Abstract
Animals with the neonatal ventral hippocampal lesion (NVHL) demonstrate altered responsiveness to stress and various drugs reminiscent of that in schizophrenia. Post-pubertal onset of abnormalities suggests the possibility of sex differences in NVHL effects that may model sex differences in schizophrenia. Here we demonstrate that novelty- and MK-801-induced hyperactivity is evident in both male and female NVHL rats, whereas only NVHL males were hyperactive in response to apomorphine. Next, we examined the sex- and NVHL-dependent differences in the activity of the ERK and Akt pathways. The basal activity of both pathways was higher in females than in males. NVHL reduces the level of phosphorylation of ERK1/2, Akt, and GSK-3 in both sexes, although males show more consistent downregulation. Females had higher levels of G-protein-coupled kinases [G-protein-coupled receptor kinase (GRK)] 3 and 5, whereas the concentrations of other GRKs and arrestins were the same. In the nucleus accumbens, the concentration of GRK5 in females was elevated by NVHL to the male level. The data demonstrate profound sex differences in the expression and activity of signalling molecules that may underlie differential susceptibility to schizophrenia.
Keywords: Akt, ERK1/2, G-protein-coupled receptor kinase, neonatal ventral hippocampal lesion, schizophrenia, sex differences
Introduction
Schizophrenia is a severe mental disorder with poorly understood aetiology. One influential concept is the neurodevelopmental hypothesis of schizophrenia. It posits that schizophrenia pathology results from neurodevelopmental abnormalities caused by genetic and/or environmental factors. The neurodevelopmental deficits lead to modifications of critical signalling pathways and ultimately result in clinical manifestations of schizophrenia. The adult onset of behavioural symptoms induced by neonatal insults is reproduced in rats that undergo neonatal lesion of the ventral hippocampus [neonatal ventral hippocampal lesion (NVHL)]. In adulthood, NVHL leads to behaviours mimicking symptoms of schizophrenia, such as exaggerated behaviour responses to novelty, stress, and direct or indirect dopamine (DA) agonists, disrupted prepulse inhibition of the acoustic startle response, impaired social behaviour, and working-memory deficits (Lipska, 2004; Lipska et al. 1993; Tseng et al. 2009). Behavioural abnormalities in NVHL animals may be linked to the abnormal activity of the mesocortical and mesolimbic dopaminergic systems. Neurons in the prefrontal cortex (PFC) and nucleus accumbens (NAc) show exaggerated responses to the stimulation of the ventral tegmental area (Goto & O'Donnell, 2002, 2004; Tseng et al. 2006, 2007). These responses may be due to altered dopaminergic modulation of cortical pyramidal neurons leading to their enhanced excitability (Tseng et al. 2007) and/or to alterations in the GABAergic cortical transmission (François et al. 2009).
Although NVHL rats show exacerbated DA-related behaviours, biochemical data about dopaminergic mechanisms affected by NVHL are inconsistent. Rats with NVHL show altered basal and stimulated DA release in NAc (Corda et al. 2006; Lillrank et al. 1999a; Wan et al. 1996). Conflicting findings regarding alterations in DA receptor expression following NVHL have been reported (Black et al. 1998; El-Rawas et al. 2009; Flores et al. 1996a, b; Lillrank et al. 1999b; Lipska et al. 2003; Schroeder et al. 1999). DA receptors modulate, directly or indirectly, multiple signalling pathways. Dopaminergic drugs and DA depletion or elevation affect the activity of the ERK pathway (Ahmed et al. 2008; Beaulieu et al. 2006; Bezard et al. 2005; Bychkov et al. 2007a; Cai et al. 2000; Chen et al. 2004; Choe et al. 2002; Gerfen et al. 2002; Nagai et al. 2007; Pavon et al. 2006; Pozzi et al. 2003; Shi & McGinty, 2007; Valjent et al. 2000, 2004; Wang et al. 2005; Zhang et al. 2004). Previous studies have documented that the activity of the Akt pathway is under the influence of the dopaminergic system (Alimohamad et al. 2005; Beaulieu et al. 2004, 2006; Brami-Cherrier et al. 2002; Bychkov et al. 2007a; Chen et al. 2007; Gil et al. 2003; McGinty et al. 2008; Novikova et al. 2005; Roh et al. 2007; Shi & McGinty, 2007; Svenningsson et al. 2003; Wei et al. 2007). In addition to G-protein-dependent modulation of the signalling pathways, arrestin-mediated deactivation of the Akt pathway via DA D2/D3 receptors has also been reported (Beaulieu et al. 2007a, b). Therefore, alterations in the dopaminergic transmission caused by NVHL may be reflected in the abnormal activity of the cortical signalling pathways. Surprisingly, the effect of NVHL on the activity of these signalling pathways has never been examined.
DA receptors belong to the G-protein-coupled receptor (GPCR) subfamily. The responsiveness of many GPCRs is controlled by a two-step desensitization mechanism involving phosphorylation of a ligand-activated receptor by a G-protein-coupled receptor kinase (GRK) and subsequent high-affinity binding of an uncoupling protein, arrestin (Gurevich & Gurevich, 2006b). Arrestin binding shields the cytoplasmic surface of the receptor precluding further G protein activation. DA D1 and D2 receptors have been shown to desensitize in a GRK- and arrestin-dependent manner (Kim et al. 2001; Macey et al. 2004, 2005), although the DA D3 receptor is desensitization-deficient due to its resistance to GRK-mediated phosphorylation (Kim et al. 2001, 2005). The availability of GRKs and arrestins is an important regulator of the rate and extent of the receptor desensitization. Increased concentration of GRKs and arrestins promotes desensitization and reduces G-protein-mediated signalling, whereas reduced availability of GRKs/arrestins enhances receptor signalling in live animals (Bohn et al. 1999, 2003; Gainetdinov et al. 1999, 2003). Arrestins, in addition to mediating homologous desensitization of GPCRs, serve as scaffolds redirecting GPCR activity to multiple signalling pathways (Gurevich & Gurevich, 2006a). It is conceivable that NVHL alters the expression of GRK and/or arrestin isoforms, which would affect the signalling of DA receptors as well as that of other GPCRs. However, this possibility has never been investigated.
The majority of mechanistic studies of the NVHL model have been performed using male animals. However, sex differences both in schizophrenia (Angermeyer et al. 1990; Grossman et al. 2008; Hafner, 2003; Hafner et al. 1993; Larsen et al. 1996) and the activity of the brain signalling pathways (Kritze & Creutz, 2008; Zhou et al. 2009) have been documented. Therefore, it is conceivable that some of the NVHL effects are sex-specific. To further explore molecular signalling mechanisms of schizophrenia, we studied alteration in the ERK and Akt signalling following NVHL in male and female rats. We also examined the expression of GRKs and arrestins in both sexes. A comprehensive comparative assessment of signalling modifications induced by neonatal stress in males and females may improve our understanding of the molecular mechanism involved in schizophrenia.
Materials and methods
Neonatal lesion
Pregnant Sprague–Dawley rats were obtained at gestational day 14 (Charles River Laboratories, USA) and housed in the Vanderbilt University's animal facility under a 12-h light/dark cycle (lights on 06:00 hours) with food and water available ad libitum. All procedures followed NIH guidelines and were approved by the institutional IACUC. On postnatal day (PD) 7, male and female pups were anaesthetized by placing them on wet ice for 10–15 min. Pups were randomly assigned to receive NVHL or sham lesion. Ibotenic acid [3 μg in 0.3 μl PBS (pH 7.4)] or an equal volume of vehicle at rate 0.1 μl/min was infused bilaterally into the ventral hippocampus at coordinates A=−3.0 mm, L=3.5 mm, H=−5.5 mm from bregma. After surgery, the pups were warmed and returned to their dams. On PD 21, the animals were weaned, separated by sex and type of infusion, and housed three to a cage. The effect of the oestrous cycle on the NVHL-induced behaviour and activity signalling pathways was not specifically investigated in this study.
Behavioural testing
On PD 60, the spontaneous and apomorphine-stimulated locomotor activity was assessed in a circular (30 cm diameter) apparatus. The detector registered the number of 15° turns in any direction, and the data were converted into centimetres travelled. Spontaneous locomotion was recorded for 30 min, then rats were removed from apparatus, treated with D1/D2 dopaminergic agonist apomorphine (0.75 mg/kg s.c.) and locomotor activity was recorded for an additional 60 min. Seventy-two hours later, the rats were treated with non-competitive NMDA antagonist MK-801 (0.2 mg/kg i.p.) and tested for locomotor activity for 120 min.
Tissue preparation and histology
All rats were sacrificed by decapitation under pentobarbital anaesthesia 72 h after the last behavioural testing [half-life of MK-801 in the brain is 2 h (Vezzani et al. 1989)]. The heads were placed in a dish with liquid nitrogen to rapidly chill the brains, then the brains were quickly removed and frozen on dry ice. Samples of the PFC (plates 8–10 according to Paxinos & Watson, 1998); included all of the prelimbic and infralimbic cortices of these plates, NAc (included all of the shell and core of plates 13–15), rostral caudate putamen (CPu; all of the CPu of plates 13–15) were prepared from frozen 100-μm-thick sections as previously described (Ahmed et al. 2007; Bychkov et al. 2007a). This process allows for good anatomical precision while keeping the tissue frozen at all times. Tissue was immediately dissolved in cold lysis solution (Ambion, USA) containing the protein denaturing agent guanidine thiocyanate that inhibits enzymatic activity. Samples were stored at −80 °C until needed. We found that phosphorylated proteins are stable in lysis solution at −80 °C for several months. This sample preparation procedure offers good preservation of the protein phosphorylation state, sufficient to detect changes in the in-vivo phosphorylation levels induced by drugs or loss of DA as described previously (Bychkov et al. 2007a). The middle portion of the brain containing the hippocampus was fixed overnight in 4% paraformaldehyde, cryoprotected in 30% sucrose, and frozen on dry ice. The 35-μm-thick sections were cut on a cryostat and stained with Cresyl Violet to verify the size and location of the lesion.
