Abstract
The efficacy of recombinant enzyme therapy for genetic diseases is limited in some patients by the generation of a humoral immune response to the therapeutic protein. Inducing immune tolerance to the protein prior to treatment has the potential to increase therapeutic efficacy. Using an AAV8 vector encoding human acid α-glucosidase (hGAA), we have evaluated direct intrathymic injection for inducing tolerance. We have also compared the final tolerogenic states achieved by intrathymic and intravenous injection. Intrathymic vector delivery induced tolerance equivalent to that generated by intravenous delivery, but at a 25-fold lower dose, the thymic hGAA expression level was 10,000-fold lower than the liver expression necessary for systemic tolerance induction. Splenic regulatory T cells (Tregs) were apparent after delivery by both routes, but with different phenotypes. Intrathymic delivery resulted in Tregs with higher FoxP3, TGFβ, and IL-10 mRNA levels. These differences may account for the differences noted in splenic T cells, where only intravenous delivery appeared to inhibit their activation. Our results imply that different mechanisms may be operating to generate immune tolerance by intrathymic and intravenous delivery of an AAV vector, and suggest that the intrathymic route may hold promise for decreasing the humoral immune response to therapeutic proteins in genetic disease indications.
Introduction
The generation of neutralizing antibodies against a potentially therapeutic protein in patients with inherited enzyme deficiency diseases could decrease treatment efficacy. For example, Pompe disease is an inherited metabolic disorder caused by deficiencies of the lysosomal enzyme acid α-glucosidase (GAA). Although enzyme replacement therapy with recombinant human GAA (rhGAA) is being used to treat patients with Pompe disease, it has been recognized that antibodies against rhGAA in some patients may decrease therapeutic efficacy.1,2
We have shown previously that immune tolerance to a subsequent challenge with rhGAA can be induced in Pompe mice by intravenous gene delivery of an AAV8 vector containing a largely liver-specific albumin promoter and bearing the cDNA for either enzymatically active hGAA3 or an inactive mutant hGAA.4 After intravenous delivery of an AAV8 vector, the expression of hGAA derives almost exclusively from hepatocytes. For hGAA as well as other transgenes, the degree of tolerance appears to be directly related to the serum expression level attained, which, in turn, may depend on the mouse strain.5,6 Liver-based expression sufficient to induce tolerance may be difficult to attain in patients with pre-existing antibodies against the vector. We have therefore evaluated intrathymic gene delivery as an alternative approach for inducing immune tolerance. This approach is essentially an extension of Waksman's pioneering studies on thymic tolerance induction.7,8 Low expression levels of promiscuous genes, tissue restricted or peripheral antigens in the thymus play a critical role in thymus-generated self-tolerance, i.e., thymic tolerance.7,9,10,11 During thymic development, T cells undergo both positive (T cells need to bind to self-major histocompatibility complex molecules) and negative selection (T cells with affinity too high for self-antigens are deleted). T cells with intermediate affinity for self-antigens that escape negative selection are induced to become regulatory T cells (Tregs)—a process essential for the maintenance of peripheral tolerance. The generation of thymic tolerance has been evaluated previously by the intrathymic delivery of cells, proteins, or genes.12,13,14,15,16 We hypothesized that expressing hGAA in the thymus at a low level by intrathymic delivery of AAV8-hGAA would induce immune tolerance to the protein through both negative selection and induction of Tregs. We also reasoned that the relatively low threshold level of hGAA expression required to generate thymic tolerance might be achievable by local thymic delivery even in the presence of pre-existing antibodies against the vector.
The primary goal of these studies was to determine whether immune tolerance could be induced against a foreign protein by direct injection of an AAV8 vector bearing the appropriate cDNA, and to evaluate any effects of pre-existing anti-AAV8 antibodies on this process. Our secondary goal was to characterize the status of any Tregs generated by intrathymic gene delivery, and to compare directly these results with those obtained after intravenous delivery of the same vector. The results of this study add to our current understanding of the tolerogenic states resulting from intrathymic and intravenous delivery, and may help develop an appropriate approach to induce tolerance that may overcome immune responses to enzyme replacement therapy in the clinic.
Results
Intrathymic injection of a low dose of AAV8-hGAA results in immune tolerance equivalent to that induced by a high intravenous dose
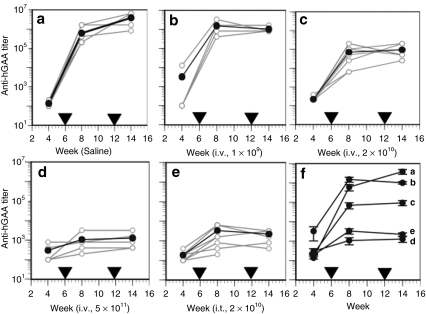
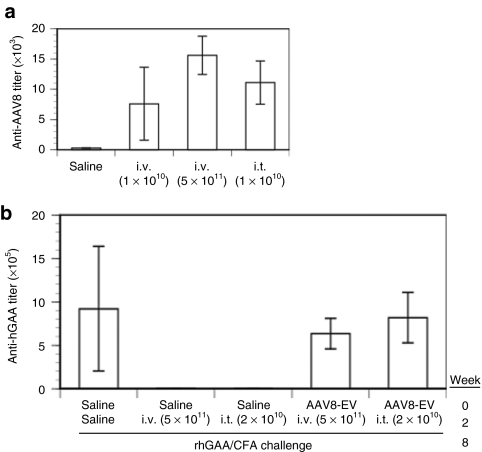
We have shown previously3 that a relatively high dose (5 × 1011 DNase-resistant particles (drp)/mouse) of AAV8-hGAA is required to induce tolerance within a few weeks using an intravenous delivery route, viz, lower doses such as 2 × 1010 or 1 × 109 drp either require much longer expression or do not lead to immune tolerance to hGAA. To ask whether tolerance could be induced by direct intrathymic delivery of the same vector but at a lower dose, 2 × 1010 drp of AAV8-hGAA was injected intrathymically followed by challenges with rhGAA in complete Freund's adjuvant (CFA) and incomplete Freund's adjuvant (IFA) at 6 and 12 weeks after vector administration, respectively. For comparison, increasing doses (1 × 109, 2 × 1010, and 5 × 1011 drp) of the same vector were administered by intravenous injection. Consistent with our previous results,3 Figure 1 shows that 2 weeks after the rhGAA/CFA challenge, anti-hGAA titers were significantly increased in the saline (Figure 1a) and all intravenous-treated groups (Figure 1b,c) except the highest dose (5 × 1011 drp) group (Figure 1d). These results were confirmed by the anti-hGAA titers observed 2 weeks after the subsequent rhGAA/IFA challenge. In contrast to these results using intravenous delivery, Figure 1e shows that low-dose (2 × 1010 drp) intrathymic delivery resulted in an antibody profile similar to that achieved with the much higher (5 × 1011 drp) intravenous dose of the same vector (Figure 1d). This equivalent degree of tolerance, and the progressive lack of tolerance at the lower intravenous doses, is more clearly depicted in the composite Figure 1f.
