Abstract
Background: Increasing evidence supports the important role of illness state and individual characteristics in insight. Methods: Insight, as measured with the Scale to Assess Unawareness of Mental Disorder, over the first 2 years of early-onset first-episode psychosis and its correlations with clinical, socio-demographic, cognitive, and structural brain variables are studied. Results: (1) insight at 2 years is poorer in schizophrenia spectrum disorders (SSDs) than in subjects with other psychoses; (2) the more severe the psychosis, the worse the insight. In SSD, depressive symptoms, poorer baseline executive functioning, lower IQ, longer duration of untreated psychosis (DUP), and poorer premorbid infancy adjustment are associated with poorer insight; frontal and parietal gray matter (GM) reductions at baseline correlate with worse insight into having psychotic symptoms at 2 years; (3) insight into having a mental disorder (Scale to Assess Unawareness of Mental Disorder [SUMD]1) at 1 year, DUP, and baseline IQ are the most consistent variables explaining different aspects of insight at 2 years in SSD patients. IQ and SUMD1 at 1 year, together with left frontal and parietal GM volumes, explain 80% of the variance of insight into having specific psychotic symptoms in SSD patients (adjusted R2 = 0.795, F = 15.576, P < .001). Conclusion: Insight is a complex phenomenon that depends both on severity of psychopathology and also on disease and subject characteristics, such as past adjustment, IQ, DUP, cognitive functioning, frontal and parietal GM volumes, and age, gender, and ethnicity.
Keywords: first-episode psychosis, insight, brain volume, depression, early-onset psychosis, awareness
Introduction
Lack of insight (usually referring to impaired awareness of having an illness) is a well-recognized and common clinical characteristic of both affective and nonaffective psychosis.1–3 Insight is a complex concept, associated with symptom severity, cognitive impairment, and neurobiological dysfunction.4–10 The most commonly and systematically used definition of insight comprises a multidimensional concept that includes,1,2,8,11 (1) awareness of having a mental disorder, (2) awareness of the need of treatment, (3) understanding the social consequences of the disorder, (4) awareness of specific signs and symptoms of the disorder, and (5) attribution of symptoms to the disorder.11,12
Although few clinicians would dispute the relevance of insight, there is no a general agreement on its clinical meaning and biological substrate. Lack of insight has been considered a core symptom of schizophrenia,13,14 a psychological coping mechanism aimed at preserving emotional well-being,15,16 a result of some cognitive or psychological dysfunction,9,17 a kind of neurological mechanism similar to anosognosia,18–20 or a combination of the above.5
Most, but not all studies that have assessed insight in patients with psychosis report a relationship between poor insight and diagnosis of schizophrenia,1,6,21–23 more severe psychotic psychopathology,5,6,21,22,24–26 lower cognitive functioning (with some specificity to executive functioning),10,26–30 lower gray matter (GM) volumes in the temporal, frontal and/or parietal regions,9,31 and poor outcome.13,32–35 Very recently, poor insight has been associated with long duration of untreated psychosis.36 In addition, poor insight has been generally related to less severe depressive symptomatology and fewer suicide attempts after a first episode of psychosis (FEP).7,22,25,28,37,38 A meta-analysis by Mintz and colleagues22 showed that, as positive, negative, and general symptoms increased, the degree of insight decreased, and as the degree of insight increased, depressive symptoms increased.22 In addition, a meta-analysis by Aleman and colleagues8 found that in patients with schizophrenia and other psychoses, although there was a significant relationship, the predictive value of neurocognition was rather modest.
Total brain, gray, and white matter volumes have been studied in relation to insight, with contradictory results.25,39 Associations between lower gray-matter volumes in prefrontal, temporal, and parietal lobes and dimensions of insight have been observed in stable schizophrenia patients,4,10,37,40 although some studies have found no association between structural data and insight.38 For a review, see Gilleen et al.20
Taken together, these findings argue in favor of insight being partially dependent on the mental status of the patient but also having some trait value, related to the diagnosis of schizophrenia.6 The study of insight in FEP patients entails crucial advantages. Indeed, clinical and neuropsychological correlates with insight in FEP are somewhat different than those reported for established schizophrenia.26,28 First, insight is not a static condition and it may be influenced by neurobiological, clinical, therapeutic, and social circumstances. Furthermore, insight tends to be better in at risk mental state for psychosis than during the FEP,41 it tends to improve after first episode,6,25 and patients with FEP are less aware of having a mental illness than multiple-episode patients.42 Second, FEP constitutes a heterogeneous group that includes different psychosis categories, the boundaries of which are now a subject of lively debate.40
Although the average age of onset of schizophrenia has been calculated as 18 in men and 25 in women,43 very little research has been conducted to assess the effect on insight of having a FEP in adolescence, before personality and neurobiological maturation are completed. An adolescent sample as the one studied in the present investigation may give valuable information about the effect of age on the different domains of insight and also lacks the effects of chronicity, multiple episodes, or medication. In a prior publication,6 we found that in adolescents with first-episode early onset psychosis (EOP), insight into having a mental disorder, its consequences, and the need for treatment improved over the early phases of treatment. Poorer insight was associated with more severe positive, negative, and overall psychotic symptoms, and with poorer global functioning, at both baseline and 6-month follow-up.6 After 6 months, poor insight correlated with final diagnosis of schizophrenia at the 1-year follow-up.
The aim of the present study was to explore prospectively the relationship between insight and depressive symptomatology, neurocognitive performance, and GM volumes. In addition, we wanted to extend our previous study6 by examining the course of insight over the first 2 years after a first episode of EOP. We analyze all these aspects splitting the whole sample into schizophrenia spectrum disorders (SSDs) and non-SSD. To our knowledge, this is the first study that prospectively assesses the relationship of insight with depressive symptoms and neuropsychological and neurobiological markers in an early onset FEP cohort. Based on our previous studies and those by other groups reviewed in this introduction, our hypotheses were that: (1) insight improves over the first 2 years after a first episode of EOP, (2) insight is poorer in schizophrenia patients than in patients with non-SSD psychoses 2 years after the psychosis onset, (3) more severe positive or negative symptoms are associated with poorer insight throughout the course of illness, (4) better insight is associated with the presence of more depressive symptomatology over the course of EOP, and (5) poorer prefrontal cognitive functioning and/or lower prefrontal volumes are associated with poorer insight irrespective of specific diagnosis (SSD or non-SSD).
Methods
Data were collected as part of a 2-year longitudinal study of FEP with onset in adolescence. The study population consisted of 110 consecutively treated FEP with early and recent onset of psychosis, recruited as part of a national longitudinal multicenter study.44 Inclusion criteria for patients included an onset of positive psychosis symptoms less than 6 months prior to baseline assessment and age between 9 and 17 years. Exclusion criteria were presence of a concomitant Axis I disorder at the time of evaluation that might account for the psychotic symptoms (such as substance abuse or dependence), mental retardation (Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV] criteria), neurological disorders, history of head trauma with loss of consciousness, and pregnancy or breast feeding. Detailed information on the characteristics of the sample and methods of the study is reported elsewhere.44,45 The study was approved by the IRBs of all participating clinical centers. All parents or legal guardians gave written informed consent before the study, and patients agreed to participate.
Clinical Assessment
Diagnosis was made according to the DSM-IV criteria46 (at baseline, 6 months, 1 year, and 2 years), using the Kiddie Schedule for Affective Disorders and Schizophrenia, Present and Lifetime version (K-SADS-PL) Spanish adaptation at baseline, and 2 years.47,48 Socioeconomic status (SES) was estimated using parental years of formal education. Psychopathology was assessed with the Spanish adaptation of the Positive and Negative Symptom Scale (PANSS).49 Depressive symptomatology was assessed with the Hamilton Rating Scale for Depression (HRSD).50 The questionnaire rates the severity of symptoms observed in depression, such as low mood, insomnia, agitation, anxiety, and weight loss. Each question has 3–5 possible responses, which increase in severity. Seventeen questions contribute to the total score (HRSD-17). Presence of suicidal thoughts or behaviors was assessed by means of the Likert Clinical Global Impressions–Severity of Suicide scale.51
Premorbid adjustment was assessed with the childhood subscale of the Premorbid Adjustment Scale (PAS).52
The Children’s Global Assessment of Functioning (C-GAF) Scale,53 which measures the severity of symptoms and the level of functioning on a scale from 1 to 100, was used for assessing general functioning.
Duration of untreated psychosis (DUP) was calculated as the time elapsed between the first positive, negative, or disorganization symptom recalled and the baseline assessment, in days.
Insight Assessment.
