Abstract
Endoscopy is a widely used diagnostic tool to detect reflux esophagitis. Although its specificity was reported to be excellent at 90%-95%, its sensitivity was only 50%. Therefore, it is quite difficult to detect these lesions under the standard white light endoscopy especially in patients with minimal change esophageal reflux disease (MERD). In recent years, endoscopic technologies have evolved tremendously; these include high resolution and magnification digital chromoendoscopy. These technologies are useful practically for detecting various subtle lesions along the gastrointestinal tract starting from esophagus to colon. Currently, these technologies can be classified in 2 systems; pre-processed system (NBI, Olympus) and post processed system (FICE and i-SCAN, Fujinon and Pentax respectively). Over a few years, there have been many emerging publications on the benefit of these systems on MERD detection. The overall sensitivities to diagnose MERD were reported as much better than controls. However, large, multi-center and randomized controlled studies comparing these new imaging modalities with the conventional white light chromoendoscopy are warranted to validate its accuracy. Standard, simple and precise endoscopic reading criteria for the identification of MERD are also required.
Keywords: Digital, Chromoendoscopy, Minimal change, Erosive reflux esophagitis, Diagnosis, Pre-processed, Post processed
INTRODUCTION
Gastroesophageal reflux disease (GERD) is a major acid related health problem which is increasing in prevalence all over the world[1]. During the last two decades, when GERD was defined by symptoms of at least weekly heartburn and/or acid regurgitation, its prevalence was approximately 20% in Western countries whereas its prevalence was less than 5% in Eastern countries[2]. However, its trend in Asia has recently increased to almost 10%[3].
Endoscopy is a widely used diagnostic tool for reflux esophagitis detection. Although its specificity was reported to be excellent at 90%-95%[4], its sensitivity was only 50%[5]. More than half of patients with GERD symptoms have been diagnosed as non-erosive reflux disease (NERD) and this is more common in Asian (59%-87%) than in Western patients (54%)[6-8]. Indeed, by careful analysis, the majority of NERD patients did not have completely normal endoscopic finding but were found to have subtle distal esophageal mucosal changes from acid refluxate such as reddish or whitish colors, edema of mucosal folds, blurring with friability of mucosal junction, increased vascularity and microerosion[9-10]. Unfortunately, it is quite difficult to detect these lesions under the standard white light endoscopy, particularly by inexperienced endoscopists (kappa value for interobserver agreement by inexperienced endoscopist for minimal changes was reported to be low and ranged between 0.19 and 0.36)[10]. Over a few years, endoscopic technologies have evolved tremendously including high resolution and magnification digital chromoendoscopy. These technologies are practically useful for detecting various subtle lesions along the gastrointestinal (GI) tract starting from esophagus to colon. Other than the detection of subtle change reflux esophagitis, these have proven to be beneficial for Barrett’s epithelium detection[11], targeted biopsy of the suspicious area in gastric intestinal metaplasia[12] and detecting early gastric and colon cancers[13-14]. This review will address the concept and provide a practical use of these novelties for the detection of subtle changes in GERD. In particular, all minimal changes reflux disease will be called “minimal change esophageal reflux disease (MERD)”.
USE OF CHROMOENDOSCOPY IN DIAGNOSING GERD AND MERD
The idea of using dye for tissue staining in gastrointestinal endoscopy is mainly to enhance the contrast of different GI mucosa. Currently, there are 2 groups of dye that are categorized by the effect on tissue staining. First is the vital stained dye that is absorbed into mucosal cells and the color of the absorbed mucosa will change depending on the type of dyes and absorptive cells. Examples of these vital stains are Lugol’s solution, methylene blue and Congo red Lugol’s solution is the most widely used dye for diagnosing reflux esophagitis. This agent is rapidly absorbed by normal squamous epithelial cells containing glycogen and it subsequently yields a dark-brown color. The unstained area is delineated as abnormal mucosa since the inflamed mucosa contains no glycogen. Another use of Lugol’s solution is to detect neoplastic squamous mucosa including high grade dysplasia and early squamous cell carcinoma of the esophagus. Another agent, methylene blue, is not absorbed into metaplastic or dysplastic epithelial cells and helps the delineation of the area of esophageal metaplasia from normal mucosa. It is mostly used for Barrett’s epithelium detection. With its pH dependent character, Congo red is another dye that is occasionally used for detection of abnormal lesions in the stomach. It turns dark when the pH of the mucosa drops to acidic level and this in turn is helpful to identify the un-dark area of atrophic gastritis which is prone to develop gastric cancer.
The other dye is non-vital dye which can not be absorbed into the cells but can emphasize the contrast enhancement between different structures by filling in the grooves and folds of the gastrointestinal mucosa. The prototypes for this dye are indigocarmine and crystal violet that are practically used for the detection of flat and depressed lesions of the GI tract. In addition, this dye can be helpful for highlighting the mucosal irregularities in Barrett’s esophagus.
Chromoendoscopy was introduced to facilitate mucosal characterization and localization of abnormal area of the esophageal mucosa in the 1970’s[15]. In 1989, Misumi et al[16] demonstrated the benefit of Lugol chromoendoscopy as a potential useful diagnostic tool in suspected GERD. Twenty one patients whose reflux esophagitis was diagnosed by conventional endoscopy were examined under Lugol’s stain and biopsies were taken from the stained and unstained mucosa for pathological comparison. The positive findings from Lugol’s unstained images were more concordant with positive histological findings. In addition, the extension and degree of reflux changes were also better defined with Lugol’s (89.9% vs 69.7%, P < 0.001).