Quantitative Western blotting
Samples for Western blotting were prepared as described previously (Ahmed et al. 2007; Bychkov et al. 2007a). Electrophoresis and transfer onto Immobilon-P (Millipore, USA) membrane were performed essentially as previously described (Bezard et al. 2005) using Criterion cassettes (Bio-Rad, USA) with 26 wells per gel. Equal amounts of protein from each animal were loaded on the gel (0.625–5 μg). Arrestins were detected with arrestin2-specific (Mundell et al. 1999) (1:9000) or arrestin3-specific (Orsini & Benovic, 1998) (1:900) affinity-purified rabbit polyclonal antibodies. We used rabbit polyclonal antibodies (Santa Cruz Biotechnology, USA) to quantify GRK2 (1:500), GRK3 (1:300), GRK5 (1:500), and GRK6 (1:300). To measure phosphorylated proteins, the following antibodies (all from Cell Signaling Technologies, USA) and dilutions were used: mouse anti-phospho-p44/42 MAPK (Thr202/Tyr204) (1:2000), rabbit anti-phospho-Akt (Thr308) (1:1000); rabbit anti-phospho-Akt (Ser473) (1:1000), rabbit anti-phospho-glycogen synthase kinase (GSK)-α/β (Ser21/9) (1:1000), rabbit anti-phospho-mTOR (Ser2448) (1:1000), rabbit anti-phospho-PDK1 (Ser241) (1:1000). To detect total proteins, anti-p44/42 MAPK, anti-Akt, anti-GSK-3α, anti-PDK1, anti-mTOR, and anti-Rictor rabbit antibodies were used (Cell Signaling Technologies), all in 1:1000 dilution. For arrestins, dilutions of standards containing a 1:1 mix of Escherichia coli-expressed purified bovine arrestin2 and arrestin3 (Gurevich et al. 2002) in sample buffer were loaded onto each gel along with samples. For quantification of GRKs, we used bovine GRK2 and GRK3 (Kim et al. 1993), human GRK5 (Kunapuli et al. 1994), and GRK6 (Loudon & Benovic, 1994), purified as described in the given references. We used purified total and phospho-ERK2 (pERK) (expressed in E. coli, purified, and measured as described by Waas et al. 2003) as standards to determine absolute ERK concentrations. To identify bands corresponding to phosphorylated MEK1/2, Akt at Thr308 and Ser473, phosphoinositide-dependent kinase 1 (PDK1) at Ser241, and mTOR at Ser2448, we used lysates prepared from HEK293 cells stimulated with FGF (10 ng/ml) for 5 min. To identify bands on the blot corresponding to GSK-3α and GSK-3β, we used lysates prepared from HEK293 cells stimulated with insulin (60 nM) for 5 min. Appropriate dilutions of proteins were loaded onto each gel alongside the samples to generate calibration curves. Arrestin2, 3; GRK2, 3, 5, 6, total and pERK are expressed in ng/mg protein. Other proteins are expressed in arbitrary units.
For Western blots the grey values of the bands were measured on X-ray film using Versadoc 4 (Bio-Rad). To account for the background, the local background subtraction algorithm (the background determined in the area immediately surrounding the band of interest) provided by Versadoc software was used. The optical densities of the bands corresponding to arrestins, GRKs, pERK and total ERK were converted into nanograms of the respective protein per milligram of protein using the calibration curves constructed with protein standards of known concentrations. For other proteins, for which no purified standard proteins were available, lysates of HEK293 cells were stimulated with either FGF (10 ng/ml, 5 min; for MEK1/2, and Akt) or insulin (50 mg/ml, 30 min; for GSK-3 and mTOR). The standards aided in the band identification, ensured linearity, and reduced blot-to-blot variability. Calibration curves were fitted to linear equations using Prism 4.0 (GraphPad Software, USA). The values for arrestins, GRKs, pERK and total ERK were expressed in ng/mg of total protein; for all other proteins in arbitrary units/mg of total protein. The arbitrary unit for MEK1/2 and Akt was defined as the amount of respective protein (phosphorylated or total) in 1 μg and for GSK-3 and mTOR in 3 μg of the total lysate protein of stimulated HEK293 cells.
Statistics
StatView software (SAS Institute, USA) was used for statistical analysis. Locomotor activity and biochemistry data were analysed by two-way ANOVA with lesion and sex as main factors. Where significant effects of factors or lesion × sex interaction were detected, the data were post-hoc analysed for male and female separately by two-tailed Student's t test. Differences of p<0.05 were considered significant. Each significant finding was verified in two independent experiments based on specific hypothesis (to account for multiple comparisons).
Results
The lesion and behaviour
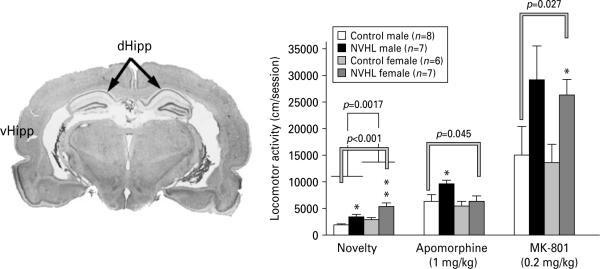
Injection of ibotenic acid in ventral hippocampus of neonatal rats resulted significant damage of the ventral hippocampus with boundaries AP 4.3–5.8 and DV 4.2–9.0 (Fig. 1a). The typical extent of the NVHL in this work corresponds to maximal or near maximal extents of the lesion seen in other studies (Tseng et al. 2007). The dorsal hippocampus, the amygdalo-hippocampal transitional area, and the entorhinal cortex regions were spared in all cases.
Fig. 1.
Neonatal ventral hippocampal lesion (NVHL) was produced by the infusion of ibotenic acid into the ventral hippocampus of neonatal (postnatal day 7; PD 7) rat pups of both sexes. (a) A Nissl-stained coronal section of the adult rat brain demonstrating a typical extent of the lesion. (b) The graph shows elevated activity in the neonatally lesioned rats in response to novel environment (novelty), apomorphine or MK-801 administration. The rats were lesioned at PD 7 and tested at PD 56. The data were analysed by two-way ANOVA with group (control vs. lesion) and sex as main factors. Single-line brackets indicate the difference between the sexes; double-line brackets indicate the differences between the control and lesioned groups; groups across sexes. * p<0.05, ** p<0.01 for respective control group by Student's t test.
NVHL significantly increased spontaneous (novelty-induced) locomotor activity in both male and female postpubertal rats [F(1, 44)=19.6, p<0.0001]. The level of spontaneous locomotion in female rats of both control and NVHL groups was significantly higher than in males [F(1, 44)=11.13, p<0.05] (Fig. 1b). However, there was no significant interaction between the factors. Apomorphine injection caused significant increase in locomotor activity. The two-way ANOVA analysis yielded significant effect of lesion on the apomorphine-induced locomotor activity [F(1, 44)=4.26, p=0.045]. There was no significant effect of sex (p=0.052) on apomorphine-stimulated locomotion and no lesion × sex interaction. A more detailed analysis demonstrated that only males with NVHL had significantly higher apomorphine-stimulated locomotor activity than control males, whereas females with NVHL were not significantly different from control females (Fig. 1b). Furthermore, whereas both control and NVHL male rats demonstrated significant increase in the locomotor activity upon apomorphine administration (p=0.023 and 0.0015 for control and NVHL, respectively, according to Wilcoxon's signed ranks test), only control females significantly increased their locomotor activity in response to apomorphine (p=0.041 and 0.48, respectively). Thus, novelty-induced locomotor activity was higher in female rats, and both sexes respond to NVHL by increasing spontaneous locomotion. In contrast, the apomorphine-stimulated locomotion was quantitatively similar in normal males and females, but only males respond to NVHL by increased apomorphine-induced locomotion. Conversely, MK-801 increased locomotor activity in control or NVHL male and female animals to a similar degree (Fig. 1b).
