Abstract
Atrial fibrillation (AF), which is the main cause of stroke, is the most common sustained arrhythmia afflicting about 2.3 million Americans. Clinical treatment and management of AF would benefit from a non-invasive and global assessment of the arrhythmia; however that avenue seems currently limited in part by our poor understanding of the arrhythmia itself. Experimental studies of AF in the isolated sheep heart demonstrated that high-frequency sources in the posterior wall of the left atrium drive the fibrillatory activity throughout both atria. Motivated by those results and by a growing body of work investigating how measurements of the cycle length of activity in patients during AF can contribute to its treatment, we focused our analysis on the dispersion of dominant frequency (DF) of the activity during AF in humans. Using electroanatomic mapping and Fourier methods we generated three-dimensional, intra-cardiac DF maps of the atria in patients prior to AF ablation procedures and identified relatively small high DF (HDF) sites. In patients with paroxysmal AF, the HDF sites are often localized to the posterior left atrium near the ostia of the pulmonary veins. In contrast, patients with permanent AF demonstrate HDF sites that are more often localized to either atria than the posterior left atrium-PV junction. In our study ablation at HDF sites resulted in significant slowing of the arrhythmia and termination of sustained AF in 87% of patients with paroxysmal AF. Furthermore, we found that abolishing by ablation a preexisting left atrium to right atrium DF gradients predicted long term freedom of AF in both paroxysmal and persistent AF patients. Overall, the analysis of intracardiac electrical recordings in the frequency domain has greatly enhanced our understanding of its underlying mechanisms and may contribute to monitoring drug effects and guide ablation procedures aiming at its termination. On the other hand, current body surface mapping methods have also suggested better correlations between surface AF frequency and intracardiac local DFs as compared to spatiotemporal activation patterns. Therefore, a further study of the correlation of spectral observables obtained from the atria and from the surface ECG during AF seems to have the potential to advance our ability to diagnose and discern mechanisms of AF non-invasively.
Introduction
About 2.3 million people in the United States are currently afflicted with Atrial Fibrillation (AF), the most common sustained cardiac arrhythmia in humans, and many more cases are predicted in the near future.1 Antiarrhythmic drugs are only partially effective in treating AF, and have the potential for serious side effects, including life-threatening pro-arrhythmia. On the other hand, it has recently been demonstrated that paroxysmal AF in patients is initiated by focal triggers localized usually to one of the pulmonary veins (PVs)2 and can be cured by a catheter-based ablation procedure.3 However, in persistent AF, the prevailing theory is that multiple random wavelets of activation coexist to create a chaotic cardiac rhythm,4 and therapy is more challenging.5 Current studies of AF in humans rely in large on intracardiac electrical mapping to provide excitation data in the patients during the ablation procedure and are mostly consisting of sequential local, or across small regions, recordings limited to the time of the procedure. Arguably, the short and long term study and management of AF would benefit from a non-invasive and global assessment of the arrhythmia, as proposed by many investigators of the surface ECG,6,7 however that avenue seems currently limited in part by our poor understanding of the arrhythmia itself.
To enhance our understanding of AF early studies on regional differences in activation cycle length during AF8,9 were followed by an increased interest in the analysis of the frequency spectrum as a measure of the activation rate in animal models.10–13 Furthermore, works demonstrating how measurements of AF cycle length in patients can contribute to its treatment,14,15 motivated my collaborators and me to translate experimental knowledge and methods into analysis of human AF in the frequency domain.16 As suggested by our recent studies,16–18 high-resolution analysis of the Fourier power spectrum with its dominant frequency (DF) offers the unique opportunity of being able to correlate systematically the spatial distribution of excitation frequency with cardiac anatomy and ablation procedures and to provide mechanistic insight into different types of AF. Thus, the central goal of this paper is to provide an intracardiac perspective into the continuing efforts toward the development of surface ECG tools based on our current understanding of the spatiotemporal and frequency patterns of atrial excitation waves during AF in animals models and humans.
