Abstract
Objectives. To determine the prevalence, the clinical predictors, and the prognostic significances of Worsening Renal Function (WRF) in hospitalized patients with Acute Heart Failure (AHF). Methods. 394 consecutively hospitalized patients with AHF were evaluated. WRF was defined as an increase in serum creatinine of ≥0.3 mg/dL from baseline to discharge. Results. Nearly 11% of patients developed WRF. The independent predictors of WRF analyzed with a multivariable logistic regression were history of chronic kidney disease (P = .047), age >75 years (P = .049), and admission heart rates ≥100 bpm (P = .004). Mortality or rehospitalization rates at 1 month, 6 months, and 1year were not significantly different between patients with WRF and those without WRF. Conclusion. Different clinical predictors at hospital admission can be used to identify patients at increased risk for developing WRF. Patients with WRF compared with those without WRF experienced no significant differences in hospital length of stay, mortality, or rehospitalization rates.
1. Introduction
In the setting of heart failure, baseline renal insufficiency is a common and well-established independent marker of poor prognosis [1–6]. Worsening Renal Function (WRF) during the hospitalization for Acute Heart Failure (AHF) occurs frequently and may also have a prognostic significance. Indeed, several studies have reported that even small changes in renal function are associated with longer length of hospital stay, increased in-hospital costs, higher in-hospital mortality, higher mortality, and rehospitalization rates at short and long term [7–12]. However, not all the published information agree that WRF is associated with a worse clinical outcomes [13, 14]. Moreover, not all the studies have adopted the same definition of WRF [7, 8, 10, 11, 13–15] and in most of them only short-term followup (in-hospital complications and 6-months follow-up) was carried on [9–11]. Lastly, the major part of the results is derived from retrospective analyses, and in several studies a multivariate analysis was not performed to identify the independent prognostic value of WRF [7, 10, 11, 15]. Therefore, the role that WRF plays in general HF population is still to be better validated.
The goal of this study is to determine the prevalence, the clinical predictors, and the prognostic significance of WRF in a consecutive series of hospitalized patients for AHF.
2. Materials and Methods
2.1. Study Population
We enrolled patients consecutively admitted for AHF at our Institute from October 2002 to May 2008. Criteria for inclusion were exacerbation of previously documented Heart Failure (HF) or new onset of AHF using standard Framingham criteria [16]. The only criterion for exclusion was the presence of terminal noncardiac illness that could influence short-term prognosis.
2.2. Study Protocol
The study was approved by the local Ethics Committee, and all patients gave informed consent to participate.
All patients underwent a complete clinical and laboratory examination at the time of hospital admission and at hospital discharge. Estimated glomerular filtration rate (eGFR) was calculated using Modification of Diet in Renal Disease (MDRD) equation. This has been shown to be the best method for the indirect assessment of renal function in HF population [17–19]. WRF was defined as an increase in serum creatinine of ≥0.3 mg/dL from baseline to discharge (WRF(CRE)). This value was chosen because it has previously been demonstrated to have the maximum sensitivity and specificity to predict the prognosis [11].
In order to verify the prognostic value of WRF, the hazard ratios for death and rehospitalization were also assessed for a decline in eGFR ≥20% from baseline to discharge (WRF(GFR)) [20].
Followup was performed by clinical visits and/or telephone calls at 1 month, 6 months, and 1 year. The main endpoints were hospital length of stay, death, and rehospitalization risks.
2.3. Statistical Analysis
Continuous variables were expressed as the arithmetic mean and the standard deviation (SD). Discrete variables were presented as a percentage. Associations between WRF and continuous variables were analyzed using Student's t-test for normal data and Wilcoxon test for not normally distributed variables. Discrete Variables were compared with the use of Chi-square analysis. A forward stepwise multivariable logistic regression analysis was performed to identify the independent variables predictive of WRF. All the variables which were significantly different (P < .1) between patients with and without WRF at univariable analysis were taken into multivariable analysis, but only retained at an exit significance value of P < .05. Associations of the development of WRF with the prognostic outcomes (hospital length of stay, hospitalization, and mortality risks) were assessed with a Cox proportional hazards analysis. Survival probability curves were constructed according to the Kaplan-Meier method.
