Abstract
Oxidative stress is a redox imbalance between pro-oxidants and antioxidants in favour of the former ones, leading to different responses depending on the level of pro-oxidants and the duration of the exposure. In this article, we discuss the damaging or cytoprotective signaling mechanisms associated with oxidative stress by addressing (1) the role of prolonged and severe oxidative stress and insulin resistance as determinant factors in the pathogenesis of non-alcoholic fatty liver disease associated with obesity, which, with the concurrence of nutritional factors, may determine the onset of fatty liver and its progression to steatohepatitis; and (2) the development of an acute and mild pro-oxidant state by thyroid hormone administration, which elicits the redox up-regulation of the expression of proteins affording cell protection, as a preconditioning strategy against ischemia-reperfusion liver injury.
Keywords: Oxidative stress, Obesity, Insulin resistance, Non-alcoholic fatty liver disease, Thyroid hormone, Liver preconditioning
INTRODUCTION
Oxidative stress is a redox disequilibrium in which the pro-oxidant/antioxidant balance is shifted in favour of the pro-oxidants[1], a phenomenon related to the aerobic nature of cellular metabolism, in which O2 reduction is a major event. The latter proceeds through electron transfer reactions due to the electronic structure of O2 in the ground state, with generation of reactive oxygen species (ROS), including (1) primary oxidants [superoxide radical (O2•−), hydrogen peroxide (H2O2), and hydroxyl radical (HO•)]; and (2) secondary oxidants [hydroperoxides or alkoxy and peroxy radicals of biomolecules, in addition to electronically excited states derived from free-radical reactions (singlet oxygen, triplet carbonyls)][2]. The detoxication of ROS is a major prerequisite of aerobic life[1], which is accomplished via several enzymatic and non-enzymatic antioxidant mechanisms that are available in different cell compartments[1,3]. Secondary mechanisms, restoring used cofactors and repairing altered biomolecules, are also required, in addition to those triggering the expression of proteins damaged by ROS or needed to attain cell survival[1,3,4]. These mechanisms need to be coupled to the intermediary metabolism for ATP, NADPH, and precursors supply, and depend on the dietary replenishment of essential components to maintain pro-oxidant reactions and cellular damage at a minimum level under basal conditions.
At the cellular level, oxidative stress leads to a wide spectrum of responses, depending on the cell type, the level of ROS achieved, and the duration of the exposure[4-6]. The moderate increase in ROS and reactive nitrogen species (RNS) in a defined time window can elicit an imbalance capable of redox regulation, as found for L-3,3',5-triiodothyronine (T3)-induced oxidative stress[7], involving important signals regulating either protein function, via reversible oxidation or nitrosation of protein sulfhydryls, and/or gene expression, through modulation of specific kinases, phosphatases, and redox-sensitive transcription factors[4-6]. However, in the case of organs subjected to ischemia-reperfusion (IR)[8] or in obesity[9] and other chronic states, large levels of ROS are attained, which may induce severe oxidation of biomolecules and dysregulation of signal transduction and gene expression, leading to cell death through necrotic and/or apoptotic mechanisms[4].
In this review article, the damaging or cytoprotective signaling mechanisms associated with oxidative stress are addressed. In particular, I will discuss (1) the role of oxidative stress and insulin resistance as contributing factors in the pathogenesis of non-alcoholic fatty liver disease (NAFLD) in obese patients, which, with the concurrence of nutritional factors, may determine the onset of fatty liver and its progression to steatohepatitis; and (2) the implications of the redox regulation of T3-induced gene expression as a preconditioning mechanism against IR liver injury.
OXIDATIVE STRESS SIGNALING UNDERLYING OBESITY-ASSOCIATED NAFLD
The onset of oxidative stress, insulin resistance, and steatosis in obese NAFLD patients
NAFLD is a rapidly growing entity that is becoming a major cause of chronic liver disease, due to the increasing incidence of obesity and type 2 diabetes in the general population. NAFLD includes simple triacylglycerol (TAG) accumulation in hepatocytes (hepatic steatosis) or steatosis with inflammation, fibrosis, and cirrhosis (non-alcoholic steatohepatitis, NASH), with oxidative stress, insulin resistance, and nutritional factors playing major contributing roles[10,11].
