Abstract
Background
The Atlantic salmon (Salmo salar) immunoglobulin heavy chain (IgH) locus possesses two parallel IgH isoloci (IGH-A and IGH-B), that are related to the genomic duplication event in the family Salmonidae. These duplicated IgH loci in Atlantic salmon provide a unique opportunity to examine the mechanisms of genome diversity and genome evolution of the IgH loci in vertebrates. In this study, we defined the structure of these loci in Atlantic salmon, and sequenced 24 bacterial artificial chromosome (BAC) clones that were assembled into the IGH-A (1.1 Mb) and IGH-B (0.9 Mb) loci. In addition, over 7,000 cDNA clones from the IgH variable (VH) region have been sequenced and analyzed.
Results
The present study shows that the genomic organization of the duplicated IgH loci in Atlantic salmon differs from that in other teleosts and other vertebrates. The loci possess multiple Cτ genes upstream of the Cμ region, with three of the Cτ genes being functional. Moreover, the duplicated loci possess over 300 VH segments which could be classified into 18 families. This is the largest number of VH families currently defined in any vertebrate. There were significant structural differences between the two loci, indicating that both IGH-A and -B loci have evolved independently in the short time after the recent genome duplication approximately 60 mya.
Conclusions
Our results indicate that the duplication of the IgH loci in Atlantic salmon significantly contributes to the increased diversity of the antibody repertoire, as compared with the single IgH locus in other vertebrates.
Background
The adaptive immune system based on somatic recombination of immune receptor genes appeared in vertebrates some 500 million years ago (mya) [1,2]. While jawless vertebrates, such as lamprey and hagfish, assemble their variable lymphocyte receptors (VLRs) through recombination of leucine-rich repeat (LRR) modular units [3-5], jawed vertebrates generate their diverse repertoire of B and T cell antigen receptors through rearrangement of variable-(diversity)-joining (V-(D)-J) gene segments [6,7].
Immunoglobulins (Igs) are key molecules within the jawed vertebrate humoral immune system that are generated by the B cells for defence against a wide variety of pathogens. Igs are composed of two heavy (H) chains and two light (L) chains that are encoded by the IgH locus and IgL locus, respectively. Two different types of genomic rearrangement of the IgH locus have evolved. In cartilaginous fishes, such as sharks and skates, closely linked individual clusters of VH-D-D-J-Constant (CH) gene segments are repeated 100 - 200 times [8,9]. In contrast, in most bony vertebrates (from teleost fishes to mammals), the VH, D, JH, and CH gene segments are in tandem arrays, also known as translocon organisation (VH)n-(D)m-(JH)x-(CH)y [10-13]. The contribution of multiple germ line VH, D and JH gene segments to antibody diversity is magnified by the random rearrangement of these segments in somatic cells [14]. In response to an antigen, mature B cells can change their expressed CH region genes, and the different CH region genes that possess different effector functions. In mammals, there are five Ig classes, named for their CH region component as IgM (μ chain), IgD (δ chain), IgG (γ chain), IgA (α chain) and IgE (ε chain) [9,15]. In teleosts, the predominant serum antibody is IgM, which was the first Ig class identified [13]. Subsequently, IgD was also found, but the teleost IgD gene is expressed as a chimeric transcript that includes the first exon of the IgM gene (Cμ1) [16-20]. It has long been believed that teleost fish possess only IgM and IgD. However, novel Ig classes (IgTs) have recently been found in zebrafish (Danio rerio) (IgZ) [21], rainbow trout (Oncorhynchus mykiss) [22], fugu (Fugu rubripes) (novel IgH) [23] and carp (Cyprinus carpio) (IgM-IgZ chimera) [24] as more sequences have become available.
In the zebrafish and rainbow trout IgH locus, a CH gene (Cτ) of the novel Ig class (IgT) exists upstream of Cμ and Cδ genes, possessing its own D and JH segments [21,22]. This organization of genes in the zebrafish and rainbow trout IgH loci resembles the mouse T cell receptor (TCR) α/δ locus (TRA/TRD) [21]. Similarly, the fugu ortholog of IgT (novel IgH) is also found upstream of Cμ and Cδ genes and this Ig has its own D and JH segments, but the gene organization of the fugu IgT differs significantly from zebrafish and rainbow trout IgTs [23]. However, in catfish, a CH region upstream of Cμ and Cδ genes similar to IgT has not been found [25]. In addition, the catfish IgH locus contains three linked pairs of Cμ and Cδ genes, but only one Cμ and possibly three Cδ genes are functional [25-27]. The three different Cδ gene regions encode heavy chains of membrane and secreted IgD, and although secreted IgD has so far been found only in catfish, it may not contain a functional V-region [26]. Recently, it has been reported that the stickleback (Gasterosteus aculeatus) IgH locus contains three tandem duplicated Cτ, Cμ and Cδ genes separated by VH, D, and JH segments, as well as a fourth Cτ gene in the 3` end of the locus [28,29]. These findings indicate that there is a large amount of variability within the IgH loci among teleosts.
One interesting feature of the Atlantic salmon (Salmo salar) IgH locus is that it possesses two parallel IgH isoloci (IGH-A and IGH-B) [12,30-32], that are related to the tetraploid ancestry of the family Salmonidae [30,33]. A recent study by Shiina et al. (2005) estimated the duplication event to have taken place approx. 60 mya based on sequence divergence of duplicated MHC class I regions of rainbow trout [34] Recently, the presence of two IgH loci have also been demonstrated in rainbow trout by in situ hybridization to rainbow trout chromosomal spreads with IgH-positive BAC clones [22]. However, only approximately 100 kb of the 3' end of one rainbow trout IgH locus has been sequenced to date. Two IgM isotypes were found in Atlantic salmon and brown trout (Salmo trutta), while it has been suggested through gel filtration analysis that rainbow trout and Arctic char (Salvelinus alpinus) possess a single IgM [35]. Moreover, only one Cμ cDNA has been found from a single homozygous rainbow trout, whereas duplicated versions of the rainbow trout Cτ and Cδ genes have been suggested from cDNA variants [22]. These findings suggest that rainbow trout and Arctic char lost an intact IgM in evolution after the genera Salmo, Oncorhynchus and Salvelinus radiated (10 - 18 mya) [35,36]. Thus, determination of the structure of the loci in Atlantic salmon provides a unique opportunity for understanding the evolution of the IgH locus in salmonids.
In this study, to define the structure of the loci in Atlantic salmon, 24 bacterial artificial chromosome (BAC) clones were sequenced and complete IGH-A (1.1 Mb) and IGH-B (0.9 Mb) loci were assembled. In addition, over 7,000 clones from the IgH variable (VH) region cDNAs have been sequenced and analyzed. The present study shows that the Atlantic salmon IgH locus represents the most complex and diverse vertebrate IgH locus characterized to date.
Results
Overall organization of IGH-A and IGH-B
Two loci were assembled from overlapping BAC sequences. IGH-A was assembled from 7 BACs, and IGH-B was assembled using 8 BACs. A few contigs, internal to BACs could not be joined, resulting in two contigs for IGH-A and four contigs for IGH-B, that are separated by small gaps of unknown length in regions of repeated sequences. The sequences in the two IgH loci containing VH and CH regions cover approximately 670 kb (IGH-A) and 710 kb (IGH-B), respectively. Part of a second allele for IGH-A was identified in the assembly of a number of BAC sequences. We noted a similarity of 99.7% over contigs spanning 190 kb sequence (data not shown).
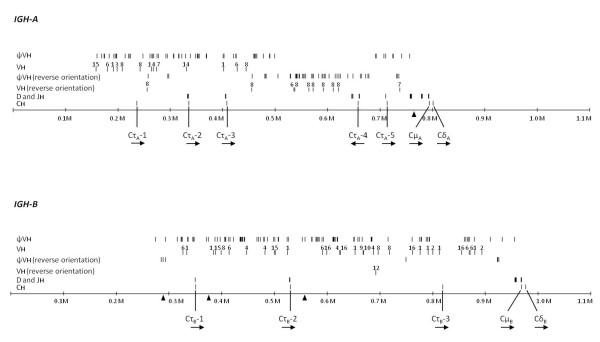
In each of the two IgH loci we identified numerous VH segments, many D and JH segments and several CH gene segments (Table 1, Figure 1, Additional file 1 and Additional file 2). The CH sequences are comprised of three classes, one Cμ and one Cδ in each locus and 3 and 5 complete or partial Cτ sequences, respectively. The Cμ and Cδ sequences are the most 3' elements in both loci while the constant Cτ sequences are distributed throughout the loci. D and JH sequences are generally 5' of the Cμ and Cτ sequences. Most interestingly, the region that contains the VH sequences coincides with the region that contains a large number of "Nhe I" elements, piggyBAC-like sequences that have also been recovered numerous times in the V region of the Atlantic salmon TRA/TRD locus [37].
Table 1.
Summary of CH, D, JH gene segments in the duplicated loci.
| CH | D | JH | in EST | comment |
|---|---|---|---|---|
| IGH-A | ||||
| τA-1 | - | - | no | exon 4 only |
| τA-2 | 2 | 2 | no | exon 1 and 2 |
| τA-3 | 0 | 2 | no | has FS |
| τA-4 | 2 (5'); 3 (3') a | 2 | yes | reverse orientation |
| τA-5 | 1 | 2 | yes | |
| μ A | 6 + 3 b | 5 | yes | |
| δ A | - | - | yes | |
| IGH-B | ||||
| τB-1 | - | 1 | no | has FS |
| τB-2 | 2 | 2 | yes | |
| τB-3 | 0 | 0 | no exon 1 | |
| μ B | 6 | 5 | yes | |
| δ B | - | - | yes | |
a 5' and 3' relative to the τA-4 gene orientation, which is in reverse orientation compared to the rest of the locus.
b The 3 D sequences are located approximately 20 kb 3' from the 6 D sequences and approximately 13 kb 5' of the J sequences.
Figure 1.
Organization of the Atlantic salmon duplicated IgH loci. The sequences in the two IgH loci containing VH and CH regions cover approximately 670 kb (IGH-A) and 710 kb (IGH-B), respectively. The positions of the small gaps are indicated by triangles (IGH-A gap at position 760.2 kb; IGH-B gaps at position 282.8 kb, 372.8 kb and 555.4 kb). The numbers near VH genes indicate the VH genes family numbers. Transcriptional directions for the CH genes are shown by arrowheads. A numerated version of Figure 1 is available in Additional file 1 (IGH-A) and Additional file 2 (IGH-B).
The IGH-A and -B loci show 81-85% sequence identity surrounding the VH sequence region, but internally less identity than 81-85%, indicating significant reorganization involving the VH gene and NheI-elements. Additional file 3 lists identified genes flanking the loci. A dotter plot of IGH-A versus IGH-B is shown in Additional file 4. It is worth noting the similarity between the sequences flanking the two loci as this is in stark contrast to the lack of similarity between the loci themselves.