Sex difference and the effect of NVHL on the activity of the MEK-ERK pathway
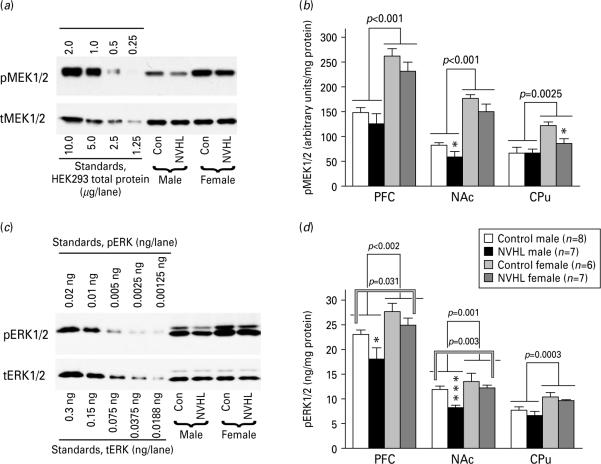
Next, we examined the activity of the MEK-ERK pathway in males and females with NVHL. The levels of phospho-MEK1/2 were higher in females than in males in all brain regions (Fig. 2a, b). We detected no effect of lesion in any region (in NAc, the lesion effect approached significance with p=0.052). However, the level of phospho-MEK1/2 was reduced by the lesion in NAc in males and CPu in females (Fig. 2b). Similarly to MEK1/2, the phosphorylation of ERK1/2 was higher in males than in females in all brain regions, effect of sex [PFC: F(1, 24)=11.61, p=0.002; NAc: 13.45, p=0.0012; CPu: 18.4, p=0.0003] (Fig. 2c, d). The phosphorylation of ERK1/2 was significantly reduced by NVHL in PFC [F(1, 24)=5.27, p=0.031], and NAc (F=10.92, p=0.003), but not in CPu (Fig. 2d). The activation of ERK1/2 was altered by NVHL in both males and females, since there was no significant lesion × sex interaction. Separate analysis in males and females demonstrated that NVHL significantly reduced the ERK1/2 phosphorylation in males only (in PFC and NAc). There were no differences in the levels of total MEK1/2 or ERK1/2 between males and females or in response to NVHL (Fig. 2a,c).
Fig. 2.
Modulation of activity of the MEK-ERK pathway by sex and neonatal ventral hippocampal lesion (NVHL). (a) Representative Western blots for phosphorylated (upper panel) and total (lower panel) MEK1/2 in the PFC. Note a much high levels of phospho-MEK1/2 in females compared to males (no differences in total MEK1/2 level). Left five lanes (standards): lysates of HEK293 stimulated with FGF (10 ng/ml for 5 min (amount of total protein in μg loaded per lane is indicated for each blot). (b) Quantification of Western blot data for phospho-MEK1/2. Brackets indicate the difference between the sexes across control and lesion groups; * p<0.05 for respective control group by Student's t test. (c) Representative Western blots for phosphorylated (upper panel) and total (lower panel) ERK1/2. Note a much high levels of pERK1/2 in females compared to males reduced concentration of pERK in neonatally lesioned animals of both sexes (no differences in the total ERK1/2 level). Standards (left five lanes) are the same as for MEK1/2 (amount of total protein in μg loaded per lane is indicated for each blot). (d) Quantification of Western blot data for pERK1/2. Single-line brackets indicate the difference between the sexes across control and lesion groups; double-line brackets indicate the differences between the control and lesioned groups; groups across sexes.* p<0.05, *** p<0.001 for respective control group by Student's t test.
Sex difference and the effect of NVHL on the Akt pathway
Akt activity
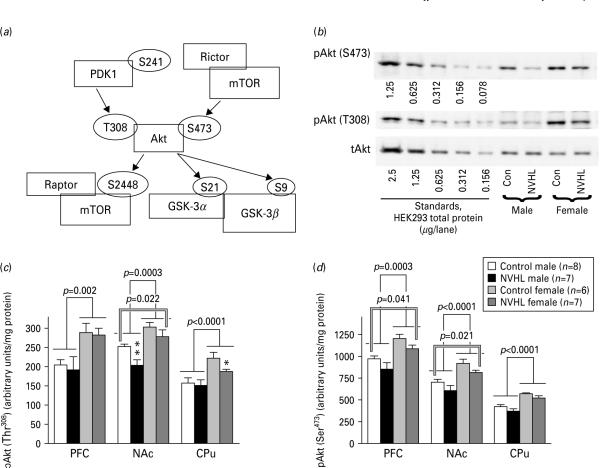
Figure 3a represents the proteins and their phosphorylation sites in the Akt pathways investigated in the study. The Akt pathway mediates the effects of DA via DA D2/D3 receptors (Beaulieu et al. 2007a, b) and is affected by DA depletion and dopaminergic drugs (Bychkov et al. 2007a). Akt is regulated by phosphorylation at multiple sites (Fig. 3a). The phosphorylation at Thr308 by PDK1 is obligatory for activation (Chan et al. 1999). The role of phosphorylation at Ser3473 by mTOR complex 2 (mTOR in complex with Rictor; Fig. 3a) remains poorly understood, but may precede phosphorylation at Thr308 and promote recognition and activation of Akt by PDK1 (Sarbassov et al. 2005; Yang et al. 2002). Furthermore, Ser3473 phosphorylation may regulate Akt substrate specificity (Guertin et al. 2006; Jacinto et al. 2006; Manning & Cantley, 2007). Female rats displayed higher level of Akt phosphorylation at Thr308 than males in all brain regions examined [PFC: F(1, 24)=12.08, p=0.002; NAc: 17.52, p=0.0003; CPu: 22.35, p<0.0001] (Fig. 3b, c). The Akt phosphorylation at Ser473 was also significantly higher in females than in males [PFC: F=18.1, NAc: 24.96, CPu: 30.2; p<0.001] (Fig. 3b, d).
Fig. 3.
Modulation of activity of the Akt pathway by sex and neonatal ventral hippocampal lesion (NVHL). (a) Simplified diagram of the Akt pathway indicating the proteins (in rectangles) and phosphorylation sites (in ellipses) examined in the study. Arrows indicate phosphorylation by upstream kinases at specific sites. (b) Representative Western blots for Akt phosphorylated at Ser473 (upper panel), Akt phosphorylated at Thr408 and total Akt (lower panel) in the NAc. Left five lanes (standards): lysate of HEK293 stimulated with FGF (10 ng/ml) for 5 min (amount of total protein in μg loaded per lane is indicated for each blot). (c) Quantification of Western blot data for phospho-Akt (Thr308) (means±S.E.M.). The data were analysed by two-way ANOVA with group (control vs. lesion) and sex as main factors. Single-line brackets indicate the difference between the sexes; double-line brackets indicate the differences between the control and lesioned groups across sexes. * p<0.05, ** p<0.01 for respective control group by Student's t test. (d) Graph showing quantification of the Western blot for data phospho-Akt (Ser473) (means±S.E.M.). Single-line brackets indicate the difference between the sexes; double-line brackets indicate the differences between the control and lesioned groups across sexes.
NVHL leads to significant reduction in the Akt phosphorylation at Thr308 in NAc [F(1, 24)=6, p=0.02] and at Ser473 in PFC (F=4.65, p=0.041) and NAc (F=6.1, p=0.021) (Fig. 3c, d). The effect of NVHL on the Akt phosphorylation at Thr308 in NAc was due to changes in males (p=0.0077) rather than females (p=0.38). In contrast, in CPu the change in females was more evident (Fig. 3c). There were no differences in the total Akt level between the groups or sexes (Fig. 3b).
Akt substrates
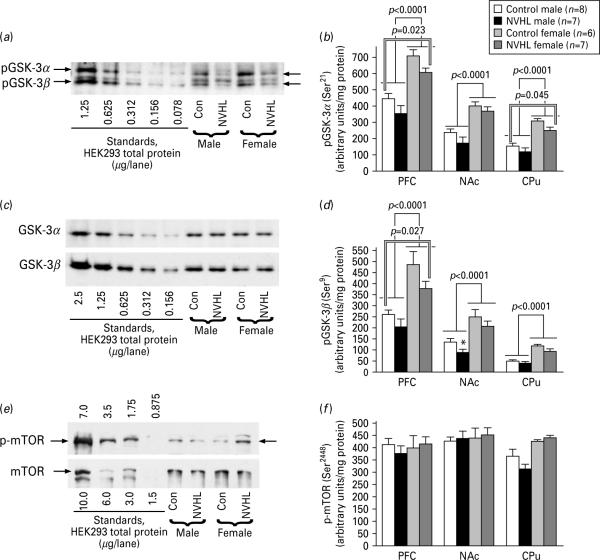
GSK-3α,β is one of the best characterized substrates of Akt (Manning & Cantley, 2007) (Fig. 3a) and is known to be involved in mediating dopaminergic signalling in the brain (Beaulieu et al. 2007a, b). The GSK-3α(Ser21) (Fig. 4a, b) and GSK-3β(Ser9) (Fig. 4a, d) phosphorylation levels were uniformly higher in females than in males in all brain regions examined. NVHL significantly reduced the phosphorylation levels of GSK-3α in PFC (F=5.9, p=0.023) and CPu (F=4.46, p=0.045). PFC, but not CPu, also showed significant reduction in the GSK-3β phosphorylation across sexes (F=5.54, p=0.02). In CPu, only males displayed significant reduction in the phosphorylation of GSK-3β (p=0.031) (Fig. 4d). No differences were seen in the total concentration of GSK-3α,β (Fig. 4c).
Fig. 4.
The effect of sex and neonatal ventral hippocampal lesion (NVHL) on the phosphorylation of GSK-3 and mTOR. (a) Representative Western blots for phosphorylated GSK-3α and GSK-3βbin the PFC. Note a much high levels of both phosphorylated GSK-3 isoforms in females compared to males and reduced concentration of phospho-GSK-3 in neonatally lesioned animals of both sexes. Left five lanes (standards): lysate of HEK293 stimulated with insulin (60 nM) for 5 min (amount of total protein in μg loaded per lane is indicated for each blot). (b) Quantification of Western blot data for phospho-GSK-3α(Ser21). Single-line brackets indicate the difference between the sexes across control and lesion groups; double-line brackets indicate the differences between the control and lesioned groups across sexes. (c) Representative Western blots for total GSK-3α (upper panel) and GSK-3β (lower panel) in the PFC. Note similar expression of both isoforms in males and females and the lack of effect of thge lesion. (d) Quantification of Western blot data for phospho-GSK-3β(Ser9). Single-line brackets indicate the difference between the sexes across control and lesion groups; double-line brackets indicate the differences between the control and lesioned groups across sexes. * p<0.05 for respective control group by Student's t test. (e) Representative Western blots for phosphorylated at Ser2448 (upper panel) and total (lower panel) mTOR. Standards (left four lanes) are lysates of HEK293 stimulated with FGF (10 ng/ml) for 5 min (amount of total protein in μg loaded per lane is indicated for each blot). Note a high level of individual variability but no differences between males and females or control and lesioned animals. (f) Quantification of Western blot data for phospho-mTOR (Ser2448). No significant differences are detected between the sexes or control vs. lesioned animals.
