One step closer to on-demand platelet production.
Abstract
The era of induced pluripotent stem (iPS) cells carries with it the promise of virtually unlimited sources of autologous cells for regenerative medicine. However, efficiently differentiating iPS cells into fully functional mature cell types remains challenging. A new study reporting the formation of fully functional platelets from human iPS (hiPS) cells improves upon recent efforts to generate this enucleated cell type, which remains in high demand for therapeutic transfusions. Notably, their lack of nucleus renders platelets unable to retain the pluripotent or tumorigenic properties of iPS cells.
A wide variety of cell types have been generated from mouse embryonic stem cells (ESCs) and human ESCs (hESCs), including blood cells, neurons, and heart cells (Olsen et al., 2006; Dhara et al., 2009; Lengerke and Daley, 2010; Yoshida and Yamanaka, 2010). This has opened up exciting prospects for the use of hESCs in regenerative medicine. However, two major limiting factors need to be addressed before pluripotent stem cell–based therapies can come to fruition. Although hESCs express low levels of major histocompatibility class (MHC) molecules, MHC expression increases with differentiation. Thus, differentiated progeny of hESCs may express MHC levels sufficient to cause rejection in genetically mismatched patients receiving hESC-derived cells. This issue may be addressed by the discovery of induced pluripotent stem (iPS) cells, which can be generated from somatic cells by de novo expression of four “reprogramming” transcription factors: Oct4, Klf4, Sox2, and c-Myc (Takahashi and Yamanaka, 2006). Because it appears that essentially all somatic cell types tested thus far can be reprogrammed into iPS cells (Jaenisch, 2009; Stadtfeld and Hochedlinger, 2010), iPS cells can be generated from individual patients, thereby avoiding the use of allogeneic cell sources for therapy.
However, because all ESCs and iPS cells are inherently tumorigenic and form teratomas in vivo, regardless of the reprogramming method (e.g., integration-free, small molecule, etc.), differentiated cell cultures intended for cell replacement therapy must be completely free of contamination with residual pluripotent cells. One of the ways in which pluripotent cells can be eliminated is by cell irradiation; however, this method is only suitable for red blood cells and platelets, which are enucleated and thus resistant to irradiation. For all other cell derivatives, alternative strategies have to be used, such as the introduction of suicide genes that ablate remaining iPS cells or the pharmacological inhibition of antiapoptotic genes specifically expressed in pluripotent, but not differentiated, ESCs (Schuldiner et al., 2003; Blum et al., 2009).
It is noteworthy that despite intensive efforts, in vitro differentiation of pluripotent stem cells into tissues of interest remains challenging, and that only a fraction of the several hundred specialized cell types in the human body have been generated. The most successful model for ESC differentiation is the hematopoietic system. To date, most mature cell types, including erythrocytes, monocytes, natural killer cells, B and T lymphocytes, and platelets have been differentiated from ESCs in vitro. Nevertheless, with the exception of erythrocytes, the process is either too inefficient for clinical use and/or the differentiated cells do not exhibit full functionality (Lengerke and Daley, 2010). In this issue of the Journal of Experimental Medicine, Takayama et al. describe a novel iPS-based approach to produce fully functional human platelets that could be useful in the clinic.