Constant (CH) regions
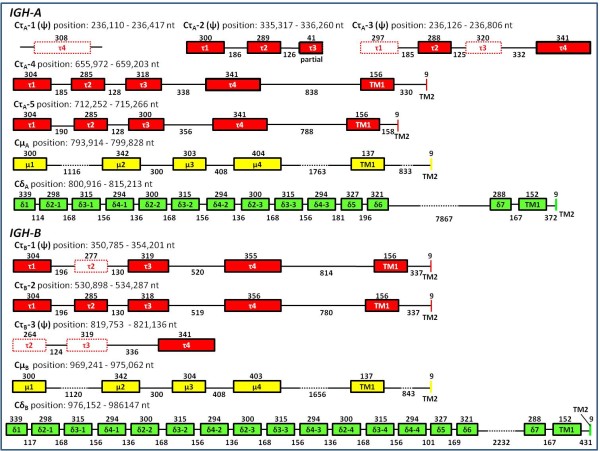
Each locus contains several CH gene sequences, Cμ, Cδ, and Cτ. At the 3' end of each locus is one Cμ sequence followed by one Cδ sequence as previously reported [17,38]. The Cμ and Cδ sequences are approximately 98% similar between loci. Surprisingly, there are several Cτ sequences, 5 in IGH-A and 3 in IGH-B, spread out over each locus (Figure 1). The Cτ sequences in the IGH-A are as follows: starting from the 5' end; 1) CτA-1, partial (most of exon 4 only), 2) CτA-2, partial (5' start to approximately 40 base pairs into exon 3), 3) CτA-3, complete but has a frameshift, 4) CτA-4, complete but in reverse orientation, and 5) CτA-5, complete (Figure 2). In IGH-B there is 1) CτB-1, a complete sequence but with a frameshift, 2) CτB-2, complete, and 3) CτB-3, a partial (missing exon 1) (Figure 2). CτB-1 and CτB-2 are >99% identical. There are a total of two intact Cτ genes in IGH-A and one intact Cτ gene in IGH-B (Table 1 and Figure 2). The alignments of the intact CH gene amino acid sequences are available in Additional file 5. We constructed a phylogenetic tree from the translated sequences of the three intact Cτ genes and other CH genes (see Additional file 6). The three intact Cτ genes clustered within a branch containing teleost Cζ/τ sequences. Interestingly, CτA (CτA-4 and CτA-5) shared a branch with the rainbow trout Cτ, and CτB-2 branched basal to the CτAs/rainbow trout Cτ clade. It has been reported that both duplicated IgD genes in Atlantic salmon have a tandem duplication of Cδ2-Cδ3-Cδ4 [17,38]. However, our present study shows that the number of times exons (Cδ2-Cδ3-Cδ4) are repeated is different between loci, three times in IGH-A and four times in IGH-B (Figure 2). The sequence identity between these repeats of the two loci is very high (>98%), suggesting gene conversion events. The three functional Cτ genes (CτB-2, CτA-4 and CτA-5), the two Cμ genes (CμA and CμB) and Cδ genes (CδA and CδB) have been submitted to Genbank and the accession numbers are listed in Additional file 7.
Figure 2.
Genomic structure of the CH and CH pseudogenes (ψ)genes (not to scale). The coding regions are boxes, and introns are lines. Dashed-line boxes indicate the frameshifts. The continuous regions are indicated by dotted lines. Values indicate the number of base positions.
Diversity (D) segments
D gene sequences were identified through the conserved pattern of their recombination signal sequences (RSS). A total of 25 D sequence genes were identified in the two IgH loci (Figure 1). All segments are flanked on each end by a consensus nonamer-heptamer combination that is separated by 12 base pairs (see Additional file 8). Nine are located in the IGH-A 5' of the Cμ gene, in a group of 6 and a group of 3. There are eight D segments associated with the three Cτ genes (0 for CτA-1 which has no 5' part; 2 for CτA-2 which has exon 1 and 2 only; 0 for CτA-3; 2 for CτA-4 with also 3 on the 3' side of the inverted CτA-4 (which is 5' in the assembly) and 1 for CτA-5) (Table 1). In IGH-B there are six D segments associated with the Cμ gene and two with one of the Cτ genes (CτB-2) (Table 1). Comparison of the sequences of the six D elements in each of the two groups associated with the Cμ genes in IGH-A and IGH-B indicates that the D elements occur in the same order in both loci. The additional group of three in IGH-A is similar to the last three of the groups of six, indicating a duplication event (see Additional file 9).
Joining (JH) segments
Each of the two Cμ genes is preceded by five JH sequences as previously reported [12]. In addition, two JH segments are associated with each Cτ gene, except CτA-1 which has no 5' region and CτB-1 which has only a single JH segment, for a total of seven JH segments for the Cτ genes in IGH-A and three JH segments for the Cτ genes in IGH-B, located just 5' upstream of their respective Cτ gene (Table 1). All segments have a fairly conserved 5' RSS, a nonamer-heptamer combination separated by 24 base pairs (see Additional file 10). A 3' AGGT splice site is found in 18 sequences and a TGGT site in 2 sequences. A translation of the coding sequence reveals a highly conserved FDYWGKGTXVTVS amino acid sequence. One of the JH sequences (JH-τB1-1) is a pseudogene as it is interrupted by a TAG stop codon.
The corresponding JH sequences in IGH-A and IGH-B are identical in coding sequence, except for JH-μA-3 and JH-μB-3 [12], and therefore cannot be distinguished in rearranged products.
Variable (VH) segments
Each locus contains a large number of VH genes and pseudogenes (Figure 1). A total of 153 sequences in IGH-A and 161 sequences in IGH-B were identified as matching VH gene sequences. 99 sequences in IGH-A and 103 sequences in IGH-B were characterized and named. Of these, 23 in IGH-A and 32 in IGH-B have a putative open reading frame (ORF). The deduced amino acid sequences of these VH genes have been submitted to Genbank and the accession numbers are listed in Additional file 7. The alignments of VH genes amino acid sequences are available in Additional file 11. Many other sequences are found only as fragments. VH genes start with a more or less consensus 5'-ATG(C/T)AAA(G/T)-3' octamer sequence [39] located 5' to the site of transcription initiation, and terminate at a nonamer-heptamer RSS. VH genes have a short exon 1 and a long exon 2 sequence. When complete exon 2 sequences (without the RSS) are aligned and 75% identity is applied, 18 families can be distinguished. Representative sequences from the 13 families that were identified in Oncorhynchus mykiss [40,41] align within 13 of these 18 families (Figure 3). The distribution of the VH families between IGH-A and -B is listed in Additional file 12. The number of sequences identified per family varies widely, from a single copy (in family 18) to 18 members (see Additional file 12). The orientation of the VH sequences indicates some rearrangement within the loci and an inversion event is evident when comparing IGH-A and IGH-B sequence (data not shown). This inversion event explains the inverse orientation of the CτA-4 sequence in IGH-A.
Figure 3.
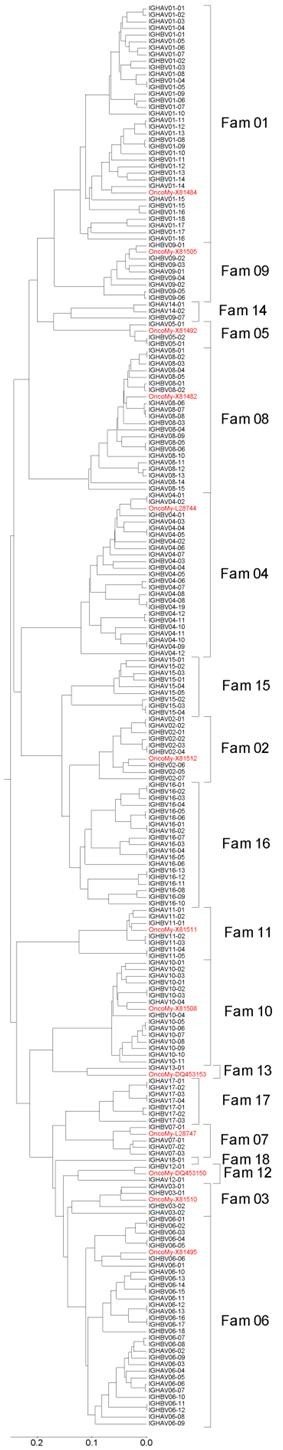
Phylogenetic tree based on nucleotide sequences of Atlantic salmon and rainbow trout VH genes. The tree was constructed from complete exon 2 sequences (without the RSS). These Atlantic salmon VH genes could be grouped into 18 families (Fam 01 - 18), based on >75% nucleotide similarity. Examples from thirteen VH families of rainbow trout [40,41] are shown in red letters.
The use of VH sequences was grouped by family for analysis. VH families are used to a different extent by different constant genes. For example, family 8 is used by CμA much more frequently than by CμB, while the opposite is true for family 6 VH genes (Figure 4). The most commonly used gene families include families 1, 6, and 8, members of which comprise 60% of the putative VH ORFs. Families that contain few members with putative ORFs are also rarely recovered in ESTs.
Figure 4.
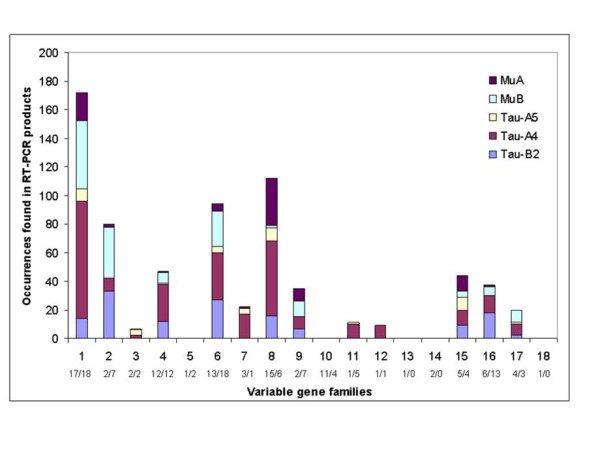
Use of VH sequence families in rearrangements. The numbers below the VH gene family numbers indicate distribution of VH genes in the IGH-A/in the IGH-B.
Rearrangements
A comprehensive set of VH specific primers (178 VH specific primers) were constructed to compliment CH specific primers and ~12 clones from all positive products sequenced to identify expression and rearrangement patterns. More than 7,000 VH-D-JH-CH cloned PCR products amplified from the kidney and spleen of two healthy individuals. We found three main types of rearrangements with VH sequences; those with a Cμ gene, those with a Cτ gene, and those with both Cμ and Cδ exons. However, not all rearrangements involving Cδ include Cμ sequence.
Of 1,872 sequences generated from Cμ-specific primers, located in exon 2, 1,794 contained a conserved sequence in exon 1 of the Cμ sequence and were further analyzed. After removal of identical sequences, confirmation of an ORF in the amplified fragment, and a minimum of 98% match (BLAST) over 30 base pairs in the variable sequence, a total of 225 unique sequences were obtained containing the CμA gene and 358 sequences with the CμB gene. The JH sequences associated with the Cμ genes are not equally used; the middle JH (Cμ - JH-3) occurs most frequently in rearrangements. In fact, the use distribution for the Cμ- JH sequences by CμA and CμB is quite similar (Figure 5).
Figure 5.