Figure 1.
Anti-hGAA antibody profiles before and after rhGAA protein challenges in mice receiving AAV8-hGAA by intravenous or intrathymic delivery. Mice were treated (week zero) with (a) saline by intravenous (N = 5) or intrathymic (N = 3) injection, or the AAV8-hGAA vector by intravenous (i.v.) injection at (b) 1 × 109 (N = 4), (c) 2 × 1010 (N = 7), or (d) 5 × 1011 DNase-resistant particles (drp) (N = 5), or (e) intrathymic (i.t.) injection at 2 × 1010 drp (N = 9). Mice were challenged with rhGAA in complete Freund's adjuvant at week 6 after AAV administration and with rhGAA in incomplete Freund's adjuvant at week 12, as indicated by the arrowheads. Anti-hGAA titers were analyzed at weeks 4, 8 (2 weeks after the 1st challenge), and 14 (2 weeks after the 2nd challenge). Open circles connected by gray lines represent anti-hGAA titers for individual mice. Filled circles connected by dark lines represent average values for each group, which are shown compiled in f with SEM.
Intrathymic and intravenous delivery generate tolerogenic states with different tissue hGAA mRNA profiles
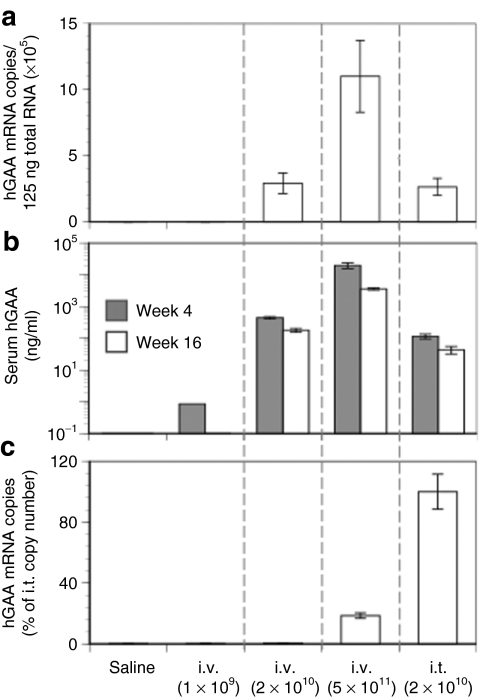
We have shown previously that hGAA expression resulting from intravenous delivery of AAV8-hGAA must attain a threshold level to achieve tolerance.3 (It is important to note that transgene expression after intravenous delivery of an AAV vector containing this albumin-driven cassette is almost exclusively derived from hepatocytes3,17,18). This threshold level of expression is consistently achieved at a vector dose of 5 × 1011 drp, but not at 1 × 1011 or 1 × 1010. Figure 2a,b shows that increasing intravenous doses of vector lead to dose-dependent increases in the following: (i) vector-specific hGAA mRNA copy number in the liver and (ii) serum hGAA levels. Figure 2c shows that as a consequence of intravenous delivery, hGAA mRNA copies are detectable in the thymus only at the highest dose (5 × 1011). These results are consistent with our previous report3 that the serum hGAA expression threshold to achieve tolerance by intravenous injection is ~1,000 ng/ml. Here, we also show that an AAV vector containing a largely hepatocyte-restricted promoter, namely, a human serum albumin promoter, can express an exogenous gene in the thymus, albeit at very low levels. To our knowledge, this is the first report documenting AAV transduction of the thymus with a tissue-specific promoter in mice, and is consistent with a previous report in transgenic mice that the albumin promoter can express an exogenous gene in liver and at a much lower level in thymus.19
Figure 2.
hGAA levels in liver, serum, and thymus after intravenous or intrathymic injection of AAV8-hGAA. Mice received different doses of the AAV8-hGAA vector by intravenous or intrathymic injection as in Figure 1. (a) Liver expression of the transgene was quantified at week 16 after AAV administration by hGAA mRNA copies. (b) Serum hGAA levels were determined by enzyme-linked immunosorbent assay at weeks 4 and 16, and (c) thymic expression was quantified at week 16 by hGAA mRNA copies. Data are expressed as means ± SEM (N = 8–10 mice/group).
Figure 2 also shows that intrathymic delivery of 2 × 1010 drp AAV8-hGAA leads to liver (Figure 2a; mRNA) and serum (Figure 2b; hGAA) expression equivalent to that observed after intravenous delivery of the same vector dose. In contrast to intravenous delivery, however, direct intrathymic injection of the vector leads to more copies of hGAA mRNA in the thymus. Similar liver mRNA and serum expression levels following both intravenous and intrathymic injection of 2 × 1010 drp suggest that the vast majority of the vector injected into the thymus escaped from that organ and traveled to the liver. Importantly, however, at this dose, only the direct intrathymic delivery resulted in tolerance induction (Figure 1). Intrathymic injection thus led to tolerance at liver and serum expression levels that would not have led to tolerance had they been achieved by intravenous injection. Thus, although most of the intrathymic injection wound up in the liver, some of that vector transduced the thymus (Figure 2c) and resulted in tolerance to hGAA. Intravenous vector delivery also resulted in some expression in the thymus (Figure 2c), but as we show below, these two delivery routes lead to very different responses by immune cells, and imply different mechanisms for tolerance induction by intravenous and intrathymic delivery.
