Abstract
During short-term sensitization, a simple form of nonassociative learning in Aplysia, the presentation of a single brief noxious stimulus results in enhancement of the defensive withdrawal reflex lasting minutes to tens of minutes. This behavioral plasticity involves presynaptic facilitation of synaptic transmission from the mechanosensory neurons that mediate the reflex to their central target cells. This facilitation is due to cAMP-dependent protein phosphorylation. To determine whether the time course of presynaptic facilitation might be due to a persistent increase in activity of adenylate cyclase (EC 4.6.1.1) itself, persistence of the transmitter, or yet other processes, we developed a perfused-membrane method to analyze the time course of activation of adenylate cyclase by transient stimuli. After stimulation by a pulse of stimulatory transmitter, activation of adenylate cyclase decayed within 60 sec. This finding indicates that the enzyme does not remain persistently active in the absence of transmitter and suggests that short-term retention is likely to be due to other mechanisms. Possible additional mechanisms include continued activation of the cyclase by transmitter, cellular factors extrinsic to the cyclase that prolong the time course of its activation, and persistence of processes downstream from the cyclase.
Full text
PDF

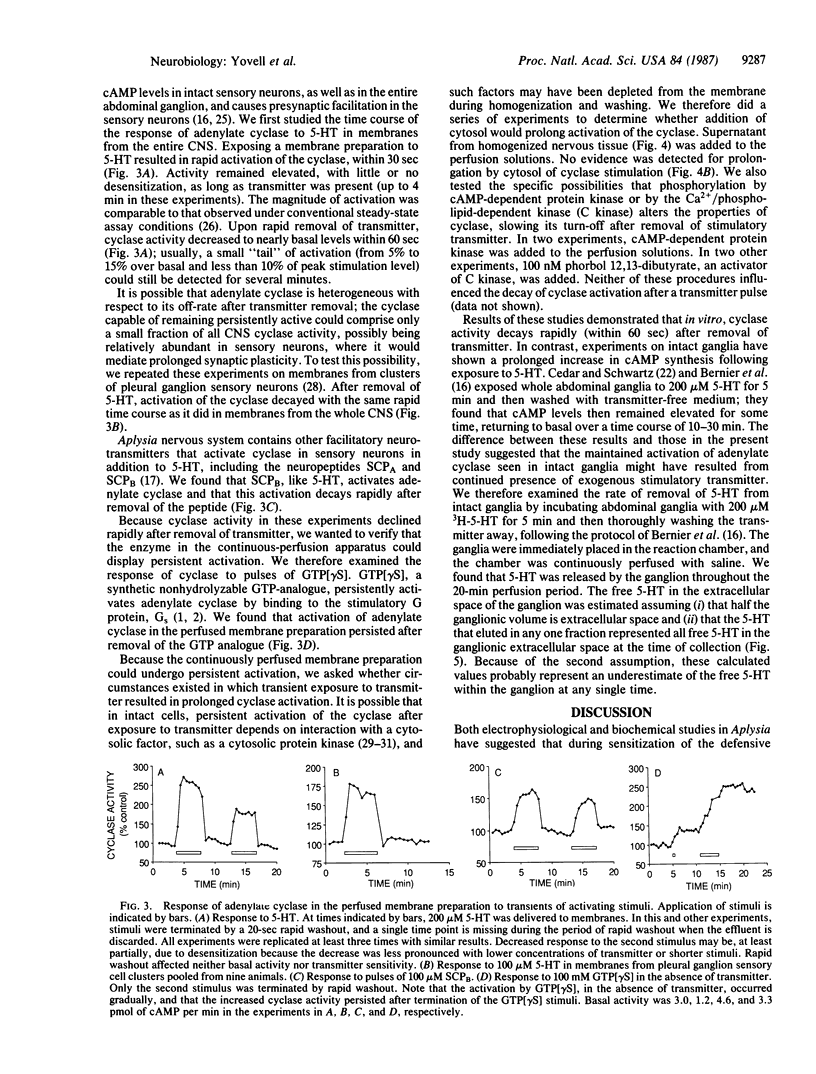
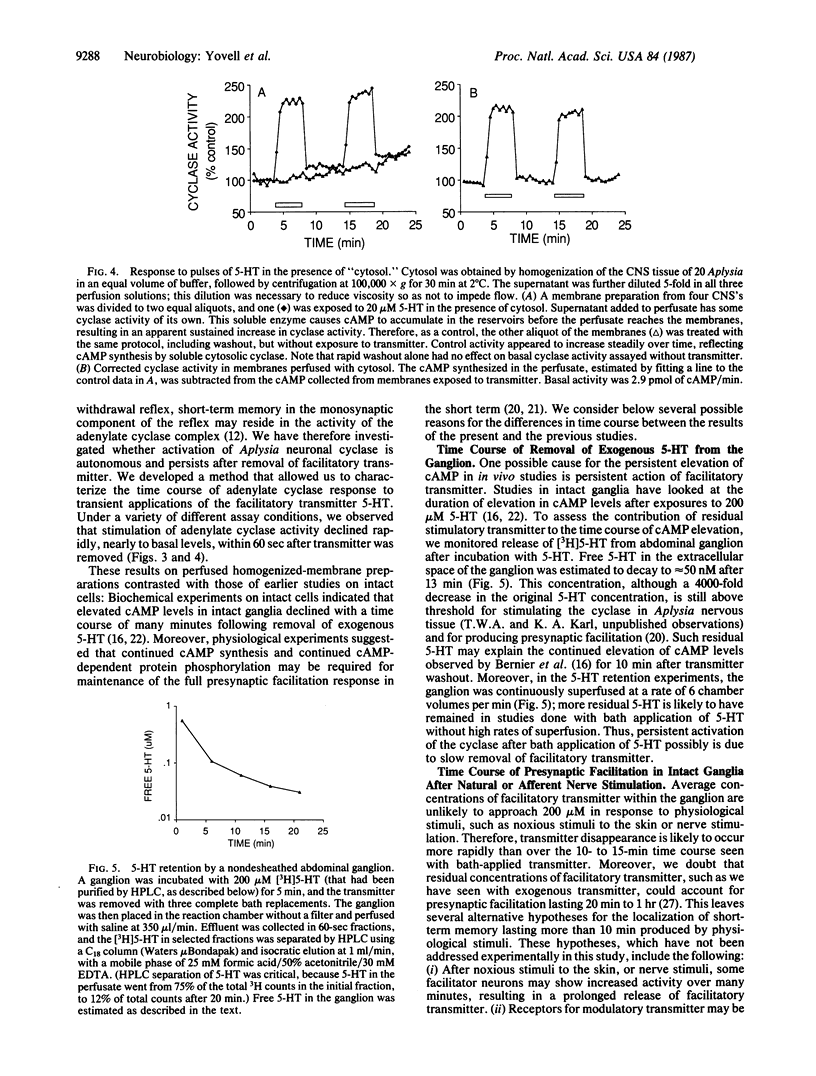

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrams T. W. Activity-dependent presynaptic facilitation: an associative mechanism in Aplysia. Cell Mol Neurobiol. 1985 Jun;5(1-2):123–145. doi: 10.1007/BF00711089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abrams T. W., Castellucci V. F., Camardo J. S., Kandel E. R., Lloyd P. E. Two endogenous neuropeptides modulate the gill and siphon withdrawal reflex in Aplysia by presynaptic facilitation involving cAMP-dependent closure of a serotonin-sensitive potassium channel. Proc Natl Acad Sci U S A. 1984 Dec;81(24):7956–7960. doi: 10.1073/pnas.81.24.7956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aceves-Piña E. O., Booker R., Duerr J. S., Livingstone M. S., Quinn W. G., Smith R. F., Sziber P. P., Tempel B. L., Tully T. P. Learning and memory in Drosophila, studied with mutants. Cold Spring Harb Symp Quant Biol. 1983;48(Pt 2):831–840. doi: 10.1101/sqb.1983.048.01.086. [DOI] [PubMed] [Google Scholar]