Activity in the left and right atria during AF in the sheep model
The notion that the spatio-temporal complexity of wave propagation during AF reflects in the local activation rate dispersion led us to determine the DF throughout both left and right atria (LA and RA, respectively) during acute fibrillation in isolated sheep hearts; high resolution optical mapping revealed them to be organized in discrete domains.11 Most notably, during the arrhythmia the activation frequencies in certain areas of the LA were consistently faster than any other areas.10–12 In search for the mechanism underlying such frequency distribution in space, a subsequent study demonstrated the manner in which interatrial pathways mediate fibrillatory conduction and the establishment of frequency gradients between the LA and RA13 along Bachmann’s bundle (BB) and the inferoposterior interatrial pathway (ipp) that underlies the coronary sinus, both of which are well-known routes of interatrial electrical communication.19,20 Propagation along these pathways was organized and occurred from left to right along the decrease in DF values. Quantification of this finding revealed that wave fronts propagated from left to right in 81% and 80% of the analyzed activations along BB and ipp, respectively. Conversely, right-to-left propagation occurred in a significantly smaller percentage of cases.13 As suggested by other studies, repetitive and fast activation may also originate from the septum21 or the pulmonary veins (PVs).2,21 Altogether, the data support the contention that AF results from rapidly successive wave fronts emanating from fast sources localized in the LA.10,12,13,22 The wave fronts propagating through both atria interact with anatomic and/or functional obstacles, leading to fragmentation and wavelet formation.23,24
The spatial distribution of DFs during AF in patients
Several recent studies have characterized the spatial distribution of DF during AF in patients.17,25,26 Using the CARTO electroanatomic mapping system, Sanders et al17 sequentially acquired 5-sec long endocardial bipolar recordings from about 120 points throughout both atria and the coronary sinus (CS) in 32 patients during sustained AF. The recorded signals were rectified27 and 3–15 Hz bandpass filtered. Then FFT was used to determine the DF of each of 120 segments lasting 4.096 sec with a resolution of 0.24 Hz.28
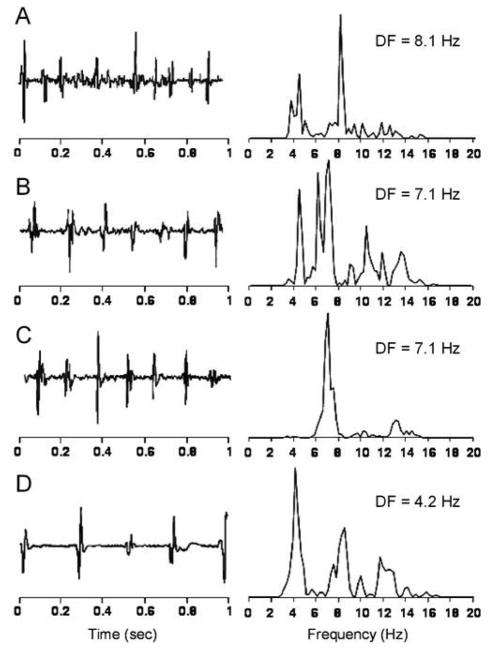
Figure 1 shows four sample bipolar electrograms with their respective power spectra acquired from endocardial sites in the LA and RA of a patient with paroxysmal AF of spontaneous onset.17 On the left are typical AF recordings with variable amplitudes and inter-beat intervals, which do not enable accurate cycle length analysis, particularly in the top recording. However, the spectra on the right show relatively narrow bands in the 3–15 Hz range with distinct peaks of DFs approximately corresponding to the activation rate and the inverse of the average cycle length in the electrograms28,29
Figure 1.
Bipolar electrograms and corresponding power spectra obtained from 4 points in a patient with spontaneous paroxysmal AF. Each site shows distinct DF. Panel A clearly demonstrates the utility of spectral analysis. The bipolar recording shows low amplitude complex signals that make accurate determination of frequency of activation difficult. The power spectrum clearly demonstrates the DF at 8.1 Hz. The DF map of this patient is shown in Figure 2A. Panel A is from the site of maximal DF at the RIPV; Panels B and C are from other HDF sites in the right superior and left superior PVs; and Panel D is from the posterior RA. Reproduced from Sanders et al, Circulation. 2005;112:789–797.17
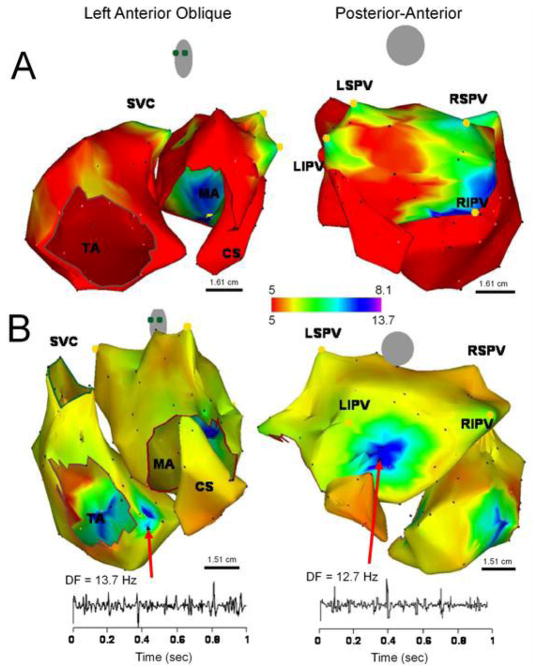