Intrathymic vector injection results in a Treg mRNA profile in thymus, but intravenous injection does not
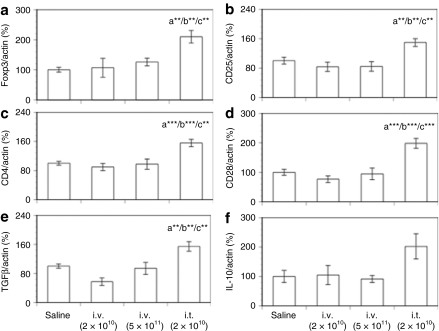
Both intrathymic and intravenous delivery of AAV8-hGAA led to detectable hGAA mRNA levels in the thymus (Figure 2c). To ask whether either delivery route led to the expression of genes in the thymus characteristic of Tregs, thymus mRNA was analyzed for the Treg markers Foxp3, CD25, CD4, and CD28 16 weeks after AAV8 hGAA administration. CD28 is required for the development of CD4+CD25+Foxp3+ cells (Tregs) in the thymus.20,21 Figure 3a–d shows that 16 weeks after vector delivery and after the subsequent hGAA protein challenges, neither a nontolerizing (2 × 1010) nor a tolerizing (5 × 1011) intravenous dose of AAV8-hGAA led to an mRNA profile any different from that obtained in animals initially dosed with saline. In contrast to these results, intrathymic delivery led to significant increases in the mRNA levels of each of these markers in the thymus, consistent with the generation of Tregs by this delivery route.
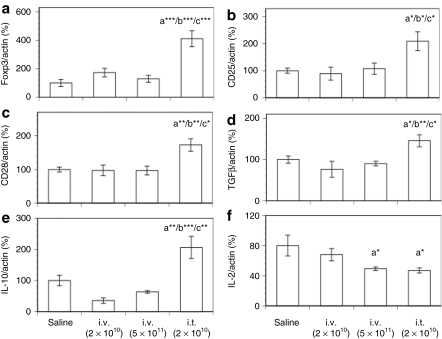
Figure 3.
Thymic mRNA levels of Foxp3, CD markers, and cytokines after intravenous or intrathymic injection of AAV8-hGAA. Thymic mRNA expression levels of (a) Foxp3, (b) CD25, (c) CD4, (d) CD28, (e) TGFβ, and (f) IL-10 were analyzed by TaqMan with preamplification (see Materials and Methods) at week 16 after AAV8-hGAA administration. Results shown are normalized to actin expression and expressed as a percentage of the saline control, and are presented as means ± SEM (N = 8–10 mice/group). Above a bar, the letter “a” represents a significant difference compared to the saline group, “b” compared to the i.v. (2 × 1010) group, and “c” compared to the i.v. (5 × 1011) group. The number of asterisks following the letter denote the level of statistical significance: **P < 0.01, ***P < 0.001.
Consistent with this view, Figure 3e,f shows that an analysis of thymus tissue for mRNA of the immune suppressive cytokines TGFβ and IL-10 demonstrated significant levels of TGFβ only in mice tolerized by intrathymic delivery; Figure 3f shows that there was a trend toward an increase in IL-10 only in the intrathymic group. Taken together, these data suggest that intrathymic vector delivery results in CD4+CD25+Foxp3+ Tregs in the thymus that may play a role in the observed tolerance. They also suggest that in terms of thymic Tregs, intravenous delivery results in a different long-term immune status. We note that the lack of intrathymic Treg markers after intravenous administration is at apparent odds with an earlier study,22 but there are significant differences between the studies, e.g., mouse models, that may contribute to this discrepancy.
In the periphery, intrathymic delivery generates CD4+CD25+ Tregs that produce suppressive cytokines, whereas intravenous delivery does not
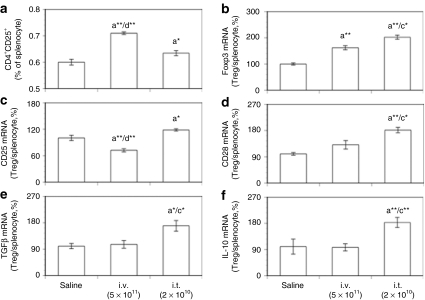
To compare directly the peripheral Tregs in mice resulting from tolerization by intrathymic and intravenous vector delivery, CD4+CD25+ cells were isolated from the spleen 16 weeks after AAV delivery and characterized for Treg markers. Figure 4a shows that a higher proportion of splenocytes were CD4+CD25+ cells in mice tolerized by an intravenous route than in mice tolerized by an intrathymic injection. Interestingly, although Figure 4b demonstrates that both intravenous and intrathymic delivery resulted in CD4+CD25+ cells with enhanced Foxp3 mRNA levels that may therefore play a role in maintaining peripheral tolerance, the Foxp3 mRNA levels were higher in the CD4+CD25+ cells resulting from intrathymic delivery.
Figure 4.
mRNA levels of Foxp3, CD markers, and cytokines by isolated CD4+CD25+ Tregs after intravenous or intrathymic injection of AAV8-hGAA. CD4+CD25+ Tregs were isolated from mouse spleen. mRNA expression by isolated CD4+CD25+ Tregs and total spleen cells at week 16 after AAV8-hGAA administration was analyzed by TaqMan with preamplification. (a) The percent of total spleen cells that are CD4+CD25+ (Tregs). Expression of mRNAs for (b) Foxp3, (c) CD25, (d) CD28, (e) TGFβ, and (f) IL-10 by these CD4+CD25+ cells relative to the corresponding mRNA in total splenocytes, normalized to actin mRNA and depicted as a percentage of the saline control. Data are shown as means ± SEM (N = 8–10 mice/group). Above a bar, the letter “a” represents a significant difference compared to the saline group, “c” compared to the i.v. (5 × 1011) group, and “d” compared to the i.t. (2 × 1010) group. The number of asterisks following the letter denote the level of statistical significance: *P < 0.05, **P < 0.01.
To characterize the splenic Tregs further, mRNA levels of the Treg markers CD25 and CD28 were determined in both isolated CD4+CD25+ cells and splenocytes. CD25 is required for Treg proliferation,23 whereas CD28 is not only required for the development of CD4+CD25+Foxp3+ cells in the thymus, but also contributes to Treg survival and homeostasis in the periphery.20,21 Figures 4c,d show that compared to cells from nontolerized (saline-treated) mice, the mRNAs for the Treg markers CD25 and CD28 were significantly increased only in mice tolerized by intrathymic delivery. Further, Figures 4e,f demonstrate that only the Tregs generated by intrathymic delivery had significantly greater TGFβ and IL-10 mRNA levels than the saline group, suggesting one mechanism by which these Tregs could exert immune suppressive activity.
