Abstract
Previous studies show that high-intensity sweeteners can stimulate weight gain in rats. The present studies examined whether caffeine, a stimulant commonly added to beverages consumed by humans, influences intake of saccharin- or glucose-sweetened solutions or body weight gain in rats and whether the nature of the maintenance diet influences the effects of caffeine. In two experiments, rats received glucose or saccharin solution mixed with 0.125 mg/g caffeine or no caffeine. Rats consumed significantly more caffeinated than noncaffeinated solutions when they were maintained on a low-fat chow diet (Experiment 1) and when maintained on a sweet, high-fat, high calorie chow diet (Experiment 2). Consumption of saccharin resulted in higher body weight gain in both experiments. Caffeine reversed this effect in Experiment 1 (low-fat diet) but not Experiment 2 (sweet, high-fat diet). The findings extend what is known about the conditions under which consumption of high intensity sweeteners promote energy dysregulation.
Keywords: Energy balance, high-intensity sweeteners, obesity
We reported previously that rats given a daily supplement to their maintenance diet that contained a non-energetic sweetener (e.g., saccharin, Acesulfame Potassium [AceK]) ate more, gained more weight, grew fatter, and were impaired their ability to compensate for calories delivered in a novel, sweet tasting food, compared to rats given the same dietary supplement sweetened with glucose (Swithers, Baker, & Davidson, 2009; Swithers & Davidson, 2008). Similarly, rats given solutions sweetened with saccharin or AceK gained significantly more weight than rats given glucose- sweetened solutions (see Swithers, Martin, & Davidson, 2010, for review). These findings question the longstanding notion that substituting high-intensity sweeteners for added sugars in food and beverages is an effective way for humans to improve weight control (Bellisle & Drewnowski, 2007).
Although high-intensity sweeteners are now being substituted for added sugars in a variety of foods, their use is especially prevalent in so-called “diet” beverages, with estimated yearly per capita consumption of diet soft drinks rising to approximately 15 gallons in 2007 (e.g. Popkin, 2010). In addition to high-intensity sweeteners, diet sodas often contain substantial amounts of caffeine. In fact, the amount of caffeine contained in diet versions is often substantially higher than in the sugar-sweetened forms of these beverages (e.g. Chou & Bell, 2007).
The effects on energy and body weight regulation of adding caffeine to beverages sweetened with high-intensity sweeteners are difficult to predict. Both humans and rats have been reported to develop preferences for flavors associated with caffeine (Chambers, Mobini, & Yeomans, 2007; Myers & Izbicki, 2006; Yeomans, et al., 2000; Yeomans, Mobini, & Chambers, 2007). Thus, the addition of caffeine could promote increased intake of diet beverages, and could thereby exacerbate the increases in energy intake and body weight gain associated with increased consumption of non-caloric sweeteners. On the other hand, there is evidence that caffeine can suppress food intake, body weight gain and adiposity in rodents (e.g. Bukowiecki, Lupien, Follea, & Jahjah, 1983; Nakabayashi, Hashimoto, Ashida, Nishiumi, & Kanazawa, 2008) and can increase insulin-independent glucose transport (Egawa, et al., 2009) and improve insulin sensitivity (Park, Jang, & Hong, 2007). In addition, caffeine has been linked to increased thermogenesis and decreased body weight in some clinical studies (Dalbo, Roberts, Stout, & Kerksick, 2010; Hursel & Westerterp-Plantenga, 2010; Westerterp-Plantenga, 2010). Thus, it is also possible that addition of caffeine to high-intensity sweetened beverages might counteract at least some of the negative effects of the non-caloric sweeteners on energy regulation.
The aim of the present experiments was to examine how the addition of caffeine to sweetened beverages influenced intake and body weight gain in rats. In the first study, four groups of rats were maintained on ad lib access to standard low-fat, laboratory chow and were given daily restricted access (3 hr per day) to a sweet-tasting solution. Solutions were sweetened with either glucose or saccharin, and either contained caffeine or did not contain caffeine. Food intake, body weight gain, and intake of the liquids were recorded for 14 days.
In the second experiment, the effects of adding caffeine to liquid diets in rats maintained on a “Westernized” chow-base diet, high in fat and sugar, on body weight gain and body composition was assessed. Thus, this experiment provided a basis for evaluating if the effects of consuming high-intensity sweeteners in conjunction with caffeine depended on whether the rats were maintained on a relatively low-fat maintenance diet or on a much higher-calorie, high-fat, maintenance diet with a macronutrient composition more similar to that consumed by humans in westernized societies. Thus, Experiment 2 assessed the effects of consuming high-intensity sweeteners on energy intake and body weight gain under dietary conditions that are similar to those prevailing in the current obesogenic human food environment.