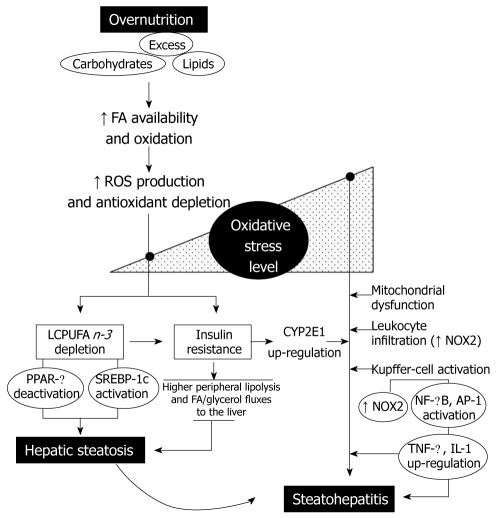
Under most circumstances, fatty acids (FA) are the major oxidative fuel in the liver. However, carbohydrate and lipid affluence induce significant changes in hepatic intermediary metabolism. In fact, high glucose and insulin levels stimulate FA synthesis from glucose and inhibit FA β-oxidation, re-directing FA towards the formation of TAG[9]. Considering that the amount of TAG exported as VLDL depends on synthesis of the protein components, FA in excess are likely to be converted to TAG and stored as lipid droplets within hepatocytes, upon consumption of calorie-enriched diets. Since non-adipose tissues have limited capacity for TAG storage, the lipids in excess that accumulate under conditions of overnutrition determine high intracellular levels of saturated FA, which can induce cell dysfunction and/or cell death, a phenomenon known as lipotoxicity[12]. Consequently, higher rates of FA oxidation and ROS generation are achieved, which might explain the increase in the oxidative stress-related parameters and antioxidant depletion found in the liver of obese patients with NAFLD (Figure 1)[9,13]. Furthermore, prolonged oxidative stress may favour: (1) liver n-3 LCPUFA depletion, which may be compounded by dietary imbalance and defective desaturation activity[14,15]; and (2) insulin resistance, in association with the redox activation of multiple stress-sensitive serine/threonine kinases that alters insulin signaling (Figure 1)[16]. The latter phenomenon is a membrane-mediated process that might be also compromised by n-3 LCPUFA depletion, due to loss of membrane polyunsaturation. Both IR and liver n-3 LCPUFA depletion can determine hepatic steatosis by different mechanisms, namely, (1) insulin resistance-dependent higher peripheral mobilization of FA and glycerol to the liver; and (2) n-3 LCPUFA depletion-induced changes in the DNA-binding activity of the peroxisome proliferator-activated receptor-α (PPAR-α) as well as of the sterol regulatory element binding protein-1c (SREBP-1c), determining a metabolic imbalance between FA oxidation and lipogenesis in favour of the latter (Figure 1). This notion is based on the findings that n-3 LCPUFA are signaling biomolecules regulating hepatic lipid metabolism through (1) down-regulation of the expression of SREBP-1c and its processing, with inhibition of the transcription of lipogenic and glycolytic genes; and (2) up-regulation of the expression of genes encoding enzymes of the oxidation of FA, which act as ligands of PPAR-α[17].
Figure 1.
Interrelationships between the level of oxidative stress and insulin resistance, leading to hepatic steatosis and its progression to steatohepatitis, associated with overnutrition. AP-1: Activating protein 1; CYP2E1: Ethanol inducible form of cytochrome P450; FA: Fatty acids; IL-1: Interleukin-1; LCPUFA; Long-chain polyunsaturated fatty acids; NF-κB: Nuclear factor-κB; NOX2: NADPH oxidase in phagocytic cells; PPAR-α: Peroxisome proliferator-activated receptor-α; ROS: Reactive oxygen species; SREBP-1c: Sterol regulatory element binding protein-1c; TNF-α: Tumor necrosis factor-α.
Exacerbation of hepatic oxidative stress and progression from steatosis to steatohepatitis
Changes in liver oxidative stress-related parameters observed in obese patients with steatosis persist in those with steatohepatitis[9,13]. In steatohepatitis, these features are observed concomitantly with (1) low catalase activity[13]; (2) high immunohistochemical reactivity to 8-hydroxydeoxyguanosine and 4-hydroxy-2-nonenal, as markers of oxidative DNA damage and lipid peroxidation, respectively[18]; (3) a further increment of both 3-nitrotyrosine immunoreactivity and production of O2•− and malondialdehyde by Kupffer cells; (4) induction of inducible nitric oxide synthase; and (5) up-regulation of cytochrome P450 2E1 (CYP2E1), as shown by the higher CYP2E1 protein expression and in vivo chlorzoxazone hydroxylation, an indicator of CYP2E1 activity (Figure 1) (for specific references see[9]).
The exacerbation of the oxidative stress status of the liver in cases of steatohepatitis, compared to livers with steatosis alone, seems to involve several mechanisms (Figure 1). First, induction of liver CYP2E1[13,19] is of particular importance in the pathogenesis of NASH, due to its poor coupling with NADPH-cytochrome P450 reductase, with substantial NADPH oxidase activity, leading to O2•−, H2O2, and consequent lipid peroxidation[20]. Second, hepatic mitochondrial dysfunction is an alternate contributing factor to the genesis of lesions in steatohepatitis, considering the lower levels of mitochondrial DNA and the decreased expression of mitochondrial DNA-encoded proteins, which might lead to reduced activity of respiratory complexes I, III, and IV, and ATP synthase complex V, thus increasing O2•− and H2O2 generation[10]. Third, mixed inflammatory-cell infiltration is a characteristic feature of NASH, including mononuclear cells, polymorphonuclear cells, or both[10], which may represent an additional mechanism of ROS generation, due to the expression and activation of NADPH oxidase (NOX2), an enzyme that produces large amounts of O2•− and H2O2[21]. NOX2 is also expressed in Kupffer cells, which, in patients with steatohepatitis, produce O2•− at rates that are 20-fold higher than normal, in agreement with the 7-fold increase in malondialdehyde levels in Kupffer cells from steatohepatitis patients[22]. Under these conditions, the oxidative stress status of the liver achieved in steatohepatitis might promote hepatocellular damage by inducing (1) severe oxidative alteration of biomolecules, with loss of their functions and impairment of cell viability; and (2) sustained activation of redox-sensitive transcription factors, such as NF-κB and AP-1, with consequent up-regulation of the expression of pro-inflammatory mediators at the Kupffer cell level (Figure 1)[23].