Intrathymic vector injection results in a Treg mRNA profile in the liver, but intravenous injection does not
Because in thymic-tolerized mice isolated splenic Tregs (Figure 4) had the same mRNA profile as seen in the thymus (Figure 3), we next asked whether intrathymic injection led to Tregs in the liver with the potential of suppressing a humoral immune response there. Figure 5a–c demonstrates that as in thymus, liver mRNA levels of the Treg markers Foxp3, CD25, and CD28 were also elevated in mice tolerized by intrathymic but not intravenous delivery. Similarly, Figure 5d,e shows that mRNAs for the suppressive cytokines TGFβ and IL-10 were also significantly increased only in mice tolerized by intrathymic injection. Importantly, this mRNA profile is identical to that seen in the thymus (Figure 3) and in Tregs isolated from the spleen (Figure 4) after intrathymic injection, and it is consistent with the notion that hepatic homing of Tregs from the thymus occurred in mice tolerized by intrathymic injection.
Figure 5.
Liver mRNA levels of Foxp3, CD markers, and cytokines after intravenous or intrathymic injection of AAV8-hGAA. Liver mRNA expression levels of (a) Foxp3, (b) CD25, (c) CD28, (d) TGFβ, (e) IL-10, and (f) IL-2 were analyzed by TaqMan with preamplification at week 16 after administration of AAV8-hGAA. Results shown are normalized to actin expression and expressed as a percent of the saline control, and are presented as means ± SEM (N = 8–10 mice/group). Above a bar, the letter “a” represents a significant difference compared to the saline group, “b” compared to the i.v. (2 × 1010) group, and “c” compared to the i.v. (5 × 1011) group. The number of asterisks following the letter denote the level of statistical significance: *P < 0.05, **P < 0.01, ***P < 0.001.
Figure 5f also shows that IL-2 mRNA levels were significantly lower in the livers of mice tolerized by both routes. For mice dosed intrathymically, this result is consistent with the enhanced immune suppressive mRNA profile of this group (Figure 5d,e), one consequence of which is likely to be the inhibition of IL-2 production by immune cells.
In thymic-tolerized mice, B-cell (but not T-cell) maturation is inhibited in the periphery
In an attempt to understand the basis of tolerization induced by intrathymic and intravenous vector delivery, splenic T-cell and B-cell maturation markers were evaluated. For T cells, CD69 is one of the earliest markers of activation,24 whereas glucocorticoid-induced tumor necrosis factor receptor ligands (GITR), a member of the tumor necrosis factor superfamily, activates its receptor on T cells to generate critical co-stimulatory signals.25 Figure 6a,b shows that the mRNA levels of these two markers are significantly lower in the spleens of mice tolerized by intravenous vector delivery than in the spleens of nontolerized mice (saline or 2 × 1010 vector given intravenously) or mice tolerized by intrathymic vector delivery. These results suggest that T-cell activation is inhibited only in mice tolerized by intravenous delivery. Conversely, by these same criteria, T-cell activation appears normal, i.e., as in the saline group, in the spleens of mice that received either a low intravenous dose of vector (and were not tolerized) or a low intrathymic dose of vector (and were tolerized).
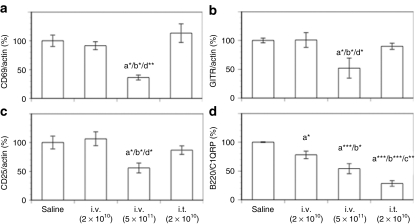
Figure 6.
Splenic mRNA levels of T- and B-cell markers after intravenous or intrathymic injection of AAV8-hGAA. Splenic mRNA expression levels of (a) CD69, (b) GITR, (c) CD25, and (d) B220/C1QRP were analyzed by TaqMan with preamplification at week 16 after administration of AAV8-hGAA. Results shown are normalized to actin expression and expressed as a percentage of the saline control, and are presented as means ± SEM (N = 8–10 mice/group). Above a bar, the letter “a” represents a significant difference compared to the saline group, “b” compared to the i.v. (2 × 1010) group, “c” compared to the i.v. (5 × 1011) group, and “d” compared to the i.t. (2 × 1010) group. The number of asterisks following the letter denote the level of statistical significance: *P < 0.05, **P < 0.01, ***P < 0.001.
The IL-2/CD25 (IL-2 receptor α) pathway is required for the clonal expansion and differentiation of T cells. Although CD4 mRNA levels were equivalent in all groups (data not shown), splenic IL-2 mRNA levels were significantly lower only in mice tolerized by intravenous delivery (data not shown). Relevant to this pathway, Figure 6c demonstrates that CD25 mRNA levels were significantly lower only in this same cohort. Taken together with the CD69 and GITR results (Figure 6a,b), these results suggest that although splenic T-cell activation and proliferation appear normal in mice tolerized by intrathymic vector injection, viz., equivalent to that of animals injected with saline (or intravenous AAV at 2 × 1010 drp) and then challenged with antigen, it is inhibited in mice tolerized by intravenous vector delivery.
For B cells, B220 expression increases with maturation, whereas C1QRP expression decreases.26 The B220/C1QRP mRNA ratio is therefore a relatively sensitive indicator that increases with B-cell maturation. Figure 6d shows that consistent with their anti-hGAA antibody titers (Figure 1), this ratio is significantly lower in tolerized (intravenous and intrathymic) than in nontolerized (saline and low-dose intravenous) mice, and implies a lower degree of B-cell maturation in mice tolerized by both delivery routes. Consistent with this interpretation, Figure 6d also shows that the low-dose intravenous group has a ratio intermediate between those of tolerized and nontolerized (saline-treated) mice, in accord with the intermediate level of anti-hGAA antibodies developed by this treatment (Figure 1). These lower B220/C1QRP mRNA ratios in the tolerized groups are a result of their lower B220 mRNA levels coupled with (for the intrathymic group) a higher C1QRP mRNA level than in the saline or low-dose intravenous groups (data not shown).
Taken together, the results depicted in Figure 6 suggest that the state of tolerance induced by intrathymic injection is characterized by a decrease in the ability of B cells to mature in response to antigen, whereas the tolerance generated by intravenous vector injection is characterized by blocks in both T- and B-cell activation and maturation.
Intrathymic injection does not result in tolerance to the AAV vector, and tolerance induction can be abrogated by pre-existing anti-AAV8 antibodies
Intrathymic injection of an adenovirus (AdV) vector led to immune tolerance to the vector itself.15 In contrast to the AdV result, Figure 7a shows that an analysis of the host humoral immune response to the AAV8 vector 2 weeks after an intrathymic injection resulted in anti-AAV8 titers equivalent to those obtained from intravenous administration of the same vector, i.e., immune tolerance to the AAV8 vector does not occur as a result of intrathymic administration. This result thus suggests that the immune tolerance induced by intrathymic delivery is antigen specific, i.e., it is directed only against the newly expressed transgene, hGAA.