Methods
Experiment 1
Subjects were 49 naïve, adult male Sprague-Dawley rats (Harlan, Indianapolis) weighing 358 – 398 g at the start of the experiment. Animals were individually housed in hanging wire cages with standard pelleted diet (Harlan 2018) and water available ad libitum except as described below. One week following arrival in the lab, animals were assigned to one of four groups matched on body weight (n=12–13 per group). Each group of animals received daily access to 50g of a flavored, sweetened liquid for 14 days; chow was removed from the cage during the 3 hr access period and returned immediately afterward. For two groups, the liquid was sweetened with 10% (w/w) glucose. For the remaining two groups, the liquid was sweetened with 0.3% saccharin. Caffeine (0.125 mg/g) was added to the sweetened liquid for one group in each sweetener condition. This dose of caffeine was chosen because it is similar to caffeine levels found in many sweetened beverages that are consumed by humans (USDA, 2009), and has also been used in previous studies of caffeine preference with rats (Fedorchak, Mesita, Plater, & Brougham, 2002; Myers & Izbicki, 2006). Flavors (0.1% w/w grape or cherry Kool-Aid) were added to mask flavor of the caffeine, were counterbalanced with the groups across animals and each received the same flavored solution every day. Thus, the four groups received sweetened liquids with glucose; glucose plus caffeine; saccharin; or saccharin plus caffeine. Intake of liquids and chow and body weight were measured daily by weighing.
Body weight gain and intake of the liquids were analyzed separately using 3-Way (Sweetener X Caffeine X Day) Repeated Measures ANOVAs with sweetener and caffeine as between-subjects factors and Day as a within-subjects factor. Total caloric intake (liquids plus chow) across the 14 days of the study was analyzed separately using a 2-Way (Sweetener X Caffeine) ANOVA. Post-hoc tests were done using Newman-Keuls tests. Alpha was set at 0.05 for all tests.
Experiment 2
Subjects were 33 naïve, male Sprague-Dawley rats (Harlan, Indianapolis) weighing 375 – 401 g at the start of the experiment. Food and water were available ad lib throughout the experiment. After arrival in the lab, rats were given one week acclimation on a standard lab chow diet (Harlan 2018) followed by one week acclimation to a powdered, unsweetened, high fat diet previously used in our lab (~5.48 kcal/g, with ~ 41% of calories provided by fat, 41% provided by carbohydrate, and 18% provided by protein; testdiet.com cat. # 25312, modified diet 5012 with 4% starch and 16% peanut oil). Body composition was then assessed using NMR (EchoMRI-900) and animals were assigned to one of four groups (n=8–9 per group), matched on body weight. Animals were then given ad lib access to the powdered high fat diet, to which 20% glucose (w/w) was added (~5.18 kcal/g, with ~33% of calories provided by fat, ~53% provided by carbohydrate and ~14% provided by protein) . On the same day, they began receiving access to 30 g of a sweetened liquid daily 6 days per week for 4 weeks. On the 7th day of each week, water and the high fat, sweet diet alone were provided. Two groups of animals received flavored 20% glucose solution (w/w) while the remaining two groups received flavored 0.3% saccharin solution (w/w). Caffeine (0.125 mg/g) was added to the sweetened liquid for one group of animals given each sweetener. Sweetened liquids were flavored with 0.1% grape or cherry Kool-Aid; animals always received the same flavor and half of the animals in each group received each flavor. Liquid intake and body weight were collected daily. Food intake data were not available due to excessive spillage of the powdered diet. Animals received the liquid supplements for four weeks, and body composition was again assessed using NMR.
Body weight gain was analyzed with a Three-Way (Sweetener X Caffeine X Day), repeated measures ANOVA with Sweetener and Caffeine as between-subjects factors and Day as a within-subjects factor. Liquid intake was analyzed with a Two-Way ANOVA (Sweetener X Caffeine). Body fat percent at the beginning and end of the study was analyzed with a 3-Way (Sweetener X Caffeine X Time) repeated measures ANOVA with Sweetener and Caffeine as between-subjects factors and Time as a within-subjects factor. Post-hoc tests were done using Newman-Keuls tests. A p < 0.05 was taken as significant for all tests.
