Abstract
The autoinflammatory diseases are characterized by seemingly unprovoked episodes of inflammation, without high-titer autoantibodies or antigen-specific T cells. The concept was proposed ten years ago with the identification of the genes underlying hereditary periodic fever syndromes. This nosology has taken root because of the dramatic advances in our knowledge of the genetic basis of both mendelian and complex autoinflammatory diseases, and with the recognition that these illnesses derive from genetic variants of the innate immune system. Herein we propose an updated classification scheme based on the molecular insights garnered over the past decade, supplanting a clinical classification that has served well but is opaque to the genetic, immunologic, and therapeutic interrelationships now before us. We define six categories of autoinflammatory disease: IL-1β activation disorders (inflammasomopathies), NF-κB activation syndromes, protein misfolding disorders, complement regulatory diseases, disturbances in cytokine signaling, and macrophage activation syndromes. A system based on molecular pathophysiology will bring greater clarity to our discourse while catalyzing new hypotheses both at the bench and at the bedside.
Keywords: innate immunity, IL-1β, inflammasome, type 2 diabetes mellitus, pulmonary fibrosis, Crohn’s disease, ankylosing spondylitis, atherosclerosis
INTRODUCTION
The possibility of maladies in which the immune system turns against its host has long held a special fascination, dating back to the time of Paul Ehrlich and “Horror Autotoxicus” (1). Inherited illnesses, so-called “experiments of nature,” extend our understanding of genes and proteins we may have thought we understood while opening windows to the heretofore unimaginable. The publication of this review marks the tenth anniversary of the concept of systemic autoinflammatory diseases. The idea was initially proposed with the identification of ectodomain mutations in the p55 tumor necrosis factor (TNF) receptor in patients with a dominantly inherited syndrome of fever and widespread inflammation (the TNF receptor–associated periodic syndrome, TRAPS) (2). This discovery followed close on the heels of the positional cloning of the gene for a similar recessively inherited illness, familial Mediterranean fever (FMF) (3, 4), and thereby raised the possibility that these disorders might be prototypes for an emerging family of inflammatory diseases. Both FMF and TRAPS are characterized by seemingly unprovoked, recurrent episodes of fever, serositis, arthritis, and cutaneous inflammation, but the usual hallmarks of autoimmunity, namely high-titer autoantibodies and antigen-specific T cells, are usually absent. The term autoinflammatory was coined to draw the distinction between this category of illnesses and the more classically recognized autoimmune diseases, such as systemic lupus erythematosus and rheumatoid arthritis, in which the hallmarks of adaptive immunity are more evident.
The following year witnessed the identification of the gene underlying yet another hereditary fever, the hyperimmunoglobulinemia D with periodic fever syndrome (HIDS) (5, 6), and the positional cloning of a dominantly inherited gene that causes a curious syndrome of fever, malaise, and hives-like rash upon generalized cold exposure (7). Neither of these latter mendelian disorders fits under the rubric of classical autoimmunity, thus further kindling the notion that autoinflammation might serve as the basis for a new taxonomy of human diseases that are in a sense complementary to the established autoimmune diseases. Based more on their clinical presentation than on a detailed understanding of their molecular basis, several other categories were added to the autoinflammatory universe, including metabolic disorders such as gout and other crystalline arthropathies, complement diatheses such as hereditary angioedema, granulomatous diseases such as Blau syndrome (chronic granulomatous synovitis with uveitis and cranial neuropathy), storage diseases such as Gaucher’s disease and Hermansky-Pudlak syndrome, fibrosing disorders such as idiopathic pulmonary fibrosis, and vasculitic syndromes such as Behçet’s disease (8). This formulation not only widened the phenotypic scope associated with autoinflammation but also extended the concept into the realm of genetically complex disease. Even more recently, several other classes of diseases have been placed under the autoinflammatory banner, including idiopathic febrile syndromes [systemic-onset juvenile idiopathic arthritis (SoJIA); adult Still’s disease; the syndrome of periodic fever with aphthous stomatitis, pharyngitis, and cervical adenitis (PFAPA)] (9, 10); pyogenic disorders such as the syndrome of pyogenic arthritis, pyoderma gangrenosum, and acne (PAPA) (11); and the autoinflammatory bone diseases (12). The notion that the inflammatory manifestations of these diseases are truly unprovoked is, of course, a relative matter, since we now know that a number of factors, including psychological stress, trauma, immunizations, cold exposure, and dietary indiscretion, may trigger some of these illnesses, but in all cases the autoinflammatory appellation implicitly posits a significant host predilection.
A truly useful disease nosology reflects not only clinical phenotype but also underlying biology, thereby suggesting previously unexpected relationships between/among illnesses, spawning new pathogenic hypotheses, and directing the clinician to novel therapeutic targets. At first in parallel and relatively independently, the biologic basis for autoinflammation was established with the recognition of innate immunity as a phylogenetically ancient, hardwired, rapid-response system distinct from but, in mammals, intertwined with adaptive immunity (13). Over time, it became clear that the myeloid effector cells and germline molecules of innate immunity play a major role in the pathogenesis of many of the illnesses clinically classified as autoinflammatory, while the lymphoid cells and somatically plastic molecules of the adaptive immune system play a more significant role in the classic autoimmune diseases. A watershed in the convergence of the clinical concept of autoinflammatory disease with the biology of innate immunity came with the discovery that three well-established autoinflammatory diseases are all caused by activating, gain-of-function mutations in NLRP3 (originally denoted CIAS1 for cold-induced autoinflammatory syndrome 1; also known as NALP3, PYPAF1, and CLR1.1), encoding what was then a newly recognized molecular linchpin in the innate immune system (7, 14, 15).
The discovery of disease-associated mutations in NLRP3 solidified the nexus between autoinflammatory disease and innate immunity for several reasons. First, the NLRP3 protein product, originally called cryopyrin, now officially denoted NLRP3, is a component of a macromolecular complex, the inflammasome, that senses various microbial products and endogenous “danger signals” (damage-associated molecular patterns, DAMPs) to activate caspase-1, thereby initiating IL-1β and IL-18 processing, a key step in the innate immune response (16). Second, the NLRP3 inflammasome may directly or indirectly interact with proteins mutated in other putative autoinflammatory diseases, including pyrin (in FMF) (17) and PSTPIP1 (in PAPA syndrome) (11). Third, the inflammasome has now been implicated in the pathogenesis of a number of diseases, such as gout (18) and pulmonary fibrosis (19–21), included in the expanded definition of autoinflammation, thus providing molecular vindication for the clinical classification. Finally, NLRP3 is a prototype for a family of proteins, now known as the NLR family (22–24), that is intimately involved in the innate immune system, and recent evidence implicates other members of this protein family in human disease.
With the recognition that these inflammatory diseases without hallmarks of adaptive immunity are in fact disorders of the innate immune system, it is possible to propose a new schema based upon underlying molecular mechanisms (Table 1). It should be stated at the outset that, as noted by McGonagle & McDermott (25), the spectrum of self-reactive immunological disease represents a continuum between autoimmune disorders based primarily on lesions of the adaptive immune system and autoinflammatory conditions rooted primarily in the innate immune system. Particularly for genetically complex disorders, multiple lesions of both branches of the immune system, with potentially self-amplifying loops, are quite possible. In keeping with the foregoing discussion, the first types of autoinflammatory disease enumerated in the Table are IL-1β inflammasomopathies, defined as disorders of macromolecular IL-1β-activating complexes, the prototypes of which are nucleated by NLRP3, but which may also include complexes of a number of related proteins. Intrinsic inflammasomopathies represent molecular lesions in the constituent proteins of the complex, while extrinsic inflammasomopathies denote disorders of various upstream or downstream regulatory elements.
Table 1.
Provisional molecular/functional classification of autoinflammatory disease
| Disease | Gene (chromosome) | Protein (synonyms) or pathogenic stimulus |
|---|---|---|
|
Type 1: IL-1β activation disorders (inflammasomopathies) | ||
| Intrinsic | ||
| FCASa, MWSb, NOMIDc/CINCAd | NLRP3/CIAS1 (1q44) | NLRP3e (cryopyrin, NALP3, PYPAF1) |
| Extrinsic | ||
| FMFf | MEFV (16p13.3) | Pyrin (marenostrin) |
| PAPAg | PSTPIP1 (15q24–25.1) | PSTPIP1h (CD2BP1i) |
| CRMOj/SAPHOk | Complex | |
| Majeed syndrome | LPIN2 (18p11.31) | Lipin-2 |
| HIDSl | MVK (12q24) | Mevalonate kinase |
| Recurrent hydatidiform mole | NLRP7 (19q13) | NLRP7 (NALP7, PYPAF3, NOD12) |
| DIRAm | IL1RN | IL-1Ra |
| Complex/acquired | ||
| Gout, pseudogout | Complex | Uric acid/CPPD |
| Fibrosing disorders | Complex | Asbestos/silica |
| Type 2 diabetes mellitus | Complex | Hyperglycemia |
| Schnitzler syndrome | Sporadic | |
|
Type 2: NF- B activation disorders | ||
| Crohn’s disease | Complex | Muramyl dipeptide |
| NOD2 (16p12) | NOD2n (CARD15) | |
| ATG16L1 (2q37.1) | ATG16L1o | |
| IRGM (5q33.1) | IRGMp | |
| Blau syndrome | NOD2 (16p12) | NOD2 (CARD15) |
| FCAS2 (Guadaloupe periodic fever) | NLRP12 (19q13.4) | NLRP12 (NALP12) |
|
Type 3: Protein folding disorders of the innate immune system | ||
| TRAPSq | TNFRSF1A (12p13) | TNFRSF1Ar (TNFR1, p55, CD120a) |
| Spondyloarthropathies | Complex | |
| HLA-B (6p21.3) | HLA-B27s | |
| ERAP1 (5q15) | ERAP1t (ARTS1) | |
|
Type 4: Complement disorders | ||
| aHUSu | CFH (1q32) | Complement factor H |
| MCP (1q32) | MCPv (CD46) | |
| CFI (4q25) | Complement factor I | |
| CFB (6p21.3) | Complement factor B | |
| Complex | Autoantibodies | |
| AMDw | Complex | |
| CFH (1q32) | Complement factor H | |
|
Type 5: Cytokine signaling disorders | ||
| Cherubism | SH3BP2 (4p16.3) | SH3-binding protein 2 |
|
Type 6: Macrophage activation | ||
| Familial HLHx | UNC13D (17q21.1) | Munc13-4 |
| PRF1 (10q22) | Perforin 1 | |
| STX11 (6q24.2) | Syntaxin 11 | |
| Complex | Virus | |
| Chediak-Higashi syndrome | LYST (1q42.3) | LYSTy (CHS1) |
| Griscelli syndrome | RAB27A (15q21.3) | RAB27A |
| X-linked lymphoproliferative syndrome | SH2D1A (Xq25) | SAPz |
| Hermansky-Pudlak syndrome | HPS1-8 | HPS1-8aa |
| Secondary HLH | Complex | |
| Atherosclerosis | Complex | Cholesterol |
Familial cold autoinflammatory syndrome;
Muckle-Wells syndrome;
neonatal-onset multisystem inflammatory disease;
chronic neurologic cutaneous and articular syndrome;
nucleotide-binding domain, leucine-rich repeat, and pyrin domain containing protein 3;
familial Mediterranean fever;
pyogenic arthritis, pyoderma gangrenosum, and acne;
proline serine threonine phosphatase-interacting protein;
CD2-binding protein 1;
chronic recurrent multifocal osteomyelitis;
synovitis acne pustulosis hyperostosis osteitis;
hyperimmunoglobinemia D with periodic fever syndrome;
deficiency of the interleukin-1 receptor antagonist;
nucleotide-binding oligomerization domain-containing;
autophagy-related 16-like 1;
immunity-related GTPase family M;
TNF receptor–associated periodic syndrome;
TNF receptor superfamily 1A;
human leukocyte antigen B27;
endoplasmic reticulum aminopeptidase 1;
atypical hemolytic uremic syndrome;
membrane cofactor protein;
age-related macular degeneration;
hemophagocytic lymphohistiocytosis;
lysosomal trafficking regulator;
SLAM-associated protein;
Hermansky-Pudlak syndrome 1–8.
Although the IL-1β inflammasome represents a major conceptual advance in our understanding of innate immunity and related human disease, it is by no means the only molecular engine of innate immunity. In the Table, the proposed Type 2 autoinflammatory diseases, such as Crohn’s disease (CD) and Blau syndrome (BS), are caused in part by sequence variants in NOD2/CARD15 (26–28). While IL-1β undeniably plays a role in both illnesses, the NOD2/CARD15 protein plays a central role in NF-κB activation in response to intracellular microbial products. Emphasizing the interplay between innate and adaptive immunity, granulomas feature prominently in both disorders. In a third member of the proposed NF-κB activation disorders, Guadaloupe variant periodic fever syndrome, mutations occur in NLRP12, encoding a regulator of NF-κB activation (29).
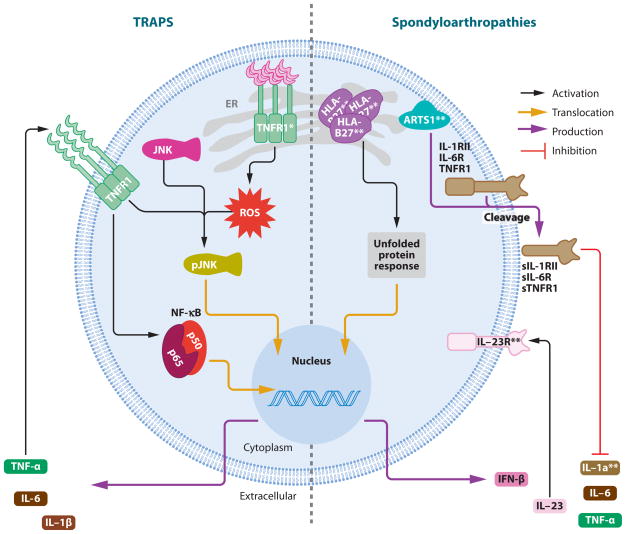
A third group of autoinflammatory diseases is due to the biologic consequences of protein misfolding in cells of the innate immune system. The mendelian prototype of this process is TRAPS, in which missense substitutions in the p55 TNF receptor lead to misfolding (30) and ligand-independent activation of kinases and aberrant cytokine production. Similarly, in the spondyloarthropathies, misfolding of HLA-B27 appears to trigger the unfolded protein response (UPR) in macrophages and consequently inappropriate cytokine secretion (31–33).
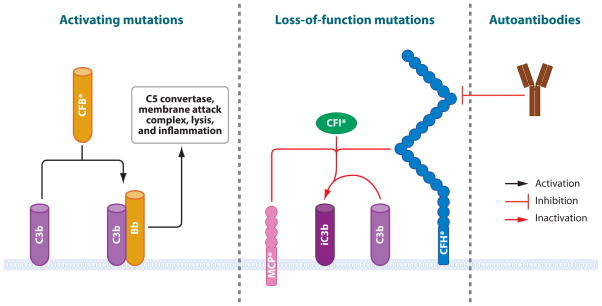
Disorders of the complement system, long recognized as a key component of innate immunity, can lead to a host of immunologic disorders. In some cases, such as the deficiency of the fourth component of complement, abnormal clearance of immune complexes leads to a classical autoimmune lupus-like picture. Here we focus on those instances in which the deficiency of complement regulatory factors produces an autoinflammatory phenotype, such as age-related macular degeneration and atypical hemolytic uremic syndrome (Type 4 disease in the Table).
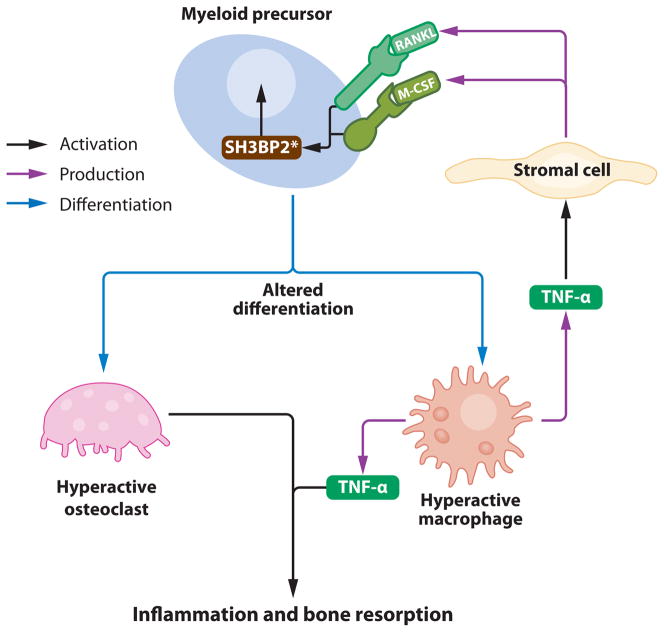
Cherubism, a relatively newly recognized autoinflammatory disorder of the bone, is caused by mutations in an SH3-binding protein (34), which in animal models leads to heightened responsiveness to the cytokines M-CSF and RANKL, and increased osteoclastogenesis (35). Given the importance of cytokine signaling in the innate immune response, additional examples of aberrant cytokine responses will likely be identified in other autoinflammatory diseases, and hence the proposal of Type 5 disorders.
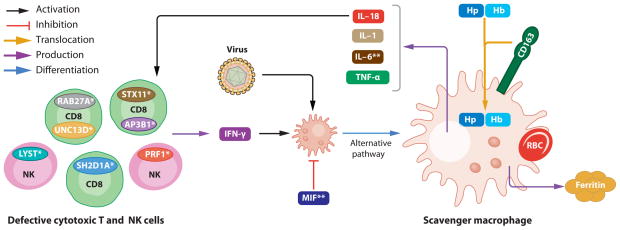
Finally, macrophage activation is a common theme among a host of inflammatory diseases, and the genetics of familial hemophagocytic lymphohistiocytosis implicates loss-of-function lesions in the adaptive immune system as one cause of this (36, 37). Other mechanisms leading to activation of effector cells in the innate immune system and elaboration of a proinflammatory cytokine milieu also characterize Type 6 autoinflammatory disease, which possibly includes the leading cause of death in the Western world, atherosclerosis (38).
There remain a number of human illnesses that are clearly autoinflammatory but do not fall neatly into any of the proposed categories. For some disorders, such as PFAPA, the molecular details are still too few to permit classification, whereas for other syndromes, such as SoJIA, multiple molecular mechanisms are emerging. In yet other instances, such as Behçet’s disease, lack of detail and heterogeneity appear paradoxically to go hand in hand. Undoubtedly, the six types of autoinflammatory disease enumerated in Table 1 represent only a beginning but should serve not only to organize our thinking about existing data, but also to stimulate hypotheses about less well-understood phenotypes.
IL-1β ACTIVATION DISORDERS (INFLAMMASOMOPATHIES)
Intrinsic Inflammasomopathies
Cryopyrin-associated periodic syndromes
As disorders of an essential inflammasome protein, the cryopyrin-associated periodic syndromes (CAPS, also known as cryopyrinopathies) encompass a spectrum of disease states. In order of severity, from mild to severe, these include familial cold autoinflammatory syndrome (FCAS), which presents with cold-induced fevers, urticaria-like rash, and constitutional symptoms; Muckle-Wells syndrome (MWS), with fevers, hives, sensorineural hearing loss, and arthritis unrelated to cold exposure; and neonatal-onset multisystem inflammatory disease (NOMID) [or chronic infantile neurologic cutaneous articular (CINCA) syndrome], which presents with fever, urticaria, epiphyseal overgrowth of the long bones, and chronic aseptic meningitis.
The cryopyrinopathies are due to autosomal dominant or de novo mutations of NLRP3 (7, 14, 15). The protein product of this gene (Figure 1) was first denoted cryopyrin, although the current convention is NLRP3 (24). To avoid confusion, we have retained the cryopyrinopathy/CAPS disease nomenclature in this review, although we use the new terminology to denote the gene and protein. The original protein name was derived from the aforementioned association of symptoms with cold exposure in some patients (cryo), and its invariant ~90 amino acid N-terminal death-fold motif, the pyrin domain (PYD) (39–41), that mediates cognate interactions with other proteins, and that it shares with the FMF protein, pyrin. Through this domain both NLRP3 and pyrin interact with an adaptor protein denoted ASC (apoptosis-associated speck-like protein with a caspase-recruitment domain). NLRP3 belongs to the family of NLR (nucleotide-binding domain and leucine-rich repeat containing) proteins (24) implicated in inflammation and apoptosis, providing an additional clue as to its function. C-terminal leucine-rich repeats (LRRs) had already been implicated in sensing bacterial components, suggesting a role in the innate immune response. The case was strengthened with the discovery that, through pyrin domain–ASC interaction, the related protein NLRP1 nucleates the first recognized IL-1β activating inflammasome (42), and the subsequent finding that NLRP3 itself participates in a somewhat different inflammasome that also exhibits pro-IL-1β processing activity (16). Although the inflammasome is also required for processing of IL-18, IL-33, and several other nontraditionally secreted proteins (43), most research has focused on how NLRP3 mutations affect IL-1β production, and certainly IL-1β is upregulated in this disease (15).
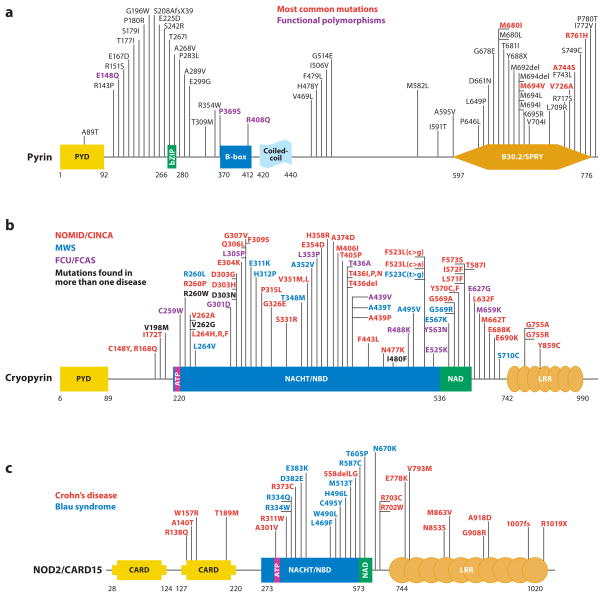
Figure 1.
Schematic representation of mutations in five proteins that cause autoinflammatory diseases. (a) For pyrin, mutations that are most frequently found to cause FMF are presented in red, while those in black are less common disease-causing variants. Residues in purple are found at approximately 1% allele frequency in the general population and may therefore represent functional polymorphisms. (b) Cryopyrin mutations cause a spectrum of disease states that range in severity from severe (NOMID/CINCA, red ), to intermediate (MWS, blue), to mild (FCAS, purple). In some instances, it is difficult to distinguish between NOMID/CINCA and MWS. Residues for which disease presentation overlaps are depicted in black. (c) NOD2/CARD15 mutations can cause Blau syndrome/early-onset sarcoidosis (blue), which cluster within the NACHT/NBD and NAD domains. Other variants that are spread throughout this protein are associated with Crohn’s disease (red ). (d ) Mevalonate kinase mutations can cause the severe metabolic disease mevalonic aciduria (red ), or the less severe autoinflammatory disease HIDS (blue). HIDS mutations are recessively inherited and often include one mild mutation coupled with a severe mutation; thus, the severe mutations can be found in both HIDS and mevalonic aciduria (black). (e) Dominantly inherited missense mutations in TNFR1 that cause TRAPS are now known to affect almost every cysteine residue within the first two cysteine-rich domains of the extracellular region of the protein (red ). These mutations appear to affect the protein folding, whereas at least two mutations, I170N and V173D, can affect ectodomain cleavage that generates the soluble form of the receptor. P46L and R92Q ( purple) are probably functional polymorphisms also present in unaffected individuals. The numbering system for TNFR1 used here begins at residue 30, which is at the N terminus after removal of the 29-residue signal peptide, as per common convention. Abbreviations used: PYD, pyrin domain; NBD, nucleotide-binding domain; NAD, NACHT-associated domain; LRR, leucine-rich repeats; CRD, cysteine-rich domain; TM, transmembrane domain.