Collectively, the discussed evidence supports the view of a functional interdependence between oxidative stress and insulin resistance (Figure 1). This may involve (1) initial ROS production due to lipotoxicity, related to the onset of insulin resistance in steatosis; and (2) a further increase in ROS generation due to CYP2E1 induction, mitochondrial dysfunction, and Kupffer cell or infiltrating leukocyte NOX2 activity, characterizing steatohepatitis. Dysregulation of pro-inflammatory cytokine, adipokine, and chemokine signaling in NAFLD may reinforce the initial mechanisms of ROS production and IR, representing key factors in the progression from steatosis to steatohepatitis, in the setting of oxidative stress-mediated hepatocyte sensitization[11,24]. In this context, antioxidants can act as insulin sensitizers by lowering ROS levels, a condition that might abrogate free-radical-mediated activation of signaling serine/threonine kinases and damage to biomolecules, as shown in cell-culture studies[25]. However, these findings remain to be confirmed in obese patients with NAFLD.
OXIDATIVE STRESS SIGNALING UNDERLYING THYROID HORMONE LIVER PRECONDITIONING
Mechanisms in thyroid hormone calorigenesis and liver oxidative stress
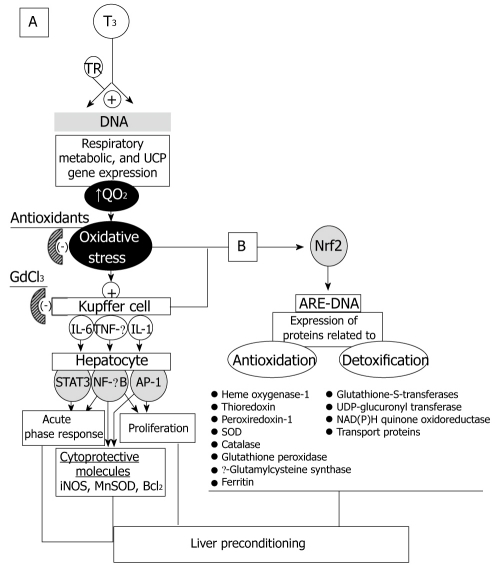
Thyroid hormones play important roles in cell growth, differentiation, and metabolism, through different and complex mechanisms of action. In mammals, major effects are exerted on cellular oxygen consumption (QO2) and metabolic rate, leading to stimulation and maintenance of basal thermogenesis[26,27]. This action of T3 is carried out via thyroid hormone receptors expressed in almost all tissues. These receptors are recognized by specific thyroid hormone response elements across the DNA, leading to ligand-dependent upregulation of the expression of respiratory, metabolic, and uncoupling protein genes (Figure 2A)[28]. In addition to the above classical genomic model of T3-dependent calorigenesis, non-genomic mechanisms may also contribute to increase cellular QO2[29], with the consequent increase of the mitochondrial capacity for oxidative phosphorylation and ROS generation[30].
Figure 2.
Oxidative stress signaling in thyroid hormone (T3) liver preconditioning as mediated by redox-sensitive transcriptional factors NF-κB, AP-1, and STAT3 (A) or Nrf2 (B). AP-1: Activating protein 1; ARE: Antioxidant responsive element; GdCl3: Gadolinium chloride; IL: Interleukin; iNOS: Inducible nitric oxide synthase; MnSOD: Manganese superoxide dismutase; NF-κB: Nuclear factor-κB; Nrf2: Nuclear factor-erythroid 2-related factor 2; QO2: Rate of oxygen consumption; TNF-α: Tumor necrosis factor-α; STAT3: Signal transducer and activator of transcription 3; TR: Thyroid hormone receptor; UCP: Uncoupling protein.
In addition to T3-induced liver mitochondrial capacity for ROS production, the induction of other enzymatic mechanisms also occurs, namely, (1) higher activity of microsomal NADPH-cytochrome P450 reductase[31] and NADPH oxidase[32], the latter representing the oxidase activity of cytochrome P450 responsible for the O2•− and H2O2 production related to the T3-mediated induction of the highly pro-oxidant cytochrome P4502E1 isoform[33]; (2) enhancement of cytosolic enzymatic mechanisms, such as the O2•−/H2O2 generator xanthine oxidase[34] and ROS production, possibly coupled to enhanced FA β-oxidation due to liver peroxisomal proliferation[30]; and (3) Kupffer-cell activation with increased respiratory burst activity, due to NADPH oxidase[35].
T3-induced liver free-radical activity is associated with depletion of antioxidant defences, leading to increased oxidative stress of the liver (Figure 2A)[7,28,36]. However, this pro-oxidant state achieved in the liver by T3-induced calorigenesis can be considered as a mild redox alteration, as suggested by the lack of occurrence of morphological changes in liver parenchyma, except for the significant hyperplasia and hypertrophy of Kupffer cells[35], the resident macrophages of the liver[37]. The latter effect of T3 might be of importance considering that Kupffer cells play a central role in the homeostatic response to liver injury, through the production and release of a wide array of mediators that provide physiologically diverse and key paracrine effects on all other liver cells[37,38].