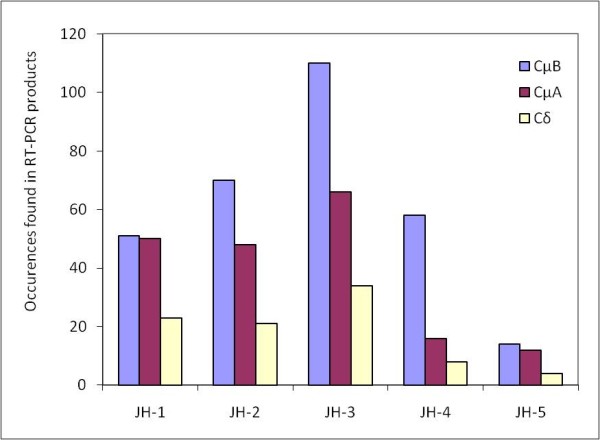
Use of JH sequences in rearrangements. Because many of the Cδ sequences are too similar in sequence to distinguish them unequivocally, the CδA and CδB were put into single columns.
Of 1,852 sequences generated from Cτ-specific primers, 1,555 contained a conserved sequence in exon 1 of the Cτ sequence and were further analyzed. After removal of identical sequences, confirmation of an ORF in the amplified fragment, and a minimum of 98% match (BLAST) over 30 base pairs of variable sequences, a total of 140 unique sequences were obtained containing the CτB-2 gene, 284 sequences with the CτA-4 gene, and 39 sequences with the CτA-5 gene. Interestingly, these three genes are the putatively functional Cτ sequences, with the most frequently used CτA-4 gene which is in an inverted orientation. Two instances were observed where rearrangement took place with CτA-1, which has only the first 2 exons. Both occurred with VH sequences that are 5' of CτA-1. The inverted CτA-4 gene is found associated with VH sequences from both 5' and 3' of the constant sequence. In those instances where the joining sequence can be uniquely identified, we note that the CτB-2 gene is associated with JH-τB1-1, rather than JH-τB2-2, 95% of the time.
cDNA sequences containing Cδ were obtained with primers located in exon 3 of the Cδ sequence. There were 832 sequences of those amplified with the Cδ specific primers that contained Cδ sequences. Approximately 90% of the sequences are chimeric transcripts that contain the first exon of Cμ (Cμ1). However, in approximately 10% of the rearrangements involving Cδ, no Cμ1 sequence and generally no use of the normal splice and recombination signals is evident. A JH and VH sequence is not obvious in all of these cases in the readable sequence and in many of these rearrangements joining takes place from somewhere inside or just after the VH sequence to various distributed sites inside exon 1, 2, and 3 of the Cδ sequence (Table 2). In one recovered rearrangement, the first RSS of the Cμ-D4 sequence and the second RSS of the next Cμ-D5 sequence is used, resulting in the use of two D sequences including a 335 bp intervening genomic sequence (est 007-171). Interestingly, in many of these atypical rearrangements, joining occurs at a short repeat sequence present at both joining ends (see Table 2 for examples), indicative of a homology-directed recombination event. Because many of the Cδ sequences are too similar in sequence to distinguish them unequivocally, we were unable to distinguish the distribution of JH sequences between the CδA and CδB. However, the data still shows a preference for the middle JH by Cδ as seen in CμA and CμB sequences (Figure 5).
Table 2.
Examples of "atypical" rearrangements involving δ.
| EST ID | Sequence repeat | Rearrangement | Join in δa |
|---|---|---|---|
| 001-084 | CTAG | V-(D?)-J-δ | 641(e2) |
| 002-029 | CTAC | V-(D?)-J-δ | 532(e2) |
| 002-179 | GANACAG | V(before RSS)b-δ | 1073(e3) |
| 003-005 | CCA | VB1-12-D-J-VB1-13-δ | 562(e2) |
| 3.007 | V-J-m-δ | 1(e1) | |
| 3.169 | V-(D)-J-δ | 1(e1) | |
| 004-006 | AGTG | V-(D)-δ | 611(e2) |
| 004-078 | C | V(before RSS)-δ | 236(e1) |
| 006-049/55 | δ only ? | 337(e1) | |
| 006.078/82 | δ only ? | 623(e2) | |
| 006-106 | CAG | V-(D?)-J-δ | begin(e1) |
| 007-025 | AGTGANGACACAG | V(before RSS)-δ | 506(e2) |
| 007-171 | CATCAG | D-genomic-D-J-δ | 647(e2) |
| 9.03 | V-?-δ | 235(e1) | |
| 009-052 | CCAC | V-(D?)-J-δ | 294(e1) |
| 11.169 | CTG | V-(D)-J-δ | 1089(e3) |
| 12.059 | GAC | V(before RSS)-δ | 242(e1) |
| 017-108 | ACACA | V(before RSS)-δ | 689(e2) |
| 017-121 | CAGAGG | V(before RSS)-δ | 253(e1) |
a Joining position in exon1, 2, or 3.
b Joining does not involve RSS but initiates earlier.
In up to 20% of the rearrangements the variable sequence was identified as from one locus and the constant sequence from the other locus. For example, Cμa was found rearranged with members of family 1 and 8 from the IGH-B, and Cμb was found rearranged with members of family 1 and 6 from the IGH-A. However, twenty analyzed EST sequences in our EST database [42] that contained a Cμ sequence, contained a VH sequence from the same locus (data not shown). The locus origin of the JH sequences in the rearrangements could not be unambiguously established. Nevertheless, in a number of these rearrangements, the point of crossover appears to be located in the amplified part of the Cμ sequence based on the five single nucleotide differences between the two loci.
Expression of the Atlantic salmon Ig genes
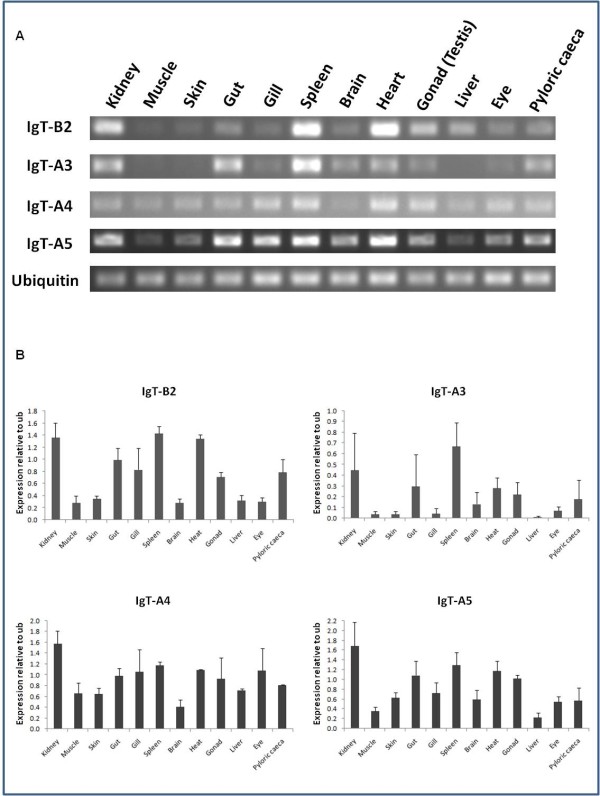
The tissue distribution for four different forms of IgTs, IgT-B2, IgT-A3, IgT-A4 and IgT-A5, was examined by RT-PCR (Figure 6). Figure 6 represents the results from analysis of 12 tissues from 3 different adult individuals. The IgT genes were expressed at high levels in the kidney and spleen. It has been reported that other teleost IgM and IgD genes were also primarily expressed in kidney and spleen [16,18,22]. The kidney and spleen are the major lymphoid organs of teleosts [43,44]. The teleost anterior kidney is a main site for B lymphogenesis, while the teleost posterior kidney provides an environment capable of inducing B cell activation and differentiation into plasma cells [45]. The teleost spleen functions as a major secondary immune organ, as in mammalian species. Mature B cells are abundant at this site, and Ig-secreting cells have been detected from splenic B cells [46]. Interestingly, the IgT genes were also highly expressed in the mucosal tissues, such as the gut or gills (Figure 6). In other tissues, different expression patterns were observed among the different Cτ genes (Figure 6). In particular, the expression pattern of IgT-A3 was quite different from the other three IgTs (Figure 6). Interestingly, the constant region of the IgT-A3 (CτA-3) has a frameshift, and does not have any D segments. In addition, we could not find the IgT-A3 in VH cDNA clones. Therefore, the functionality of IgT-A3 must be questioned. It should also be noted that the expression of IgT-A4 was not detected in one fish. The expression of IgT-B2, IgT-A4 and IgT-A5 was highly expressed in the heart. Hansen et al. (2005) have suggested that the expression of Ig genes in the heart is most likely due to circulating B cells, because salmonid blood is a rich source of leukocytes [22]. In the present study, fish were bled before isolation of tissues but were not exsanguinated. It is therefore assumed that blood remained in the heart.
Figure 6.
Detection of Ig genes in various Atlantic salmon tissues by RT-PCR analysis. (A) Agarose gel electrophoresis with PCR products of the Ig genes. Ubiquitin is an internal control. (B) The level of expression is calculated relative to the ubiquitin (ub) expression level. Data are expressed as mean ± SE of three fish.
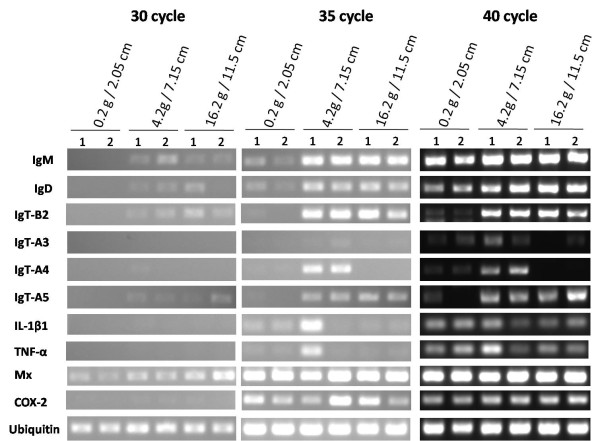
We also examined the expression of Ig genes during three early developmental stages of Atlantic salmon by RT-PCR (Figure 7). IgM and IgD were weakly detected in the earliest stage (0.2 g/2.05 cm), and the expression of both genes increased at later stages of development. Similarly, the expression of both IgT-B2 and IgT-A5 was increased in these stages; however, the expression of these genes was negative or very weak in the earliest stage. Interestingly, the expression of IgT-A4 was not detected in the 16.2 g/11.5 cm fish, and at especially high levels in the 4.2 g/7.15 cm fish. Similarly, IgT-A3 was highly expressed in the 4.2 g/7.15 cm fish, and very weak expression of the gene was observed in only one individual of the 16.2 g/11.5 cm fish.
Figure 7.
Expression of Ig genes during three early developmental stages. PCR amplifications of each primer set were performed for 30, 35 and 40 cycles. Two individuals were studied in each stage. Four immune related genes, il-1β1 (IL-1β1), tnf-α (TNF-α), mx (Mx) and cox-2 (COX-2), were also examined. Ubiquitin is an internal control.
Four immune related genes, il-1β1 (IL-1β1), tnf-α (TNF-α), mx (Mx) and cox-2 (COX-2), were also examined (Figure 7). IL-1β, TNF-α and COX-2 are key mediators of the inflammatory response [47], while Mx proteins are members of the type I interferon (IFN)-inducible genes, and play a role in anti-viral defenses in teleosts [48]. The expression of IL-1β1 and TNF-α was quite similar with strong expression observed in only one individual of the 4.2 g/7.15 cm fish. Only very weak expression of these genes was observed in other fishes at different stages. The expression of Mx increased during the three developmental stages examined, while the expression of COX-2 was quite variable among individual fishes and development stage.