Results
Experiment 1
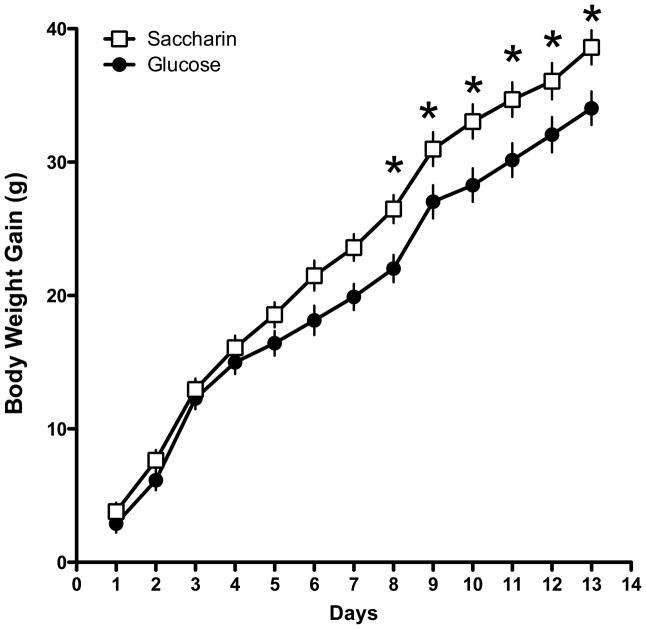
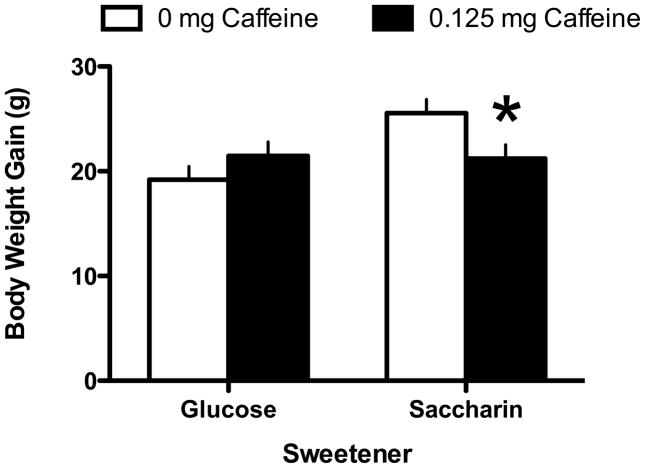
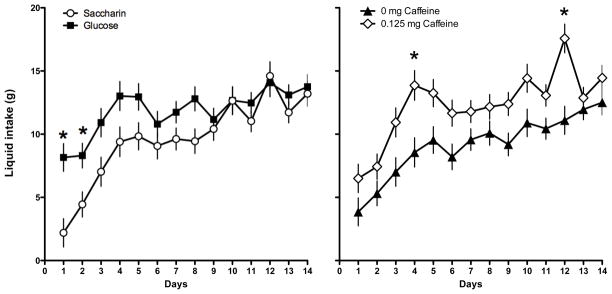
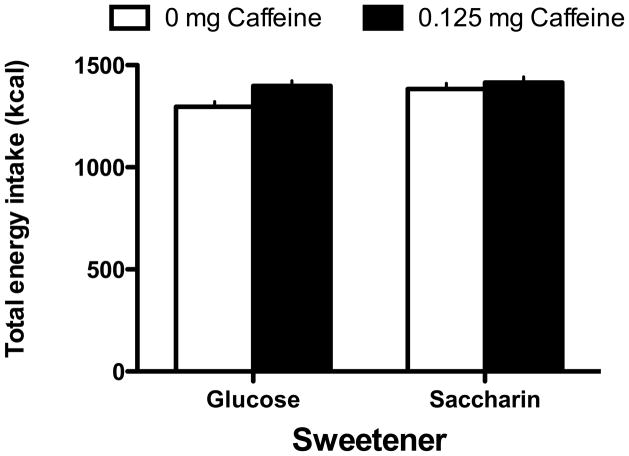
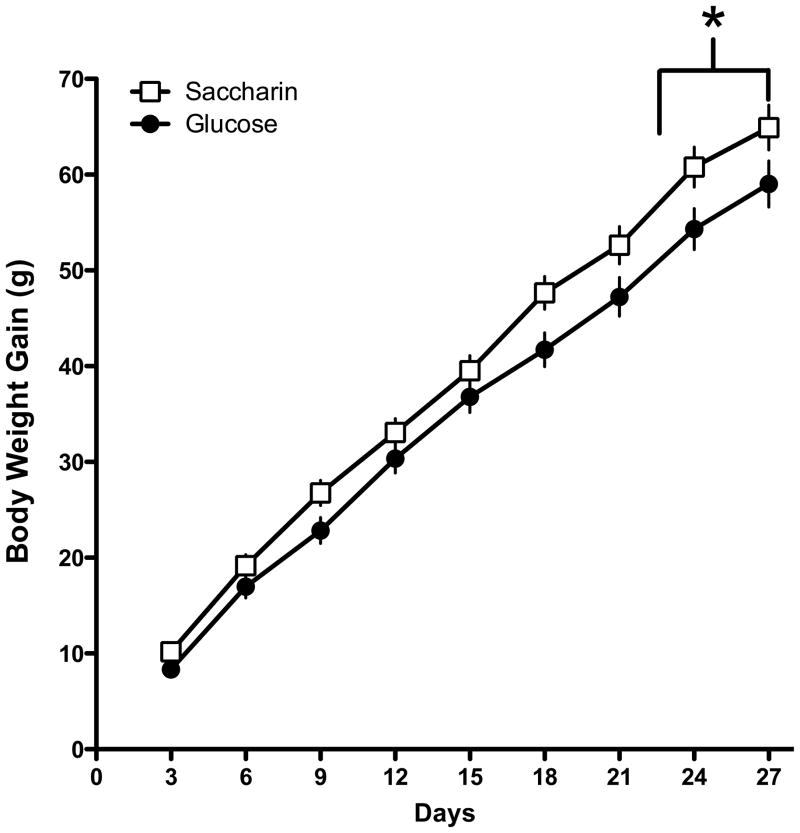
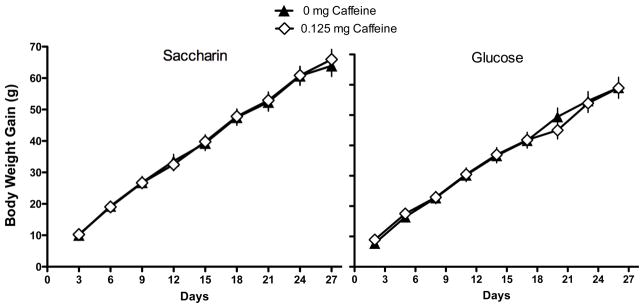
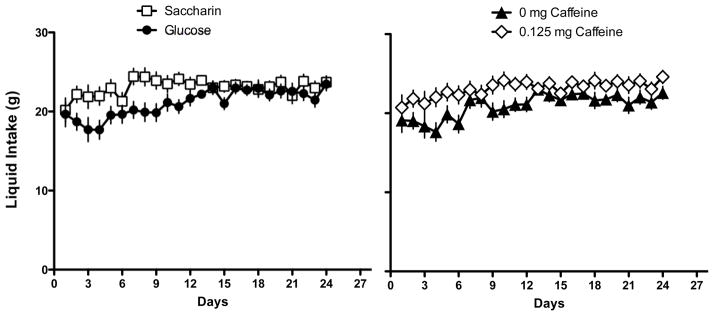
The results indicated that rats that were maintained on a standard lab-chow diet and given access to a liquid sweetened with saccharin gained significantly more weight than animals given access to a liquid sweetened with glucose (Main effect of Sweetener; F 1, 45 = 5.49, p = 0.0023; Day X Sweetener interaction; F 12, 540 = 3.704, p =0.000022; Figure 1). Post-hoc tests indicated that the difference in body weight gain across the two sweeteners was significant on Days 9–14 of access to the sweetened liquid. However, the effect of the sweetener also interacted with the presence of caffeine (Caffeine X Sweetener interaction; F 1, 45 = 6.48, p=0.014; Figure 2). Post-hoc tests revealed that in animals given the glucose-sweetened liquid, caffeine did not affect weight gain, while in animals given the saccharin-sweetened liquid, weight gain was significantly lower in animals when the liquid also contained caffeine. Analysis of intake data revealed that intake of the liquids was significantly affected by the sweetener and the addition of caffeine (Main effect of Sweetener, F 1, 45 = 4.41, p = 0.04; Main effect of Caffeine, F 1, 45 = 