Another domain of the NLRP3 protein is the ATP-binding cassette (nucleotide binding domain, NBD, or NACHT domain; Figure 1), which has now been confirmed as such both in a cell-free system (44) and in a number of other in vitro settings (45), and is the major locus of CAPS-associated mutations. This leads to the intriguing possibility that these sequence variants may confer activation of the protein without the usual ATP costimulus, and certainly many of the mutations are structurally proximate to a hypothetical ATP-binding site (46). Indeed, Gattorno et al. in a recent publication, showed that mutation-positive NOMID and MWS patient peripheral blood mononu-clear cells (PBMCs) secrete higher levels of IL-1β in response to LPS alone than healthy controls, but that with the addition of ATP, secreted IL-1β levels are comparable (47). This suggests that the mutations in NLRP3 have bypassed a requirement for ATP in IL-1β production. However, inactivating mutations at the ATP-binding site ablate the constitutive IL-1β production associated with MWS mutations in an overexpression system (45). This emphasizes the importance of ATP binding to NLRP3 activation and indicates that the ATP independence of MWS mutations may not be related to the requirement of ATP binding to the NACHT domain. Instead, NLRP3 mutations may function independently of exogenous ATP due to heightened downstream sensitivity to the effect of ATP on the P2X7 receptor at the cell surface, which is a well-documented stimulus for the processing and secretion of IL-1β (48, 49). It would be interesting to test these hypotheses in a cell-free system and to know the exact mechanism behind the gain of function of NLRP3 mutations.
In 2006, four laboratories published data describing the role of NLRP3 in vivo from analysis of knockout mice. These findings are presented in greater depth in another review appearing in this issue of the Annual Review of Immunology (50). The results that are most relevant to the current discussion include the requirement of NLRP3 for macrophage IL-1β production in response to Toll-like receptor (TLR) agonists plus ATP (51, 52), to Gram-positive bacteria such as Staphylococcus aureus or Listeria monocytogenes (51), to bacterial RNA (53), to dsRNA and viral RNA (54), and to uric acid crystals (18). While NLRP3 is required to produce IL-1β in response to these many and varied insults, the disease-causing mutations in this protein do not seem to render CAPS patients clinically overresponsive when faced with these challenges in natura, although some patients anecdotally report increased resistance to common viral infections. Sutterwala and colleagues also observed that, like IL-1β, NLRP3 was required to generate contact hypersensitivity to the hapten trinitrophenylchloride (TNP-CL) (52). This observation agrees with a recent body of work highlighting the critical role of the innate immune system in contact hypersensitivity, which is predominantly a T cell–mediated disease (55). Again, we have not observed that CAPS patients manifest an increased delayed-type hypersensitivity reaction.
One further aspect of the function of NLRP3 was indicated by the reduced level of macrophage cell death observed for cells of knockout mice in response to Gram-positive bacteria (Staphylococcus) (51). Willingham et al. showed that this was also true for infection with Shigella flexneri, and independent of the inflammasome component ASC and of IL-1β (56). Monocyte/macrophage cell death has also been examined in overexpression systems (57), and with cells from patients directly (56, 58); in both cases, LPS-induced necrosis appears to be associated with the mutations in NLRP3 that cause CAPS. This comports with previous work that reported on a CAPS patient with low-level somatic mosaicism who did not inherit a mutated NLRP3 allele from either parent (59). Because the mutated cells are only a fraction of the total, being able to identify and sort them could facilitate better detection of this cause of disease. Indeed, by looking at just those cells undergoing necrotic cell death, Saito et al. found somatic mosaicism in three of four previously mutation-negative patients (58). However, this approach would seem to rely heavily on the genetic fidelity of a dead or dying population of cells, which has not been established. Indeed, the very nature of a mutation undergoing selection in a small population of cells that proceed to rapid cell death seems somewhat contradictory, without some driving force for their continued production.
Further lines of investigation that are sure to attract interest include the elucidation of possible genotype-phenotype correlations in the cryopyrinopathies and investigation of the structural basis for disease-causing mutations. While such studies would provide important mechanistic insight, it is very fortunate that therapy for these diseases based on current understanding is remarkably effective. For example, the IL-1 receptor antagonist, anakinra, has a very significant impact in even the most severe cryopyrinopathy, NOMID/CINCA, with dramatic effects on not only the rash and acute-phase proteins, but also on the aseptic meningitis and cochlear inflammation, which often lead to severe disability (60). Improved therapy for CAPS patients now focuses on new biologics with more favorable treatment schedules, such as rilonacept (administered once per week) and canakinumab (administered once every eight weeks). Unlike anakinra, which blocks IL-1β signaling at the receptor, rilonacept and canakinumab target IL-1β directly, and in the studies completed to date, these agents show great promise (61–63). It is interesting that patients with all of the clinical features of CAPS but no demonstrable NLRP3 mutations also respond to therapy targeting IL-1β. This suggests an excellent opportunity for discovering additional disease-causing genes on the IL-1β axis.
Extrinsic Inflammasomopathies
Familial Mediterranean fever (FMF)
FMF is characterized by 1- to 3-day episodes of fever with sterile peritonitis, pleural inflammation, arthritis, and/or rash, sometimes complicated by systemic amyloidosis. While FMF is the prototypic autoinflammatory disease, the effect of the underlying disease-causing mutations on IL-1β production may be primarily extrinsic to the inflammasome (Figure 2). Linkage analysis (64) and positional cloning based on an autosomal recessive model of inheritance allowed two independent consortia to identify mutations in a single gene that cause this disease (3, 4). The gene, denoted MEFV for MEditerranean FeVer, encodes a protein product (Figure 1) alternatively termed pyrin (after the Greek for fever) or marenostrin (after the Latin for the Mediterranean Sea). The mutations causing FMF are present at a very high frequency in several populations, and for this reason the disease is more prevalent in the Mediterranean basin and Middle East (65).
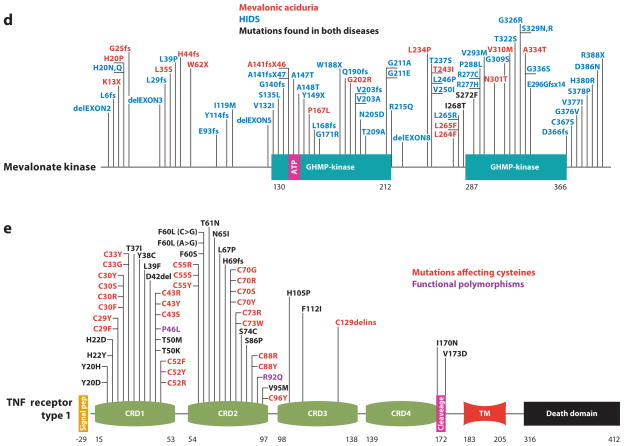
Figure 2.
Mechanisms of autoinflammatory disease regulated by IL-1β (inflammasomopathies) and NF-κB. The NLRP3 protein interacts with ASC and caspase-1 to form a complex termed the inflammasome, which is a macromolecular complex that processes IL-1β into its active form. Mutations (denoted by asterisks) in proteins that affect the function of this complex such as pyrin, PSTPIP1, mevalonate kinase (MK), and NLRP7 thus represent extrinsic inflammasomopathies. Mutations in NLRP3 clearly activate the molecule; however, the precise mechanism by which pyrin and PSTPIP1 mutations regulate inflammasome activity has not been determined. As NLRP7 is a negative regulator of IL-1β production, these mutations are likely to inactivate the protein, and it has been shown that MK mutations are also loss of function, although the pathway that leads from these mutations to Rac1/PI3K/PKB activation is not yet formally described. Preliminary data from our laboratory suggest that mutations in the IL-1 receptor antagonist (IL-1Ra) cause another extrinsic inflammasomopathy. For NF-κB activation disorders, NOD2 could become activated by mutations that may relieve autorepression of the molecule; however, there are also persuasive data showing that mutations inactivate the protein from tolerizing the host to the bacterial cell wall component muramyl dipeptide (MDP). NLRP12 is a negative regulator of NF-κB, and thus a proinflammatory disease would be caused by what appear to be loss-of-function mutations. Abbreviations used: CPPD, calcium pyrophosphate dihydrate; Aβ, amyloid-β; ROS, reactive oxygen species; PKB, protein kinase B.
In the years following the discovery of mutations in MEFV, much has changed in our understanding of this disease. Perhaps most strikingly, as the sequencing of this gene has become widespread in patients and families outside those studied in the initial linkage analysis, we have discovered many patients with compatible symptoms, sometimes even classic manifestations, but only a single MEFV mutation. It is also true that many of the obligate carriers for single MEFV mutations may actually have subclinical evidence of inflammation (66), or even be periodically symptomatic for FMF (67) but not to the extent that would prompt medical evaluation or genetic diagnosis. Although most FMF patients inherit two pyrin mutations recessively, it appears that in some instances a more broadly defined FMF is inherited in a dominant fashion, and in fact there are select mutations for which this does seem to be the case (68). However, for the remaining people with only one pyrin mutation, what determines if disease will present or not? An attractive hypothesis is that there is digenic or even multi-genic inheritance of FMF. In such a scenario, a single pyrin mutation may require permissive alleles at one or more additional loci before FMF is manifest.
Mutations are found throughout the MEFV gene (http://fmf.igh.cnrs.fr/infevers), but the most severe are clustered in exon 10 (69, 70), which encodes a motif known as the B30.2/SPRY domain (PRYSPRY), at the C terminus of the protein (Figure 1). Pyrin is the exemplar for the N-terminal domain that bears its name, and also contains B-box, bZIP basic, and coiled-coil domains. Pyrin is also known as TRIM20, as it is a part of a larger family termed the TRIpartite Motif (TRIM) proteins that typically have a RING domain (not present in pyrin), B-box, and coiled-coil domain, and frequently also the B30.2/SPRY domain (71). Initial speculation concerning the function of pyrin hypothesized that it was a transcription factor (3). Although DNA binding activity has not been ascribed to pyrin, the protein does contain two nuclear localization motifs (3, 72), and the endogenous protein does localize to the nucleus in granulocytes and dendritic cells (73). We have also observed the translocation of a specific N-terminal fragment of pyrin to the nucleus after cleavage by caspase-1 (74). N-terminal pyrin appears to activate NF-κB through increased calpain-mediated degradation of IκB-α, and was also observed in patient leukocytes. Pyrin can also be demonstrated in the cytoplasm of monocytes, and much more has now been published regarding the role of the protein in this locale. One interesting finding was that pyrin interacts with tubulin and colocalizes with microtubules (75), suggesting a rationale for current highly efficacious treatment of the disease using colchicine, a microtubule-destabilizing agent (76).
More recently, pyrin was found to interact with ASC, the apoptosis-associated speck-like protein with a caspase-recruitment domain (CARD), through cognate pyrin domain association. When this interaction was first described, it was the apoptotic effect of ASC that was addressed, and indeed wild-type pyrin, when overexpressed in HeLa cells, appeared to increase ASC speck formation and, paradoxically, increase the survival of these cells (77). Macrophages from mice harboring a truncated form of pyrin have subsequently been shown to manifest a defect in apoptosis (17). In addition to its role in apoptosis, ASC also nucleates inflammasome complexes through the homotypic interactions of its pyrin domain and CARD with NLRP proteins and inflammatory caspases, respectively (78), thus activating IL-1β (Figure 2). The direct interaction of pyrin with ASC suggests potential molecular mechanisms for the inflammatory hallmarks of FMF, either if pyrin inhibits IL-1β activation by competing with caspase-1 for ASC (17) or if pyrin itself forms an inflammasome complex (79).
Depending on experimental conditions, there are data to support both of these formulations and the corresponding antiinflammatory and proinflammatory effects for wild-type pyrin. The initial observation to support an antiinflammatory role for pyrin concerned mice deficient for MEFV that exhibit increased cytokine production and subsequent lethality induced by LPS (17). Because murine MEFV lacks a significant portion of exon 10, including the region where FMF mutations reside, many experiments using human constructs have subsequently been performed. We have shown that the B30.2 domain of pyrin interacts with caspase-1 and that this inhibits the production of mature IL-1β (80). Papin et al. have verified this interaction and the consequent inhibition of IL-1β processing (81), but the effect of FMF-associated mutations in the B30.2 domain remains controversial. On the other hand, Yu et al. reported that overexpression of pyrin in 293T cells that stably express ASC and procaspase-1 appears to activate an inflammasome complex, although overexpressed mutant forms of pyrin did not result in higher levels of caspase-1 activation (79). A different approach to address this issue is to study the effect of a reduction in the level of pyrin. Using siRNA to decrease the level of pyrin in THP-1 cells resulted in increased IL-1β production in vitro (80, 81). Nevertheless, under different experimental conditions ectopic silencing of pyrin resulted in decreased IL-1β production (82), suggesting that the function of pyrin may be context dependent, with dual roles that are physiologically relevant.
Additional studies of peripheral blood leukocytes from FMF patients and healthy controls have been performed to identify a predominant role for pyrin in vivo, but again the results are contradictory. In favor of a proinflammatory effect for wild-type pyrin, the ability of macrophages from healthy donors to process IL-1β compared to the monocytes from which they are derived is significantly reduced, corresponding to the reduced amount of pyrin that these cells express (82). In contrast, the same laboratory showed an association of reduced mRNA levels of pyrin, IL-10, and IL-1 receptor–associated kinase (IRAK)-M with survival in pediatric multiple organ dysfunction syndrome, consistent with an antiinflammatory role for pyrin in cytokine production (83). In FMF patients, Ustek et al. observed reduced levels of MEFV message at baseline, with further reductions during disease flares (84), whereas our preliminary analysis of pyrin protein expression in peripheral blood neutrophils shows an increase in FMF patients. Further complicating the picture, it is not clear whether changes in mRNA or protein expression reflect the underlying pathophysiology of FMF or a compensatory mechanism.
Given the autoinflammatory phenotype in FMF, one might associate an antiinflammatory effect of the wild-type protein and loss-of-function mutations with recessively inherited disease, or a proinflammatory role of the wild-type protein and gain-of-function mutations with dominantly inherited disease. At present, neither the effect of the pyrin protein in experimental models, nor the effect of mutations on gene expression, nor the mode of inheritance of the disease is entirely unambiguous. It has therefore been difficult to construct a detailed molecular account of FMF pathogenesis, and we have provisionally opted to denote FMF as an extrinsic inflammasomopathy, recognizing that further refinements may be necessary.
These points notwithstanding, pyrin does play an important role in the regulation of IL-1β in experimental systems, suggesting that parenteral administration of the recombinant IL-1 receptor antagonist, anakinra, would ameliorate the inflammatory attacks of FMF. Indeed, there are now several anecdotal reports vindicating this concept (80, 85–89), although anakinra appears not to have as uniformly dramatic an effect as that seen in the cryopyrinopathies. The microtubule-inhibitor colchicine, which is given orally and is markedly less expensive than anakinra, remains the drug of choice in FMF. IL-1β inhibitors may play an adjunctive role in the treatment of the 5–10% of FMF patients for whom colchicine is ineffective or not tolerated. The proposed mechanisms by which colchicine prevents the attacks of FMF relate both to the mutant protein, pyrin (which binds to microtubules), and to the granulocytes that are a major locus of pyrin expression. A more complete understanding of how colchicine works in FMF and why it sometimes does not work would likely shed additional light on the function of pyrin, reinforcing the possibility of as yet undiscovered additional functions for this protein.
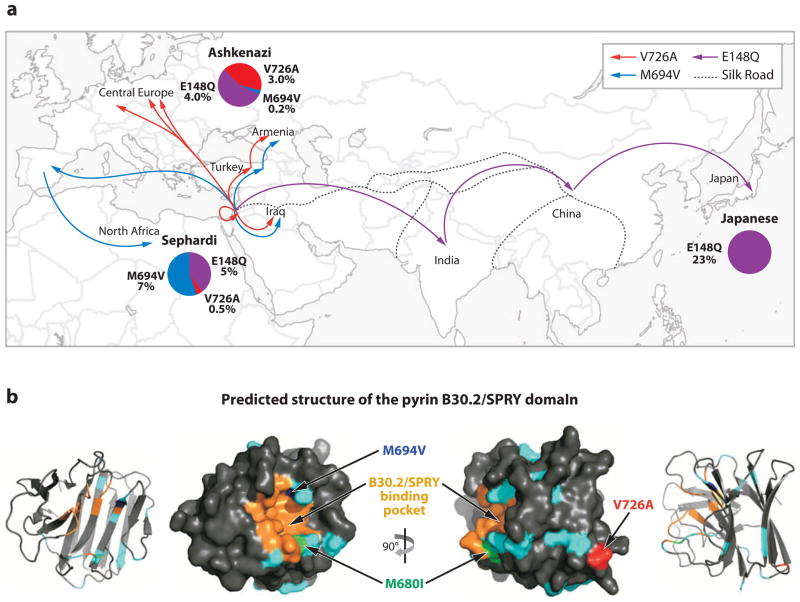
Perhaps the most engaging concept pertinent to any discussion of FMF is the evolution of disease-causing mutations. Several aspects in particular are noteworthy. First, carriers of FMF mutations are far more frequent in certain populations than would be expected. Under the simplifying assumption of autosomal recessive inheritance, even if homozygotes for disease mutations all die before reproductive age, their frequency varies with the square of the mutant allele frequency, which is much less than the frequency of heterozygous carriers, and thus natural selection would have increased the frequency of mutations only if they conferred a heterozygote advantage. Second, different mutations predominate in different populations (Figure 3a). This reinforces the possibility of a selective advantage, as genetic drift would be unlikely to account for the increased carrier rate of several different FMF mutations in these various populations. Third, the most common and potentially most severe mutations cluster in one domain of the protein, the B30.2/SPRY domain. This domain is not conserved among lower species (90), which highlights the relatively recent evolution of pyrin. This is also true for the majority of the TRIM protein family that has rapidly expanded in higher organisms. More than 20 TRIM family proteins exhibit the ability to affect the retroviral life cycle (to date, pyrin has not been found to have such an effect) and some of these TRIM proteins confer innate immune resistance to retroviruses (91). Finally, in nonhuman primates the wild-type pyrin protein includes several residues that are associated with disease in humans (92). For humans, FMF mutations often represent a reversion to ancestral amino acids, and modeling suggests episodic positive selection for the current human wild-type sequence. This argues that nonhuman primates may live with endemic pathogens against which pyrin mutations confer resistance, and that FMF mutations may also confer resistance against a similar, perhaps geographically restricted, pathogen for humans.
Figure 3.
The evolution and structural ramifications of ancestral mutations in pyrin. (a) This figure highlights both that pyrin mutations are very common in several Mediterranean populations and that it is not the same mutation that is expanded in each population. Blue and red lines indicate the migration of populations carrying the M649V and V726A mutations, respectively, while the E148Q mutation ( purple) is common to countries along the medieval trade route (the Silk Road) that extended from the Middle East through to Japan. Pie diagrams indicate the carrier frequencies of selected mutations in Ashkenazi Jewish, Sephardi Jewish, and Japanese populations. (b) A model for the structure of the B30.2/SPRY domain of pyrin (based on that of TRIM21) indicates the preponderance of FMF mutations (cyan) affecting one side of the molecule that is thought to present a protein-binding pocket. A phenotype-genotype correlation is proposed that links the more severe disease-causing mutations (M694V blue, M680I green) to the putative binding site, whereas a less severe mutation (V726A red ) is located further away from this region.
Recent NMR and crystallographic data suggest a possible structural basis for the evolutionary data. The first structure for the B30.2/SPRY domain was an NMR model derived from a murine protein SSB-2 (93). Even in this first model, it was apparent that FMF mutations predominantly affected one face of the molecule, and mutational analysis of the SSB-2 protein showed that a protein-binding site was present at this region. Further information has come from crystal structures of other B30.2/SPRY domains in complex with their binding partners (94, 95). This more definitively described the protein interaction surface of the domain, and models of the pyrin B30.2/SPRY domain show where FMF-associated residues map close to the binding pocket (Figure 3b). Although several FMF mutations do not map near this region, the initial observation that the least severe FMF mutations map further from the binding interface gives a structural understanding to the potential genotype-phenotype correlation in FMF (93). Together, these evolutionary and structural data point to the possibility that pyrin is capable of directly binding to a pathogen, or to another moiety that acts as a surrogate marker of infection. Identification of such a mechanism would represent a major advance in our understanding of the innate immune host defense as it relates to the mechanisms of autoinflammatory disease.
Pyogenic arthritis with pyoderma gangrenosum and acne (PAPA) syndrome
PAPA syndrome is a dramatic hereditary autoinflammatory disorder of the skin and joints for which there is clear evidence of an important, although not necessarily exclusive, pathogenic role for IL-1β. PAPA syndrome was initially described by two different groups, several years apart (96, 97). Both groups observed families with recurrent arthritis, sterile but purulent synovial fluid, and cutaneous manifestations such as pyoderma gangrenosum (large, open purulent lesions) and severe cystic acne. The genetic basis for this dominantly inherited condition was found to be mutations of CD2-binding protein 1 (CD2BP1) (98). This protein is now usually denoted by the name of its murine ortholog, proline serine threonine phosphatase-interacting protein 1 (PSTPIP1), which interacts with a PEST [rich in proline (P), glutamic acid (E), serine (S), and threonine (T)] type protein tyrosine phosphatase (PTP-PEST). Initially, it was shown that the mutations that cause PAPA syndrome diminish the interaction of PSTPIP1 with PTP-PEST, but the relevance of this finding for the promotion of an autoinflammatory disease was unclear.
Subsequent data from our laboratory established that PSTPIP1 interacts with the prototypic autoinflammatory protein, pyrin (11), thus suggesting potential molecular mechanisms for the proinflammatory aspects of PAPA syndrome (Figure 2). Furthermore, the PAPA mutations of PSTPIP1 lead to a stronger interaction with pyrin (FMF mutations in pyrin, however, do not have a similar effect). The increased interaction with pyrin is most likely because the PSTPIP1 variants bind PTP-PEST less avidly and are therefore hyperphosphorylated, and because the avidity of PSTPIP1 for pyrin varies with PSTPIP1 phosphorylation status. Both in vitro and ex vivo, PAPA-associated variants were associated with increased IL-1β production. If one takes the view that pyrin inhibits inflammation, PSTPIP1 mutations could be seen to function by increased sequestration and impairment of pyrin function (11). On the other hand, Yu and colleagues (99) have shown that the domain of pyrin to which PSTPIP1 binds (the B-box) is an autoinhibitory domain that constrains ASC binding and thereby prevents the formation of the pyroptosome, a large molecular assembly of ASC and pyrin molecules of which there is only one per cell (100). Pyroptosome formation rapidly proceeds to cell death and the release of proinflammatory cytokines such as IL-1β. By this formulation, the stronger interaction of the PAPA mutants with the B-box of pyrin would relieve the autoinhibitory constraint and promote inflammation via pyrin engagement in pyroptosomes. While the mechanisms proposed by Shoham et al. (11) and Yu et al. (99) differ respectively as to whether wild-type pyrin is an intermolecular inhibitor of IL-1β production or is itself prevented from activating IL-1β by intramolecular interactions, both agree that increased binding by PSTPIP1 mutants would work against this constraint.
