Abstract
Mutations in the dysferlin gene underlie a group of autosomal recessive muscle-wasting disorders denoted as dysferlinopathies. Dysferlin has been shown to play roles in muscle membrane repair and muscle regeneration, both of which require vesicle-membrane fusion. However, the mechanism by which muscle becomes dystrophic in these disorders remains poorly understood. Although muscle inflammation is widely recognized in dysferlinopathy and dysferlin is expressed in immune cells, the contribution of the immune system to the pathology of dysferlinopathy remains to be fully explored. Here, we show that the complement system plays an important role in muscle pathology in dysferlinopathy. Dysferlin deficiency led to increased expression of complement factors in muscle, while muscle-specific transgenic expression of dysferlin normalized the expression of complement factors and eliminated the dystrophic phenotype present in dysferlin-null mice. Furthermore, genetic disruption of the central component (C3) of the complement system ameliorated muscle pathology in dysferlin-deficient mice but had no significant beneficial effect in a genetically distinct model of muscular dystrophy, mdx mice. These results demonstrate that complement-mediated muscle injury is central to the pathogenesis of dysferlinopathy and suggest that targeting the complement system might serve as a therapeutic approach for this disease.
Introduction
The dysferlin gene is located at chromosome 2p13 and contains over 55 exons. The major transcript is 8.5 kb in length and is expressed strongly in striated muscle and the placenta (1, 2). Mutations in the dysferlin gene lead to several types of muscle-wasting diseases denoted as dysferlinopathies — limb-girdle muscular dystrophy type 2B (LGMD2B) (1, 2), Miyoshi myopathy (MM) (2), and a distal anterior compartment myopathy (DACM) (3). Dysferlinopathies have also been shown to be associated with a late-onset dilative cardiomyopathy (4–6). Dysferlin is a 230-kDa protein that is widely expressed in different tissues and cells including striated muscle (7) and immune cells (8–10). Previous work using dysferlin mutant mice demonstrated that dysferlin plays an essential role in cell membrane repair of striated muscle (5, 11–15). However, it is not fully understood what physiological events lead to muscle pathogenesis when the membrane repair is compromised in dysferlinopathy.
Lengthening contractions (LCs) may cause muscle damage, but several lines of evidence suggest that LCs are not a unique or critical factor for muscle pathogenesis in dysferlinopathy. First, many dysferlinopathy patients were reported to be athletically gifted at a young age and they did not show significant muscle pathology during these sporting activities (16, 17). Second, increased susceptibility to contraction-induced injury of the muscle membrane is usually caused by the loss of the dystrophin-glycoprotein complex (DGC); however, dysferlin-deficient skeletal muscle possesses a structurally intact and stable DGC (11). Indeed, recent data showed that dysferlin-deficient skeletal muscles are equal to normal skeletal muscles in resistance to contraction-induced injury (18). However, following either 15 repetitive large-strain LCs or 150 small-strain LCs, dysferlin-null muscles were observed to experience a strong inflammatory response that delayed their recovery from injury caused by LCs (19, 20). Although the presence of inflammatory infiltrates in muscle is a characteristic of the inflammatory myopathies such as polymyositis and dermatomyositis, several studies have suggested a prominent inflammatory response in the muscle of dysferlinopathy patients (21–23). In about 25% of the cases, the dysferlinopathy patients were initially misdiagnosed as having polymyositis (24, 25). It is enigmatic that dysferlinopathy triggers a prominent inflammatory response, but recent findings have revealed some potential causes. Dysferlin-deficient monocytes from SJL/J mice were reported to have increased phagocytic activity (26), and dysferlin deficiency induces an upregulation of inflammasome (27). However, a more recent study (18) did not find a difference in phagocytic activity of dysferlin-deficient monocytes using C57BL/10-SJL.Dysf mice, which have a more controlled genetic background. Instead, the authors reported an impaired secretion of chemotactic molecules in dysferlin-deficient myocytes, thus reducing neutrophil recruitment at an early stage of regeneration with subsequent incomplete muscle remodeling and ultimate inflammatory responses and development of muscular dystrophy (18). All of these previous findings implicate a complex involvement of the immune system in the pathogenesis of dysferlinopathy.
Our present study identifies the complement system, an important part of the innate immune system that promotes inflammation, as a key player for the pathogenesis of dysferlinopathy. We have shown that the expression of the complement factors are induced in dysferlin-deficient skeletal muscle and that genetic ablation of the complement factor C3 ameliorates muscular dystrophy in dysferlin-null mice.
Results
Contractile properties and muscle pathophysiology in response to LCs.
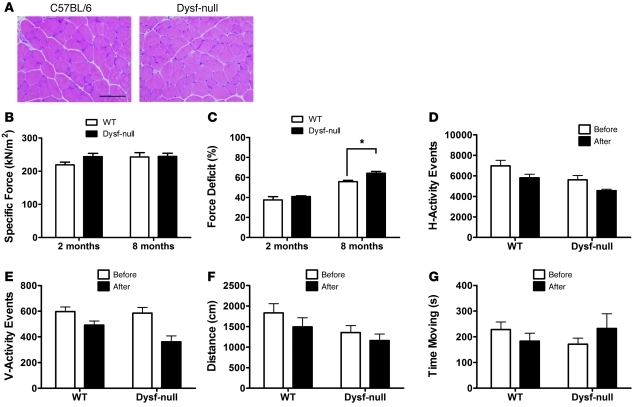
Muscular dystrophy shows 2 important features: reduced muscle force generation and increased susceptibility to LC-induced damage. We examined the effect of dysferlin deficiency on force production and force deficit in response to LC–induced muscle injury by measuring the in vitro contractile properties of the extensor digitorum longus (EDL) muscles (28) of dysferlin-deficient mice and age-matched controls (WT). Unlike quadriceps, the EDL muscle in dysferlin-null mice even at 8 months of age had a limited overt pathology (Figure 1A), which allowed us to study the consequence of dysferlin deficiency on contractile properties without preexisting dystrophic alterations. The specific force (kN/m2) produced by the dysferlin-deficient EDL muscles was not significantly different from that in control muscles at either 2 months (219 ± 8 in WT vs. 244 ± 10 in dysferlin-null; n = 4 for each; P = 0.10) or 8 months (243 ± 13 in WT vs. 245 ± 10 in dysferlin-null; n = 8 for each; P = 0.92) of age (Figure 1B). These results indicate that dysferlin is not directly involved in the generation of force by skeletal muscles. Previously, Chiu et al. (18) showed that dysferlin-deficient tibialis anterior muscle was not susceptible to 1 or 2 LCs with 40% stretch. To detect any subtle difference in the susceptibility to LC-induced damage, we delivered seven 30% stretches to a maximally activated EDL muscles (29), which resulted in a force deficit (percentage of force loss after the stretch protocol) of approximately 37% in WT EDL muscle (Figure 1C). The force deficits in the dysferlin-deficient EDL muscle (41% ± 1% in dysferlin-null vs. 37% ± 3% in WT; n = 4 for each; P = 0.35) were not significantly different from those in WT EDL muscle at 2 months of age. Even with the stretch level of 40%, the force deficit in the dysferlin-deficient EDL muscle at 8 months of age was only slightly increased (56% ± 1% in WT vs. 64% ± 2% in dysferlin-null; n = 4 for each; P = 0.01) (Figure 1C).