In 2005, Yoshikawa et al[17] reported the usefulness of endoscopy with Lugol’s iodine solution for the diagnosis of NERD. Sixty-one patients with typical reflux symptoms and 42 controls underwent a standard white light upper endoscopy. Twenty-two of 61 (36%) reflux patients and 4 of 42 controls had visible esophageal injuries by standard endoscopy. Thirty-nine patients with endoscopy-negative but positive reflux symptoms (NERD) and 38 endoscopy negative asymptomatic controls later underwent Lugol chromoendoscopy. Unstained streaks in the distal esophagus were observed in 19 of 39 (49%) patients with NERD whereas only one out of 38 controls (2.6%) was positive. The biopsy specimens were obtained from the Lugol unstained areas and the adjacent stained mucosa for histopathological comparison which served as the gold standard. When compared to the stained mucosa, the unstained areas significantly contained more typical pathologic changes related to reflux esophagitis, namely thicker basal cell layers (30.9 ± 7.6% vs 12.3 ± 4.5% of total epithelial thickness, P < 0.001), longer papillae (57.9 ± 12.6% vs 38.1 ± 12.6% of total epithelial thickness, P < 0.01), and higher numbers of intraepithelial lymphocytes (9.6 ± 5.7 vs 6.0 ± 4.0 per 3 high-power fields, P < 0.01). Although this method was helpful in diagnosing microerosions in NERD patients and claimed to be safe and easy, it was time consuming and needed additional spraying equipment. In addition, the adverse reactions of Lugol’s solutions which include esophageal mucosal irritation causing retrosternal chest pain or discomfort[18], acute necrotizing esophagitis[19] and allergic reactions (laryngospasm, bronchospasm and cardiac arrest)[20] need to be considered.
USE OF HIGH-RESOLUTION AND MAGNIFICATION ENDOSCOPY IN DIAGNOSING MERD
The recent advances in optical engineering that include charge couple device (CCD) and zoom lens, provide high resolution and magnifying (up to 150-fold) images. This technology can enhance the visualization of mucosal details. Kiesslich et al[21] compared the endoscopic results by magnifying endoscopy (EG3470ZK, Pentax, Hamburg, Germany and 160Z, Olympus, Hamburg, Germany) in NERD patients with the results in asymptomatic controls. The positive endoscopic criteria included increased vascular markings at proximal and distal to Z-line, punctate erythema at proximal to Z-line, squamous islands at distal to Z-line and villous-appearing mucosa adjacent to distal Z-line. They reported that the punctate erythema was the only criterion found more often in NERD patients than controls (44% vs 10%, P = 0.009). When all criteria were combined, the sensitivity and specificity of magnifying endoscopy were 64% and 85% respectively. The positive predictive value (PPV) was 80%, the negative predictive value (NPV) was 70% and the accuracy was 74% for the diagnosis of MERD.
COMBINATION OF CHROMOENDOSCOPY AND HIGH-RESOLUTION MAGNIFYING ENDOSCOPY
The diagnostic criteria for MERD using the combination of Lugol chromoendoscopy with high-resolution magnifying system was proposed by Tam et al[22]. Ten patients with reflux symptoms were examined by conventional upper endoscopy followed by high resolution magnifying endoscopy (Fujinon 850K CCD, Omiya, Japan). Lugol’s dye was sprayed during the procedure. The examinations were performed before and after a 4 wk course of oral omeprazole. In patients with unremarkable examination by conventional endoscopy, they found that certain subtle changes at the distal esophagus including pin-point capillaries, loss of longitudinal vessels and branching blood vessels were very useful to increase the yield for MERD diagnoses. Another study was performed by Edebo et al[23]. Eleven NERD patients diagnosed by questionnaire or conventional endoscopy and 10 controls were examined by high resolution magnifying chromoendoscopy (optical magnification of x35 and digital magnification of x2, EG485-ZH, Fujinon Co., Omiya Japan) and 24 h pH monitoring was also used as another mode for reflux diagnosis. Six of 8 control subjects had negative results on pH test whereas 8 of 11 symptomatic patients had positive results on the pH test. The patients were reexamined with magnifying endoscopy after 4 wk of PPI treatment. Still images were assessed by experienced endoscopists. Seven endoscopic criteria for diagnosis of MERD were proposed as follows: triangular indentations of villiform columnar mucosa into the squamous mucosa, apical mucosal breaks at the vertex of a triangular indentation, obscured pallisade blood vessels above the squamocolumnar junction (SCJ), pin-point blood vessels above the SCJ, branching blood vessels below the SCJ, serrated SCJ and villiform mucosa below the SCJ. When only symptomatic criteria were used as a gold standard for reflux diagnosis, triangular lesions, apical mucosal breaks and the presence of pin-point blood vessels above the SCJ were found more frequently in MERD patients than controls (86% vs 63%, 57% vs 38% and 55% vs 38% respectively). Moreover, the prevalence of triangular lesions, apical mucosal breaks and the presence of pin-point blood vessels above the SCJ decreased in MERD patients after PPI treatment (43%, 10% and 22%, respectively). When the pH monitoring test was used as the gold standard for reflux diagnosis, these 3 findings presented significantly more frequently in patients than in controls (92% vs 45%, 60% vs 20% and 67% vs 25% respectively, P < 0.05). Likewise, in the symptomatic criteria cases, the frequencies of these positive criteria were significantly decreased after PPI treatment to 45%, 10% and 21% respectively in the reflux group. Unfortunately, the interobserver agreement was quite low for each criterion (kappa value, 0.18-0.28). Therefore, these criteria are still too premature to be used as standard criteria to diagnose MERD. Easier MERD diagnostic criteria are necessary for practicing endoscopists for a short learning curve and better interobserver agreement value.