Despite anecdotal evidence for the efficacy of the IL-1 receptor antagonist anakinra in the articular attacks of PAPA syndrome (101), broader experience suggests that IL-1 inhibition is probably not as effective a strategy in PAPA syndrome as for CAPS, and thus raises the possibility that PSTPIP1 mutations may have additional pathophysiologic effects. That PSTPIP1 is also very highly expressed in T cells lends itself to the hypothesis that PAPA syndrome may at some level arise due to improper activation of the adaptive immune response. PSTPIP1 interacts with a number of T cell proteins, including the Wiskott-Aldrich syndrome protein (WASp), CD2, and Fas ligand (FasL) (102–104). PSTPIP1/PTP-PEST binding inhibits WASp phosphorylation, which is required for activated T cell transcriptional activity, actin polymerization, and immunologic synapse formation. Although PAPA-associated PSTPIP1 mutations did not affect WASp binding in yeast, in mammalian cells such mutations may decrease binding as a result of PSTPIP1 hyperphosphorylation and could thereby potentiate T cell activation (98). PTP-PEST/PSTPIP1 interaction with CD2 inhibits T cell activation (103), and, if PAPA mutations inhibit this interaction, T cell activation could ensue. PSTPIP1 also interacts with FasL in T cells, and when PSTPIP1 is overexpressed, FasL is retained in the cytoplasm of the T cell, thus preventing its cytotoxic activity (104). Although the net effect of PAPA-associated PSTPIP1 mutants on T cell function in vivo is unknown, the adaptive immune response could provide at least a trigger for PAPA flares, notwithstanding the fact that the neutrophil is by far the predominant effector cell in PAPA lesions.
Among a number of unanswered questions is the relative contribution of innate and adaptive immunity in the pathogenesis of PAPA. The dominant mode of inheritance suggests a gain of function for mutant PSTPIP1, consistent with its documented increased binding to pyrin, but which may extend to its interactions with binding partners in T cells or other lymphocyte sub-populations. Careful examination of protein interactions in patient cells, and the development of knockout and knockin animal models, may help address basic mechanistic issues.
Chronic recurrent multifocal osteomyelitis (CRMO) and synovitis acne pustulosis hyperostosis osteitis (SAPHO) syndrome
In the cmo mouse model, spontaneous recessive mutations in Pstpip2 (a murine gene encoding a protein homologous to the PAPA syndrome protein PSTPIP1) cause autoinflammatory bone disease (105, 106). This most closely resembles CRMO, a human disorder presenting as bone pain, lytic bone lesions on radiographs, and culture-negative inflammatory infiltrates of the bone, with or without fever (12, 107, 108). CRMO usually presents early in life (~10 years of age), and in most patients treatment with NSAIDs can ameliorate symptoms. CRMO is often associated with other inflammatory syndromes, such as palmar-plantar pustulosis, psoriasis vulgaris, and inflammatory bowel disease. SAPHO syndrome is characterized by the conditions abbreviated in its acronym (synovitis, acne, pustulosis, hyperostosis, and osteitis), and most of these are coincident with CRMO, although skin involvement plays a larger role in SAPHO. Therefore, CRMO and SAPHO syndrome probably form a spectrum of disease with one difference being that CRMO is usually a pediatric disease, whereas for the most part SAPHO syndrome is not.
A genetic influence is clear in some families, with affected siblings, concordant monozygotic twins, and indeed an autosomal recessive form of CRMO with congenital dyserythropoietic anemia, known as Majeed syndrome, that is caused by mutations in the gene LPIN2. Although little is known about the function of LPIN2, it has been suggested that its upregulation in macrophages with oxidative stress could relate to a possible role in regulating the innate immune response (107). By this hypothesis, disease-associated LPIN2 mutations might disturb its protective function against oxidative stress, thus causing tissue damage and polymorphonuclear (PMN) cell influx. This is also how pathology in the spontaneous cmo murine disease proceeds, with neutrophilic osteomyelitis and ensuing bone resorption that closely mirrors both Majeed syndrome and CRMO as a whole (105, 106).
A second mouse model, termed Lupo, exhibiting a similar phenotype primarily in distal appendages, was subsequently generated by random mutagenesis and found to have a different missense mutation in Pstpip2 (106, 109). Lupo is intrinsic to the hematopoietic system as documented by adoptive transfer of affected bone marrow, and the phenotype is unchanged when examined in mice lacking both T and B cells. This characteristic agrees well with the expression of human PSTPIP2 predominantly in monocytes and macrophages and, because expression of mutant Pstpip2 is much lower in Lupo mice relative to controls, suggests that the function of the gene is antiinflammatory. The functional relationship between PSTPIP2 and PSTPIP1 remains unclear, particularly because PSTPIP2 lacks the C-terminal SH3 domain that mediates interactions with many different binding partners for PSTPIP1 including pyrin. The human PSTPIP2 gene is encoded within a genomic interval found to be associated with sporadic CRMO by transmission disequilibrium testing (110), although to date no PSTPIP2 mutations have been identified in sporadic cases.
The classification of CRMO and SAPHO as disorders of IL-1β inflammasome regulation is conjectural at the present juncture, but it does immediately suggest additional lines of investigation both in mice and humans. In the animal model, breeding of intercrosses of Pstpip2 mutant mice with mice deficient in various IL-1β inflammasome components will be of great interest. In humans, the potential efficacy of IL-1 inhibition in patients with the CRMO/SAPHO disease spectrum may provide additional data to support or refute the importance of this cytokine in disease pathogenesis.
Hyperimmunoglobulinemia D with periodic fever syndrome (HIDS)/mevalonate kinase deficiency (MKD)
Initially, mevalonate kinase (MVK), the gene responsible for HIDS (5, 6), was enigmatic as a gene causing autoinflammatory disease without clear links to the immune system. The mevalonate kinase (MK) enzyme is ubiquitously expressed, and by converting mevalonic acid to 5-phosphomevalonic acid it catalyzes an early step in the biosynthetic pathway for cholesterol and nonsterol isoprenes. The most severe loss-of-function mutations in MVK, when present on both chromosomes, result in mevalonic aciduria, with pathology including recurrent fever, mental retardation, developmental abnormalities, and often early death (111). In HIDS there may be approximately 5% residual enzymatic activity (often due to a mild mutation in trans with a more severe mutation), and these patients have the recurrent fever, lymphadenopathy, abdominal pain, and rash associated with HIDS, but not the more severe manifestations of mevalonic aciduria. Whereas the mutations associated with mevalonic aciduria tend to be clustered in sites important to the catalytic activity of the enzyme, HIDS-associated mutations are more broadly distributed throughout the protein sequence (Figure 1). Preliminary hypotheses suggested opposing mechanisms for the disease pathogenesis in HIDS, one being that mevalonic acid is present in excess and is therefore toxic, the other that isoprenoid biosynthesis is deficient, leading to perturbations in signaling pathways (5, 6). Defective apoptosis of lymphocytes from HIDS patients is also suggested to be a cause of disease, although the molecular basis of this is uncertain (112).
More recently, investigation into the signaling pathways that may be affected by alterations in the MK pathway has implicated the small GTPase Rac1, phosphoinositide 3-kinase (PI3K), and protein kinase B (PKB) as key molecules that could give rise to activated caspase-1 (and hence IL-1β production and inflammation) due to isoprenoid deficiency (113). These data come from a model of isoprenoid deficiency created using simvastatin, an inhibitor of HMG-CoA reductase that should mirror the effect of loss-of-function mutations in MK by lowering mevalonate levels. Simvastatin treatment of THP-1 cells after exposure to LPS resulted in increased IL-1β secretion that was abrogated by Rac1 and PI3K inhibition, but further increased when constitutively active PKB was overexpressed (Figure 2). Replication of these findings in vivo, perhaps using the recently developed murine model of HIDS from a mouse lacking one Mvk allele (114), is necessary. Nevertheless, it is very encouraging to see that inhibition of Rac-1 lowers the levels of IL-1β produced by HIDS PBMCs ex vivo, as this suggests a new avenue for treatment of HIDS that may be directly targeted to the disease mechanism (113).
In the aforementioned murine model (114), heterozygous knockout mice for the Mvk gene have a clear accumulation of mevalonic acid in several organs and significantly increased serum concentration of IgD. These mice also have a higher incidence of hepatomegaly (25%) and splenomegaly (33%) and a higher serum concentration of TNF-α. The involvement of IgD in the pathogenesis of HIDS is unlikely (115). Symptoms can occur in children some years before the elevation of serum IgD concentration is found, and IgD concentrations do not correlate with disease activity or severity. In addition, a number of mutation-positive patients have a normal concentration of IgD, and occasional patients with other recurrent fever syndromes have modestly elevated serum IgD (116).
There is not yet consensus in the literature about treatment of HIDS, although preliminary reports on the efficacy of anakinra are consistent with the involvement of IL-1β in this disease (117, 118). Furthermore, it seems that, especially for adult HIDS patients with infrequent and/or less severe attacks, anakinra need not be used prophylactically but instead can be administered at symptom onset to lessen the severity and duration of attacks. For this reason, and because of the new biochemical data linking MK to IL-1β, we feel comfortable discussing HIDS as an extrinsic inflammasomopathy. An argument could also be made to classify HIDS as a protein-folding disorder because the mutations in MK appear to affect protein folding (119). Culture of patient fibroblasts in circumstances that promote a more controlled protein folding increased the residual mevalonate kinase enzyme activity. Intercrossing Mvk heterozygous mice with ASC- and/or Nlrp3-deficient mice should confirm the role of the inflammasome in disease or suggest an alternative mechanism of disease pathogenesis.
Recurrent hydatidiform mole
Recurrent hydatidiform mole is a relatively rare condition that underscores the important connections between reproductive biology and innate immunity. In general, hydatidiform mole is a benign trophoblastic tumor presenting as a pregnancy with the absence of embryonic development and with cystic degeneration of chorionic villi (120). This condition may occur in as many as 1 in 1500 pregnancies, although recurrent hydatiform mole (RHM) comprises only about 2% of the total. RHM has a strong genetic contribution, and multiple cases within individual families have been described (120). Although there is biparental inheritance of RHM, DNA methylation at imprinted loci is abnormal, with either a defective maternal contribution or paternal-specific methylation patterns on maternally inherited alleles (121). Therefore it was somewhat surprising when the first genetic cause of RHM was identified as NLRP7 (NALP7, PYPAF3) (122), an innate immune gene without a known direct role in DNA methylation. Women with RHM are homozygous or compound heterozygous for NLRP7 mutations, whereas males homozygous for the same mutation have normal reproduction. The NLRP7 protein is similar in structure to NLRP3 and NLRP1, with an N-terminal pyrin domain, a NACHT/NBD motif, and a C-terminal LRR domain. NLRP7 is not known to function as NLRP1 and NLRP3, to activate IL-1β, but instead is thought to act as a negative regulator of IL-1β, perhaps induced by inflammatory cytokines as part of a negative feedback loop (123). NLRP7 was also found to be expressed in target organs such as uterus, endometrium, and ovary (122), and therefore the regulation of the involvement of inflammatory processes during pregnancy becomes the new hypothetical mechanism of disease, and DNA methylation differences may instead only be as a result of tissue inflammation.
The role of the innate and adaptive immune systems during pregnancy is a fascinating topic, as they control a very delicate equilibrium between fetal rejection and defense against infection. The balance appears to change during the course of pregnancy, with a predominance of pro- or antiinflammatory cytokines required at different stages (124). For brevity, we discuss only IL-1, although functional data implicate many other cytokines, chemokines, receptors, and complement molecules (125). Regarding IL-1, it has been shown that IL-1β induces preterm delivery in mice and that an intronic polymorphism in the IL-1 receptor antagonist (IL-1RN) may be associated with preterm birth of humans (126). The relevant IL-1RN polymorphism is also associated with increased IL-1β production in vitro (127), and thus one could hypothesize that inactivating mutations in NLRP7, a protein that negatively regulates IL-1β, could similarly shift the balance in favor of increased IL-1β activity in vivo. Perhaps this speculation is somewhat premature because the key biochemical experiments looking at IL-1β production in the presence of mutant NLRP7 molecules have not yet been reported, let alone the in vivo correlates of this in animal models. It is interesting that two NLRP7 mutations may preclude the possibility of normal pregnancy (128), whereas heterozygous mutations in females predispose to still birth and spontaneous abortions (129). The emerging phenotypic picture may be a complex interplay of NLRP7 genotypes with other genetic and environmental effects. Understanding how innate immunity, IL-1β, and NLRP7 are regulated to allow successful pregnancy is certain to advance over the next few years.
Complex/Acquired Inflammasomopathies
Gout/pseudogout
Gout and pseudogout are common rheumatic diseases caused by the respective deposition of monosodium urate (MSU) and calcium pyrophosphate dihydrate (CPPD) crystals in the joints and periarticular tissues, leading to acute and chronic inflammation. In the sense that metabolic perturbation provokes inflammatory activation, the crystal deposition diseases could be considered autoinflammatory (8), but the molecular details, particularly regarding the innate immune system, were at first lacking. Martinon and colleagues (18) subsequently made a key breakthrough by showing that NLRP3 plays a pivotal role in the inflammatory complications of MSU and CPPD crystal deposition (Figure 2). Both MSU and CPPD crystals increased caspase-1 activation and IL-1β secretion from macrophages stimulated with LPS, but this did not occur in ASC- or Nlrp3-deficient macrophages (18). Furthermore, the neutrophil influx generated in a mouse model of crystal-induced peritonitis was reduced in mice deficient for inflammasome components.
More evidence for a role of IL-1 in crystalline arthropathies comes from mice deficient in the adaptor protein MyD88, which are resistant to MSU-initiated gouty inflammation (130). MyD88 transduces both TLR and IL-1 receptor signaling, and although mice deficient for various TLRs still exhibit inflammation, those deficient for the IL-1β receptor do not, suggesting that IL-1 signaling specifically controls disease progression in this model. Bone marrow reconstitution experiments demonstrated that the IL-1 receptor was required in nonhematopoietic cells, but not hematopoietic cells, for acute gouty inflammation.
Small pilot studies of anakinra suggest that this research is translatable into the clinic (131, 132). While this supports the role of the IL-1β inflammasome in the crystalline arthropathies, IL-1 inhibition is unlikely to have the same impact in these illnesses as in CAPS, given the availability of alternative approaches. Lifestyle change and nonbiological agents such as colchicine will likely remain the mainstay of treatment. This is not to say that IL-1 inhibition will not become increasingly useful as the drugs targeting this pathway are improved for half-life, cost, or oral absorption. Even with currently available agents, randomized controlled studies may support the efficacy and cost-effectiveness of IL-1 inhibition in the crystalline arthropathies.
Fibrosing disorders
The fibrosing disorders are a heterogeneous group of conditions that include idiopathic pulmonary fibrosis, cryptogenic cirrhosis, retroperitoneal fibrosis, sclerosing cholangitis, and scleroderma. There are a few well-documented cases of fibrosing disease secondary to environmental insults, as was recently demonstrated for nephrogenic systemic sclerosis induced by the magnetic resonance contrast agent gadolinium (133). Genetic factors, such as mutations at the telomerase locus, have also been associated with rare familial cases of pulmonary fibrosis (134). While there are anecdotes and small series suggesting that the fibrosing disorders may be manifestations of a broader susceptibility to inflammation (135), until recently the details have remained elusive. However, recent data point to an important role for the inflammasome in two environmentally induced forms of pulmonary fibrosis.
Asbestos and silica can cause severe inflammatory and fibrotic disease when inhaled. Alveolar macrophages lavaged from individuals with prolonged asbestos exposure secrete enhanced basal amounts of IL-1β in vitro (136). Several labs have now reported that IL-1β production in response to these particles is abrogated in Nlrp3-deficient mice (19–21) (Figure 2). One paper further suggests that NLRP3-activating crystals/fibers share the ability to destabilize lysosomes (21). Similarly, phagocytosis of amyloid-β in the central nervous system may also lead to lysosomal destabilization, inflammasome activation, and IL-1β release (137). While the role of IL-1β in the pathogenesis of Alzheimer’s disease is still not conclusive, there is now a growing rationale for clinical trials targeting IL-1β in the fibrosing disorders caused by substance inhalation.
By analogy, it is tempting to hypothesize that idiopathic pulmonary fibrosis (IPF) is caused by a similar process, triggered perhaps by other inhalants or environmental exposures. Genetics appears to play an important role in susceptibility to IPF (138), with some studies suggesting an association with a polymorphism in the IL-1 receptor antagonist gene (139). In mice, direct administration of IL-1β by inhalation results in fibrotic inflammation that is comparable to that induced by bleomycin (140), a murine model for IPF. Bleomycin-induced pulmonary fibrosis is attenuated by genetic deletion of the IL-1 receptor and the inflammasome adaptor ASC genes, and by exogenously administered IL-1 receptor antagonist. Bearing in mind that IPF is likely a genetically complex disorder involving a number of inflammatory pathways, data from human and animal models suggest a pathogenic role for IL-1β and raise the possibility of pilot studies of IL-1 inhibitors in this life-threatening disease.
Type 2 diabetes mellitus
Diabetes mellitus is characterized by elevated fasting and postprandial blood sugar levels due to the relative or absolute deficiency of insulin. In type 1 diabetes (T1DM) there is strong evidence of pancreatic islet cell destruction, often early in life, eventually leading to marked reduction in insulin production. The large majority (type 1A) are caused by anti-islet cell autoantibodies (141). In contrast, type 2 diabetes mellitus (T2DM) tends to occur later in life and is due to the combination of peripheral insulin resistance and impaired islet cell insulin secretion, which are thought to be metabolic consequences of excessive weight (142). In addition, an emerging body of data suggests that the innate immune system plays an important role in the pathogenesis of T2DM.
T2DM was first proposed as a disease of the innate immune system over a decade ago (143). This work suggested that T2DM is associated with persistent elevation of acute-phase reactants and inflammatory mediators, and this has now been further refined to a subset of proinflammatory cytokines, particularly IL-1β and IL-6 (144). IL-1β is a key cytokine that induces the apoptosis of pancreatic beta cells. Hyperglycemia creates a vicious circle by inducing the production of IL-1β by the β cells themselves (145), thus leading to further islet cell dropout and ever-increasing blood glucose levels. In a mouse model of diabetes, administration of the IL-1 receptor antagonist protected animals fed a high-fat/high-sucrose diet from hyperglycemia and β cell apoptosis (146). Moreover, in a randomized, double-blind, placebo-controlled trial in 70 patients with T2DM, anakinra improved glycemia and β cell secretory function (147). Although to date variants in the genes encoding inflammasome proteins have not been associated with T2DM in whole-genome analyses, the biologic and clinical evidence places the inflammasome at a critical crossroads in the pathogenesis of T2DM. Particularly as longer-acting IL-1 inhibitors become available, this pathway could provide an important target in the treatment of this common but debilitating illness.
Schnitzler syndrome
Our discussion of Schnitzler syndrome as a disease mediated primarily by the innate immune system may still be premature. This illness is characteristically associated with monoclonal IgM gammopathy and sometimes bone pain and radiographic findings (148), which might indicate involvement of the B cell lineage, and the mean age of onset is 51 years. However, the remaining constellation of urticaria, intermittent fever, arthralgia or arthritis, and elevated acute-phase reactants is reminiscent of the hereditary periodic fever syndromes, and of the cryopyrinopathy MWS in particular. The biochemical data available to date do indeed suggest a prominent influence of proinflammatory cytokines from the innate immune system (149), and there are now several reports indicating that inhibiting IL-1β signaling is also very beneficial in the treatment of Schnitzler syndrome (149–153). Gammopathy may persist after anakinra despite remission of fever, rash, and constitutional symptoms (149), suggesting more proximal molecular events in the pathogenesis of Schnitzler syndrome.
NF-κB ACTIVATION DISORDERS
Crohn’s Disease (CD)
NOD2/CARD15 was placed center stage in the field of immunology when two simultaneous publications documented susceptibility alleles that predispose to CD (26, 27). CD and ulcerative colitis (UC) are the two most common forms of inflammatory bowel disease (IBD), but there are several clinical as well as genetic features that distinguish between them (163). In CD, inflammation is typically transmural and can be found discontinuously throughout the gut, whereas UC primarily affects the mucosal and submucosal layers of the rectum and colon in a continuous pattern. NOD2 appears to be a genetic discriminating factor as a locus that is only associated with CD (163–167). This is in contrast to single nucleotide polymorphisms (SNPs) in ECM1 (an intestinal glycoprotein that activates NF-κB) and IL-10 associated just with UC (165, 167), and to SNPs in IL-23R and IL-12B (encoding the p40 subunit of IL-12 and IL-23) that are associated with IBD in general (163–167). These genes highlight the involvement of both the innate and adaptive immune system with features of both autoinflammatory and autoimmune disease. Of note, the cellular distribution of NOD2 expression is almost exclusively within the myeloid lineage of innate immune cells, while the associations with IL-10, IL-12B, and IL-23R suggest an important pathogenic role for T cells. A unifying theory might posit that NOD2-associated SNPs cause inappropriate activation of innate immune myeloid cells to normal intestinal flora, thus triggering a response influenced by genes of the adaptive immune system. Alternatively, NOD2 may normally function as an innate signal to tolerize the host adaptive immune system, for which loss-of-function mutations may render the host sensitive to normal enteric bacteria.
The NOD2 protein bears some structural similarities to the NLRP3 protein (Figure 1). Whereas NLRP3 has a single N-terminal pyrin domain, NOD2 has two N-terminal CARDs, but both pyrin domains and CARDs belong to the same family of motifs, which assume a three-dimensional structure that permits cognate interactions (39–41). Through homotypic CARD interactions, NOD2 interacts with RICK/RIP2 to activate the NF-κB and mitogen-activated protein (MAP) kinase signaling pathways (168). Like NLRP3, NOD2 also has a NACHT/NBD domain with ATP-binding activity, and ten C-terminal LRRs through which it mediates, directly or indirectly, intracellular recognition of the bacterial cell wall component MDP (157, 158) (Figure 2). While CD mutations are found throughout NOD2, three mutations in the C-terminal third account for 80% of NOD2 CD variants (169).
The mechanism by which CD mutations in NOD2 cause IBD remains controversial. One major problem is that the effect of these mutations on innate immunity is highly dependent on the type of cells being assayed. Thus, whereas NF-κB activation is increased in the lamina propria of individuals with CD (170), PBMCs exhibit decreased MDP-induced activation (171, 172). Bone marrow–derived macrophages from mice harboring a NOD2 CD mutation exhibit increased NF-κB activity and IL-1β in response to MDP (173), consistent with the patient data from intestinal tissue. On the other hand, CD patients exhibited an impaired inflammatory response to topical application of IL-8, which was normalized by the addition of MDP, unless the patient was homozygous for a NOD2 CD variant (174).
Mice deficient in the murine Nod2 are susceptible to Listeria monocytogenes infection via the oral route and express reduced amounts of a subgroup of intestinal antimicrobial peptides known as cryptdins (175). More recent data from CD patients suggest that this latter effect is due to inflammation and is not a primary pathogenic event (176). However, neither the aforementioned mice nor another line deficient in Nod2 develop spontaneous IBD-like disease (175, 177). Data from yet another experimental model suggest that MDP activation of NOD2 downregulates multiple TLR responses and that the absence of such tolerization with CD mutations might increase susceptibility to intestinal inflammation (178). Alternatively, it has been suggested that the LRR of NLR proteins is autoinhibitory and that inactivating CD mutations would prevent autoinhibition and thus promote inflammation (Figure 2).
Another important consideration is that NOD2 is not the only innate immune gene associated with CD, and thus it is possible that animal model systems should take into account more of the >30 genetic factors that have been associated with disease susceptibility in humans (164). Several autophagy-related genes, such as ATG16L1, are strongly associated with CD, which further suggests a role for the presentation of bacterial components to intracellularly expressed proteins such as NOD2. Paneth cells from mice deficient in the murine Atg16L1 manifest abnormalities in granule exocytosis similar to those seen in CD patients (179) and exhibit heightened susceptibility to chemically induced colitis in an IL-1β-dependent fashion (180). Mice deficient in a second CD-associated autophagy gene, Irgm, have an impaired capacity to eliminate intracellular pathogens. Such data suggest a complex interaction between NOD2, various autophagy genes, and a number of other loci controlling innate and adaptive immune function to confer susceptibility to CD.