T3-induced Kupffer cell-dependent up-regulation of cytokine expression and hepatocyte proteins related to antioxidation, anti-apoptosis, acute-phase response, and cell proliferation
Kupffer cell hyperplasia is a major finding after in vivo T3 administration, an effect that may involve the expansion of Kupffer cell precursors by means of circulating monocyte recruitment, the differentiation of pre-existing local Kupffer cell precursors into mature liver macrophages, or both[39]. Under these conditions, assessment of Kupffer cell function revealed a significant increase in the rate of carbon phagocytosis and the associated carbon-induced O2 uptake, representing the respiratory burst activity of Kupffer cells, a process that is largely dependent on the activity of the ROS-generator NADPH oxidase and abolished by pretreatment with the Kupffer cell inactivator gadolinium chloride (GdCl3) (Figure 2A)[35].
The interdependence between T3-induced calorigenesis, liver QO2, and ROS production is associated with a significant increase in the hepatic DNA binding of the transcription factors NF-κB[40], STAT3[41], and AP-1[42] (Figure 2A). Activation of these transcription factors by T3 administration is suppressed by in vivo pretreatment with GdCl3, whereas NF-κB and STAT3 activation by T3 is also abolished by pretreatment with antioxidants[40,41], thus supporting the view that T3 induces the redox activation of hepatic NF-κB, STAT3, and AP-1 by actions primarily exerted at the Kupffer cell level (Figure 2A). T3 administration involving significant NF-κB and AP-1 activation induced mRNA expression of the NF-κB/AP-1-responsive genes for TNF-α, with increased serum levels of the cytokine[40] that are abolished by pretreatment with the antisense oligonucleotide TJU-2755, targeting the primary RNA transcript of TNF-α[43]. T3 also elicited an increase in the serum levels of IL-6[41] and in the hepatic mRNA expression and serum levels of IL-1[40]. In addition to NF-κB and AP-1 activation, the enhancement in STAT3 DNA binding by T3 administration[41] may be associated with the proliferation of macrophage precursors and their differentiation into Kupffer cells[39], considering the central role of STAT3 in gp130-mediated cell growth, differentiation, and survival[44].
The effects of cytokines released from Kupffer cells are exerted through their interaction with specific surface receptors of liver target cells, mediating the signaling transduction from the cell membrane to the nucleus[37]. In agreement with the above view, the transient TNF-α response elicited by T3 administration correlates with the substantial increase in liver IκB-α phosphorylation[45,46], leading to the activation of the IKK complex that in turn activates NF-κB, after coupling with the TNF-α receptor and associating with different adaptor proteins[47]. T3-induced TNF-α response, liver IKK phosphorylation, and NF-κB activation are abolished by pretreatment with either α-tocopherol or GdCl3, supporting the role of ROS production and Kupffer-cell activation in T3-dependent signaling leading to up-regulation of hepatic gene expression[45,46]. This is shown by the increased expression of the NF-κB-responsive genes encoding for inducible NOS (iNOS)[45], manganese superoxide dismutase (MnSOD), and the anti-apoptotic protein Bcl-2[46] (Figure 2A). Thus, T3 administration elicits the redox up-regulation of iNOS, MnSOD, and Bcl-2 in the liver, in association with the Kupffer cell-dependent release of TNF-α and activation of the IKK/NF-κB cascade, representing antioxidant and anti-apoptotic responses triggered by the underlying oxidative stress (Figure 2A).
In addition to the above responses, T3 administration up-regulate the acute-phase response (APR) of the liver and the hepatocyte proliferation. The APR is a major pathophysiologic reaction in which normal homeostatic mechanisms are replaced by new set-points, contributing to defensive or adaptive capabilities against inflammation and oxidative stress[48,49]. In fact, T3 induced the Kupffer-cell-dependent release of IL-6 and activation of hepatic STAT3 controlling both type I (haptoglobin) and type II (β-fibrinogen) acute-phase protein (APP) genes[41]. In addition, this response may be contributed by the T3-induced TNF-α/IKK/NF-κB pathway[45,46], which controls type I APP genes, considering that NF-κB activation can synergistically enhance the effects of STAT3 and C/EBPβ upon C-reactive protein induction[50]. Furthermore, the in vivo effects of T3 as a primary hepatic mitogen, leading to hepatocyte proliferation in intact liver, are well established (Figure 2A)[42]. This process involves a large number of genes and requires the concurrence of cytokines, growth factors and metabolic networks[51]. Resting hepatocytes, i.e. in the G0 phase of the cell cycle, need to be primed by TNF-α and IL-6 before they can respond to growth factors, with the concomitant activation of NF-κB, STAT3, AP-1, and E/EBPβ, enter the G1 phase and initiate cell cycle progression. T3 administration has been associated with increased liver cyclin-dependent kinase 2 expression and hepatocyte proliferation, as shown by the increase of Ki-67, a nuclear cell proliferation-associated protein expressed in all active parts of the cell cycle, and of the proliferating cell nuclear antigen (PCNA)[42].
Collectively, data reported by our group indicate that T3 triggers cytoprotection in the liver through redox- and Kupffer cell-dependent signaling mechanisms, namely, (1) antioxidant responses (iNOS, MnSOD); (2) anti-apoptosis (Bcl-2); (3) immune, transport, and antioxidant (haptoglobin, ceruloplasmin, ferritin) functions fulfilled by APR induction; and (4) hepatocyte proliferation (Figure 2A), the metabolic demands of which being met by acceleration of energy metabolism due to T3-induced calorigenesis[7,28,52].