8.80, p =0.0048, Day X Sweet interaction, F 13, 585 = 3.29, p = 0.000073, Day X Caffeine Interaction, F 13, 585 = 2.08, p = 0.013). Post-hoc tests revealed that animals consumed more glucose-sweetened liquid than saccharin-sweetened liquid on Days 1 and 2, while animals given the caffeinated liquid consumed more than the animals given the non-caffeinated liquid on Days 4 and 12 (Figure 3). Analysis of total caloric intake indicated that animals given the caffeinated liquids consumed significantly more calories than animals given the liquids without caffeine and that animals given the saccharin-sweetened liquids consumed significantly more calories than animals given the glucose-sweetened liquids (Figure 4; Main effect of Caffeine, F 1, 45 = 6.76, p 1, 45 = 0.012; Main effect of Sweet, F 1, 45 = 4.14, p = 0.048).
Figure 1.
Body weight gain in rats maintained on a standard lab chow diet and given daily, 3-hr, access to saccharin-sweetened liquids was significantly greater than body weight gain in rats given similar access to glucose-sweetened liquids.
* p< 0.05 compared to Glucose group
Figure 2.
Body weight gain in rats maintained on a standard lab chow diet and given daily, 3-hr, access to saccharin-sweetened liquids to which caffeine (0.125 mg/g) had been added gained significantly less weight than animals given similar access to saccharin-sweetened liquids without caffeine.
* p < 0.05 compared to 0 mg Caffeine group
Figure 3.