Electrograms that were collected from multiple sites in patients during AF had their corresponding DFs superimposed on the atrial geometry to generate color DF maps,17 as illustrated in Figure 2. On each DF map, those sites that demonstrated frequency activity 20% or higher than the surrounding atrial tissue were defined as high DF (HDF) sites. In this study most HDF sites involved a single point, but in some the HDF site extended over 2–3 adjacent points. Figure 2A reproduces left anterior oblique (left) and postero-anterior (right) views17 of the same map showing fibrillatory activity at mean DF of ~4.8 Hz in the paroxysmal AF patient whose electrograms are shown in Figure 1. While frequency in the majority of the atria and CS was relatively slow (≤5 Hz), the posterior wall of the LA was activated at faster rates (7–8 Hz), with notable HDF sites at each of the PVs. In this patient, focal radiofrequency ablation applied to the HDF site near the right inferior PV (RIPV) effectively terminated AF. Figure 2B shows a DF map from another patient with permanent AF. Compared with the patient with paroxysmal AF, this patient not only had a higher frequency at the maximal HDF site (13.7 Hz), but also both atria demonstrated higher global frequency of activity and in addition, many of the HDF sites were located in the atria rather in the PV region.17,18 It has been concluded that in patients, paroxysmal AF is characterized by the hierarchical spatial distribution of DFs where the LA and PVs were always the fastest regions. By contrast, in persistent AF, a more uniform distribution of higher DF values was observed, where the highest DFs could not be found primarily in the PV region.17,18,26,30
Figure 2.
Dominant frequency maps of human AF. A, DF map in a patient with paroxysmal AF (6 hours). Note HDF sites in each of the PVs. Ablation sequence in this patient was LSPV, LIPV RSPV and RIPV (site of AF termination); AF CL increased by 10 ms, 25 ms, 9 ms and 75 ms, respectively, before termination. B, DF map in a patient with permanent AF (24 months). The maximal DF and atrial frequency are higher than the patient in panel A. In addition, many of the HDF sites are located outside the PVs. Ablation sequence in this patient was RIPV, RSPV, LSPV and LIPV; AFCL increased by 5 ms, 2 ms, 0 ms and 5 ms respectively. The electrogram of the site of maximal DF in the LA and RA is presented showing significant fractionation. LSPV, left superior PV; LIPV, left inferior PV; RSPV, right superior PV; RIPV, right inferior PV; SVC, superior vena cava; MA, mitral annulus; TA, tricuspid annulus. Color-bar, scale in Hz. Reproduced from Sanders et al, Circulation. 2005;112:789–797.17
The high DF sites and maintenance of AF
AF mapping studies have recognized the presence of temporally and spatially periodic activity12,31–33 emanating from the PV region with regularity,10 suggesting that these structures may have a role in maintaining AF,12 by harboring either localized short cycle length reentrant sources and/or focal automatic activity.22,34 Sequential ablation of sites showing the shortest cycle length has been associated with a progressive slowing of AF frequency culminating in termination in 75% of patients with paroxysmal AF.15
Using a blind correlation between atrial DF distribution and ablation, without any attempt at identifying potentially arrhythmogenic sites at the time of the procedure, Sanders et al17 found that ablation at PVs harboring HDF sites resulted in an increase in AF CL (≥5 ms) within the CS in 89% of cases. The latter was true in patients with either paroxysmal or permanent AF. However, eventual arrhythmia termination occurred during ablation in 15 (88%) out of 17 patients with paroxysmal but none with permanent AF (p<0.0001). In 13 (87%) of the 15 paroxysmal AF patients arrhythmia termination was associated with ablation at a HDF site; 11 localized to a PV and 2 to the LA roof and the fossa ovalis. A more recent study by Atienza et al35 using a real-time mapping of DF allows targeting specifically HDF sites and further demonstrates that abolishing by ablation a preexisting LA-to-RA DF gradients predicted long term freedom of AF in both paroxysmal and persistent AF patients. In persistent AF patients, Yoshida et al36 recently demonstrated that isolation of the PV region eliminated high DF components and transform the fibrillation to tachycardia with a slower frequency. Interestingly, for reasons which are not entirely clear the frequency of the emerging tachycardia waves had a significantly high power during the preceding fibrillation.36 The aforementioned data, together with those of previous studies by Atienza et al18 and Lazar et al,37 clearly indicate that the HDF sites play a role in the maintenance of AF in a significant number of patients.