Discussion
Structure of the duplicated IgH loci, IGH-A and -B
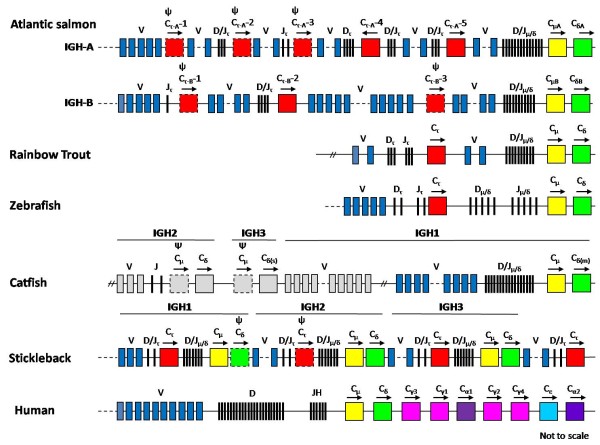
Mammalian IgH loci do not have any CH genes located upstream of Cμ genes. Recently, however, a novel CH gene located upstream of Cμ and Cδ genes has been found in the IgH locus of zebrafish, rainbow trout and fugu [21-23]. In the stickleback IgH locus, a cluster of Cτ-Cμ-Cδ was found duplicated three times in tandem, with an additional Cτ gene in the 3' end of the locus [28,29]. Similarly, the catfish IgH locus contains three linked pairs of Cμ and Cδ genes, but a CH region upstream of Cμ and Cδ genes similar to IgT has not been found in that locus [25,26] (Figure 8). In this study we confirmed that two duplicated IgH loci can functionally coexist and further found that several novel CH (Cτ) genes exist between the VH and Cμ region of the duplicated Atlantic salmon IgH loci, five in IGH-A and three in IGH-B. Therefore, the Atlantic salmon duplicated IgH loci are the only IgH loci so far known, in which multiple Cτ genes are spread out over the region upstream of Cμ. Of these Cτ genes, two Cτ genes in IGH-A (CτA-4 and CτA-5) and one Cτ gene in IGH-B (CτB-2) are functional (Figure 8). These three genes were recovered in cDNA clones associated with VH, D and JH sequences. Interestingly, the CτA-4 gene is in the inverse orientation as has also been observed with the Cα genes in duck and chicken [49,50]. In addition, the inverted CτA-4 gene is found associated with VH and D sequences both 5' and 3' of the constant sequence, and the CτA-4 gene was the most frequently used Cτ gene in our present analysis of VH rearranged cDNAs. Thus, the Atlantic salmon expresses seven kinds of CH genes, three functional Cτ genes (CτB-2, CτA-4 and CτA-5), in addition to the two previously known Cμ genes (CμA and CμB) and Cδ genes (CδA and CδB)[17,31] (Figure 2 and Figure 8).
Figure 8.
Schematic structures of IgH loci of the Atlantic salmon and other vertebrates (not to scale). The Atlantic salmon duplicated IgH loci, IGH-A (670 kb) and IGH-B (710 kb), were completely sequenced in this study. The diagram of the IgH locus of zebrafish (175 kb), stickleback (175 kb) and human (1, 250 kb) are modified from references, [21,28,29] and [14], respectively. To date, only 3' end regions of approximately 100 kb in the rainbow trout [22] and 260 kb in the catfish [25]IgH locus have been sequenced. In the catfish locus, linkage among IGH1, IGH2 and IGH3 was established by restriction mapping and Southern blot analyses [26,82]. The complete sequences of the catfish IGH2, IGH3 and the upstream of IGH1 VH region have not yet been reported. Therefore, these regions are shown in gray boxes. The continuous regions are indicated by dotted lines, while the gap regions are indicated by double slashes (//). Dashed-line boxes indicate the CH pseudogenes (ψ). Transcription directions are shown by arrowhead.
In the zebrafish and fugu IgH loci, VH genes are located upstream of the Dζ1 (zebrafish) or DH1 (fugu) segment [21,23]. However, both IgH loci in Atlantic salmon (Figure 1 and Figure 8) and the rainbow trout IgH locus [22] also have VH genes between the Cτ and Cμ genes, suggesting that these additional VH regions arose in the family Salmonidae. Interestingly, the Atlantic salmon VH region that contains the VH sequences coincides with the region that contains a large number of "Nhe I" elements, piggyBAC-like sequences (Figure 1) that are also highly concentrated in the V region of the Atlantic salmon TRA/TRD locus [37]. We postulate that these elements are involved in the generation and diversification of the large number of V segments of the Atlantic salmon IgH loci and TRA/TRB locus.
The two Atlantic salmon IgH loci contain over 300 VH genes, 99 VH sequences in IGH-A and 103 VH sequences in IGH-B were characterized in this study. These Atlantic salmon VH genes could be grouped into 18 families, based on >75% nucleotide identity (Figure 3). This is the largest number of VH families currently defined in any vertebrate. However, it includes a high number of pseudogenes (>68%). The proportion of pseudogenes is quite different among vertebrate species. The mouse locus contains 56% (110/195) functional genes [51], while human have 36% (44/123) [52]. In teleosts, the zebrafish locus contains 47 VH genes, of which 36 are presumed functional (77%) [21]. In catfish, there are 165 - 200 VH segments as estimated by Southern blotting, and the analysis of 10% of the germline VH genes suggested that approximately 50% are pseudogenes [53]. Although a large number of VH genes in the Atlantic salmon are pseudogenes, some of these pseudo VH genes were expressed. It is unclear whether these pseudo VH genes may play a functional role.
Structural differences between IGH-A and -B
We found some structural differences between the two parallel IgH isoloci in Atlantic salmon. The orientation of the VH sequences indicates some rearrangement within the loci and an approximately 200 kb inversion event is evident in the IGH-A locus when comparing the IGH-A and IGH-B sequence (Figure 1 and Additional file 4). This inversion event, which ranges from approximately position 480 kb to 680 kb (in Figure 1) explains the inverse orientation of the CτA-4 sequence in IGH-A. In addition, three VH families, families 13, 14 and 18 were found only in IGH-A (Figure 3). An additional group of three D segments associated with the Cμ gene exists in the IGH-A locus (see Additional file 9). The amino acid sequence of CμB has an extra cysteine residue near the C-terminal end as described previously by Hordvik et al. (2002) [35] (see Additional file 5). This additional cysteine in CμB is absent in Cμ of rainbow trout [35]. Moreover, the amino acid sequence identities between the rainbow trout Cτ and the Atlantic salmon CτAs (CτA-4 and CτA-5) is higher (80 - 82%) than the similarity between the rainbow trout Cτ and the Atlantic salmon CτB (CτB-2) (75%). In addition, phylogenetic analysis indicates that CτAs are more closely related to the rainbow trout Cτ than to CτB (see Additional file 6). It has been reported that both duplicated IgD genes in Atlantic salmon have a tandem duplication of Cδ2-Cδ3-Cδ4 [17,38]. While our present study shows that these three exons (Cδ2-Cδ3-Cδ4) are indeed repeated three times in IGH-A, they are repeated four times in IGH-B (Figure 2). These observations indicate that both IGH-A and -B loci have evolved independently in the short time after the recent genome duplication. Thus, the existence of two parallel IgH isoloci in Atlantic salmon has contributed to the extensive diversity of the antibody repertoire.
Atypical VH -D-JH -Cδ rearrangements in Atlantic salmon
Analysis of the RT-PCR products amplified using primers specific for Cδ and for VH genes showed that there were a large number of different (unexpected) band sizes and some multiple bands in PCR product from one set of primers (data not shown), indicating that various types of VH-D-JH-Cδ rearrangements exist in Atlantic salmon. The teleost IgD gene is expressed as a chimeric transcript that includes the first exon of IgM gene (Cμ1), because the teleost first exon of IgD (Cδ1) does not contain an appropriate cysteine expected to form the disulfide bond with the L chain [16-20]. Unexpectedly, analysis of the Atlantic salmon IgD VH cDNA clones revealed that approximately 10% of the IgD transcripts do not include the Cμ1 sequence, showing that the Atlantic salmon IgD can be expressed as both a chimera and without the inclusion of the Cμ1 sequence. Recently, it has been reported that either of the Cμ1 and Cδ1 exons could be observed in expressed porcine IgD cDNA sequences (VDJ-Cμ1-hinge region (H)-Cδ2-Cδ3 or VDJ-C δ1-H-Cδ2-C δ3) [54]. The porcine genomic Cδ1 exon is highly similar to the Cμ1 exon with only 4 nucleotides difference. Both the Cδ1 and Cμ1 exons contain three cysteines, only one of which becomes part of the IgD transcript and interacts with the L chain. In contrast, the amino acid sequences of the Atlantic salmon Cδ1 exons are quite different from that of the Cμ1 exons. In addition, the Cδ1 exon lacks the cysteine, similar to other teleosts [17]. Therefore, the functionality of these non-chimeric IgD transcripts is questionable. In fact, these non-chimeric IgD transcripts generally do not use the normal splice and recombination signals. In many of these non-chimeric IgD transcripts, joining takes place from somewhere inside or just after the VH sequence to various distributed sites inside exon 1, 2, or 3 of the Cδ sequence. In many of these instances, a short repeat sequence is present at the two joined ends (Table 2). We cannot at this time exclude the possibility of PCR artifacts contribute to these atypical VH-D-JH-Cδ sequences. However, because they are only observed with Cδ and not with Cτ and Cμ, we assume that it is not very likely. It will be of interest, in future studies, to discover the functions of these unusual IgD transcripts.
Ig genes in Atlantic salmon
Teleost IgM molecules exist in both secreted and membrane-bound forms. The membrane forms of teleost IgM splice the transmembrane (TM1) exon directly to the Cμ3 exon splice site because of lack of a cryptic splice site within Cμ4 exon [55]. The Atlantic salmon and other teleost IgD have been identified as membrane IgD transcripts only [16,18,19,38]. In contrast, the catfish IgH locus encodes both membrane and secreted Cδ genes, and a secreted IgD molecule is identified in the serum of catfish [56]. However, the catfish secreted IgD molecule is encoded by a pseudo IGHM-IGHD locus and may not contain a functional VH region [26]. We found cDNAs encoding secreted and membrane-bound forms of the three functional IgTs in the Atlantic salmon EST database [42], indicating that these three IgTs exist in both the secreted and membrane-bound forms as has also been observed with the IgTs of zebrafish, rainbow trout and fugu [21-23]. Unlike teleost membrane-bound IgM, the membrane-bound forms of these three IgTs transcripts include Cτ4 exons as found in the IgTs of zebrafish and rainbow trout [21,22].