Intake of glucose-sweetened liquids was greater on the first two days of exposure in rats maintained on a standard lab chow diet and given daily, 3-hr, access compared to intake of saccharin-sweetened liquids (left panel). Intake of sweetened liquids containing caffeine was greater than intake of sweetened liquids without caffeine (right panel).
* p < 0.05 compared to saccharin (left panel) or 0 mg caffeine (right panel)
Figure 4.
Total energy intake in rats maintained on a standard lab chow diet and given daily, 3-hr, access to saccharin-sweetened liquids was significantly greater compared to rats given similar access to glucose-sweetened liquids. In addition, total energy intake was significantly higher in rats consuming sweetened liquids with caffeine compared to liquids with no caffeine.
Experiment 2
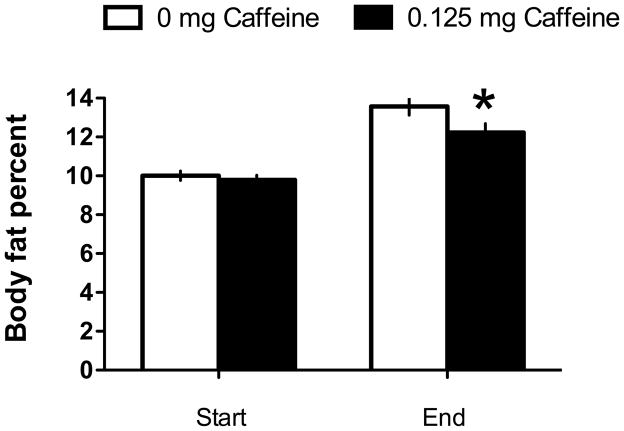
As seen in chow-fed animals in Experiment 1, body weight gain in animals maintained on a sweet, high-fat, high-energy chow diet was significantly higher in animals given access to the saccharin-sweetened liquid compared to the glucose-sweetened liquid (Figure 5; Main effect of Sweetener, F 1, 29 =5.96, p = 0.021; Sweetener X Day interaction; F 26, 754 = 2.29, p = 0.00027). However, unlike the effects observed in Experiment 1, there were no significant effects or interactions of caffeine on body weight gain in animals given saccharin-sweetened liquids, nor were there any effects of caffeine on body weight gain in animals given glucose-sweetened liquids (Figure 6; F’s < 1 for main effects and interactions with caffeine). Intake of the liquids was significantly affected by sweetener and by caffeine (Figure 7; Main effect of Sweetener, p 1, 29 = 4.23, p = 0.048; Main effect of Caffeine, p 1, 29 = 4.97, p = 0.034). Post-hoc analyses indicated that animals consumed more caffeinated than non-caffeinated liquids and that consumption of saccharin-sweetened liquids was greater than consumption of glucose-sweetened liquids. Analysis of changes in body composition revealed that there were no differences in body fat across the groups at the start of the experiment, however, animals given caffeine during the experiment gained significantly less fat than animals that were not given caffeine (Figure 8; Main effect of Time; F 1, 29 = 164.34, p < 0.0000001; Caffeine X Time interaction, F 1, 29 = 5.73, p = 0.023).
Figure 5.
Body weight gain in rats maintained on a high-fat, sweetened chow diet was significantly greater in animals given 24 hr access to saccharin-sweetened liquids compared to those given glucose-sweetened liquids. The data are presented at 3 day intervals, although body weight was measured daily.
* differences between the saccharin and glucose groups were significant on each of the last 5 days of testing (p < 0.05).
Figure 6.
There were no effects of caffeine (0.125 mg/g) on body weight gain in rats maintained on a high-fat, sweetened chow diet and given 24 hr access to saccharin-sweetened liquids (left) or glucose-sweetened liquids (right).
Figure 7.
Intake of saccharin-sweetened liquids was significantly higher than glucose-sweetened liquids in rats maintained on a high-fat, sweetened chow diet and given 24 hr access (left panel). In addition, intake of caffeinated liquids was significantly higher than intake of non-caffeinated liquids (right panel).
Figure 8.
Body fat percent was significantly greater in rats maintained on a high-fat, sweetened chow diet and given 24 hr access to sweetened liquids that did not contain caffeine compared to rats with similar access to sweetened liquids with caffeine.
* p < 0.05 compared to 0 mg caffeine
Discussion
The results of Experiment 1 confirmed our previous findings (see Swithers, et al., 2010) by showing that daily consumption of a solution sweetened with saccharin promoted greater total caloric intake and increased body weight gain relative to consuming a solution sweetened with glucose. The findings of this study also showed that adding caffeine to either the saccharin- or glucose sweetened solutions stimulated intake of these fluids by rats compared to when these solutions were given without caffeine. However, in Experiment 1 the addition of caffeine did not augment the effects of saccharin on weight gain. Rats that consumed saccharin-sweetened solutions gained no more weight than rats given glucose solutions when each solution contained caffeine. Elevated body weight gain was not observed in the saccharin-caffeine group despite significant increases in overall caloric intake in animals given caffeinated beverages.