Figure 7.
Antibody responses in naive and preimmunized mice receiving AAV8-hGAA by intravenous or intrathymic injection. (a) Antibody titers against AAV8 were determined in naive mice 2 weeks after receiving saline or AAV8-hGAA by intravenous delivery of 1 × 1010 or 5 × 1011 DNase-resistant particles (drp) or intrathymic delivery of 2 × 1010 drp. (b) Antibody titers against hGAA were determined at week 10 in (i) mice given intravenous saline at week 0, given 5 × 1011 drp AAV8-hGAA intravenously or 2 × 1010 intrathymically at week 2, and then challenged at week 8 with rhGAA in complete Freund's adjuvant (CFA), and (ii) mice preimmunized with 1× 1010 drp AAV8-EV at week 0, given 5 × 1011 drp AAV8-hGAA intravenously or 2 × 1010 intrathymically at week 2 and then challenged at week 8 with rhGAA in CFA. Data are expressed as means ± SEM (N = 5 mice/group).
As a therapeutic means of generating tolerance, an intrathymic vector administration might be advantageous relative to a systemic administration if it could more readily evade pre-existing anti-vector antibodies. To evaluate this possibility, mice were first immunized against AAV8 by intravenous administration of an AAV8 empty vector (AAV8-EV). After 2 weeks, they were dosed with AAV8-hGAA by intravenous (5 × 1011 drp) or intrathymic (2 × 1010 drp) delivery. Six weeks after the AAV8-GAA dose, they were challenged with rhGAA in CFA. In contrast to analogous deliveries of the same vector doses in naive animals (Figure 2b,c), neither delivery route led to detectable serum or thymic expression of hGAA in these AAV8-preimmunized mice (data not shown). Figure 7b demonstrates that AAV8-hGAA vector delivery by either delivery route in mice with pre-existing anti-AAV8 antibodies led to anti-hGAA antibody titers equivalent to those obtained by challenging naive animals with hGAA in CFA. These results thus demonstrate that pre-existing anti-AAV8 antibodies can block not only AAV8-mediated liver transduction by an intravenous injection, but also transduction of the thymus by a direct injection.
Discussion
In an effort to evaluate an AAV-based approach to generating immune tolerance for the purpose of making subsequent administrations of protein therapeutics more efficacious, we directly injected low-dose (2 × 1010 drp) vector into mouse thymus. In terms of humoral immune responses to protein challenge, the resulting tolerogenic state was found to be equivalent to that induced by an intravenous administration of a relatively high dose (5 × 1011 drp) of the same vector. The threshold of hGAA mRNA expression in the thymus required to generate tolerance was found to be >10,000-fold lower than the corresponding liver expression required for liver-based tolerance. It is worth pointing out that the promoter-driving transgene expression was originally designed for hepatic expression, and that tolerance was induced despite the relatively very low hGAA mRNA expression in the thymus. Thus, at least in a mouse naive to AAV8, direct injection of an AAV8 vector into the thymus can induce a state of immune tolerance to the gene product.
We have also compared the long-term (16 weeks postvector) state of immune tolerance to a foreign transgene product achieved by intrathymic injection of a low dose of AAV to that generated by a high systemic dose of the same vector. After intrathymic vector injection, mRNA for the markers CD4, CD25, CD28, and FoxP3 were seen to be elevated in the thymus, consistent with the presence of Tregs in this tissue. The mRNA levels of TGFβ and IL-10 were also increased in thymus, consistent with Tregs that have a suppressive phenotype mediated, at least in part, through these cytokines. The relatively high levels of IL-10 and TGFβ mRNA found in thymus, liver, and CD4+CD25+ splenocytes resulting from intrathymic vector delivery are consistent with an “activated” phenotype of mouse Tregs. Thus, for example, the levels of IL-10 mRNA are significantly increased in activated FoxP3-cleaved CD4+ T cells,27 and resting mouse Tregs that have been activated appear to display membrane-bound TGFβ28 that can exert suppressive effects on the innate immune system.29,30
Tregs that originate in the thymus characteristically express high levels of FoxP3 and CD28 and are immune suppressive. Intrathymic vector delivery resulted in the generation of not only thymic tissue with these characteristics (Figure 3), but also relatively high levels of these same markers in liver tissue (Figure 5) and in the (CD4+CD25+) Tregs isolated from the spleen (Figure 4), and support the notion that Tregs were generated in the thymus that were capable of subsequently migrating from the thymus to these secondary organs. In the spleen, our results (Figure 6) suggest that inhibiting anti-hGAA antibody production in the thymic-tolerized mice is not due to a decrease in splenic T-cell activation, but rather appears to be due to a decrease in B-cell maturation, possibly caused (directly or indirectly) by the thymically generated Tregs, and is consistent with previously recognized Treg inhibition of B-cell function.31
In contrast to this picture of tolerance resulting from intrathymic vector delivery, our results also imply that intravenous delivery results in a state of tolerance with quite different characteristics. For example, unlike the situation (16 weeks) after intrathymic delivery, we found no increase in Treg markers (Figure 5a–e), and low IL-2 mRNA levels (Figure 5f) in the livers of mice tolerized by intravenous delivery. However, the tolerance state generated by intravenous delivery appears to have resulted in Tregs in spleen (Figure 4a), and these cells are characterized by an apparent increase in the mRNA for FoxP3 (Figure 4b), although the per cell levels of these markers are lower than those resulting from intrathymic delivery. Again, in contrast to the Treg cells resulting from intrathymic delivery, the Tregs resulting from intravenous delivery do not have elevated levels of the mRNAs for TGFβ and IL-10, suggesting a different Treg phenotype. Nonetheless, previous adoptive transfer studies have shown that these Tregs are competent for inducing immune tolerance.17,22
Consistent with this different Treg phenotype resulting from intravenous vector delivery, the analysis of splenic T- and B-cell markers depicted in Figure 6 implies that in addition to some inhibition of B-cell maturation, both T-cell activation and proliferation are inhibited. In contrast, these T-cell processes do not appear to be affected as a result of intrathymic delivery. These intravenous delivery results are most consistent with an ongoing immune state of relative T-cell depletion. We speculate that T-cell depletion following intravenous delivery of vector to the liver could occur by apoptotic mechanisms involving antigen presentation by liver antigen presenting cells, viz., Kupffer cells, dendritic cells, and sinusoidal endothelial cells.32,33,34
From a preclinical point of view, direct intrathymic delivery of an AAV vector has been achieved in nonhuman primates using endoscopic guidance.35 In humans, direct intrathymic injection would be easiest in younger patients, in which the thymus is relatively large, and before it undergoes normal age-dependent atrophy. However, to use an intrathymic AAV approach in a clinical setting, it would be important to be able to transduce the thymus in the presence of anti-AAV antibodies; it would also be important that such an approach not lead to immune tolerance to the AAV vector itself. In contrast to a previous report15 in which intrathymic delivery of an AdV vector induced tolerance to the vector, we did not observe tolerance to the AAV8 vector (Figure 7a). This difference in outcome could be a result of the difference in ages of the mice in the two studies, viz, intrathymic injection of AAV8 was in 9- to 10-week-old mice, whereas intrathymic injection of AdV was in neonatal mice. In mice preimmunized with AAV8, neither thymic nor intravenous vector delivery resulted in tolerance to rhGAA (Figure 7b), which presumably results from the vector being neutralized by the high levels of pre-existing anti-AAV8 antibodies. Although it can be argued that these high anti-AAV8 antibody titers are not normally encountered in humans, it will be important to improve the ability of the vector to transduce the thymus, for example, by optimizing the details of the intrathymic delivery.