3. Results
3.1. Baseline Characteristics and Prevalence of WRF
Patients' characteristics are presented in Table 1. We initially enrolled in the study 402 consecutive patients. Eight of them were lost to follow-up and were excluded from the analysis. No one of the lost to follow-up patients had developed the WRF during the index hospitalization. Therefore, the study population consists of 394 consecutive patients. The mean age of the cohort was 77.9 (SD 10.1) years, with nearly 70% of the patients over 75 years. The majority of the patients were male (67.5%). Almost 60% of the total population had a history of prior hypertension (58.4%), heart failure (61.7%), and acute coronary syndrome (57.4%). Relatively high percentages of diabetes (33%) and anemia (42.4%) were present in the population. The mean ejection fraction was 39.6% (SD 12.2) with 241 patients (61.2%) having EF < 45%. On admission, 159 (40.3%) patients were in NYHA class IV, 194 (49.2%) patients were in NYHA class III, and only 41 (10.4%) patients were in NYHA class II. At time of the admission, more than half of the patients were on diuretics (72.3%) and on ACE inhibitors (54.7%) treatments; 109 (27.7%) patients were receiving beta-blockers. The mean serum creatinine was 1.5 (SD 0.8) mg/dL with 111(28.2%) of the patients having values >1.5 mg/dL. WRF(CRE) and WRF(GFR) occurred, respectively, in 10.9% and in 11.6% of the population.
Table 1.
Characteristics at admission of the whole population and of patients with and without WRF (CRE).
| Total | WRF absent | WRF present | P-value | |
|---|---|---|---|---|
| (n = 394) | (n = 351) | (n = 43) | ||
| Demographics | ||||
| Age (mean, SD) | 77.9 (10.1) | 77.8 (10.3) | 79.2 (8.4) | .380 |
| Age >75 years | 274 (69.5) | 239 (68.1) | 35 (81.4) | .074 |
| Males | 266 (67.5) | 236 (67.2) | 30 (69.8) | .738 |
| Medical history | ||||
| Prior heart failure | 243 (61.7) | 218 (62.1) | 25 (58.1) | .613 |
| Diabetes | 130 (33.0) | 108 (30.8) | 12 (27.9) | .700 |
| Valvular disease | 108 (27.4) | 99 (28.2) | 9 (20.9) | .313 |
| COPD | 122 (30.9) | 111 (31.6) | 11 (25.6) | .418 |
| Hypertension | 230 (58.4) | 201 (57.3) | 29 (67.4) | .201 |
| Peripheral vascular disease (PVD) | 74 (18.8) | 67 (19.1) | 7 (16.3) | .656 |
| Previus acute coronary syndrome | 226 (57.4) | 204 (58.1) | 22 (51.2) | .384 |
| Prior renal failure | 95 (24.1) | 76 (21.6) | 19 (44.2) | .001 |
| Clinical and laboratory parameters | ||||
| Systolic blood pressure, mm Hg (mean, SD) | 132 (25.5) | 131 (25.1) | 137 (29.1) | .138 |
| Systolic blood pressure >160 mm Hg | 43 (10.9) | 36 (10.3) | 7 (16.3) | .230 |
| Serum creatinine, mg/dL (mean, SD) | 1.5 (0.8) | 1.47 (0.8) | 1.71 (0.9) | .066 |
| Creatinine >1,5 mg/dL | 111 (28.2) | 92 (26.2) | 19 (44.2) | .013 |
| GFR, mL/min (mean, SD) | 54 (23.5) | 55 (22.8) | 51 (28.4) | .070 |
| GFR <60 mL/min | 239 (60.7) | 207 (59.0) | 32 (74.4) | .055 |
| Sodium, mEq/L (mean, SD) | 140 (4.4) | 140 (4.5) | 140 (3.5) | .854 |
| Potassium, mEq/L (mean, SD) | 4.0 (0.6) | 4.0 (0.6) | 3.9 (0.6) | .219 |
| Glycemia, mg/dL (mean, SD) | 76 (73.0) | 78 (74.6) | 58 (56.6) | .114 |
| Haemoglobin, gr/dL (mean, SD) | 12.6 (6.5) | 12.7 (1.8) | 12.1 (6.9) | .387 |
| Anaemia (hemoglobin <12 gr/dL) | 167 (42.4) | 147 (41.9) | 20 (46.5) | .562 |
| Haematocrit (mean, SD) | 38 (6.1) | 39 (6.2) | 38 (5.2) | .502 |