Thyroid hormone-induced liver preconditioning
Organ preconditioning, including that involving the liver, consists in strategies protecting the organ from detrimental effects of subsequent noxious events, such as those underlying chemically-induced injury or IR[8,53]. In general terms, IR injury refers to tissue damage produced by blood perfusion to a previously ischemic organ. In the case of the liver, this occurs in the clinical settings of hepatic resection, transplantation, low-blood pressure states, and abdominal surgery requiring hepatic vascular occlusion. IR liver injury assessed in a model involving 1 h of partial ischemia, as induced by vascular clamping, and followed by reperfusion for 20 h, elicited minimal mortality but substantial liver damage, with increased serum transaminase and TNF-α levels as well as metabolic changes, namely, (1) a drastic increase in the oxidative stress status of the liver; (2) loss in the DNA binding of NF-κB and STAT3, implying loss of cytoprotective potential, as shown by the concomitant diminution in the expression of the APR protein haptoglobin, controlled by both these transcription factors; and (3) increase of the hepatic AP-1 DNA binding activity, which may constitute a major determinant of hepatotoxicity under conditions of reduced NF-κB activation and TNF-α response[54,55]. These changes were normalized by T3 treatment given 48 h before the IR protocol, a preconditioning effect that was sensitive to the antioxidant N-acetylcysteine given prior to T3[55], with enhanced hepatocyte proliferation compensating for liver cells lost due to IR-induced hepatocellular necrosis[42].
In conclusion, the data discussed above indicate that redox regulation of gene transcription by T3 involves antioxidant-sensitive NF-κB, AP-1, and STAT3 activation and up-regulation of the expression of cytoprotective proteins affording liver preconditioning (Figure 2A)[52]. T3 liver preconditioning may also involve the activation of the Nrf2-Keap1 defense pathway, up-regulating antioxidant proteins and phase-2 detoxifying enzymes (Figure 2B)[56], which is currently under study in our laboratory.
CONCLUSION
Data analyzed indicate that development of extreme levels of oxidative stress in the liver determines opposite cellular responses, depending on the period of exposure to ROS. Development of a progressive and severe pro-oxidant state in the liver of obese patients with NAFLD is associated with the onset of steatosis and its progression to steatohepatitis, as a chronic model of nutritional oxidative stress. The molecular pathogenesis of NAFLD in obese patients seems to be multifactorial, with oxidative stress and insulin resistance as major pathophysiological mechanisms, which may be interdependent[24]. Considering the lack of an effective drug therapy for NAFLD at present[10], further studies of potentially attractive therapeutic targets are required. These may include the expression and activation status of metabolic (PPAR-α and SREBP-1c) and pro-inflammatory (NF-κB and AP-1) transcription factors, and the activity of enzymes associated with insulin resistance, such as serine/threonine stress kinases and protein tyrosine phosphatases, which will undoubtedly contribute to understand the role of chronic and progressive oxidative stress and insulin resistance in determining steatosis and its progression to NASH. It is now increasingly accepted that bariatric surgery is the most effective method of achieving long-term weight control for patients with morbid obesity[10], with the consequent improvement of the key features of NAFLD and NASH. Thus, weight loss, as a central therapeutic measure, might be combined with antioxidants, in order to minimize or prevent the onset of oxidative stress-induced inflammatory response and insulin resistance, and/or n-3 LCPUFA, to improve the efficiency of signaling cascades related to hepatic lipid metabolism and insulin resistance.
Following acute T3 administration, induction of a mild pro-oxidant state within a time window of 48 h triggers liver preconditioning[54]. This preconditioning strategy has clinical potential, considering that (1) pharmacological and other liver preconditioning maneuvers have not been transferred to clinical applications[53]; and that (2) T3 is an endobiotic substance, and a widely used and well-tolerated therapeutic agent, which, at low doses, has either no significant or minimal adverse effects that can be readily controlled. However, prevention of IR injury in humans during liver surgery and liver transplantation, using reduced-size grafts from living donors, awaits further experimental and clinical studies.
ACKNOWLEDGMENTS
The author wishes to thank Drs. V Fernández, G Tapia, P Varela, P Romanque, P Cornejo, R Rodrigo, J Araya, and J Poniachik for valuable discussion, and whose contributions are cited in this review article.