Blau Syndrome (BS)
Whereas certain variants in NOD2 are risk factors for CD, a different subset of NOD2 sequence substitutions are high-penetrance, dominantly inherited mutations that cause BS (28). BS is characterized by a triad of granulomatous uveitis, arthritis, and skin rash with camptodactyly (flexion contractures of the fingers). NOD2 mutations have also been found in early-onset sarcoidosis (181), an entity that some regard as the same disease as BS, and among patients with a somewhat broader phenotype than that typical for BS (182, 183). BS mutations almost exclusively target the NBD of the protein, whereas CD mutations predominantly affect its LRR (Figure 1). The central NBD of other NLR proteins is known to bind and hydrolyze ATP, and this is required for signaling. It has not yet been determined that NBD mutations in BS result in altered ATP processing, but it is known that basal NF-κB activation is upregulated (181). Mutations causing cryopyrinopathies also cluster around the NBD of NLRP3. Comparison of the predicted three-dimensional structure of NLRP3 with that of NOD2 indicates that the R260W mutation of NLRP3 and the BS-associated R334W mutation of NOD2 encode the same amino acid substitution at a homologous, structurally conserved residue (184).
It is intriguing why CD-associated NOD2 variants predominantly reside in or adjacent to the LRRs and induce gastrointestinal disease, while BS-associated NOD2 mutations occur mostly in the NBD/NACHT domain and produce a broader distribution of affected tissues. As is the case for NLRP3, NBD mutations in BS may produce a protein that is constitutively active. On the other hand, LRR mutations may alter thresholds for PAMP-induced signaling, which for NOD2 may occur mostly in the gastrointestinal tract, but would not lead to constitutive, ATP-independent activation.
Guadeloupe Variant Periodic Fever Syndrome (FCAS2)
We have provisionally placed a newly reported periodic fever syndrome, first described in two families from Guadeloupe (29), among the NF-κB activation disorders, although at first glance the clinical picture suggests an IL-1β-related disorder. Patients present with week-long episodic fevers triggered by cold exposure and associated with arthralgia, myalgia, and other constitutional symptoms. Two affected members of one family had sensorineural hearing loss. Although clinically similar to FCAS or MWS, mutational screening in NLRP3 and other known periodic fever genes was negative. A subsequent examination of NLRP12 (NALP12, PYPAF7, MONARCH-1), chosen because of its similarity to NLRP3 and expression in myelomonocytic cells, revealed dominantly inherited nonsense and splice-site mutations in the two families. These variants were not found in a panel of 104 ethnically matched control chromosomes, a relatively small number, but the functional case for their pathogenic role is fairly strong (see below). Based on the clinical similarities with FCAS, but unique genetics, this condition has also been termed FCAS2.
NLRP12 was the first example of an NLR protein capable of negatively regulating NF-κB activation (185). The mutations identified in NLRP12 affect the protein significantly, with the nonsense mutation truncating the protein within the NBD and the splice mutation deleting the C-terminal LRRs (29). This differs from the NLRP3 mutations in CAPS, which are predominantly missense mutations of the NBD. In overexpression systems, the NBD truncation substantially impacted normal NLRP12 inhibition of NF-κB activation, with the LRR mutation showing a less convincing effect (29). It is not known whether NLRP12 can participate in an inflammasome complex regulating IL-1β production.
PROTEIN FOLDING DISORDERS OF THE INNATE IMMUNE SYSTEM
TNF Receptor–Associated Periodic Syndrome (TRAPS)
Dominantly inherited heterozygous mutations in TNFRSF1A, encoding the TNF receptor 1 (TNFR1, also known as TNFRSF1A, p55, and CD120a) cause TRAPS (2). Patients experience recurrent, often prolonged fevers that can be accompanied by severe abdominal pain, pleurisy, arthritis, a migratory skin rash with underlying fasciitis, and/or periorbital edema (186, 187). Some TRAPS patients eventually develop systemic AA amyloidosis. In contrast to FMF, there is no ethnic predilection, and corticosteroids are effective for the acute attacks, whereas colchicine is not.
TRAPS-associated mutations exclusively affect the extracellular region of the receptor (Figure 1), and in many cases the cysteine residues that participate in disulfide bonds result in the most severe and penetrant disease phenotype with the highest risk of amyloidosis (188). In the initial description of TRAPS, we considered the possibility that interchain disul-fide binding of unpaired cysteine residues might lead to constitutive activation but rejected this hypothesis based on studies of cytokine production from patients’ PBMCs (2). Experiments examining the binding of radiolabeled TNF to leukocytes from patients and controls showed no increased TNF binding (2). An alternate hypothesis was that TNFR1 mutations might lead to a defect in the ability of metalloproteases to cleave TNFR1 from the cell membrane, also called receptor shedding, which is the most common way of inactivating TNFR1 (189). Despite the fact that most TRAPS-associated TNFR1 mutations are remote from the known cleavage site, initial observations, including increased cell surface TNFR1 and decreased soluble TNFR1 in patients’ blood, were consistent with impaired ectodomain cleavage (2). By this hypothesis, TRAPS-associated mutations would lead to impaired receptor shedding, thereby permitting repeated stimulation through membrane-bound receptors and a decreased pool of potentially inhibitory soluble receptors. Implicit in this hypothesis was the concept that cell-bound mutant receptors would signal normally and that TNF signaling would be the central pathogenic mechanism. However, not all TRAPS-associated TNFR1 mutations have an effect on shedding (188), and even for TNFR1 mutations where an effect on shedding has been demonstrated, this is confined to certain cell types (190).
Our understanding of how the TRAPS phenotype develops has recently been advanced by in vitro analysis of mutant receptors. Overexpression experiments in cell lines suggest that the mutations cause TNFR1 to aggregate and inhibit its trafficking to the cell membrane (30, 191, 192). The TNFR1 mutations also cause decreased binding of TNF (191). Lobito et al. demonstrated that the mutant receptors no longer oligomerized with wild-type receptors, instead forming disulfide-linked homo-oligomers (30). Furthermore, the mutant receptors were retained intracellularly where they colocalized with markers for the endoplasmic reticulum (ER), and their cell surface expression was also prevented in vivo in mice homozygous for a knockin of the T50M mutation (30). This finding is also in agreement with computational modeling studies that suggest improper folding of the mutated protein, leading to different conformations of the TNFR1 ectodomain (193), and with studies of neutrophils from C33Y TRAPS patients that show minimal surface staining for the mutant receptor (192).
These recent findings are a significant departure from our initial understanding of the disease. Undoubtedly, the structural changes induced by TRAPS mutations would interfere with recognition by proteases and thereby inhibit cleavage of whatever mutant receptor makes it to the cell surface. Moreover, there are rare patients with the TRAPS phenotype who do have mutations near the cleavage site at the transmembrane region whose leukocytes exhibit impaired receptor shedding (194, 195). However, the evidence for impaired trafficking of a number of mutant receptors adds an important new dimension to the discourse on the underlying cause of TRAPS.
From a theoretical view, there are at least two constraints on potential hypotheses regarding the mechanism of TRAPS. First, any proposal must explain how mutations that lead to a grossly misfolded protein, with impaired ligand binding, aberrant intracellular trafficking, and impaired aggregation with wild-type receptors, still lead to an autoinflammatory rather than an immunodeficiency phenotype. Second, any viable theory must explain the dominant mode of inheritance of TRAPS. Initially (when it was still assumed that mutant TNFR1 would reach the cell surface normally), the inheritance of TRAPS was explained by the fact that TNFR1 molecules must trimerize to signal, and that 7/8 possible trimers composed of mutant and wild-type receptors would contain at least one mutant receptor. However, in light of the recent data in transfection systems that mutant receptors seem not to associate with wild-type molecules (30), this explanation seems much less likely. Similarly, dominant-negative interference with apoptotic signaling through the wild-type TNFR1 would seem unlikely if the wild-type and mutant species do not interact. Instead, various gain-of-function hypotheses become more likely. Two such formulations that comport with the data include the notion that misfolded intracellular receptors might trigger ER stress and the UPR (196), or that intracellular aggregates of misfolded protein might constitutively induce intracellular signaling through aggregation of their death domains and subsequent recruitment of downstream signaling molecules. In contrast, certain other theories, such as the proposal that TNFR1 mutations might lead to shunting through the 75-kDa TNFR2, seem less likely because they do not explain the dominant inheritance of TRAPS.
Our recent data suggest that the accumulation of mutant TNFR1 results in spontaneously increased activation of MAPK such as JNK and p38 (Figure 4). This activation may prime TRAPS cells to become more susceptible to low doses of inflammatory stimuli such as LPS (A. Simon, H. Park, R. Maddipati, A. Bulua, A. Jackson, et al., submitted). Increased MAPK activation was mediated by reactive oxygen species (ROS), and could be blocked by inhibition of ROS. Further exploration of a mouse model of TRAPS showed that homozygous mutant mice, which lack the wild-type TNFR1 cell surface receptor, do not develop the full phenotype seen in heterozygous mice. We hypothesize that the wild-type cell surface TNFR1 is necessary for the autocrine and paracrine feedback loops of TNF-α (Figure 4), which in turn result in a marked increase in cytokine production.
Figure 4.
Mechanisms of autoinflammatory protein folding disorders. Left: TNF-receptor associated periodic syndrome (TRAPS). TNF-receptor type 1 (TNFR1) accumulates in the endoplasmic reticulum (ER) when mutated (indicated by asterisk). This accumulation leads to increased reactive oxygen species (ROS) activation, and subsequent MAPK phosphorylation ( JNK and p38), which makes the cells more susceptible to inflammatory stimuli. TRAPS patients, who are heterozygous for TNFR1 mutations, still carry the wild-type TNFR1, which is thought to play a role in propagating the inflammatory cascade. Right: spondyloarthropathies, ankylosing spondylitis. An HLA-B27 variant is strongly associated with ankylosing spondylitis and may accumulate in the ER, leading to an unfolded protein response. Weaker gene associations include ARTS1 (ER-associated aminopeptidase 1, ERAP1), which could affect the pathogenic presentation of antigenic peptide fragments or the cleavage of membrane bound receptors, and IL-23R/IL-1α, which argue for the role of proinflammatory cytokines in this disease.
Initially, treatment of TRAPS with the soluble p75:Fc fusion protein (etanercept) seemed to be the ideal substitute for a deficiency of soluble p55 TNFR1 that was implicated by the shedding hypothesis (186). It did not come as a surprise therefore that this treatment ameliorated disease in a large number of cases. However, as our understanding of TRAPS has become more sophisticated, with ligand-independent intracellular signaling taking on new prominence, the limitations of etanercept have become clearer, and there are even anecdotal reports of the efficacy of anakinra in this disease (197–199). This raises the possibility that cytokine inhibitors may work in TRAPS by mechanisms much less specific than originally envisaged. On the other hand, the fact that etanercept has provided at least some benefit to a number of patients is consistent with the hypothesis that wild-type TNFR1 is required to develop the disease phenotype.
Ankylosing Spondylitis
Ankylosing spondylitis (AS) is a systemic disease manifested by chronic arthritis of the spine and sacroiliac joints, leading eventually to the loss of spinal mobility. There are a number of extra-articular manifestations, including uveitis, aortitis, enthesitis, and dactylitis, and spinal fusion may lead to restrictive lung disease and consequent pulmonary hypertension. The association of ankylosing spondylitis susceptibility with the class I molecule HLA-B27 is one of the strongest known HLA disease associations and has been recognized for over 30 years (200). Initial theories of disease pathogenesis have revolved around the possible abnormal presentation of self or microbial peptides selectively by HLA-B27, but the identification of so-called arthritogenic peptides has remained elusive (201).
The inability to explain the association of HLA-B27 with AS on the basis of antigen presentation and models of adaptive immunity has led to consideration of alternative hypotheses. One such line of investigation is based on the tendency of HLA-B27 heavy chains to mis-fold in the ER and to form disulfide-linked heavy chain dimers (202, 203). In transgenic rats, disulfide-linked heavy chain complexes are much more prone to form in animals harboring the AS-associated B27 allele rather than the disease-resistant HLA-B7 allele (204). Moreover, bone marrow macrophages from B27, but not B7, rats exhibit evidence of the UPR, either during the inflammatory phase of illness or with cytokine stimulation in premorbid animals (31, 205). When stimulated with TLR ligands, bone marrow macrophages from HLA-B27 transgenic rats produce increased amounts of type I interferons (32) and possibly IL-23 (33). While these data have formed the basis for a new paradigm for understanding the pathogenesis of AS, considerable complexities remain in correlating the many subtypes of HLA-B27 with misfolding and disease susceptibility (206, 207).
Although the molecular mechanism may still require some refinement, genetic studies are providing additional insights. Candidate gene analyses have implicated the IL-1 cluster of genes on chromosome 2q13 as an important AS susceptibility locus (208). The genetic contribution to AS has recently been further clarified by whole-genome analysis that identified a variant ARTS1 allele as a risk factor for the disease (209). The ARTS1 protein (also known as ERAP1) has at least two immunologic functions, one related to adaptive immunity, the second more related to innate immunity. In the ER, ARTS trims peptides to optimal length for presentation to MHC class I molecules, while at the cell surface it cleaves receptors for the proinflammatory cytokines IL-1, IL-6, and TNF (Figure 4). Data from this large study also implicated the gene encoding the IL-23 receptor as an AS susceptibility locus. This is of especial note, given the aforementioned observations on IL-23 production by HLA-B27 transgenic rat macrophages, the already established role for IL-23R in susceptibility to CD (164) and psoriasis (210), and the emerging role for IL-23 in the differentiation of the Th17 subset of T lymphocytes (211). IL-23 may therefore provide yet another connection between innate and adaptive immunity in the pathogenesis of autoinflammatory disease.
COMPLEMENT DISORDERS
Hemolytic Uremic Syndrome
Hemolytic uremic syndrome (HUS) is characterized by Coombs-negative hemolytic anemia, thrombocytopenia, and renal failure. The underlying pathological process is thrombotic microangiopathy, with uncontrolled small-vessel blood clots leading to erythrocyte fragmentation, platelet consumption, and glomerular hypoperfusion (212). HUS is most commonly caused by a strain of Escherichia coli expressing Shiga toxin, and is characterized by diarrhea followed by the aforementioned triad of clinical findings (213). With the exception of diarrhea, these clinical features are also common to the atypical form of the disease (aHUS), which is an inherited condition for which the genetic causes are now becoming understood. At first deficiency of, and then simple mutations in, the complement factor H (CFH) gene was identified (214, 215), and it is now known that mutations in the gene encoding the membrane cofactor protein (MCP or CD46, which is in the same complement gene cluster as CFH on chromosome 1q32) (216, 217) and complement factor I (CFI) gene (218) also cause disease when mutated (Figure 5).
Figure 5.
Pathogenesis of the complement-mediated autoinflammatory disease atypical hemolytic uremic syndrome. Activating gain-of-function mutations in CFB increase C3bBb convertase stability and lead to permanent activation of the alternative pathway of complement. Autoantibodies against, and inactivating mutations within negative regulators of this process have also been described, such as for complement factor H (CFH) and membrane cofactor protein (MCP), which are cofactors for the inactivation of C3b by complement factor I (CFI). Asterisks denote proteins that carry mutations with known disease associations.
The third component of complement is a critical molecule at the convergence of the classical, alternative, and lectin pathways, and its activation is tightly regulated (212, 219). CFH binds C3b to prevent the formation of the C3bBb convertase complex that further activates C3, it accelerates the dissociation of Bb from the convertase complex, and it acts as a cofactor for CFI in degrading C3b (215). Over 100 CFH mutations have been identified in aHUS. MCP/CD46 is a transmembrane glycoprotein that acts as a membrane-bound cofactor for C3I-mediated degradation of C3b. CFH is particularly important in protecting structures such as the glomerulus, which lack MCP/CD46, from C3 activation (220). CFI is a serine protease that inactivates C3b. Unchecked C3 activation leads to the production of the anaphylatoxins C3a and C5a, which are chemotactic factors that elicit the influx of neutrophils and other inflammatory cells, and C3b, which acts as an opsonin (219). Neutrophil-mediated endothelial cell damage can, in turn, lead to the release of thrombin and the development of the microangiopathic cascade.
Age-Related Macular Degeneration
Complement factor H variants confer risk of developing age-related macular degeneration, a leading cause of blindness in developed nations (221–223). Homozygosity at one specific variant, the substitution of histidine for tyrosine at residue 402 (Y402H), may increase the risk of developing this condition late in life by as much as sevenfold. The hallmark of this disease is the presence of drusen (protein and cell debris containing immune and complement-associated molecules) in and around the macula, the area of the retina that confers central vision and maximal visual acuity. Recent studies have focused on delineating the mechanism by which CFH variants lead to the deposition of drusen and consequent macular degeneration. In mice, a very close approximation of the human disease can be generated by immunization with carboxyethylpyrrole, an oxidation fragment of docosahexaenoic acid that has been found attached to protein in the drusen and in the serum of people with AMD (age-related macular degeneration) (224). Thus one possible scenario is that oxidative damage in the outer retina where docosahexaenoic acid is abundant can perturb the complement system, and that in individuals with high-risk variants of CFH, this leads to the development of pathogenic drusen.
CYTOKINE SIGNALING DISORDERS
Cherubism
Cherubism is a dominantly inherited disorder characterized by bony swelling of the upper and lower jaws during childhood (12). Patients often develop the characteristic cherubic round face after which the disease was named. Radiographs show multiple, symmetrical radiolucent cystic bone lesions of the mandibula and maxilla. The pathologic process is one of inflammatory cyst formation and bone replacement with fibrous tissue composed of stromal cells and multi-nucleated osteoclast-like cells. Although jaw swelling regresses in puberty, cherubism is not only disfiguring during the active phase but causes significant permanent dental problems. A relatively recent addition to the family of autoinflammatory diseases, cherubism is caused by mutations in SH3BP2, which encodes an SH3-binding protein expressed in multinucleated osteoclasts and in stromal cells from cherubism lesions (34).
Recently, the development of a knockin mouse model has provided important insight into the molecular pathophysiology of cherubism (35, 225). In this study, Ueki and colleagues developed a mouse line in which the P416R SH3BP2 cherubism mutation was knocked in to the mouse ortholog. Somewhat unexpectedly, the heterozygous mice exhibited only minimal trabecular bone loss in the long bones, even though cherubism is dominantly inherited in humans. However, mice homozygous for the knockin mutation developed an inflammatory phenotype affecting not only the bone, but also the skin, muscle, liver, lung, and stomach. Bone lesions included trabecular bone loss and pits in the mandible, with increased osteoclasts and areas of fibrosis in the affected bone.
Bone marrow transplantation experiments demonstrated that disease could be transferred from mutant to irradiated wild-type mice with hematopoietic cells. In order to further characterize the cellular and molecular basis of disease in the knockin mice, animals were bred that were doubly homozygous for cherubism knockin and Rag1 deficiency (which lack functional lymphocytes) or for the knockin and Op-deficiency (which lack functional myeloid cells). Bone loss occurred in the Rag1-deficient mice, but not the Op-deficient animals, thus establishing a key role for cells of the myeloid lineage. Moreover, upon stimulation with macrophage colony-stimulating factor (M-CSF) and receptor activator of NF-κB ligand (RANKL), myeloid osteoclast progenitors from homozygous or heterozygous knockin mice formed unusually large osteoclasts and macrophages that secreted excessive TNF-α (Figure 6). The mechanism of increased responsiveness to M-CSF and RANKL appears to be increased ERK and Syk phosphorylation.
Figure 6.
Autoinflammatory disease caused by aberrant cytokine signaling. Osteoclast and macrophage differentiation is controlled by the cytokines M-CSF and RANKL. Dominantly inherited mutations in SH3BP2 alter the signals that normally come from these cytokines, which results in the differentiation of hyperactive osteoclasts and macrophages that can cause the autoinflammatory disease cherubism. Excess TNF-α produced by these hyperactive macrophages could also feed back through the stroma to increase M-CSF and RANKL levels, although this is still hypothetical.
In this model TNF-α plays an important role both as an effector and amplifier of bone resorption. This cytokine is a potent activator of osteoclasts, and has been implicated in the bone loss associated with a number of inflammatory arthritides. It is therefore likely that hyperre-sponsiveness to M-CSF and RANKL may set up a positive feedback loop mediated by TNF-α. In fact, the homozygous knockin mouse had elevated levels of TNF-α in the serum, and crossing this mouse to one deficient for TNF-α rescued it from disease, although bone marrow cells from the double mutant mice did maintain an enhanced response to M-CSF and RANKL.
The data from the SH3BP2 knockin mouse support the concept that the molecular pathophysiology of cherubism involves the aberrant response of myeloid progenitors to M-CSF and RANKL, which then induces a TNF-α-mediated vicious circle of osteoclast stimulation. There remain a number of unanswered questions, including the explanation for the difference in inheritance between human (autosomal dominant) and murine (autosomal recessive) disease, the mechanism by which the positive feedback loop is short-circuited at puberty, and the question of whether therapy with TNF inhibitors will ameliorate disease. Nevertheless, we regard cherubism as the prototype for a new category of autoinflammatory disease characterized by aberrant cytokine responsiveness.
MACROPHAGE ACTIVATION
Primary and Secondary Hemophagocytic Lymphohistiocytosis
Although triggered by the adaptive immune system, the major effector cell of both primary and secondary HLH (hemophagocytic lymphohistiocytosis) is the macrophage, an agent of innate immunity. Primary (familial) HLH is a potentially life-threatening disorder caused by mutations in several genes important to cytotoxic T cell and natural killer (NK) cell function (36, 37). Clinical manifestations include prolonged fever, splenomegaly, and cytopenias involving multiple lineages in the peripheral blood (erythrocytes, platelets, leukocytes). Although not required for the diagnosis, some patients have evidence of hemophagocytosis (macrophage-mediated phagocytosis of erythrocytes) in the bone marrow, spleen, lymph nodes, or cerebrospinal fluid (226). Other laboratory findings include hypofibrinogenemia (due to coagulopathy), hypertriglyceridemia, elevated serum ferritin, low or absent NK cell function, and elevated levels of soluble IL-2Rα. Because of the hypofibrinogenemia, the erythrocyte sedimentation rate is usually unexpectedly low relative to other inflammatory measures, such as the C-reactive protein.
Exacerbations of primary HLH are sometimes associated with viral infection, and current concepts on pathophysiology revolve around (a) the inability of cytotoxic cells to clear virally infected cells, leading to persistent antigen-driven activation of macrophages, and (b) cytokine-mediated differentiation and amplification of activated macrophages for which there are insufficient apoptotic signals to remove (226, 227). The resultant hypercytokinemia leads to the expansion and activation of a subset of scavenger macrophages expressing high levels of CD163, a scavenger receptor that binds hemoglobin-haptoglobin complexes (228) and that attaches hematopoietic cells to macrophages, facilitating hemophagocytosis (226). These cells, in turn, secrete ferritin, an acute-phase protein that binds free iron to prevent oxidative damage (229), and a number of proinflammatory cytokines (Figure 7).
Figure 7.
Molecular lesions that affect cytotoxic adaptive immune system cells lead to unregulated macrophage activation. Familial hemophagocytic lymphohistiocytosis (HLH) is caused by mutations in genes encoding proteins associated with vesicular transport and granule shedding, such as Munc13-4 (UNC13D), Perforin 1 (PERF1), and Syntaxin11 (STX11). The functions of NK and cytotoxic T cells are also controlled by several other genes that cause individual monogenic diseases when mutated (LYST, RAB27A, SH2D1A, AP3B1). Impairment of the normal efficacy of cytotoxic T cells for virally infected targets and upregulation of IFN-κ production leads to the compensatory development of a subset of scavenger macrophages that mediate the macrophage activation syndrome (MAS). They secrete proinflammatory cytokines, phagocytose erythrocytes, express the CD163 receptor for haptoglobin-hemoglobin complexes, and produce ferritin. Their production of IL-18 in particular could amplify the production of IFN-κ in a positive feedback loop. Secondary HLH is most commonly observed as a sequella of systemic-onset juvenile arthritis, for which variants of IL-6 and MIF are risk factors (double asterisks). A clinical hallmark of MAS is profound hyperferritinemia.