The novel Ig class (IgT), found in zebrafish, rainbow trout and fugu, possesses its own complement of D and JH segments [21-23]. This organization resembles that seen in the mouse TRA/TRD locus [21]. Similarly, each of the three functional Atlantic salmon IgTs has its own complement of D and JH segments (Figure 1 and Figure 8). In mammals, antigen-reactive B cells make antibodies of a single type as according to the "one cell-one antibody" rule [57]. If this rule applies to Atlantic salmon, a single B cell should only express one kind of Ig class from the three different IgTs or IgM, because these Igs have different D and JH segments. Li et al. (2006) found that the gene encoding the rainbow trout IgT was expressed only in IgM- peripheral blood leukocytes (PBLs), indicating that those IgT+IgM- cells represent a unique subset of lymphocytes [58]. Therefore, the Atlantic salmon may possess three different IgT+IgM- B cell and IgT-IgM+ B cell populations. If so, the mechanism of expression of the Atlantic salmon IgTs differs from the mammalian "class switch recombination" mechanism. However, it is not known whether or not the Atlantic salmon B cells only express single IgT or IgM. Further study on the Atlantic salmon B cells will provide new insights into the evolution of B cells in vertebrates.
Expression of the three novel IgT isotypes in Atlantic salmon
The expression of the four innate-immune related genes was variable among individual fishes and development stages, while the expression of Ig genes were quite similar among individual fishes and developmental stages (Figure 7). The expression of the IgM, IgD and four innate immune-related genes were detected in the earliest stage (0.2 g/2.05 cm fish). On the other hand, the expression of the three IgTs was negative or very weak in the earliest stage (Figure 7), suggesting that the Atlantic salmon IgTs are involved in the more mature developmental stage than the IgM and IgD.
As mentioned above, three intact Cτ genes (CτA-4, CτA-5 and CτB-2,) were found in Atlantic salmon. The Cμ and Cδ genes, respectively, show a high degree of amino acid identities between IGH-A and IGH-B (96% ~) [17,31]. In contrast, the three intact Cτ genes (CτB-2, CτA-4 and CτA-5) exhibit significant sequence divergence at the amino acid level not only between loci but also within a locus (IGH-A) (see Additional file 5). The per cent amino acid identities between the CτA (CτA-4 and CτA-5) and CτB (CτB-2) sequences is 75-76, and between CτA-4 and CτA-5 is 87. RT-PCR analyses revealed different tissue distribution patterns among these three IgTs (Figure 6). In addition, the expression pattern of IgT-A4 was quite different from the other two IgTs during the three early developmental stages tested. The expression of IgT-A4 was not detected in the 16.2 g/11.5 cm fish (Figure 7). The high degree of amino acid diversity and the differential expression patterns among three Cτ genes suggest that the three different IgTs may have different functions.
Interestingly, in our present RT-PCR analysis, the IgTs, especially IgT-A5, were highly expressed in the mucosal tissues, including gut or gills (Figure 6). The fugu IgT mRNA was also strongly expressed in goblet cells of the intestine and gill epithelium [23]. In mammals, the mucosal surfaces constitute the first defensive line against invading microbial pathogens. The specific immunological defense at this site is primarily mediated by IgA antibodies [59,60]. Although the structure of the Xenopus laevis IgX is quite different from the mammalian IgA, the IgX is considered an analog of IgA because its association with the mucosae of the intestine resembles that of IgA [61]. In teleosts, no typical mucosal Ig class such as the IgA and IgX has been identified, but small amounts of IgM is present in gut mucus of several teleosts [62-64]. However, Hatten et al. (2001) have reported that IgM is not present in gut mucus of Atlantic salmon, and the gut mucus contains a large amount of proteolytic enzymes able to degrade serum IgM [65]. Their results suggested that antibodies related to the gut of the Atlantic salmon should be of another, yet unidentified, Ig class. Because the three new IgTs identified in our present study were found to be highly expressed in mucosal tissues, these IgTs might form the mucosal Ig class in Atlantic salmon.
Conclusions
The present study shows that the genomic organization of the duplicated IgH loci in Atlantic salmon differs from that in other teleosts. The loci possess multiple Cτ genes upstream of the Cμ region, with three of the Cτ genes being functional. Moreover, the duplicated loci possess over 300 VH segments which could be classified into 18 families. This is the largest number of VH families currently defined in any vertebrate. Our results indicate that the duplication of the IgH loci in Atlantic salmon contributes heavily to the increases in diversity of the antibody repertoire, as compared with the single IgH locus in other vertebrates. Previous studies of the Atlantic salmon TRA/TRD and TRG loci revealed that Atlantic salmon clearly has one of the largest TCR repertoires known for any vertebrate [66,67]. Much more comprehensive analyses of Ig and TCR repertoires in Atlantic salmon can use the method of Warren et al. (2009) [68]. Atlantic salmon have both freshwater and saltwater phases in their life cycles. Therefore, Atlantic salmon is exposed to a wider variety of pathogens from these two different environments. Thus, the large diversity of antigen receptors in Atlantic salmon may have evolved to protect against such a wide variety of pathogens. Further study on the biological significance of the Igs and TCRs will provide unique insight into the evolution of the adaptive immune system in vertebrates.
Methods
Sequencing of Atlantic salmon IGH-A and -B loci
The Atlantic salmon IGH-A and -B loci were isolated and sequenced as previously described [66,67]. An Atlantic salmon BAC library (CHORI-214), constructed from a Norwegian aquaculture male strain, was obtained from BACPAC Resources, Children's Hospital Oakland Research Institute (CHORI) [69]. Six BAC library filters were hybridized with three (Cμ, Cδ and Cτ) 70-mer oligo probes (Integrated DNA Technologies) that were 5`-end-labeled with 32P-ATP using T4 polynucleotide kinase (Invitrogen). The labeled probes were added to BAC filters that had been pre-hybridized at 65°C for 4 h (5 × SSC, 5 × Denhardt's, 0.1% SDS). The hybridization was carried out overnight at 65°C. Three washes were performed, each for 30 min at 50°C; the first consisting of 2 × SSC and 0.1% SDS, and the second and third each consisting of 1 × SSC and 0.1% SDS. Filters were visualized using BioMax film (Kodak). BAC clones were chosen based on the physical BAC fingerprint map for Atlantic salmon [70] that is publicly available on the internet Contig Explorer (iCE) version 3.5 [71]. The BAC end sequence information, that is available in ASalBase [72], was also used for selection of the BAC clones. BAC shotgun libraries were constructed and sequenced on an ABI 3730 DNA sequencer, each of which was assembled using PHRED and PHRAP [73,74] and Consed [75].
The Dotter program [76] was used extensively to identify sequence elements. Sequence alignments were performed with ClustalW [77] and phylogenetic trees generated with MEGA3.1 [78] using the Unweighted Pair Group Method with Arithmetic Mean (UPGMA), pairwise deletion, and a p-distance model. Gene families were defined at 75% identity, as per the World Health Organization-International Union of Immunological Societies (WHO-IUIS) Nomenclature Subcommittee guidelines [79]. Genes flanking the loci were identified for IGH-B with the Digit Web Server [80].
The sequence of the IGH-A and IGH-B loci were deposited in Genbank under accession numbers, IGH-A [GenBank:GU129139], IGH-B [GenBank:GU129140] and the other IGH-A allele [GenBank:GU321975 - GU321980]. The nucleotide sequences for the Cμ, Cδ and Cτ probes were based on EST sequence data and are provided in Additional file 13.
Cloning and sequencing of VH cDNAs
Adult Atlantic salmon (Mowi stock) tissues were obtained from the Department of Fisheries and Oceans (Robert Devlin, WestVan Lab., West Vancouver, British Columbia). Adult fish were euthanized, followed by rapid dissection of tissues. Tissues were flash frozen in liquid nitrogen or dry ice and stored at -80°C until RNA extraction. Total RNA was extracted from the kidney and spleen of two healthy individuals using TRIzol reagent (Invitrogen). Purified total RNA (1.0 μg) was reverse transcribed with SuperScript™ II (Invitrogen) using oligo (dT)15 primer as described in the manufacture's protocol. The cDNAs were synthesized in 25 μl reactions incubated at 42°C for 90 min and the transcriptase heat-inactivated at 70°C for 30 min. Equal amounts of each cDNA were combined and the mixture used as PCR template.
One hundred seventy eight (178) forward primers were designed from VH sequences identified for one or several VH sequences per primer (generally as part of a family). The reverse primers were designed from the consensus sequence of Cμ, Cδ, and Cτ. For increased specificity, nested primers were also designed from the consensus sequence of IGH-A and -B locus for Cμ and Cδ genes, and from the four different forms of Cτ genes. These nested primers were located in the first exon of τ (Cτ1), and in the second exon of μ (Cμ2) and δ (Cδ2). The PCR primers used in this study are shown in Additional file 13. PCRs were performed using GoTaq DNA polymerase (Promega) with an initial denaturation of 2 min at 95°C and then 30 cycles at 30 s of denaturation at 95°C, 30 s of annealing at 55°C, and 1 min of extension at 72°C. PCR products were cloned into pCR2.1 (TA Cloning Kit, Invitrogen) according to the manufacturer's protocol. Twelve (12) clones from each positive PCR product were sequenced as described above. Sequences obtained from Cμ and Cτ genes were sequenced in single forward direction, while the longer sequences that include the Cδ gene were sequenced in both forward and reverse directions. We only analyzed the results where the two reads could form a contig with a 100% overlap. The cDNA sequences were BLAST searched against the VH, JH and CH gene sequences to identify their presence in the clones.
RT-PCR analysis of Ig genes expression
Twelve different Atlantic salmon tissues from three different adult fish were provided by the Department of Fisheries and Oceans. Immature and juvenile stages of fish were provided by Marine Harvest Canada (Big Tree Creek Hatchery, Sayward, B.C.) Total RNA was extracted and reverse transcribed using the method described above for tissues (kidney and spleen) from two individuals for three different development stages, as well as tissues from three individual adult fish (kidney, muscle, skin, gut, gill, spleen, brain, heart, gonad, liver, eye and pyloric caeca). PCR primers were designed from the consensus sequence of IGH-A and IGH-B locus for Cμ and Cδ genes, and four different forms of Cτ genes. The primers correspond to sequences in the first and third exon for Cμ and Cτ genes, and in the fifth and seventh exon for Cδ gene. Ubiquitin was used as internal positive control. In addition, four immune related genes, interleukin (IL)-1β1, tumour necrosis factor (TNF)-α, Mx and cyclo-oxygenase (COX)-2, were also examined. The PCR primers used in this study are shown in Additional file 13. PCR was performed using GoTaq DNA polymerase (Promega) with an initial denaturation step of 2 min at 95°C and then 30 or 35 cycles as follows: 30 s of denaturation at 95°C, 30 s of annealing at 55°C and 1 min of extension at 72°C. The PCR products derived from each primer set were TA-cloned and confirmed by sequencing. The PCR products were electrophoresed on a 1.0% agarose gel. The intensity of the amplification bands was semi-quantitatively measured using ImageJ software [81], and divided by the intensity of the respective ubiquitin signals.
Abbreviations
IgH: immunoglobulin heavy chain locus; IgL: Immunoglobulin light chain locus; IGH-A and -B: Atlantic salmon duplicated immunoglobulin heavy chain loci A and B; VH: Variable region of immunoglobulin heavy chains; D: Diversity region of immunoglobulin heavy chains; JH: Joining region of immunoglobulin heavy chains; CH: Constant region of immunoglobulin heavy chains; Cμ: μ chain constant region; Cδ: δ chain constant region; Cγ: γ chain constant region; Cα: α chain constant region; Cε: ε chain constant region; Cζ: ζ chain constant region; Cτ: τ chain constant region; TRB/TRD: T cell receptor (TCR) α/δ locus, TRG, TCR γ locus.