Finally, to translate this approach from mice to patients, it will also be important to (i) improve our ability to transduce the thymus while minimizing vector leakage, e.g., to the liver, (ii) further restrict expression to the thymus using a thymus-restricted promoter, e.g., the autoimmune regulator,36,37 and in this context (iii) demonstrate that physiologically relevant pre-existing levels of anti-AAV antibodies can be overcome.
Materials and Methods
AAV8 vectors. The AAV2/8-hGAA vector (AAV8-hGAA) contains the DC190 expression cassette and hGAA described previously.3,18 Briefly, DC190 contains a human serum albumin promoter to which are appended two copies of the human prothrombin enhancer. This cassette is largely hepatocyte-restricted. The AAV8 pseudotyped vector was purified by iodixanol gradient centrifugation followed by ion exchange chromatography over HiTrap Q HP Columns (GE Healthcare Bio-Science, Piscataway, NJ). TaqMan, a real-time PCR (RT-PCR) assay (ABI PRISM 7700; Applied Biosystems, Foster City, CA) with primers designed to amplify the vector-specific bovine growth hormone polyadenylation sequence, was used to determine the number of virus particles containing genomes in a sample, and is expressed as drp. An AAV2/8 vector bearing no transgene (AAV8-EV) was constructed and purified using the same procedures.
Animal studies. Six- to seven-week-old male C57BL/6N mice were purchased from Charles River (Wilmington, MA) and were acclimated for 1 week prior to being placed on study. Mice were housed at an AAALAC-accredited facility in accordance with the guidelines established by the National Research Council. All experiments were conducted under an approved IACUC protocol. After acclimation, mice received AAV8-EV for preimmunization or saline by intravenous delivery. After 2 weeks, mice received an intrathymic or intravenous injection of saline or AAV8-hGAA (in saline). Intrathymic injection was performed under surgical microscopy and required 5–10 minutes. Briefly, the anesthetized mouse was placed on its back with the head toward the investigator. A small incision (< 1 cm) was made to expose the apex of the thymus and one lobe injected with 10 µl of a solution containing AAV8-hGAA or saline; the incision was then sutured and analgesics provided for 48 hours.
Blood was collected from the orbital venous plexus of mice under anesthesia (2–3% isoflurane) using microhematocrit capillary tubes and transferred to serum separating tubes. Serum was separated by centrifugation at 10,000g for 10 minutes and then stored at −80 °C until assayed for hGAA, anti-hGAA, and anti-AAV8.
Immune tolerance to hGAA was evaluated in mice at 6 and 12 weeks after AAV8-hGAA administration by immune challenge with an intraperitoneal injection of 50 µg of purified rhGAA (Genzyme, Cambridge, MA) in 100 µl of saline emulsified with 100 µl of either CFA (Sigma-Aldrich, St Louis, MO) or IFA (Sigma-Aldrich). To assess immune tolerance to hGAA, serum was collected 2 weeks after each challenge and assayed for anti-hGAA antibody titers.
Measurement of serum hGAA levels and antibody titers. Serum levels of hGAA, anti-hGAA, and anti-AAV8 antibody titers were determined by an enzyme-linked immunosorbent assay as described previously.3 Briefly, 96-well high-protein-binding microtiter plates (Costar, New York, NY) were coated with the following: (i) a monoclonal antibody specific for hGAA (Genzyme) at 1 µg/ml to quantify serum hGAA, (ii) rhGAA (Genzyme) at 5 µg/ml to quantify antibodies against hGAA, or (iii) AAV8 (Genzyme) at 1 µg/ml to quantify antibodies against AAV8. After blocking, diluted serum samples or standards were added to the wells in duplicate, followed by a secondary, biotinylated monoclonal antibody at 1 µg/ml and streptavidin-horseradish peroxidase at 2 µg/ml (Pierce Biotechnology, Rockford, IL) for serum hGAA, and horseradish peroxidase–conjugated goat anti-mouse immunoglobulin G (IgG, IgM, and IgA; Invitrogen Zymed, South San Francisco, CA) for anti-hGAA and anti-AAV8 antibody titers. Color was developed with 3,3′,5,5′-tetramethylbenzidine (BioFX Laboratories, Owings Mills, MD), the reaction stopped with BioFX stop solution, and the plates read using absorbance at 450 nm on a Spectromax Plus plate reader (Molecular Devices, Sunnyvale, CA).
