenotype affects social interactions in an animal network. Proc. R. Soc. B 275, 2515–2520 10.1098/rspb.2008.0744 (doi:10.1098/rspb.2008.0744) [DOI] [PMC free article] [PubMed] [Google Scholar]
- R Development Core Team 2009R: a language and environment for statistical computing. [Google Scholar]
- Raihani N. J., Ridley A. R., Browning L. E., Nelson-Flower M. J., Knowles S.2008Juvenile female aggression in cooperatively breeding pied babblers: causes and contexts. Ethology 114, 452–458 10.1111/j.1439-0310.2008.01482.x (doi:10.1111/j.1439-0310.2008.01482.x) [DOI] [Google Scholar]
- Réale D., Reader S. M., Sol D., McDougall P. T., Dingemanse N. J.2007Integrating animal temperament within ecology and evolution. Biol. Rev. Camb. Phil. Soc. 82, 291–318 10.1111/j.1469-185X.2007.00010.x (doi:10.1111/j.1469-185X.2007.00010.x) [DOI] [PubMed] [Google Scholar]
- Ryder T. B., McDonald D. B., Blake J. G., Parker P. G., Loiselle B. A.2008Social networks in the lek-mating wire-tailed manakin (Pipra filicauda). Proc. R. Soc. B 275, 1367–1374 10.1098/rspb.2008.0205 (doi:10.1098/rspb.2008.0205) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schürch R., Heg D.2010aVariation in helper type affects group stability and reproductive decisions in a cooperative breeder. Ethology 116, 257–269 10.1111/j.1439-0310.2009.01738.x (doi:10.1111/j.1439-0310.2009.01738.x) [DOI] [Google Scholar]
- Schürch R., Heg D.2010bLife history and behavioral type in the highly social cichlid Neolamprologus pulcher. Behav. Ecol. 21, 588–598 10.1093/beheco/arq024 (doi:10.1093/beheco/arq024) [DOI] [Google Scholar]
- Sih A., Bell A., Johnson J. C.2004Behavioral syndromes: an ecological and evolutionary overview. Trends Ecol. Evol. 19, 372–378 10.1016/j.tree.2004.04.009 (doi:10.1016/j.tree.2004.04.009) [DOI] [PubMed] [Google Scholar]
- Stiver K. A., Dierkes P., Taborsky M., Balshine S.2004Dispersal patterns and status change in a co-operatively breeding cichlid Neolamprologus pulcher: evidence from microsatellite analyses and behavioural observations. J. Fish Biol. 65, 91–105 10.1111/j.0022-1112.2004.00427.x (doi:10.1111/j.0022-1112.2004.00427.x) [DOI] [Google Scholar]
- Stiver K. A., Fitzpatrick J., Desjardins J. K., Balshine S.2006Sex differences in rates of territory joining and inheritance in a cooperatively breeding cichlid fish. Anim. Behav. 71, 449–456 10.1016/j.anbehav.2005.06.011 (doi:10.1016/j.anbehav.2005.06.011) [DOI] [Google Scholar]
- Stiver K. A., Desjardins J. K., Fitzpatrick J. L., Neff B., Quinn J. S., Balshine S.2007Evidence for size and sex-specific dispersal in a cooperatively breeding cichlid fish. Mol. Ecol. 16, 2974–2984 10.1111/j.1365-294X.2007.03350.x (doi:10.1111/j.1365-294X.2007.03350.x) [DOI] [PubMed] [Google Scholar]
- Stiver K. A., Fitzpatrick J. L., Desjardins J. K., Balshine S.2009Mixed parentage in Neolamprologus pulcher groups. J. Fish Biol. 74, 1129–1135 10.1111/j.1095-8649.2009.02173.x (doi:10.1111/j.1095-8649.2009.02173.x) [DOI] [PubMed] [Google Scholar]
- Taborsky M.1984Broodcare helpers in the cichlid fish Lamprologus brichardi: their costs and benefits. Anim. Behav. 32, 1236–1252 10.1016/S0003-3472(84)80241-9 (doi:10.1016/S0003-3472(84)80241-9) [DOI] [Google Scholar]
- Taborsky M., Limberger D.1981Helpers in fish. Behav. Ecol. Sociobiol. 8, 143–145 10.1007/BF00300826 (doi:10.1007/BF00300826) [DOI] [Google Scholar]
- van de Pol M., Pen I., Heg D., Weissing F. J.2007Variation in habitat choice and delayed reproduction: adaptive queuing strategies or individual quality differences? Am. Nat. 170, 530–541 10.1086/521237 (doi:10.1086/521237) [DOI] [PubMed] [Google Scholar]
- van Oers K., Drent P. J., Dingemanse N. J., Kempenaers B.2008Personality is associated with extrapair paternity in great tits, Parus major. Anim. Behav. 76, 555–563 10.1016/j.anbehav.2008.03.011 (doi:10.1016/j.anbehav.2008.03.011) [DOI] [Google Scholar]
- Webster M., Ward A., Hart P.2009Individual boldness affects interspecific interactions in sticklebacks. Behav. Ecol. Sociobiol. 63, 511–520 10.1007/s00265-008-0685-2 (doi:10.1007/s00265-008-0685-2) [DOI] [Google Scholar]
- Werner N. Y., Balshine S., Leach B., Lotem A.2003Helping opportunities and space segregation in cooperatively breeding cichlids. Behav. Ecol. 14, 749–756 10.1093/beheco/arg067 (doi:10.1093/beheco/arg067) [DOI] [Google Scholar]
- Whitehead H.2008Analyzing animal societies: quantitative methods for vertebrate social analysis. Chicago, IL: The University of Chicago Press [Google Scholar]
- Wolf M., van Doorn G. S., Leimar O., Weissing F. J.2007Life-history trade-offs favour the evolution of animal personalities. Nature 447, 581–584 10.1038/nature05835 (doi:10.1038/nature05835) [DOI] [PubMed] [Google Scholar]