Activation frequency and driver mechanisms
Overall, it seems that the dynamic nature of AF keeps the spatio-temporal variability of the DF within a limit that allows consistent identification of HDF sites as discrete AF drivers despite mapping limitations. However, to this date, whether such sites are automatic, triggered or reentrant and whether changes in the driver activity would alter spatial frequency gradients, remains unresolved. Identifying rotors as drivers of AF directly in patients is very difficult with the existing mapping techniques.38,39 Yet, the ACh dose dependent acceleration of rotor frequency40 offered an idea that enabled translating animal experiments to the patients and gave us the opportunity to obtain evidence, albeit indirect, for the presence of rotors as drivers through pharmacologic means. Translation was made possible also by the fact that adenosine, which is widely used in the clinic, is known to activate the same Kir3.x subfamily of inward rectifier potassium channels as ACh.41–43 By increasing K+ conductance in the atrium, both ACh and adenosine hyperpolarize the cell membrane, abbreviate the action potential duration and the refractory period, and inhibit spontaneous pacemaker discharge as well as early and delayed depolarizations.41,42 On the other hand they both accelerate reentrant activity.40 In a recent study18 Atienza et al18 analyzed the effect of adenosine on the activation rate in specific regions at the junction of the PV and the LA (PV-LAJ), the roof of the RA and the CS. That study found that adenosine infusion increased frequency primarily at sites that activated at the highest rate at baseline. In paroxysmal AF patients, adenosine increased activation frequency in the PV–LAJ. In persistent AF patients, the highest-frequency sources accelerated by adenosine were located in either atria but not at PV sites. Thus, the response to adenosine is consistent with reentrant drivers maintaining AF that have different locations in paroxysmal compared with persistent AF patients.18
Surface ECG and frequency analysis
Intracardiac mapping and Fourier methods generated whole-atrial DF maps in patients undergoing AF ablation procedures and identified relatively small HDF sites. Surface ECG analysis that captures that information would potentially be relevant to the guidance into AF mechanisms as well as its pharmaceutical and ablative therapies. To what extent one should expect to be able to determine the distribution and role of the HDF sites in AF maintenance in population using surface ECG at large? To address that question, consideration should be given to the relatively low resolution and small fields of currently available mapping systems, with the substantial temporal limitation imposed by the need to sequentially acquire the electrograms to generate the wide area intracardiac DF maps. At the same time, a study using surface mapping with multiple electrodes across the body upper torso has described surface activity during atrial arrhythmias.44 Despite the smoothing effect of the torso and relatively low atrial signal, activation maps were constructed showing inter-patient variability but short term reproducibility during AF.44 The paucity of invasive data in that study nevertheless prevents a direct correlation of the activation maps with actual atrial sequences of excitation. Another approach used a high density surface mapping in combination with individual torso-heart reconstruction and the inverse solution to generate activation maps on the atrial epicardium.45 The method, named ECGi has been validated in a few cases of atrial flutter and tachycardia,46,47 Based on these studies it is not clear yet whether the body surface mapping will provide the details relevant to the complex spatiotemporal patterns of activation that underlie AF in patients.