Akt phosphorylates mTOR at Ser2448 (Yang & Guan, 2007) (Fig. 3a). We measured the level of mTOR (Ser2448) phosphorylation and found no significant difference between males and females and no effect of NVHL (Fig. 4e, f). There were also no differences in the total levels of mTOR (Fig. 4e).
Akt activation
We examined the phosphorylation of PDK1 at Ser241, which is required for the kinase activation (Casamayor et al. 1999). However, in contrast to Akt, which is turned on by PDK1-dependent phosphorylation, the PDK1 activity is regulated largely by the substrate conformation and subcellular relocalization (Toker & Newton, 2000). We found no differences between sexes and no effect of NVHL on the level of the PDK1 phosphorylation at Ser241 or on the total PDK1 concentration (Fig. 5) suggesting that Ser241 phosphorylation is not rate-limiting for PDK1 activity in these conditions. The second regulatory residue in Akt, Ser473, is phosphorylated by mTOR in complex with Rictor (Yang & Guan, 2007). We found no differences in the concentration of Rictor between males and females in any of the brain regions examined. There was also no effect of NVHL on the concentration of Rictor in either sex (Fig. 5).
Fig. 5.
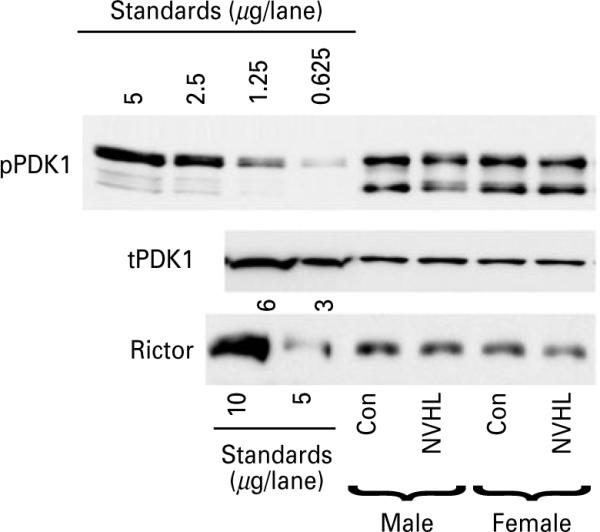
The effect of sex and neonatal ventral hippocampal lesion (NVHL) on the phosphorylation and expression of PDK1 and expression of Rictor. Representative Western blots for phospho-PDK1 (Ser241) (upper panel), total PDK1 (middle panel), and Rictor (lower panel) in the PFC. Left five lanes on the phospho-PDK1 blot: lysate of HEK293 stimulated with FGF (10 ng/ml) for 5 min (amount of total protein in μg loaded per lane is indicated for each blot) used as standards. Note that phospho-PDK1 (Ser241) runs as multiple bands. For total PDK1 and Rictor, lysates of normal HEK293 cells were used for standards (amount of total protein in μg loaded per lane is indicated for each blot). Note the lack of sex differences or NVHL effects.
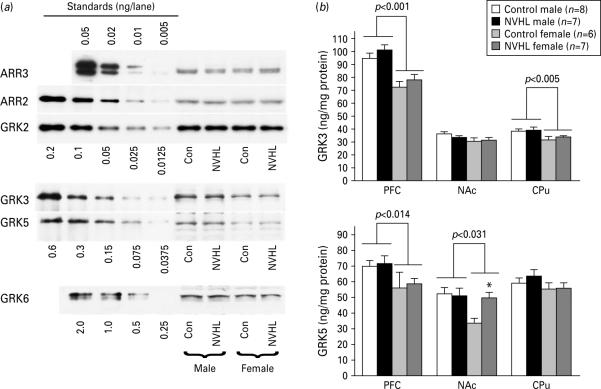
Sex and region difference in the expression of arrestins and GRKs
No significant differences in the concentration of arrestin2 or 3 and in GRK2 or 6 (Fig. 6a) were detected in males and females or following NVHL in any of the brain structures examined. The expression of GRK3 was significantly higher in males than in females in PFC and CPu, but not in NAc [F(1, 24)=30.54 and 9.67, p<0.0001 and p=0.005, respectively] regardless of the lesion (Fig. 6a, b). There was no effect of lesion or significant sex × lesion interaction in any of the forebrain structures. Similarly to GRK3, the expression of GRK5 in PFC and NAc, but not in CPu, was significantly higher in males than in females [F(1, 24)=6.97 and 5.23, p=−0.014 and p=0.031, respectively] (Fig. 6a, b). In NAc, NVHL significantly elevated the expression of GRK5 in females to the male level, but no changes were seen in males (Fig. 6b).
Fig. 6.
The differences between males and females in the expression of G-protein-coupled receptor kinases (GRKs). (a) Representative Western blots for arrestin and GRK isoforms in the PFC. Left five lanes show purified recombinant arrestin and GRK proteins of known concentration (amount loaded per lane in ng is indicated for each isoform). Note even expression of arrestins, GRK2 and GRK6 in males and females and in control and lesioned rats. Also note much higher levels of GRK3 and GRK5 in males compared to females. (b) Quantification of Western blot data for GRK3 (upper panel) and GRK5 (lower panel) isoforms. The data were analysed by two-way ANOVA with group (control vs. lesion) and sex as main factors. Brackets indicate the difference between the sexes across control and lesion groups; * p<0.05 for respective control group by Student's t test.
Discussion
The aim of present study was to examine alterations in the signalling pathways induced by NVHL in adult animals that may be important in schizophrenia. The neonatal destruction of the ventral hippocampus induces behavioural deficits resembling those seen in schizophrenia (Tseng et al. 2009). Molecular mechanisms responsible for the NVHL-induced pathology remain poorly understood. PFC seems to be the predominant site of functional deficits induced by NVHL (Tseng et al. 2006, 2008), although abnormalities in NAc and CPu have also been noted (Flores et al. 2005; Laplante et al. 2005). Adult rats with NVHL exhibit enhanced sensitivity to dopaminergic and glutamatergic stimuli as well as to stimuli known to induce DA release in the mesolimbic system, such as stress and exposure to novel environment (Tseng et al. 2007, 2009). Recently, adult supersensitivity of animals with NVHL to α1-adrenergic and muscarinic stimulation has been reported (Kamath et al. 2008; Laplante et al. 2005). These alterations suggest that NVHL induces specific modification of signalling pathways in the affected brain regions responsible for enhanced sensitivity to specific stimuli.
Here we demonstrate, in agreement with previous reports (Tseng et al. 2009), that NVHL significantly increased spontaneous locomotor activity in post-pubertal rats. Interestingly, females displayed the overall higher novelty-induced locomotor activity, but showed similar increase following NVHL. In our study, the novelty and MK-801 effects were similar in males and females, whereas the apomorphine-induced hyperlocomotion was evident in male rats only. Previous studies showed increased locomotion produced by novelty and amphetamine in adult male as well as female NVHL rats (Black et al. 1998; Silva-Gomez et al. 2003). The reason for this disagreement may be the drug used (amphetamine vs. apomorphine) or differences in oestrous cycle of examined female rats. Another explanation may be the difference in the way locomotion was measured: we recorded locomotion in a confined area as opposed to the open-field-type apparatus commonly used. It is possible that the experimental setting devoid of stress/anxiety component helped to reveal sex differences. Spontaneous locomotion was shown to be independent of hormonal fluctuations, whereas DA-induced locomotion is affected by the oestrous cycle (Kazandjian et al. 1987; Steiner et al. 1980). In any case, the hyperactivity in NVHL males develops earlier and is more pronounced than in females (Black et al. 1998). The sexually dimorphic behavioural and physiological effects of NVHL may derive from the sexual dimorphism of the dopaminergic system. Male and female rats exhibit differences in the anatomy of the dopaminergic system (Kritze & Creutz, 2008), sensitivity to dopaminergic drugs (Zhou et al. 2009), and responsiveness of the dopaminergic system to stress (Sullivan et al. 2009). However, gender-specific molecular mechanisms involved in the NVHL pathology remain largely unexplored.
An increase in the level of ERK protein and mRNA in the thalamus, but not in other brain regions, in schizophrenia has been reported (Kyosseva, 2004). However, it remains unclear whether these data reflect the increased MEK-ERK activation in schizophrenia or the elevated expression is actually not accompanied by excessive activation, since no phosphorylation measurements were performed. Due to long post-mortem delay, it is uncertain whether it is possible to reliably estimate the activity of the MEK-ERK pathway in human post-mortem samples. Therefore, animal models are of particular value for examining the functional role of signalling pathways where the activity of signalling molecules is regulated mostly by phosphorylation. The NVHL-induced reduction in the activity of the MEK-ERK pathways found in the present study argues that schizophrenia might be associated with a suppression of MEK-ERK activity. The fact that clinically effective antipsychotic drugs such as clozapine increase ERK1/2 activation in the PFC (Ahmed et al. 2008; Browning et al. 2005) indicates that elevated activity of the ERK pathway may be beneficial in schizophrenia. Furthermore, lower vulnerability of females in comparison to males to behavioural effects of NVHL correlates with the elevated basal activity of the MEK-ERK pathway coupled with less consistent reduction of this activity by NVHL. Taken together, these data provide evidence that schizophrenia pathology may be associated with reduced MEK-ERK activity.