- Bernier L., Castellucci V. F., Kandel E. R., Schwartz J. H. Facilitatory transmitter causes a selective and prolonged increase in adenosine 3':5'-monophosphate in sensory neurons mediating the gill and siphon withdrawal reflex in Aplysia. J Neurosci. 1982 Dec;2(12):1682–1691. doi: 10.1523/JNEUROSCI.02-12-01682.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brostrom C. O., Huang Y. C., Breckenridge B. M., Wolff D. J. Identification of a calcium-binding protein as a calcium-dependent regulator of brain adenylate cyclase. Proc Natl Acad Sci U S A. 1975 Jan;72(1):64–68. doi: 10.1073/pnas.72.1.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunelli M., Castellucci V., Kandel E. R. Synaptic facilitation and behavioral sensitization in Aplysia: possible role of serotonin and cyclic AMP. Science. 1976 Dec 10;194(4270):1178–1181. doi: 10.1126/science.186870. [DOI] [PubMed] [Google Scholar]
- Castellucci V. F., Nairn A., Greengard P., Schwartz J. H., Kandel E. R. Inhibitor of adenosine 3':5'-monophosphate-dependent protein kinase blocks presynaptic facilitation in Aplysia. J Neurosci. 1982 Dec;2(12):1673–1681. doi: 10.1523/JNEUROSCI.02-12-01673.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellucci V., Kandel E. R. Presynaptic facilitation as a mechanism for behavioral sensitization in Aplysia. Science. 1976 Dec 10;194(4270):1176–1178. doi: 10.1126/science.11560. [DOI] [PubMed] [Google Scholar]
- Cech S. Y., Broaddus W. C., Maguire M. E. Adenylate cyclase: the role of magnesium and other divalent cations. Mol Cell Biochem. 1980 Dec 10;33(1-2):67–92. doi: 10.1007/BF00224572. [DOI] [PubMed] [Google Scholar]