On the other hand surface frequency analysis during AF found that surface frequency mapping is feasible with global ECG frequencies ranging between 4 and 8 Hz, values similar to average DF values of local activity obtained by intracardiac bipolars.17,25,35 Using the standard 12 lead ECG configuration, recent studies analyzed the vectorial representation of surface potentials and showed a correlation between its loop morphology and stability to the AF frequency as measured on the surface.48,49 Notably though is the fact taht the physiologic meaning of a single vector representation of the atrial activation during fibrillation remains to be studied. Indeed, while the degree of wavefront consistency provided insights into the temporal variability of the activation order, this aspect was not reflected by the body surface parameters in another study.50 However, the same study found that AF frequency on the surface was able to distinguish between recordings with different degrees of intra-atrial signal organization.50 Other studies also found high correlations between surface dominant frequency and intracardiac DFs at the left and right atria49 and the coronary sinus.51 Because the relationship between the spatiotemporal patterns of activation and the local DF during AF at the intracardiac level is not fully understood, it remains a challenge to utilize efficiently the surface ECG for a mechanistic AF diagnosis or management. Nevertheless, the fact that various studies linked the spectral content of the AF to either its underlying mechanisms17,18 or prediction of long term freedom of AF35,51 makes the frequency analysis of the surface ECG a rational approach to achieve that important goal.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Reference List
- 1.Chen LY, Shen WK. Epidemiology of atrial fibrillation: a current perspective. Heart Rhythm. 2007;4:S1–S6. doi: 10.1016/j.hrthm.2006.12.018. [DOI] [PubMed] [Google Scholar]
- 2.Haissaguerre M, Jais P, Shah DC, Takahashi A, Hocini M, Quiniou G, Garrigue S, Le Mouroux A, Le Metayer P, Clementy J. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998;339:659–666. doi: 10.1056/NEJM199809033391003. [DOI] [PubMed] [Google Scholar]
- 3.Haissaguerre M, Shah DC, Jais P, Hocini M, Yamane T, Deisenhofer I, Garrigue S, Clementy J. Mapping-guided ablation of pulmonary veins to cure atrial fibrillation. American Journal of Cardiology. 2000;86:9K–19K. doi: 10.1016/s0002-9149(00)01186-3. [DOI] [PubMed] [Google Scholar]
- 4.Moe GK, Abildskov JA. Atrial fibrillation as a self-sustaining arrhythmia independent of focal discharges. American Heart Journal. 1959;58:59–70. doi: 10.1016/0002-8703(59)90274-1. [DOI] [PubMed] [Google Scholar]
- 5.Oral H, Pappone C, Chugh A, Good E, Bogun F, Pelosi F, Jr, Bates ER, Lehmann MH, Vicedomini G, Augello G, Agricola E, Sala S, Santinelli V, Morady F. Circumferential pulmonary-vein ablation for chronic atrial fibrillation. N Engl J Med. 2006;354:934–941. doi: 10.1056/NEJMoa050955. [DOI] [PubMed] [Google Scholar]
- 6.Bollmann A, Husser D, Mainardi L, Lombardi F, Langley P, Murray A, Rieta JJ, Millet J, Olsson SB, Stridh M, Sornmo L. Analysis of surface electrocardiograms in atrial fibrillation: techniques, research, and clinical applications. Europace. 2006;8:911–926. doi: 10.1093/europace/eul113. [DOI] [PubMed] [Google Scholar]
- 7.Childers R. Electrophysiology of the electrocardiographic changes of atrial fibrillation. J Electrocardiol. 2006;39:S174–S179. doi: 10.1016/j.jelectrocard.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 8.Morillo CA, Klein GJ, Jones DL, Guiraudon CM. Chronic rapid atrial pacing: Structural, functional, and electrophysiological characteristics of a new model of sustained atrial fibrillation. Circulation. 1995;91:1588–1595. doi: 10.1161/01.cir.91.5.1588. [DOI] [PubMed] [Google Scholar]
- 9.Harada A, Sasaki K, Fukushima T, Ikeshita M, Asano T, Yamauchi S, Shoji T. Atrial activation during chronic atrial fibrillation in patients with isolated mitral valve disease. Ann Thorac Surg. 1996;61:104–112. doi: 10.1016/0003-4975(95)00824-1. [DOI] [PubMed] [Google Scholar]
- 10.Skanes AC, Mandapati R, Berenfeld O, Davidenko JM, Jalife J. Spatiotemporal periodicity during atrial fibrillation in the isolated sheep heart. Circulation. 1998;98:1236–1248. doi: 10.1161/01.cir.98.12.1236. [DOI] [PubMed] [Google Scholar]