| Azotemia, mg/dL (mean, SD) | 46 (50.0) | 45.5 (49.1) | 50.2 (60.5) | .561 |
| NYHA class (mean, SD) | 3.3 (0.7) | 3.3 (0.6) | 3.3 (0.7) | .778 |
| NYHA class III-IV | 353 (89.6) | 316 (90.0) | 37 (86.0) | .420 |
| KILLIP (mean, SD) | 2.4 (0.6) | 2.3 (0.6) | 2.4 (0.6) | .363 |
| Doppler echocardiography | ||||
| Ejection fraction (mean, SD) | 39.6 (12.2) | 39.5 (12.2) | 39.7 (12.4) | .942 |
| Ejection fraction <45% | 241 (61.2) | 215 (61.3) | 26 (60.5) | .869 |
| Left atrial size, mm (mean, SD) | 46.3 (7.3) | 46.4 (7.5) | 45.2 (6.0) | .343 |
| Left ventricular size, mm (mean, SD) | 58.5 (9.5) | 58.4 (10.0) | 58.1 (6.7) | .982 |
| Left atrial dilatation | 232 (58.9) | 211 (60.1) | 21 (48.8) | .279 |
| Left ventricular dilatation | 265 (67.3) | 237 (67.5) | 28 (65.1) | .880 |
| Elettrocardiogram | ||||
| LBBB | 45 (11.4) | 40 (11.4) | 5 (11.6) | .960 |
| Heart rate, bpm (mean, SD) | 85.3 (17.0) | 85 (17.0) | 88 (16.3) | .276 |
| Heart rate ≥100 bpm | 80 (20.3) | 64 (18.2) | 16 (37.2) | .003 |
| Atrial fibrillation | 164 (41.6) | 149 (42.4) | 15 (34.9) | .342 |
| Medical treatments | ||||
| Aldosterone antagonists | 88 (22.3) | 76 (21.6) | 12 (27.9) | .352 |
| Diuretics | 285 (72.3) | 255 (72.5) | 30 (69.8) | .690 |
| Beta-blockers | 109 (27.7) | 98 (27.9) | 11 (25.6) | .746 |
| Calcium channell blockers | 60 (15.3) | 49 (14.0) | 11 (25.6) | .045 |
| ACE inhibitors | 215 (54.7) | 190 (54.1) | 25 (58.1) | .618 |
| ARBs | 37 (9.4) | 30 (8.5) | 7 (16.3) | .101 |
| Statins | 46 (11.7) | 40 (11.4) | 6 (14.0) | .865 |
| ASA | 163 (41.4) | 143 (40.7) | 20 (46.5) | .468 |
| Warfarin | 101 (25.6) | 91 (25.9) | 10 (23.3) | .705 |
| Nitrates | 144 (36.5) | 133 (37.9) | 11 (25.6) | .114 |
| Digoxin | 99 (25.1) | 95 (27.1) | 4 (9.3) | .006 |
ACE: angiotensin-converting enzyme; ARBs: angiotensin receptor blockers; ASA: acetylsalycilic acid; LBBB: left bundle branch block; COPD: chronic obstructive pulmunary disease; NYHA: New York Health Association.
3.2. Predictors of WRF
In the univariable analysis, patients who experienced WRF(CRE) compared with those without WRF(CRE) were more likely to be older than 75 years, to have a history of preexisting Chronic Renal Failure (CRF), and to have higher heart rate (atrial or sinus arrhythmia with a heart rate >100 bpm). Higher serum creatinine (Scr) level and lower eGFR value at admission were also found to be significantly associated with the development of WRF. Moreover, patients with WRF(CRE) were more likely to be on calcium channel blockers and less likely to be on digoxin treatment. There were no significant differences in the other considered variables.
When a multivariable analysis was conducted (results listed in Table 2), preexisting CRF, admission heart rate (≥100 bpm), and age (>75 years) remained independent risk factors for the development of WRF(CRE) (resp., P = .047; .004; .049). Conversely, digoxin treatment resulted to have a protective effect against WRF(CRE) (P = .024).
Table 2.
Predictors of WRF (CRE): multivariable analysis.
| Predictors | Odds ratio | 95% CI | P-value |
|---|---|---|---|
| Age >75 years | 2.34 | 1.00–5.46 | .049 |
| Calcium channel blockers | 1.53 | 0.68–3.44 | .300 |
| Creatinine >1,5 mg/dL | 0.90 | 0.33–2.50 | .840 |
| Digoxin | 0.29 | 0.10–0.85 | .024 |
| Prior renal failure | 2.80 | 1.01–7.79 | .047 |
| Heart rate ≥100 bpm | 2.89 | 1.40–5.94 | .004 |
CI: Confidence Interval.