Concept of digital chromoendoscopy
Generally, white light can be visibly shaded into 7 colors. Different colors carry different wavelengths. The depth of light penetration into gastrointestinal mucosa depends conversely on the wavelength. For instance, violet has the shortest wavelength at 400 nm. Therefore, violet is not able to penetrate the mucosa as deep as red which has a longer wavelength at 700 nm. Blue, green and yellow are colors that have wavelengths in between violet and red. Thus, the depths of penetration of these colors are ranged from 0.15 mm to 0.30 mm[24].
Hemoglobin is the main substance that is responsible for visible light absorption with the peak absorption in the blue part (415 nm). Mucosal structures containing hemoglobin such as blood vessels can absorb 415 nm light and produce a dark brown color that is different to the brighter surrounding mucosa that reflects the blue light. Hypervascularized tissues including malignant or inflammatory masses absorb this blue light and can be identified in a dark brown area.
Currently, digital chromoendoscopy can be categorized into 2 systems; the system that filters only the blue wave length to spot on the object can be called the pre processed whereas the post processed system is the system that reprocesses only certain wave lengths from the objective picture after white light illumination. The representative of the pre processed is a narrow band imaging (NBI) that has been introduced by Olympus Company. Currently, Olympus provides 2 systems for the market; non-sequential system (Excera II 180 series) and sequential system (Lucera II 280 series). Excera system uses a color charge coupling device (CCD) and uses an additional filter that selects only blue band (415 nm). This system is widely available outside Japan. Another system, Lucera system, contains 2 filters; one is an RGB filter for black and white CCD and the other is an NBI filter. This system is mainly available in Japan, South Korea, the UK and some tertiary academic research centers. By contrast, the post processed system relies on the computer system that enhances the image contrast by reprocessing the captured images. By selecting only the preferred spectrum of wavelength, the computer can generate a new picture that can highlight the area of inflammation and cancer. There are 2 models currently available; Intelligent Color Enhancement [Fujinon (FICE)] and I-scan (Pentax). FICE generates a very large number of wavelength permutations with increments of 5 nm. There are 10 stations that the factory assigned for an instant use. These stations are adjustable according to users’ preference. The advantage of the post processed system is that all preferred wavelengths are adjustable. Since there has been no standard guideline that advocates the use of these different colors another question to be answered is what wavelength is more preferred for the detection of different gastrointestinal tract lesions and locations.
i-SCAN is a new computerized dynamic digital image processor which provides high resolution enhanced images. There are currently 4 modes available; standard (HD+) mode, contrast enhancement (CE) mode, surface enhancement (SE) mode and tone enhancement (TE) mode. Each mode provides different images features and can be used alone or combined with the others. CE mode highlights the mucosal surface and pit pattern and SE mode enhances the mucosal surface, pit pattern, vascularity and tissue topography. Both CE and SE mode provide pictures in natural color tone whereas TE mode provides images like those obtained from chromoendoscopy. Other than the use for diagnosis of MERD, preliminary data also reported its utility for detection of early gastric cancer lesions[25].
Utility of NBI for detection of MERD
NBI is an innovative technique that uses the optical narrow band filters of blue light for object illumination. As mentioned above, the depth of penetration depends conversely on the wavelength; hence the blue light which is the short wavelength (450 nm) can attain only the superficial layers. This in turn can provide the images of the superficial structure of the mucosa including the capillaries and details of the mucosal patterns. In conjunction with the magnification system, it enables highlighting the patterns of “intrapapillary capillary loops (IPCLs)” which contains abnormal figures indicating the inflammatory process and neoplasia[26-27] (Figure 1). This is considered a great advantage over the simple chromoendoscopy that can not clearly detail the vascular patterns because they are concealed by the stain agents.
Figure 1.
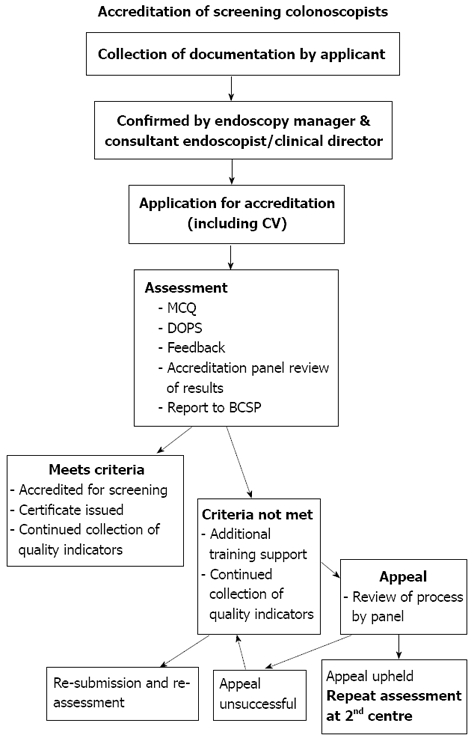
Images from magnifying non-sequential NBI show increased number and dilated and tortuous of intrapapillary capillary loops.