- 11.Berenfeld O, Mandapati R, Dixit S, Skanes AC, Chen J, Mansour M, Jalife J. Spatially distributed dominant excitation frequencies reveal hidden organization in atrial fibrillation in the Langendorff-perfused sheep heart. J Cardiovasc Electrophysiol. 2000;11:869–879. doi: 10.1111/j.1540-8167.2000.tb00066.x. [DOI] [PubMed] [Google Scholar]
- 12.Mandapati R, Skanes A, Chen J, Berenfeld O, Jalife J. Stable microreentrant sources as a mechanism of atrial fibrillation in the isolated sheep heart. Circulation. 2000;101:194–199. doi: 10.1161/01.cir.101.2.194. [DOI] [PubMed] [Google Scholar]
- 13.Mansour M, Mandapati R, Berenfeld O, Chen J, Samie FH, Jalife J. Left-to-right gradient of atrial frequencies during acute atrial fibrillation in the isolated sheep heart. Circulation. 2001;103:2631–2636. doi: 10.1161/01.cir.103.21.2631. [DOI] [PubMed] [Google Scholar]
- 14.Pappone C, Rosanio S. Pulmonary Vein Isolation for Atrial Fibrillation. In: Zipes DP, Jalife J, editors. Cardiac Electrophysiology - From Cell to Bedside. Philadelphia: Saunders; 2004. pp. 1039–1052. [Google Scholar]
- 15.Haissaguerre M, Sanders P, Hocini M, Hsu LF, Shah DC, Scavee C, Takahashi Y, Rotter M, Pasquie JL, Garrigue S, Clementy J, Jais P. Changes in atrial fibrillation cycle length and inducibility during catheter ablation and their relation to outcome. Circulation. 2004;109:3007–3013. doi: 10.1161/01.CIR.0000130645.95357.97. [DOI] [PubMed] [Google Scholar]
- 16.Berenfeld O. Quantifying activation frequency in atrial fibrillation to establish underlying mechanisms and ablation guidance. Heart Rhythm. 2007;4:1225–1234. doi: 10.1016/j.hrthm.2007.05.004. [DOI] [PubMed] [Google Scholar]
- 17.Sanders P, Berenfeld O, Hocini M, Jais P, Vaidyanathan R, Hsu LF, Garrigue S, Takahashi Y, Rotter M, Sacher F, Scavee C, Ploutz-Snyder R, Jalife J, Haissaguerre M. Spectral analysis identifies sites of high-frequency activity maintaining atrial fibrillation in humans. Circulation. 2005;112:789–797. doi: 10.1161/CIRCULATIONAHA.104.517011. [DOI] [PubMed] [Google Scholar]
- 18.Atienza F, Almendral J, Moreno J, Vaidyanathan R, Talkachou A, Kalifa J, Arenal A, Villacastin JP, Torrecilla EG, Sanchez A, Ploutz-Snyder R, Jalife J, Berenfeld O. Activation of inward rectifier potassium channels accelerates atrial fibrillation in humans: evidence for a reentrant mechanism. Circulation. 2006;114:2434–2442. doi: 10.1161/CIRCULATIONAHA.106.633735. [DOI] [PubMed] [Google Scholar]
- 19.Antz M, Otomo K, Arruda M, Scherlag B, Pitha J, Tondo C, Lazzara R, Jackman W. Electrical connections between the right atrium and the left atrium via the musculature of the coronary sinus. Circulation. 1998;98:1790–1795. doi: 10.1161/01.cir.98.17.1790. [DOI] [PubMed] [Google Scholar]
- 20.Dolber PC, Spach MS. Structure of the canine Bachmann’s bundle related to propagation of excitation. Am J Physiol. 1989;257:H1446–H1457. doi: 10.1152/ajpheart.1989.257.5.H1446. [DOI] [PubMed] [Google Scholar]
- 21.Kumagai K, Khrestian C, Waldo AL. Simultaneous multisite mapping studies during induced atrial fibrillation in the sterile pericarditis model. Insights into the mechanisms of its maintenance. Circulation. 1997;95:511–521. doi: 10.1161/01.cir.95.2.511. [DOI] [PubMed] [Google Scholar]
- 22.Kalifa J, Jalife J, Zaitsev AV, Bagwe S, Warren M, Moreno J, Berenfeld O, Nattel S. Intra-atrial pressure increases rate and organization of waves emanating from the superior pulmonary veins during atrial fibrillation. Circulation. 2003;108:668–671. doi: 10.1161/01.CIR.0000086979.39843.7B. [DOI] [PubMed] [Google Scholar]
- 23.Berenfeld O, Zaitsev AV, Mironov SF, Pertsov AM, Jalife J. Frequency-dependent breakdown of wave propagation into fibrillatory conduction across the pectinate muscle network in the isolated sheep right atrium. Circ Res. 2002;90:1173–1180. doi: 10.1161/01.res.0000022854.95998.5c. [DOI] [PubMed] [Google Scholar]