- Cedar H., Schwartz J. H. Cyclic adenosine monophosphate in the nervous system of Aplysia californica. II. Effect of serotonin and dopamine. J Gen Physiol. 1972 Nov;60(5):570–587. doi: 10.1085/jgp.60.5.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudai Y. Some properties of adenylate cyclase which might be important for memory formation. FEBS Lett. 1985 Oct 28;191(2):165–170. doi: 10.1016/0014-5793(85)80001-6. [DOI] [PubMed] [Google Scholar]
- Dudaí Y., Uzzan A., Zvi S. Abnormal activity of adenylate cyclase in the Drosophila memory mutant rutabaga. Neurosci Lett. 1983 Dec 2;42(2):207–212. doi: 10.1016/0304-3940(83)90408-1. [DOI] [PubMed] [Google Scholar]
- Eckstein F., Cassel D., Levkovitz H., Lowe M., Selinger Z. Guanosine 5'-O-(2-thiodiphosphate). An inhibitor of adenylate cyclase stimulation by guanine nucleotides and fluoride ions. J Biol Chem. 1979 Oct 10;254(19):9829–9834. [PubMed] [Google Scholar]
- Greenberg S. M., Castellucci V. F., Bayley H., Schwartz J. H. A molecular mechanism for long-term sensitization in Aplysia. Nature. 1987 Sep 3;329(6134):62–65. doi: 10.1038/329062a0. [DOI] [PubMed] [Google Scholar]
- Jakobs K. H., Bauer S., Watanabe Y. Modulation of adenylate cyclase of human platelets by phorbol ester. Impairment of the hormone-sensitive inhibitory pathway. Eur J Biochem. 1985 Sep 2;151(2):425–430. doi: 10.1111/j.1432-1033.1985.tb09119.x. [DOI] [PubMed] [Google Scholar]
- Kandel E. R., Abrams T., Bernier L., Carew T. J., Hawkins R. D., Schwartz J. H. Classical conditioning and sensitization share aspects of the same molecular cascade in Aplysia. Cold Spring Harb Symp Quant Biol. 1983;48(Pt 2):821–830. doi: 10.1101/sqb.1983.048.01.085. [DOI] [PubMed] [Google Scholar]
- Kandel E. R., Schwartz J. H. Molecular biology of learning: modulation of transmitter release. Science. 1982 Oct 29;218(4571):433–443. doi: 10.1126/science.6289442. [DOI] [PubMed] [Google Scholar]
- Livingstone M. S. Genetic dissection of Drosophila adenylate cyclase. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5992–5996. doi: 10.1073/pnas.82.17.5992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livingstone M. S., Sziber P. P., Quinn W. G. Loss of calcium/calmodulin responsiveness in adenylate cyclase of rutabaga, a Drosophila learning mutant. Cell. 1984 May;37(1):205–215. doi: 10.1016/0092-8674(84)90316-7. [DOI] [PubMed] [Google Scholar]