Authors' contributions
MY performed the BAC library preparation, VH cDNA cloning, RT-PCR studies and drafted the manuscript. JdB performed the data analysis and drafted the manuscript. KRVS performed the VH cDNA cloning and RT-PCR studies. GAC, LM, AM, SS performed the BAC library preparation and DNA sequencing for the project. WSD contributed to the project planning and direction. BFK contributed to the planning, design, and direction of the project. All authors read and approved the final manuscript.
Supplementary Material
Features of the IGH-A. Table listing genes and pseudogenes identified in the IGH-A.
Features of the IGH-B. Table listing genes and pseudogenes identified in the IGH-B.
The identified genes flanking the loci. Table listing genes flanking the loci identified with the Digit Web Server (http://synthetic-biology.jp/sw/pic/en/crib151s2rib151s72i/).
Dotter plot of locus A (IGH-A) versus locus B (IGH-B). This file contains a dotter plot of IGH-A versus IGH-B.
Alignment of amino acid sequences encoded by (A) Cτ, (B) C μ and (C) Cδ. This file contains multiple sequence alignments of amino acid sequences encoded by (A) Cτ, (B) C μ and (C) Cδ obtained from ClustalW. Identical residues are shown as dots (.) and gaps are shown as hyphens (-).
Phylogenic relationships for the CH genes in various species. Phylogenic tree showing the relationship of the CH genes amino acid sequences of CH2 and CH3 domains of α, human δ and γ; CH3 and CH4 of μ, ζ/τ, ε, and duck α; CH4 and CH5 of new antigen receptor (NAR); CH5 and CH6 of ω, NARC and teleost δ. The tree was constructed with the MEGA 4 package by neighbor-joining (NJ) method and bootstrap values for replicated 1,000 were represented by percentages on the edge of node. The bootstrap values greater than 50% are presented. The scale bar indicates the branch length. Genbank accession numbers are as follows: α: duck [GenBank:AAA68606], human [GenBank:AAC82528]. δ: Atlantic salmon δA and δB [GenBank:AF278717; AF141605], catfish [GenBank:T18537], fugu [GenBank:BAD34542], zebrafish [GenBank:CAI11477], Xenopus [GenBank:DQ350886], human [GenBank:AAA52771]. ε: human [GenBank:AAB59395], opossum (Monodelphis domestica) [GenBank:AAC79674]. γ1: human [GenBank:AAC82527]. γ3: mouse [GenBank:AAB59697]. μ: Atlantic salmon μA and μB, [GenBank:AAB24064; AAF69490], bowfin (Amia calva) [GenBank:ACU12456], carp [GenBank:AB004105], catfish [GenBank:M27230], gar (Lepisosteus osseus) [GenBank:U12455], ladyfish (Elops saurus) [GenBank:M26182] lungfish (Protopterus aethiopicus) [GenBank:AF437724] nurse shark (Ginglymostoma cirratum) [GenBank:M92851], rainbow trout [GenBank:X83372], skate (Leucoraja erinacea) [GenBank:M29679], sturgeon (Acipenser baeri) [GenBank:Y13253], zebrafish [GenBank:AY643753], Xenopus [GenBank:M20484], chicken [GenBank:X01613], mouse [GenBank:J00443], human [GenBank:X14940]. υ: Xenopus [GenBank:X15114]. ω: lungfish [GenBank:AF437727], sandbar shark (Carcharhinus plumbeus) [GenBank:CPU40560]. NAR: nurse shark [GenBank:GCU51450]. NARC: nurse shark [GenBank:GCU18701]. ζ/τ: grass carp (Ctenopharyngodon idella) [GenBank:DQ489733], rainbow trout τ1 and τ2 [GenBank:AAW66978] and [GenBank:AAW66981], perch (Siniperca chuatsi), [GenBank:DQ016660], zebrafish [GenBank:AY643752].
Genbank accession numbers for deduced amino acid sequences of CH and VH domains. Table listing the accession numbers for deduced amino acid sequences of CH and VH domains.
Alignment of D sequences. This file contains a multiple sequence alignment of D sequences obtained from ClustalW.
Phylogenic trees showing the relationship between the (A) D and (B) JH sequences. This file contains phylogenic trees for (A) D and (B) JH genes.
Alignment of JH sequences. This file contains a multiple sequence alignment of JH sequences obtained from ClustalW.
Alignment of amino acid VH sequences. This file contains a multiple sequence alignment of amino acid VH sequences obtained from ClustalW. Identical residues are shown as dots (.) and gaps are shown as hyphens (-).
Distribution of variable (VH) families in the two IgH loci. Table showing the number of sequences identified per family.
PCR primers and oligo probes. Table listing the PCR primers and oligo probes used in this study.
Contributor Information
Motoshige Yasuike, Email: myasuike@uvic.ca.
Johan de Boer, Email: jdboer@uvic.ca.
Kristian R von Schalburg, Email: krvs@uvic.ca.
Glenn A Cooper, Email: glennacopp@gmail.com.
Linda McKinnel, Email: mckinl@uvic.ca.
Amber Messmer, Email: amber_messmer@hotmail.com.
Stacy So, Email: stacs1@shaw.ca.
William S Davidson, Email: wdavidso@sfu.ca.
Ben F Koop, Email: bkoop@uvic.ca.
Acknowledgements
This work was supported by Genome Canada, Genome BC and by the Natural Sciences and Engineering Research Council of Canada (B.K., W.D). We would like to thank Robert Devlin (Department of Fisheries and Oceans) for providing adult Atlantic salmon tissues; Marine Harvest Canada for providing immature and juvenile stages of fish; Krzysztof P. Lubieniecki (Simon Fraser University) for his assistance in construction of BAC shotgun libraries; Eric Rondeau (University of Victoria) for his assistance in obtaining tissues for RT-PCR analysis; Dan Sanderson (University of Victoria) for his help in preparing the manuscript.
References
- Cannon PJ, Haire NR, Rast PJ, Litman GW. The phylogenetic origins of the antigen-binding receptors and somatic diversification mechanisms. Immunol Rev. 2004;200(1):12–22. doi: 10.1111/j.0105-2896.2004.00166.x. [DOI] [PubMed] [Google Scholar]
- Pancer Z, Cooper MD. The evolution of adaptive immunity. Annu Rev Immunol. 2006;24(1):497–518. doi: 10.1146/annurev.immunol.24.021605.090542. [DOI] [PubMed] [Google Scholar]
- Alder MN, Rogozin IB, Iyer LM, Glazko GV, Cooper MD, Pancer Z. Diversity and function of adaptive immune receptors in a jawless vertebrate. Science. 2005;310(5756):1970–1973. doi: 10.1126/science.1119420. [DOI] [PubMed] [Google Scholar]
- Pancer Z, Amemiya CT, Ehrhardt GRA, Ceitlin J, Larry Gartland G, Cooper MD. Somatic diversification of variable lymphocyte receptors in the agnathan sea lamprey. Nature. 2004;430(6996):174–180. doi: 10.1038/nature02740. [DOI] [PubMed] [Google Scholar]
- Pancer Z, Saha NR, Kasamatsu J, Suzuki T, Amemiya CT, Kasahara M, Cooper MD. Variable lymphocyte receptors in hagfish. Proc Natl Acad Sci USA. 2005;102(26):9224–9229. doi: 10.1073/pnas.0503792102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassing CH, Swat W, Alt FW. The mechanism and regulation of chromosomal V(D)J recombination. Cell. 2002;109(2):S45–S55. doi: 10.1016/S0092-8674(02)00675-X. [DOI] [PubMed] [Google Scholar]
- Schlissel MS. Regulating antigen-receptor gene assembly. Nat Rev Immunol. 2003;3(11):890–899. doi: 10.1038/nri1225. [DOI] [PubMed] [Google Scholar]
- Dooley H, Flajnik MF. Antibody repertoire development in cartilaginous fish. Dev Comp Immunol. 2006;30(1-2):43–56. doi: 10.1016/j.dci.2005.06.022. [DOI] [PubMed] [Google Scholar]
- Hsu E, Pulham N, Rumfelt LL, Flajnik MF. The plasticity of immunoglobulin gene systems in evolution. Immunol Rev. 2006;210:8–26. doi: 10.1111/j.0105-2896.2006.00366.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amemiya CT, Litman GW. Complete nucleotide sequence of an immunoglobulin heavy-chain gene and analysis of immunoglobulin gene organization in a primitive teleost species. Proc Natl Acad Sci USA. 1990;87(2):811–815. doi: 10.1073/pnas.87.2.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghaffari SH, Lobb CJ. Organization of immunoglobulin heavy chain constant and joining region genes in the channel catfish. Mol Immunol. 1992;29(2):151–159. doi: 10.1016/0161-5890(92)90096-G. [DOI] [PubMed] [Google Scholar]
- Hordvik I, De Vries Lindstrom C, Voie AM, Jacob ALJ, Endresen C. Structure and organization of the immunoglobulin m heavy chain genes in Atlantic Salmon, Salmo salar. Mol Immunol. 1997;34(8-9):631–639. doi: 10.1016/S0161-5890(97)00071-0. [DOI] [PubMed] [Google Scholar]
- Warr GW. The immunoglobulin genes of fish. Dev Comp Immunol. 1995;19(1):1–12. doi: 10.1016/0145-305X(94)00052-H. [DOI] [PubMed] [Google Scholar]
- Abbas KA, Lichtman HA, Pillai S. Cellular and Mol Immunol. 6. Philadelphia: WB Saunders; 2007. Lymphocyte development and the rearrangement and expression of antigen receptor genes; pp. 153–187. 6th edition edn. [Google Scholar]
- Snapper CM, Marcu KB, Zelazowski P. The immunoglobulin class switch: beyond "accessibility". Immunity. 1997;6(3):217–223. doi: 10.1016/S1074-7613(00)80324-6. [DOI] [PubMed] [Google Scholar]
- Hirono I, Nam BH, Enomoto J, Uchino K, Aoki T. Cloning and characterisation of a cDNA encoding Japanese flounder Paralichthys olivaceus IgD. Fish Shellfish Immunol. 2003;15(1):63–70. doi: 10.1016/S1050-4648(02)00139-0. [DOI] [PubMed] [Google Scholar]
- Hordvik I, Thevarajan J, Samdal I, Bastani N, Krossøy B. Molecular cloning and phylogenetic analysis of the Atlantic salmon immunoglobulin D gene. Scand J Immunol. 1999;50(2):202–210. doi: 10.1046/j.1365-3083.1999.00583.x. [DOI] [PubMed] [Google Scholar]
- Saha NR, Suetake H, Kikuchi K, Suzuki Y. Fugu immunoglobulin D: a highly unusual gene with unprecedented duplications in its constant region. Immunogenetics. 2004;56(6):438–447. doi: 10.1007/s00251-004-0693-y. [DOI] [PubMed] [Google Scholar]