The utility of NBI magnification system as the diagnostic tool of GERD has been increasingly explored[28-30]. The landmark study on NERD was recently conducted in 50 symptomatic GERD patients and 30 controls by Sharma et al[29]. The still images of 4 quadrants of the distal 5 cm of esophagus were captured by both conventional endoscopy and sequential NBI with magnification (Olympus GIF Q240 Z, 115x, Olympus Evis Lucera system). Patients with mucosal breaks identified by standard upper endoscopy were classified as erosive esophagitis based on the Los Angeles classification. Patients with no visible mucosal break on standard upper endoscopy were classified as NERD. Positive endoscopic criteria to diagnose reflux esophagitis were proposed as follows: microerosions, increased vascularity at SCJ, ridge or villous pattern below SCJ, columnar islands in the distal esophagus and increased/tortuous/dilated IPCLs. They showed that the increased number of IPCLs was a predictor of the presence of erosive esophagitis (OR 17.8; 95% CI: 4.7-67.8). The increased number (OR 4.7; 95% CI: 1.1-21.2) and dilated IPCLs (OR 6.2; 95% CI: 1.5-25.3) were shown to be the best parameters for the diagnosis of MERD. The average numbers of 117 IPCLs per field yielded 90% sensitivity and 70% specificity to determine minor change of reflux disease (MERD) and the average count of 123 IPCLs per field provided a sensitivity of 86% and a specificity of 76% for the diagnosis of significant erosive esophagitis (GERD). Additionally, good interobserver and moderate intraobserver agreements among the investigators were noted with kappa value ranged from 0.48-0.88 and 0.39-0.52 respectively. Although this technique is very promising, there are significant limitations for practical use. Firstly, it was impractical to use some of these endoscopic criteria since they are too ideal. For instance, dilated and/or tortuous IPCLs are very subjective whereas the objective criteria, particularly the number of IPCLs, are quite difficult to accurately count. In addition, the non- magnified standard scope cannot be used because the zoom mode is mandatory to allow optimal inspection of IPCLs and the scope tip needs to be focused closer to the mucosa than the white light system. Therefore, a transparent plastic cap attachment is a requisite to maintain the precise distance between scope tip and the mucosa. As a result, it requires tremendous familiarity and a long learning curve to adjust the scope use. Secondly, the examination time required during the procedure was not mentioned. This method is anticipated to be time-consuming and quite complicated because only a small area can be magnified and examined in each scope touch. Therefore, adequate duration of examination is required for a complete study of the whole esophageal circumference and for a complete manual count of the IPCLs amount in each field. Thirdly, the original article used only two investigators for the assessment of the images and the differences in the values of intraobserver agreement rates were not clearly explained. Therefore, the validation of interobserver agreements and simpler criteria are mandatory before applying this method for the routine clinical practice. Lastly, the sequential NBI system that was used in this study is available only in some countries and research centers. We need to clarify that the results can be reproducible with the commercial non-sequential system.
Very recently, the role of NBI in the diagnosis of MERD has been re-evaluated by Fock et al[30]. They compared the conventional endoscopy and non-sequential NBI (Olympus CLV-160B, Olympus Evis Exera II system) under normal and digital magnification of 1.5 x in symptomatic GERD patients and asymptomatic controls. Erosive esophagitis was diagnosed by using the Los Angeles Classification whereas NERD was defined by the presence of reflux symptoms without mucosal breaks on standard white light endoscopy and the response to PPI treatment. Mucosal morphology at SCJ from conventional endoscopy and NBI were compared among 41 erosive reflux disease (ERD), 36 NERD and 30 control patients. The endoscopic criteria comprised of the presence of micro-erosions (mucosal breaks only visible on NBI but not on standard white light endoscopy), increased vascularity at the SCJ (vascular spots or punctuate erythema above the SCJ), increased erythema just below the SCJ and the presence of columnar epithelium island above the SCJ. When comparing this with the study using magnifying scope with the Olympus sequential system, the punctuate erythema by Olympus non-sequential system corresponded with the IPCLs described by Sharma et al[29]. The results showed that the presence of micro-erosions and the increase in vascularity at SCJ were significantly more common in NERD patients compared to controls (52.8% vs 23.3% and 91.7% vs 36.7% respectively). The investigators also reported a significant difference in mucosal pit patterns at the distal esophagus between NERD patients and control. The round pit pattern which represents the gastric epithelial mucosa was found less frequently in NERD patients than in the control group (5.6% vs 70%, P < 0.001). Interestingly, the prevalence of the increase in vascularity and round pit pattern was similar in patients with ERD and NERD. Therefore, the increase in vascularity with the absence of round pit pattern was helpful to differentiate NERD from controls with a sensitivity of 86.1% and specificity of 83.3% respectively. In addition, interobserver agreement determined among 3 endoscopists who performed the en-doscopic procedures appeared to be very good for the presence of micro-erosions (kappa value, 0.89; SE, 0.08), increased vascularity at SCJ (kappa value, 0.95; SE, 0.08) and good for round pit pattern (kappa value, 0.80). The most beneficial part of this study is the practical use that may be duplicated by others[28-29]. The criteria used in this study were much simpler since there was no need for magnification system. However, the limitation was the nature of preliminary study, the reproducibility of the results conducted from multi-centers and good interobserver agreement were required before applying this technique into daily practice.
Usefulness of FICE for detecting MERD
FICE is another evolution in digital chromoendoscopy which has emerged for the evaluation of MERD[31]. This technique combines a high-resolution endoscopy with adjustable wavelength to improve visualization of subtle lesions of GERD. We recently conducted a study in 21 patients with typical symptoms of heartburn and/or acid regurgitation compared with 9 controls. The endoscopy was performed under the conventional white light endoscopy and the FICE system that included 2 stations; station 0 and 4 which representing the RGB wavelength of 540, 415 and 415 nm and of 560, 500 and 475 nm respectively. Patients with esophageal erosions diagnosed by the standard white light endoscopy were excluded from the study. Gold standard for the diagnosis was based on typical symptoms of reflux. The “triangular lesion”, defined as a triangular indentation into the squamous mucosa extended from the villiform columnar at the Z-line, was used as the endoscopic diagnostic criterion (Figure 2). The preliminary data showed that FICE provided higher sensitivity, NPV and accuracy than the standard white light endoscopy. Sensitivity, specificity, NPV, PPV and accuracy of each station are shown in Table 1[31]. This technique appears to provide similar results comparable to the NBI system[31]. However, the values of interobserver agreement are still suboptimal and need to be improved. It is speculated that the optimal wavelength has not been standardized yet and more learning curves may be also required. Moreover, further validation of the results in a larger population is also necessary.