Generation of platelets in vitro
Platelet transfusions reduce the risk of severe bleeding in a wide range of patients and are a commonly practiced clinical procedure (Wallace et al., 1995). However, platelet transfusions are costly and may lead to immune system complications such as sepsis (Kruskall, 1997), underlining a real need for alternative platelet therapies. Platelets are the progeny of megakaryocytes, a rare population of hyperploid cells that reside in the adult bone marrow, and are generated by the extension of pro-platelet–like filopodia into the vasculature; these extensions shed thousands of cell fragments into the circulation under the shear force of blood flow (Schulze and Shivdasani, 2005; Junt et al., 2007). The formation of megakaryocytes and platelets requires specific transcription factors, including GATA-1, NF-E2, and SCL, which also play key roles in erythropoiesis (Shivdasani and Orkin, 1995; Mikkola et al., 2003). In addition, megakaryocyte numbers, maturation, and pro-platelet formation are dependent on thrombopoietin (TPO; Kaushansky, 2005; Gaur et al., 2006)
Using mouse ESCs, several groups managed to produce platelets and/or megakaryocytes in vitro (Eto et al., 2003; Fujimoto et al., 2003; Kennedy and Keller, 2003). The generation of mature megakaryocytes from hESCs was first reported by Gaur et al. (2006). The hESCs were differentiated on OP9, a bone marrow stromal cell line derived from mice lacking macrophage colony-stimulating factor (Nakano et al., 1994), which has been widely used to support differentiation of both mouse ESCs and hESCs into hematopoietic cells in vitro. The ESC-derived megakaryocytes obtained after 15–17 d of co-culture supplemented with TPO expressed specific surface antigens, including the lineage markers CD41a (integrin αIIb or GPIIb) and CD42b (GPIbα). However, platelets were not detected (Gaur et al., 2006). More recently, Takayama et al. (2008) improved upon the protocol of Gaur et al. (2006) by co-culturing ESCs on either OP9 or C3H10T1/2 stroma for an initial period of 14–15 d with vascular endothelial growth factor (VEGF). This led to the formation of so-called ES-sacs, which are sac-like structures consisting of a morphologically distinct outer layer of cells with endothelial-like properties. ESC sacs also contain a robust population of round hematopoietic-like cells expressing shared markers of blood and endothelium, such as VE-cadherin and CD31, as well as the hematopoietic cell-specific markers CD34, CD41a, and CD45 (Takayama et al., 2008). These hematopoietic cells have the capacity to form hematopoietic colonies in vitro, suggesting that ESC sacs serve as an endothelial-type niche for early blood cell progenitors. When cultured for 24 d in medium that was first supplemented with VEGF and then with TPO plus other cytokines (interleukin [IL]-6, IL-11, and stem cell factor [SCF]), the hematopoietic progenitors from ESC sacs formed megakaryocytes. In addition, megakaryocytes obtained in this manner released platelets that expressed CD41a and CD61 (integrin β3 or GPIIIa) and could be activated in response to ADP and thrombin. However, hESC-derived megakaryocytes contained fewer granules and yielded far fewer platelets than their normal counterparts (2–3 compared with ∼1,000-3,000 per megakaryocyte; Stenberg and Levin, 1989; Takayama et al., 2008). In addition, the in vivo functionality of the ES sac–derived platelets was not demonstrated.
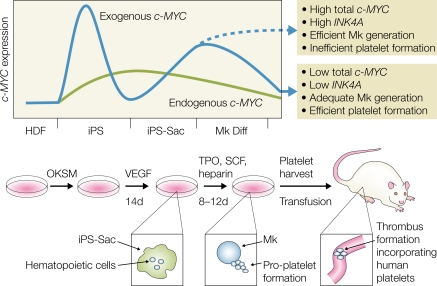
Takayama et al. (2010) now report the generation of hiPS cell–derived megakaryocytes and platelets with in vivo functionality. The main difference between this study and their previous work is the use of hiPS cells derived from human dermal fibroblasts instead of hESCs. In brief, hiPS cells were co-cultured on C3H10T1/2 feeder cells in the presence of VEGF for 14–15 d, and the resulting hiPS sacs were replated in the presence of human TPO and other cytokines (IL-6, IL-11, and SCF) for an additional 8–12 d to form megakaryocytes (Fig. 1). The platelet yield per megakaryocyte under these conditions was improved by approximately two- to threefold compared with their previous study (Takayama et al., 2008). The hiPS-derived platelets were detectable in the circulation for at least 24 h after transfusion into irradiated NOD-SCID-Il2rg−/− mice. Importantly, intravital microscopy revealed that upon laser-induced vessel injury, the transfused platelets participated in the formation of blood clots, showing for the first time the formation of functional platelets from pluripotent human cells in an in vivo assay.
Figure 1.