Several genes encoding proteins important in cytotoxic cell function have been implicated in recessively inherited familial HLH. These include PRF1, which encodes perforin, a soluble pore-forming protein that permeabilizes the target cell membrane (230); UNC13D, which encodes Munc13-4, a protein essential for cytolytic granule fusion with the cell membrane during degranulation (231); and STX11, encoding syntaxin 11, a member of the SNARE protein family that facilitates fusion in intracellular membrane trafficking (232).
HLH may also be seen sporadically in a number of immunodeficiency syndromes, usually caused by genes that impact the innate immune system through their roles in vesicular transport and granule shedding in cytotoxic T cells and NK cells (Figure 7). In Chediak-Higashi syndrome, mutations in LYST (CHS1) appear to have an effect on membrane fission or fusion events (233), while in Griscelli syndrome, RAB27A mutations lead to impaired function of a small Rab GTPase involved in exocytosis of cytotoxic vesicles (36). Hermansky-Pudlak syndrome type II, caused by mutations in AP3B1 (encoding a component of the AP-3 complex, involved in vesicle formation), may also manifest defects in cytotoxic T cell and NK activity leading to HLH (234), while in X-linked lymphoproliferative syndrome there are hemizygous mutations in SH2D1A that cause abnormal NK responses and NKT cell deficiency (226).
Secondary HLH is seen in childhood rheumatic diseases, most commonly SoJIA (227), and is often termed macrophage activation syndrome (MAS) under these circumstances. Clinical features of MAS are similar to those seen in primary HLH, including low NK cell function and reduced perforin expression in cytotoxic cells (235). Gene expression profiling in peripheral blood leukocytes suggests that a subset of untreated SoJIA patients may have some of the early features of MAS (228). Although gene variants associated with risk of primary HLH appear not to be susceptibility loci for SoJIA (236), in a panel of 18 patients with SoJIA/MAS, a genetic association with UNC13D was observed, first for biallelic mutations that have been observed in familial HLH, and also for a series of 12 SNPs that are likely to be inherited as an extended haplotype (237). Cytokine profiling consistently shows that IFN-κ is elevated in HLH, and for primary HLH it is predominantly made by malfunctioning CD8 and NK cells. In secondary HLH, the effect of IFN-κ could also be amplified by increased levels of IL-18 that have been observed (Figure 7). This scenario does reveal two contradictions: One is that there is no IFN-κ gene expression signature despite these differences in cytokine levels, and the other is that in vitro, IFN-κ skews toward inflammatory macrophage differentiation rather than the scavenger macrophage typically observed in MAS.
Taken together, these data suggest a fundamental relationship between primary HLH and MAS. In both, compensatory mechanisms elicited because of impaired cytolytic T cell and NK activity lead to runaway activation of macrophages, truly leading to a horror autoinflammaticus that, if not treated quickly and aggressively, can even lead to death.
Atherosclerosis
Cardiovascular disease has become the leading cause of death in the Western world, and is predominantly caused by atherosclerosis (localized arterial inflammation). Preceding an atherosclerotic lesion, lipid-laden macrophages and some T cells accumulate beneath the endothelium, and this nidus can go on to form the core of the plaque where characteristic foam cells (activated, lipid-laden macrophages) are found (238). The top of the plaque is a mix of smooth muscle cells and collagen-rich matrix. Between and around this, T cells, macrophages, dendritic cells, and mast cells infiltrate and can destabilize the lesion, leading to rupture, which accounts for 60%–70% of coronary thromboses (38). The contribution of inflammatory cytokines and chemokines to these processes (for example, TNF-α, MCP-1, IL-1, IL-6) cannot be understated, both in the generation and effect after rupture. Aside from innate immune cells, genes of the innate immune system are also implicated in atherosclerosis. This has been shown using an ApoE-deficient mouse that spontaneously develops atherosclerotic disease, which can be partially rescued by deletion of TLR2, TLR4, and MyD88 (239). In the same mouse model, overexpression or administration of LOX-1 or IL-18, respectively, dramatically worsens disease progression. These findings implicate TLR/C-type lectin activation and IL-1/IL-18 signaling pathways in atherosclerosis.
As a clear-cut inflammatory disease, further refinement of atherosclerosis as an autoinflammatory or autoimmune disease is not straightforward. Findings that argue the autoimmune point of view include a report that adoptively transferred CD4-positive T cells promote atherosclerotic disease in ApoE-deficient mice (240), and another report demonstrating that polymorphisms in the T cell cos-timulator TNFSF4 are associated with coronary atherosclerosis in man, and that mice with targeted mutations in the Tnfsf4 homolog had smaller atherosclerotic lesions than control mice when fed a high-fat diet (241). Also, although foam cells are characteristic of disease, it is unclear if they represent an innate immune response gone awry, or if they could actually be beneficial in disease. Mouse studies of macrophages lacking the scavenging receptors that mediate uptake of oxidized low-density lipoprotein (LDL) have conflicting results, some progressing, others ameliorating ApoE mediated disease (242, 243). Some of this difference might be accounted for by the observation that foam cells are still generated in the scavenger receptor–deficient mice, indicating that alternate mechanisms of LDL uptake may exist.
Given the numerous factors that may contribute to the formation of atheromatous lesions, it is unlikely that any one cytokine or pathway will predominate, and hence the judgment to group atherosclerosis among the syndromes of macrophage activation. Nevertheless, it is intriguing to consider whether at least some component of susceptibility would be mediated through the inflammasome and IL-1β. By this line of reasoning, the uncontrolled accumulation of cholesterol could potentially act as a danger signal to an innate immune cell. Several years ago, studies of IL-1 receptor antagonist-deficient mice demonstrated increased foam cell lesion area relative to controls when fed a high-cholesterol/cholate diet. Conversely, mice transgenic for the IL-1 receptor antagonist on an LDL receptor–deficient background were moderately protected from cholesterol/cholate lesions (244). A clinical trial is currently under way to assess the efficacy of anakinra, the recombinant IL-1 receptor antagonist, in controlling inflammatory markers in patients with coronary artery disease (245).
THE NEXT FRONTIER: AUTOINFLAMMATORY DISEASES OF UNKNOWN ETIOLOGY
As noted in the introduction, there remain a number of disorders that appear clearly to be autoinflammatory, but for which there are insufficient data to place them into one of the six categories described in this review. In this final section, we briefly discuss three of these illnesses, SoJIA, Behçet’s disease, and PFAPA. With the current burgeoning interest in autoinflammatory diseases, each of these illnesses is coming under considerable scrutiny.
SoJIA is a systemic disease of children that may present with a daily fever, a characteristic salmon-colored skin rash, anemia, and hepatosplenomegaly; eventually arthritis develops, but systemic symptoms may precede the onset of arthritis by weeks or months. Autoanti-bodies are generally not observed in SoJIA patients. Pioneering studies by Virginia Pascual and colleagues have demonstrated an important role for IL-1β in the pathogenesis of SoJIA (9). This group found that serum from SoJIA patients induces the transcription of innate immunity genes, including IL-1, in PBMCs from healthy donors, and that SoJIA PBMCs release large amounts of IL-1β when stimulated. They went on to show that nine patients refractory to other therapies responded to anakinra, and they have subsequently identified a gene expression profile characteristic of SoJIA (246). Nevertheless, a substantial number of SoJIA patients do not respond to anakinra treatment (247), and there are also data implicating other cytokines in the pathogenesis of SoJIA. For example, a SNP in the promoter region of the IL-6 gene has been associated with SoJIA (248), as well as a promoter haplotype in the gene encoding macrophage migration inhibitory factor (249). For these reasons we have resisted the temptation to group SoJIA with the inflammasomopathies for the time being, though the data on IL-1β are clearly encouraging.
Behçet’s disease (BD) is a genetically complex disorder that usually presents in adults rather than children, and is characterized by oral and genital ulcerations, uveitis, acneiform and papulopustular skin lesions, arthritis, and vasculitis involving both the arteries and veins (250). Some patients with BD also exhibit the phenomenon of pathergy, in which a pustule forms following a needle prick. Autoantibodies are generally not seen in patients with BD. BD is most common among populations spread along the historical Silk Road, which extended from the Mediterranean basin, through Turkey and central Asia, to Japan and Korea. Although BD is clearly not inherited as a mendelian trait, there is a strong familial tendency, and susceptibility has also been associated with the HLA-BW51 allele. The BD phenotype likely represents a complex interaction of multiple genes and the environment. Just as has been the case for AS and IBD, genome-wide association studies may yield important new insights into the important host factors and pathways that underlie this fascinating disorder.
PFAPA, the syndrome of periodic fever, aphthous stomatitis, pharyngitis, and cervical adenopathy, is the most common periodic fever syndrome seen in childhood (10). Important additional features include the remarkable clockwork periodicity of the attacks, the responsiveness of attacks to abortive doses of corticosteroids (although in some patients the episodes then recur more frequently), and the almost predictable remission that occurs in adolescence. Like BD, PFAPA is not inherited as a mendelian trait, but does exhibit some familial tendency.
At the molecular level, current data highlight a role for proinflammatory cytokines IL-1β, IL-6, and TNF, which were elevated in PFAPA patients even between attacks (251). In addition, the antiinflammatory cytokine IL-4 was decreased compared to controls at all the time-points measured, with a strong increase of IFN-κ during fever episodes. One unresolved question is whether PFAPA is triggered by a viral or other infectious agent; certainly children of this age are regularly encountering many foreign antigens, some of them for the first time (252). While the exact cellular determinants and pathogenic molecular mediators remain elusive, the clinical picture and cytokine profile of PFAPA outline a very complex, perhaps heterogeneous, but nevertheless autoinflammatory, disease.
CONCLUDING REMARKS
The autoinflammatory diseases are the consequence of natural variation in the innate immune system that is severe enough to cause illness, but not so severe as to be embryonic lethal. These disorders represent a selective sampling from the universe of what can go wrong, a reminder of the precarious equilibrium that defines human health, and a vindication of the nonredundant role of specific genes in human biology. Monogenic illnesses such as the cryopyrinopathies, caused by rare mutations conferring no apparent selective advantage, have profoundly informed our understanding of each of the six categories of autoinflammatory disease discussed in this review. Diseases such as FMF, in which disease-associated recessive variants appear to have been selected in certain geographic regions perhaps by endemic pathogens, promise a more nuanced understanding of gene-environment and gene-gene interactions. Genetically complex illnesses such as CD and the spondyloarthropathies have been associated with multiple common variants of innate immune genes and extend the reach of autoinflammation squarely into the realm of everyday experience.
Regrettably, chance and the inherent fallibility of DNA replication provide an inexhaustible font of new subjects for this science. During the course of the preparation of the present manuscript, we have found evidence for a new autoinflammatory disorder with severe skin and bone manifestations, caused by recessive loss-of-function mutations in the IL-1 receptor antagonist. This illness promises yet deeper insights into the role of IL-1 in human and the importance of its soluble inhibitor, the absence of which can be fatal. In other cases, rare recessive mutations and incompletely penetrant dominant mutations may underlie disorders that initially appear not to be genetic at all. In the era of genome-wide association studies and targeted biologics, it is now also possible not only to discover new etiologic pathways in the genetically complex diseases in a completely hypothesis-neutral fashion, but also to interrogate the role of one’s favorite mediator with exquisite specificity.
Table 1 also underscores the prominence of the inflammasome and its regulation in a large number of autoinflammatory diseases. It remains to be seen whether IL-1β occupies some special place in the firmament of cytokines, unique in an apparent ability to trigger innate immunity without major provocation of the adaptive immune system, or whether the prominence of IL-1β among the autoinflammatory diseases merely reflects the bias of increased vigilance. The latter possibility notwithstanding, the number of molecular stimuli for the NLRP3 inflammasome has become quite large, raising questions as to whether the LRR of NLRP3 could actually bind such a disparate group of partners. This has given rise to a formulation termed the guard hypothesis, whereby NLR proteins monitor homeostatic pathways that can be perturbed by individual PAMPs or DAMPs to trigger activity of the inflammasome. If true, the proteins comprising these homeostatic pathways would become prime suspects in the search for new autoinflammatory disease loci. The recently recognized molecular consequences of protein misfolding in innate immune cells may represent yet another opportunity for gene discovery in unexplained syndromes.
Another theme that emerges from our analysis of autoinflammatory disease is the opportunity for the innate immune system to make mischief in sites of immunologic privilege. The central nervous system, eye, and pregnant uterus are three such anatomic sites, yet each is a major site of involvement for NOMID (central nervous system, eye), age-related macular degeneration (eye), and recurrent hydatidiform mole (pregnant uterus). Possibly, the restraints that place these anatomic compartments off limits for the adaptive immune system do not similarly confine the cells or molecules of innate immunity.
As diseases of human beings, the autoinflammatory disorders are not merely guideposts to the innate immunome, but also opportunities to better the human condition. The use of IL-1 inhibitors in NOMID and DIRA (deficiency of the interleukin-1 receptor antagonist) represent triumphs of molecular medicine, but the application of similar therapies to conditions like T2DM and atherosclerosis may represent the next breakthrough, and the development of small molecule inhibitors, if effective, would be revolutionary. It is our hope that as the study of autoinflammatory disease moves into its second decade, we stand on the threshold of understanding and treating a broad spectrum of human afflictions, both rare and common.
INNATE IMMUNE GENES AS DETERMINANTS OF AUTOIMMUNE DISEASE.
Recent data implicate genes of the NLR and TLR families in the development of a number of diseases that more appropriately fit into the autoimmune (as opposed to the autoinflammatory) spectrum. One interesting example is the association of NLRP1 not only with vitiligo, but also with a range of other autoimmune diseases that affect the relatives of vitiligo probands (154). NLRP1 is highly expressed in T cells, but also in a range of innate immune cells such as monocytes and dendritic cells. In allogeneic stem cell transplantation, recipient variants in NLRP1, as well as donor variants at NLRP2 and NLRP3, have been shown to be important prognostic factors (155). Variants at another NLR gene, NOD2/CARD15, in both donor and recipient are associated with graft-versus-host disease and mortality (156). Although we have distinguished between the IL-1β activating role of the NLRP proteins, and the NF-κB regulatory effect of NOD2, both link pathogen-associated molecular patterns (PAMPs) with proinflammatory cytokine activation. NOD2 may directly sense an exogenous bacterial component like muramyl dipeptide (MDP) (157, 158), whereas NLRP1, NLRP2, and NLRP3 may respond to changes in homeostatic proteins due to danger signals from molecules such as uric acid that would be released with a variety of natural or iatrogenic insults. In contrast to this, TLR7 and TLR9 are activated directly by nucleic acids, and this activation is implicated in the pathogenesis of systemic lupus erythematosus (159–162). These receptors usually alert the innate immune system to viral RNA/DNA, but inappropriate presentation of self RNA/DNA may lead to autoimmune disease. Although our understanding of innate immune genes in autoimmune diseases is still rudimentary, these pathways are becoming increasingly attractive targets for therapeutic intervention.
SUMMARY POINTS.
Intrinsic inflammasomopathies: NLRP3 nucleates a complex including ASC and caspase-1 called the inflammasome that processes pro-IL-1β into a mature, active form. To date, mutations in only one component, NLRP3, have been identified, and they cause a spectrum of diseases known as the cryopyrinopathies (CAPS).
Extrinsic inflammasomopathies: These diseases are caused by mutations in proteins that regulate the production of IL-1β, or can gain the function to do so, but are not constituents of the inflammasome complex.
Complex/acquired inflammasomopathies: Multigenic or environmentally predisposed diseases result in increased secretion of IL-1β by the inflammasome.
NF-κB activation disorders: These diseases are mediated predominantly by improper regulation of NF-κB within the innate immune system.
Protein folding disorders of the innate immune system: Multiple mechanisms can lead from the buildup of misfolded proteins to the production of proinflammatory cytokines by innate immune cells.
Complement disorders: Complement was the first described arm of the innate immune system, and it is tightly regulated to prevent autoinflammatory diseases.
Cytokine signaling disorders: Cytokines control the differentiation and activation of innate immune cells, so perturbations at the cytokine receptor or of downstream signaling pathways can cause autoinflammatory disease.
Macrophage activation: Diseases have been identified in which the activation of macrophages occurs indirectly, perhaps as a result of defective adaptive immune cells or environmental and inflammatory pathways.
FUTURE ISSUES.
How exactly do mutations all clustered in the NACHT domain of NLRP3 activate the inflammasome and lead to a wide spectrum of inflammation as observed in CAPS patients?
What are the intermediate(s) and direct upstream activator(s) of NLRP3, given that NLRP3 is probably incapable of directly detecting all the known agents that stimulate its activity?
What is the role of pyrin in humans, pro- or antiinflammatory, and what is the effect of mutations that cause FMF?
What are other genes mutated in patients with monogenic autoinflammatory diseases, and what susceptibility alleles are yet to be found in patients with complex autoinflammatory diseases?
What are the DAMPs or PAMPs that activate the inflammasome in IPF, T2DM, atherosclerosis, and other complex inflammasomopathies?
Are IL-1 and IL-23, which participate in the differentiation of naive T cells into Th17 cells, present and perhaps pathogenic in autoinflammatory diseases that are caused by increased production of IL-1β or associated with polymorphisms in IL-23?
Does the NOD2 molecule normally tolerize or sensitize the host to bacteria and what is the effect of NOD2 mutations on this function?
Why does the activation of IL-1 in autoinflammatory disease not predispose patients to autoimmune disease, given that this pathway is thought to prime an adaptive immune response and account for the adjuvant effect of alum?
Is there cross talk between the different mechanisms causing autoinflammatory disease? We know that IL-1 plays a role beyond the bounds of the inflammasomopathies. Might ER stress likewise play a role in the other categories of disease such as T2DM and UC/IBD?
Acknowledgments
This work was supported by the Intramural Research Program of the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
- autoinflammatory
innate immune activation by endogenous pathways
- TRAPS
TNF receptor–associated periodic syndrome
- FMF
familial Mediterranean fever
- autoimmune
adaptive immune activation by endogenous pathways
- HIDS
hyperimmunoglobulinemia D with periodic fever syndrome
- crystalline arthropathies
diseases caused by uric acid or calcium pyrophosphate dihydrate crystal deposition
- fibrosing disorder
inflammatory disease that leads to fibrosis
- DAMP
danger-associated molecular patterns
- NLR
nucleotide-binding domain and leucine-rich repeat containing
- UPR
unfolded protein response
- CAPS
cryopyrin-associated periodic syndrome
- cryopyrinopathy
a spectrum of diseases caused by mutations in NLRP3 (cryopyrin)
- inflammasomopathy
diseases that result from increased levels of secreted IL-1β, either directly or indirectly
- Pyroptosome
macromolecular complex of ASC and pyrin molecules, of which there is only one per cell
- IPF
idiopathic pulmonary fibrosis
- T2DM
type 2 diabetes mellitus
- IBD
inflammatory bowel disease
- PAMP
pathogen-associated molecular pattern
- aHUS
atypical hemolytic uremic syndrome
- AMD
age-related macular degeneration
- HLH
hemophagocytic lymphohistiocytosis
- Scavenger macrophage
activated macrophage that scavenges haptoglobin-hemoglobin complexes via the CD163 receptor, phagocytoses erythrocytes, and secretes proinflammatory cytokines and ferritin
- MAS
macrophage activation syndrome
- foam cell
lipid-laden macrophage characteristically found in atherosclerotic plaques
- Guard hypothesis
mechanism of innate immune protection based on the detection of alterations in homeostatic parameters as a proxy for pathogenic insults
Footnotes
The U.S. Government has the right to retain a nonexclusive, royalty-free license in and to any copyright covering this paper.
DISCLOSURE STATEMENT
The authors are not aware of any affiliations, memberships, funding, or financial holdings that might be perceived as affecting the objectivity of this review.
Contributor Information
Seth L. Masters, Email: masterss@mail.nih.gov.
Anna Simon, Email: a.simon@aig.umcn.nl.
Ivona Aksentijevich, Email: aksentii@mail.nih.gov.
Daniel L. Kastner, Email: kastnerd@mail.nih.gov.