Insight was measured by the abbreviated version of the Scale to Assess Unawareness of Mental Disorder (SUMD).2 The SUMD has been widely used to assess insight in schizophrenia and its relationship to psychopathology11 and takes into account the aforesaid multidimensional approach. It gives more information than the G12 item of the PANSS in this population.6 The patients were interviewed at baseline when they were in condition to cooperate with a clinical interview (those patients acutely psychotic or too disorganized were interviewed after the improvement of symptoms allowed for a clinical interview in which the SUMD could be assessed). The SUMD was completed by the clinician in the context of an interview and using all the information from the complete history, mental state examination, and chart. The first 3 items assess, in turn, the subject’s general insight into having a mental disorder (SUMD1), the effects of medication on the disorder (SUMD2), and general understanding of the consequences of the disorder (SUMD3). These 3 items were completed for all subjects, and their level of insight was rated on a 5-point Likert scale (1 = full insight and 5 = no insight). Items 4–9 pertain to specific symptoms and are asked only if it has been established that the patient experienced any of these symptoms (score of 3 or more on the PANSS for that symptom). The same 5-point Likert scale was used. If the subject showed insight into a symptom, defined as a score between 1 (full insight) and 3 (partial insight), the subject’s attribution of the symptom to the psychotic illness was assessed and rated on a 5-point Likert scale (1 = correct attribution and 5 = incorrect attribution). Along with SUMD items 1–3, insight and attribution of symptoms (SUMDTot1 and SUMDTot2, respectively) can be considered the 5 dimensions of insight. In all items, there was the option to score 0 = nonrelevant, which was not counted in subsequent analyses.
Neuropsychological Measures.
For the purpose of the present study, we selected those measures considered to be related to executive function (EF) and dorsolateral prefrontal cortex (DLPC) functioning54,55 because of their possible relationship with insight.56 EF was measured by a composite score formed by a group of measures from different tests that were previously transformed into z-scores. That is to say, we used the total errors, perseverative errors, and conceptual-level responses from the Wisconsin Card Sorting Test (WCST),57 the interference score of the Stroop test,58,59 number of correct words from the verbal fluency test (Controlled Verbal Fluency Task),60 and the time to complete the task and the number of mistakes committed from the Trail Making Test-B61,62 to calculate an arithmetic mean index for the EF domain.63,64
The index measure related to DLPC functioning was calculated as a composite score grouping different results of the following tasks that have been related to the corresponding cognitive functions as follows: attention (measured using reaction time and correct responses from the Continuous Performance Test, CPT-II65; Digit Span Forward, Trail Making Test A61; words, and colors parts of the Stroop test58,59), working memory (Digit Span Backward and Letter–Number Sequencing from the Wechsler Adult Intelligence Scale),66 and adding the composite score for EF itself. All tests were administered and scored according to standardized published instructions by 6 previously trained research psychologists. Interrater reliability for some scales (eg, WCST) was determined by interclass correlation coefficient, which was between 0.95 and 0.99. All subjects performed the same battery of tests in a fixed order. To estimate general intellectual functioning, we used one of the most widely used short forms from the full Wechsler Intelligence Scale, selecting 2 specific subtests: block design and vocabulary.
Neuroimaging Assessment.
Magnetic resonance imaging (MRI) scans were acquired at baseline, less than 3 months after the onset of psychotic symptoms. MRIs were performed with 5 different scanners: 2 Siemens Symphony, 2 General Electric Signa, and one Philips ACS Gyroscan, all 1.5T. Data were collected from each center and processed at one site. Sequences were acquired in axial orientation for each subject, a T1-weighted 3D gradient echo (voxel size 1 × 1 × 1.5 mm) and a T2-weighted Turbo-Spin-Echo (voxel size 1 × 1 × 3.5 mm). MRI images were processed using locally developed software incorporating a variety of image processing and quantification tools.67,68 To obtain volume measurements of the main brain lobes, a method for semiautomated segmentation of the brain based on the Talairach proportional grid system was used.59,60 The regions of interest included in the analysis were the frontal, parietal, and temporal lobes.59 Volume of whole brain GM and that of frontal, parietal, temporal, and occipital lobes were obtained for each hemisphere. These areas were included due to the extensive deficits in GM observed in studies in first episodes of early-onset psychoses.44,69 Full details on the neuroimaging methodology are reported elsewhere.70
Baseline Assessment.
The patients in this study had their first lifetime acute psychotic episode when recruited. The majority of the patients (83%) were admitted to the hospital at the time of first evaluation. A baseline assessment was performed shortly after admission (either in the inpatient unit or outpatient clinic) or at the time of first assessment for those not hospitalized, except for the SUMD scale and the neuropsychological assessment, which were administered prior to discharge or at the time of best mental state during the episode.
Longitudinal Assessment.
Six months, 1 year, and 2 years after the baseline visit, the clinical, insight, and functional outcome assessments were repeated. The type of psychosis was determined after the 2-year administration of the K-SADS. Taking into account the last reliable diagnosis available, the sample was divided into a group of SSDs (including schizophrenia and schizophreniform disorders)71 and a group of non-SSDs.
Statistical Analysis
We performed repeated measures tests to analyze change in insight and other clinical variables in each of the diagnostic groups (SSD and non-SSD). We then cross-sectionally compared both groups. A Kolmogorov–Smirnov test was used to test for the normality of variables. Age, DUP, CGI, and SUMD subscales did not follow a normal distribution (except for insight into having psychosis symptoms). The rest of the variables were normally distributed. Therefore, nonparametric association tests were used to examine the relationship between insight dimensions and socio-demographic variables, premorbid adjustment, and psychopathology. Mann–Whitney tests were used to evaluate the differences in insight measurements between the diagnostic groups (24-month diagnosis). Repeated-measures t-tests (for the PANSS and Hamilton) and Wilcoxon rank tests (for the SUMD) were used to assess the changes in variables over time. Spearman correlations were used to assess the relationship between insight and psychopathological, neurocognitive, and neuroimaging scores.
A series of analyses (stepwise linear regression) were performed to assess which state or trait characteristics predicted the different domains of insight over the long term. The variables included in the models were those expected from the literature to predict the assessed dependent variable or those that had correlations with the dependent variable in the univariate analysis with a significance level of P < .25.72 Thus, the dependent variables were the different domains of insight at 2 years and their independent variables were: diagnosis of SSD, gender, baseline executive functioning (devised z-score based composite index), estimated IQ, age, premorbid adjustment in infancy (infancy subscale of the PAS scale), DUP, general functioning and SUMD at 1 year, and negative symptoms (negative subscale of the PANSS) and depressive symptoms (HRSD) at 6 months (due to the fact that, in this sample, insight was very dependent on acute symptomatology at baseline and that after 6 months, the symptomatology did not further improve).6 We included the measure of insight into having a disorder at 1 year because insight improved over the first year (see later in the text and table 2). Baseline left and right frontal and parietal GM were also included in the models.
Table 2.
Insight Means Over Time
| Baseline | 6 Months | 1 Year | 2 Years | Baseline–1 Year, Z, P | 1 Year–2 Years, Z, P | ||
| SSD | SUMD1 | 3.38 ± 1.51 (52) | 2.74 ± 1.36 (50) | 2.57 ± 1.45 (46) | 2.80 ± 1.48 (40) | −2.999, <.001 | −2.269, .02 |
| SUMD2 | 3.23 ± 1.51 (52) | 2.42 ± 1.30 (48) | 2.16 ± 1.36 (43) | 2.36 ± 1.51 (39) | −3.578, <.001 | −1.936, .05 | |
| SUMD3 | 3.52 ± 1.50 (52) | 3.16 ± 1.41 (49) | 2.67 ± 1.47 (45) | 2.80 ± 1.50 (40) | −3.052, <.01 | ||
| TOT1 | 3.26 ± 1.46 (52) | 3.34 ± 1.27 (38) | 3.06 ± 1.25 (29) | 3.54 ± 1.02 (22) | −1.939, .05 | ||
| TOT2 | 1.98 ± 1.73 (52) | 2.92 ± 0.87 (26) | 2.67 ± 0.75 (19) | 3.13 ± 0.85 (12) | |||
| Non-SSD | SUMD1 | 3.31 ± 1.54 (54) | 2.00 ± 1.38 (48) | 1.89 ± 1.28 (44) | 1.91 ± 1.40 (34) | −3.741, <.001 | |
| SUMD2 | 2.91 ± 1.62 (54) | 2.20 ± 1.45 (49) | 1.90 ± 1.26 (42) | 1.82 ± 1.33 (33) | −2.438, .01 | ||
| SUMD3 | 3.37 ± 1.40 (54) | 2.33 ± 1.42 (49) | 2.16 ± 1.31 (44) | 1.97 ± 1.35 (34) | −3.671, <.001 | ||
| TOT1 | 2.3 ± 1.66 (54) | 3.11 ± 1.50 (31) | 2.41 ± 1.10 (22) | 2.91 ± 1.38 (14) | |||
| TOT2 | 1.40 ± 1.39 (54) | 2.43 ± 1.31 (19) | 2.62 ± 1.13 (20) | 2.64 ± 1.12 (11) | −2.833, <.01 |
Note: Results are presented as means and SDs. Wilcoxon rank tests. Sample size in parentheses. Results < 0.1 are shown. SUMD1, general awareness of a mental disorder; SUMD2, the effects of medication on the disorder; SUMD3, general understanding of the consequences of the disorder (SUMD3); TOT1, awareness of specific psychotic symptoms; SUMD Tot2, attribution of symptoms. Abbreviations are explained in the first footnote to table 1.
