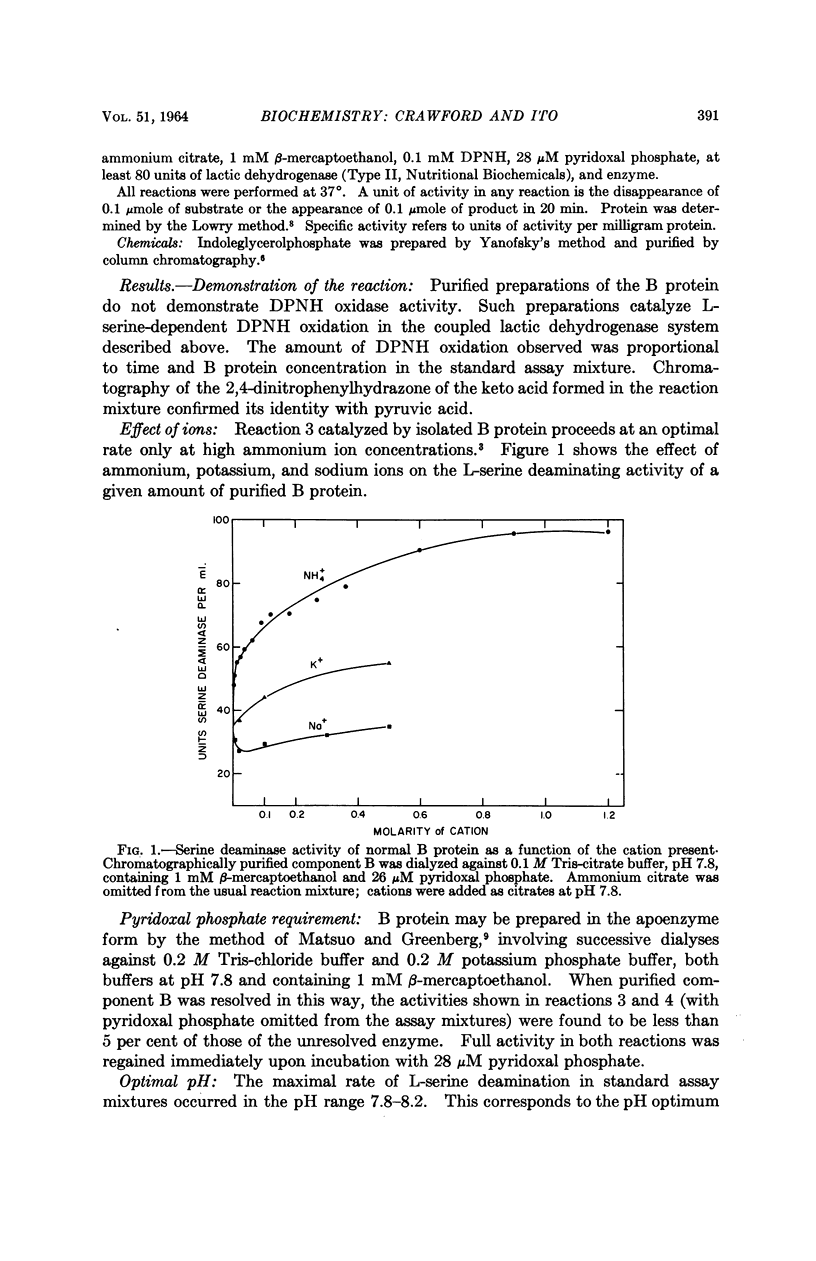
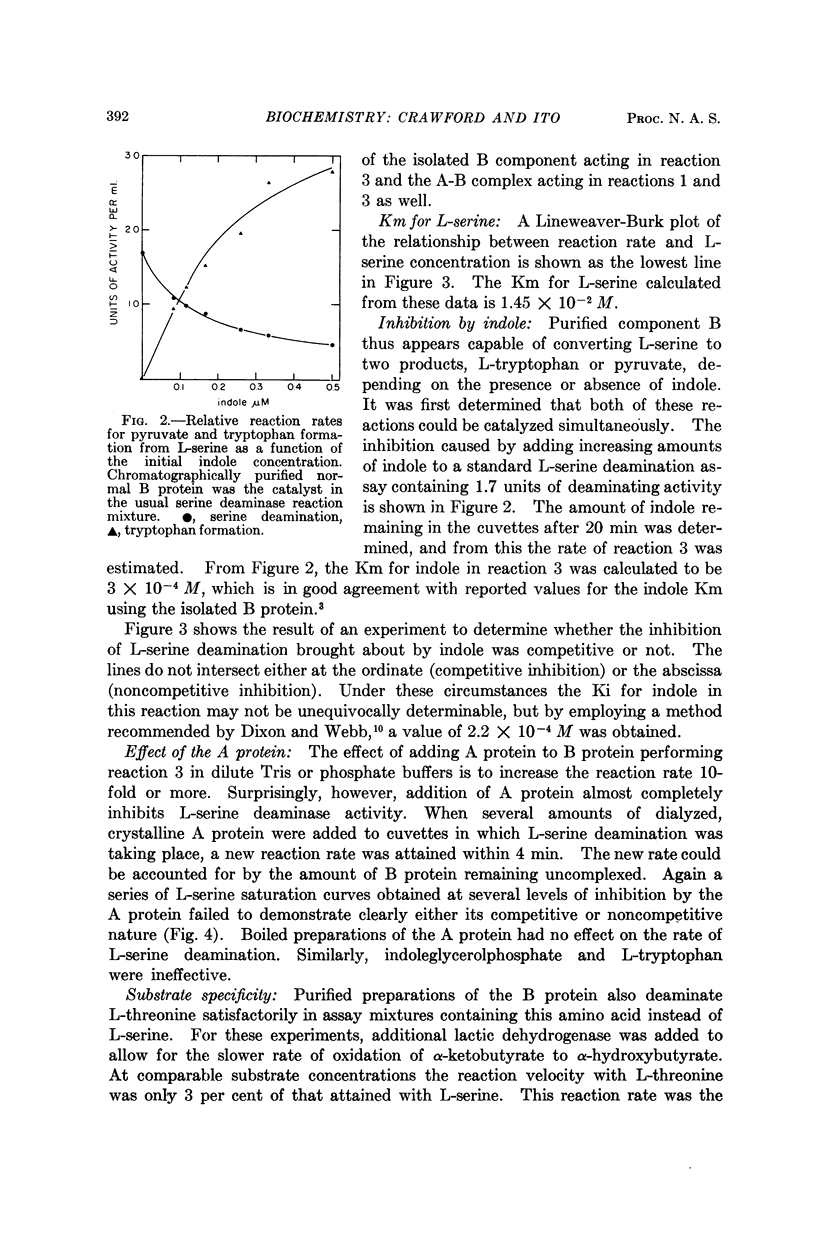
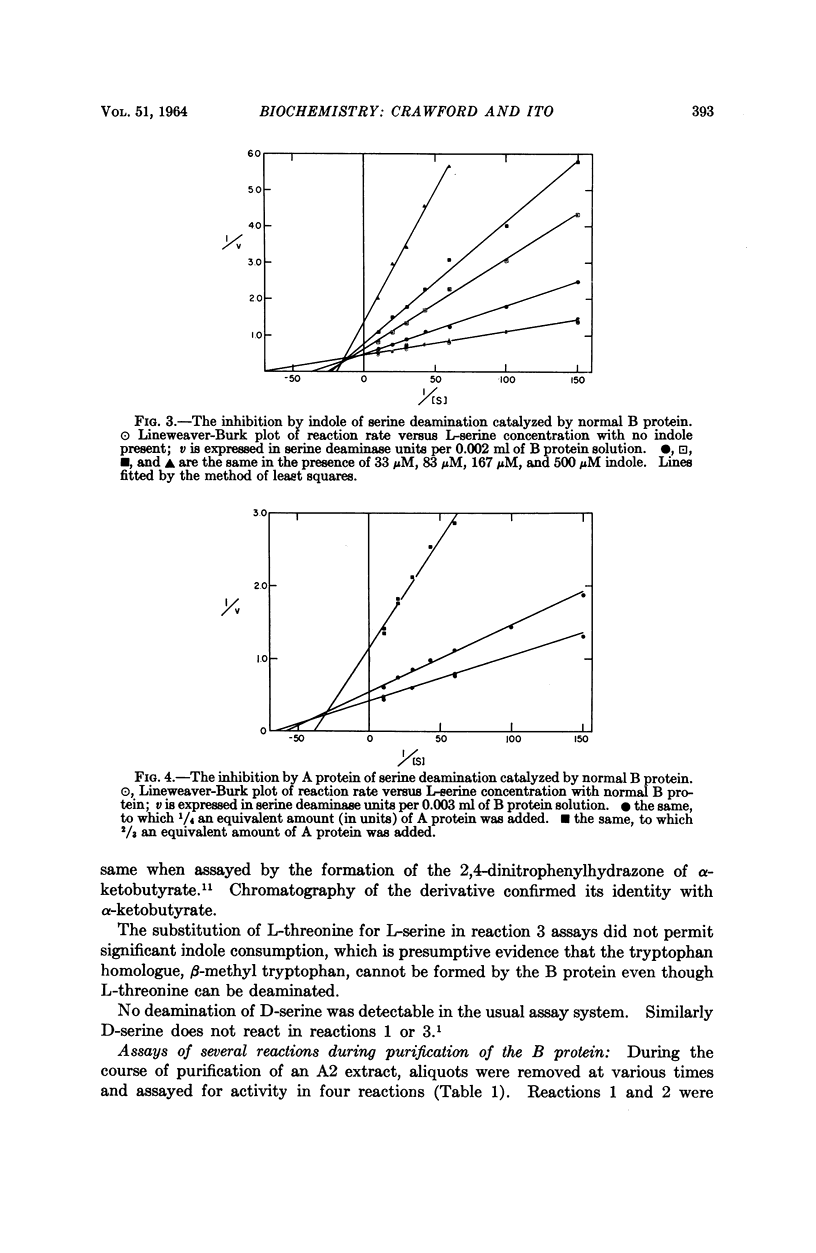
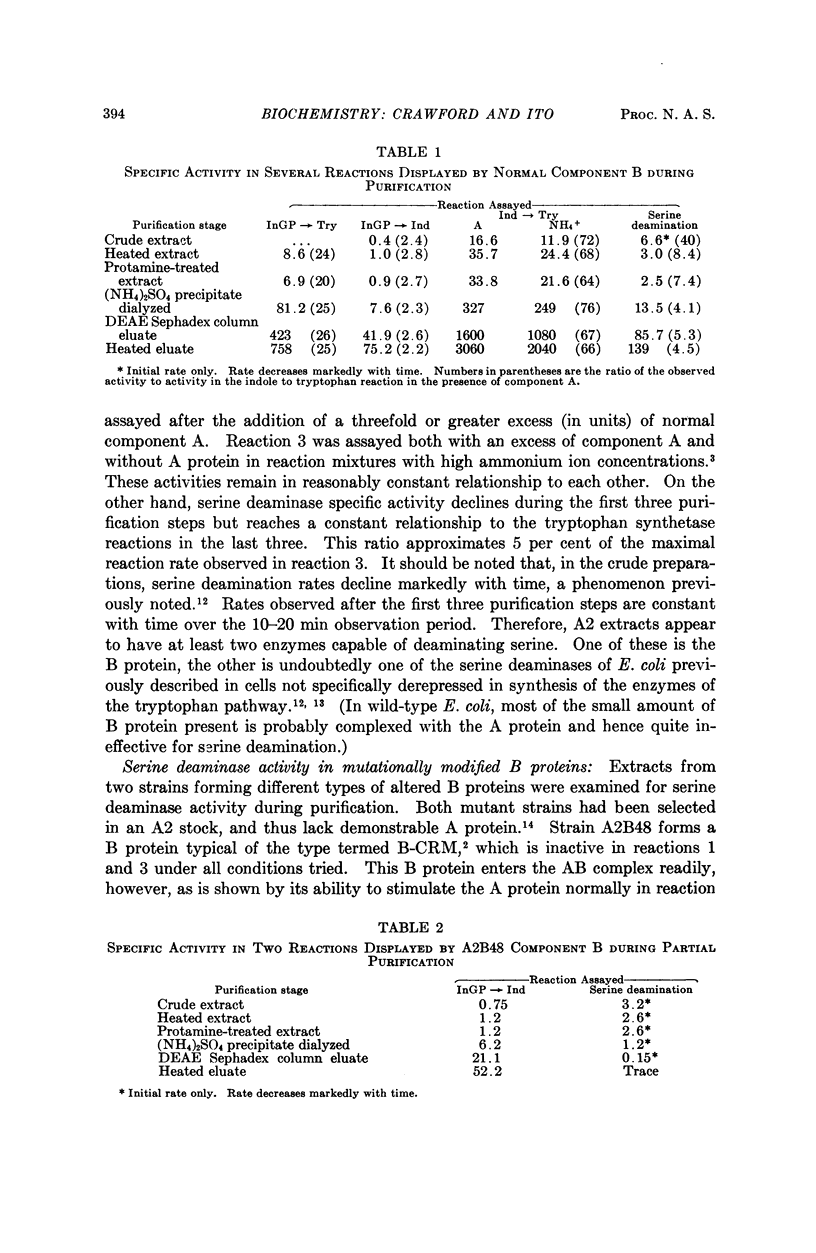
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- CRAWFORD I. P. Identification of the triose phosphate formed in the tryptophan synthetase reaction. Biochim Biophys Acta. 1960 Dec 4;45:405–407. doi: 10.1016/0006-3002(60)91474-8. [DOI] [PubMed] [Google Scholar]
- CRAWFORD I. P., JOHNSON L. M. Mutants of Escherichia coli defective in the B protein of tryptophan synthetase. I. Selection and classification. Genetics. 1963 May;48:725–736. doi: 10.1093/genetics/48.5.