- Stenvik J, Jørgensen TØ. Immunoglobulin D (IgD) of Atlantic cod has a unique structure. Immunogenetics. 2000;51(6):452–461. doi: 10.1007/s002510050644. [DOI] [PubMed] [Google Scholar]
- Wilson M, Bengten E, Miller NW, Clem LW, Du Pasquier L, Warr GW. A novel chimeric Ig heavy chain from a teleost fish shares similarities to IgD. Proc Natl Acad Sci USA. 1997;94(9):4593–4597. doi: 10.1073/pnas.94.9.4593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danilova N, Bussmann J, Jekosch K, Steiner LA. The immunoglobulin heavy-chain locus in zebrafish: identification and expression of a previously unknown isotype, immunoglobulin Z. Nat Immunol. 2005;6(3):295–302. doi: 10.1038/ni1166. [DOI] [PubMed] [Google Scholar]
- Hansen JD, Landis ED, Phillips RB. Discovery of a unique Ig heavy-chain isotype (IgT) in rainbow trout: Implications for a distinctive B cell developmental pathway in teleost fish. Proc Natl Acad Sci USA. 2005;102(19):6919–6924. doi: 10.1073/pnas.0500027102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savan R, Aman A, Sato K, Yamaguchi R, Sakai M. Discovery of a new class of immunoglobulin heavy chain from fugu. European Journal of Immunology. 2005;35(11):3320–3331. doi: 10.1002/eji.200535248. [DOI] [PubMed] [Google Scholar]
- Savan R, Aman A, Nakao M, Watanuki H, Sakai M. Discovery of a novel immunoglobulin heavy chain gene chimera from common carp (Cyprinus carpio L.) Immunogenetics. 2005;57(6):458–463. doi: 10.1007/s00251-005-0015-z. [DOI] [PubMed] [Google Scholar]
- Bengtén E, Quiniou S, Hikima J, Waldbieser G, Warr G, Miller N, Wilson M. Structure of the catfish IGH locus: analysis of the region including the single functional IGHM gene. Immunogenetics. 2006;58(10):831–844. doi: 10.1007/s00251-006-0139-9. [DOI] [PubMed] [Google Scholar]
- Bengtén E, Quiniou SMA, Stuge TB, Katagiri T, Miller NW, Clem LW, Warr GW, Wilson M. The IgH locus of the channel catfish, Ictalurus punctatus, contains multiple constant region gene sequences: different genes encode heavy chains of membrane and secreted IgD. J Immunol. 2002;169(5):2488–2497. doi: 10.4049/jimmunol.169.5.2488. [DOI] [PubMed] [Google Scholar]
- Bengtén E, Clem LW, Miller NW, Warr GW, Wilson M. Channel catfish immunoglobulins: Repertoire and expression. Dev Comp Immunol. 2006;30(1-2):77–92. doi: 10.1016/j.dci.2005.06.016. [DOI] [PubMed] [Google Scholar]
- Bao Y, Wang T, Guo Y, Zhao Z, Li N, Zhao Y. The immunoglobulin gene loci in the teleost Gasterosteus aculeatus. Fish Shellfish Immunol. 2010;28:40–8. doi: 10.1016/j.fsi.2009.09.014. [DOI] [PubMed] [Google Scholar]
- Gambón-Deza F, Sánchez-Espinel C, Magadán-Mompó S. Presence of an unique IgT on the IGH locus in three-spined stickleback fish (Gasterosteus aculeatus) and the very recent generation of a repertoire of VH genes. Dev Comp Immunol. 2010;34:114–22. doi: 10.1016/j.dci.2009.08.011. [DOI] [PubMed] [Google Scholar]
- Hordvik I. The impact of ancestral tetraploidy on antibody heterogeneity in salmonid fishes. Immunol Rev. 1998;166(1):153–157. doi: 10.1111/j.1600-065X.1998.tb01260.x. [DOI] [PubMed] [Google Scholar]
- Hordvik I, Voie AM, Glette J, Male R, Endresen C. Cloning and sequence analysis of two isotypic IgM heavy chain genes from Atlantic salmon, Salmo salar L. Eur J Immunol. 1992;22(11):2957–2962. doi: 10.1002/eji.1830221130. [DOI] [PubMed] [Google Scholar]
- Solem ST, Hordvik I, Killie JEA, Warr GW, Jørgensen TØ. Diversity of the immunoglobulin heavy chain in the Atlantic salmon (Salmo salar L.) is contributed by genes from two parallel IgH isoloci. Dev Comp Immunol. 2001;25(5-6):403–417. doi: 10.1016/S0145-305X(01)00008-8. [DOI] [PubMed] [Google Scholar]
- Ohno S. Evolution by gene duplication. New York Springer-Verlag; 1970. [Google Scholar]
- Shiina T, Dijkstra JM, Shimizu S, Watanabe A, Yanagiya K, Kiryu I, Fujiwara A, Nishida-Umehara C, Kaba Y, Hirono I. et al. Interchromosomal duplication of major histocompatibility complex class I regions in rainbow trout (Oncorhynchus mykiss), a species with a presumably recent tetraploid ancestry. Immunogenetics. 2005;56(12):878–893. doi: 10.1007/s00251-004-0755-1. [DOI] [PubMed] [Google Scholar]
- Hordvik I, Berven FS, Solem ST, Hatten F, Endresen C. Analysis of two IgM isotypes in Atlantic salmon and brown trout. Mol Immunol. 2002;39(5-6):313–321. doi: 10.1016/S0161-5890(02)00114-1. [DOI] [PubMed] [Google Scholar]
- Oakley TH, Phillips RB. Phylogeny of Salmonine fishes based upon growth hormone introns: Atlantic (Salmo) and Pacific (Oncorhynchus) salmon are not sister taxa. Mol Phylogenet Evol. 1999;11:381–393. doi: 10.1006/mpev.1998.0599. [DOI] [PubMed] [Google Scholar]
- de Boer J, Yazawa R, Davidson WS, Koop BF. Bursts and horizontal evolution of DNA transposons in the speciation of pseudotetraploid salmonids. BMC Genomics. 2007;8(1):422. doi: 10.1186/1471-2164-8-422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hordvik I. Identification of a novel immunoglobulin δ transcript and comparative analysis of the genes encoding IgD in Atlantic salmon and Atlantic halibut. Mol Immunol. 2002;39(1-2):85–91. doi: 10.1016/S0161-5890(02)00043-3. [DOI] [PubMed] [Google Scholar]
- Landolfi NF, Yin XM, Capra JD, Tucker PW. A conserved heptamer upstream of the IgH promoter region octamer can be the site of a coordinate protein-DNA interaction. Nucl Acids Res. 1988;16(12):5503–5514. doi: 10.1093/nar/16.12.5503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown G, Kaattari I, Kaattari S. Two new Ig VH gene families in Oncorhynchus mykiss. Immunogenetics. 2006;58(11):933–936. doi: 10.1007/s00251-006-0149-7. [DOI] [PubMed] [Google Scholar]
- Roman T, Charlemagne J, Matsunaga T, Andersson E, Bengtén E, Pilström L, Hansen J, Kaattari S. Unified nomenclature of Ig VH genes in rainbow trout (Oncorhynchus mykiss): definition of elevenVH families. Immunogenetics. 1996;43(5):325–326. doi: 10.1007/BF02441003. [DOI] [PubMed] [Google Scholar]
- Koop BF, von Schalburg KR, Leong J, Walker N, Lieph R, Cooper GA, Robb A, Beetz-Sargent M, Holt R, Moore R. et al. A salmonid EST genomic study: genes, duplications, phylogeny and microarrays. BMC Genomics. 2008;9:545. doi: 10.1186/1471-2164-9-545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Press CM, Evensen Ø. The morphology of the immune system in teleost fishes. Fish Shellfish Immunol. 1999;9(4):309–318. doi: 10.1006/fsim.1998.0181. [DOI] [Google Scholar]
- Zapata A, Diez B, Cejalvo T, Gutiérrez-de Frías C, Cortés A. Ontogeny of the immune system of fish. Fish Shellfish Immunol. 2006;20(2):126–136. doi: 10.1016/j.fsi.2004.09.005. [DOI] [PubMed] [Google Scholar]
- Zwollo P, Cole S, Bromage E, Kaattari S. B cell heterogeneity in the teleost kidney: evidence for a maturation gradient from anterior to posterior kidney. J Immunol. 2005;174(11):6608–6616. doi: 10.4049/jimmunol.174.11.6608. [DOI] [PubMed] [Google Scholar]
- Zwollo P, Haines A, Rosato P, Gumulak-Smith J. Molecular and cellular analysis of B-cell populations in the rainbow trout using Pax5 and immunoglobulin markers. Dev Comp Immunol. 2008;32(12):1482–1496. doi: 10.1016/j.dci.2008.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Secombes CJ, Wang T, Hong S, Peddie S, Crampe M, Laing KJ, Cunningham C, Zou J. Cytokines and innate immunity of fish. Dev Comp Immunol. 2001;25(8-9):713–723. doi: 10.1016/S0145-305X(01)00032-5. [DOI] [PubMed] [Google Scholar]
- Leong CJA, Trobridge DG, Kim HYC, Johnson M, Simon B. Interferon-inducible Mx proteins in fish. Immunol Rev. 1998;166(1):349–363. doi: 10.1111/j.1600-065X.1998.tb01275.x. [DOI] [PubMed] [Google Scholar]
- Lundqvist ML, Middleton DL, Hazard S, Warr GW. The immunoglobulin heavy chain locus of the duck. Genomic organization and expression of D, J, and C region genes. J Biol Chem. 2001;276(50):46729–46736. doi: 10.1074/jbc.M106221200. [DOI] [PubMed] [Google Scholar]
- Zhao Y, Rabbani H, Shimizu A, Hammarström L. Mapping of the chicken immunoglobulin heavy-chain constant region gene locus reveals an inverted α gene upstream of a condensed upsilon gene. Immunology. 2000;101(3):348–353. doi: 10.1046/j.1365-2567.2000.00106.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston CM, Wood AL, Bolland DJ, Corcoran AE. Complete sequence assembly and characterization of the C57BL/6 mouse Ig heavy chain V region. J Immunol. 2006;176(7):4221–4234. doi: 10.4049/jimmunol.176.7.4221. [DOI] [PubMed] [Google Scholar]
- Matsuda F, Ishii K, Bourvagnet P, Kuma K, Hayashida H, Miyata T, Honjo T. The complete nucleotide sequence of the human immunoglobulin heavy chain variable region locus. J Exp Med. 1998;188(11):2151–2162. doi: 10.1084/jem.188.11.2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang F, Ventura-Holman T, Waldbieser GC, Lobb CJ. Structure, genomic organization, and phylogenetic implications of six new VH families in the channel catfish. Mol Immunol. 2003;40(5):247–260. doi: 10.1016/S0161-5890(03)00143-3. [DOI] [PubMed] [Google Scholar]
- Zhao Y, Pan-Hammarstrom Q, Kacskovics I, Hammarstrom L. The porcine Ig δ gene: Unique chimeric splicing of the first constant region domain in its heavy chain transcripts. J Immunol. 2003;171(3):1312–1318. doi: 10.4049/jimmunol.171.3.1312. [DOI] [PubMed] [Google Scholar]
- David AR, Melanie RW, Norman WM, Clem LW, Gregory WW, David AR, Melanie RW, Normon WM, Clem LW, Gregory WW. Evolutionary variation of immunoglobulin μ heavy chain RNA processing pathways: origins, effects, and implications. Immunol Rev. 1998;166(1):143–151. doi: 10.1111/j.1600-065X.1998.tb01259.x. [DOI] [PubMed] [Google Scholar]