Footnotes
Supported by Grant 1090020 From FONDECYT, Chile
Peer reviewer: Shivendra Shukla, Professor, Department of Medical Pharmacology and Physiology, University of Missouri School of Medicine, 1 Hospital Drive, M530 Medical Sciences Bldg., Columbia, MO 65212, United States
S- Editor Zhang HN L- Editor Negro F E- Editor Ma WH
References
- 1.Sies H. Biochemistry of oxidative stress. Angew Chem Int Ed Engl. 1986;25:1058–1071. [Google Scholar]
- 2.Videla LA, Fernandez V. Biochemical aspects of cellular oxidative stress. Arch Biol Med Exp (Santiago) 1988;21:85–92. [PubMed] [Google Scholar]
- 3.Fernandez V, Videla LA. Biochemical aspects of cellular antioxidant systems. Biol Res. 1996;29:177–182. [PubMed] [Google Scholar]
- 4.Droge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002;82:47–95. doi: 10.1152/physrev.00018.2001. [DOI] [PubMed] [Google Scholar]
- 5.Martindale JL, Holbrook NJ. Cellular response to oxidative stress: signaling for suicide and survival. J Cell Physiol. 2002;192:1–15. doi: 10.1002/jcp.10119. [DOI] [PubMed] [Google Scholar]
- 6.Poli G, Leonarduzzi G, Biasi F, Chiarpotto E. Oxidative stress and cell signalling. Curr Med Chem. 2004;11:1163–1182. doi: 10.2174/0929867043365323. [DOI] [PubMed] [Google Scholar]
- 7.Varela P, Tapia G, Fernandez V, Videla LA. The role of thyroid hormone calorigenesis in the redox regulation of gene expression. Biol Res. 2006;39:611–617. doi: 10.4067/s0716-97602006000500004. [DOI] [PubMed] [Google Scholar]
- 8.Romanque UP, Uribe M M, Videla LA. [Molecular mechanisms in liver ischemic-reperfusion injury and ischemic preconditioning] Rev Med Chil. 2005;133:469–476. doi: 10.4067/s0034-98872005000400012. [DOI] [PubMed] [Google Scholar]
- 9.Videla LA. Oxidative stress and insulin resistance as interdependent pathogenic mechanisms in non-alcoholic fatty liver disease associated with obesity. In: Alvarez S, Evelson P, editors. Free Radical Pathophysiology. Kerala, India: Transworld Research Network; 2008. pp. 369–385. [Google Scholar]
- 10.Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221–1231. doi: 10.1056/NEJMra011775. [DOI] [PubMed] [Google Scholar]
- 11.Videla LA, Rodrigo R, Araya J, Poniachik J. Oxidative stress and depletion of hepatic long-chain polyunsaturated fatty acids may contribute to nonalcoholic fatty liver disease. Free Radic Biol Med. 2004;37:1499–1507. doi: 10.1016/j.freeradbiomed.2004.06.033. [DOI] [PubMed] [Google Scholar]
- 12.Aronis A, Madar Z, Tirosh O. Mechanism underlying oxidative stress-mediated lipotoxicity: exposure of J774.2 macrophages to triacylglycerols facilitates mitochondrial reactive oxygen species production and cellular necrosis. Free Radic Biol Med. 2005;38:1221–1230. doi: 10.1016/j.freeradbiomed.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 13.Videla LA, Rodrigo R, Orellana M, Fernandez V, Tapia G, Quinones L, Varela N, Contreras J, Lazarte R, Csendes A, et al. Oxidative stress-related parameters in the liver of non-alcoholic fatty liver disease patients. Clin Sci (Lond) 2004;106:261–268. doi: 10.1042/CS20030285. [DOI] [PubMed] [Google Scholar]
- 14.Araya J, Rodrigo R, Videla LA, Thielemann L, Orellana M, Pettinelli P, Poniachik J. Increase in long-chain polyunsaturated fatty acid n - 6/n - 3 ratio in relation to hepatic steatosis in patients with non-alcoholic fatty liver disease. Clin Sci (Lond) 2004;106:635–643. doi: 10.1042/CS20030326. [DOI] [PubMed] [Google Scholar]
- 15.Elizondo A, Araya J, Rodrigo R, Signorini C, Sgherri C, Comporti M, Poniachik J, Videla LA. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol Res. 2008;41:59–68. [PubMed] [Google Scholar]
- 16.Evans JL, Maddux BA, Goldfine ID. The molecular basis for oxidative stress-induced insulin resistance. Antioxid Redox Signal. 2005;7:1040–1052. doi: 10.1089/ars.2005.7.1040. [DOI] [PubMed] [Google Scholar]
- 17.Clarke SD. The multi-dimensional regulation of gene expression by fatty acids: polyunsaturated fats as nutrient sensors. Curr Opin Lipidol. 2004;15:13–18. doi: 10.1097/00041433-200402000-00004. [DOI] [PubMed] [Google Scholar]
- 18.Seki S, Kitada T, Yamada T, Sakaguchi H, Nakatani K, Wakasa K. In situ detection of lipid peroxidation and oxidative DNA damage in non-alcoholic fatty liver diseases. J Hepatol. 2002;37:56–62. doi: 10.1016/s0168-8278(02)00073-9. [DOI] [PubMed] [Google Scholar]