Figure 1. Contractile properties of the dysferlin-deficient EDL muscles and muscle pathophysiology in response to LCs in vivo.
(A) Representative micrographs of H&E-stained EDL muscle sections from C57BL/6 (WT) and dysferlin-null (Dysf-null) mice at 8 months of age. Scale bar: 100 μm. (B) Muscle-specific force and (C) force deficit after 7 LCs were measured for EDL muscles isolated from WT and dysferlin-null mice (n = 4 for each strain at 2 or 8 months of age). Quantified horizontal activity (D), vertical activity (E), travel distance (F), and the moving time (G) before (white columns) and after (black columns) exercise for 2-month-old WT (n = 5) and dysferlin-null mice (n = 4). *P < 0.05. Data are expressed as mean ± SEM.
In order to determine the effect of LC-induced muscle damage in vivo, 2-month-old (prepathological) dysferlin-deficient mice were subjected to downhill running. Interestingly, we did not observe a dramatic change in serum creatine kinase (CK) levels (data not shown) or post-run activity (Figure 1, D–G) as compared with mdx mice (30), a mouse model for Duchenne muscular dystrophy (DMD), which carry a mutation in the dystrophin gene (31). Importantly, the examiner was blinded to the genotype of the test animals and used a protocol similar to that reported previously (30). These data suggest that dysferlin-deficient skeletal muscle is not prone to LC-induced muscle damage as dystrophin-deficient skeletal muscle is, and are consistent with the finding that dysferlin-deficient skeletal muscle possesses a structurally intact and stable DGC (11). Therefore, in agreement with recent findings (18), our data demonstrate that LC-induced muscle injury alone is not sufficient to cause muscle necrosis in dysferlinopathy.
Increased proinflammatory gene expression in dysferlin-deficient skeletal muscle.
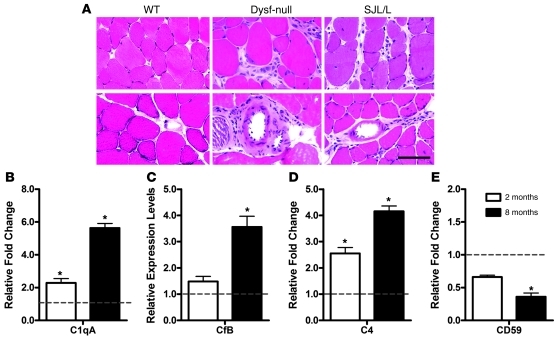
Similarly to human patients, the dysferlin-deficient SJL/J mouse, a natural mouse model of dysferlin deficiency (32), also develops muscular dystrophy with prominent inflammation (33). In addition to developing spontaneous myopathy with inflammation, SJL/J mice are uniquely susceptible to developing experimentally induced myositis (34) and several other autoimmune conditions including experimental autoimmune encephalitis (35). But recently it was shown that the susceptibility to experimental autoimmune encephalomyelitis does not appear to be related to dysferlin deficiency (36). To confirm the correlation of muscle inflammation with dysferlin deficiency, we examined inflammatory features in the skeletal muscle of dysferlin-null mice (in C57BL/6 background) through histological and immunofluorescence analyses. Muscle isolated from the mice younger than 4 months of age with little or no muscle pathology did not show inflammatory infiltration (data not shown). However, muscle isolated from 8-month-old dysferlin-null mice had small numbers of infiltrating lymphocytes (Figure 2A). Perivascular as well as endomysial leukocytes were observed in dysferlin-null mice. Consistent with previous observations of muscle infiltrate cell populations in dysferlin-deficient patients (21–23), approximately 80%–90% of these leukocytes were positive for the macrophage marker Mac-1 (data not shown).
Figure 2. Muscle inflammation and expression of complement factors in dysferlin-deficient mice.
(A) H&E-stained muscle section of 8-month-old WT, dysferlin-null (Dysf-null), and SJL/J mice. Endomysial and perivascular infiltration was observed in both dysferlin-null and SJL/J mice. Scale bar: 100 μm. (B–E) Real-time quantitative RT-PCR was performed on quadriceps muscles of 2- or 8-month-old dysferlin-null versus WT mice (n = 3). The mRNA levels of the complement genes were normalized to Gapdh mRNA. Expression of C1qa (B), CFB (C), and C4 (D) were elevated during the early and active phases of muscle pathology in dysferlin-deficient skeletal muscle compared with levels in skeletal muscle taken from age-matched control mice, whereas the complement inhibitor CD59 (E) was significantly downregulated in old mice. *P < 0.05. Data are expressed as mean ± SEM.
To understand the molecular mechanism of the muscle inflammation in dysferlinopathy, we next sought to identify the proinflammatory genes induced by dysferlin deficiency. We analyzed gene expression profiling data published previously with the use of either dysferlin-deficient mouse or human skeletal muscles (14, 37–40). Four of five such microarray studies (14, 37–40) identified the complement factors, including C1qa, C1qb, C1qc, C4, Cfd, and DAF, to be differentially expressed. In order to validate these gene array results, we directly measured the RNA levels of complement factors in skeletal muscle. We performed quantitative real-time RT-PCR on skeletal muscle RNA extracted from 2-month-old (early stage of disease) and 8-month-old (active phase of disease) dysferlin-null mice as well as age-matched C57BL/6 controls. The RNA level of the initiating complement factor C1qa for the classical complement pathway was elevated in skeletal muscle from the dysferlin-deficient mice. The elevation was observed even in the absence of obvious pathology, with levels increased up to 6-fold (when normalized to GAPDH RNA) (Figure 2B). Expression of the complement factors CFB (alternative pathway-specific component) and C4 (common component for classical and lectin-mediated pathway) were also elevated in 8-month-old dysferlin-deficient skeletal muscle compared with those in age-matched controls (Figure 2, C and D). However, the lectin pathway-specific factor MASP2 was not significantly affected in dysferlin-null muscle from young mice and was slightly downregulated in older mice (Supplemental Figure 1; supplemental material available online with this article; doi: 10.1172/JCI42390DS1). In addition, the complement inhibitor CD59 was significantly downregulated in skeletal muscle from old dysferlin-null mice (Figure 2E). These data suggest that upregulation of classical and alternative complement pathways and downregulation of complement inhibitors may play a major role in the muscle damage in dysferlinopathy.