3.3. WRF and Prognosis
The mean hospital length of stay during the index hospitalization was 8.8 ± 4.8 days [median: 7 days; interquartile range(iqr): 6–10] for the whole group. Patients who develop WRF(CRE) and patients without WRF(CRE) were similar in mean and median hospital length of stay (resp., mean: 8.5 ± 4.3 days; median: 7 days; iqr: 6–10; Versus mean: 8.9 ± 4.9 days; median: 7 days; iqr: 6–10; P = .64). Patients with and patients without WRF(GFR) experienced no significant differences in hospital length of stay (resp., mean: 8.0 ± 3.6 days; median: 7 days; iqr: 5–10; Versus mean: 8.9 ± 5 days; median: 7 days; iqr: 6–10; P = .29).
There were no statistically significant differences in rehospitalization risk between patients with WRF(CRE) and patients without WRF(CRE) at either 1 month, 6 months, or 12 months (results shown in Table 3). Patients with WRF(CRE) experienced no significantly higher risk of death at 1-, 6-, and 12-month followup (Table 3).
Table 3.
Association of WRF (CRE) with mortality and rehospitalizations risks.
| HR | 95% CI | P-value | |
|---|---|---|---|
| Mortality | |||
| 1 month | 1.01 | 0.13–8.07 | .99 |
| 6 months | 0.80 | 0.32–1.99 | .63 |
| 12 months | 1.03 | 0.49–2.15 | .94 |
| Rehospitalization | |||
| 1 month | 1.32 | 0.51–3.38 | .56 |
| 6 months | 1.00 | 0.59–1.70 | .99 |
| 12 months | 1.02 | 0.63–1.63 | .95 |
CI: Confidence Interval.
Similar results were observed when the WRF(GFR) definition was adopted (results shown in Table 4).
Table 4.
Association of WRF (GFR) with mortality and rehospitalizations risks.
| HR | 95% | P-value | |
|---|---|---|---|
| Mortality | |||
| 1 month | 0.93 | 0.12–7.43 | .94 |
| 6 months | 0.89 | 0.38–2.08 | .79 |
| 12 months | 1.09 | 0.54–2.12 | .81 |
| Rehospitalization | |||
| 1 month | 1.21 | 0.47–3.01 | .69 |
| 6 months | 0.82 | 0.47–1.43 | .48 |
| 12 months | 0.89 | 0.55–1.43 | .63 |
CI: Confidence Interval.
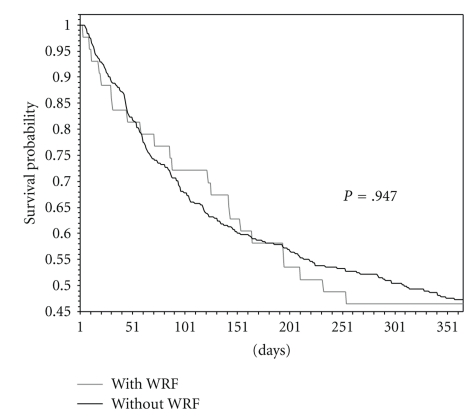
The combined endpoint death/rehospitalization was considered for the construction of Kaplan-Meier survival-free curves of patients with and without WRF(CRE). As it is possible to observe in Figure 1, the two curves were almost similar (log-rank test: P = .947).
Figure 1.
Kaplan-Meire hospitalization and morality free survival curves for patients with and without WRF.
4. Discussion
Several previous studies have reported a relatively high prevalence of WRF (around 25%) among patients hospitalized with acute heart failure [10, 12, 14, 21]. In our study, the prevalence of WRF is lower (11%). This is probably due to the fact that in our study patients with a transitory increase in Scr or decrease in eGFR, which did not persist at the moment of discharge, were not considered as WRF-patients.
The mechanisms which may cause WRF in patients with HF are multiple and are not completely understood [22]. Several predictors of WRF have been reported in the literature. One of the most acknowledged predictors is renal dysfunction either as a preexisting renal disease or as admission renal failure [8, 9, 13]. In a retrospective study carried on by Forman et al [10] on more 1000 patients, history of prior chronic heart failure, diabetes, systolic blood pressure >160 mmHg, and serum creatinine >1.5 mg/dL were identified as the most important predictors of WRF and used to elaborate a score to stratify the risk of developing WRF. Other reported important risk factors of WRF were: advanced age [7, 21], high systolic blood pressure [10, 14], diabetes [8, 10, 14], pulmonary edema [13], NYHA class [8], ejection fraction [8], use of high doses of furosemide [8, 9], and use of calcium channel blockers [9].