Results
Sample
The sample consisted of 110 adolescents, 9–17 years of age, with a first episode of nontoxic psychosis. Table 1 presents socio-demographic and clinical characteristics of the sample at baseline. The attrition rate was 24.55% at 2 years. We compared the group that completed the follow up visits as established with those unavailable at 2 years, and there were no differences in baseline socio-demographic or clinical characteristics between them with the exception that the group lost for follow up had a slight better executive functioning than the other group (P = 0 < .01). At least some of the variables were available for 110 patients at baseline, 99 at 6 months, 92 at 1 year, and 83 at 2 years. The diagnosis used was the last one available.
Table 1.
Subject Sociodemographic, Clinical, and Brain Volumes Baseline Characteristics
| N = 110 | SSD, (n = 53) | Non-SSD, (n = 57) | Test | P |
| Gender (male/female) | 39/14 | 35/22 | χ2 = 1.85 | .12 |
| Race (caucasian/others) | 46/7 | 47/10 | χ2 = 2.51 | .35 |
| Mean (SD) | Mean (SD) | |||
| Age (years) | 15.43 (1.95) | 15.61 (1.58) | U = 1498.5 | .94 |
| DUP (days) | 66.75 (54.654) | 63.82 (49.98) | U = 1505.7 | .98 |
| Parental studies (years) | 11.92 (4.15) | 11.02 (3.81) | t = 0.17 | .25 |
| Estimated IQ | 80.89 (16.95) | 80.70 (18.16) | t = −0.05 | .96 |
| DLPC | −0.72 (0.95) | −0.58 (0.77) | t = −0.87 | .38 |
| Executive functioning | 0.865 (0.82) | −0.85 (0.78) | t = −0.13 | .96 |
| PAS infancy (0–24) | 7.81 (4.84) | 6.74 (4.75) | t = 1.17 | .24 |
| GAF (0–100) | 33.25 (15.82) | 33.72 (13.99) | t = −0.17 | .87 |
| CGI (1–7) | 5.62 (1.04) | 5.54 (1.01) | U = 1428.5 | .72 |
| PANSS positive (7–49) | 23.47 (5.76) | 24.28 (7.15) | t = −0.650 | .52 |
| PANSS negative (7–49) | 20.96 (8.58) | 19.14 (9.03) | t = 1.083 | .28 |
| PANSS total score (30–210) | 88.26 (17.46) | 89.67 (22.28) | t = −0.36 | .71 |
| Hamilton (0–65) | 15.77 (7) | 20.75 (9.57) | t = −3.097 | <.01 |
| CGI-S (1–4) | 1.22 (0.60) | 1.35 (0.79) | U = 1416.5 | .4 |
| ICV (cc) | 1521.6 (155.8) | 1490.70 (152.86) | t = 0.95 | .34 |
| LFGM (cc) | 78.8 (8.1) | 78.52 (8.13) | t = 0.16 | .87 |
| RFGM (cc) | 81.3 (8.3) | 80.60 (8.37) | t = 0.41 | .68 |
| LPGM (cc) | 65.3 (7.6) | 65.07 (7.91) | t = 0.13 | .89 |
| RPGM (cc) | 65.5 (7.6) | 65.18 (7.95) | t = 0.20 | .84 |
| LTGM (cc) | 84.2 (10.2) | 80.80 (9.12) | t = 1.67 | .09 |
| RTGM (cc) | 83.1 (10.0) | 80.57 (9.48) | t = 1.22 | .23 |
| OGM (cc) | 97.1 (14.8) | 94.22 (14.29) | t = 0.94 | .35 |
Note: Chi-square test for categorical variables; t-test for normally distributed continuous variables, and Mann–Whitney U for nonnormal continuous variables. Possible ranges in parentheses after the variable name. SSD, schizophrenia spectrum disorder; non-SSD, nonschizophrenia spectrum disorder; DLPC, dorsolateral prefrontal cortex; IQ, intelligence quotient; PAS, premorbid adjustment scale; GAF, global assessment functioning; CGI, clinical general impression; PANSS, Positive and Negative Syndrome Scale; ICV, intracranial volume; LFGM, left frontal gray matter; RFGM, right frontal gray matter; LPGM, left parietal gray matter; RPGM, right parietal gray matter; LTGM, left temporal gray matter; RTGM, right temporal gray matter; OGM, occipital gray matter; DUP, duration of untreated psychosis.
Within the SSD (total of 53 patients), 44 had a diagnosis of schizophrenia and 9 of schizophreniform disorder. The non-SSD group was a heterogeneous group composed of 57 patients, including 22 patients with bipolar disorder, 16 patients with Psychosis-NOS, 9 patients with a depressive disorder with psychotic symptoms, 3 patients with a brief psychotic disorder, and 7 patients with schizoaffective disorder.
Extended demographic data are reported elsewhere.45 The 2 groups were similar in their socio-demographic characteristics, their baseline cognitive functioning, and the severity of their psychopathology (for details, see table 1). Only depressive symptomatology was significantly less severe in the SSD group in comparison with the non-SSD group.
Of the baseline socio-demographic variables, age, gender, and ethnicity correlated with some aspects of insight at 2 years. Parental education did not. In the whole group of patients, the older the patient, the better the insight into having a disorder, into the consequences of the disorder, into having psychotic symptoms, and their correct attribution (Spearman rho SUMD1 = −0.263, P = .02; SUMD2 rho = −0.211, P = .08; SUMD3 rho = −0.270, P = .02; SUMD Tot1 rho = −0.341, P = .04; SUMD Tot2 rho = −0.420, P = .05). Females had better insight into having psychotic symptoms, only in the non-SSD group (Mann–Whitney U = 7.5, P = .02), and Caucasians had better attribution of the origin of the symptoms in the whole group (U = 9.00, P = .03).
Course of Insight Over the First 2 Years of Psychosis
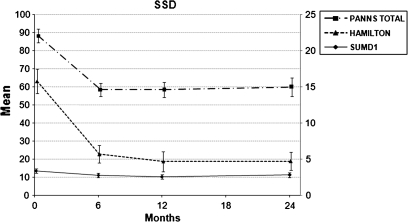
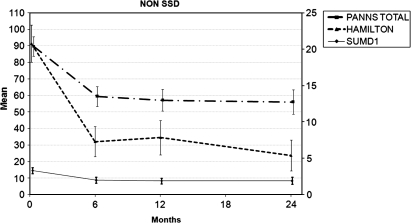
In the SSD group, insight into having a disorder, the need for treatment, the consequences of the disorder, and insight into psychotic symptoms all improved over the first year after the first episode (see table 2 and figure 1) with deterioration in the 2 first subscales between the first and second year. In the non-SSD group, insight into having a disorder, the need for treatment, and the consequences of the disorder also improved during the first year. After that, no further significant change was observed (see figure 2).
Fig. 1.
Insight into Having a Disorder (Scale to Assess Unawareness of Mental Disorder [SUMD]1), Depressive Symptomatology (Hamilton Score), and Total Psychopathology (Total Positive and Negative Symptom Scale [PANSS] Score), in Schizophrenia Spectrum Disorder Patients.
Mean and SE represented. Axis to the left represents the score in the PANSS. Axis to the right represents score in SUMD1 and Hamilton.
Fig. 2.
Insight into Having a Disorder (Scale to Assess Unawareness of Mental Disorder [SUMD]1), Depressive Symptomatology (Hamilton Score), and Total Psychopathology (Total Positive and Negative Symptom Scale [PANSS] Score), in Non-Schizophrenia Spectrum Disorder Patients.
Mean and SE represented. Axis to the left represents the score in the PANSS. Axis to the right represents score in SUMD1 and Hamilton.
Long-term Insight and Socio-Demographic, Cognitive, and Clinical Correlates
At 2 years, insight into having a disorder and the consequences of the disorder were worse in the group of schizophrenia patients compared with the non-SSD patients (U = 435.5, P < .01 and U = 449.5, P < .01, respectively).
Between the first and second year of illness, no change was found in the severity of psychotic symptoms (measured by the PANSS) in any of the groups. No change was observed in general functioning. Changes in psychopathological symptoms over the first year of the disorder are reported elsewhere.6
Table 3 shows the correlations between insight at 2 years and concurrent and 1-year psychopathological and global functioning and insight at 2 years and baseline psychopathological and individual characteristics (age, IQ, cognitive functioning, and premorbid adjustment) by group.
Table 3.