Figure 2.
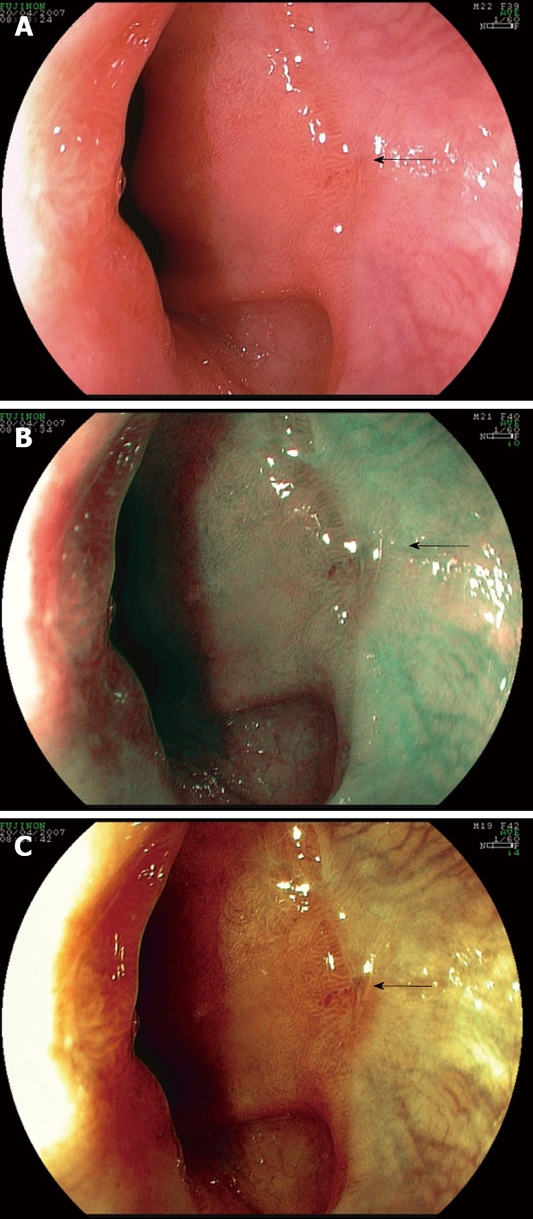
Triangular lesion (arrow). A: Conventional white light endoscopy; B: FICE station 0 (RGB wavelength of 540, 415 and 415 nm); C: FICE station 4 (RGB wavelength of 560, 500 and 475 nm).
Table 1.
Comparison of the outcomes from various types of standard and digital chromoendoscopy for minimal change reflux disease diagnosis
| Endoscopy with magnification[21] | NBI with magnification[29] | NBI without magnification[30] | FICE without magnification[31] | |
| Sensitivity | 64 | 55 | 86.6 | 77.8 |
| Specificity | 85 | 87 | 83.3 | 83.3 |
| NPV | 70 | NA | 83.3 | 55.6 |
| PPV | 80 | NA | 86.1 | 93.3 |
| Accuracy | 74 | NA | NA | 79.2 |
| Criteria for the diagnosis | Punctuate erythema | Increased IPCLs | Increased vascularity with absence of round pit patterns | Triangular lesions |
| Interobserver1 agreement | NA | Fair | Good | Poor |
| Intraobserver1 agreement | NA | Fair | NA | NA |
NA: Not available;
Gold standards for GERD diagnosis were different in each study.
In addition, the magnification system can be added as a combination to FICE. This may help to enhance the details of vascular pattern of esophageal mucosal of MERD. The increased numbers of tortuous and dilated IPCLs can be visualized by magnifying FICE similar to magnifying NBI (Figure 3). However, the same limitations of using magnifying FICE in practice are similar to magnifying NBI that was mentioned earlier.
Figure 3.
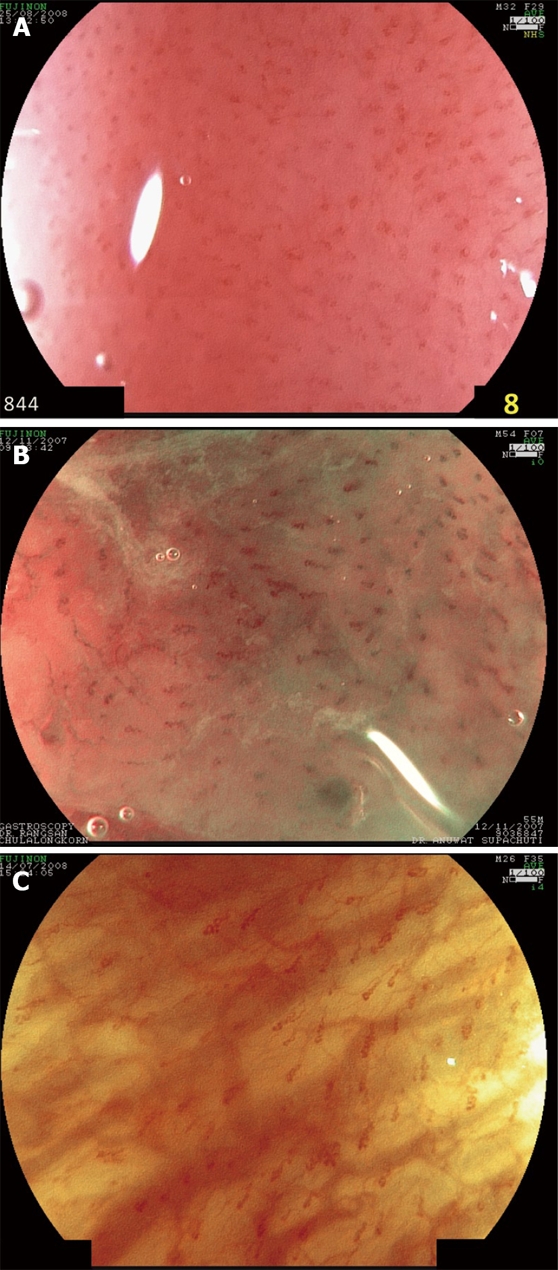
Images from magnifying FICE of × 100 show increased number and dilated and tortuous of intrapapillary capillary loops (IPCL). A: Conventional white light endoscopy; B: FICE station 0 (RGB wavelength of 540, 415 and 415 nm); C: FICE station 4 (RGB wavelength of 560, 500 and 475 nm).