The rats in Experiment 1 were maintained on a standard lab chow, which contained very low levels of fat, did not have sugar added, and was relatively low in energy content. Experiment 2 also evaluated the effects on body weight gain of consuming saccharin-sweetened or glucose-sweetened solutions with or without added caffeine. However, in Experiment 2 the maintenance diet was energy rich compared to the diet in Experiment 1, containing higher amounts of fat with sugar added. The results of Experiment 2 showed that for rats maintained on this westernized diet, consuming the saccharin solution was accompanied by greater weight gain compared to consuming glucose solution, regardless of whether these sweetened solutions also contained caffeine, even when the concentration of the glucose solution was increased to 20%.
What types of mechanisms could operate to produce this seemingly complex pattern of results, where the effects of saccharin and caffeine on body weight gain varied as function of the sweetness, fat content, and/ or caloric density of the maintenance diet? Previously, we proposed that efficient energy regulation relies on the ability of animals, including humans to use sweet taste and other orosensory cues to predict the impending postingestive consequences of eating. Consuming high-intensity sweeteners degrades this predictive relationship by separating sweet taste from energetic postingestive outcomes, thereby making it more difficult to reduce intake in anticipation of those consequences when sweet-tasting and high calorie foods and beverages are consumed (Davidson & Swithers, 2004).
The results of Experiment 1 showed that animals given the saccharin-sweetened liquids consumed more calories overall than animals given glucose-sweetened liquids, regardless of whether the liquids contained caffeine. This pattern suggests that caffeine did not prevent the detrimental effects of weakening the normal sweet taste - calorie relationship; consuming saccharin-sweetened liquids stimulated overall food intake. However, in Experiment 1, the presence of caffeine did prevent the augmented body weight gain typically observed when this relationship is weakened; rats given saccharin plus caffeine did not gain more weight than rats given glucose plus caffeine. One way this outcome could have been obtained is if caffeine elevated energy expenditure enough to counteract the excess caloric intake produced by consuming the saccharin solution. For example, caffeine has been demonstrated to produce thermogenic effects in rats (Bukowiecki, et al., 1983) and these effects could be sufficient to offset the increased energy intake observed Experiment 1. Caffeine could also have stimulated behavioral activity (Haldi, Wynn, & Ensor, 1947), which could also contribute to increased energy expenditure and decreased overall body weight gain.
Within this analysis, the results of Experiment 2 suggest that the amount of caffeine used in these studies was not sufficient to offset weight gain produced by saccharin when the maintenance diet was energy dense and sweetened with added sugar, despite the increased intake of the caffeinated saccharin-sweetened liquid in Experiment 2. It is plausible that the effects of consuming saccharin on body weight gain could be an increasing function of the sweetness of the maintenance diet (e.g. Davidson, Martin, and Swithers, 2010, under review). If saccharin consumption promotes excess intake by degrading the ability of rats to use sweet taste to signal energetic outcomes, then failure to accurately predict the caloric consequences of eating a sweetened high-energy maintenance diet might be greater than failing to anticipate the consequences of eating less sweet and less energy dense food.
Differences in the outcomes of the two experiments could also reflect differences in the duration of access to the liquids (3 hr in Experiment 1 versus 24 hr in Experiment 2). Because animals did consume more of the sweetened liquids in Experiment 2 relative to Experiment 1, addition of caffeine could have been ineffective at moderating the effects of this increased exposure. However, consumption was increased in both the caffeinated and uncaffeinated liquid groups in Experiment 2 compared to Experiment 1; as a result, animals in Experiment 2 got not only increased exposure to the sweeteners, but increased exposure to the caffeine. While this increased exposure is difficult to reconcile with the reduced impact on body weight gain, the potential impact of differences in consumption of the liquids across the experiments cannot be ruled out.
Despite the failure of the caffeine to reduce body weight gain in animals on the high fat-sweet maintenance diet, caffeine did suppress increases in adiposity in both the group that consumed glucose-sweetened liquids and the group that consumed saccharin-sweetened liquids. These results are consistent with previous reports of beneficial effects of caffeine on adiposity in animals maintained on high fat diets (e.g. (Inoue, et al., 2006; Kobayashi-Hattori, Mogi, Matsumoto, & Takita, 2005), and suggest that even when the diets are high in energy, caffeine can reduce potentially negative consequences of consuming sweetened beverages on body composition.
Although additional research is needed to pinpoint the underlying mechanisms, the results of these studies indicate that consuming liquids sweetened with the high-intensity sweetener saccharin results in increased food intake and body weight gain in rats relative to animals consuming liquids sweetened with glucose. These effects occur when the liquids are available for a limited time, or when ad lib access is provided, and emerge in animals maintained on a standard low-fat, laboratory chow as well as those consuming a high-energy, high-fat, sweetened chow diet. Addition of caffeine appears to mitigate the negative consequences of consuming the high-intensity sweetener, but only if the diet consumed along with the liquid is relatively low in fat and calories.