- 24.Kalifa J, Tanaka K, Zaitsev AV, Warren M, Vaidyanathan R, Auerbach D, Pandit S, Vikstrom KL, Ploutz-Snyder R, Talkachou A, Atienza F, Guiraudon G, Jalife J, Berenfeld O. Mechanisms of wave fractionation at boundaries of high-frequency excitation in the posterior left atrium of the isolated sheep heart during atrial fibrillation. Circulation. 2006;113:626–633. doi: 10.1161/CIRCULATIONAHA.105.575340. [DOI] [PubMed] [Google Scholar]
- 25.Lazar S, Dixit S, Marchlinski FE, Callans DJ, Gerstenfeld EP. Presence of left-to-right atrial frequency gradient in paroxysmal but not persistent atrial fibrillation in humans. Circulation. 2004;110:3181–3186. doi: 10.1161/01.CIR.0000147279.91094.5E. [DOI] [PubMed] [Google Scholar]
- 26.Sahadevan J, Ryu K, Peltz L, Khrestian CM, Stewart RW, Markowitz AH, Waldo AL. Epicardial Mapping of Chronic Atrial Fibrillation in Patients: Preliminary Observations. Circulation. 2004;110:3293–3299. doi: 10.1161/01.CIR.0000147781.02738.13. [DOI] [PubMed] [Google Scholar]
- 27.Botteron GW, Smith JM. Quantitative assessment of the spatial organization of atrial fibrillation in the intact human heart. Circulation. 1996;93:513–518. doi: 10.1161/01.cir.93.3.513. [DOI] [PubMed] [Google Scholar]
- 28.Fischer G, Stuhlinger MC, Nowak CN, Wieser L, Tilg B, Hintringer F. On computing dominant frequency from bipolar intracardiac electrograms. IEEE Trans Biomed Eng. 2007;54:165–169. doi: 10.1109/TBME.2006.883739. [DOI] [PubMed] [Google Scholar]
- 29.Ng J, Kadish AH, Goldberger JJ. Effect of electrogram characteristics on the relationship of dominant frequency to atrial activation rate in atrial fibrillation. Heart Rhythm. 2006;3:1295–1305. doi: 10.1016/j.hrthm.2006.07.027. [DOI] [PubMed] [Google Scholar]
- 30.Schuessler RB, Kay MW, Melby SJ, Branham BH, Boineau JP, Damiano RJ., Jr Spatial and temporal stability of the dominant frequency of activation in human atrial fibrillation. J Electrocardiol. 2006 doi: 10.1016/j.jelectrocard.2006.04.009. [DOI] [PubMed] [Google Scholar]
- 31.Wu TJ, Doshi RN, Huang HLA, Blanche C, Kass RM, Trento A, Cheng W, Karagueuzian HS, Peter CT, Chen PS. Simultaneous biatrial computerized mapping during permanent atrial fibrillation in patients with organic heart disease. J Cardiovasc Electrophysiol. 2002;13:571–577. doi: 10.1046/j.1540-8167.2002.00571.x. [DOI] [PubMed] [Google Scholar]
- 32.Sih HJ, Zipes DP, Berbari EJ, Adams DE, Olgin JE. Differences in organization between acute and chronic atrial fibrillation in dogs. Journal of the American College of Cardiology. 2000;36:924–931. doi: 10.1016/s0735-1097(00)00788-9. [DOI] [PubMed] [Google Scholar]
- 33.Wu TJ, Ong JJC, Chang CM, Doshi RN, Yashima M, Huang HLA, Fishbein MC, Ting CT, Karagueuzian HS, Chen PS. Pulmonary veins and ligament of Marshall as sources of rapid activations in a canine model of sustained atrial fibrillation. Circulation. 2001;103:1157–1163. doi: 10.1161/01.cir.103.8.1157. [DOI] [PubMed] [Google Scholar]
- 34.Arora R, Verheule S, Scott L, Navarrete A, Katari V, Wilson E, Vaz D, Olgin JE. Arrhythmogenic substrate of the pulmonary veins assessed by high-resolution optical mapping. Circulation. 2003;107:1816–1821. doi: 10.1161/01.CIR.0000058461.86339.7E. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Atienza F, Almendral J, Jalife J, Zlochiver S, Ploutz-Snyder R, Torrecilla EG, Arenal A, Kalifa J, Fernandez-Aviles F, Berenfeld O. Real-time dominant frequency mapping and ablation of dominant frequency sites in atrial fibrillation with left-to-right frequency gradients predicts long-term maintenance of sinus rhythm. Heart Rhythm. 2009;6:33–40. doi: 10.1016/j.hrthm.2008.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yoshida K, Chugh A, Ulfarsson M, Good E, Kuhne M, Crawford T, Sarrazin JF, Chalfoun N, Wells D, Boonyapisit W, Veerareddy S, Billakanty S, Wong WS, Jongnarangsin K, Pelosi F, Jr, Bogun F, Morady F, Oral H. Relationship between the spectral characteristics of atrial fibrillation and atrial tachycardias that occur after catheter ablation of atrial fibrillation. Heart Rhythm. 2009;6:11–17. doi: 10.1016/j.hrthm.2008.09.031. [DOI] [PubMed] [Google Scholar]