At the moment, FICE system may be useful for targeted biopsy in reflux patients with negative study on conventional endoscopy.
Feasibility of i-SCAN for the detection of MERD
High-definition endoscopy with i-SCAN, a post-processing digital filter which enhances the detailed mucosal surface and vasculatures, has been recently evaluated for detection of mucosal breaks in reflux disease[32]. The distal esophagus of 50 patients with reflux symptoms were inspected by a high-definition (HD) endoscopy, with i-SCAN and Lugol’s staining method in stepwise manner. The examination with i-SCAN was initially performed under surface enhancement (SE) mode and clearly identified the mucosal changes. Subsequently it was performed under pattern mode (P-mode) and vascular mode (V-mode) which were able to characterize the details of the lesions. Mucosal breaks were detected in 9 of 50 patients with HD endoscopy. The detection rate was slightly increased to 12 of 50 patients with i-SCAN and significantly increased to 25 of 50 patients with Lugol chromoendoscopy (Figure 4). The degree of esophagitis was upgraded in 5 and 14 patients by using i-SCAN and chromoendoscopy respectively whereas one patient with esophagitis LA grade B diagnosed by i-SCAN was downgraded to LA grade A by Lugol chromoendoscopy. In addition, when compared to standard HD endoscopy, both enhancing methods demonstrated more significant numbers of small circumscribed lesions (58 lesions diagnosed by i-SCAN, 85 lesions diagnosed by chromoendoscopy vs only 21 lesions diagnosed by HD endoscopy). Compared to the previous studies, the criteria used for MERD diagnosis in this study were different from those used in NBI and FICE studies that were mentioned earlier. We can assume that i-SCAN can improve detection rates of minute mucosal breaks in smaller numbers of patients than HD chromoendoscopy. However, more studies using the combination of i-SCAN and magnification system is required to demonstrate the practical benefit.
Figure 4.
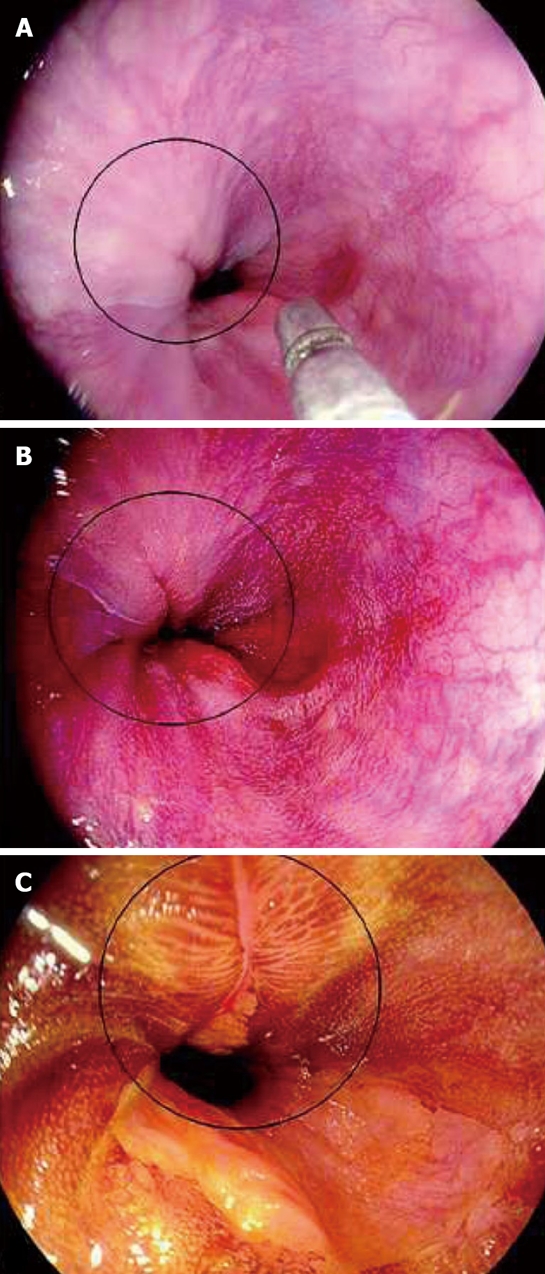
Images from i-SCAN. A: HD+: A mucosal break is not clearly visible; B: i-SCAN can better depict the mucosal break; C: Lugol’s stain reveals a larger extent of the mucosal break which can help upgrade the degree of esophagitis according to Los Angeles classification. (with permission from Georg Thieme Verlag KG).
CONCLUSION
Digital chromoendoscopy is an important tool with a high potential for GERD diagnosis particularly for MERD since it provides the endoscopist with a simple, safe and rapid method for a better detection of the subtle esophageal lesions. In our speculation, over the next few years it may become a standard tool for GERD and MERD diagnosis. However, large, multi-center and randomized controlled studies comparing these new imaging modalities with the conventional white light chromoendoscopy are warranted to validate its accuracy. Standard, simple and precise endoscopic reading criteria for the identification of minor mucosal changes in reflux disease are required. Lastly, intra- and interobserver agreements have to be improved before utilizing these innovations in clinical practice.