- 19.Orellana M, Rodrigo R, Varela N, Araya J, Poniachik J, Csendes A, Smok G, Videla LA. Relationship between in vivo chlorzoxazone hydroxylation, hepatic cytochrome P450 2E1 content and liver injury in obese non-alcoholic fatty liver disease patients. Hepatol Res. 2006;34:57–63. doi: 10.1016/j.hepres.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 20.Caro AA, Cederbaum AI. Oxidative stress, toxicology, and pharmacology of CYP2E1. Annu Rev Pharmacol Toxicol. 2004;44:27–42. doi: 10.1146/annurev.pharmtox.44.101802.121704. [DOI] [PubMed] [Google Scholar]
- 21.Babior BM. NADPH oxidase. Curr Opin Immunol. 2004;16:42–47. doi: 10.1016/j.coi.2003.12.001. [DOI] [PubMed] [Google Scholar]
- 22.Malaguarnera L, Di Rosa M, Zambito AM, dell’Ombra N, Nicoletti F, Malaguarnera M. Chitotriosidase gene expression in Kupffer cells from patients with non-alcoholic fatty liver disease. Gut. 2006;55:1313–1320. doi: 10.1136/gut.2005.075697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Videla LA, Tapia G, Rodrigo R, Pettinelli P, Haim D, Santibanez C, Araya AV, Smok G, Csendes A, Gutierrez L, et al. Liver NF-kappaB and AP-1 DNA binding in obese patients. Obesity (Silver Spring) 2009;17:973–979. doi: 10.1038/oby.2008.601. [DOI] [PubMed] [Google Scholar]
- 24.Videla LA, Rodrigo R, Araya J, Poniachik J. Insulin resistance and oxidative stress interdependency in non-alcoholic fatty liver disease. Trends Mol Med. 2006;12:555–558. doi: 10.1016/j.molmed.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 25.Houstis N, Rosen ED, Lander ES. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature. 2006;440:944–948. doi: 10.1038/nature04634. [DOI] [PubMed] [Google Scholar]
- 26.Schwartz HL, Oppenheimer JH. Physiologic and biochemical actions of thyroid hormone. Pharmacol Ther B. 1978;3:349–376. doi: 10.1016/s0306-039x(78)80002-6. [DOI] [PubMed] [Google Scholar]
- 27.Yen PM. Physiological and molecular basis of thyroid hormone action. Physiol Rev. 2001;81:1097–1142. doi: 10.1152/physrev.2001.81.3.1097. [DOI] [PubMed] [Google Scholar]
- 28.Videla LA, Fernandez V, Tapia G, Varela P. Thyroid hormone calorigenesis and mitochondrial redox signaling: upregulation of gene expression. Front Biosci. 2007;12:1220–1228. doi: 10.2741/2140. [DOI] [PubMed] [Google Scholar]
- 29.Moreno M, Lombardi A, Beneduce L, Silvestri E, Pinna G, Goglia F, Lanni A. Are the effects of T3 on resting metabolic rate in euthyroid rats entirely caused by T3 itself? Endocrinology. 2002;143:504–510. doi: 10.1210/endo.143.2.8613. [DOI] [PubMed] [Google Scholar]
- 30.Fernandez V, Videla LA. Influence of hyperthyroidism on superoxide radical and hydrogen peroxide production by rat liver submitochondrial particles. Free Radic Res Commun. 1993;18:329–335. doi: 10.3109/10715769309147500. [DOI] [PubMed] [Google Scholar]
- 31.Ram PA, Waxman DJ. Thyroid hormone stimulation of NADPH P450 reductase expression in liver and extrahepatic tissues. Regulation by multiple mechanisms. J Biol Chem. 1992;267:3294–3301. [PubMed] [Google Scholar]
- 32.Fernandez V, Barrientos X, Kipreos K, Valenzuela A, Videla LA. Superoxide radical generation, NADPH oxidase activity, and cytochrome P-450 content of rat liver microsomal fractions in an experimental hyperthyroid state: relation to lipid peroxidation. Endocrinology. 1985;117:496–501. doi: 10.1210/endo-117-2-496. [DOI] [PubMed] [Google Scholar]
- 33.Fernandez V, Massa L, Quinones L, Simon-Giavarotti KA, Giavarotti L, D'Almeida V, Azzalis LA, Junqueira VB, Videla LA. Effects of gamma-hexachlorocyclohexane and L-3,3’,5-triiodothyronine on rat liver cytochrome P4502E1-dependent activity and content in relation to microsomal superoxide radical generation. Biol Res. 2003;36:359–365. doi: 10.4067/s0716-97602003000300007. [DOI] [PubMed] [Google Scholar]
- 34.Huh K, Kwon TH, Kim JS, Park JM. Role of the hepatic xanthine oxidase in thyroid dysfunction: effect of thyroid hormones in oxidative stress in rat liver. Arch Pharm Res. 1998;21:236–240. doi: 10.1007/BF02975281. [DOI] [PubMed] [Google Scholar]
- 35.Tapia G, Pepper I, Smok G, Videla LA. Kupffer cell function in thyroid hormone-induced liver oxidative stress in the rat. Free Radic Res. 1997;26:267–279. doi: 10.3109/10715769709097805. [DOI] [PubMed] [Google Scholar]
- 36.Venditti P, Di Meo S. Thyroid hormone-induced oxidative stress. Cell Mol Life Sci. 2006;63:414–434. doi: 10.1007/s00018-005-5457-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tsukamoto H. Redox regulation of cytokine expression in Kupffer cells. Antioxid Redox Signal. 2002;4:741–748. doi: 10.1089/152308602760598882. [DOI] [PubMed] [Google Scholar]
- 38.Bilzer M, Roggel F, Gerbes AL. Role of Kupffer cells in host defense and liver disease. Liver Int. 2006;26:1175–1186. doi: 10.1111/j.1478-3231.2006.01342.x. [DOI] [PubMed] [Google Scholar]