The generation of human platelets from hiPS cells and the role of c-MYC. iPS cells are generated by the reprogramming of human dermal fibroblasts (HDFs) with the transcription factors OCT4, KLF4, SOX2, and c-MYC (OKSM). In hiPS cells, exogenous c-MYC levels are initially high and activate expression of endogenous c-MYC. Exogenous c-MYC expression subsequently decreases. Co-culture with stromal cells in the presence of VEGF drives formation of an iPS sac containing hematopoietic progenitors. Further culture in megakaryocyte (Mk) conditions (TPO, SCF, and heparin) reactivates exogenous c-MYC expression, leading to the differentiation of megakaryocytes. Down-regulation of exogenous c-MYC expression leads to the efficient production of platelets, whereas sustained exogenous c-MYC expression inhibits platelet generation. Platelets obtained with this protocol, when transferred into mice, can participate in thrombus formation after vessel injury.
Role of c-MYC in iPS and platelet generation
Takayama et al. (2010) also provide insight into the role of c-MYC in the formation of megakaryocytes and platelets. Previous work using transgenic mice overexpressing c-Myc under the control of the platelet-specific promoter PF4 showed a small but significant increase in the number of low ploidy megakaryocytes and a reduction in platelet numbers (Thompson et al., 1996). In support of this observation, c-Myc expression is up-regulated by TPO in megakaryocytes (Chanprasert et al., 2006). Paradoxically, two recent studies reported that the inactivation of c-Myc, either by ablation of the gene, or more indirectly by deletion of the c-Myc regulator RBM15, likewise leads to a cell autonomous increase in the number of low ploidy megakaryocytes (Guo et al., 2009; Niu et al., 2009). The reasons for this apparent discrepancy are not clear, but could be explained or by the assumption that c-Myc has both positive and negative targets that modulate megakaryopoiesis, and that these targets are regulated in a manner sensitive to different transcription factor concentrations.
The first hint about a role of c-MYC in the process of platelet generation from hiPS cells came from the finding that hiPS clones generated with all four reprogramming transcription factors yielded larger numbers of megakaryocytes than hiPS cells generated with only three factors (SOX2, OCT4, and KLF4) in the absence of c-MYC. Further analyses revealed that the four-factor clones reactivated expression of exogenous c-MYC (as well as the other reprogramming factors) at the megakaryocyte stage and that the clone that yielded the largest numbers of platelets showed a decrease of total (exogenous plus endogenous) c-MYC expression after day 15. This suggested that although c-MYC is beneficial for megakaryocyte formation, its sustained expression inhibits platelet formation (Fig. 1), a hypothesis that is supported by two lines of experiments. First, overexpression of c-MYC, but not of OCT4, SOX2, or KLF4, in ESCs led to an increase in megakaryocyte formation. Megakaryocytes generated by c-MYC overexpression exhibited impaired pro-platelet formation, lower ploidy, and a differentiation block and reduced levels of the platelet adhesion molecule GP1bα. Second, hiPS cells expressing an inducible form of c-MYC together with OCT4, KLF4, and SOX2 using a nonintegrating vector (Sendai virus) efficiently generated platelets when c-MYC was inactivated at day 22, but not when c-MYC expression was extended to day 26. It is also noteworthy that overexpression of c-MYC in ESCs to levels that are inhibitory for platelet formation activate the expression of the senescence-inducing p14 (ARF) and p16 (INK4A) genes. p14 (ARF) and p16 (INK4A) expression was not activated in the iPS clone that transiently expressed c-MYC and showed the highest platelet production efficiency. This indicates that a narrow window of c-MYC expression is required for efficient platelet development. Which, if any, of the observed effects of sustained c-MYC expression is the cause of the observed inhibition of platelet formation remains to be determined.
Thus, optimal platelet formation from patient-derived somatic cells requires two waves of c-MYC expression; the first contributes to the efficient reprogramming of somatic cells into hiPS cells (together with the other reprogramming factors) and the second mediates ES/iPS sac and megakaryocyte formation (Fig. 1). However, c-MYC expression has to be shut off to permit efficient platelet formation. In line with this interpretation is the finding that distinct threshold levels of c-Myc govern its output in vivo. Although low levels of deregulated c-Myc are sufficient to induce oncogenesis and ectopic proliferation of somatic cells, activation of the apoptotic and ARF/p53 intrinsic tumor surveillance pathways requires high c-Myc expression (Murphy et al., 2008).