Footnotes
Peer reviewer: Rajvinder Singh, MBBS, MRCP, Senior Medical Practitioner/Gastroenterologist, The Lyell McEwin Hospital, SA 5081, Australia
S- Editor Zhang HN L- Editor Roemmele A E- Editor Liu N
References
- 1.El-Serag HB. Time trends of gastroesophageal reflux disease: a systematic review. Clin Gastroenterol Hepatol. 2007;5:17–26. doi: 10.1016/j.cgh.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 2.Dent J, El-Serag HB, Wallander MA, Johansson S. Epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut. 2005;54:710–717. doi: 10.1136/gut.2004.051821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ho KY, Chan YH, Kang JY. Increasing trend of reflux esophagitis and decreasing trend of Helicobacter pylori infection in patients from a multiethnic Asian country. Am J Gastroenterol. 2005;100:1923–1928. doi: 10.1111/j.1572-0241.2005.50138.x. [DOI] [PubMed] [Google Scholar]
- 4.Richter JE. Diagnostic tests for gastroesophageal reflux disease. Am J Med Sci. 2003;326:300–308. doi: 10.1097/00000441-200311000-00006. [DOI] [PubMed] [Google Scholar]
- 5.Dent J, Brun J, Fendrick AM, Fennerty MB, Janssens J, Kahrilas PJ, Lauritsen K, Reynolds JC, Shaw M, Talley NJ. An evidence-based appraisal of reflux disease management--the Genval Workshop Report. Gut. 1999;44 Suppl 2:S1–S16. doi: 10.1136/gut.44.2008.s1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rosaida MS, Goh KL. Gastro-oesophageal reflux disease, reflux oesophagitis and non-erosive reflux disease in a multiracial Asian population: a prospective, endoscopy based study. Eur J Gastroenterol Hepatol. 2004;16:495–501. doi: 10.1097/00042737-200405000-00010. [DOI] [PubMed] [Google Scholar]
- 7.Labenz J, Jaspersen D, Kulig M, Leodolter A, Lind T, Meyer-Sabellek W, Stolte M, Vieth M, Willich S, Malfertheiner P. Risk factors for erosive esophagitis: a multivariate analysis based on the ProGERD study initiative. Am J Gastroenterol. 2004;99:1652–1656. doi: 10.1111/j.1572-0241.2004.30390.x. [DOI] [PubMed] [Google Scholar]
- 8.Ang TL, Fock KM, Ng TM, Teo EK, Chua TS, Tan J. A comparison of the clinical, demographic and psychiatric profiles among patients with erosive and non-erosive reflux disease in a multi-ethnic Asian country. World J Gastroenterol. 2005;11:3558–3561. doi: 10.3748/wjg.v11.i23.3558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nakamura T, Shirakawa K, Masuyama H, Sugaya H, Hiraishi H, Terano A. Minimal change oesophagitis: a disease with characteristic differences to erosive oesophagitis. Aliment Pharmacol Ther. 2005;21 Suppl 2:19–26. doi: 10.1111/j.1365-2036.2005.02469.x. [DOI] [PubMed] [Google Scholar]
- 10.Armstrong D, Bennett JR, Blum AL, Dent J, De Dombal FT, Galmiche JP, Lundell L, Margulies M, Richter JE, Spechler SJ, et al. The endoscopic assessment of esophagitis: a progress report on observer agreement. Gastroenterology. 1996;111:85–92. doi: 10.1053/gast.1996.v111.pm8698230. [DOI] [PubMed] [Google Scholar]
- 11.Sharma P, Bansal A, Mathur S, Wani S, Cherian R, McGregor D, Higbee A, Hall S, Weston A. The utility of a novel narrow band imaging endoscopy system in patients with Barrett’s esophagus. Gastrointest Endosc. 2006;64:167–175. doi: 10.1016/j.gie.2005.10.044. [DOI] [PubMed] [Google Scholar]
- 12.Imraporn B, Jutaghokiat S, Wisedopas N, Rerknimitr R, Treeprasertsuk S, Mahachai V, Kullavanijaya P. Validity of magnifying NBI for gastric intestinal metaplasia targeted biopsy. Gastrointest Endosc. 2008;67:AB280. [Google Scholar]
- 13.Osawa H, Yoshizawa M, Yamamoto H, Kita H, Satoh K, Ohnishi H, Nakano H, Wada M, Arashiro M, Tsukui M, et al. Optimal band imaging system can facilitate detection of changes in depressed-type early gastric cancer. Gastrointest Endosc. 2008;67:226–234. doi: 10.1016/j.gie.2007.06.067. [DOI] [PubMed] [Google Scholar]
- 14.Rastogi A, Bansal A, Wani S, Callahan P, McGregor DH, Cherian R, Sharma P. Narrow-band imaging colonoscopy--a pilot feasibility study for the detection of polyps and correlation of surface patterns with polyp histologic diagnosis. Gastrointest Endosc. 2008;67:280–286. doi: 10.1016/j.gie.2007.07.036. [DOI] [PubMed] [Google Scholar]
- 15.Nothmann BJ, Wright JR, Schuster MM. In vivo vital staining as an aid to identification of esophagogastric mucosal junction in man. Am J Dig Dis. 1972;17:919–924. doi: 10.1007/BF02239531. [DOI] [PubMed] [Google Scholar]
- 16.Misumi A, Kondou H, Murakami A, Arima K, Honmyou U, Baba K, Akagi M. Endoscopic diagnosis of reflux esophagitis by the dye-spraying method. Endoscopy. 1989;21:1–6. doi: 10.1055/s-2007-1012883. [DOI] [PubMed] [Google Scholar]