LITERATURE CITED
- 1.Ehrlich P. Studies in Immunity. London: Wiley; 1910. [Google Scholar]
- 2.McDermott MF, Aksentijevich I, Galon J, McDermott EM, Ogunkolade BW, et al. Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell. 1999;97:133–44. doi: 10.1016/s0092-8674(00)80721-7. Demonstrated that mutations in TNFR1 cause TRAPS and coined the phrase autoinflammatory. [DOI] [PubMed] [Google Scholar]
- 3.International FMF Consortium. Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. Int FMF Consort Cell. 1997;90:797–807. doi: 10.1016/s0092-8674(00)80539-5. [DOI] [PubMed] [Google Scholar]
- 4.French FMF Consortium. A candidate gene for familial Mediterranean fever. The French FMF Consort. Nat Genet. 1997;17:25–31. doi: 10.1038/ng0997-25. [DOI] [PubMed] [Google Scholar]
- 5.Houten SM, Kuis W, Duran M, de Koning TJ, van Royen-Kerkhof A, et al. Mutations in MVK, encoding mevalonate kinase, cause hyperimmunoglobulinaemia D and periodic fever syndrome. Nat Genet. 1999;22:175–77. doi: 10.1038/9691. [DOI] [PubMed] [Google Scholar]
- 6.Drenth JP, Cuisset L, Grateau G, Vasseur C, van de Velde-Visser SD, et al. Mutations in the gene encoding mevalonate kinase cause hyper-IgD and periodic fever syndrome. International Hyper-IgD Study Group. Nat Genet. 1999;22:178–81. doi: 10.1038/9696. [DOI] [PubMed] [Google Scholar]
- 7.Hoffman HM, Mueller JL, Broide DH, Wanderer AA, Kolodner RD. Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle-Wells syndrome. Nat Genet. 2001;29:301–5. doi: 10.1038/ng756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Galon J, Aksentijevich I, McDermott MF, O’Shea JJ, Kastner DL. TNFRSF1A mutations and autoinflammatory syndromes. Curr Opin Immunol. 2000;12:479–86. doi: 10.1016/s0952-7915(00)00124-2. [DOI] [PubMed] [Google Scholar]
- 9.Pascual V, Allantaz F, Arce E, Punaro M, Banchereau J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J Exp Med. 2005;201:1479–86. doi: 10.1084/jem.20050473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thomas KT, Feder HM, Jr, Lawton AR, Edwards KM. Periodic fever syndrome in children. J Pediatr. 1999;135:15–21. doi: 10.1016/s0022-3476(99)70321-5. [DOI] [PubMed] [Google Scholar]
- 11.Shoham NG, Centola M, Mansfield E, Hull KM, Wood G, et al. Pyrin binds the PSTPIP1/CD2BP1 protein, defining familial Mediterranean fever and PAPA syndrome as disorders in the same pathway. Proc Natl Acad Sci USA. 2003;100:13501–6. doi: 10.1073/pnas.2135380100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferguson PJ, El-Shanti HI. Autoinflammatory bone disorders. Curr Opin Rheumatol. 2007;19:492–98. doi: 10.1097/BOR.0b013e32825f5492. [DOI] [PubMed] [Google Scholar]
- 13.Medzhitov R, Janeway CA., Jr Innate immunity: the virtues of a nonclonal system of recognition. Cell. 1997;91:295–98. doi: 10.1016/s0092-8674(00)80412-2. [DOI] [PubMed] [Google Scholar]
- 14.Feldmann J, Prieur AM, Quartier P, Berquin P, Certain S, et al. Chronic infantile neurological cutaneous and articular syndrome is caused by mutations in CIAS1, a gene highly expressed in polymorphonuclear cells and chondrocytes. Am J Hum Genet. 2002;71:198–203. doi: 10.1086/341357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aksentijevich I, Nowak M, Mallah M, Chae JJ, Watford WT, et al. De novo CIAS1 mutations, cytokine activation, and evidence for genetic heterogeneity in patients with neonatal-onset multisystem inflammatory disease (NOMID): a new member of the expanding family of pyrin-associated autoinflammatory diseases. Arthritis Rheum. 2002;46:3340–48. doi: 10.1002/art.10688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Agostini L, Martinon F, Burns K, McDermott MF, Hawkins PN, Tschopp J. NALP3 forms an IL-1β-processing inflammasome with increased activity in Muckle-Wells autoinflammatory disorder. Immunity. 2004;20:319–25. doi: 10.1016/s1074-7613(04)00046-9. [DOI] [PubMed] [Google Scholar]
- 17.Chae JJ, Komarow HD, Cheng J, Wood G, Raben N, et al. Targeted disruption of pyrin, the FMF protein, causes heightened sensitivity to endotoxin and a defect in macrophage apoptosis. Mol Cell. 2003;11:591–604. doi: 10.1016/s1097-2765(03)00056-x. [DOI] [PubMed] [Google Scholar]
- 18.Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–41. doi: 10.1038/nature04516. [DOI] [PubMed] [Google Scholar]
- 19.Dostert C, Petrilli V, Van Bruggen R, Steele C, Mossman BT, Tschopp J. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science. 2008;320:674–77. doi: 10.1126/science.1156995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cassel SL, Eisenbarth SC, Iyer SS, Sadler JJ, Colegio OR, et al. The Nalp3 inflammasome is essential for the development of silicosis. Proc Natl Acad Sci USA. 2008;105:9035–40. doi: 10.1073/pnas.0803933105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hornung V, Bauernfeind F, Halle A, Samstad EO, Kono H, et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat Immunol. 2008;9:847–56. doi: 10.1038/ni.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tschopp J, Martinon F, Burns K. NALPs: a novel protein family involved in inflammation. Nat Rev Mol Cell Biol. 2003;4:95–104. doi: 10.1038/nrm1019. [DOI] [PubMed] [Google Scholar]
- 23.Martinon F, Tschopp J. NLRs join TLRs as innate sensors of pathogens. Trends Immunol. 2005;26:447–54. doi: 10.1016/j.it.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 24.Ting JP, Lovering RC, Alnemri ES, Bertin J, Boss JM, et al. The NLR gene family: a standard nomenclature. Immunity. 2008;28:285–87. doi: 10.1016/j.immuni.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McGonagle D, McDermott MF. A proposed classification of the immunological diseases. PLoS Med. 2006;3:e297. doi: 10.1371/journal.pmed.0030297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hugot JP, Chamaillard M, Zouali H, Lesage S, Cezard JP, et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature. 2001;411:599–603. doi: 10.1038/35079107. Discovery that variants of NOD2 are associated with CD and dominantly inherited in BS. [DOI] [PubMed] [Google Scholar]
- 27.Ogura Y, Bonen DK, Inohara N, Nicolae DL, Chen FF, et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn’s disease. Nature. 2001;411:603–6. doi: 10.1038/35079114. Discovery that variants of NOD2 are associated with CD and dominantly inherited in BS. [DOI] [PubMed] [Google Scholar]
- 28.Miceli-Richard C, Lesage S, Rybojad M, Prieur AM, Manouvrier-Hanu S, et al. CARD15 mutations in Blau syndrome. Nat Genet. 2001;29:19–20. doi: 10.1038/ng720. Discovery that variants of NOD2 are associated with CD and dominantly inherited in BS. [DOI] [PubMed] [Google Scholar]
- 29.Jeru I, Duquesnoy P, Fernandes-Alnemri T, Cochet E, Yu JW, et al. Mutations in NALP12 cause hereditary periodic fever syndromes. Proc Natl Acad Sci USA. 2008;105:1614–19. doi: 10.1073/pnas.0708616105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lobito AA, Kimberley FC, Muppidi JR, Komarow H, Jackson AJ, et al. Abnormal disulfide-linked oligomerization results in ER retention and altered signaling by TNFR1 mutants in TNFR1-associated periodic fever syndrome (TRAPS) Blood. 2006;108:1320–27. doi: 10.1182/blood-2005-11-006783. Suggested that an alternative mechanism for the pathogenesis of TRAPS mutations in TNFR1 would exist. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Turner MJ, Delay ML, Bai S, Klenk E, Colbert RA. HLA-B27 up-regulation causes accumulation of misfolded heavy chains and correlates with the magnitude of the unfolded protein response in transgenic rats: Implications for the pathogenesis of spondylarthritis-like disease. Arthritis Rheum. 2007;56:215–23. doi: 10.1002/art.22295. [DOI] [PubMed] [Google Scholar]
- 32.Smith JA, Turner MJ, DeLay ML, Klenk EI, Sowders DP, Colbert RA. Endoplasmic reticulum stress and the unfolded protein response are linked to synergistic IFN-β induction via X-box binding protein 1. Eur J Immunol. 2008;38:1194–203. doi: 10.1002/eji.200737882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Layh-Schmitt G, Colbert RA. The interleukin-23/interleukin-17 axis in spondyloarthritis. Curr Opin Rheumatol. 2008;20:392–97. doi: 10.1097/BOR.0b013e328303204b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ueki Y, Tiziani V, Santanna C, Fukai N, Maulik C, et al. Mutations in the gene encoding c-Abl-binding protein SH3BP2 cause cherubism. Nat Genet. 2001;28:125–26. doi: 10.1038/88832. [DOI] [PubMed] [Google Scholar]
- 35.Ueki Y, Lin CY, Senoo M, Ebihara T, Agata N, et al. Increased myeloid cell responses to M-CSF and RANKL cause bone loss and inflammation in SH3BP2 “cherubism” mice. Cell. 2007;128:71–83. doi: 10.1016/j.cell.2006.10.047. Mice with cherubism mutations in SH3BP2 were made and found to differentiate hyperactive macrophages and osteoclasts in response to normal RankL and M-CSF stimulation. [DOI] [PubMed] [Google Scholar]
- 36.Zur Stadt U, Beutel K, Kolberg S, Schneppenheim R, Kabisch H, et al. Mutation spectrum in children with primary hemophagocytic lymphohistiocytosis: molecular and functional analyses of PRF1, UNC13D, STX11, and RAB27A. Hum Mutat. 2006;27:62–68. doi: 10.1002/humu.20274. [DOI] [PubMed] [Google Scholar]
- 37.Janka GE. Familial and acquired hemophagocytic lymphohistiocytosis. Eur J Pediatr. 2007;166:95–109. doi: 10.1007/s00431-006-0258-1. [DOI] [PubMed] [Google Scholar]
- 38.Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–95. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 39.Bertin J, DiStefano PS. The PYRIN domain: a novel motif found in apoptosis and inflammation proteins. Cell Death Differ. 2000;7:1273–74. doi: 10.1038/sj.cdd.4400774. [DOI] [PubMed] [Google Scholar]
- 40.Martinon F, Hofmann K, Tschopp J. The pyrin domain: a possible member of the death domain-fold family implicated in apoptosis and inflammation. Curr Biol. 2001;11:R118–20. doi: 10.1016/s0960-9822(01)00056-2. [DOI] [PubMed] [Google Scholar]
- 41.Fairbrother WJ, Gordon NC, Humke EW, O’Rourke KM, Starovasnik MA, et al. The PYRIN domain: a member of the death domain-fold superfamily. Protein Sci. 2001;10:1911–18. doi: 10.1110/ps.13801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol Cell. 2002;10:417–26. doi: 10.1016/s1097-2765(02)00599-3. First description of the macromolecular inflammasome complex. [DOI] [PubMed] [Google Scholar]
- 43.Keller M, Rüegg A, Werner S, Beer H-D. Active caspase-1 is a regulator of unconventional protein secretion. Cell. 2008;132:818–31. doi: 10.1016/j.cell.2007.12.040. [DOI] [PubMed] [Google Scholar]
- 44.Faustin B, Lartigue L, Bruey JM, Luciano F, Sergienko E, et al. Reconstituted NALP1 inflammasome reveals two-step mechanism of caspase-1 activation. Mol Cell. 2007;25:713–24. doi: 10.1016/j.molcel.2007.01.032. [DOI] [PubMed] [Google Scholar]
- 45.Duncan JA, Bergstralh DT, Wang Y, Willingham SB, Ye Z, Zimmermann AG, Ting JP. Cryopyrin/NALP3 binds ATP/dATP, is an ATPase, and requires ATP binding to mediate inflammatory signaling. Proc Natl Acad Sci USA. 2007;104:8041–46. doi: 10.1073/pnas.0611496104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Aksentijevich I, CDP, Remmers EF, Mueller JL, Le J, et al. The clinical continuum of cryopyrinopathies: novel CIAS1 mutations in North American patients and a new cryopyrin model. Arthritis Rheum. 2007;56:1273–85. doi: 10.1002/art.22491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gattorno M, Tassi S, Carta S, Delfino L, Ferlito F, et al. Pattern of interleukin-1β secretion in response to lipopolysaccharide and ATP before and after interleukin-1 blockade in patients with CIAS1 mutations. Arthritis Rheum. 2007;56:3138–48. doi: 10.1002/art.22842. [DOI] [PubMed] [Google Scholar]
- 48.Ferrari D, Pizzirani C, Adinolfi E, Lemoli RM, Curti A, et al. The P2X7 receptor: a key player in IL-1 processing and release. J Immunol. 2006;176:3877–83. doi: 10.4049/jimmunol.176.7.3877. [DOI] [PubMed] [Google Scholar]
- 49.Pelegrin P, Barroso-Gutierrez C, Surprenant A. P2X7 receptor differentially couples to distinct release pathways for IL-1β in mouse macrophage. J Immunol. 2008;180:7147–57. doi: 10.4049/jimmunol.180.11.7147. [DOI] [PubMed] [Google Scholar]
- 50.Martinon F, Mayor A, Tschopp J. The inflammasomes: guardians of the body. Annu Rev Immunol. 2009;27:229–65. doi: 10.1146/annurev.immunol.021908.132715. [DOI] [PubMed] [Google Scholar]
- 51.Mariathasan S, Weiss DS, Newton K, McBride J, O’Rourke K, et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature. 2006;440:228–32. doi: 10.1038/nature04515. [DOI] [PubMed] [Google Scholar]
- 52.Sutterwala FS, Ogura Y, Szczepanik M, Lara-Tejero M, Lichtenberger GS, et al. Critical role for NALP3/CIAS1/Cryopyrin in innate and adaptive immunity through its regulation of caspase-1. Immunity. 2006;24:317–27. doi: 10.1016/j.immuni.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 53.Kanneganti TD, Ozoren N, Body-Malapel M, Amer A, Park JH, et al. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature. 2006;440:233–36. doi: 10.1038/nature04517. [DOI] [PubMed] [Google Scholar]
- 54.Kanneganti TD, Body-Malapel M, Amer A, Park JH, Whitfield J, et al. Critical role for Cryopyrin/Nalp3 in activation of caspase-1 in response to viral infection and double-stranded RNA. J Biol Chem. 2006;281:36560–68. doi: 10.1074/jbc.M607594200. [DOI] [PubMed] [Google Scholar]
- 55.Edele F, Esser PR, Lass C, Laszczyk MN, Oswald E, et al. Innate and adaptive immune responses in allergic contact dermatitis and autoimmune skin diseases. Inflamm Allergy Drug Targets. 2007;6:236–44. doi: 10.2174/187152807783334292. [DOI] [PubMed] [Google Scholar]
- 56.Willingham SB, Bergstralh DT, O’Connor W, Morrison AC, Taxman DJ, et al. Microbial pathogen-induced necrotic cell death mediated by the inflammasome components CIAS1/cryopyrin/NLRP3 and ASC. Cell Host Microbe. 2007;2:147–59. doi: 10.1016/j.chom.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fujisawa A, Kambe N, Saito M, Nishikomori R, Tanizaki H, et al. Disease-associated mutations in CIAS1 induce cathepsin B-dependent rapid cell death of human THP-1 monocytic cells. Blood. 2007;109:2903–11. doi: 10.1182/blood-2006-07-033597. [DOI] [PubMed] [Google Scholar]
- 58.Saito M, Nishikomori R, Kambe N, Fujisawa A, Tanizaki H, et al. Disease-associated CIAS1 mutations induce monocyte death, revealing low-level mosaicism in mutation-negative cryopyrin-associated periodic syndrome patients. Blood. 2008;111:2132–41. doi: 10.1182/blood-2007-06-094201. [DOI] [PubMed] [Google Scholar]
- 59.Saito M, Fujisawa A, Nishikomori R, Kambe N, Nakata-Hizume M, et al. Somatic mosaicism of CIAS1 in a patient with chronic infantile neurologic, cutaneous, articular syndrome. Arthritis Rheum. 2005;52:3579–85. doi: 10.1002/art.21404. [DOI] [PubMed] [Google Scholar]
- 60.Goldbach-Mansky R, Dailey NJ, Canna SW, Gelabert A, Jones J, et al. Neonatal-onset multisystem inflammatory disease responsive to interleukin-1β inhibition. N Engl J Med. 2006;355:581–92. doi: 10.1056/NEJMoa055137. Shows that NOMID is remarkably well treated with IL-1 blockade. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Goldbach-Mansky R, Shroff SD, Wilson M, Snyder C, Plehn S, et al. A pilot study to evaluate the safety and efficacy of the long-acting interleukin-1 inhibitor rilonacept (interleukin-1 Trap) in patients with familial cold autoinflammatory syndrome. Arthritis Rheum. 2008;58:2432–42. doi: 10.1002/art.23620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hoffman HM, Throne ML, Amar NJ, Sebai M, Kivitz AJ, et al. Efficacy and safety of rilonacept (interleukin-1 Trap) in patients with cryopyrin-associated periodic syndromes: results from two sequential placebo-controlled studies. Arthritis Rheum. 2008;58:2443–52. doi: 10.1002/art.23687. [DOI] [PubMed] [Google Scholar]
- 63.Alten R, Gram H, Joosten LA, Van Den Berg WB, Sieper J, et al. The human anti-IL-1β monoclonal antibody ACZ885 is effective in joint inflammation models in mice and in a proof-of-concept study in patients with rheumatoid arthritis. Arthritis Res Ther. 2008;10:R67. doi: 10.1186/ar2438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Pras E, Aksentijevich I, Gruberg L, Balow JE, Jr, Prosen L, et al. Mapping of a gene causing familial Mediterranean fever to the short arm of chromosome 16. N Engl J Med. 1992;326:1509–13. doi: 10.1056/NEJM199206043262301. [DOI] [PubMed] [Google Scholar]
- 65.Kastner DL. Familial Mediterranean fever: the genetics of inflammation. Hosp Pract. 1998;33:131–34. 9–40, 43–46. doi: 10.3810/hp.1998.04.90. passim. [DOI] [PubMed] [Google Scholar]
- 66.Lachmann HJ, Sengul B, Yavuzsen TU, Booth DR, Booth SE, et al. Clinical and subclinical inflammation in patients with familial Mediterranean fever and in heterozygous carriers of MEFV mutations. Rheumatology. 2006;45:746–50. doi: 10.1093/rheumatology/kei279. [DOI] [PubMed] [Google Scholar]
- 67.Kalyoncu M, Acar BC, Cakar N, Bakkaloglu A, Ozturk S, et al. Are carriers for MEFV mutations “healthy”? Clin Exp Rheumatol. 2006;24:S120–22. [PubMed] [Google Scholar]
- 68.Booth DR, Gillmore JD, Lachmann HJ, Booth SE, Bybee A, et al. The genetic basis of autosomal dominant familial Mediterranean fever. QJM. 2000;93:217–21. doi: 10.1093/qjmed/93.4.217. [DOI] [PubMed] [Google Scholar]
- 69.Touitou I, Lesage S, McDermott M, Cuisset L, Hoffman H, et al. Infevers: an evolving mutation database for autoinflammatory syndromes. Hum Mutat. 2004;24:194–98. doi: 10.1002/humu.20080. [DOI] [PubMed] [Google Scholar]
- 70.Milhavet F, Cuisset L, Hoffman HM, Slim R, El-Shanti H, et al. The infevers autoinflammatory mutation online registry: update with new genes and functions. Hum Mutat. 2008;29:803–8. doi: 10.1002/humu.20720. [DOI] [PubMed] [Google Scholar]
- 71.Nisole S, Stoye JP, Saib A. TRIM family proteins: retroviral restriction and antiviral defence. Nat Rev Microbiol. 2005;3:799–808. doi: 10.1038/nrmicro1248. [DOI] [PubMed] [Google Scholar]
- 72.Centola M, Aksentijevich I, Kastner DL. The hereditary periodic fever syndromes: molecular analysis of a new family of inflammatory diseases. Hum Mol Genet. 1998;7:1581–88. doi: 10.1093/hmg/7.10.1581. [DOI] [PubMed] [Google Scholar]
- 73.Diaz A, Hu C, Kastner DL, Schaner P, Reginato AM, et al. Lipopolysaccharide-induced expression of multiple alternatively spliced MEFV transcripts in human synovial fibroblasts: a prominent splice isoform lacks the C-terminal domain that is highly mutated in familial Mediterranean fever. Arthritis Rheum. 2004;50:3679–89. doi: 10.1002/art.20600. [DOI] [PubMed] [Google Scholar]
- 74.Chae JJ, Wood G, Richard K, Jaffe H, Colburn NT, et al. The familial Mediterranean fever protein, pyrin, is cleaved by caspase-1 and activates NF-κB through its N-terminal fragment. Blood. 2008;112:1794–803. doi: 10.1182/blood-2008-01-134932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mansfield E, Chae JJ, Komarow HD, Brotz TM, Frucht DM, et al. The familial Mediterranean fever protein, pyrin, associates with microtubules and colocalizes with actin filaments. Blood. 2001;98:851–59. doi: 10.1182/blood.v98.3.851. [DOI] [PubMed] [Google Scholar]
- 76.Goldfinger SE. Colchicine for familial Mediterranean fever. N Engl J Med. 1972;287:1302. doi: 10.1056/NEJM197212212872514. [DOI] [PubMed] [Google Scholar]
- 77.Richards N, Schaner P, Diaz A, Stuckey J, Shelden E, et al. Interaction between pyrin and the apoptotic speck protein (ASC) modulates ASC-induced apoptosis. J Biol Chem. 2001;276:39320–29. doi: 10.1074/jbc.M104730200. [DOI] [PubMed] [Google Scholar]
- 78.Martinon F, Tschopp J. Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell. 2004;117:561–74. doi: 10.1016/j.cell.2004.05.004. [DOI] [PubMed] [Google Scholar]
- 79.Yu JW, Wu J, Zhang Z, Datta P, Ibrahimi I, et al. Cryopyrin and pyrin activate caspase-1, but not NF-κB, via ASC oligomerization. Cell Death Differ. 2006;13:236–49. doi: 10.1038/sj.cdd.4401734. [DOI] [PubMed] [Google Scholar]
- 80.Chae JJ, Wood G, Masters SL, Richard K, Park G, et al. The B30.2 domain of pyrin, the familial Mediterranean fever protein, interacts directly with caspase-1 to modulate IL-1β production. Proc Natl Acad Sci USA. 2006;103:9982–87. doi: 10.1073/pnas.0602081103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Papin S, Cuenin S, Agostini L, Martinon F, Werner S, et al. The SPRY domain of Pyrin, mutated in familial Mediterranean fever patients, interacts with inflammasome components and inhibits proIL-1β processing. Cell Death Differ. 2007;14:1457–66. doi: 10.1038/sj.cdd.4402142. [DOI] [PubMed] [Google Scholar]
- 82.Seshadri S, Duncan MD, Hart JM, Gavrilin MA, Wewers MD. Pyrin levels in human monocytes and monocyte-derived macrophages regulate IL-1β processing and release. J Immunol. 2007;179:1274–81. doi: 10.4049/jimmunol.179.2.1274. [DOI] [PubMed] [Google Scholar]
- 83.Hall MW, Gavrilin MA, Knatz NL, Duncan MD, Fernandez SA, Wewers MD. Monocyte mRNA phenotype and adverse outcomes from pediatric multiple organ dysfunction syndrome. Pediatr Res. 2007;62:597–603. doi: 10.1203/PDR.0b013e3181559774. [DOI] [PubMed] [Google Scholar]
- 84.Ustek D, Ekmekci CG, Selcukbiricik F, Cakiris A, Oku B, et al. Association between reduced levels of MEFV messenger RNA in peripheral blood leukocytes and acute inflammation. Arthritis Rheum. 2007;56:345–50. doi: 10.1002/art.22320. [DOI] [PubMed] [Google Scholar]
- 85.Belkhir R, Moulonguet-Doleris L, Hachulla E, Prinseau J, Baglin A, Hanslik T. Treatment of familial Mediterranean fever with anakinra. Ann Intern Med. 2007;146:825–26. doi: 10.7326/0003-4819-146-11-200706050-00023. [DOI] [PubMed] [Google Scholar]