Muscle-specific dysferlin transgenic expression rescues muscle pathology in dysferlin-null mice.
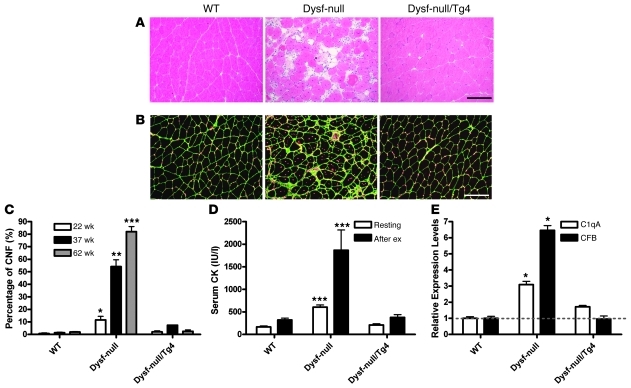
Interestingly, dysferlin has been reported to be expressed in cells of the immune system such as peripheral blood monocytes (8) and neutrophils (41). Although the function of dysferlin in these cells is not clear, dysferlin deficiency was reported recently to increase phagocytic activity in monocytes/macrophages and thus has been proposed as a potential mechanism for the onset of muscle inflammation seen in dysferlinopathy (26). To determine whether the increased phagocytic activity in dysferlin-deficient immune systems is sufficient to cause muscle damage, we generated muscle-specific dysferlin transgenic mice (Supplemental Figure 2). We crossed the dysferlin-null mice with a transgenic line (designated as Tg4) in which a single copy of the dysferlin transgene is expressed in a skeletal muscle–specific fashion under the control of mouse muscle CK promoter. Dysferlin was overexpressed by approximately 8-fold in these transgenic mice, and they did not show any muscle pathology, while 3 high expression lines showed muscle atrophy (Supplemental Figure 2). The dysferlin-null mice at 8 months presented typical myopathic changes, such as central nuclei, fat replacement, necrosis, and regeneration (Figure 3A). These myopathic changes were abrogated in the dysferlin-null/Tg4 mice of the same age except that a low percentage of central nuclei were still present (Figure 3A). The centrally nucleated fibers were counted after staining the muscle section with laminin and DAPI (Figure 3B). At 5 months of age, the quadriceps of male dysferlin-null mice had 11.5% ± 5.8% centrally nucleated fibers, while the quadriceps of the control and dysferlin-null/Tg4 mice had only 0.6% ± 1.1% and 2.0% ± 1.6% (Figure 3C). However, at 8 months of age, the percentage of the central nucleated fibers in the quadriceps of male dysferlin-null mice was dramatically increased to 72.7% ± 11.4%, and this number was only slightly increased to 9.3% ± 3.9% in dysferlin-null/Tg4 mice (Figure 3C). Serum CK levels of the dysferlin-null/Tg4 mice were normal before or after exercise, but those of the dysferlin-null were elevated as reported before (Figure 3D). Consistent with a recent report (42), these data suggest that the presence of dysferlin in skeletal muscle protects muscle from damage even in the presence of a dysferlin-deficient immune system. Furthermore, quantitative RT-PCR examination of the quadriceps muscles from the dysferlin-null/Tg4 mice suggests that transgenic expression of dysferlin normalized the expression of the complement factors C1qA and CFB (Figure 3E). Taken together, the loss of dysferlin in skeletal muscle induces complement upregulation that may lead to the development of muscle pathology accompanied by inflammation.
Figure 3. Muscle-specific transgenic expression of dysferlin rescues muscle pathology in dysferlin-null mice.
(A) H&E-stained quadriceps muscle sections of 38-week-old WT, dysferlin-null, and dysferlin-null/Tg4 mice. The muscle section from dysferlin-null showed typical myopathic changes including central nucleation, fiber size variation, inflammation, fibrosis, and fat replacement. No prominent myopathic changes were observed in the dysferlin-null/Tg4 muscle. (B) Laminin-stained (green) and DAPI-stained (red) quadriceps muscle section of WT, dysferlin-null, and dysferlin-null/Tg4 mice. Scale bars: 200 μm. (C) Percentage of fibers containing central nuclei (CNF) in the quadriceps muscles of 22-, 37-, and 62-week-old WT, dysferlin-null, and dysferlin-null/Tg4 mice (n = 3 or 4 for each group; all males). (D) Measurement of serum CK levels in WT, dysferlin-null, and dysferlin-null/Tg4 mice aged 32–45 weeks before (n = 10, 12, and 15, respectively) and after (n = 4, 3, and 9, respectively) downhill running exercise (after ex). (E) Quantitative RT-PCR examination of complement factors C1qA and CFB expression in the quadriceps muscles of WT, dysferlin-null and dysferlin-null/Tg4 mice (n = 3 for each group). *P < 0.05; **P < 0.01; ***P < 0.001 when compared with the data in either WT or dysferlin-null/Tg4. Data are expressed as mean ± SEM.
Complement deposits in dysferlin-deficient skeletal muscle.
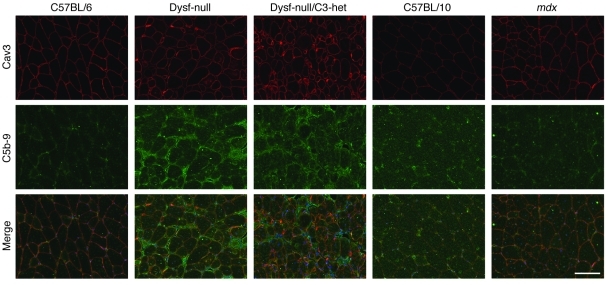
To further determine whether increased complement expression induces complement deposition in dysferlin-deficient skeletal muscle, we stained muscle sections of mice with the antibody against C5b-9 to detect dysferlin expression and the formation of the terminal complement activation product — membrane attack complex (MAC). In skeletal muscles of WT mice, no surface deposition of MAC was detected (Figure 4). However, deposition of MAC at the surface of muscle fibers could be discerned in dysferlin-null mice (Figure 4). To determine whether the MAC deposition observed in dysferlin-null muscle is the consequence of muscular dystrophy, we looked at skeletal muscle from the mdx mouse, a mouse model for DMD (31). We did not detect surface MAC staining in skeletal muscle fibers of mdx mice, although these mice developed more severe muscular dystrophy than dysferlin-null mice (Figure 4).