The functional significance of the Akt-GSK-3 signalling pathway in schizophrenia is highlighted by human post-mortem studies that demonstrated altered levels of these signalling proteins in patients with schizophrenia at post-mortem, although there are notable inconsistencies in published reports. Thus, the concentrations of Akt, phospho-Akt (Ser473), and phospho-GSK-3β (Ser9) were found to decrease in the frontal cortex and hippocampus (Amar et al. 2008; Emamian et al. 2004; Zhao et al. 2006). These data suggest an increase in the GSK-3 activity in schizophrenia. In contrast, other studies found reduced levels of GSK-3β protein (Beasley et al. 2001; Kozlovsky et al. 2000) coupled with reduced GSK-3 activity (Kozlovsky et al. 2001) and unchanged Akt concentration (Amar et al. 2008; Ide et al. 2006) in the frontal cortex of patients with schizophrenia. Our data on decreased levels of Akt and GSK-3 phosphorylation in NVHL animals are in agreement with the human studies suggesting increased GSK-3 activity in schizophrenia. The elevated locomotor activity of NVHL animals may be due to enhanced GSK-3 activity (seen as reduced levels of Akt-GSK-3 phosphorylation compared to sham-operated animals). Previous studies demonstrated that enhanced activity of GSK-3 caused by overexpression of GSK-3 in the brain results in an increased locomotion (Prickaerts et al. 2006), whereas locomotion is attenuated in animals treated with GSK-3 inhibitors (Beaulieu et al. 2004; Gould et al. 2004) and in GSK-3 knockout mice (Beaulieu et al. 2004). Typical and atypical antipsychotics have been shown to inhibit GSK-3 activity (increased the level of phosphorylated GSK-3) in rat brain (Alimohamad et al. 2005; Emamian et al. 2004; Li et al. 2007), although opposite results have also been reported (Kozlovsky et al. 2006). Interestingly, GSK-3 as a substrate of Akt seems to be specifically involved, since the level of Akt-dependent phosphorylation of another substrate, mTOR, was unaffected by NVHL and not significantly different between males and females. Therefore, the available evidence supports the role of elevated GSK-3 activity in schizophrenia. Moreover, it is conceivable that reduced basal GSK-3 activity in females may confer resistance to some behavioural manifestations of schizophrenia. The critical importance of the Akt-GSK-3 pathway in schizophrenia is further underscored by the recent evidence that the pathway is linked to the activity of DISC1, the key susceptibility genes in schizophrenia (Kim et al. 2009; Mao et al. 2009). Furthermore, since DISC1-dependent regulation of Akt signalling plays a role in brain development (Mao et al. 2009), this may be one of the molecular mechanisms involved in the effects of NVHL and sex on the Akt signalling.
We found that females have significantly lower concentrations of GRK3 and GRK5 isoforms, whereas the concentration of GRK2 and GRK6 isoforms and arrestins were the same in both sexes. We did not detect any effect of NVHL on the expression of arrestins or GRKs in adult males. However, in females, the level of GRK5 in the NAc, which was normally significantly lower than that in males, was increased by NVHL to the male level. The role of the GRK/arrestin proteins in schizophrenia pathology is poorly defined. We recently found reduced expression of several GRK/arrestin isoforms in the cortical regions of schizophrenia patients, with GRK3 and GRK5 being specifically affected in younger and GRK6 in older patients with schizophrenia (Bychkov et al. 2007b). Furthermore, chronic treatment with haloperidol and clozapine induced drug-specific alterations in the concentration of arrestin and GRK isoforms in key brain regions (Ahmed et al. 2008). These data suggest that GRKs may be involved in the pathology of schizophrenia. The present finding of sexually dimorphic expression of GRK3 and GRK5 isoforms suggest these proteins may play a role in sex differences in the basal activity of signalling pathways and in differential vulnerability to the neonatal insult.
Basal differences in the concentration of signalling molecules and activity of the signalling pathways between males and females seen in the present study could presage differential responsiveness, quantitatively or qualitatively, of males and females to a combination of genes and life events that trigger schizophrenia. They could also play a decisive role in differential responsiveness of sexes to psychotropic drugs. Cataloguing and understanding sex differences in the basal and evoked activity of the signalling pathways may shed light on the pathophysiology of schizophrenia and lead to the design of highly effective antipsychotic drugs.
Acknowledgements
We are grateful to Dr Jeffrey Benovic (Thomas Jefferson University, Philadelphia, PA) for the gift of purified GRKs. We thank Dr Vsevolod Gurevich (Vanderbilt University, Nashville, TN) for the gift of purified arrestins. We are indebted to Dr Kevin Dalby (University of Texas at Austin, Austin, TX) for the gift of purified total and phosphorylated ERK. This work was supported by NS45117 (to E.V.G.).
Footnotes
Statement of Interest None.
References
- Ahmed MR, Bychkov E, Gurevich VV, Benovic JL, et al. Altered expression and subcellular distribution of GRK subtypes in the dopamine-depleted rat basal ganglia is not normalized by L-DOPA treatment. Journal of Neurochemistry. 2007;104:1622–1636. doi: 10.1111/j.1471-4159.2007.05104.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed MR, Gurevich VV, Dalby KN, Benovic JL, et al. Haloperidol and clozapine differentially affect the expression of arrestins, receptor kinases, and extracellular signal-regulated kinase activation. Journal of Pharmacology and Experimental Therapeutics. 2008;325:276–283. doi: 10.1124/jpet.107.131987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alimohamad H, Rajakumar N, Seah YH, Rushlow W. Antipsychotics alter the protein expression levels of beta-catenin and GSK-3 in the rat medial prefrontal cortex and striatum. Biological Psychiatry. 2005;57:533–542. doi: 10.1016/j.biopsych.2004.11.036. [DOI] [PubMed] [Google Scholar]
- Amar S, Shaltiel G, Mann L, Shamir A, et al. Possible involvement of post-dopamine D2 receptor signalling components in the pathophysiology of schizophrenia. International Journal of Neuropsychopharmacology. 2008;11:197–205. doi: 10.1017/S1461145707007948. [DOI] [PubMed] [Google Scholar]
- Angermeyer MC, Kuhn L, Goldstein JM. Gender and the course of schizophrenia: differences in treated outcomes. Schizophrenia Bulletin. 1990;16:293–307. doi: 10.1093/schbul/16.2.293. [DOI] [PubMed] [Google Scholar]
- Beasley C, Cotter D, Khan N, Pollard C, et al. Glycogen synthase kinase-3beta immunoreactivity is reduced in the prefrontal cortex in schizophrenia. Neuroscience Letters. 2001;302:117–120. doi: 10.1016/s0304-3940(01)01688-3. [DOI] [PubMed] [Google Scholar]
- Beaulieu J-M, Tirotta E, Sotnikova TD, Masri B, et al. Regulation of Akt signaling by D2 and D3 dopamine receptors in vivo. Journal of Neuroscience. 2007a;27:881–885. doi: 10.1523/JNEUROSCI.5074-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaulieu JM, Gainetdinov RR, Caron MG. The Akt-GSK-3 signaling cascade in the actions of dopamine. Trends of Pharmacological Sciences. 2007b;28:166–172. doi: 10.1016/j.tips.2007.02.006. [DOI] [PubMed] [Google Scholar]
- Beaulieu JM, Sotnikova TD, Gainetdinov RR, Caron MG. Paradoxical striatal cellular signaling responses to psychostimulants in hyperactive mice. Journal of Biological Chemistry. 2006;281:32072–32080. doi: 10.1074/jbc.M606062200. [DOI] [PubMed] [Google Scholar]
- Beaulieu JM, Sotnikova TD, Yao WD, Kockeritz L, et al. Lithium antagonizes dopamine-dependent behaviors mediated by an AKT/glycogen synthase kinase 3 signaling cascade. Proceedings of the National Academy of Sciences USA. 2004;101:5099–5104. doi: 10.1073/pnas.0307921101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezard E, Gross CE, Qin L, Gurevich VV, et al. L-DOPA reverses the MPTP-induced elevation of the arrestin2 and GRK6 expression and enhanced ERK activation in monkey brain. Neurobiology of Disease. 2005;18:323–335. doi: 10.1016/j.nbd.2004.10.005. [DOI] [PubMed] [Google Scholar]
- Black MD, Lister S, Hitchcock JM, Van Giersbergen P, et al. Neonatal hippocampal lesion model of schizophrenia in rats: sex differences and persistence of effects into maturity. Drug Development and Research. 1998;43:206–213. [Google Scholar]