In our study, we report that the history of preexisting renal failure is one of the strongest independent predictors of WRF. The age was found to be another independent predictor of WRF. Patients who are >75 years old were more likely to develop WRF.
In the univariable analysis, baseline serum creatinine and baseline eGFR were associated with WRF; however these links disappeared when the multivariable analysis were conducted. Although the results of some studies [10, 13] are not consistent with these findings, in the ESCAPE study baseline renal insufficiency was not predictive of WRF even in the univariable analysis [14], and similar results were observed in the prospective study of Metra et al. [8].
In our investigation, digoxin use was shown to have a protective effect against WRF. This result has not been reported previously. The effect does not disappear on multivariable analysis. Since the small number of patients on digoxin use, this observation is likely to be due only to chance.
Heart rate >100 bpm was another independent risk factor of WRF that was not previously reported. This finding may be due to an underlying more severe cardiac disease or to an underuse of medications in these patients.
Worsening renal function during the hospitalization for acute heart failure has been shown to be associated with lengthier hospitalization. However, in our study, the patients with WRF had almost equal mean hospital length of stay to those who did not develop WRF.
Similar short- and long-term rehospitalization and mortality rates were found in patients with and without WRF. Similar findings were observed adopting both definitions of WRF (WRF(CRE) and WRF(GFR)) to assess the prognostic significance of WRF. These data are in agreement with the results reported in a European multicenter prospective study (POSH study) [13] and in the ESCAPE study [14]. On the other hand, several studies have reported that even small changes in serum creatinine during the hospitalization for acute heart failure are associated with higher rehospitalization risk and mortality rate. These conflicting results highlight the need of a better comprehension of the prognostic significances of WRF in patients with AHF. The fear of WRF may have important clinical consequences, since the physicians could tend to reduce diuretics dosages and to underuse important life-prolonging drugs such as aldosterone antagonists and ACE inhibitors.
Further prospective studies are needed to elucidate whether WRF, in the setting of AHF, is a justified fear or just a marker of intrinsic renal disease that is inevitable in patients with several risk factors.
References
- 1.Hillege HL, Girbes ARJ, De Kam PJ, et al. Renal function, neurohormonal activation, and survival in patients with chronic heart failure. Circulation. 2000;102(2):203–210. doi: 10.1161/01.cir.102.2.203. [DOI] [PubMed] [Google Scholar]
- 2.Smilde TDJ, Hillege HL, Voors AA, Dunselman PHJ, Van Veldhuisen DJ. Prognostic importance of renal function in patients with early heart failure and mild left ventricular dysfunction. American Journal of Cardiology. 2004;94(2):240–243. doi: 10.1016/j.amjcard.2004.03.075. [DOI] [PubMed] [Google Scholar]
- 3.Smith GL, Lichtman JH, Bracken MB, et al. Renal impairment and outcomes in heart failure: systematic review and meta-analysis. Journal of the American College of Cardiology. 2006;47(10):1987–1996. doi: 10.1016/j.jacc.2005.11.084. [DOI] [PubMed] [Google Scholar]
- 4.Dries DL, Exner DV, Domanski MJ, Greenberg B, Stevenson LW. The prognostic implications of renal insufficiency in asymptomatic and symptomatic patients with left ventricular systolic dysfunction. Journal of the American College of Cardiology. 2000;35(3):681–689. doi: 10.1016/s0735-1097(99)00608-7. [DOI] [PubMed] [Google Scholar]
- 5.McAlister FA, Ezekowitz J, Tonelli M, Armstrong PW. Renal insufficiency and heart failure: prognostic and therapeutic implications from a prospective cohort study. Circulation. 2004;109(8):1004–1009. doi: 10.1161/01.CIR.0000116764.53225.A9. [DOI] [PubMed] [Google Scholar]
- 6.Shlipak MG, Massie BM. The clinical challenge of cardiorenal syndrome. Circulation. 2004;110(12):1514–1517. doi: 10.1161/01.CIR.0000143547.55093.17. [DOI] [PubMed] [Google Scholar]