- 86.Calligaris L, Marchetti F, Tommasini A, Ventura A. The efficacy of anakinra in an adolescent with colchicine-resistant familial Mediterranean fever. Eur J Pediatr. 2008;167:695–96. doi: 10.1007/s00431-007-0547-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gattringer R, Lagler H, Gattringer KB, Knapp S, Burgmann H, et al. Anakinra in two adolescent female patients suffering from colchicine-resistant familial Mediterranean fever: effective but risky. Eur J Clin Invest. 2007;37:912–14. doi: 10.1111/j.1365-2362.2007.01868.x. [DOI] [PubMed] [Google Scholar]
- 88.Kuijk LM, Govers AM, Frenkel J, Hofhuis WJ. Effective treatment of a colchicine-resistant familial Mediterranean fever patient with anakinra. Ann Rheum Dis. 2007;66:1545–46. doi: 10.1136/ard.2007.071498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Roldan R, Ruiz AM, Miranda MD, Collantes E. Anakinra: new therapeutic approach in children with familial Mediterranean fever resistant to colchicine. Joint Bone Spine. 2008;75:504–5. doi: 10.1016/j.jbspin.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 90.Chae JJ, Centola M, Aksentijevich I, Dutra A, Tran M, et al. Isolation, genomic organization, and expression analysis of the mouse and rat homologs of MEFV, the gene for familial Mediterranean fever. Mamm Genome. 2000;11:428–35. doi: 10.1007/s003350010082. [DOI] [PubMed] [Google Scholar]
- 91.Uchil PD, Quinlan BD, Chan WT, Luna JM, Mothes W. TRIM E3 ligases interfere with early and late stages of the retroviral life cycle. PLoS Pathog. 2008;4:e16. doi: 10.1371/journal.ppat.0040016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Schaner P, Richards N, Wadhwa A, Aksentijevich I, Kastner D, et al. Episodic evolution of pyrin in primates: human mutations recapitulate ancestral amino acid states. Nat Genet. 2001;27:318–21. doi: 10.1038/85893. [DOI] [PubMed] [Google Scholar]
- 93.Masters SL, Yao S, Willson TA, Zhang JG, Palmer KR, et al. The SPRY domain of SSB-2 adopts a novel fold that presents conserved Par-4-binding residues. Nat Struct Mol Biol. 2006;13:77–84. doi: 10.1038/nsmb1034. [DOI] [PubMed] [Google Scholar]
- 94.Keeble AH, Khan Z, Forster A, James LC. TRIM21 is an IgG receptor that is structurally, thermodynamically, and kinetically conserved. Proc Natl Acad Sci USA. 2008;105:6045–50. doi: 10.1073/pnas.0800159105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Woo JS, Suh HY, Park SY, Oh BH. Structural basis for protein recognition by B30.2/SPRY domains. Mol Cell. 2006;24:967–76. doi: 10.1016/j.molcel.2006.11.009. [DOI] [PubMed] [Google Scholar]
- 96.Lindor NM, Arsenault TM, Solomon H, Seidman CE, McEvoy MT. A new autosomal dominant disorder of pyogenic sterile arthritis, pyoderma gangrenosum, and acne: PAPA syndrome. Mayo Clin Proc. 1997;72:611–15. doi: 10.1016/S0025-6196(11)63565-9. [DOI] [PubMed] [Google Scholar]
- 97.Wise CA, Bennett LB, Pascual V, Gillum JD, Bowcock AM. Localization of a gene for familial recurrent arthritis. Arthritis Rheum. 2000;43:2041–45. doi: 10.1002/1529-0131(200009)43:9<2041::AID-ANR15>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 98.Wise CA, Gillum JD, Seidman CE, Lindor NM, Veile R, et al. Mutations in CD2BP1 disrupt binding to PTP PEST and are responsible for PAPA syndrome, an autoinflammatory disorder. Hum Mol Genet. 2002;11:961–69. doi: 10.1093/hmg/11.8.961. [DOI] [PubMed] [Google Scholar]
- 99.Yu JW, Fernandes-Alnemri T, Datta P, Wu J, Juliana C, et al. Pyrin activates the ASC pyroptosome in response to engagement by autoinflammatory PSTPIP1 mutants. Mol Cell. 2007;28:214–27. doi: 10.1016/j.molcel.2007.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fernandes-Alnemri T, Wu J, Yu JW, Datta P, Miller B, et al. The pyroptosome: a supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ. 2007;14:1590–604. doi: 10.1038/sj.cdd.4402194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Dierselhuis MP, Frenkel J, Wulffraat NM, Boelens JJ. Anakinra for flares of pyogenic arthritis in PAPA syndrome. Rheumatology. 2005;44:406–8. doi: 10.1093/rheumatology/keh479. [DOI] [PubMed] [Google Scholar]
- 102.Badour K, Zhang J, Shi F, Leng Y, Collins M, Siminovitch KA. Fyn and PTP-PEST-mediated regulation of Wiskott-Aldrich syndrome protein (WASp) tyrosine phosphorylation is required for coupling T cell antigen receptor engagement to WASp effector function and T cell activation. J Exp Med. 2004;199:99–112. doi: 10.1084/jem.20030976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Yang H, Reinherz EL. CD2BP1 modulates CD2-dependent T cell activation via linkage to protein tyrosine phosphatase (PTP)-PEST. J Immunol. 2006;176:5898–907. doi: 10.4049/jimmunol.176.10.5898. [DOI] [PubMed] [Google Scholar]
- 104.Baum W, Kirkin V, Fernandez SB, Pick R, Lettau M, et al. Binding of the intracellular Fas ligand (FasL) domain to the adaptor protein PSTPIP results in a cytoplasmic localization of FasL. J Biol Chem. 2005;280:40012–24. doi: 10.1074/jbc.M502222200. [DOI] [PubMed] [Google Scholar]
- 105.Byrd L, Grossmann M, Potter M, Shen-Ong GL. Chronic multifocal osteomyelitis, a new recessive mutation on chromosome 18 of the mouse. Genomics. 1991;11:794–98. doi: 10.1016/0888-7543(91)90002-v. [DOI] [PubMed] [Google Scholar]
- 106.Ferguson PJ, Bing X, Vasef MA, Ochoa LA, Mahgoub A, et al. A missense mutation in pstpip2 is associated with the murine autoinflammatory disorder chronic multifocal osteomyelitis. Bone. 2006;38:41–47. doi: 10.1016/j.bone.2005.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.El-Shanti HI, Ferguson PJ. Chronic recurrent multifocal osteomyelitis: a concise review and genetic update. Clin Orthop Relat Res. 2007;462:11–19. doi: 10.1097/BLO.0b013e3180986d73. [DOI] [PubMed] [Google Scholar]
- 108.Jansson A, Renner ED, Ramser J, Mayer A, Haban M, et al. Classification of nonbacterial osteitis: retrospective study of clinical, immunological and genetic aspects in 89 patients. Rheumatology. 2007;46:154–60. doi: 10.1093/rheumatology/kel190. [DOI] [PubMed] [Google Scholar]
- 109.Grosse J, Chitu V, Marquardt A, Hanke P, Schmittwolf C, et al. Mutation of mouse Mayp/Pstpip2 causes a macrophage autoinflammatory disease. Blood. 2006;107:3350–58. doi: 10.1182/blood-2005-09-3556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Golla A, Jansson A, Ramser J, Hellebrand H, Zahn R, et al. Chronic recurrent multifocal osteomyelitis (CRMO): evidence for a susceptibility gene located on chromosome 18q21.3–18q22. Eur J Hum Genet. 2002;10:217–21. doi: 10.1038/sj.ejhg.5200789. [DOI] [PubMed] [Google Scholar]
- 111.Hoffmann G, Gibson KM, Brandt IK, Bader PI, Wappner RS, Sweetman L. Mevalonic aciduria—an inborn error of cholesterol and nonsterol isoprene biosynthesis. N Engl J Med. 1986;314:1610–14. doi: 10.1056/NEJM198606193142504. [DOI] [PubMed] [Google Scholar]
- 112.Bodar EJ, Van Der Hilst JC, van Heerde W, Van Der Meer JW, Drenth JP, Simon A. Defective apoptosis of peripheral-blood lymphocytes in hyper-IgD and periodic fever syndrome. Blood. 2007;109:2416–18. doi: 10.1182/blood-2005-10-039578. [DOI] [PubMed] [Google Scholar]
- 113.Kuijk LM, Beekman JM, Koster J, Waterham HR, Frenkel J, Coffer PJ. HMG-CoA reductase inhibition induces IL-1β release through Rac1/PI3K/PKB-dependent caspase-1 activation. Blood. 2008;112:3563–73. doi: 10.1182/blood-2008-03-144667. [DOI] [PubMed] [Google Scholar]
- 114.Hager EJ, Tse HM, Piganelli JD, Gupta M, Baetscher M, et al. Deletion of a single mevalonate kinase (Mvk) allele yields a murine model of hyper-IgD syndrome. J Inherit Metab Dis. 2007;30:888–95. doi: 10.1007/s10545-007-0776-7. [DOI] [PubMed] [Google Scholar]
- 115.Simon A, Bijzet J, Voorbij HA, Mantovani A, Van Der Meer JW, Drenth JP. Effect of inflammatory attacks in the classical type hyper-IgD syndrome on immunoglobulin D, cholesterol and parameters of the acute phase response. J Intern Med. 2004;256:247–53. doi: 10.1111/j.1365-2796.2004.01359.x. [DOI] [PubMed] [Google Scholar]
- 116.Simon A, van Deuren M, Tighe PJ, Van Der Meer JW, Drenth JP. Genetic analysis as a valuable key to diagnosis and treatment of periodic fever. Arch Intern Med. 2001;161:2491–93. doi: 10.1001/archinte.161.20.2491. [DOI] [PubMed] [Google Scholar]
- 117.Bodar EJ, Van Der Hilst JC, Drenth JP, Van Der Meer JW, Simon A. Effect of etanercept and anakinra on inflammatory attacks in the hyper-IgD syndrome: introducing a vaccination provocation model. Neth J Med. 2005;63:260–64. [PubMed] [Google Scholar]
- 118.Cailliez M, Garaix F, Rousset-Rouviere C, Bruno D, Kone-Paut I, et al. Anakinra is safe and effective in controlling hyperimmunoglobulinaemia D syndrome-associated febrile crisis. J Inherit Metab Dis. 2006;29:763. doi: 10.1007/s10545-006-0408-7. [DOI] [PubMed] [Google Scholar]
- 119.Mandey SH, Schneiders MS, Koster J, Waterham HR. Mutational spectrum and genotype-phenotype correlations in mevalonate kinase deficiency. Hum Mutat. 2006;27:796–802. doi: 10.1002/humu.20361. [DOI] [PubMed] [Google Scholar]
- 120.Slim R, Mehio A. The genetics of hydatidiform moles: new lights on an ancient disease. Clin Genet. 2007;71:25–34. doi: 10.1111/j.1399-0004.2006.00697.x. [DOI] [PubMed] [Google Scholar]
- 121.El-Maarri O, Slim R. Familial hydatidiform molar pregnancy: the germline imprinting defect hypothesis? Curr Top Microbiol Immunol. 2006;301:229–41. doi: 10.1007/3-540-31390-7_8. [DOI] [PubMed] [Google Scholar]
- 122.Murdoch S, Djuric U, Mazhar B, Seoud M, Khan R, et al. Mutations in NALP7 cause recurrent hydatidiform moles and reproductive wastage in humans. Nat Genet. 2006;38:300–2. doi: 10.1038/ng1740. [DOI] [PubMed] [Google Scholar]
- 123.Kinoshita T, Wang Y, Hasegawa M, Imamura R, Suda T. PYPAF3, a PYRIN-containing APAF-1-like protein, is a feedback regulator of caspase-1-dependent interleukin-1β secretion. J Biol Chem. 2005;280:21720–25. doi: 10.1074/jbc.M410057200. [DOI] [PubMed] [Google Scholar]
- 124.Mor G. Inflammation and pregnancy: the role of Toll-like receptors in trophoblast-immune interaction. Ann NY Acad Sci. 2008;1127:121–28. doi: 10.1196/annals.1434.006. [DOI] [PubMed] [Google Scholar]
- 125.Caucheteux SM, Kanellopoulos-Langevin C, Ojcius DM. At the innate frontiers between mother and fetus: linking abortion with complement activation. Immunity. 2003;18:169–72. doi: 10.1016/s1074-7613(03)00028-1. [DOI] [PubMed] [Google Scholar]
- 126.Murtha AP, Nieves A, Hauser ER, Swamy GK, Yonish BA, et al. Association of maternal IL-1 receptor antagonist intron 2 gene polymorphism and preterm birth. Am J Obstet Gynecol. 2006;195:1249–53. doi: 10.1016/j.ajog.2006.09.002. [DOI] [PubMed] [Google Scholar]
- 127.Santtila S, Savinainen K, Hurme M. Presence of the IL-1RA allele 2 (IL1RN*2) is associated with enhanced IL-1β production in vitro. Scand J Immunol. 1998;47:195–98. doi: 10.1046/j.1365-3083.1998.00300.x. [DOI] [PubMed] [Google Scholar]
- 128.Kou YC, Shao L, Peng HH, Rosetta R, del Gaudio D, et al. A recurrent intragenic genomic duplication, other novel mutations in NLRP7 and imprinting defects in recurrent biparental hydatidiform moles. Mol Hum Reprod. 2008;14:33–40. doi: 10.1093/molehr/gam079. [DOI] [PubMed] [Google Scholar]
- 129.Qian J, Deveault C, Bagga R, Xie X, Slim R. Women heterozygous for NALP7/NLRP7 mutations are at risk for reproductive wastage: report of two novel mutations. Hum Mutat. 2007;28:741. doi: 10.1002/humu.9498. [DOI] [PubMed] [Google Scholar]
- 130.Chen CJ, Shi Y, Hearn A, Fitzgerald K, Golenbock D, et al. MyD88-dependent IL-1 receptor signaling is essential for gouty inflammation stimulated by monosodium urate crystals. J Clin Invest. 2006;116:2262–71. doi: 10.1172/JCI28075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.McGonagle D, Tan AL, Shankaranarayana S, Madden J, Emery P, McDermott MF. Management of treatment resistant inflammation of acute or chronic tophaceous gout with anakinra. Ann Rheum Dis. 2007;66:1683–84. doi: 10.1136/ard.2007.073759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.So A, De Smedt T, Revaz S, Tschopp J. A pilot study of IL-1 inhibition by anakinra in acute gout. Arthritis Res Ther. 2007;9:R28. doi: 10.1186/ar2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Cowper SE, Kuo PH, Bucala R. Nephrogenic systemic fibrosis and gadolinium exposure: association and lessons for idiopathic fibrosing disorders. Arthritis Rheum. 2007;56:3173–75. doi: 10.1002/art.22926. [DOI] [PubMed] [Google Scholar]
- 134.Armanios MY, Chen JJ, Cogan JD, Alder JK, Ingersoll RG, et al. Telomerase mutations in families with idiopathic pulmonary fibrosis. N Engl J Med. 2007;356:1317–26. doi: 10.1056/NEJMoa066157. [DOI] [PubMed] [Google Scholar]
- 135.Afeltra A, Gentilucci UV, Rabitti C, Amoroso A, Caricato M, et al. Retroperitoneal fibrosis and ankylosing spondylitis: which links? Semin Arthritis Rheum. 2005;35:43–48. doi: 10.1016/j.semarthrit.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 136.Perkins RC, Scheule RK, Hamilton R, Gomes G, Freidman G, Holian A. Human alveolar macrophage cytokine release in response to in vitro and in vivo asbestos exposure. Exp Lung Res. 1993;19:55–65. doi: 10.3109/01902149309071080. [DOI] [PubMed] [Google Scholar]
- 137.Halle A, Hornung V, Petzold GC, Stewart CR, Monks BG, et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nat Immunol. 2008;9:857–65. doi: 10.1038/ni.1636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lawson WE, Loyd JE. The genetic approach in pulmonary fibrosis: Can it provide clues to this complex disease? Proc Am Thorac Soc. 2006;3:345–49. doi: 10.1513/pats.200512-137TK. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Whyte M, Hubbard R, Meliconi R, Whidborne M, Eaton V, et al. Increased risk of fibrosing alveolitis associated with interleukin-1 receptor antagonist and tumor necrosis factor-α gene polymorphisms. Am J Respir Crit Care Med. 2000;162:755–58. doi: 10.1164/ajrccm.162.2.9909053. [DOI] [PubMed] [Google Scholar]
- 140.Gasse P, Mary C, Guenon I, Noulin N, Charron S, et al. IL-1R1/MyD88 signaling and the inflammasome are essential in pulmonary inflammation and fibrosis in mice. J Clin Invest. 2007;117:3786–99. doi: 10.1172/JCI32285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Eisenbarth GS. Update in type 1 diabetes. J Clin Endocrinol Metab. 2007;92:2403–7. doi: 10.1210/jc.2007-0339. [DOI] [PubMed] [Google Scholar]
- 142.Stumvoll M, Goldstein BJ, van Haeften TW. Type 2 diabetes: principles of pathogenesis and therapy. Lancet. 2005;365:1333–46. doi: 10.1016/S0140-6736(05)61032-X. [DOI] [PubMed] [Google Scholar]
- 143.Pickup JC, Mattock MB, Chusney GD, Burt D. NIDDM as a disease of the innate immune system: association of acute-phase reactants and interleukin-6 with metabolic syndrome X. Diabetologia. 1997;40:1286–92. doi: 10.1007/s001250050822. [DOI] [PubMed] [Google Scholar]
- 144.Spranger J, Kroke A, Mohlig M, Hoffmann K, Bergmann MM, et al. Inflammatory cytokines and the risk to develop type 2 diabetes: results of the prospective population-based European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Diabetes. 2003;52:812–17. doi: 10.2337/diabetes.52.3.812. [DOI] [PubMed] [Google Scholar]
- 145.Maedler K, Sergeev P, Ris F, Oberholzer J, Joller-Jemelka HI, et al. Glucose-induced β cell production of IL-1β contributes to glucotoxicity in human pancreatic islets. J Clin Invest. 2002;110:851–60. doi: 10.1172/JCI15318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Sauter NS, Schulthess FT, Galasso R, Castellani LW, Maedler K. The antiinflammatory cytokine interleukin-1 receptor antagonist protects from high-fat diet-induced hyperglycemia. Endocrinology. 2008;149:2208–18. doi: 10.1210/en.2007-1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Larsen CM, Faulenbach M, Vaag A, Volund A, Ehses JA, et al. Interleukin-1-receptor antagonist in type 2 diabetes mellitus. N Engl J Med. 2007;356:1517–26. doi: 10.1056/NEJMoa065213. A clinical trial showed that T2DM is responsive to therapy with IL-1 blockade. [DOI] [PubMed] [Google Scholar]
- 148.de Koning HD, Bodar EJ, Van Der Meer JW, Simon A. Schnitzler syndrome: beyond the case reports: review and follow-up of 94 patients with an emphasis on prognosis and treatment. Semin Arthritis Rheum. 2007;37:137–48. doi: 10.1016/j.semarthrit.2007.04.001. [DOI] [PubMed] [Google Scholar]
- 149.Ryan JG, de Koning HD, Beck LA, Booty MG, Kastner DL, Simon A. IL-1 blockade in Schnitzler syndrome: ex vivo findings correlate with clinical remission. J Allergy Clin Immunol. 2008;121:260–62. doi: 10.1016/j.jaci.2007.09.021. [DOI] [PubMed] [Google Scholar]
- 150.Dybowski F, Sepp N, Bergerhausen HJ, Braun J. Successful use of anakinra to treat refractory Schnitzler’s syndrome. Clin Exp Rheumatol. 2008;26:354–57. [PubMed] [Google Scholar]
- 151.Frischmeyer-Guerrerio PA, Rachamalla R, Saini SS. Remission of Schnitzler syndrome after treatment with anakinra. Ann Allergy Asthma Immunol. 2008;100:617–19. doi: 10.1016/S1081-1206(10)60064-6. [DOI] [PubMed] [Google Scholar]
- 152.Gilson M, Abad S, Larroche C, Dhote R. Treatment of Schnitzler’s syndrome with anakinra. Clin Exp Rheumatol. 2007;25:931. [PubMed] [Google Scholar]
- 153.Wastiaux H, Barbarot S, Gagey-Caron V, Berthelot JM, Hamidou M, Stalder JF. Schnitzler syndrome: a dramatic improvement with anakinra. J Eur Acad Dermatol Venereol. 2008 doi: 10.1111/j.1468-3083.2008.02708.x. In press. [DOI] [PubMed] [Google Scholar]
- 154.Jin Y, Mailloux CM, Gowan K, Riccardi SL, LaBerge G, et al. NALP1 in vitiligo-associated multiple autoimmune disease. N Engl J Med. 2007;356:1216–25. doi: 10.1056/NEJMoa061592. [DOI] [PubMed] [Google Scholar]
- 155.Granell M, Urbano-Ispizua A, Pons A, Arostegui JI, Gel B, et al. Common variants in NLRP2 and NLRP3 genes are strong prognostic factors for the outcome of HLA-identical sibling allogeneic stem cell transplantation. Blood. 2008;112:4337–42. doi: 10.1182/blood-2007-12-129247. [DOI] [PubMed] [Google Scholar]
- 156.Holler E, Rogler G, Herfarth H, Brenmoehl J, Wild PJ, et al. Both donor and recipient NOD2/CARD15 mutations associate with transplant-related mortality and GvHD following allogeneic stem cell transplantation. Blood. 2004;104:889–94. doi: 10.1182/blood-2003-10-3543. [DOI] [PubMed] [Google Scholar]
- 157.Inohara N, Ogura Y, Chen FF, Muto A, Nunez G. Human Nod1 confers responsiveness to bacterial lipopolysaccharides. J Biol Chem. 2001;276:2551–54. doi: 10.1074/jbc.M009728200. [DOI] [PubMed] [Google Scholar]
- 158.Inohara N, Ogura Y, Fontalba A, Gutierrez O, Pons F, et al. Host recognition of bacterial muramyl dipeptide mediated through NOD2. Implications for Crohn’s disease. J Biol Chem. 2003;278:5509–12. doi: 10.1074/jbc.C200673200. [DOI] [PubMed] [Google Scholar]
- 159.Barrat FJ, Meeker T, Gregorio J, Chan JH, Uematsu S, et al. Nucleic acids of mammalian origin can act as endogenous ligands for Toll-like receptors and may promote systemic lupus erythematosus. J Exp Med. 2005;202:1131–39. doi: 10.1084/jem.20050914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pisitkun P, Deane JA, Difilippantonio MJ, Tarasenko T, Satterthwaite AB, Bolland S. Autoreactive B cell responses to RNA-related antigens due to TLR7 gene duplication. Science. 2006;312:1669–72. doi: 10.1126/science.1124978. [DOI] [PubMed] [Google Scholar]
- 161.Subramanian S, Tus K, Li QZ, Wang A, Tian XH, et al. A Tlr7 translocation accelerates systemic autoimmunity in murine lupus. Proc Natl Acad Sci USA. 2006;103:9970–75. doi: 10.1073/pnas.0603912103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Krieg AM. The Toll of too much TLR7. Immunity. 2007;27:695–97. doi: 10.1016/j.immuni.2007.11.001. [DOI] [PubMed] [Google Scholar]
- 163.Cho JH. The genetics and immunopathogenesis of inflammatory bowel disease. Nat Rev Immunol. 2008;8:458–66. doi: 10.1038/nri2340. [DOI] [PubMed] [Google Scholar]
- 164.Barrett JC, Hansoul S, Nicolae DL, Cho JH, Duerr RH, et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn’s disease. Nat Genet. 2008;40:955–62. doi: 10.1038/NG.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Fisher SA, Tremelling M, Anderson CA, Gwilliam R, Bumpstead S, et al. Genetic determinants of ulcerative colitis include the ECM1 locus and five loci implicated in Crohn’s disease. Nat Genet. 2008;40:710–12. doi: 10.1038/ng.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Franke A, Balschun T, Karlsen TH, Hedderich J, May S, et al. Replication of signals from recent studies of Crohn’s disease identifies previously unknown disease loci for ulcerative colitis. Nat Genet. 2008;40:713–15. doi: 10.1038/ng.148. [DOI] [PubMed] [Google Scholar]
- 167.Franke A, Balschun T, Karlsen TH, Sventoraityte J, Nikolaus S, et al. Sequence variants in IL10, ARPC2 and multiple other loci contribute to ulcerative colitis susceptibility. Nat Genet. 2008;40:1319–23. doi: 10.1038/ng.221. [DOI] [PubMed] [Google Scholar]
- 168.Inohara N, Chamaillard M, McDonald C, Nuñez G. NOD-LRR proteins: role in host-microbial interactions and inflammatory disease. Annu Rev Biochem. 2005;74:355–83. doi: 10.1146/annurev.biochem.74.082803.133347. [DOI] [PubMed] [Google Scholar]
- 169.Lesage S, Zouali H, Cezard JP, Colombel JF, Belaiche J, et al. CARD15/NOD2 mutational analysis and genotype-phenotype correlation in 612 patients with inflammatory bowel disease. Am J Hum Genet. 2002;70:845–57. doi: 10.1086/339432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Schreiber S, Nikolaus S, Hampe J. Activation of nuclear factor kB inflammatory bowel disease. Gut. 1998;42:477–84. doi: 10.1136/gut.42.4.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Li J, Moran T, Swanson E, Julian C, Harris J, et al. Regulation of IL-8 and IL-1β expression in Crohn’s disease associated NOD2/CARD15 mutations. Hum Mol Genet. 2004;13:1715–25. doi: 10.1093/hmg/ddh182. [DOI] [PubMed] [Google Scholar]