- Bohn LM, Gainetdinov RR, Sotnikova TD, Medvedev IO, et al. Enhanced rewarding properties of morphine, but not cocaine, in beta(arrestin)-2 knock-out mice. Journal of Neuroscience. 2003;23:10265–10273. doi: 10.1523/JNEUROSCI.23-32-10265.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohn LM, Lefkowitz RJ, Gainetdinov RR, Peppel K, et al. Enhanced morphine analgesia in mice lacking beta-arrestin2. Science. 1999;286:2495–2498. doi: 10.1126/science.286.5449.2495. [DOI] [PubMed] [Google Scholar]
- Brami-Cherrier K, Valjent E, Garcia M, Pages C, et al. Dopamine induces a PI3-kinase-independent activation of Akt in striatal neurons: a new route to cAMP response element-binding protein phosphorylation. Journal of Neuroscience. 2002;22:8911–8921. doi: 10.1523/JNEUROSCI.22-20-08911.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browning JL, Patel T, Brandt PC, Young KA, et al. Clozapine and the mitogen-activated protein kinase signal transduction pathway: implications for antipsychotic actions. Biological Psychiatry. 2005;57:617–623. doi: 10.1016/j.biopsych.2004.12.002. [DOI] [PubMed] [Google Scholar]
- Bychkov E, Ahmed MR, Dalby KN, Gurevich EV. Dopamine depletion and subsequent treatment with L-DOPA, but not the long-lived dopamine agonist pergolide, enhances activity of the Akt pathway in the rat striatum. Journal of Neurochemistry. 2007a;102:699–711. doi: 10.1111/j.1471-4159.2007.04586.x. [DOI] [PubMed] [Google Scholar]
- Bychkov ER, Ahmed MR, Haroutunian V, Gurevich EV. Expression of arrestins and G protein-coupled receptor kinases in schizophrenia and schizoaffective disorder. Society for Neuroscience Annual Meeting; San Diego, CA. 2007b. [Google Scholar]
- Cai G, Zhen X, Uryu K, Friedman E. Activation of extracellular signal-regulated protein kinases is associated with a sensitized locomotor response to D(2) dopamine receptor stimulation in unilateral 6-hydroxydopamine-lesioned rats. Journal of Neuroscience. 2000;20:1849–1857. doi: 10.1523/JNEUROSCI.20-05-01849.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casamayor A, Morrice NA, Alessi DR. Phosphorylation of Ser-241 is essential for the activity of 3-phosphoinositide-dependent protein kinase-1: identification of five sites of phosphorylation in vivo. Biochemical Journal. 1999;342:287–292. [PMC free article] [PubMed] [Google Scholar]
- Chan TO, Rittenhouse SE, Tsichlis PN. AKT/PKB and other D3 phosphoinositide-regulated kinases: kinase activation by phosphoinositide-dependent phosphorylation. Annual Review of Biochemistry. 1999;68:965–1014. doi: 10.1146/annurev.biochem.68.1.965. [DOI] [PubMed] [Google Scholar]
- Chen J, Rusnak M, Luedtke RR, Sidhu A. D1 dopamine receptor mediates dopamine-induced cytotoxicity via the ERK signal cascade. Journal of Biological Chemistry. 2004;279:39317–39330. doi: 10.1074/jbc.M403891200. [DOI] [PubMed] [Google Scholar]
- Chen PC, Lao CL, Chen JC. Dual alteration of limbic dopamine D1 receptor-mediated signalling and the Akt/GSK3 pathway in dopamine D3 receptor mutants during the development of methamphetamine sensitization. Journal of Neurochemistry. 2007;100:225–241. doi: 10.1111/j.1471-4159.2006.04203.x. [DOI] [PubMed] [Google Scholar]
- Choe ES, Chung KT, Mao L, Wang JQ. Amphetamine increases phosphorylation of extracellular signal-regulated kinase and transcription factors in the rat striatum via group I metabotropic glutamate receptors. Neuropsychopharmacology. 2002;27:565–575. doi: 10.1016/S0893-133X(02)00341-X. [DOI] [PubMed] [Google Scholar]
- Corda MG, Piras G, Giorgi O. Neonatal ventral hippocampal lesions potentiate amphetamine-induced increments in dopamine efflux in the core, but not the shell, of the nucleus accumbens. Biological Psychiatry. 2006;60:1188–1195. doi: 10.1016/j.biopsych.2006.03.047. [DOI] [PubMed] [Google Scholar]
- El-Rawas R, Saadé NE, Thiriet N, Atweh S, et al. Developmental changes in the mRNA expression of neuropeptides and dopamine and glutamate receptors in neonates and adult rats after ventral hippocampal lesion. Schizophenia Research. 2009;113:298–307. doi: 10.1016/j.schres.2009.05.009. [DOI] [PubMed] [Google Scholar]
- Emamian ES, Hall D, Birnbaum MJ, Karayiorgou M, et al. Convergent evidence for impaired AKT1-GSK3beta signaling in schizophrenia. Nature Genetics. 2004;36:131–137. doi: 10.1038/ng1296. [DOI] [PubMed] [Google Scholar]
- Flores G, Alquicer G, Silva-Gómez AB, Zaldivar G, et al. Alterations in dendritic morphology of prefrontal cortical and nucleus accumbens neurons in post-pubertal rats after neonatal excitotoxic lesions of the ventral hippocampus. Neuroscience. 2005;133:463–470. doi: 10.1016/j.neuroscience.2005.02.021. [DOI] [PubMed] [Google Scholar]
- Flores G, Barbeau D, Quirion R, Srivastava LK. Decreased binding of dopamine D3 receptors in limbic subregions after neonatal bilateral lesion of rat hippocampus. Journal of Neuroscience. 1996a;16:2020–2026. doi: 10.1523/JNEUROSCI.16-06-02020.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flores G, Wood GK, Liang J-J, Quirion R, et al. Enhanced amphetamine sensitivity and increased expression of dopamine D2 receptors in postpubertal rats after neonatal excitotoxic lesions of the medial prefrontal cortex. Journal of Neuroscience. 1996b;16:7366–7375. doi: 10.1523/JNEUROSCI.16-22-07366.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- François J, Ferrandon A, Koning E, Angs MJ, et al. Selective reorganization of GABAergic transmission in neonatal ventral hippocampal-lesioned rats. Intertational Journal of Neuropsychopharmacology. 2009;12:1097–1110. doi: 10.1017/S1461145709009985. [DOI] [PubMed] [Google Scholar]
- Gainetdinov RR, Bohn LM, Sotnikova TD, Cyr M, et al. Dopaminergic supersensitivity in G protein-coupled receptor kinase 6-deficient mice. Neuron. 2003;38:291–303. doi: 10.1016/s0896-6273(03)00192-2. [DOI] [PubMed] [Google Scholar]
- Gainetdinov RR, Bohn LM, Walker JK, Laporte SA, et al. Muscarinic supersensitivity and impaired receptor desensitization in G protein-coupled receptor kinase 5-deficient mice. Neuron. 1999;24:1029–1036. doi: 10.1016/s0896-6273(00)81048-x. [DOI] [PubMed] [Google Scholar]
- Gerfen CR, Miyachi S, Paletzki R, Brown P. D1 dopamine receptor supersensitivity in the dopamine-depleted striatum results from a switch in the regulation of ERK1/2/MAP kinase. Journal of Neuroscience. 2002;22:5042–5054. doi: 10.1523/JNEUROSCI.22-12-05042.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil M, Zhen X, Friedman E. Prenatal cocaine exposure alters glycogen synthase kinase-3beta (GSK3beta) pathway in select rabbit brain areas. Neuroscience Letters. 2003;349:143–146. doi: 10.1016/s0304-3940(03)00852-8. [DOI] [PubMed] [Google Scholar]
- Goto Y, O'Donnell P. Delayed mesolimbic system alteration in a developmental animal model of schizophrenia. Journal of Neuroscience. 2002;22:9070–9077. doi: 10.1523/JNEUROSCI.22-20-09070.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goto Y, O'Donnell P. Prefrontal lesion reverses abnormal mesoaccumbens response in an animal model of schizophrenia. Biological Psychiatry. 2004;55:172–176. doi: 10.1016/s0006-3223(03)00783-2. [DOI] [PubMed] [Google Scholar]
- Gould TD, Einat H, Bhat R, Manji HK. AR-A014418, a selective GSK-3 inhibitor, produces antidepressant-like effects in the forced swim test. International Journal of Neuropsychopharmacology. 2004;7:387–390. doi: 10.1017/S1461145704004535. [DOI] [PubMed] [Google Scholar]
- Grossman LS, Harrow M, Rosen C, Faull R, et al. Sex differences in schizophrenia and other psychotic disorders: a 20-year longitudinal study of psychosis and recovery. Comprarative Psychiatry. 2008;49:523–529. doi: 10.1016/j.comppsych.2008.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guertin DA, Stevens DM, Thoreen CC, Burds AA, et al. Ablation in mice of the mTORC components raptor, rictor, or mLST8 reveals that mTORC2 is required for signaling to Akt-FOXO and PKCalpha, but not S6K1. Developmental Cell. 2006;11:859–871. doi: 10.1016/j.devcel.2006.10.007. [DOI] [PubMed] [Google Scholar]
- Gurevich EV, Benovic JL, Gurevich VV. Arrestin2 and arrestin3 are differentially expressed in the rat brain during postnatal development. Neuroscience. 2002;109:421–436. doi: 10.1016/s0306-4522(01)00511-5. [DOI] [PubMed] [Google Scholar]