Our findings with rats may have important implications for understanding intake and body weight regulation in humans. The diets consumed by humans in westernized societies is often characterized as “energy-rich” because they contain high amounts of fat and processed sugar. The wide availability of these diets has been linked to the emergence of the current global obesity epidemic and to a variety of other disorders (e.g., Type II diabetes, cardiovascular disease, hypertension, cognitive functions) that are related to excess caloric intake (Fung, et al., 2009; Fung, et al., 2001; van Dam, Rimm, Willett, Stampfer, & Hu, 2002).
Replacing added sugars with noncaloric sweeteners has been proposed as one way to help address these types of problems (“Position of the American Dietetic Association: use of nutritive and nonnutritive sweeteners,” 2004). The results of the present research question the effectiveness of this strategy. The present findings confirmed that rats that consumed non-caloric sweetened solutions along with their maintenance diet gained more weight that rats that consumed calorically-sweetened solutions. The addition of caffeine to the solutions abolished this effect only when the maintenance diet was already low-calorie. It should be noted that even with caffeine and a relatively low calorie diet, substituting a saccharin-sweetened solution for a solution sweetened with glucose did not produce weight loss. Of greater potential concern, increased weight gain produced by consuming saccharin compared to glucose solutions was unabated by caffeine when the composition of the rat’s maintenance diet was sweet and energy dense—similar to the diets consumed by millions of people who are currently battling obesity and its related disorders. One implication of the present findings is that for these people, consuming non-calorically sweetened beverages, with or without caffeine, may not be an effective aid to weight control.
Acknowledgments
We thank Melissa McCurley, Isaac Ratcliffe, Stephanie and Mary Reher for assistance with performing the studies. Supported by NIH grants R01DK076078 and P01HD052112 to S.E.S and T.L.D. The funding agencies had no involvement in the study design, collection, analysis or interpretation of the results, writing of the report or decision to submit the paper for publication.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References Cited
- Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691–700. doi: 10.1038/sj.ejcn.1602649. [DOI] [PubMed] [Google Scholar]
- Bukowiecki LJ, Lupien J, Follea N, Jahjah L. Effects of sucrose, caffeine, and cola beverages on obesity, cold resistance, and adipose tissue cellularity. Am J Physiol. 1983;244:R500–507. doi: 10.1152/ajpregu.1983.244.4.R500. [DOI] [PubMed] [Google Scholar]
- Chambers L, Mobini S, Yeomans MR. Caffeine deprivation state modulates expression of acquired liking for caffeine-paired flavours. Q J Exp Psychol (Colchester) 2007;60:1356–1366. doi: 10.1080/17470210601154545. [DOI] [PubMed] [Google Scholar]
- Chou KH, Bell LN. Caffeine content of prepackaged national-brand and private-label carbonated beverages. J Food Sci. 2007;72:C337–342. doi: 10.1111/j.1750-3841.2007.00414.x. [DOI] [PubMed] [Google Scholar]
- Dalbo VJ, Roberts MD, Stout JR, Kerksick CM. Effect of gender on the metabolic impact of a commercially available thermogenic drink. J Strength Cond Res. 2010;24:1633–1642. doi: 10.1519/JSC.0b013e3181db9bbd. [DOI] [PubMed] [Google Scholar]
- Davidson TL, Swithers SE. A Pavlovian approach to the problem of obesity. Int J Obes Relat Metab Disord. 2004;28:933–935. doi: 10.1038/sj.ijo.0802660. [DOI] [PubMed] [Google Scholar]
- Egawa T, Hamada T, Kameda N, Karaike K, Ma X, Masuda S, et al. Caffeine acutely activates 5'adenosine monophosphate-activated protein kinase and increases insulin-independent glucose transport in rat skeletal muscles. Metabolism. 2000;58:1609–1617. doi: 10.1016/j.metabol.2009.05.013. [DOI] [PubMed] [Google Scholar]
- Fedorchak PM, Mesita J, Plater SA, Brougham K. Caffeine-reinforced conditioned flavor preferences in rats. Behav Neurosci. 2002;116:334–346. [PubMed] [Google Scholar]