- Mahaffee D. D., Ontjes D. A. The role of calcium in the control of adrenal adenylate cyclase. Enhancement of enzyme activation by guanyl-5'-yl imidodiphosphate. J Biol Chem. 1980 Feb 25;255(4):1565–1571. [PubMed] [Google Scholar]
- McPherson J. M., Whitehouse S., Walsh D. A. Possibility of shape conformers of the protein inhibitor of the cyclic adenosine monophosphate dependent protein kinase. Biochemistry. 1979 Oct 30;18(22):4835–4845. doi: 10.1021/bi00589a011. [DOI] [PubMed] [Google Scholar]
- Salomon Y. Adenylate cyclase assay. Adv Cyclic Nucleotide Res. 1979;10:35–55. [PubMed] [Google Scholar]
- Schramm M., Selinger Z. Message transmission: receptor controlled adenylate cyclase system. Science. 1984 Sep 21;225(4668):1350–1356. doi: 10.1126/science.6147897. [DOI] [PubMed] [Google Scholar]
- Schwartz J. H., Bernier L., Castellucci V. F., Palazzolo M., Saitoh T., Stapleton A., Kandel E. R. What molecular steps determine the time course of the memory for short-term sensitization in Aplysia? Cold Spring Harb Symp Quant Biol. 1983;48(Pt 2):811–819. doi: 10.1101/sqb.1983.048.01.084. [DOI] [PubMed] [Google Scholar]
- Smigel M. D., Ferguson K. M., Gilman A. G. Control of adenylate cyclase activity by G proteins. Adv Cyclic Nucleotide Protein Phosphorylation Res. 1985;19:103–111. [PubMed] [Google Scholar]
- Sugden D., Vanecek J., Klein D. C., Thomas T. P., Anderson W. B. Activation of protein kinase C potentiates isoprenaline-induced cyclic AMP accumulation in rat pinealocytes. 1985 Mar 28-Apr 3Nature. 314(6009):359–361. doi: 10.1038/314359a0. [DOI] [PubMed] [Google Scholar]
- Walters E. T., Byrne J. H., Carew T. J., Kandel E. R. Mechanoafferent neurons innervating tail of Aplysia. I. Response properties and synaptic connections. J Neurophysiol. 1983 Dec;50(6):1522–1542. doi: 10.1152/jn.1983.50.6.1522. [DOI] [PubMed] [Google Scholar]
- Yovell Y., Dudai Y. Possible involvement of adenylate cyclase in learning and short-term memory. Experimental data and some theoretical considerations. Isr J Med Sci. 1987 Jan-Feb;23(1-2):49–60. [PubMed] [Google Scholar]