- 7.Weinfeld MS, Chertow GM, Stevenson LW. Aggravated renal dysfunction during intensive therapy for advanced chronic heart failure. American Heart Journal. 1999;138(2):285–290. doi: 10.1016/s0002-8703(99)70113-4. [DOI] [PubMed] [Google Scholar]
- 8.Metra M, Nodari S, Parrinello G, et al. Worsening renal function in patients hospitalised for acute heart failure: clinical implications and prognostic significance. European Journal of Heart Failure. 2008;10(2):188–195. doi: 10.1016/j.ejheart.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 9.Butler J, Forman DE, Abraham WT, et al. Relationship between heart failure treatment and development of worsening renal function among hospitalized patients. American Heart Journal. 2004;147(2):331–338. doi: 10.1016/j.ahj.2003.08.012. [DOI] [PubMed] [Google Scholar]
- 10.Forman DE, Butler J, Wang Y, et al. Incidence, predictors at admission, and impact of worsening renal function among patients hospitalized with heart failure. Journal of the American College of Cardiology. 2004;43(1):61–67. doi: 10.1016/j.jacc.2003.07.031. [DOI] [PubMed] [Google Scholar]
- 11.Gottlieb SS, Abraham W, Butler J, et al. The prognostic importance of different definitions of worsening renal function in congestive heart failure. Journal of Cardiac Failure. 2002;8(3):136–141. doi: 10.1054/jcaf.2002.125289. [DOI] [PubMed] [Google Scholar]
- 12.Krumholz HM, Chen Y-T, Vaccarino V, et al. Correlates and impact on outcomes of worsening renal function in patients ≥65 years of age with heart failure. American Journal of Cardiology. 2000;85(9):1110–1113. doi: 10.1016/s0002-9149(00)00705-0. [DOI] [PubMed] [Google Scholar]
- 13.Cowie MR, Komajda M, Murray-Thomas T, Underwood J, Ticho B. Prevalence and impact of worsening renal function in patients hospitalized with decompensated heart failure: results of the prospective outcomes study in heart failure (POSH) European Heart Journal. 2006;27(10):1216–1222. doi: 10.1093/eurheartj/ehi859. [DOI] [PubMed] [Google Scholar]
- 14.Nohria A, Hasselblad V, Stebbins A, et al. Cardiorenal interactions. Insights from the ESCAPE trial. Journal of the American College of Cardiology. 2008;51(13):1268–1274. doi: 10.1016/j.jacc.2007.08.072. [DOI] [PubMed] [Google Scholar]
- 15.Akhter MW, Aronson D, Bitar F, et al. Effect of elevated admission serum creatinine and its worsening on outcome in hospitalized patients with decompensated heart failure. American Journal of Cardiology. 2004;94(7):957–960. doi: 10.1016/j.amjcard.2004.06.041. [DOI] [PubMed] [Google Scholar]
- 16.Ho KKL, Pinsky JL, Kannel WB, Levy D. The epidemiology of heart failure: the Framingham study. Journal of the American College of Cardiology. 1993;22(4):6A–13A. doi: 10.1016/0735-1097(93)90455-a. [DOI] [PubMed] [Google Scholar]
- 17.O’Meara E, Chong KS, Gardner RS, Jardine AG, Neilly JB, McDonagh TA. The Modification of Diet in Renal Disease (MDRD) equations provide valid estimations of glomerular filtration rates in patients with advanced heart failure. European Journal of Heart Failure. 2006;8(1):63–67. doi: 10.1016/j.ejheart.2005.04.013. [DOI] [PubMed] [Google Scholar]
- 18.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Annals of Internal Medicine. 1999;130(6):461–470. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- 19.Smilde TDJ, Van Veldhuisen DJ, Navis G, Voors AA, Hillege HL. Drawbacks and prognostic value of formulas estimating renal function in patients with chronic heart failure and systolic dysfunction. Circulation. 2006;114(15):1572–1580. doi: 10.1161/CIRCULATIONAHA.105.610642. [DOI] [PubMed] [Google Scholar]
- 20.Testani JM, McCauley BD, Chen J, Shumski M, Shannon RP. Worsening renal function defined as an absolute increase in serum creatinine is a biased metric for the study of cardio-renal interactions. Cardiology. 2010;116(3):206–212. doi: 10.1159/000316038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Belziti CA, Bagnati R, Ledesma P, Vulcano N, Fernández S. Worsening renal function in patients adamite with acute decompensated heart failure: incidence, risk factors and prognostic implications. Revista Espanola de Cardiologia. 2010;63(3):294–302. doi: 10.1016/s1885-5857(10)70062-1. [DOI] [PubMed] [Google Scholar]
- 22.Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal syndrome. Journal of the American College of Cardiology. 2008;52(19):1527–1539. doi: 10.1016/j.jacc.2008.07.051. [DOI] [PubMed] [Google Scholar]