- Gurevich EV, Gurevich VV. Arrestins: ubiquitous regulators of cellular signaling pathways. Genome Biology. 2006a;7:236. doi: 10.1186/gb-2006-7-9-236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurevich VV, Gurevich EV. The structural basis of arrestin-mediated regulation of G-protein-coupled receptors. Pharmacology and Therapeutics. 2006b;110:465–502. doi: 10.1016/j.pharmthera.2005.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hafner H. Gender differences in schizophrenia. Psychoneuroendocrinology. 2003;28(Suppl. 2):17–54. doi: 10.1016/s0306-4530(02)00125-7. [DOI] [PubMed] [Google Scholar]
- Hafner H, Maurer K, Loffler W, Riecher-Rossler A. The influence of age and sex on the onset of early course of schizophrenia. British Journal of Psychiatry. 1993;162:80–86. doi: 10.1192/bjp.162.1.80. [DOI] [PubMed] [Google Scholar]
- Ide M, Ohnishi T, Murayama M, Matsumoto I, et al. Failure to support a genetic contribution of AKT1 polymorphisms and altered AKT signaling in schizophrenia. Journal of Neurochemistry. 2006;99:277–287. doi: 10.1111/j.1471-4159.2006.04033.x. [DOI] [PubMed] [Google Scholar]
- Jacinto E, Facchinetti V, Liu D, Soto N, et al. SIN1/MIP1 maintains rictor-mTOR complex integrity and regulates Akt phosphorylation and substrate specificity. Cell. 2006;127:125–137. doi: 10.1016/j.cell.2006.08.033. [DOI] [PubMed] [Google Scholar]
- Kamath A, Al-Khairi I, Bhardwaj S, Srivastava LK. Enhanced alpha1 adrenergic sensitivity in sensorimotor gating deficits in neonatal ventral hippocampus-lesioned rats. International Journal of Neuropsychopharmacology. 2008;11:1085–1096. doi: 10.1017/S1461145708008845. [DOI] [PubMed] [Google Scholar]
- Kazandjian A, Spyraki C, Sfikakis A, Varonos DD. Apomorphine-induced behaviour during the oestrous cycle of the rat. Neuropharmacology. 1987;26:1037–1045. doi: 10.1016/0028-3908(87)90246-2. [DOI] [PubMed] [Google Scholar]
- Kim CM, Dion SB, Onorato JJ, Benovic JL. Expression and characterization of two beta-adrenergic receptor kinase isoforms using the baculovirus expression system. Receptor. 1993;3:39–55. [PubMed] [Google Scholar]
- Kim JY, Duan X, Liu CY, Jang MH, et al. DISC1 regulates new neuron development in the adult brain via modulation of AKT-mTOR signaling through KIAA1212. Neuron. 2009;63:761–773. doi: 10.1016/j.neuron.2009.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KM, Gainetdinov RR, Laporte SA, Caron MG, et al. G protein-coupled receptor kinase regulates dopamine D3 receptor signaling by modulating the stability of a receptor-filamin-beta-arrestin complex. A case of autoreceptor regulation. Journal of Biological Chemistry. 2005;280:12774–12780. doi: 10.1074/jbc.M408901200. [DOI] [PubMed] [Google Scholar]
- Kim KM, Valenzano KJ, Robinson SR, Yao WD, et al. Differential regulation of the dopamine D2 and D3 receptors by G protein-coupled receptor kinases and beta-arrestins. Journal of Biological Chemistry. 2001;276:37409–37414. doi: 10.1074/jbc.M106728200. [DOI] [PubMed] [Google Scholar]
- Kozlovsky N, Amar S, Belmaker RH, Agam G. Psychotropic drugs affect Ser9-phosphorylated GSK-3 beta protein levels in rodent frontal cortex. International Journal of Neuropsychopharmacology. 2006;9:337–342. doi: 10.1017/S1461145705006097. [DOI] [PubMed] [Google Scholar]
- Kozlovsky N, Belmaker RH, Agam G. Low GSK-3beta immunoreactivity in postmortem frontal cortex of schizophrenic patients. American Journal of Psychiatry. 2000;157:831–833. doi: 10.1176/appi.ajp.157.5.831. [DOI] [PubMed] [Google Scholar]
- Kozlovsky N, Belmaker RH, Agam G. Low GSK-3 activity in frontal cortex of schizophrenic patients. Schizophenia Research. 2001;52:101–105. doi: 10.1016/s0920-9964(00)00174-2. [DOI] [PubMed] [Google Scholar]
- Kritze RMF, Creutz LM. Region and sex differences in constituent dopamine neurons and immunoreactivity for intracellular estrogen and androgen receptors in mesocortical projections in rats. Journal of Neuroscience. 2008;28:9525–9535. doi: 10.1523/JNEUROSCI.2637-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunapuli P, Onorato JJ, Hosey MM, Benovic JL. Expression, purification, and characterization of the G protein-coupled receptor kinase GRK5. Journal of Biological Chemistry. 1994;269:1099–1105. [PubMed] [Google Scholar]
- Kyosseva SV. Differential expression of mitogen-activated protein kinases and immediate early genes fos and jun in thalamus in schizophrenia. Progress in Neuropsychopharmacology and Biological Psychiatry. 2004;28:997–1006. doi: 10.1016/j.pnpbp.2004.05.017. [DOI] [PubMed] [Google Scholar]
- Laplante F, Nakagawasai O, Srivastava LK, Quirion R. Alterations in behavioral responses to a cholinergic agonist in post-pubertal rats with neonatal ventral hippocampal lesions : relationship to changes in muscarinic receptor levels. Neuropsychopharmacology. 2005;30:1076–1087. doi: 10.1038/sj.npp.1300640. [DOI] [PubMed] [Google Scholar]
- Larsen TK, McGlashan TH, Johannessen JO, Vlbe-Hansen LD. First-episode schizophrenia : II. Premorbid patterns by gender. Schizophrenia Bulletin. 1996;22:257–269. doi: 10.1093/schbul/22.2.257. [DOI] [PubMed] [Google Scholar]
- Li X, Rosborough KM, Friedman AB, Zhu W, et al. Regulation of mouse brain glycogen synthase kinase-3 by atypical antipsychotics. International Journal of Neuropsychopharmacology. 2007;10:7–19. doi: 10.1017/S1461145706006547. [DOI] [PubMed] [Google Scholar]
- Lillrank SM, Lipska BK, Kolachana BS, Weinberger DR. Attenuated extracellular dopamine levels after stress and amphetamine in the nucleus accumbens of rats with neonatal ventral hippocampal damage. Journal of Neural Transmission. 1999a;106:183–196. doi: 10.1007/s007020050150. [DOI] [PubMed] [Google Scholar]
- Lillrank SM, Lipska BK, Weinberger DR, Fredholm BB, et al. Adenosine and dopamine receptor antagonist binding in the rat ventral and dorsal striatum : lack of changes after a neonatal bilateral lesion of the ventral hippocampus. Neurochemistry International. 1999b;34:235–244. doi: 10.1016/s0197-0186(99)00008-x. [DOI] [PubMed] [Google Scholar]
- Lipska BK. Using animal models to test a neurodevelopmental hypothesis of schizophrenia. Journal of Psychiatry and Neuroscience. 2004;29:282–286. [PMC free article] [PubMed] [Google Scholar]
- Lipska BK, Jaskiw GE, Weinberger DR. Postpubertal emergence of hyperresponsiveness to stress and to amphetamine after neonatal excitotoxic hippocampal damage: a potential animal model of schizophrenia. Neuropsychopharmacology. 1993;9:67–75. doi: 10.1038/npp.1993.44. [DOI] [PubMed] [Google Scholar]
- Lipska BK, Lerman DN, Khaing ZZ, Weickert CS, et al. Gene expression in dopamine and GABA systems in an animal model of schizophrenia : effects of antipsychotic drugs. European Journal of Neuroscience. 2003;18:391–402. doi: 10.1046/j.1460-9568.2003.02738.x. [DOI] [PubMed] [Google Scholar]
- Loudon RP, Benovic JL. Expression, purification, and characterization of the G protein-coupled receptor kinase GRK6. Journal of Biological Chemistry. 1994;269:22691–22697. [PubMed] [Google Scholar]
- Macey TA, Gurevich VV, Neve KA. Preferential Interaction between the dopamine D2 receptor and Arrestin2 in neostriatal neurons. Molecular Pharmacology. 2004;66:1635–1642. doi: 10.1124/mol.104.001495. [DOI] [PubMed] [Google Scholar]
- Macey TA, Liu Y, Gurevich VV, Neve KA. Dopamine D1 receptor interaction with arrestin3 in neostriatal neurons. Journal of Neurochemistry. 2005;93:128–134. doi: 10.1111/j.1471-4159.2004.02998.x. [DOI] [PubMed] [Google Scholar]
- Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129:1261–1274. doi: 10.1016/j.cell.2007.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao Y, Ge X, Frank CL, Madison JM, et al. Disrupted in schizophrenia 1 regulates neuronal progenitor proliferation via modulation of GSK3beta/beta-catenin signaling. Cell. 2009;136:1017–1031. doi: 10.1016/j.cell.2008.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGinty JF, Shi XD, Schwendt M, Saylor A, et al. Regulation of psychostimulant-induced signaling and gene expression in the striatum. Journal of Neurochemistry. 2008;104:1440–1449. doi: 10.1111/j.1471-4159.2008.05240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mundell SJ, Loudon RP, Benovic JL. Characterization of G protein-coupled receptor regulation in antisense mRNA-expressing cells with reduced arrestin levels. Biochemistry. 1999;38:8723–8732. doi: 10.1021/bi990361v. [DOI] [PubMed] [Google Scholar]