No matter what roles c-Myc plays in the formation of megakaryocyte and platelets, the new work represents a step closer to the use of a highly desired cell type derived from iPS cells for cell replacement therapies. This prospect is all the more exciting because platelets are enucleated cells that can be irradiated, thereby eliminating safety concerns caused by the presence of contaminating iPS cells with the potential to cause tumors.
References
- Blum B., Bar-Nur O., Golan-Lev T., Benvenisty N. 2009. The anti-apoptotic gene survivin contributes to teratoma formation by human embryonic stem cells. Nat. Biotechnol. 27:281–287 10.1038/nbt.1527 [DOI] [PubMed] [Google Scholar]
- Chanprasert S., Geddis A.E., Barroga C., Fox N.E., Kaushansky K. 2006. Thrombopoietin (TPO) induces c-myc expression through a PI3K- and MAPK-dependent pathway that is not mediated by Akt, PKCzeta or mTOR in TPO-dependent cell lines and primary megakaryocytes. Cell. Signal. 18:1212–1218 10.1016/j.cellsig.2005.09.010 [DOI] [PubMed] [Google Scholar]
- Dhara S.K., Gerwe B.A., Majumder A., Dodla M.C., Boyd N.L., Machacek D.W., Hasneen K., Stice S.L. 2009. Genetic manipulation of neural progenitors derived from human embryonic stem cells. Tissue Eng. Part A. 15:3621–3634 10.1089/ten.tea.2009.0155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eto K., Leavitt A.L., Nakano T., Shattil S.J. 2003. Development and analysis of megakaryocytes from murine embryonic stem cells. Methods Enzymol. 365:142–158 10.1016/S0076-6879(03)65010-X [DOI] [PubMed] [Google Scholar]
- Fujimoto T.T., Kohata S., Suzuki H., Miyazaki H., Fujimura K. 2003. Production of functional platelets by differentiated embryonic stem (ES) cells in vitro. Blood. 102:4044–4051 10.1182/blood-2003-06-1773 [DOI] [PubMed] [Google Scholar]
- Gaur M., Kamata T., Wang S., Moran B., Shattil S.J., Leavitt A.D. 2006. Megakaryocytes derived from human embryonic stem cells: a genetically tractable system to study megakaryocytopoiesis and integrin function. J. Thromb. Haemost. 4:436–442 10.1111/j.1538-7836.2006.01744.x [DOI] [PubMed] [Google Scholar]
- Guo Y., Niu C., Breslin P., Tang M., Zhang S., Wei W., Kini A.R., Paner G.P., Alkan S., Morris S.W., et al. 2009. c-Myc-mediated control of cell fate in megakaryocyte-erythrocyte progenitors. Blood. 114:2097–2106 10.1182/blood-2009-01-197947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaenisch R. 2009. Stem cells, pluripotency and nuclear reprogramming. J. Thromb. Haemost. 7:21–23 10.1111/j.1538-7836.2009.03418.x [DOI] [PubMed] [Google Scholar]
- Junt T., Schulze H., Chen Z., Massberg S., Goerge T., Krueger A., Wagner D.D., Graf T., Italiano J.E., Jr, Shivdasani R.A., von Andrian U.H. 2007. Dynamic visualization of thrombopoiesis within bone marrow. Science. 317:1767–1770 10.1126/science.1146304 [DOI] [PubMed] [Google Scholar]
- Kaushansky K. 2005. The molecular mechanisms that control thrombopoiesis. J. Clin. Invest. 115:3339–3347 10.1172/JCI26674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M., Keller G.M. 2003. Hematopoietic commitment of ES cells in culture. Methods Enzymol. 365:39–59 10.1016/S0076-6879(03)65003-2 [DOI] [PubMed] [Google Scholar]
- Kerenyi M.A., Orkin S.H. 2010. Networking erythropoiesis. J. Exp. Med. 207:2543–2550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruskall M.S. 1997. The perils of platelet transfusions. N. Engl. J. Med. 337:1914–1915 10.1056/NEJM199712253372609 [DOI] [PubMed] [Google Scholar]