Assay for hGAA, mouse Foxp3, CD marker, and cytokine mRNAs. Spleen cells were collected from mouse spleen using two filters (cell strainer 70 and 40 µm; BD Falcon, San Jose, CA). Total cells were counted after red blood cells had been lysed. CD4+CD25+ cells were isolated using the mouse CD4+CD25+ Treg isolation kit (Miltenyi Biotec, Auburn, CA). Total RNA was extracted from mouse thymus, spleen, liver, isolated splenocytes, and isolated CD4+CD25+ cells using the RNeasy Lipid Tissue Mini kit (Qiagen, Valencia, CA). Total RNA concentrations were quantified by A260 (NanoDrop ND-1000 Spectrophotometer; NanoDrop Technologies, Wilmington, DE). cDNA was synthesized and preamplificated (except for hGAA in liver) using the TaqMan PreAmplification master mix kit (Applied Biosystems), amplifying 15 cycles for hGAA in thymus (without preamplification thymic hGAA mRNA copies were below the detection limit of 100 copies/125 ng total RNA; for increased accuracy, the resulting copy numbers are expressed as a percentage of copies found after intrathymic delivery), and 14 cycles for other genes. A pooled TaqMan assay for the preamplification step contained TaqMan Gene Expression Assay (Applied Biosystems) for various genes at 0.2× dilution as recommended by the kit, with the exception of a 0.002× dilution for mouse actin. After diluting the preamplified cDNA products from 1× to 1000×, the resulting solutions were used to quantify mRNA expression using the 7500 RT-PCR system (Applied Biosystems) and the standard protocol. The optimal dilution for each gene was adjusted based on prescreening results from tested samples to detect signal between cycle thresholds 20–26. Standard copy number curves were generated using hGAA plasmid DNA for hGAA, and total RNA for other genes. Except for hGAA, mRNA expression from other genes was normalized to actin expression and presented as a percent of the saline control. To minimize variability, mRNA expression by CD4+CD25+ cells isolated from splenocytes was presented as a ratio of the mRNA expression by CD4+CD25+ Tregs over that of total splenocytes, expressed as a percentage of the saline control value.
Statistics/error analysis. Results comparing multiple groups were analyzed by one-way analysis of variance followed by a Newman–Keuls test. Values shown represent means, and error bars represent standard error of the mean. Asterisks are used to denote results with significant differences at the levels of *P < 0.05, **P < 0.01, and ***P < 0.001. The letter “a” represents significant differences compared to the saline group, “b” to the intravenous (2 × 1010 drp), “c” to the intravenous (5 × 1011 drp), and “d” to the intrathymic (2 × 1010 drp) group.
Acknowledgments
We express our sincere thanks to Joszef Karman for a critical reading of the manuscript, Jennifer Nietupski and the Vector Production group for the AAV vectors, Scott Bercury and Robin Ziegler for helpful discussions, and the Department of Comparative Medicine for assistance with the animal studies.
REFERENCES
- Joseph A, Munroe K, Housman M, Garman R., and, Richards S. Immune tolerance induction to enzyme-replacement therapy by co-administration of short-term, low-dose methotrexate in a murine Pompe disease model. Clin Exp Immunol. 2008;152:138–146. doi: 10.1111/j.1365-2249.2008.03602.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishnani PS, Goldenberg PC, DeArmey SL, Heller J, Benjamin D, Young S, et al. Cross-reactive immunologic material status affects treatment outcomes in Pompe disease infants. Mol Genet Metab. 2010;99:26–33. doi: 10.1016/j.ymgme.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler RJ, Bercury SD, Fidler J, Zhao MA, Foley J, Taksir TV, et al. Ability of adeno-associated virus serotype 8-mediated hepatic expression of acid alpha-glucosidase to correct the biochemical and motor function deficits of presymptomatic and symptomatic Pompe mice. Hum Gene Ther. 2008;19:609–621. doi: 10.1089/hum.2008.010. [DOI] [PubMed] [Google Scholar]
- Zhu Y, Jiang JL, Gumlaw NK, Zhang J, Bercury SD, Ziegler RJ, et al. Glycoengineered acid alpha-glucosidase with improved efficacy at correcting the metabolic aberrations and motor function deficits in a mouse model of Pompe disease. Mol Ther. 2009;17:954–963. doi: 10.1038/mt.2009.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu Q, Joseph M, Przybylska M, Yew NS., and, Scheule RK. Transient siRNA-mediated attenuation of liver expression from an alpha-galactosidase A plasmid reduces subsequent humoral immune responses to the transgene product in mice. Mol Ther. 2005;12:264–273. doi: 10.1016/j.ymthe.2005.04.007. [DOI] [PubMed] [Google Scholar]
- Mingozzi F, Liu YL, Dobrzynski E, Kaufhold A, Liu JH, Wang Y, et al. Induction of immune tolerance to coagulation factor IX antigen by in vivo hepatic gene transfer. J Clin Invest. 2003;111:1347–1356. doi: 10.1172/JCI16887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waksman BH. Tolerance, the thymus, and suppressor T cells. Clin Exp Immunol. 1977;28:363–374. [PMC free article] [PubMed] [Google Scholar]
- Staples PJ, Gery I., and, Waksman BH. Role of the thymus in tolerance. 3. Tolerance to bovine gamma globulin after direct injection of antigen into the shielded thymus of irradiated rats. J Exp Med. 1966;124:127–139. doi: 10.1084/jem.124.2.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derbinski J, Gäbler J, Brors B, Tierling S, Jonnakuty S, Hergenhahn M, et al. Promiscuous gene expression in thymic epithelial cells is regulated at multiple levels. J Exp Med. 2005;202:33–45. doi: 10.1084/jem.20050471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa Cardoso R, Magalhães DA, Baião AM, Junta CM, Macedo C, Marques MM, et al. Onset of promiscuous gene expression in murine fetal thymus organ culture. Immunology. 2006;119:369–375. doi: 10.1111/j.1365-2567.2006.02441.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magalhães DA, Silveira EL, Junta CM, Sandrin-Garcia P, Fachin AL, Donadi EA, et al. Promiscuous gene expression in the thymus: the root of central tolerance. Clin Dev Immunol. 2006;13:81–99. doi: 10.1080/17402520600877091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Posselt AM, Barker CF, Tomaszewski JE, Markmann JF, Choti MA., and, Naji A. Induction of donor-specific unresponsiveness by intrathymic islet transplantation. Science. 1990;249:1293–1295. doi: 10.1126/science.2119056. [DOI] [PubMed] [Google Scholar]