Long-term Insight Correlations with Concurrent Psychopathology and Baseline Individual Characteristics and Psychopathology
| SSD 2 Year |
Non-SSD 2 Year |
||||||||||
| SUMD 1 | SUMD 2 | SUMD 3 | TOT 1 | TOT 2 | SUMD 1 | SUMD 2 | SUMD 3 | TOT 1 | TOT 2 | ||
| 2 Years | PANSS positive | 0.445** | 0.422** | 0.544** | 0.373 | −0.157 | 0.574** | 0.509** | 0.530** | 0.423 | 0.320 |
| PANSS negative | 0.413** | 0.342* | 0.460** | 0.528* | 0.507 | 0.067 | 0.078 | 0.114 | −0.012 | 0.465 | |
| PANSS total | 0.559** | 0.450** | 0.601** | 0.668** | 0.38 | 0.500** | 0.482** | 0.410* | 0.352 | 0.444 | |
| DLPC | 0.065 | 0.114 | −0.015 | 0.587* | −0.183 | −0.383* | −0.483** | −0.479** | −0.792** | −0.473 | |
| GAF | −0.433** | −0.281 | −0.571** | −0.399 | −0.205 | −0.484** | −0.634** | −0.458** | −0.343 | −0.429 | |
| 1 year | PANSS positive | 0.365* | 0.293 | 0.390* | 0.118 | −0.015 | 0.328 | 0.236 | 0.224 | 0.175 | 0.440 |
| PANSS negative | 0.388* | 0.274 | 0.440** | 0.224 | −0.215 | 0.204 | 0.180 | 0.123 | 0.354 | 0.480 | |
| PANSS total | 0.538** | 0.409* | 0.595** | 0.378 | −0.035 | 0.299 | 0.241 | 0.192 | 0.114 | 0.438 | |
| GAF | −0.386* | −0.174 | −0.561** | −0.297 | −0.191 | −0.568** | −0.659** | −0.442** | −0.321 | −0.730* | |
| Baseline | AGE | −0.490** | −0.332* | −0.460** | −0.385 | −0.165 | 0.072 | −0.032 | −0.038 | −0.325 | −0.723* |
| DLPC | 0.087 | 0.187 | −0.018 | −0.019 | −0.492 | −0.272 | −0.364* | −0.283 | −0.679** | −0.563 | |
| Executive function | −0.383* | −0.272 | −0.313* | −0.391 | 0.088 | −0.003 | −0.074 | −0.051 | −0.529 | −0.105 | |
| PAS infancy | 0.345* | 0.164 | 0.392* | −0.002 | −0.094 | 0.041 | 0.071 | 0.012 | 0.369 | 0.641* | |
| DUP | 0.411** | 0.413** | 0.379** | 0.516** | 0.374 | 0.296 | 0.354* | 0.340* | −0.181 | 0.456 | |
| IQ | −0.455** | −0.237 | −0.467** | −0.497* | 0.27 | −0.095 | 0.002 | −0.186 | −0.109 | −0.130 | |
In SSD, concurrent severity of psychotic symptomatology positively correlated with long-term (2 year) insight (the more severe the psychotic symptoms, the worse the insight). Both concurrent and 1-year better global functioning correlated with better insight into having a mental disorder and its consequences. As in the whole group of patients, the younger the patient at baseline, the worse the insight at 2 years. Poorer baseline executive functioning, lower IQ, and poorer premorbid infancy adjustment correlated with poorer insight into having a disorder and its consequences, at the 2-year follow-up. Regarding insight into having symptoms of a psychotic disorder (SUMD Tot1), SSD patients had poorer insight if they had more severe concurrent total or negative psychopathology and lower baseline IQ. No correlations were found between correct attribution of symptoms and any of the variables measured.
Within the non-SSD group, some insight domains at 2 years correlated with concurrent positive and total psychopathology, better concurrent and 1-year global functioning, and better DLPC functioning (both baseline and at 2 years). Non-SSD patients had poorer insight into having symptoms of a psychotic disorder if they had poorer baseline or 2-year prefrontal cognitive functioning (DLPC measure). No correlations were found between correct attribution of symptoms and any of the variables measured.
Insight and Depression
Depressive symptoms, as measured by the Hamilton scale, improved over the first 6 months after the first episode in both groups of patients (t = 9.83, P < .001 in SSD and t = 7.543, P < .001 in non-SSD) and did not change thereafter (see figures 1 and 2). There were positive correlations between depressive symptoms and negative symptoms, in SSD at 1 and 2 years (Pearson rho = 0.463, P = .001 and 0.626, P < .001, respectively) and in non-SSD at baseline, 1 year, and 2 years (Pearson rho = 0.273, P = .04 and 0.598, P < .001 and 0.369, P = .02, respectively).
In SSD patients, depressive symptoms correlated with some aspects of insight at 6 months and 1 year but not at 2 years (see table 4); the higher the patient’s depression score, the worse the insight. These correlations did not occur in the group of non-SSD patients. Figures 1 and 2 show the course of insight into having a mental disorder and depressive symptomatology (together with the PANSS total score) over time in the SSD and non-SSD groups.
Table 4.
Correlations of Insight and Depressive Symptoms at Each Point in Time
| SSD |
Non-SSD |
|||||||||
| SUMD 1 | SUMD 2 | SUMD 3 | TOT 1 | TOT 2 | SUMD 1 | SUMD 2 | SUMD 3 | TOT 1 | TOT 2 | |
| Baseline Hamilton | 0.164 | 0.341* | 0.114 | 0.087 | −0.161 | 0.305* | 0.433** | 0.219 | 0.147 | 0.028 |
| 6-month Hamilton | 0.305* | 0.433** | 0.219 | 0.147 | 0.028 | 0.075 | 0.223 | 0.016 | −0.311 | 0.075 |
| 1-year Hamilton | 0.334* | 0.373* | 0.456** | 0.336 | −0.217 | 0.238 | 0.343 | 0.099 | 0.303 | 0.591 |
| 2-year Hamilton | 0.261 | 0.206 | 0.303 | 0.171 | 0.261 | 0.292 | 0.264 | 0.028 | −0.259 | 0.647 |
Neuroimaging and Insight
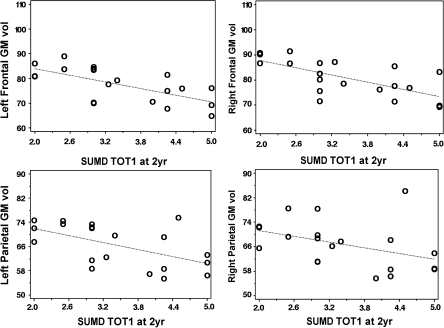
Brain images were available for 91 patients. No significant relationship between volumetric variables and insight measures (any of the 5 domains studied) were found considering the whole group of patients. However, in SSD patients, significant partial Spearman correlations, removing the effect of age on insight and brain volumes, were observed in the GM volume of frontal and parietal lobes, bilaterally (left frontal Spearman rho = −0.575, P < .01; right frontal Spearman rho = −0.611, P < .01; left parietal Spearman rho = −0.495, P = .03; and right parietal Spearman rho = −0.46, P = .04). Thus, more frontal and parietal GM at baseline was correlated with better insight into having psychotic symptoms at 2 years (see figure 3). No other aspects of insight correlated with the structural measurements.
Fig. 3.
Spearman Partial Correlations between Baseline Frontal and Parietal (Left and Right) Gray Matter (GM) and Insight into Having Psychosis Symptoms at 2 Years (SUMDTOT1).
Multivariate Analysis
Table 5 shows the explanatory variables for the different models that used different insight domains at 2 years as dependent variables.
Table 5.
Explanatory Models in SSD and Non-SSD Patients, With Insight Domains at 2 Years as Dependent Variables
| Adjusted |
Standardized |
Adjusted |
Standardized |
||||||
| SSD | R2 | F (df) | Beta | P | Non-SSD | R2 | F | Beta | P |
| Insight into having a mental illness | 0.598 | 16.894 (32) | <.001 | Insight into having a mental illness | 0.268 | 8.690 (21) | <.01 | ||
| 1-year SUMD1 | 0.608 | <.001 | 1-year GAF | −0.550 | <.01 | ||||
| IQ | −0.316 | <.01 | |||||||
| DUP | −0.265 | .03 | |||||||
| Insight into the need of treatment | 0.167 | 7.208 (31) | .01 | Insight into the need of treatment | 0.501 | 11.041 (20) | <.01 | ||
| DUP | 0.440 | .01 | 1-year GAF | −0.527 | <.01 | ||||
| 1-year SUMD1 | 0.431 | .01 | |||||||
| Insight into the consequences of illness | 0.694 | 15.540 (32) | <.001 | Insight into the consequences of illness | 0.415 | 8.458 (21) | <.01 | ||
| 1-year SUMD1 | 0.609 | <.001 | 1-year GAF | −0.757 | <.01 | ||||
| IQ | −0.350 | <.01 | 6-month Hamilton | −0.434 | .03 | ||||
| RPGM | 0.594 | <.01 | |||||||
| DUP | 0.286 | <.01 | |||||||
| LPGM | −0.376 | .04 | |||||||
| Insight into having psychotic symptoms | 0.795 | 15.576 (15) | <.001 | Insight into having psychotic symptoms | .436 | 7.970 (9) | .02 | ||
| IQ | −0.658 | <.001 | Gender | 0.706 | .02 | ||||
| 1-year SUMD1 | 0.738 | <.001 | |||||||
| LFGM | 0.406 | <.01 | |||||||
| LPGM | 0.402 | .01 | |||||||
| Attribution of psychotic symptoms | 0.436 | 7.970 (9) | .02 | Attribution of psychotic symptoms | 0.930 | 31.953 (7) | <.01 | ||
| Gender | 0.706 | .02 | PAS infancy | 0.769 | <.01 | ||||
| 6-month PANSS negative | 0.612 | <.01 | |||||||
| Gender | 0.385 | .03 | |||||||
Insight into having a mental disorder at 1 year, DUP, and IQ emerged as the most significant contributors to explain insight domains at 2 years in SSD patients. In addition, left and right parietal GM added some explanatory effect on the SUMD3 and left frontal and parietal GM on SUMDT1. In SSD patients, IQ, DUP, and SUMD1 at 1 year explained 60% of the variance of having a mental illness; these variables, together with the right and left parietal GM volume, explained 70% of the variance of insight into the consequences of illness and IQ, SUMD1 at 1 year, together with left frontal and left parietal GM volumes, explained 80% of the variance of insight into having specific psychotic symptoms. Only DUP entered in the model to explain insight into the need of treatment (17% of the variance) and only gender in the model of the correct attribution of symptoms (44% of the variance).