Figure 4. Immunofluorescence analysis of complement C5b-9 deposition in mouse skeletal muscles.
Immunofluorescence analysis did not detect complement C5b-9 expression in the muscle sections from WT mice (C57BL/6 and C57BL/10) and mdx mice but showed that complement C5b-9 was deposited onto the muscle cell membrane in dysferlin-null mice. All samples were prepared and processed equally at the same time, and all images were taken at the same exposure level. Scale bar: 100 μm.
To test the possibility that the complement deposition shown in dysferlin-null skeletal muscle is species specific, we also examined muscle sections from human patients for complement deposition. Dysferlin-deficient patient muscle sections were stained with an antibody against dysferlin and an antibody against C5b-9, respectively (Figure 5, A–J). Dysferlin was not detected in the muscles from MM (Figure 5B) and LGMD2B patients (Figure 5D). As reported previously, increased cytoplasmic dysferlin expression was observed in the muscles from DMD and LGMD2I patients (Figure 5, C and E), as compared with control muscle (Figure 5A). Interestingly, MAC was observed to be widely deposited onto the sarcolemma of skeletal muscle fibers from MM (Figure 5G) and LGMD2B (Figure 5I) patients while not observed in control muscle (Figure 5F). Although positive MAC staining was also observed in DMD muscle sections, it was only observed in necrotic myofibers where it stained the entire cell (Figure 5H). The nonnecrotic fibers are spared for MAC deposition in DMD (Figure 5H). Similar staining for MAC is observed in nondysferlin forms of LGMD such as LGMD2I (Figure 5J). These data suggest that the surface complement deposition is specific for dysferlin-deficient skeletal muscle and does not appear to be merely a secondary effect after muscle damage.
Figure 5. Immunofluorescence analysis of complement C5b-9 deposition in human patient skeletal muscles.
Muscle sections from control (A and F), MM (B and G), DMD (C and H), LGMD2B (D and I), and LGMD2I (E and J) patients were stained with antibodies against dysferlin (A–E) or C5b-9 (F–J). Complement C5b-9 was not present in control patient muscle fibers, but deposited onto the sarcolemma of MM and LGMD2B muscles. In DMD and LGMD2I muscle sections, complement C5b-9 was detected only in the necrotic muscle fibers (H and J). All samples were prepared and processed equally at the same time, and all images were taken at the same exposure level. Scale bar: 100 μm. Asterisks indicate necrotic muscle fibers.
Genetic ablation of C3 ameliorates muscle pathology in dysferlin-null mice.
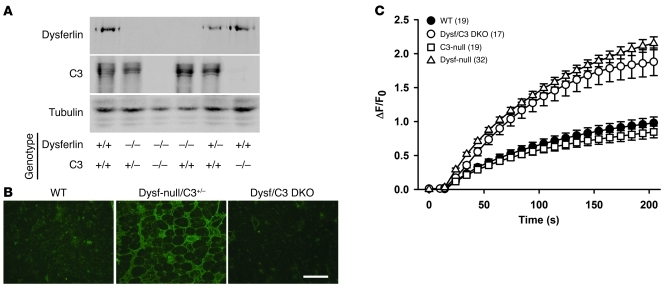
If the complement activation contributes to the muscle damage in dysferlinopathy, we hypothesized that disruption of the complement pathway may ameliorate the dystrophic phenotype in dysferlin-null mice. To test this hypothesis, we examined the effect of genetic deletion of the central component (C3) of the complement system on muscle pathology in dysferlin-null mice. Dysferlin/C3 double-mutant mice were generated by crossing dysferlin-null mice with C3-deficient mice, which were made in the laboratory of Michael C. Carroll (43). Both dysferlin-null mice and C3-null mice had been backcrossed with C57BL/6 mice for more than 6 generations before they were bred together to generate the double-mutant mice. Disruption of both dysferlin and C3 in dysferlin/C3 double-mutant mice was confirmed by Western blotting analysis with the antibodies against dysferlin and C3 (Figure 6A). C3 immunofluorescence analysis of quadriceps muscles showed C3 is widely deposited onto the sarcolemma of dysferlin-deficient muscle, while this staining pattern was not observed in either WT or dysferlin/C3 double-null mice (Figure 6B). To determine whether C3 ablation affects the membrane repair capacity of muscle fibers, we performed the laser-induced membrane damage and repair assay (5, 11, 44) on the muscle fibers isolated from either WT, dysferlin-null, C3-null, or dysferlin/C3 double-null mice. As expected, we found that C3 ablation did not have any significant effect on the membrane-repair capacity of muscle fibers (Figure 6C).
Figure 6. Generation and characterization of dysferlin/C3 double-mutant (Dysf/C3 DKO) mice.
(A) Western blotting analysis of skeletal muscle microsomes prepared from the mice of different genotypes as designated. The expression of dysferlin and C3 was disrupted in the dysferlin/C3 double-null mice. (B) C3 immunofluorescence staining of quadriceps muscle sections from WT, dysferlin-null/C3+/–, and dysferlin/C3 double-null mice. Scale bar: 200 μm. (C) Quantitative analysis of FM 1-43 fluorescence intensity changes (ΔF/F0) following laser-induced membrane damage showed that the membrane repair capacity of muscle fibers was compromised in dysferlin-null mice (triangles) compared with WT (black circles), but disruption of C3 did not significantly affect the membrane repair efficiency in either WT (squares) or dysferlin-null background (white circles). The numbers in parentheses are the numbers of fibers examined. Data are expressed as mean ± SEM.