- 172.van Heel DA, Ghosh S, Butler M, Hunt KA, Lundberg AM, et al. Muramyl dipeptide and Toll-like receptor sensitivity in NOD2-associated Crohn’s disease. Lancet. 2005;365:1794–96. doi: 10.1016/S0140-6736(05)66582-8. [DOI] [PubMed] [Google Scholar]
- 173.Maeda S, Hsu LC, Liu H, Bankston LA, Iimura M, et al. Nod2 mutation in Crohn’s disease potentiates NF-κB activity and IL-1β processing. Science. 2005;307:734–38. doi: 10.1126/science.1103685. [DOI] [PubMed] [Google Scholar]
- 174.Marks DJ, Harbord MW, MacAllister R, Rahman RZ, Young J, et al. Defective acute inflammation in Crohn’s disease: a clinical investigation. Lancet. 2006;367:668–78. doi: 10.1016/S0140-6736(06)68265-2. [DOI] [PubMed] [Google Scholar]
- 175.Kobayashi KS, Chamaillard M, Ogura Y, Henegariu O, Inohara N, et al. Nod2-dependent regulation of innate and adaptive immunity in the intestinal tract. Science. 2005;307:731–34. doi: 10.1126/science.1104911. [DOI] [PubMed] [Google Scholar]
- 176.Simms LA, Doecke JD, Walsh MD, Huang N, Fowler EV, Radford-Smith GL. Reduced alpha-defensin expression is associated with inflammation and not NOD2 mutation status in ileal Crohn’s disease. Gut. 2008;57:903–10. doi: 10.1136/gut.2007.142588. [DOI] [PubMed] [Google Scholar]
- 177.Pauleau AL, Murray PJ. Role of nod2 in the response of macrophages to Toll-like receptor agonists. Mol Cell Biol. 2003;23:7531–39. doi: 10.1128/MCB.23.21.7531-7539.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Watanabe T, Asano N, Murray PJ, Ozato K, Tailor P, et al. Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. J Clin Invest. 2008;118:545–59. doi: 10.1172/JCI33145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Cadwell K, Liu JY, Brown SL, Miyoshi H, Loh J, et al. A key role for autophagy and the autophagy gene Atg16l1 in mouse and human intestinal Paneth cells. Nature. 2008;456:259–63. doi: 10.1038/nature07416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Saitoh T, Fujita N, Jang MH, Uematsu S, Yang BG, et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature. 2008;456:264–68. doi: 10.1038/nature07383. [DOI] [PubMed] [Google Scholar]
- 181.Kanazawa N, Okafuji I, Kambe N, Nishikomori R, Nakata-Hizume M, et al. Early-onset sarcoidosis and CARD15 mutations with constitutive nuclear factor-κB activation: common genetic etiology with Blau syndrome. Blood. 2005;105:1195–97. doi: 10.1182/blood-2004-07-2972. [DOI] [PubMed] [Google Scholar]
- 182.Wang X, Kuivaniemi H, Bonavita G, Mutkus L, Mau U, et al. CARD15 mutations in familial granulomatosis syndromes: a study of the original Blau syndrome kindred and other families with large-vessel arteritis and cranial neuropathy. Arthritis Rheum. 2002;46:3041–45. doi: 10.1002/art.10618. [DOI] [PubMed] [Google Scholar]
- 183.Arostegui JI, Arnal C, Merino R, Modesto C, Antonia Carballo M, et al. NOD2 gene-associated pediatric granulomatous arthritis: clinical diversity, novel and recurrent mutations, and evidence of clinical improvement with interleukin-1 blockade in a Spanish cohort. Arthritis Rheum. 2007;56:3805–13. doi: 10.1002/art.22966. [DOI] [PubMed] [Google Scholar]
- 184.Albrecht M, Lengauer T, Schreiber S. Disease-associated variants in PYPAF1 and NOD2 result in similar alterations of conserved sequence. Bioinformatics. 2003;19:2171–75. doi: 10.1093/bioinformatics/btg370. [DOI] [PubMed] [Google Scholar]
- 185.Williams KL, Lich JD, Duncan JA, Reed W, Rallabhandi P, et al. The CATERPILLER protein monarch-1 is an antagonist of Toll-like receptor-, tumor necrosis factor α-, and Mycobacterium tuberculosis-induced proinflammatory signals. J Biol Chem. 2005;280:39914–24. doi: 10.1074/jbc.M502820200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Hull KM, Drewe E, Aksentijevich I, Singh HK, Wong K, et al. The TNF receptor-associated periodic syndrome (TRAPS): emerging concepts of an autoinflammatory disorder. Medicine. 2002;81:349–68. doi: 10.1097/00005792-200209000-00002. [DOI] [PubMed] [Google Scholar]
- 187.Hull KM, Wong K, Wood GM, Chu WS, Kastner DL. Monocytic fasciitis: a newly recognized clinical feature of tumor necrosis factor receptor dysfunction. Arthritis Rheum. 2002;46:2189–94. doi: 10.1002/art.10448. [DOI] [PubMed] [Google Scholar]
- 188.Aksentijevich I, Galon J, Soares M, Mansfield E, Hull K, et al. The tumor-necrosis-factor receptor-associated periodic syndrome: new mutations in TNFRSF1A, ancestral origins, genotype-phenotype studies, and evidence for further genetic heterogeneity of periodic fevers. Am J Hum Genet. 2001;69:301–14. doi: 10.1086/321976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Mullberg J, Durie FH, Otten-Evans C, Alderson MR, Rose-John S, et al. A metalloprotease inhibitor blocks shedding of the IL-6 receptor and the p60 TNF receptor. J Immunol. 1995;155:5198–205. [PubMed] [Google Scholar]
- 190.Huggins ML, Radford PM, McIntosh RS, Bainbridge SE, Dickinson P, et al. Shedding of mutant tumor necrosis factor receptor superfamily 1A associated with tumor necrosis factor receptor-associated periodic syndrome: differences between cell types. Arthritis Rheum. 2004;50:2651–59. doi: 10.1002/art.20380. [DOI] [PubMed] [Google Scholar]
- 191.Todd I, Radford PM, Draper-Morgan KA, McIntosh R, Bainbridge S, et al. Mutant forms of tumour necrosis factor receptor I that occur in TNF-receptor-associated periodic syndrome retain signalling functions but show abnormal behaviour. Immunology. 2004;113:65–79. doi: 10.1111/j.1365-2567.2004.01942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Todd I, Radford PM, Daffa N, Bainbridge SE, Powell RJ, Tighe PJ. Mutant tumor necrosis factor receptor associated with tumor necrosis factor receptor-associated periodic syndrome is altered antigenically and is retained within patients’ leukocytes. Arthritis Rheum. 2007;56:2765–73. doi: 10.1002/art.22740. [DOI] [PubMed] [Google Scholar]
- 193.Rebelo SL, Bainbridge SE, Amel-Kashipaz MR, Radford PM, Powell RJ, et al. Modeling of tumor necrosis factor receptor superfamily 1A mutants associated with tumor necrosis factor receptor-associated periodic syndrome indicates misfolding consistent with abnormal function. Arthritis Rheum. 2006;54:2674–87. doi: 10.1002/art.21964. [DOI] [PubMed] [Google Scholar]
- 194.Kriegel MA, Huffmeier U, Scherb E, Scheidig C, Geiler T, et al. Tumor necrosis factor receptor-associated periodic syndrome characterized by a mutation affecting the cleavage site of the receptor: implications for pathogenesis. Arthritis Rheum. 2003;48:2386–88. doi: 10.1002/art.11169. [DOI] [PubMed] [Google Scholar]
- 195.Stojanov S, Dejaco C, Lohse P, Huss K, Duftner C, et al. Clinical and functional characterisation of a novel TNFRSF1A c. 605T>A/V173D cleavage site mutation associated with tumour necrosis factor receptor-associated periodic fever syndrome (TRAPS), cardiovascular complications and excellent response to etanercept treatment. Ann Rheum Dis. 2008;67:1292–98. doi: 10.1136/ard.2007.079376. [DOI] [PubMed] [Google Scholar]
- 196.Xu C, Bailly-Maitre B, Reed JC. Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest. 2005;115:2656–64. doi: 10.1172/JCI26373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Simon A, Bodar EJ, Van Der Hilst JC, Van Der Meer JW, Fiselier TJ, et al. Beneficial response to interleukin 1 receptor antagonist in traps. Am J Med. 2004;117:208–10. doi: 10.1016/j.amjmed.2004.02.039. [DOI] [PubMed] [Google Scholar]
- 198.Gattorno M, Pelagatti MA, Meini A, Obici L, Barcellona R, et al. Persistent efficacy of anakinra in patients with tumor necrosis factor receptor-associated periodic syndrome. Arthritis Rheum. 2008;58:1516–20. doi: 10.1002/art.23475. [DOI] [PubMed] [Google Scholar]
- 199.Sacre K, Brihaye B, Lidove O, Papo T, Pocidalo MA, et al. Dramatic improvement following interleukin 1β blockade in tumor necrosis factor receptor-1-associated syndrome (TRAPS) resistant to anti-TNF-α therapy. J Rheumatol. 2008;35:357–58. [PubMed] [Google Scholar]
- 200.Sims AM, Wordsworth BP, Brown MA. Genetic susceptibility to ankylosing spondylitis. Curr Mol Med. 2004;4:13–20. doi: 10.2174/1566524043479284. [DOI] [PubMed] [Google Scholar]
- 201.Turner MJ, Colbert RA. HLA-B27 and pathogenesis of spondyloarthropathies. Curr Opin Rheumatol. 2002;14:367–72. doi: 10.1097/00002281-200207000-00006. [DOI] [PubMed] [Google Scholar]
- 202.Dangoria NS, DeLay ML, Kingsbury DJ, Mear JP, Uchanska-Ziegler B, et al. HLA-B27 misfolding is associated with aberrant intermolecular disulfide bond formation (dimerization) in the endoplasmic reticulum. J Biol Chem. 2002;277:23459–68. doi: 10.1074/jbc.M110336200. Evidence that misfolded HLAB27 could account for its association with ankylosing spondylitis. [DOI] [PubMed] [Google Scholar]
- 203.Colbert RA. The immunobiology of HLA-B27: variations on a theme. Curr Mol Med. 2004;4:21–30. doi: 10.2174/1566524043479293. [DOI] [PubMed] [Google Scholar]
- 204.Tran TM, Satumtira N, Dorris ML, May E, Wang A, et al. HLA-B27 in transgenic rats forms disulfide-linked heavy chain oligomers and multimers that bind to the chaperone BiP. J Immunol. 2004;172:5110–19. doi: 10.4049/jimmunol.172.8.5110. [DOI] [PubMed] [Google Scholar]
- 205.Turner MJ, Sowders DP, DeLay ML, Mohapatra R, Bai S, et al. HLA-B27 misfolding in transgenic rats is associated with activation of the unfolded protein response. J Immunol. 2005;175:2438–48. doi: 10.4049/jimmunol.175.4.2438. [DOI] [PubMed] [Google Scholar]
- 206.Taurog JD. The mystery of HLA-B27: If it isn’t one thing, it’s another. Arthritis Rheum. 2007;56:2478–81. doi: 10.1002/art.22807. [DOI] [PubMed] [Google Scholar]
- 207.Galocha B, de Castro JA. Folding of HLA-B27 subtypes is determined by the global effect of polymorphic residues and shows incomplete correspondence to ankylosing spondylitis. Arthritis Rheum. 2008;58:401–12. doi: 10.1002/art.23164. [DOI] [PubMed] [Google Scholar]
- 208.Sims AM, Timms AE, Bruges-Armas J, Burgos-Vargas R, Chou CT, et al. Prospective meta-analysis of interleukin 1 gene complex polymorphisms confirms associations with ankylosing spondylitis. Ann Rheum Dis. 2008;67:1305–9. doi: 10.1136/ard.2007.081364. [DOI] [PubMed] [Google Scholar]
- 209.Burton PR, Clayton DG, Cardon LR, Craddock N, Deloukas P, et al. Association scan of 14500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat Genet. 2007;39:1329–37. doi: 10.1038/ng.2007.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Cargill M, Schrodi SJ, Chang M, Garcia VE, Brandon R, et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am J Hum Genet. 2007;80:273–90. doi: 10.1086/511051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Chen Z, O’Shea JJ. Th17 cells: a new fate for differentiating helper T cells. Immunol Res. 2008;41:87–102. doi: 10.1007/s12026-007-8014-9. [DOI] [PubMed] [Google Scholar]
- 212.Loirat C, Noris M, Fremeaux-Bacchi V. Complement and the atypical hemolytic uremic syndrome in children. Pediatr Nephrol. 2008;23:1957–72. doi: 10.1007/s00467-008-0872-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Tarr PI, Gordon CA, Chandler WL. Shigatoxin-producing Escherichia coli and haemolytic uraemic syndrome. Lancet. 2005;365:1073–86. doi: 10.1016/S0140-6736(05)71144-2. [DOI] [PubMed] [Google Scholar]
- 214.Ohali M, Shalev H, Schlesinger M, Katz Y, Kachko L, et al. Hypocomplementemic autosomal recessive hemolytic uremic syndrome with decreased factor H. Pediatr Nephrol. 1998;12:619–24. doi: 10.1007/s004670050515. [DOI] [PubMed] [Google Scholar]
- 215.Warwicker P, Goodship TH, Donne RL, Pirson Y, Nicholls A, et al. Genetic studies into inherited and sporadic hemolytic uremic syndrome. Kidney Int. 1998;53:836–44. doi: 10.1111/j.1523-1755.1998.00824.x. [DOI] [PubMed] [Google Scholar]
- 216.Noris M, Brioschi S, Caprioli J, Todeschini M, Bresin E, et al. Familial haemolytic uraemic syndrome and an MCP mutation. Lancet. 2003;362:1542–47. doi: 10.1016/S0140-6736(03)14742-3. [DOI] [PubMed] [Google Scholar]
- 217.Richards A, Kemp EJ, Liszewski MK, Goodship JA, Lampe AK, et al. Mutations in human complement regulator, membrane cofactor protein (CD46), predispose to development of familial hemolytic uremic syndrome. Proc Natl Acad Sci USA. 2003;100:12966–71. doi: 10.1073/pnas.2135497100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kavanagh D, Kemp EJ, Mayland E, Winney RJ, Duffield JS, et al. Mutations in complement factor I predispose to development of atypical hemolytic uremic syndrome. J Am Soc Nephrol. 2005;16:2150–55. doi: 10.1681/ASN.2005010103. [DOI] [PubMed] [Google Scholar]
- 219.Gros P, Milder FJ, Janssen BJ. Complement driven by conformational changes. Nat Rev Immunol. 2008;8:48–58. doi: 10.1038/nri2231. [DOI] [PubMed] [Google Scholar]
- 220.Jokiranta TS, Jaakola VP, Lehtinen MJ, Parepalo M, Meri S, Goldman A. Structure of complement factor H carboxyl-terminus reveals molecular basis of atypical haemolytic uremic syndrome. EMBO J. 2006;25:1784–94. doi: 10.1038/sj.emboj.7601052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Edwards AO, Ritter R, III, Abel KJ, Manning A, Panhuysen C, Farrer LA. Complement factor H polymorphism and age-related macular degeneration. Science. 2005;308:421–24. doi: 10.1126/science.1110189. Variants of CFH predispose to AMD. [DOI] [PubMed] [Google Scholar]
- 222.Haines JL, Hauser MA, Schmidt S, Scott WK, Olson LM, et al. Complement factor H variant increases the risk of age-related macular degeneration. Science. 2005;308:419–21. doi: 10.1126/science.1110359. Variants of CFH predispose to AMD. [DOI] [PubMed] [Google Scholar]
- 223.Klein RJ, Zeiss C, Chew EY, Tsai JY, Sackler RS, et al. Complement factor H polymorphism in age-related macular degeneration. Science. 2005;308:385–89. doi: 10.1126/science.1109557. Variants of CFH predispose to AMD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Hollyfield JG, Bonilha VL, Rayborn ME, Yang X, Shadrach KG, et al. Oxidative damage-induced inflammation initiates age-related macular degeneration. Nat Med. 2008;14:194–98. doi: 10.1038/nm1709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Novack DV, Faccio R. Jawing about TNF: new hope for cherubism. Cell. 2007;128:15–17. doi: 10.1016/j.cell.2006.12.019. [DOI] [PubMed] [Google Scholar]
- 226.Filipovich AH. Hemophagocytic lymphohistiocytosis and other hemophagocytic disorders. Immunol Allergy Clin North Am. 2008;28:293–313. viii. doi: 10.1016/j.iac.2008.01.010. [DOI] [PubMed] [Google Scholar]
- 227.Kelly A, Ramanan AV. Recognition and management of macrophage activation syndrome in juvenile arthritis. Curr Opin Rheumatol. 2007;19:477–81. doi: 10.1097/BOR.0b013e32825a6a79. [DOI] [PubMed] [Google Scholar]
- 228.Fall N, Barnes M, Thornton S, Luyrink L, Olson J, et al. Gene expression profiling of peripheral blood from patients with untreated new-onset systemic juvenile idiopathic arthritis reveals molecular heterogeneity that may predict macrophage activation syndrome. Arthritis Rheum. 2007;56:3793–804. doi: 10.1002/art.22981. [DOI] [PubMed] [Google Scholar]
- 229.Schaer DJ, Schleiffenbaum B, Kurrer M, Imhof A, Bachli E, et al. Soluble hemoglobin-haptoglobin scavenger receptor CD163 as a lineage-specific marker in the reactive hemophagocytic syndrome. Eur J Haematol. 2005;74:6–10. doi: 10.1111/j.1600-0609.2004.00318.x. [DOI] [PubMed] [Google Scholar]
- 230.Stepp SE, Dufourcq-Lagelouse R, Le Deist F, Bhawan S, Certain S, et al. Perforin gene defects in familial hemophagocytic lymphohistiocytosis. Science. 1999;286:1957–59. doi: 10.1126/science.286.5446.1957. [DOI] [PubMed] [Google Scholar]
- 231.Feldmann J, Callebaut I, Raposo G, Certain S, Bacq D, et al. Munc13-4 is essential for cytolytic granules fusion and is mutated in a form of familial hemophagocytic lymphohistiocytosis (FHL3) Cell. 2003;115:461–73. doi: 10.1016/s0092-8674(03)00855-9. [DOI] [PubMed] [Google Scholar]
- 232.Bryceson YT, Rudd E, Zheng C, Edner J, Ma D, et al. Defective cytotoxic lymphocyte degranulation in syntaxin-11 deficient familial hemophagocytic lymphohistiocytosis 4 (FHL4) patients. Blood. 2007;110:1906–15. doi: 10.1182/blood-2007-02-074468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Westbroek W, Adams D, Huizing M, Koshoffer A, Dorward H, et al. Cellular defects in Chediak-Higashi syndrome correlate with the molecular genotype and clinical phenotype. J Invest Dermatol. 2007;127:2674–77. doi: 10.1038/sj.jid.5700899. [DOI] [PubMed] [Google Scholar]
- 234.Enders A, Zieger B, Schwarz K, Yoshimi A, Speckmann C, et al. Lethal hemophagocytic lymphohistiocytosis in Hermansky-Pudlak syndrome type II. Blood. 2006;108:81–87. doi: 10.1182/blood-2005-11-4413. [DOI] [PubMed] [Google Scholar]
- 235.Grom AA. Natural killer cell dysfunction: A common pathway in systemic-onset juvenile rheumatoid arthritis, macrophage activation syndrome, and hemophagocytic lymphohistiocytosis? Arthritis Rheum. 2004;50:689–98. doi: 10.1002/art.20198. [DOI] [PubMed] [Google Scholar]
- 236.Donn R, Ellison S, Lamb R, Day T, Baildam E, Ramanan AV. Genetic loci contributing to hemophagocytic lymphohistiocytosis do not confer susceptibility to systemic-onset juvenile idiopathic arthritis. Arthritis Rheum. 2008;58:869–74. doi: 10.1002/art.23270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Zhang K, Biroschak J, Glass DN, Thompson SD, Finkel T, et al. Macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis is associated with MUNC13-4 polymorphisms. Arthritis Rheum. 2008;58:2892–96. doi: 10.1002/art.23734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med. 1999;340:115–26. doi: 10.1056/NEJM199901143400207. Argues for inflammation in the pathogenesis of atherosclerosis. [DOI] [PubMed] [Google Scholar]
- 239.Yan ZQ, Hansson GK. Innate immunity, macrophage activation, and atherosclerosis. Immunol Rev. 2007;219:187–203. doi: 10.1111/j.1600-065X.2007.00554.x. [DOI] [PubMed] [Google Scholar]
- 240.Zhou X, Robertson AK, Hjerpe C, Hansson GK. Adoptive transfer of CD4+ T cells reactive to modified low-density lipoprotein aggravates atherosclerosis. Arterioscler Thromb Vasc Biol. 2006;26:864–70. doi: 10.1161/01.ATV.0000206122.61591.ff. [DOI] [PubMed] [Google Scholar]
- 241.Wang X, Ria M, Kelmenson PM, Eriksson P, Higgins DC, et al. Positional identification of TNFSF4, encoding OX40 ligand, as a gene that influences atherosclerosis susceptibility. Nat Genet. 2005;37:365–72. doi: 10.1038/ng1524. [DOI] [PubMed] [Google Scholar]
- 242.Febbraio M, Podrez EA, Smith JD, Hajjar DP, Hazen SL, et al. Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. J Clin Invest. 2000;105:1049–56. doi: 10.1172/JCI9259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Moore KJ, Kunjathoor VV, Koehn SL, Manning JJ, Tseng AA, et al. Loss of receptor-mediated lipid uptake via scavenger receptor A or CD36 pathways does not ameliorate atherosclerosis in hyperlipidemic mice. J Clin Invest. 2005;115:2192–201. doi: 10.1172/JCI24061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Devlin CM, Kuriakose G, Hirsch E, Tabas I. Genetic alterations of IL-1 receptor antagonist in mice affect plasma cholesterol level and foam cell lesion size. Proc Natl Acad Sci USA. 2002;99:6280–85. doi: 10.1073/pnas.092324399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Crossman DC, Morton AC, Gunn JP, Greenwood JP, Hall AS, et al. Investigation of the effect of Interleukin-1 receptor antagonist (IL-1ra) on markers of inflammation in non-ST elevation acute coronary syndromes (The MRC-ILA-HEART Study) Trials. 2008;9:8. doi: 10.1186/1745-6215-9-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Allantaz F, Chaussabel D, Stichweh D, Bennett L, Allman W, et al. Blood leukocyte microarrays to diagnose systemic onset juvenile idiopathic arthritis and follow the response to IL-1 blockade. J Exp Med. 2007;204:2131–44. doi: 10.1084/jem.20070070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Lequerre T, Quartier P, Rosellini D, Alaoui F, De Bandt M, et al. Interleukin-1 receptor antagonist (anakinra) treatment in patients with systemic-onset juvenile idiopathic arthritis or adult onset Still disease: preliminary experience in France. Ann Rheum Dis. 2008;67:302–8. doi: 10.1136/ard.2007.076034. [DOI] [PubMed] [Google Scholar]
- 248.Fishman D, Faulds G, Jeffery R, Mohamed-Ali V, Yudkin JS, et al. The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. J Clin Invest. 1998;102:1369–76. doi: 10.1172/JCI2629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Donn R, Alourfi Z, Zeggini E, Lamb R, Jury F, et al. A functional promoter haplotype of macrophage migration inhibitory factor is linked and associated with juvenile idiopathic arthritis. Arthritis Rheum. 2004;50:1604–10. doi: 10.1002/art.20178. [DOI] [PubMed] [Google Scholar]
- 250.Gul A. Behcet’s disease as an autoinflammatory disorder. Curr Drug Targets Inflamm Allergy. 2005;4:81–83. doi: 10.2174/1568010053622894. [DOI] [PubMed] [Google Scholar]
- 251.Stojanov S, Hoffmann F, Kery A, Renner ED, Hartl D, et al. Cytokine profile in PFAPA syndrome suggests continuous inflammation and reduced anti-inflammatory response. Eur Cytokine Netw. 2006;17:90–97. [PubMed] [Google Scholar]
- 252.Long SS. Syndrome of Periodic Fever, Aphthous stomatitis, Pharyngitis, and Adenitis (PFAPA)—What it isn’t. What is it? J Pediatr. 1999;135:1–5. doi: 10.1016/s0022-3476(99)70316-1. [DOI] [PubMed] [Google Scholar]