- 17.Yoshikawa I, Yamasaki M, Yamasaki T, Kume K, Otsuki M. Lugol chromoendoscopy as a diagnostic tool in so-called endoscopy-negative GERD. Gastrointest Endosc. 2005;62:698–703; quiz 752, 754. doi: 10.1016/j.gie.2005.06.027. [DOI] [PubMed] [Google Scholar]
- 18.Kondo H, Fukuda H, Ono H, Gotoda T, Saito D, Takahiro K, Shirao K, Yamaguchi H, Yoshida S. Sodium thiosulphate solution spray for relief of irritation caused by Lugol’s stain in chromoendoscopy. Gastrointest Endosc. 2001;53:199–202. doi: 10.1067/mge.2001.110730. [DOI] [PubMed] [Google Scholar]
- 19.Myung Park J, Seok Lee I, Young Kang J, Nyol Paik C, Kyung Cho Y, Woo Kim S, Choi MG, Chung IS. Acute esophageal and gastric injury: complication of Lugol’s solution. Scand J Gastroenterol. 2007;42:135–137. doi: 10.1080/00365520600825141. [DOI] [PubMed] [Google Scholar]
- 20.Stevens PD, Lightdale CJ, Green PH, Siegel LM, Garcia-Carrasquillo RJ, Rotterdam H. Combined magnification endoscopy with chromoendoscopy for the evaluation of Barrett’s esophagus. Gastrointest Endosc. 1994;40:747–749. [PubMed] [Google Scholar]
- 21.Kiesslich R, Kanzler S, Vieth M, Moehler M, Neidig J, Thanka Nadar BJ, Schilling D, Burg J, Nafe B, Neurath MF, et al. Minimal change esophagitis: prospective comparison of endoscopic and histological markers between patients with non-erosive reflux disease and normal controls using magnifying endoscopy. Dig Dis. 2004;22:221–227. doi: 10.1159/000080323. [DOI] [PubMed] [Google Scholar]
- 22.Tam W, Dent J, Conroy-Hiller TA, Schoeman MN. Proposed new endoscopic criteria for minimal change reflux esophagitis based on magnification endoscopy. Gastrointest Endosc. 2001;53:AB119. [Google Scholar]
- 23.Edebo A, Tam W, Bruno M, Van Berkel AM, Jönson C, Schoeman M, Tytgat G, Dent J, Lundell L. Magnification endoscopy for diagnosis of nonerosive reflux disease: a proposal of diagnostic criteria and critical analysis of observer variability. Endoscopy. 2007;39:195–201. doi: 10.1055/s-2006-945112. [DOI] [PubMed] [Google Scholar]
- 24.Kuznetsov K, Lambert R, Rey JF. Narrow-band imaging: potential and limitations. Endoscopy. 2006;38:76–81. doi: 10.1055/s-2005-921114. [DOI] [PubMed] [Google Scholar]
- 25.Kawahara Y, Okada H, Kawano S, Inoue M, Tsuzuki T, Tanioka D, Hori K, Yamamoto K. New digital imaging technology I-Scan for upper GI endoscopy: a pilot study. Gastrointest Endosc. 2009;69:AB377. [Google Scholar]
- 26.Kumagai Y, Inoue H, Nagai K, Kawano T, Iwai T. Magnifying endoscopy, stereoscopic microscopy, and the microvascular architecture of superficial esophageal carcinoma. Endoscopy. 2002;34:369–375. doi: 10.1055/s-2002-25285. [DOI] [PubMed] [Google Scholar]
- 27.Kumagai Y, Toi M, Inoue H. Dynamism of tumour vasculature in the early phase of cancer progression: outcomes from oesophageal cancer research. Lancet Oncol. 2002;3:604–610. doi: 10.1016/s1470-2045(02)00874-4. [DOI] [PubMed] [Google Scholar]
- 28.Lee YC, Lin JT, Chiu HM, Liao WC, Chen CC, Tu CH, Tai CM, Chiang TH, Chiu YH, Wu MS, et al. Intraobserver and interobserver consistency for grading esophagitis with narrow-band imaging. Gastrointest Endosc. 2007;66:230–236. doi: 10.1016/j.gie.2006.10.056. [DOI] [PubMed] [Google Scholar]
- 29.Sharma P, Wani S, Bansal A, Hall S, Puli S, Mathur S, Rastogi A. A feasibility trial of narrow band imaging endoscopy in patients with gastroesophageal reflux disease. Gastroenterology. 2007;133:454–464; quiz 674. doi: 10.1053/j.gastro.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 30.Fock KM, Teo EK, Ang TL, Tan JY, Law NM. The utility of narrow band imaging in improving the endoscopic diagnosis of gastroesophageal reflux disease. Clin Gastroenterol Hepatol. 2009;7:54–59. doi: 10.1016/j.cgh.2008.08.030. [DOI] [PubMed] [Google Scholar]
- 31.Chaiteerakij R, Geratikornsupuk N, Tangmankongworakoon N, Gonlachanvit S, Treeprasertsuk S, Rerknimitr R, Kullavanijaya P. Efficacy of intelligent chromo endoscopy for detection of minimal mucosal breaks in patients with typical reflux symptoms of gastroesophageal reflux disease. Gastrointest Endosc. 2008;67:AB86. [Google Scholar]
- 32.Hoffman A, Basting N, Goetz M, Tresch A, Mudter J, Biesterfeld S, Galle PR, Neurath MF, Kiesslich R. High-definition endoscopy with i-Scan and Lugol’s solution for more precise detection of mucosal breaks in patients with reflux symptoms. Endoscopy. 2009;41:107–112. doi: 10.1055/s-0028-1119469. [DOI] [PubMed] [Google Scholar]