In non-SSD, GAF at 1 year explained part of the main insight domains. Gender explained 44% of the variance of insight into psychotic symptoms and together with PAS, and the negative score of the PANSS at 6 months explained 93% of correct attribution of symptoms. Taking the whole group of patients together, diagnosis did not contribute to explain insight at 2 years.
Discussion
In this 2-year longitudinal study of insight after a FEP occurring in adolescents, we have shown that: (1) Older age is associated with better insight at 2 years; (2) Insight at 2 years is poorer in adolescents with SDD than in subjects with non-SSD psychoses; (3) The improvement of most aspects of insight found in the initial stages of the disorder does not continue after 1 year. In fact, insight into having a mental disorder and into the need for treatment deteriorates between years 1 and 2 in SSD patients; (4) Different correlates of long-term insight were found in SSD and non-SSD patients. The more severe the psychotic symptomatology, the poorer the insight in both groups. Worse depressive symptoms were associated with poorer insight at baseline in both groups. However, depressive symptoms were associated with poorer insight after baseline only in SSD subjects (at 6 months and 1 year). In addition, age, IQ, executive functioning, and premorbid adjustment were associated with insight only in SSD group; (5) Insight at 1 year, DUP and IQ explain a moderate amount of the variance in different aspects of insight at 2 years, in SSD patients. Frontal and parietal GM volumes also contributed to explain part of the long-term insight in this group. Different clinical aspects explained part of the insight at 2 years in non-SSD, general functioning at 1 year being the most pervasive across different domains. Gender explained part of the insight into psychotic symptoms in non-SSD patients and their correct attribution in both groups.
Trait and State Attributes of Insight
The age range of the patients in the present study coincides with a stage in life important for the construction of personal identity. The impact of a psychotic episode on the attribution of mental states to others and oneself at this stage is largely unknown.73,74 Older age, female gender, and white ethnicity were also shown to be associated with more insight in a sample of more than 250 patients experiencing their FEP.25 In regards to gender, typically girls have been shown to have better mentalizing ability than boys75,76 and many studies have shown the better performance of women and/or girls in phenomena, such as autobiographical memory,77,78 which is associated with concepts like self-awareness or theory of mind, also shown to explain a substantial amount of variance of insight in some studies.79 The result that insight is poorer in SSD patients than in non-SSD patients is in agreement with the literature.1,6,21,22,26,80 We had previously shown that insight improved over the early phases of the illness, in parallel with psychopathological improvement and that schizophrenia patients had poorer insight than patients with bipolar disorder at 6 and 12 months but not at baseline.6 Contrary to our hypothesis, insight worsens after 1 year in the case of SSD patients. How the intensity of follow-up, presence of psychoeducational interventions, chronicity, medication, treatment adherence, and methodological issues related to the studies, such as attrition, influence the course of insight need further exploration. In any event, the fact that insight improves during the first year after the FEP, and the predictive value of insight at 1 year for insight at 2 years suggests that the first year is crucial for improving later insight and possibly general functioning (as insight correlates with general functioning at 2 years).
As shown in many other studies,6,25,36 multiple factors contribute to insight. Positive psychotic psychopathology is a major contributor to insight, irrespective of the diagnosis. This relationship has been shown in many studies22. In addition, in SSD patients, insight is poorer when baseline cognitive function is worse, negative symptoms are more severe, and in patients with poorer premorbid adjustment and longer DUP. The relationship between insight and neurocognitive functioning and with negative symptoms has also been shown in adults with schizophrenia.9,17,28,81,82 Also, better premorbid adjustment has previously been associated with better insight.6,26,82 Poor premorbid adjustment, negative symptoms, and poor neurocognitive functioning have been classically considered intrinsic to schizophrenia and probably form part of the disorder itself. In fact, the prediction analysis shows that cognitive functioning is a better predictor of long-term insight than the diagnosis itself. The cumulative effect of moderators of poor insight in SSD patients (such as poor premorbid adjustment and negative symptoms) may explain why younger age is associated with poorer insight only in SSD patients. In addition, some of the negative results in the non-SSD group could be due to the heterogeneity of the patients included in the latter.
Depression and Insight
The relationship between depressive symptoms and insight is complex. Some authors83,84 have not found an association between insight and depression, while others have shown an inverse relationship between depression and insight (more insight, less depression), as in this study in the SSD group. This was also shown at admission but not at discharge in a group of acutely ill patients with schizophrenia85 and also, in a modest association, in a group of chronic patients.86 Our group also showed such a relationship in the early stages of FEP.87 Owen et al88 argued that severe depressive symptomatology could be associated with lack of insight via irrationality. Most previous studies, however, have shown that better insight is associated with more depressive symptoms in first episodes.1,12,21,22,24,25,28,37,38,63,82 (an explanation being that better insight would be secondary to the hyperrealism that frequently accompanies depressive symptoms). In fact, the meta-analysis by Mintz22 showed positive associations between better insight and more depressive symptomatology (with small effect sizes), for all domains of insight. An association between more depressive symptoms and more insight into psychotic symptoms7,87 has also been shown in a sample of adolescents. However, the subjects in that study were assessed soon after hospital admission (less than 1 month) and were therefore in the acute psychosis stage, while our study also explored insight when patients were stable (2 years after the first episode). In our sample, no difference was found in depressive symptoms between SSD and non-SSD, when subjects were in the acute psychosis stage (see table 1), although the SSD group scored higher on the Hamilton scale, probably because a significant number of patients in the non-SSD group were manic at baseline (there were 22 bipolars in this group, most of them manic). As shown in table 4, our results are consistent in showing higher Hamilton scores related to less insight (higher insight scores) in SSD patients. However, the scores for the Hamilton scale are low (mean 4.70 ± 4.95 for SSD, 5.39 ± 6.52 for non-SSD at 2 years),89 and there is a possibility that scores were produced by symptoms more related to negative schizophrenia symptoms than to depression (such as apathy or lack of volition). However, a correlation was found between negative and depressive symptoms both in SSD and non-SSD while the association between poorer insight and depressive symptoms was found only in SSD. The more severe negative symptomatology in SSD could be one mediator. The possibility that cognitive dysfunction was also an intermediate mechanism was tested and discarded, as cognitive function did not correlate with depressive symptomatology. Given the discrepancies with the literature, we further assessed the temporal relationship between insight and depression, which is an aspect basically neglected in previous studies.22 Cross-sectionally, depressive symptoms were more intense in poor insight patients at the 6-month and 1-year assessments (table 4). We also looked at the possible association between the change in insight into having a mental disorder with the change in depressive score, in the periods from 6 months to 1 year and from 1 to 2 years, and there was no such correlation.