- Nagai T, Takuma K, Kamei H, Ito Y, et al. Dopamine D1 receptors regulate protein synthesis-dependent long-term recognition memory via extracellular signal-regulated kinase 1/2 in the prefrontal cortex. Learning and Memory. 2007;14:117–125. doi: 10.1101/lm.461407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novikova SI, He F, Bai J, Badan I, et al. Cocaine-induced changes in the expression of apoptosis-related genes in the fetal mouse cerebral wall. Neurotoxicology and Teratology. 2005;27:3–14. doi: 10.1016/j.ntt.2004.08.004. [DOI] [PubMed] [Google Scholar]
- Orsini MJ, Benovic JL. Characterization of dominant negative arrestins that inhibit beta-2-adrenergic receptor internalization by distinct mechanisms. Journal of Biological Chemistry. 1998;273:34616–34622. doi: 10.1074/jbc.273.51.34616. [DOI] [PubMed] [Google Scholar]
- Pavon N, Martin AB, Mendialdua A, Moratalla R. ERK phosphorylation and FosB expression are associated with L-DOPA-induced dyskinesia in hemiparkinsonian mice. Biological Psychiatry. 2006;59:64–74. doi: 10.1016/j.biopsych.2005.05.044. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. Academic Press; San Diego, CA: 1998. [Google Scholar]
- Pozzi L, Hakansson K, Usiello A, Borgkvist A, et al. Opposite regulation by typical and atypical anti-psychotics of ERK1/2, CREB and Elk-1 phosphorylation in mouse dorsal striatum. Journal of Neurochemistry. 2003;86:451–459. doi: 10.1046/j.1471-4159.2003.01851.x. [DOI] [PubMed] [Google Scholar]
- Prickaerts J, Moechars D, Cryns K, Lenaerts I, et al. Transgenic mice overexpressing glycogen synthase kinase 3beta : a putative model of hyperactivity and mania. Journal of Neuroscience. 2006;26:9022–9029. doi: 10.1523/JNEUROSCI.5216-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roh MS, Seo MS, Kim Y, Kim SH, et al. Haloperidol and clozapine differentially regulate signals upstream of glycogen synthase kinase 3 in the rat frontal cortex. Experimental and Molecular Medicine. 2007;39:353–360. doi: 10.1038/emm.2007.39. [DOI] [PubMed] [Google Scholar]
- Sarbassov DD, Guertin DA, Ali SM, Sabatini DM. Phosphorylation and regulation of Akt/PKB by the Rictor-mTOR complex. Science. 2005;307:1098–1101. doi: 10.1126/science.1106148. [DOI] [PubMed] [Google Scholar]
- Schroeder H, Grecksch G, Becker A, Bogerts B, et al. Alterations of the dopaminergic and glutamatergic neurotransmission in adult rats with postnatal ibotenic acid hippocampal lesion. Psychopharmacology (Berlin) 1999;145:61–66. doi: 10.1007/s002130051032. [DOI] [PubMed] [Google Scholar]
- Shi X, McGinty JF. Repeated amphetamine treatment increases phosphorylation of extracellular signal-regulated kinase, protein kinase B, and cyclase response element-binding protein in the rat striatum. Journal of Neurochemistry. 2007;103:706–713. doi: 10.1111/j.1471-4159.2007.04760.x. [DOI] [PubMed] [Google Scholar]
- Silva-Gomez AB, Bermudez M, Quirion R, Srivastava LK, et al. Comparative behavioral changes between male and female postpubertal rats following neonatal excitotoxic lesions of the ventral hippocampus. Brain Research. 2003;973:285–292. doi: 10.1016/s0006-8993(03)02537-x. [DOI] [PubMed] [Google Scholar]
- Steiner M, Katz RJ, Carroll BJ. Behavioral effects of dopamine agonists across the estrous cycle in rats. Psychopharmacology (Berlin) 1980;71:147–151. doi: 10.1007/BF00434403. [DOI] [PubMed] [Google Scholar]
- Sullivan RM, Dufresne MM, Waldron J. Lateralized sex differences in stress-induced dopamine release in the rat. Neuroreport. 2009;20:229–232. doi: 10.1097/WNR.0b013e3283196b3e. [DOI] [PubMed] [Google Scholar]
- Svenningsson P, Tzavara ET, Carruthers R, Rachleff I, et al. Diverse psychotomimetics act through a common signaling pathway. Science. 2003;302:1412–1415. doi: 10.1126/science.1089681. [DOI] [PubMed] [Google Scholar]
- Toker A, Newton AC. Cellular signaling: pivoting around PDK-1. Cell. 2000;103:185–188. doi: 10.1016/s0092-8674(00)00110-0. [DOI] [PubMed] [Google Scholar]
- Tseng KY, Amin F, Lewis BL, O'Donnell P. Altered prefrontal cortical metabolic response to mesocortical activation in adult animals with a neonatal ventral hippocampal lesion. Biological Psychiatry. 2006;60:585–590. doi: 10.1016/j.biopsych.2006.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng KY, Chambers RA, Lipska BK. The neonatal ventral hippocampal lesion as a heuristic neurodevelopmental model of schizophrenia. Behavioral Brain Research. 2009;204:295–305. doi: 10.1016/j.bbr.2008.11.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng KY, Lewis BL, Hashimoto T, Sesack SR, et al. A neonatal ventral hippocampal lesion causes functional deficits in adult prefrontal cortical interneurons. Journal of Neuroscience. 2008;28:12691–12699. doi: 10.1523/JNEUROSCI.4166-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng KY, Lewis BL, Lipska BK, O'Donnell P. Post-pubertal disruption of medial prefrontal cortical dopamine-glutamate interactions in a developmental animal model of schizophrenia. Biological Psychiatry. 2007;62:730–738. doi: 10.1016/j.biopsych.2006.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valjent E, Corvol JC, Pages C, Besson MJ, et al. Involvement of the extracellular signal-regulated kinase cascade for cocaine-rewarding properties. Journal of Neuroscience. 2000;20:8701–8709. doi: 10.1523/JNEUROSCI.20-23-08701.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valjent E, Pages C, Herve D, Girault JA, et al. Addictive and non-addictive drugs induce distinct and specific patterns of ERK activation in mouse brain. European Journal of Neuroscience. 2004;19:1826–1836. doi: 10.1111/j.1460-9568.2004.03278.x. [DOI] [PubMed] [Google Scholar]
- Vezzani A, Serafini R, Stasi MA, Caccia S, et al. Kinetics of MK-801 and its effect on quinolinic acid-induced seizures and neurotoxicity in rats. Journal of Pharmacology and Experimental Therapeutics. 1989;249:278–283. [PubMed] [Google Scholar]
- Waas WF, Rainey MA, Szafranska AE, Dalby KN. Two rate-limiting steps in the kinetic mechanism of the serine/threonine specific protein kinase ERK2: a case of fast phosphorylation followed by fast product release. Biochemistry. 2003;42:12273–12286. doi: 10.1021/bi0348617. [DOI] [PubMed] [Google Scholar]
- Wan RQ, Giovanni A, Kafka SH, Corbett R. Neonatal hippocampal lesions induced hyperresponsiveness to amphetamine: behavioral and in vivo microdialysis studies. Behavioral Brain Research. 1996;78:211–223. doi: 10.1016/0166-4328(95)00251-0. [DOI] [PubMed] [Google Scholar]
- Wang C, Buck DC, Yang R, Macey TA, et al. Dopamine D2 receptor stimulation of mitogen-activated protein kinases mediated by cell type-dependent transactivation of receptor tyrosine kinases. Journal of Neurochemistry. 2005;93:899–909. doi: 10.1111/j.1471-4159.2005.03055.x. [DOI] [PubMed] [Google Scholar]
- Wei Y, Williams JM, Dipace C, Sung U, et al. Dopamine transporter activity mediates amphetamine-induced inhibition of Akt through a Ca2+/calmodulin-dependent kinase II-dependent mechanism. Molecular Pharmacology. 2007;71:835–842. doi: 10.1124/mol.106.026351. [DOI] [PubMed] [Google Scholar]
- Yang J, Cron P, Thompson V, Good VM, et al. Molecular mechanism for the regulation of protein kinase B/Akt by hydrophobic motif phosphorylation. Molecular Cell. 2002;9:1227–1240. doi: 10.1016/s1097-2765(02)00550-6. [DOI] [PubMed] [Google Scholar]
- Yang Q, Guan KL. Expanding mTOR signaling. Cell Research. 2007;17:666–681. doi: 10.1038/cr.2007.64. [DOI] [PubMed] [Google Scholar]
- Zhang L, Lou D, Jiao H, Zhang D, et al. Cocaine-induced intracellular signaling and gene expression are oppositely regulated by the dopamine D1 and D3 receptors. Journal of Neuroscience. 2004;24:3344–3354. doi: 10.1523/JNEUROSCI.0060-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Z, Ksiezak-Reding H, Riggio S, Haroutunian V, et al. Insulin receptor deficits in schizophrenia and in cellular and animal models of insulin receptor dysfunction. Schizophrenia Research. 2006;84:1–14. doi: 10.1016/j.schres.2006.02.009. [DOI] [PubMed] [Google Scholar]
- Zhou L, Nazarian A, Sun WL, Jenab S, et al. Basal and cocaine-induced sex differences in the DARPP-32-mediated signaling pathway. Psychopharmacology (Berlin) 2009;203:175–183. doi: 10.1007/s00213-008-1388-0. [DOI] [PMC free article] [PubMed] [Google Scholar]