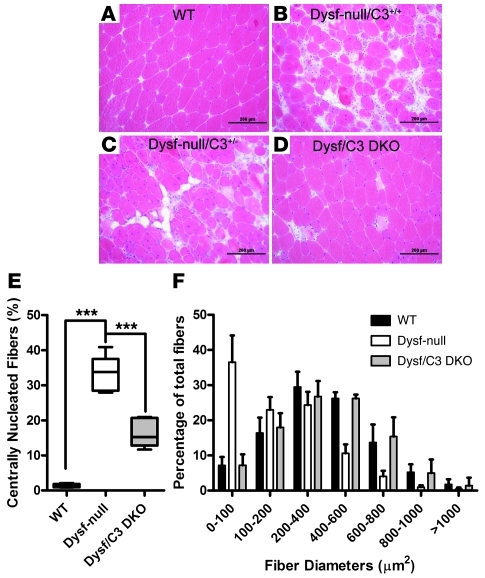
Since contractile dysfunction could hardly be discerned in EDL muscles of dysferlin-null mice even at 8 months of age (Figure 1B), we did not examine the effect of C3 ablation on the force development of EDL muscles. To assess the effect of C3 ablation on muscle pathology in dysferlin-null mice, we compared the quadriceps muscle pathology among the mice with different genotypes using histological analysis. By 5 months, the quadriceps muscles from female dysferlin-null mice developed an overt dystrophic phenotype including central nucleation, fiber size variation, hypertrophic myofibers, fibrosis, and fat replacement (Figure 7A). Deletion of the C3 gene decelerated this process significantly and maintained relatively normal muscle histology (Figure 7C). Disruption of only 1 copy of the C3 gene was not sufficient to alleviate the dystrophic features in dysferlin-null mice (Figure 7B). To more carefully analyze the phenotype of dysferlin/C3 double-mutant mice, we quantified the myofiber areas and the percentage of central nucleation of myofibers. Quadriceps muscle from dysferlin-null mice showed the characteristic increase in central nucleation of myofibers owing to ongoing degeneration and regeneration, and this degeneration/regeneration cycle was significantly reduced in the double-mutant mice (Figure 7D). Compared with the muscles from dysferlin-null mice, the muscles from dysferlin/C3 double-mutant mice showed a more uniform profile of myofiber areas, fewer smaller fibers, and a greater number of larger fibers than in WT mice (Figure 7E). To determine whether the effect of C3 ablation is common across other dystrophic models, we also crossed the C3-null mice with mdx mice to generate dystrophin/C3 double-mutant mice (mdx/C3-null) and studied the muscle pathology of the breeding progeny. Disruption of dystrophin and C3 expression in the mdx/C3-null mice was confirmed by Western blotting analysis of total muscle extracts (Supplemental Figure 3). Quadriceps muscle pathology among the progeny was similarly analyzed, as mentioned above. No significant improvements in muscle pathology were detected in mdx/C3-null mice when compared with mdx mice (Supplemental Figure 4).
Figure 7. Amelioration of muscle pathology in dysferlin-deficient mice by genetic ablation of complement factor C3.
(A–D) Representative H&E-stained histology of quadriceps muscles from WT (A), dysferlin-null/C3+/+ (B), dysferlin-null/C3+/– (C), and dysferlin/C3 double-null littermates (D) at 23 weeks of age. Scale bars: 200 μm. (E) Percentage of fibers containing central nuclei in the quadriceps muscles of 23-week-old WT, dysferlin-null, and dysferlin/C3 double-null mice (n = 5 for each group). (F) Fiber areas (grouped into size ranges) from quadriceps muscles of 23-week-old WT, dysferlin-null, and dysferlin/C3 double-null mice (n = 5 for each group). ***P < 0.01. Data are expressed as mean ± SEM.
The pathological effects of complement activation are mediated directly by MAC and indirectly by C3a and C5a factors that stimulate a range of proinflammatory responses from mast cells and leukocytes. To further determine whether MAC formation is the cause of muscle damage in dysferlinopathy, we generated dysferlin/C5 double-mutant mice. Histological examination of quadriceps muscle from the double-mutant mice did not reveal remarkable improvement in dystrophic features (Supplemental Figure 5). Quantitative analysis showed the same size distribution pattern and similar percentage of central nucleated myofibers in dysferlin/C5 double-mutant mice as in dysferlin single-mutant mice (Supplemental Figure 5). These data suggest that the terminal activation of complement has minimal effect on disease progression in dysferlinopathy.
Discussion
Collectively, our data show that loss of dysferlin in skeletal muscle induces the expression of complement factors, with the largest expression changes observed in the initiating factors C1q in the classical pathway and the alternative pathway factor CFB. Furthermore, disruption of the complement system mitigates muscle pathology in dysferlin-deficient mice. The complement system not only plays an important role in the immune defense system, but also contributes to the amplification of inflammation, if activated in excess, or is inappropriately controlled. Activation of the complement system causes tissue injury in animal models of autoimmune diseases, such as glomerulonephritis, hemolytic anemia, myasthenia gravis, and in 2 nonimmunologically mediated forms of primary tissue damage, burn, and ischemia (reviewed in ref. 45). In the present study, we showed that complement activation contributes to muscle injury in dysferlinopathy. Disruption of the central component (C3) of the complement system attenuates the pathological alterations in dysferlin-deficient skeletal muscle, but genetic ablation of the terminal component (C5) of the complement system has minimal effect on muscle pathology in dysferlin-deficient mice. These results suggest that it is not the terminal activation of the complement system but the activation of C3 that accelerates muscle injury in dysferlin-null mice.
Dysferlin has been shown to play roles in various cellular activities of muscle including membrane repair (5, 11–15), muscle differentiation (46), muscle regeneration, and cytokine release (18), all of which may rely on the function of dysferlin in promoting membrane fusion. It is still not clear why dysferlinopathy shows such a broad clinical spectrum and such extensive muscle inflammation. The age of onset of dysferlinopathy varies widely, from 12 to 59 years (47). Recently, an unusual case was reported in which a patient with complete loss of dysferlin showed the first symptoms at the age of 73 years and remained fully ambulant even at 85 years old (48). These observations are in contrast to a mean age of 34 years at the first use of wheelchair and nearly 30% of dysferlinopathy patients being wheelchair dependent after 15 years of disease diagnosis (25). Our study identified the complement immune system as an important factor affecting the progression of muscle pathology in dysferlinopathy. This finding also provides important insights into the design of novel therapeutic strategies for these patients.