- 39.Gomes LF, Lorente S, Simon-Giavarotti KA, Areco KN, Araujo-Peres C, Videla LA. Tri-iodothyronine differentially induces Kupffer cell ED1/ED2 subpopulations. Mol Aspects Med. 2004;25:183–190. doi: 10.1016/j.mam.2004.02.018. [DOI] [PubMed] [Google Scholar]
- 40.Tapia G, Fernandez V, Varela P, Cornejo P, Guerrero J, Videla LA. Thyroid hormone-induced oxidative stress triggers nuclear factor-kappaB activation and cytokine gene expression in rat liver. Free Radic Biol Med. 2003;35:257–265. doi: 10.1016/s0891-5849(03)00209-0. [DOI] [PubMed] [Google Scholar]
- 41.Tapia G, Fernandez V, Pino C, Ardiles R, Videla LA. The acute-phase response of the liver in relation to thyroid hormone-induced redox signaling. Free Radic Biol Med. 2006;40:1628–1635. doi: 10.1016/j.freeradbiomed.2005.12.033. [DOI] [PubMed] [Google Scholar]
- 42.Fernandez V, Reyes S, Bravo S, Sepulveda R, Romanque P, Santander G, Castillo I, Varela P, Tapia G, Videla LA. Involvement of Kupffer cell-dependent signaling in T3-induced hepatocyte proliferation in vivo. Biol Chem. 2007;388:831–837. doi: 10.1515/BC.2007.101. [DOI] [PubMed] [Google Scholar]
- 43.Fernandez V, Videla LA, Tapia G, Israel Y. Increases in tumor necrosis factor-alpha in response to thyroid hormone-induced liver oxidative stress in the rat. Free Radic Res. 2002;36:719–725. doi: 10.1080/10715760290032566. [DOI] [PubMed] [Google Scholar]
- 44.Hirano T, Ishihara K, Hibi M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene. 2000;19:2548–2556. doi: 10.1038/sj.onc.1203551. [DOI] [PubMed] [Google Scholar]
- 45.Fernandez V, Tapia G, Varela P, Videla LA. Redox regulation of thyroid hormone-induced Kupffer cell-dependent IkappaB-alpha phosphorylation in relation to inducible nitric oxide synthase expression. Free Radic Res. 2005;39:411–418. doi: 10.1080/10715760400029637. [DOI] [PubMed] [Google Scholar]
- 46.Fernandez V, Tapia G, Varela P, Castillo I, Mora C, Moya F, Orellana M, Videla LA. Redox up-regulated expression of rat liver manganese superoxide dismutase and Bcl-2 by thyroid hormone is associated with inhibitor of kappaB-alpha phosphorylation and nuclear factor-kappaB activation. J Endocrinol. 2005;186:539–547. doi: 10.1677/joe.1.06261. [DOI] [PubMed] [Google Scholar]
- 47.Gloire G, Legrand-Poels S, Piette J. NF-kappaB activation by reactive oxygen species: fifteen years later. Biochem Pharmacol. 2006;72:1493–1505. doi: 10.1016/j.bcp.2006.04.011. [DOI] [PubMed] [Google Scholar]
- 48.Ramadori G, Christ B. Cytokines and the hepatic acute-phase response. Semin Liver Dis. 1999;19:141–155. doi: 10.1055/s-2007-1007106. [DOI] [PubMed] [Google Scholar]
- 49.Gruys E, Toussaint MJM, Niewold TA, Koopmans SJ. Acute phase reaction and acute phase proteins. J Zhejiang Univ SCI. 2005;6B:1045–1056. doi: 10.1631/jzus.2005.B1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Agrawal A, Cha-Molstad H, Samols D, Kushner I. Overexpressed nuclear factor-kappaB can participate in endogenous C-reactive protein induction, and enhances the effects of C/EBPbeta and signal transducer and activator of transcription-3. Immunology. 2003;108:539–547. doi: 10.1046/j.1365-2567.2003.01608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45–S53. doi: 10.1002/hep.20969. [DOI] [PubMed] [Google Scholar]
- 52.Fernández V, Videla LA. Kupffer cell-dependent signaling in thyroid hormone calorigenesis: possible applications for liver preconditioning. Curr Signal Trans Ther. 2009;4:144–141. [Google Scholar]
- 53.Casillas-Ramirez A, Mosbah IB, Ramalho F, Rosello-Catafau J, Peralta C. Past and future approaches to ischemia-reperfusion lesion associated with liver transplantation. Life Sci. 2006;79:1881–1894. doi: 10.1016/j.lfs.2006.06.024. [DOI] [PubMed] [Google Scholar]
- 54.Fernandez V, Castillo I, Tapia G, Romanque P, Uribe-Echevarria S, Uribe M, Cartier-Ugarte D, Santander G, Vial MT, Videla LA. Thyroid hormone preconditioning: protection against ischemia-reperfusion liver injury in the rat. Hepatology. 2007;45:170–177. doi: 10.1002/hep.21476. [DOI] [PubMed] [Google Scholar]
- 55.Fernandez V, Tapia G, Varela P, Gaete L, Vera G, Mora C, Vial MT, Videla LA. Causal role of oxidative stress in liver preconditioning by thyroid hormone in rats. Free Radic Biol Med. 2008;44:1724–1731. doi: 10.1016/j.freeradbiomed.2008.01.010. [DOI] [PubMed] [Google Scholar]
- 56.Kobayashi M, Yamamoto M. Molecular mechanisms activating the Nrf2-Keap1 pathway of antioxidant gene regulation. Antioxid Redox Signal. 2005;7:385–394. doi: 10.1089/ars.2005.7.385. [DOI] [PubMed] [Google Scholar]