- Miller N, Wilson M, Bfufltén E, Stuge T, Warr G, Ciem W. Functional and molecular characterization of teleost leukocytes. Immunol Rev. 1998;166(1):187–197. doi: 10.1111/j.1600-065X.1998.tb01263.x. [DOI] [PubMed] [Google Scholar]
- Viret C, Gurr W. The origin of the "one cell-one antibody" rule. J Immunol. 2009;182(3):1229–1230. doi: 10.4049/jimmunol.182.3.1229. [DOI] [PubMed] [Google Scholar]
- Li J, Barreda DR, Zhang YA, Boshra H, Gelman AE, LaPatra S, Tort L, Sunyer JO. B lymphocytes from early vertebrates have potent phagocytic and microbicidal abilities. Nat Immunol. 2006;7(10):1116–1124. doi: 10.1038/ni1389. [DOI] [PubMed] [Google Scholar]
- Jenny MW, Jiri M. Mucosal immunoglobulins. Immunol Rev. 2005;206(1):64–82. doi: 10.1111/j.0105-2896.2005.00290.x. [DOI] [PubMed] [Google Scholar]
- Underdown BJ, Schiff JM. Immunoglobulin A: Strategic defense initiative at the mucosal surface. Annu Rev Immunol. 1986;4(1):389–417. doi: 10.1146/annurev.iy.04.040186.002133. [DOI] [PubMed] [Google Scholar]
- Rainer M, Louis Du P, Ellen H. Is Xenopus IgX an analog of IgA? Eur J Immunol. 1996;26(12):2823–2830. doi: 10.1002/eji.1830261205. [DOI] [PubMed] [Google Scholar]
- Solem ST, Stenvik J. Antibody repertoire development in teleosts-a review with emphasis on salmonids and Gadus morhua L. Dev Comp Immunol. 2006;30(1-2):57–76. doi: 10.1016/j.dci.2005.06.007. [DOI] [PubMed] [Google Scholar]
- Maki JL, Dickerson HW. Systemic and cutaneous mucus antibody responses of channel catfish immunized against the protozoan parasite Ichthyophthirius multifiliis. Clin Diagn Lab Immunol. 2003;10(5):876–881. doi: 10.1128/CDLI.10.5.876-881.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, Findly RC, Dickerson HW. Cutaneous antibody-secreting cells and B cells in a teleost fish. Dev Comp Immunol. 2008;32(5):500–508. doi: 10.1016/j.dci.2007.08.009. [DOI] [PubMed] [Google Scholar]
- Hatten F, Fredriksen A, Hordvik I, Endresen C. Presence of IgM in cutaneous mucus, but not in gut mucus of Atlantic salmon, Salmo salar: Serum IgM is rapidly degraded when added to gut mucus. Fish Shellfish Immunol. 2001;11(3):257–268. doi: 10.1006/fsim.2000.0313. [DOI] [PubMed] [Google Scholar]
- Yazawa R, Cooper GA, Hunt P, Beetz-Sargent M, Robb A, Conrad M, McKinnel L, So S, Jantzen S, Phillips RB. et al. Striking antigen recognition diversity in the Atlantic salmon T-cell receptor α/δ locus. Dev Comp Immunol. 2008;32(3):204–212. doi: 10.1016/j.dci.2007.05.002. [DOI] [PubMed] [Google Scholar]
- Yazawa R, Cooper GA, Beetz-Sargent M, Robb A, McKinnel L, Davidson WS, Koop BF. Functional adaptive diversity of the Atlantic salmon T-cell receptor gamma locus. Mol Immunol. 2008;45(8):2150–2157. doi: 10.1016/j.molimm.2007.12.007. [DOI] [PubMed] [Google Scholar]
- Warren RL, Nelson BH, Holt RA. Profiling model T-cell metagenomes with short reads. Bioinformatics. 2009;25(4):458–464. doi: 10.1093/bioinformatics/btp010. [DOI] [PubMed] [Google Scholar]
- Thorsen J, Zhu B, Frengen E, Osoegawa K, de Jong P, Koop BF, Davidson WS, Hoyheim B. A highly redundant BAC library of Atlantic salmon (Salmo salar): an important tool for salmon projects. BMC Genomics. 2005;6:50. doi: 10.1186/1471-2164-6-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng SHS, Artieri CG, Bosdet IE, Chiu R, Danzmann RG, Davidson WS, Ferguson MM, Fjell CD, Hoyheim B, Jones SJM. et al. A physical map of the genome of Atlantic salmon, Salmo salar. Genomics. 2005;86:396–404. doi: 10.1016/j.ygeno.2005.06.001. [DOI] [PubMed] [Google Scholar]
- Fjell CD, Bosdet I, Schein JE, Jones SJM, Marra MA. Internet Contig Explorer (iCE) - A Tool for Visualizing Clone Fingerprint Maps. Genome Res. 2003;13(6a):1244–1249. doi: 10.1101/gr.819303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ASalBase. http://www.asalbase.org
- Ewing B, Green P. Base-calling of automated sequencer traces using PHRED. II. Error probabilities. Genome Res. 1998;8:186–194. [PubMed] [Google Scholar]
- Ewing B, Hillier L, Wendl MC, Green P. Base-calling of automated sequencer traces using PHRED. I. Accuracy assessment. Genome Res. 1998;8:175–185. doi: 10.1101/gr.8.3.175. [DOI] [PubMed] [Google Scholar]
- Gordon D, Abajian C, Green P. A graphical tool for sequence finishing. Genome Res. 1998;8(3):195–202. doi: 10.1101/gr.8.3.195. [DOI] [PubMed] [Google Scholar]
- Sonnhammer ELL, Durbin R. A dot-matrix program with dynamic threshold control suited for genomic DNA and protein sequence analysis. Gene. 1995;167(1-2):GC1–GC10. doi: 10.1016/0378-1119(95)00714-8. [DOI] [PubMed] [Google Scholar]
- Higgins DG, Bleasby AJ, Fuchs R. CLUSTAL V: improved software for multiple sequence alignment. Comput Appl Biosci. 1992;8(2):189–191. doi: 10.1093/bioinformatics/8.2.189. [DOI] [PubMed] [Google Scholar]
- Kumar S, Tamura K, Nei M. MEGA3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform. 2004;5(2):150–163. doi: 10.1093/bib/5.2.150. [DOI] [PubMed] [Google Scholar]
- Lefranc MP. WHO-IUIS Nomenclature Subcommittee for immunoglobulins and T cell receptors report. Immunogenetics. 2007;59(12):899–902. doi: 10.1007/s00251-007-0260-4. [DOI] [PubMed] [Google Scholar]
- Genes identified with Digit Web Server. http://synthetic-biology.jp/sw/pic/en/crib151s2rib151s72i/
- ImageJ. http://rsbweb.nih.gov/ij/
- Ventura-Holman T, Lobb CJ. Structural organization of the immunoglobulin heavy chain locus in the channel catfish: the IgH locus represents a composite of two gene clusters. Mol Immunol. 2002;38(7):557–564. doi: 10.1016/S0161-5890(01)00075-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Features of the IGH-A. Table listing genes and pseudogenes identified in the IGH-A.
Features of the IGH-B. Table listing genes and pseudogenes identified in the IGH-B.
The identified genes flanking the loci. Table listing genes flanking the loci identified with the Digit Web Server (http://synthetic-biology.jp/sw/pic/en/crib151s2rib151s72i/).
Dotter plot of locus A (IGH-A) versus locus B (IGH-B). This file contains a dotter plot of IGH-A versus IGH-B.
Alignment of amino acid sequences encoded by (A) Cτ, (B) C μ and (C) Cδ. This file contains multiple sequence alignments of amino acid sequences encoded by (A) Cτ, (B) C μ and (C) Cδ obtained from ClustalW. Identical residues are shown as dots (.) and gaps are shown as hyphens (-).
Phylogenic relationships for the CH genes in various species. Phylogenic tree showing the relationship of the CH genes amino acid sequences of CH2 and CH3 domains of α, human δ and γ; CH3 and CH4 of μ, ζ/τ, ε, and duck α; CH4 and CH5 of new antigen receptor (NAR); CH5 and CH6 of ω, NARC and teleost δ. The tree was constructed with the MEGA 4 package by neighbor-joining (NJ) method and bootstrap values for replicated 1,000 were represented by percentages on the edge of node. The bootstrap values greater than 50% are presented. The scale bar indicates the branch length. Genbank accession numbers are as follows: α: duck [GenBank:AAA68606], human [GenBank:AAC82528]. δ: Atlantic salmon δA and δB [GenBank:AF278717; AF141605], catfish [GenBank:T18537], fugu [GenBank:BAD34542], zebrafish [GenBank:CAI11477], Xenopus [GenBank:DQ350886], human [GenBank:AAA52771]. ε: human [GenBank:AAB59395], opossum (Monodelphis domestica) [GenBank:AAC79674]. γ1: human [GenBank:AAC82527]. γ3: mouse [GenBank:AAB59697]. μ: Atlantic salmon μA and μB, [GenBank:AAB24064; AAF69490], bowfin (Amia calva) [GenBank:ACU12456], carp [GenBank:AB004105], catfish [GenBank:M27230], gar (Lepisosteus osseus) [GenBank:U12455], ladyfish (Elops saurus) [GenBank:M26182] lungfish (Protopterus aethiopicus) [GenBank:AF437724] nurse shark (Ginglymostoma cirratum) [GenBank:M92851], rainbow trout [GenBank:X83372], skate (Leucoraja erinacea) [GenBank:M29679], sturgeon (Acipenser baeri) [GenBank:Y13253], zebrafish [GenBank:AY643753], Xenopus [GenBank:M20484], chicken [GenBank:X01613], mouse [GenBank:J00443], human [GenBank:X14940]. υ: Xenopus [GenBank:X15114]. ω: lungfish [GenBank:AF437727], sandbar shark (Carcharhinus plumbeus) [GenBank:CPU40560]. NAR: nurse shark [GenBank:GCU51450]. NARC: nurse shark [GenBank:GCU18701]. ζ/τ: grass carp (Ctenopharyngodon idella) [GenBank:DQ489733], rainbow trout τ1 and τ2 [GenBank:AAW66978] and [GenBank:AAW66981], perch (Siniperca chuatsi), [GenBank:DQ016660], zebrafish [GenBank:AY643752].
Genbank accession numbers for deduced amino acid sequences of CH and VH domains. Table listing the accession numbers for deduced amino acid sequences of CH and VH domains.
Alignment of D sequences. This file contains a multiple sequence alignment of D sequences obtained from ClustalW.
Phylogenic trees showing the relationship between the (A) D and (B) JH sequences. This file contains phylogenic trees for (A) D and (B) JH genes.
Alignment of JH sequences. This file contains a multiple sequence alignment of JH sequences obtained from ClustalW.
Alignment of amino acid VH sequences. This file contains a multiple sequence alignment of amino acid VH sequences obtained from ClustalW. Identical residues are shown as dots (.) and gaps are shown as hyphens (-).
Distribution of variable (VH) families in the two IgH loci. Table showing the number of sequences identified per family.
PCR primers and oligo probes. Table listing the PCR primers and oligo probes used in this study.