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HATANAKA M., WHITE E. A., HORIBATA K., CRAWFORD I. P. A study of the catalytic properties of Escherichia coli tryptophan synthetase, a two-component enzyme. Arch Biochem Biophys. 1962 Jun;97:596–606. doi: 10.1016/0003-9861(62)90129-7. [DOI] [PubMed] [Google Scholar]
- HENNING U., HELINSKI D. R., CHAO F. C., YANOFSKY C. The A protein of the tryptophan synthetase of Escherichia coli. Purification, crystallization, and composition studies. J Biol Chem. 1962 May;237:1523–1530. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MATSUO Y., GREENBERG D. M. A crystalline enzyme that cleaves homoserine and cystathionine. III. Coenzyme resolution, activators, and inhibitors. J Biol Chem. 1959 Mar;234(3):507–515. [PubMed] [Google Scholar]
- METZLER D. E., SNELL E. E. Deamination of serine. II. D-Serine dehydrase, a vitamin B6 enzyme from Escherichia coli. J Biol Chem. 1952 Sep;198(1):363–373. [PubMed] [Google Scholar]
- NEWTON W. A., SNELL E. E. CATALYTIC PROPERTIES OF TRYPTOPHANASE, A MULTIFUNCTIONAL PYRIDOXAL PHOSPHATE ENZYME. Proc Natl Acad Sci U S A. 1964 Mar;51:382–389. doi: 10.1073/pnas.51.3.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- UMBARGER H. E., BROWN B. Threonine deamination in Escherichia coli. II. Evidence for two L-threonine deaminases. J Bacteriol. 1957 Jan;73(1):105–112. doi: 10.1128/jb.73.1.105-112.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOOD W. A., GUNSALUS I. C. Serine and threonine desaminaes of Escherichia coli; activators for a cell-free enzyme. J Biol Chem. 1949 Nov;181(1):171–182. [PubMed] [Google Scholar]
- YANOFSKY C. The tryptophan synthetase system. Bacteriol Rev. 1960 Jun;24(2):221–245. doi: 10.1128/br.24.2.221-245.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanofsky C., Crawford I. P. THE EFFECTS OF DELETIONS, POINT MUTATIONS, REVERSIONS AND SUPPRESSOR MUTATIONS ON THE TWO COMPONENTS OF THE TRYPTOPHAN SYNTHETASE OF ESCHERICHIA COLI. Proc Natl Acad Sci U S A. 1959 Jul;45(7):1016–1026. doi: 10.1073/pnas.45.7.1016. [DOI] [PMC free article] [PubMed] [Google Scholar]