It is not known how complement is activated in dysferlinopathy. At least 3 potential mechanisms are involved (Supplemental Figure 6). First, damaged cells release a number of constitutively expressed proteins, such as heat shock proteins (49), the chromatin HMGB1, and mitochondrial peptides bearing the N-formal group that are characteristic of prokaryotic proteins (50), all of which have been shown to activate the complement system. The heat shock proteins can activate the complement system in both an antibody-dependent and -independent manner, and this occurs in the absence of pathogens (51). HMGB1, which is secreted by activated monocytes and macrophages and passively released by necrotic or damaged cells, also potently induces complement activation and inflammation (50). Therefore, even aseptic tissue injury activates the complement cascade. The loss of dysferlin-mediated membrane repair would be expected to lead to a continuous leakage of cytosolic contents following any incurred membrane damage, and this in turn would result in continuous complement activation. In this scenario, the complement pathway would function as an amplifier of muscle membrane damage that occurs due to the absence of dysferlin. Second, failure of the release of cytokines from damaged muscle in dysferlinopathy (18) may account for the impairment of inflammatory cell recruitment at an early stage of muscle regeneration, with subsequent incomplete muscle remodeling, resulting in abnormal activation of the complement system. Finally, both dysferlin (11, 14) and synaptotagmin VII (52, 53) have been reported to play a role in membrane repair. Synaptotagmin VII primarily mediates the fusion of lysosomes with plasma membrane (45), while the vesicle compartments utilized by dysferlin-mediated membrane repair have not as yet been identified. It is likely that both dysferlin-mediated membrane repair and synaptotagmin VII–mediated membrane repair play roles in normal skeletal muscle maintenance, as genetic disruption of either dysferlin or synaptotagmin VII leads to the development of myopathies in mice (11, 52). In the absence of dysferlin, skeletal muscle may thus primarily utilize the synaptotagmin VII–mediated membrane repair pathway with lysosomes to reseal membrane damage. A recent study (54) showed that persistent fusion of lysosomes with plasma membrane during the suicidal membrane repair prior to apoptosis causes the externalization of phosphatidylserine (PS) on the outer leaflet of the cell membrane. The PS exposure induces inflammatory responses that preclude the development of tumor- and patient-specific immune responses during chemotherapy and radiotherapy. Interestingly, C1q, the recognition unit of the C1 complex of complement, was shown to bind PS and likely acts as a multi-ligand–bridging molecule in apoptotic cell recognition (55). Therefore, it is likely that persistent utilization of lysosomes for membrane repair in dysferlin-deficient skeletal muscle induces PS externalization that activates complement pathways through binding C1q and triggers muscle damage.
Methods
For details of mice, antibodies, reagents, and analysis, see Supplemental Methods.
Mouse models.
Mice (dysferlin-null, SJL/J, mdx, dysferlin/C3-null, dysferlin/C5-null, mdx/C3-null, and WT littermate control mice) were maintained at The University of Iowa Animal Care Unit in accordance with animal use guidelines. All animal studies were authorized by the Animal Care, Use, and Review Committee of The University of Iowa.
Measurement of contractile properties.
Muscle mass, fiber length, and maximum force were measured on 8 EDL muscles from 2- and 8-month-old WT and dysferlin-null mice (n = 4 for each group). Total cross-sectional area (CSA, cm2) and specific force Po (kN/m2) were determined (28, 44). The susceptibility of muscles to contraction-induced injury was assessed by 7 LCs with a strain of 30% of fiber length (30, 49). The differences between the experimental and WT samples were assessed by a 2-tailed Student’s t test, with the assumption of 2-sample equal variance.
Treadmill exercise and activity monitoring.
Animals were mildly exercised using an adjustable variable speed belt treadmill from AccuPacer. Activity based on ambulatory behavior was assessed in an open-field test as described in Supplemental Methods.
H&E staining and immunofluorescence analysis.
Seven-micron muscle cryosections were prepared from quadriceps and iliopsoas muscles of the aforementioned mice. Routine H&E staining and immunofluorescence staining were used to examine the muscle histopathology. The areas of muscle fibers and the percentage of central nucleated muscle fibers were calculated using Image-Pro Plus 6.
Statistics.
Unless otherwise stated, the data were calculated according to an analysis of variance and expressed as mean ± SEM. Where appropriate, the significance of differences between multiple genetically defined mouse models and their WT counterparts was assessed using 1-way ANOVA with Bonferroni’s post-tests, and the significance of differences between 2 experimental groups was assessed by unpaired 2-tailed Student’s t test. P < 0.05 was accepted as significant.
Supplementary Material
Acknowledgments
We thank Keith Garringer, Sally Prouty, Taylor Peterson, and Robert Crawford for technical support and all members of the Campbell laboratory for fruitful discussions. This work was supported in part by an American Heart Association Scientist Development grant (10SDG4140138 to R. Han), a Muscular Dystrophy Association Research grant (MDA171667 to R. Han), a Paul D. Wellstone Muscular Dystrophy Cooperative Research Center grant (1U54NS053672 to K.P. Campbell), a Muscular Dystrophy Association grant (MDA3936, to K.P. Campbell), and a U.S. Department of Defense grant (W81XWH-05-1-0079). We also thank the University of Iowa Roy J. and Lucille A. Carver College of Medicine, the Senator Paul D. Wellstone Muscular Dystrophy Cooperative Research Center, and the University of Iowa Central Microscopy Research Facility. Transgenic mice were generated at the University of Iowa Transgenic Animal Facility directed by Curt D. Sigmund, and this facility was supported in part by grants from the NIH and from the Roy J. and Lucille A. Carver College of Medicine. We wish to thank Norma Sinclair, Patricia Yarolem, and Joanne Schwarting for their technical expertise in generating transgenic mice. K.P. Campbell is an investigator of the Howard Hughes Medical Institute. J.D. Lueck is supported by a Myotonic Dystrophy Foundation postdoctoral fellowship (MDF-FF-2009-0002). J.R. Levy is supported by NIH grant T32HL007121 through NHLBI. E.P. Rader is supported by a Muscular Dystrophy Association development grant (award ID 67814).
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2010;120(12):4366–4374. doi:10.1172/JCI42390.