The question about the relationship between depressive symptoms and insight in schizophrenia remains open. The nature of depressive symptoms in stable schizophrenia and how to assess them needs to be further assessed. Studies have used different instruments to study depressive symptoms in schizophrenia; some of them have used just an item from a general scale, such as the PANSS or the Brief Psychiatric Rating Scale. The complexity of the relationship between depressive symptoms and insight and the difficulty discriminating between depressive, negative, and other cognitive difficulties in patients with schizophrenia deserves a specifically targeted study. However, studies that have used the Calgary scale, specifically meant to measure depression in schizophrenia, both in acute and remission stages, have also yielded mixed results.7
Cognitive Functioning and Insight
Regarding cognitive function, general cognitive functioning (estimated IQ) and baseline prefrontal cognitive dysfunction were associated with poorer long-term insight in SSD patients but not in non-SSD patients. Other studies have shown concurrent associations between poorer cognitive performance and insight,10,25,65 but we looked for long-term associations, on the basis that cognitive functioning after a first episode mainly stays stable90 and that insight during acute episodes is highly dependent on psychopathology.6
Early Predictors of Long-term Insight
In addition, the prediction analysis shows that, apart from previous insight, baseline general cognitive functioning and DUP are the best predictors of insight at 2 years, in SSD patients. These factors have been associated with insight in other studies.25,28,36 Our study shows that estimated IQ is a consistent predictor of almost all domains of insight in SSD patients. More GM volume in frontal and parietal lobes also explain part of the different insight domains, although the contribution they make is weak or modest; this is in agreement with previous studies.4,37 Anosognosia, which some consider to be a neurological analog of poor insight,1,27 is associated with lesions in subcortical, parietal lobe, and temporal lobe structures, as well as with frontal lesions.91 The strongest relationship found in the study by Cooke et al4 was between the Awareness of Problems score and the volume of the left precuneus, located in the medial parietal lobe. Shad et al10 propose a model in which the neurological underpinnings of insight would involve a neural network comprising subregions of prefrontal and parietal cortex mediating specific insight deficits into having psychotic symptoms and their attribution.27
The correlation analysis and the lack of predictive validity of diagnosis into insight at 2 years support the idea that the categorical classification of psychosis has important limitations regarding aspects of validity of diagnosis such as the prediction of outcome. Cognitive functioning and previous insight, but not diagnosis, predict long-term insight, which in turn is very much related with general functioning. These data support a dimensional view of psychosis, where insight adds to genetic and other neurobiological data in distributing itself across different psychotic diagnoses, with no further value when establishing nosological cutoffs.
Altogether, our neurocognitive and neuroimaging data support involvement of the frontal and parietal lobes. In agreement with our previous findings,6 insight appears to be a complex symptom that, at least in schizophrenia, depends both on current psychopathological (state) and long-term individual characteristics, some of which are unrelated to the disease (age, gender, and ethnicity) and others probably partially dependent on the individual and the disorder itself (past adjustment, IQ, brain structure, and cognitive functioning). The fact that this sample is younger than most samples that have been used to assess the correlations of insight may explain part of the differences in the results, particularly regarding depressive symptoms and insight.
Limitations
The main limitations of the study are that: (1) we have not studied aspects of personality traits such as self-awareness, self-concept, autobiographical memory, adjustment abilities, or coping styles, which could also influence the development of insight after a FEP; (2) we had an attrition rate of 25% over 2 years (quite standard for this type of study); (3) we have not conducted our own factor analysis study of the cognitive measures to define an EF separated from the others, which would have been the ideal approach; we rather followed the literature (including our own studies) to create our cognitive indexes, due to space constraints and the fact that the focus is set on insight and not on cognition. The main advantages of the study sample are its early age and the longitudinal follow-up. Adolescence is a developmental period in which cognitive, personality, and brain development are still in progress. In addition to diagnosis and psychopathological status, we have been able to assess the impact on the course of insight of some individual characteristics, such as demographics, cognitive functioning, and brain maturation indicators.
Funding
CIBER de Salud Mental (CIBERSAM); Instituto de Salud Carlos III; Spanish Ministry of Science and Innovation.
Acknowledgments
The Authors have declared that there are no conflicts of interest in relation to the subject of this study.
References
- 1.Amador XF, Flaum M, Andreasen NC, et al. Awareness of illness in schizophrenia and schizoaffective and mood disorders. Arch Gen Psychiatry. 1994;51:826–836. doi: 10.1001/archpsyc.1994.03950100074007. [DOI] [PubMed] [Google Scholar]
- 2.Amador XF, Strauss DH, Yale SA, et al. Assessment of insight in psychosis. Am J Psychiatry. 1993;150:873–879. doi: 10.1176/ajp.150.6.873. [DOI] [PubMed] [Google Scholar]
- 3.Peralta V, Cuesta MJ. Lack of insight: its status within schizophrenic psychopathology. Biol Psychiatry. 1994;36:559–561. doi: 10.1016/0006-3223(94)90620-3. [DOI] [PubMed] [Google Scholar]
- 4.Cooke MA, Fannon D, Kuipers E, et al. Neurological basis of poor insight in psychosis: a voxel-based MRI study. Schizophr Res. 2008;103:40–51. doi: 10.1016/j.schres.2008.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Osatuke K, Ciesla J, Kasckow JW, Zisook S, Mohamed S. Insight in schizophrenia: a review of etiological models and supporting research. Compr Psychiatry. 2008;49:70–77. doi: 10.1016/j.comppsych.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 6.Parellada M, Fraguas D, Bombin I, et al. Insight correlates in child- and adolescent-onset first episodes of psychosis: results from the CAFEPS study. Psychol Med. 2009;39:1433–1445. doi: 10.1017/S0033291708004868. [DOI] [PubMed] [Google Scholar]
- 7.Schwartz-Stav O, Apter A, Zalsman G. Depression, suicidal behavior and insight in adolescents with schizophrenia. Eur Child Adolesc Psychiatry. 2006;15:352–359. doi: 10.1007/s00787-006-0541-8. [DOI] [PubMed] [Google Scholar]
- 8.Aleman A, Agrawal N, Morgan KD, David AS. Insight in psychosis and neuropsychological function: meta-analysis. Br J Psychiatry. 2006;189:204–212. doi: 10.1192/bjp.189.3.204. [DOI] [PubMed] [Google Scholar]
- 9.Laroi F, Fannemel M, Ronneberg U, et al. Unawareness of illness in chronic schizophrenia and its relationship to structural brain measures and neuropsychological tests. Psychiatry Res. 2000;100:49–58. doi: 10.1016/s0925-4927(00)00063-9. [DOI] [PubMed] [Google Scholar]
- 10.Shad MU, Tamminga CA, Cullum M, Haas GL, Keshavan MS. Insight and frontal cortical function in schizophrenia: a review. Schizophr Res. 2006;86:54–70. doi: 10.1016/j.schres.2006.06.006. [DOI] [PubMed] [Google Scholar]
- 11.Amador XF, Gorman JM. Psychopathologic domains and insight in schizophrenia. Psychiatr Clin North Am. 1998;21:27–42. doi: 10.1016/s0193-953x(05)70359-2. [DOI] [PubMed] [Google Scholar]
- 12.Markova IS, Berrios GE. The meaning of insight in clinical psychiatry. Br J Psychiatry. 1992;160:850–860. doi: 10.1192/bjp.160.6.850. [DOI] [PubMed] [Google Scholar]
- 13.Cuesta MJ, Peralta V. Lack of insight in schizophrenia. Schizophr Bull. 1994;20:359–366. doi: 10.1093/schbul/20.2.359. [DOI] [PubMed] [Google Scholar]
- 14.Carpenter WT, Jr., Strauss JS, Bartko JJ. Flexible system for the diagnosis of schizophrenia: report from the WHO International Pilot Study of Schizophrenia. Science. 1973;182:1275–1278. doi: 10.1126/science.182.4118.1275. [DOI] [PubMed] [Google Scholar]