- Fung TT, Malik V, Rexrode KM, Manson JE, Willett WC, Hu FB. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037–1042. doi: 10.3945/ajcn.2008.27140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung TT, Rimm EB, Spiegelman D, Rifai N, Tofler GH, Willett WC, et al. Association between dietary patterns and plasma biomarkers of obesity and cardiovascular disease risk. Am J Clin Nutr. 2001;73:61–67. doi: 10.1093/ajcn/73.1.61. [DOI] [PubMed] [Google Scholar]
- Haldi J, Wynn W, Ensor C. Modification of the action of caffeine on the spontaneous activity of the white rat by the concomitant administration of various food materials. J Nutr. 1947;34:389–399. doi: 10.1093/jn/34.4.389. [DOI] [PubMed] [Google Scholar]
- Hursel R, Westerterp-Plantenga MS. Thermogenic ingredients and body weight regulation. Int J Obes (Lond) 2010;34:659–669. doi: 10.1038/ijo.2009.299. [DOI] [PubMed] [Google Scholar]
- Inoue H, Kobayashi-Hattori K, Horiuchi Y, Oishi Y, Arai S, Takita T. Regulation of the body fat percentage in developmental-stage rats by methylxanthine derivatives in a high-fat diet. Biosci Biotechnol Biochem. 2006;70:1134–1139. doi: 10.1271/bbb.70.1134. [DOI] [PubMed] [Google Scholar]
- Kobayashi-Hattori K, Mogi A, Matsumoto Y, Takita T. Effect of caffeine on the body fat and lipid metabolism of rats fed on a high-fat diet. Biosci Biotechnol Biochem. 2005;69:2219–2223. doi: 10.1271/bbb.69.2219. [DOI] [PubMed] [Google Scholar]
- Myers KP, Izbicki EV. Reinforcing and aversive effects of caffeine measured by flavor preference conditioning in caffeine-naive and caffeine-acclimated rats. Physiol Behav. 2006;88:585–596. doi: 10.1016/j.physbeh.2006.05.010. [DOI] [PubMed] [Google Scholar]
- Nakabayashi H, Hashimoto T, Ashida H, Nishiumi S, Kanazawa K. Inhibitory effects of caffeine and its metabolites on intracellular lipid accumulation in murine 3T3-L1 adipocytes. Biofactors. 2008;34:293–302. doi: 10.3233/BIO-2009-1083. [DOI] [PubMed] [Google Scholar]
- Park S, Jang JS, Hong SM. Long-term consumption of caffeine improves glucose homeostasis by enhancing insulinotropic action through islet insulin/insulin-like growth factor 1 signaling in diabetic rats. Metabolism. 2007;56(5):599–607. doi: 10.1016/j.metabol.2006.12.004. [DOI] [PubMed] [Google Scholar]
- Popkin BM. Patterns of beverage use across the lifecycle. Physiol Behav. 2010;100:4–9. doi: 10.1016/j.physbeh.2009.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Position of the American Dietetic Association: use of nutritive and nonnutritive sweeteners. J Am Diet Assoc. 2004;104:255–275. doi: 10.1016/j.jada.2003.12.001. [DOI] [PubMed] [Google Scholar]
- Swithers SE, Baker CR, Davidson TL. General and persistent effects of high-intensity sweeteners on body weight gain and caloric compensation in rats. Behav Neurosci. 2009;123:772–780. doi: 10.1037/a0016139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swithers SE, Davidson TL. A role for sweet taste: calorie predictive relations in energy regulation by rats. Behav Neurosci. 2008;122:161–173. doi: 10.1037/0735-7044.122.1.161. [DOI] [PubMed] [Google Scholar]
- Swithers SE, Martin AA, Davidson TL. High-intensity sweeteners and energy balance. Physiol Behav. 2010;100:55–62. doi: 10.1016/j.physbeh.2009.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- USDA Agricultural Research Service. [Accessed 7/23/2010];USDA National nutrient database for standard reference, Release 22. 2009 http://www.ars.usda.gov/ba/bhnrc/ndl.
- van Dam RM, Rimm EB, Willett WC, Stampfer MJ, Hu FB. Dietary patterns and risk for type 2 diabetes mellitus in U.S. men. Ann Intern Med. 2002;136:201–209. doi: 10.7326/0003-4819-136-3-200202050-00008. [DOI] [PubMed] [Google Scholar]
- Westerterp-Plantenga MS. Green tea catechins, caffeine and body-weight regulation. Physiol Behav. 2010;100:42–46. doi: 10.1016/j.physbeh.2010.02.005. [DOI] [PubMed] [Google Scholar]
- Yeomans MR, Jackson A, Lee MD, Steer B, Tinley E, Durlach P, et al. Acquisition and extinction of flavour preferences conditioned by caffeine in humans. Appetite. 2000;35:131–141. doi: 10.1006/appe.2000.0339. [DOI] [PubMed] [Google Scholar]
- Yeomans MR, Mobini S, Chambers L. Additive effects of flavour-caffeine and flavour-flavour pairings on liking for the smell and flavour of a novel drink. Physiol Behav. 2007;92:831–839. doi: 10.1016/j.physbeh.2007.06.006. [DOI] [PubMed] [Google Scholar]