- 37.Lazar S, Dixit S, Callans DJ, Lin D, Marchlinski FE, Gerstenfeld EP. Effect of pulmonary vein isolation on the left-to-right atrial dominant frequency gradient in human atrial fibrillation. Heart Rhythm. 2006;3:889–895. doi: 10.1016/j.hrthm.2006.04.018. [DOI] [PubMed] [Google Scholar]
- 38.Sanders P, Hocini M, Jais P, Hsu LF, Takahashi Y, Rotter M, Scavee C, Pasquie JL, Sacher F, Rostock T, Nalliah CJ, Clementy J, Haissaguerre M. Characterization of focal atrial tachycardia using high-density mapping. J Am Coll Cardiol. 2005;46:2088–2099. doi: 10.1016/j.jacc.2005.08.044. [DOI] [PubMed] [Google Scholar]
- 39.Hsu LF, Jais P, Hocini M, Sanders P, Rotter M, Takahashi Y, Scavee C, Sacher F, Clementy J, Haissaguerre M. High-density circumferential pulmonary vein mapping with a 20-pole expandable circular mapping catheter. Pacing Clin Electrophysiol. 2005;28 (Suppl 1):S94–S98. doi: 10.1111/j.1540-8159.2005.00061.x. [DOI] [PubMed] [Google Scholar]
- 40.Sarmast F, Kolli A, Zaitsev A, Parisian K, Dhamoon AS, Guha PK, Warren M, Anumonwo JMB, Taffet SM, Berenfeld O, Jalife J. Cholinergic atrial fibrillation: I-K,I-ACh gradients determine unequal left/right atrial frequencies and rotor dynamics. Cardiovascular Research. 2003;59:863–873. doi: 10.1016/s0008-6363(03)00540-6. [DOI] [PubMed] [Google Scholar]
- 41.Kabell G, Buchanan LV, Gibson JK, Belardinelli L. Effects of adenosine on atrial refractoriness and arrhythmias. Cardiovasc Res. 1994;28:1385–1389. doi: 10.1093/cvr/28.9.1385. [DOI] [PubMed] [Google Scholar]
- 42.Belardinelli L, Shryock JC, Song Y, Wang D, Srinivas M. Ionic basis of the electrophysiological actions of adenosine on cardiomyocytes. FASEB Journal. 1995;9:359–365. doi: 10.1096/fasebj.9.5.7896004. [DOI] [PubMed] [Google Scholar]
- 43.Khositseth A, Clapham DE, Ackerman MJ. Intracellular signaling and regulation of cardiac ion channels. In: Zipes DP, Jalife J, editors. Cardiac Electrophysiology - From Cell to Bedside. Philadelphia, PA: W.B. Saunders; 2004. pp. 33–41. [Google Scholar]
- 44.Guillem MS, Climent AM, Castells F, Husser D, Millet J, Arya A, Piorkowski C, Bollmann A. Noninvasive mapping of human atrial fibrillation. J Cardiovasc Electrophysiol. 2009;20:507–513. doi: 10.1111/j.1540-8167.2008.01356.x. [DOI] [PubMed] [Google Scholar]
- 45.Ramanathan C, Ghanem RN, Jia P, Ryu K, Rudy Y. Noninvasive electrocardiographic imaging for cardiac electrophysiology and arrhythmia. Nat Med. 2004;10:422–428. doi: 10.1038/nm1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang Y, Schuessler RB, Damiano RJ, Woodard PK, Rudy Y. Noninvasive electrocardiographic imaging (ECGI) of scar-related atypical atrial flutter. Heart Rhythm. 2007;4:1565–1567. doi: 10.1016/j.hrthm.2007.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang Y, Cuculich PS, Woodard PK, Lindsay BD, Rudy Y. Focal atrial tachycardia after pulmonary vein isolation: noninvasive mapping with electrocardiographic imaging (ECGI) Heart Rhythm. 2007;4:1081–1084. doi: 10.1016/j.hrthm.2007.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Richter U, Stridh M, Bollmann A, Husser D, Sornmo L. Spatial characteristics of atrial fibrillation electrocardiograms. J Electrocardiol. 2008;41:165–172. doi: 10.1016/j.jelectrocard.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 49.Dibs SR, Ng J, Arora R, Passman RS, Kadish AH, Goldberger JJ. Spatiotemporal characterization of atrial activation in persistent human atrial fibrillation: multisite electrogram analysis and surface electrocardiographic correlations--a pilot study. Heart Rhythm. 2008;5:686–693. doi: 10.1016/j.hrthm.2008.01.027. [DOI] [PubMed] [Google Scholar]
- 50.Richter U, Bollmann A, Husser D, Stridh M. Right atrial organization and wavefront analysis in atrial fibrillation. Med Biol Eng Comput. 2009;47:1237–1246. doi: 10.1007/s11517-009-0540-2. [DOI] [PubMed] [Google Scholar]
- 51.Yoshida K, Chugh A, Good E, Crawford T, Myles J, Veerareddy S, Billakanty S, Wong WS, Ebinger M, Pelosi F, Jongnarangsin K, Bogun F, Morady F, Oral H. A critical decrease in dominant frequency and clinical outcome after catheter ablation of persistent atrial fibrillation. Heart Rhythm. 2010;7:295–302. doi: 10.1016/j.hrthm.2009.11.024. [DOI] [PubMed] [Google Scholar]