- 15.Lysaker PH, Bryson GJ, Lancaster RS, Evans JD, Bell MD. Insight in schizophrenia: associations with executive function and coping style. Schizophr Res. 2003;59:41–47. doi: 10.1016/s0920-9964(01)00383-8. [DOI] [PubMed] [Google Scholar]
- 16.Martens WH. The role of self-complexity in reducing harmful insight among persons with schizophrenia. Theoretical and therapeutic implications. Am J Psychother. 2009;63:53–68. doi: 10.1176/appi.psychotherapy.2009.63.1.53. [DOI] [PubMed] [Google Scholar]
- 17.McEvoy JP, Hartman M, Gottlieb D, et al. Common sense, insight, and neuropsychological test performance in schizophrenia patients. Schizophr Bull. 1996;22:635–641. doi: 10.1093/schbul/22.4.635. [DOI] [PubMed] [Google Scholar]
- 18.Arango C, Adami H, Sherr JD, Thaker GK, Carpenter WT., Jr Relationship of awareness of dyskinesia in schizophrenia to insight into mental illness. Am J Psychiatry. 1999;156:1097–1099. doi: 10.1176/ajp.156.7.1097. [DOI] [PubMed] [Google Scholar]
- 19.Lele MV, Joglekar AS. Poor insight in schizophrenia: neurocognitive basis. J Postgrad Med. 1998;44:50–55. [PubMed] [Google Scholar]
- 20.Gilleen J, Greenwood K, David A. Anosognosia in schizophrenia and other neuropsychiatric disorders: similarities and differences. In: Prigatano G, editor. Advances in the Study of Anosognosia. New York, NY: Oxford Psychiatric Press; 2010. pp. 255–290. [Google Scholar]
- 21.Smith TE, Hull JW, Israel LM, Willson DF. Insight, symptoms, and neurocognition in schizophrenia and schizoaffective disorder. Schizophr Bull. 2000;26:193–200. doi: 10.1093/oxfordjournals.schbul.a033439. [DOI] [PubMed] [Google Scholar]
- 22.Mintz AR, Dobson KS, Romney DM. Insight in schizophrenia: a meta-analysis. Schizophr Res. 2003;61:75–88. doi: 10.1016/s0920-9964(02)00316-x. [DOI] [PubMed] [Google Scholar]
- 23.David AS. Illness and insight. Br J Hosp Med. 1992;48:652–654. [PubMed] [Google Scholar]
- 24.Sanz M, Constable G, Lopez-Ibor I, Kemp R, David AS. A comparative study of insight scales and their relationship to psychopathological and clinical variables. Psychol Med. 1998;28:437–446. doi: 10.1017/s0033291797006296. [DOI] [PubMed] [Google Scholar]
- 25.McEvoy JP, Johnson J, Perkins D, et al. Insight in first-episode psychosis. Psychol Med. 2006;36:1385–1393. doi: 10.1017/S0033291706007793. [DOI] [PubMed] [Google Scholar]
- 26.Keshavan MS, Rabinowitz J, DeSmedt G, Harvey PD, Schooler N. Correlates of insight in first episode psychosis. Schizophr Res. 2004;70:187–194. doi: 10.1016/j.schres.2003.11.007. [DOI] [PubMed] [Google Scholar]
- 27.Shad MU, Keshavan MS, Tamminga CA, Cullum CM, David A. Neurobiological underpinnings of insight deficits in schizophrenia. Int Rev Psychiatry. 2007;19:439–448. doi: 10.1080/09540260701486324. [DOI] [PubMed] [Google Scholar]
- 28.Mutsatsa SH, Joyce EM, Hutton SB, Barnes TR. Relationship between insight, cognitive function, social function and symptomatology in schizophrenia: the West London first episode study. Eur Arch Psychiatry Clin Neurosci. 2006;256:356–363. doi: 10.1007/s00406-006-0645-7. [DOI] [PubMed] [Google Scholar]
- 29.Ritsner MS, Blumenkrantz H. Predicting domain-specific insight of schizophrenia patients from symptomatology, multiple neurocognitive functions, and personality related traits. Psychiatry Res. 2007;149:59–69. doi: 10.1016/j.psychres.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 30.Varga M, Magnusson A, Flekkoy K, David AS, Opjordsmoen S. Clinical and neuropsychological correlates of insight in schizophrenia and bipolar I disorder: does diagnosis matter? Compr Psychiatry. 2007;48:583–591. doi: 10.1016/j.comppsych.2007.06.003. [DOI] [PubMed] [Google Scholar]
- 31.Varga M, Babovic A, Flekkoy K, et al. Reduced insight in bipolar I disorder: neurofunctional and neurostructural correlates: a preliminary study. J Affect Disord. 2009;116:56–63. doi: 10.1016/j.jad.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 32.McEvoy JP, Freter S, Everett G, et al. Insight and the clinical outcome of schizophrenic patients. J Nerv Ment Dis. 1989;177:48–51. doi: 10.1097/00005053-198901000-00008. [DOI] [PubMed] [Google Scholar]
- 33.Soyka M, Graz C, Bottlender R, Dirschedl P, Schoech H. Clinical correlates of later violence and criminal offences in schizophrenia. Schizophr Res. 2007;94:89–98. doi: 10.1016/j.schres.2007.03.027. [DOI] [PubMed] [Google Scholar]
- 34.Arango C, Calcedo Barba A, Gonzalez S, Calcedo Ordonez A. Violence in inpatients with schizophrenia: a prospective study. Schizophr Bull. 1999;25:493–503. doi: 10.1093/oxfordjournals.schbul.a033396. [DOI] [PubMed] [Google Scholar]
- 35.Perkins DO. Predictors of noncompliance in patients with schizophrenia. J Clin Psychiatry. 2002;63:1121–1128. doi: 10.4088/jcp.v63n1206. [DOI] [PubMed] [Google Scholar]
- 36.Saravanan B, Jacob KS, Johnson S, et al. Outcome of first-episode schizophrenia in India: longitudinal study of effect of insight and psychopathology. Br J Psychiatry. 2010;196:454–459. doi: 10.1192/bjp.bp.109.068577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Drake RJ, Pickles A, Bentall RP, et al. The evolution of insight, paranoia and depression during early schizophrenia. Psychol Med. 2004;34:285–292. doi: 10.1017/s0033291703008821. [DOI] [PubMed] [Google Scholar]
- 38.Crumlish N, Whitty P, Kamali M, et al. Early insight predicts depression and attempted suicide after 4 years in first-episode schizophrenia and schizophreniform disorder. Acta Psychiatr Scand. 2005;112:449–455. doi: 10.1111/j.1600-0447.2005.00620.x. [DOI] [PubMed] [Google Scholar]
- 39.Rossell SL, Coakes J, Shapleske J, Woodruff PWR, David AS. Insight: its relationship with cognitive function, brain volume and symptoms in schizophrenia. Psychol Med. 2003;33:111–119. doi: 10.1017/s0033291702006803. [DOI] [PubMed] [Google Scholar]
- 40.Flashman LA, Roth RM. Neural correlates of unawareness of illness in psychosis. In: Amador XF, David AS, editors. Insight and Psychosis: Awareness of Illness in Schizophrenia and Related Disorders. 2nd ed. New York, NY: Oxford University Press; 2004. pp. 157–176. [Google Scholar]
- 41.Lappin JM, Morgan KD, Valmaggia LR, et al. Insight in individuals with an At Risk Mental State. Schizophr Res. 2007;90:238–244. doi: 10.1016/j.schres.2006.11.018. [DOI] [PubMed] [Google Scholar]
- 42.Thompson KN, McGorry PD, Harrigan SM. Reduced awareness of illness in first-episode psychosis. Compr Psychiatry. 2001;42:498–503. doi: 10.1053/comp.2001.27900. [DOI] [PubMed] [Google Scholar]
- 43.Sham P, MacLean C, Kendler K. A typological model of schizophrenia based on age at onset, sex and familial morbidity. Acta Psychiatr Scand. 1994;89:135–141. doi: 10.1111/j.1600-0447.1994.tb01501.x. [DOI] [PubMed] [Google Scholar]
- 44.Reig S, Moreno C, Moreno D, et al. Progression of brain volume changes in adolescent-onset psychosis. Schizophr Bull. 2009;35:233–243. doi: 10.1093/schbul/sbm160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Castro-Fornieles J, Parellada M, Gonzalez-Pinto A, et al. The child and adolescent first-episode psychosis study (CAFEPS): design and baseline results. Schizophr Res. 2007;91:226–237. doi: 10.1016/j.schres.2006.12.004. [DOI] [PubMed] [Google Scholar]
- 46.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, (DSM-IV) Washington, DC: American Psychological Association Press; 1994. [Google Scholar]
- 47.Kaufman J, Birmaher B, Brent D, et al. Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry. 1997;36:980–988. doi: 10.1097/00004583-199707000-00021. [DOI] [PubMed] [Google Scholar]
- 48.Soutullo C. Traducción al Español de la Entrevista Diagnóstica: Kiddie-Schedule for Affective Disorders & Schizophrenia. Present & Lifetime Version (K-SADS-PL) (1996) http://www.cun.es/fileadmin/Departamentos/Psiquiatria%20y%20Psicologia%20Medica/PDF/KSADSEsp.pdf (updated September 21, 2010). Accessed September 21, 2010. [Google Scholar]
- 49.Kay SR, Opler LA, Fishbein A. Positive and Negative Syndrome Scale (PANSS) Rating Manual. Social and Behavioral Sciences Documents. San Rafael, CA: Social and Behavioral Sciences Documents; 1987. [Google Scholar]
- 50.Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lindenmayer JP, Czobor P, Alphs L, et al. InterSePT Study Group. The InterSePT scale for suicidal thinking reliability and validity. Schizophr Res. 2003;63:161–170. doi: 10.1016/s0920-9964(02)00335-3. [DOI] [PubMed] [Google Scholar]