References
- 1.Bashir R, et al. A gene related to Caenorhabditis elegans spermatogenesis factor fer-1 is mutated in limb-girdle muscular dystrophy type 2B. Nat Genet. 1998;20(1):37–42. doi: 10.1038/1689. [DOI] [PubMed] [Google Scholar]
- 2.Liu J, et al. Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nat Genet. 1998;20(1):31–36. doi: 10.1038/1682. [DOI] [PubMed] [Google Scholar]
- 3.Illa I, et al. Distal anterior compartment myopathy: a dysferlin mutation causing a new muscular dystrophy phenotype. Ann Neurol. 2001;49(1):130–134. doi: 10.1002/1531-8249(200101)49:1<130::AID-ANA22>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- 4.Kuru S, et al. A patient with limb girdle muscular dystrophy type 2B (LGMD2B) manifesting cardiomyopathy [in Japanese]. Rinsho Shinkeigaku. 2004;44(6):375–378. [PubMed] [Google Scholar]
- 5.Han R, et al. Dysferlin-mediated membrane repair protects the heart from stress-induced left ventricular injury. J Clin Invest. 2007;117(7):1805–1813. doi: 10.1172/JCI30848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chase TH, Cox GA, Burzenski L, Foreman O, Shultz LD. Dysferlin deficiency and the development of cardiomyopathy in a mouse model of limb-girdle muscular dystrophy 2B. Am J Pathol. 2009;175(6):2299–2308. doi: 10.2353/ajpath.2009.080930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anderson LV, et al. Dysferlin is a plasma membrane protein and is expressed early in human development. . Hum Mol Genet. 1999;8(5):855–861. doi: 10.1093/hmg/8.5.855. [DOI] [PubMed] [Google Scholar]
- 8.Ho M, Gallardo E, McKenna-Yasek D, De Luna N, Illa I, Brown RH., Jr A novel, blood-based diagnostic assay for limb girdle muscular dystrophy 2B and Miyoshi myopathy. Ann Neurol. 2002;51(1):129–133. doi: 10.1002/ana.10080. [DOI] [PubMed] [Google Scholar]
- 9.Jethwaney D, et al. Proteomic analysis of plasma membrane and secretory vesicles from human neutrophils. Proteome Sci. 2007;5:12. doi: 10.1186/1477-5956-5-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Luna N, et al. Dysferlin expression in monocytes: a source of mRNA for mutation analysis. Neuromuscul Disord. 2007;17(1):69–76. doi: 10.1016/j.nmd.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 11.Bansal D, et al. Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature. 2003;423(6936):168–172. doi: 10.1038/nature01573. [DOI] [PubMed] [Google Scholar]
- 12.Cenacchi G, Fanin M, De Giorgi LB, Angelini C. Ultrastructural changes in dysferlinopathy support defective membrane repair mechanism. J Clin Pathol. 2005;58(2):190–195. doi: 10.1136/jcp.2004.018978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cai C, et al. Membrane repair defects in muscular dystrophy are linked to altered interaction between MG53, caveolin-3, and dysferlin. J Biol Chem. 2009;284(23):15894–15902. doi: 10.1074/jbc.M109.009589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lennon NJ, Kho A, Bacskai BJ, Perlmutter SL, Hyman BT, Brown RH., Jr Dysferlin interacts with annexins A1 and A2 and mediates sarcolemmal wound-healing. . J Biol Chem. 2003;278(50):50466–50473. doi: 10.1074/jbc.M307247200. [DOI] [PubMed] [Google Scholar]
- 15.Hino M, et al. Insufficient membrane fusion in dysferlin-deficient muscle fibers after heavy-ion irradiation. Cell Struct Funct. 2009;34(1):11–15. doi: 10.1247/csf.08033. [DOI] [PubMed] [Google Scholar]
- 16.Klinge L, et al. New aspects on patients affected by dysferlin deficient muscular dystrophy. J Neurol Neurosurg Psychiatry. 2010;81(9):946–953. doi: 10.1136/jnnp.2009.178038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mahjneh I, Marconi G, Bushby K, Anderson LV, Tolvanen-Mahjneh H, Somer H. Dysferlinopathy (LGMD2B): a 23-year follow-up study of 10 patients homozygous for the same frameshifting dysferlin mutations. Neuromuscul Disord. 2001;11(1):20–26. doi: 10.1016/S0960-8966(00)00157-7. [DOI] [PubMed] [Google Scholar]
- 18.Chiu YH, et al. Attenuated muscle regeneration is a key factor in dysferlin-deficient muscular dystrophy. . Hum Mol Genet. 2009;18(11):1976–1989. doi: 10.1093/hmg/ddp121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Roche JA, Lovering RM, Roche R, Ru LW, Reed PW, Bloch RJ. Extensive mononuclear infiltration and myogenesis characterize the recovery of dysferlin-null skeletal muscle from contraction-induced injuries. Am J Physiol Cell Physiol. 2010;298(2):C298–C312. doi: 10.1152/ajpcell.00122.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Roche JA, Lovering RM, Bloch RJ. Impaired recovery of dysferlin-null skeletal muscle after contraction-induced injury in vivo. Neuroreport. 2008;19(16):1579–1584. doi: 10.1097/WNR.0b013e328311ca35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gallardo E, Rojas-Garcia R, de Luna N, Pou A, Brown RH, Jr, Illa I. Inflammation in dysferlin myopathy: immunohistochemical characterization of 13 patients. Neurology. 2001;57(11):2136–2138. doi: 10.1212/wnl.57.11.2136. [DOI] [PubMed] [Google Scholar]
- 22.Fanin M, Angelini C. Muscle pathology in dysferlin deficiency. Neuropathol Appl Neurobiol. 2002;28(6):461–470. doi: 10.1046/j.1365-2990.2002.00417.x. [DOI] [PubMed] [Google Scholar]
- 23.Confalonieri P, et al. Muscle inflammation and MHC class I up-regulation in muscular dystrophy with lack of dysferlin: an immunopathological study. J Neuroimmunol. 2003;142(1–2):130–136. doi: 10.1016/S0165-5728(03)00255-8. [DOI] [PubMed] [Google Scholar]
- 24.Selva-O’Callaghan A, Labrador-Horrillo M, Gallardo E, Herruzo A, Grau-Junyent JM, Vilardell-Tarres M. Muscle inflammation, autoimmune Addison’s disease and sarcoidosis in a patient with dysferlin deficiency. . Neuromuscul Disord. 2006;16(3):208–209. doi: 10.1016/j.nmd.2006.01.005. [DOI] [PubMed] [Google Scholar]
- 25.Nguyen K, et al. Phenotypic study in 40 patients with dysferlin gene mutations: high frequency of atypical phenotypes. Arch Neurol. 2007;64(8):1176–1182. doi: 10.1001/archneur.64.8.1176. [DOI] [PubMed] [Google Scholar]
- 26.Nagaraju K, et al. Dysferlin deficiency enhances monocyte phagocytosis: a model for the inflammatory onset of limb-girdle muscular dystrophy 2B. . Am J Pathol. 2008;172(3):774–785. doi: 10.2353/ajpath.2008.070327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rawat R, Cohen TV, Ampong B, Francia D, Henriques-Pons A, Hoffman EP, Nagaraju K. Inflammasome up-regulation and activation in dysferlin-deficient skeletal muscle. Am J Pathol. 2010;176(6):2891–2900. doi: 10.2353/ajpath.2010.090058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brooks SV, Faulkner JA. Contractile properties of skeletal muscles from young, adult and aged mice. J Physiol. 1988;404:71–82. doi: 10.1113/jphysiol.1988.sp017279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.DelloRusso C, Crawford RW, Chamberlain JS, Brooks SV. Tibialis anterior muscles in mdx mice are highly susceptible to contraction-induced injury. . J Muscle Res Cell Motil. 2001;22(5):467–475. doi: 10.1023/A:1014587918367. [DOI] [PubMed] [Google Scholar]