- Madoiwa S, Yamauchi T, Kobayashi E, Hakamata Y, Dokai M, Makino N, et al. Induction of factor VIII-specific unresponsiveness by intrathymic factor VIII injection in murine hemophilia A. J Thromb Haemost. 2009;7:811–824. doi: 10.1111/j.1538-7836.2009.03314.x. [DOI] [PubMed] [Google Scholar]
- Marodon G, Fisson S, Levacher B, Fabre M, Salomon BL., and, Klatzmann D. Induction of antigen-specific tolerance by intrathymic injection of lentiviral vectors. Blood. 2006;108:2972–2978. doi: 10.1182/blood-2006-03-010900. [DOI] [PubMed] [Google Scholar]
- DeMatteo RP, Chu G, Ahn M, Chang E, Barker CF., and, Markmann JF. Long-lasting adenovirus transgene expression in mice through neonatal intrathymic tolerance induction without the use of immunosuppression. J Virol. 1997;71:5330–5335. doi: 10.1128/jvi.71.7.5330-5335.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marodon G., and, Klatzmann D. In situ transduction of stromal cells and thymocytes upon intrathymic injection of lentiviral vectors. BMC Immunol. 2004;5:18. doi: 10.1186/1471-2172-5-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler RJ, Lonning SM, Armentano D, Li C, Souza DW, Cherry M, et al. AAV2 vector harboring a liver-restricted promoter facilitates sustained expression of therapeutic levels of alpha-galactosidase A and the induction of immune tolerance in Fabry mice. Mol Ther. 2004;9:231–240. doi: 10.1016/j.ymthe.2003.11.015. [DOI] [PubMed] [Google Scholar]
- Chu Q, Moreland R, Yew NS, Foley J, Ziegler R., and, Scheule RK. Systemic Insulin-like growth factor-1 reverses hypoalgesia and improves mobility in a mouse model of diabetic peripheral neuropathy. Mol Ther. 2008;16:1400–1408. doi: 10.1038/mt.2008.115. [DOI] [PubMed] [Google Scholar]
- Voehringer D, Blaser C, Grawitz AB, Chisari FV, Buerki K., and, Pircher H. Break of T cell ignorance to a viral antigen in the liver induces hepatitis. J Immunol. 2000;165:2415–2422. doi: 10.4049/jimmunol.165.5.2415. [DOI] [PubMed] [Google Scholar]
- Guo F, Iclozan C, Suh WK, Anasetti C., and, Yu XZ. CD28 controls differentiation of regulatory T cells from naive CD4 T cells. J Immunol. 2008;181:2285–2291. doi: 10.4049/jimmunol.181.4.2285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golovina TN, Mikheeva T, Suhoski MM, Aqui NA, Tai VC, Shan X, et al. CD28 costimulation is essential for human T regulatory expansion and function. J Immunol. 2008;181:2855–2868. doi: 10.4049/jimmunol.181.4.2855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao O, Dobrzynski E, Wang L, Nayak S, Mingle B, Terhorst C, et al. Induction and role of regulatory CD4+CD25+ T cells in tolerance to the transgene product following hepatic in vivo gene transfer. Blood. 2007;110:1132–1140. doi: 10.1182/blood-2007-02-073304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornton AM. Signal transduction in CD4+CD25+ regulatory T cells: CD25 and IL-2. Front Biosci. 2006;11:921–927. doi: 10.2741/1848. [DOI] [PubMed] [Google Scholar]
- Santis AG, López-Cabrera M, Sánchez-Madrid F., and, Proudfoot N. Expression of the early lymphocyte activation antigen CD69, a C-type lectin, is regulated by mRNA degradation associated with AU-rich sequence motifs. Eur J Immunol. 1995;25:2142–2146. doi: 10.1002/eji.1830250804. [DOI] [PubMed] [Google Scholar]
- Esparza EM., and, Arch RH. Glucocorticoid-induced TNF receptor functions as a costimulatory receptor that promotes survival in early phases of T cell activation. J Immunol. 2005;174:7869–7874. doi: 10.4049/jimmunol.174.12.7869. [DOI] [PubMed] [Google Scholar]
- Rolink AG, Andersson J., and, Melchers F. Molecular mechanisms guiding late stages of B-cell development. Immunol Rev. 2004;197:41–50. doi: 10.1111/j.0105-2896.2004.0101.x. [DOI] [PubMed] [Google Scholar]
- de Zoeten EF, Lee I, Wang L, Chen C, Ge G, Wells AD, et al. Foxp3 processing by proprotein convertases and control of regulatory T cell function. J Biol Chem. 2009;284:5709–5716. doi: 10.1074/jbc.M807322200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura K, Kitani A., and, Strober W. Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med. 2001;194:629–644. doi: 10.1084/jem.194.5.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghiringhelli F, Ménard C, Martin F., and, Zitvogel L. The role of regulatory T cells in the control of natural killer cells: relevance during tumor progression. Immunol Rev. 2006;214:229–238. doi: 10.1111/j.1600-065X.2006.00445.x. [DOI] [PubMed] [Google Scholar]
- Smyth MJ, Teng MW, Swann J, Kyparissoudis K, Godfrey DI., and, Hayakawa Y. CD4+CD25+ T regulatory cells suppress NK cell-mediated immunotherapy of cancer. J Immunol. 2006;176:1582–1587. doi: 10.4049/jimmunol.176.3.1582. [DOI] [PubMed] [Google Scholar]
- Lim HW, Hillsamer P, Banham AH., and, Kim CH. Cutting edge: direct suppression of B cells by CD4+ CD25+ regulatory T cells. J Immunol. 2005;175:4180–4183. doi: 10.4049/jimmunol.175.7.4180. [DOI] [PubMed] [Google Scholar]
- Crispe IN. Hepatic T cells and liver tolerance. Nat Rev Immunol. 2003;3:51–62. doi: 10.1038/nri981. [DOI] [PubMed] [Google Scholar]
- Crispe IN, Giannandrea M, Klein I, John B, Sampson B., and, Wuensch S. Cellular and molecular mechanisms of liver tolerance. Immunol Rev. 2006;213:101–118. doi: 10.1111/j.1600-065X.2006.00435.x. [DOI] [PubMed] [Google Scholar]
- Huang L, Soldevila G, Leeker M, Flavell R., and, Crispe IN. The liver eliminates T cells undergoing antigen-triggered apoptosis in vivo. Immunity. 1994;1:741–749. doi: 10.1016/s1074-7613(94)80016-2. [DOI] [PubMed] [Google Scholar]
- Moreau A, Vicente R, Dubreil L, Adjali O, Podevin G, Jacquet C, et al. Efficient intrathymic gene transfer following in situ administration of a rAAV serotype 8 vector in mice and nonhuman primates. Mol Ther. 2009;17:472–479. doi: 10.1038/mt.2008.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein L, Roettinger B., and, Kyewski B. Sampling of complementing self-antigen pools by thymic stromal cells maximizes the scope of central T cell tolerance. Eur J Immunol. 2001;31:2476–2486. doi: 10.1002/1521-4141(200108)31:8<2476::aid-immu2476>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- Anderson MS, Venanzi ES, Klein L, Chen Z, Berzins SP, Turley SJ, et al. Projection of an immunological self shadow within the thymus by the aire protein. Science. 2002;298:1395–1401. doi: 10.1126/science.1075958. [DOI] [PubMed] [Google Scholar]