- 52.Cannon-Spoor HE, Potkin SG, Wyatt RJ. Measurement of premorbid adjustment in chronic schizophrenia. Schizophr Bull. 1982;8:470–484. doi: 10.1093/schbul/8.3.470. [DOI] [PubMed] [Google Scholar]
- 53.Endicott J, Spitzer RL, Fleiss JL, Cohen J. The Global Assessment Scale. Arch Gen Psychiatry. 1976;33:766–777. doi: 10.1001/archpsyc.1976.01770060086012. [DOI] [PubMed] [Google Scholar]
- 54.Strauss E, Spreen O, Sherman EMS. A Compendium of Neuropsychological Tests: Administration, Norms and Commentary. New York, NY: Oxford University Press; 2006. [Google Scholar]
- 55.Lezak MD, Howieson BH, Loring DW. Neuropsychological Assessment. 4th ed. New York, NY: Oxford University Press; 2004. [Google Scholar]
- 56.Shad M, Muddasani S, Prasad K, Sweeney J, Keshavan M. Insight and prefrontal cortex in first-episode schizophrenia. Neuroimage. 2004;22:1315–1320. doi: 10.1016/j.neuroimage.2004.03.016. [DOI] [PubMed] [Google Scholar]
- 57.Heaton RK, Chelune GJ, Talley JL, Kay GG, Custiss GC. Test de Clasificación de Tarjetas de Wisconsin. Madrid, Spain: TEA Ediciones SA; 2001. [Google Scholar]
- 58.Golden CJ. Stroop Color and Word Test. A Manual for Clinical and Experimental Uses. Wood Dale, IL: Stoelting Co: 1978. [Google Scholar]
- 59.Golden CJ. STROOP: Test de colores y palabras. Madrid, Spain: TEA Ediciones SA; 2001. [Google Scholar]
- 60.Benton AL, Hamsher K. Multilingual Aphasia Examination. Iowa City, IA: AJA Associates, Inc; 1989. [Google Scholar]
- 61.Reitan RM, Wolfson D. The Halstead-Reitan Neuropsychological Test Battery. Tucson, AZ: Neuropsychology Press; 1985. [Google Scholar]
- 62.Strauss E, Elisabeth MS, Spreen O. A compendium of neuropsychological tests. 3rd ed. New York, NY: Oxford University Press; 2006. [Google Scholar]
- 63.De la Serna E, Mayoral M, Baeza I, et al. Cognitive functioning in children and adolescents in their first episode of psychosis: differences between previous cannabis users and nonusers. J Nerv Ment Dis. 2010;198:159–162. doi: 10.1097/NMD.0b013e3181cc0d41. [DOI] [PubMed] [Google Scholar]
- 64.Zabala A, Rapado M, Arango C, et al. Neuropsychological functioning in early-onset first-episode psychosis: comparison of diagnostic subgroups. Eur Arch Psychiatry Clin Neurosci. 2010;260:225–233. doi: 10.1007/s00406-009-0046-9. [DOI] [PubMed] [Google Scholar]
- 65.Conners CK. Connerś Continuous Performance Test Computer Program; User’s Manual. Toronto, Canada: Multi Health Systems Inc; 1995. [Google Scholar]
- 66.Wechsler D. Wechsler Adult Intelligence Scale-Third Edition. San Antonio, TX: The Psychological Corporation; 1997. [Google Scholar]
- 67.Desco M, Pascau J, Reig S, et al. Multimodality image quantification using Talairach grid. Paper presented at: Proceedings of SPIE Medical Imaging; February 18–20, 2001; San Diego, CA. [Google Scholar]
- 68.Reig S, Sánchez-González J, Arango C, et al. Assessment of the increase in variability when combining volumetric data from different scanners. Hum Brain Mapp. 2009;30:355–368. doi: 10.1002/hbm.20511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Moreno D, Burdalo M, Reig S, et al. Structural neuroimaging in adolescents with a first psychotic episode. J Am Acad Child Adolesc Psychiatry. 2005;44:1151–1157. doi: 10.1097/01.chi.0000179055.46795.3f. [DOI] [PubMed] [Google Scholar]
- 70.Reig S, Parellada M, Castro-Fornieles J, et al. Multicenter study of brain volume abnormalities in children and adolescent-onset psychosis. Schizophr Bull. doi: 10.1093/schbul/sbq044. May 16, 2010; doi: 10.1093/schbul/sbq044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Haahr U, Friis S, Larsen TK, et al. First-episode psychosis: diagnostic stability over one and two years. Psychopathology. 2008;41:322–329. doi: 10.1159/000146070. [DOI] [PubMed] [Google Scholar]
- 72.Bendel RB, Afifi AA. Comparison of stopping rules in forward "stepwise" regression. J Am Stat Assoc. 1977;72:848–854. [Google Scholar]
- 73.Bora E, Yücel M, Pantelis C. Theory of mind impairment: a distinct trait-marker for schizophrenia spectrum disorders and bipolar disorder? Acta Psychiatr Scand. 2009;120:253–264. doi: 10.1111/j.1600-0447.2009.01414.x. [DOI] [PubMed] [Google Scholar]
- 74.Marjoram D, Gardner C, Burns J, et al. Symptomatology and social inference: a theory of mind study of schizophrenia and psychotic affective disorder. Cogn Neuropsychiatry. 2005;10:347–359. doi: 10.1080/13546800444000092. [DOI] [PubMed] [Google Scholar]
- 75.Bauer PJ, Stennes L, Haight JC. Representation of the inner self in autobiography: women's and men's use of internal states language in personal narratives. Memory. 2003;11:27–42. doi: 10.1080/741938176. [DOI] [PubMed] [Google Scholar]
- 76.Bosacki SL. Theory of mind and self-concept in preadolescents: links with gender and language. J Educ Psychol. 2000;92:709–717. [Google Scholar]
- 77.Buckner JP, Fivush R. Gender and self in children's autobiographical narratives. Appl Cogn Psychol. 1998;12:407–429. [Google Scholar]
- 78.Davis PJ. Gender differences in autobiographical memory for childhood emotional experiences. J Pers Soc Psychol. 1999;76:498–510. doi: 10.1037//0022-3514.76.3.498. [DOI] [PubMed] [Google Scholar]
- 79.Bora E, Sehitoglu G, Aslier M, Atabay I, Veznedaroglu B. Theory of mind and unawareness of illness in schizophrenia: is poor insight a mentalizing deficit? Eur Arch Psychiatry Clin Neurosci. 2007;257:104–111. doi: 10.1007/s00406-006-0681-3. [DOI] [PubMed] [Google Scholar]
- 80.Morgan K, David A. Neuropsychological studies of insight in patients with psychotic disorders. In: Amador X, David A, editors. Insight and Psychosis: Awareness of Illness in Schizophrenia and Related Disorders. 2nd ed. Oxford, UK: Oxford University Press: 2004:177–193. [Google Scholar]
- 81.Lysaker PH, Bell MD, Bryson GJ, Kaplan E. Neurocognitive function and insight in schizophrenia: support for an association with impairments in prefrontal function but not with impairments in global function. Acta Psychiatr Scand. 1998;9:297–301. doi: 10.1111/j.1600-0447.1998.tb10003.x. [DOI] [PubMed] [Google Scholar]
- 82.Wiffen BD, Rabinowitz J, Lex A, David AS. Correlates, change and ‘state or trait’ properties of insight in schizophrenia. Schizophr Res. 2010;122:94–103. doi: 10.1016/j.schres.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 83.Cooke MA, Peters ER, Greenwood KE, Fisher PL, Kumari V, Kuipers E. Insight in psychosis: influence of cognitive ability and self-esteem. Br J Psychiatry. 2007;191:234–237. doi: 10.1192/bjp.bp.106.024653. [DOI] [PubMed] [Google Scholar]
- 84.Kaiser SL, Snyder JA, Corcoran R, Drake RJ. The relationships among insight, social support, and depression in psychosis. J Nerv Ment Dis. 2006;194:905–908. doi: 10.1097/01.nmd.0000242975.96932.4c. [DOI] [PubMed] [Google Scholar]
- 85.Kemp RA, Lambert TJR. Insight in schizophrenia and its relationship to psychopathology. Schizophr Res. 1995;18:21–28. doi: 10.1016/0920-9964(95)00018-6. [DOI] [PubMed] [Google Scholar]
- 86.Collins AA, Remington GJ, Coulter K, Birkett K. Insight, neurocognitive function and symptom clusters in chronic schizophrenia. Schizophr Res. 1997;27:37–44. doi: 10.1016/S0920-9964(97)00075-3. [DOI] [PubMed] [Google Scholar]
- 87.Rapado-Castro M, Soutullo C, Fraguas D, et al. Predominance of symptoms over time in early-onset psychosis: a principal component factor analysis of the Positive and Negative Syndrome Scale. J Clin Psychiatry. 2010;71:327–337. doi: 10.4088/JCP.08m04845yel. [DOI] [PubMed] [Google Scholar]
- 88.Owen GS, David AS, Richardson G, et al. Mental capacity, diagnosis and insight in psychiatric in-patients: a cross-sectional study. Psychol Med. 2009;39:1389–1398. doi: 10.1017/S0033291708004637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ballenger JC. Clinical guidelines for establishing remission in patients with depression and anxiety. J Clin Psychiatry. 1999;60:29–34. [PubMed] [Google Scholar]
- 90.Mayoral M, Zabala A, Robles O, et al. Neuropsychological functioning in adolescents with first episode psychosis: a two-year follow-up study. Eur Psychiatry. 2008;23:375–383. doi: 10.1016/j.eurpsy.2008.01.1420. [DOI] [PubMed] [Google Scholar]
- 91.Pia L, Tamietto M. Unawareness in schizophrenia: neuropsychological and neuroanatomical findings. Psychiatry Clin Neurosci. 2006;60:531–537. doi: 10.1111/j.1440-1819.2006.01576.x. [DOI] [PubMed] [Google Scholar]