- 30.Kobayashi YM, et al. Sarcolemma-localized nNOS is required to maintain activity after mild exercise. Nature. 2008;456(7221):511–515. doi: 10.1038/nature07414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sicinski P, Geng Y, Ryder-Cook AS, Barnard EA, Darlison MG, Barnard PJ. The molecular basis of muscular dystrophy in the mdx mouse: a point mutation. Science. 1989;244(4912):1578–1580. doi: 10.1126/science.2662404. [DOI] [PubMed] [Google Scholar]
- 32.Bittner RE, et al. Dysferlin deletion in SJL mice (SJL-Dysf) defines a natural model for limb girdle muscular dystrophy 2B. Nat Genet. 1999;23(2):141–142. doi: 10.1038/13770. [DOI] [PubMed] [Google Scholar]
- 33.Nemoto H, Konno S, Nakazora H, Miura H, Kurihara T. Histological and immunohistological changes of the skeletal muscles in older SJL/J mice. Eur Neurol. 2007;57(1):19–25. doi: 10.1159/000097005. [DOI] [PubMed] [Google Scholar]
- 34.Rosenberg NL, Ringel SP, Kotzin BL. Experimental autoimmune myositis in SJL/J mice. Clin Exp Immunol. 1987;68(1):117–129. [PMC free article] [PubMed] [Google Scholar]
- 35.Bernard CC, Carnegie PR. Experimental autoimmune encephalomyelitis in mice: immunologic response to mouse spinal cord and myelin basic proteins. J Immunol. 1975;114(5):1537–1540. [PubMed] [Google Scholar]
- 36.Hochmeister S, Bittner RE, Hoger H, Lassmann H, Bradl M. The susceptibility to experimental autoimmune encephalomyelitis is not related to dysferlin-deficiency. Autoimmunity. 2009;42(3):235–241. doi: 10.1080/08916930802716542. [DOI] [PubMed] [Google Scholar]
- 37.Campanaro S, et al. Gene expression profiling in dysferlinopathies using a dedicated muscle microarray. Hum Mol Genet. 2002;11(26):3283–3298. doi: 10.1093/hmg/11.26.3283. [DOI] [PubMed] [Google Scholar]
- 38.Suzuki N, et al. Expression profiling with progression of dystrophic change in dysferlin-deficient mice (SJL). Neurosci Res. 2005;52(1):47–60. doi: 10.1016/j.neures.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 39.von der Hagen M, et al. The differential gene expression profiles of proximal and distal muscle groups are altered in pre-pathological dysferlin-deficient mice. Neuromuscul Disord. 2005;15(12):863–877. doi: 10.1016/j.nmd.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 40.Wenzel K, et al. Increased susceptibility to complement attack due to down-regulation of decay-accelerating factor/CD55 in dysferlin-deficient muscular dystrophy. J Immunol. 2005;175(9):6219–6225. doi: 10.4049/jimmunol.175.9.6219. [DOI] [PubMed] [Google Scholar]
- 41.Feuk-Lagerstedt E, Movitz C, Pellme S, Dahlgren C, Karlsson A. Lipid raft proteome of the human neutrophil azurophil granule. Proteomics. 2007;7(2):194–205. doi: 10.1002/pmic.200600482. [DOI] [PubMed] [Google Scholar]
- 42.Millay DP, et al. Genetic manipulation of dysferlin expression in skeletal muscle: novel insights into muscular dystrophy. Am J Pathol. 2009;175(5):1817–1823. doi: 10.2353/ajpath.2009.090107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wessels MR, Butko P, Ma M, Warren HB, Lage AL, Carroll MC. Studies of group B streptococcal infection in mice deficient in complement component C3 or C4 demonstrate an essential role for complement in both innate and acquired immunity. Proc Natl Acad Sci U S A. 1995;92(25):11490–11494. doi: 10.1073/pnas.92.25.11490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Han R, et al. Basal lamina strengthens cell membrane integrity via the laminin G domain-binding motif of alpha-dystroglycan. Proc Natl Acad Sci U S A. 2009;106(31):12573–12579. doi: 10.1073/pnas.0906545106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Morgan BP. Complement membrane attack on nucleated cells: resistance, recovery and non-lethal effects. Biochem J. 1989;264(1):1–14. doi: 10.1042/bj2640001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.de Luna N, et al. Absence of dysferlin alters myogenin expression and delays human muscle differentiation “in vitro”. J Biol Chem. 2006;281(25):17092–17098. doi: 10.1074/jbc.M601885200. [DOI] [PubMed] [Google Scholar]
- 47.Ueyama H, Kumamoto T, Horinouchi H, Fujimoto S, Aono H, Tsuda T. Clinical heterogeneity in dysferlinopathy. Intern Med. 2002;41(7):532–536. doi: 10.2169/internalmedicine.41.532. [DOI] [PubMed] [Google Scholar]
- 48.Klinge L, et al. Late onset in dysferlinopathy widens the clinical spectrum. Neuromuscul Disord. 2008;18(4):288–290. doi: 10.1016/j.nmd.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 49.Basu S, Binder RJ, Suto R, Anderson KM, Srivastava PK. Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-kappa B pathway. Int Immunol. 2000;12(11):1539–1546. doi: 10.1093/intimm/12.11.1539. [DOI] [PubMed] [Google Scholar]
- 50.Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418(6894):191–195. doi: 10.1038/nature00858. [DOI] [PubMed] [Google Scholar]
- 51.Prohaszka Z, et al. Heat shock protein 70 is a potent activator of the human complement system. Cell Stress Chaperones. 2002;7(1):17–22. doi: 10.1379/1466-1268(2002)007<0017:HSPIAP>2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chakrabarti S, et al. Impaired membrane resealing and autoimmune myositis in synaptotagmin VII-deficient mice. J Cell Biol. 2003;162(4):543–549. doi: 10.1083/jcb.200305131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Reddy A, Caler EV, Andrews NW. Plasma membrane repair is mediated by Ca(2+)-regulated exocytosis of lysosomes. Cell. 2001;106(2):157–169. doi: 10.1016/S0092-8674(01)00421-4. [DOI] [PubMed] [Google Scholar]
- 54.Mirnikjoo B, Balasubramanian K, Schroit AJ. Suicidal Membrane Repair Regulates Phosphatidylserine Externalization during Apoptosis. J Biol Chem. 2009;284(34):22512–22516. doi: 10.1074/jbc.C109.022913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Paidassi H, et al. C1q binds phosphatidylserine and likely acts as a multiligand-bridging molecule in apoptotic cell recognition. J Immunol. 2008;180(4):2329–2338. doi: 10.4049/jimmunol.180.4.2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.