- Lengerke C., Daley G.Q. 2010. Autologous blood cell therapies from pluripotent stem cells. Blood Rev. 24:27–37 10.1016/j.blre.2009.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikkola H.K., Klintman J., Yang H., Hock H., Schlaeger T.M., Fujiwara Y., Orkin S.H. 2003. Haematopoietic stem cells retain long-term repopulating activity and multipotency in the absence of stem-cell leukaemia SCL/tal-1 gene. Nature. 421:547–551 10.1038/nature01345 [DOI] [PubMed] [Google Scholar]
- Murphy D.J., Junttila M.R., Pouyet L., Karnezis A., Shchors K., Bui D.A., Brown-Swigart L., Johnson L., Evan G.I. 2008. Distinct thresholds govern Myc’s biological output in vivo. Cancer Cell. 14:447–457 10.1016/j.ccr.2008.10.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakano T., Kodama H., Honjo T. 1994. Generation of lymphohematopoietic cells from embryonic stem cells in culture. Science. 265:1098–1101 10.1126/science.8066449 [DOI] [PubMed] [Google Scholar]
- Niu C., Zhang J., Breslin P., Onciu M., Ma Z., Morris S.W. 2009. c-Myc is a target of RNA-binding motif protein 15 in the regulation of adult hematopoietic stem cell and megakaryocyte development. Blood. 114:2087–2096 10.1182/blood-2009-01-197921 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen A.L., Stachura D.L., Weiss M.J. 2006. Designer blood: creating hematopoietic lineages from embryonic stem cells. Blood. 107:1265–1275 10.1182/blood-2005-09-3621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuldiner M., Itskovitz-Eldor J., Benvenisty N. 2003. Selective ablation of human embryonic stem cells expressing a “suicide” gene. Stem Cells. 21:257–265 10.1634/stemcells.21-3-257 [DOI] [PubMed] [Google Scholar]
- Schulze H., Shivdasani R.A. 2005. Mechanisms of thrombopoiesis. J. Thromb. Haemost. 3:1717–1724 10.1111/j.1538-7836.2005.01426.x [DOI] [PubMed] [Google Scholar]
- Shivdasani R.A., Orkin S.H. 1995. Erythropoiesis and globin gene expression in mice lacking the transcription factor NF-E2. Proc. Natl. Acad. Sci. USA. 92:8690–8694 10.1073/pnas.92.19.8690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadtfeld M., Hochedlinger K. 2010. Induced pluripotency: history, mechanisms, and applications. Genes Dev. 24:2239–2263 10.1101/gad.1963910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenberg P.E., Levin J. 1989. Mechanisms of platelet production. Blood Cells. 15:23–47 [PubMed] [Google Scholar]
- Takahashi K., Yamanaka S. 2006. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 126:663–676 10.1016/j.cell.2006.07.024 [DOI] [PubMed] [Google Scholar]
- Takayama N., Nishikii H., Usui J., Tsukui H., Sawaguchi A., Hiroyama T., Eto K., Nakauchi H. 2008. Generation of functional platelets from human embryonic stem cells in vitro via ES-sacs, VEGF-promoted structures that concentrate hematopoietic progenitors. Blood. 111:5298–5306 10.1182/blood-2007-10-117622 [DOI] [PubMed] [Google Scholar]
- Takayama N., Nishimura S., Nakamura S., Shimizu T., Ohnishi R., Endo H., Yamaguchi T., Otsu M., Nishimura K., Nakanishi M., et al. 2010. Transient activation of c-MYC expression is critical for efficient platelet generation from human induced pluripotent stem cells. J. Exp. Med. 207:2817–2830 10.1084/jem.20100844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson A., Zhang Y., Kamen D., Jackson C.W., Cardiff R.D., Ravid K. 1996. Deregulated expression of c-myc in megakaryocytes of transgenic mice increases megakaryopoiesis and decreases polyploidization. J. Biol. Chem. 271:22976–22982 10.1074/jbc.271.38.22976 [DOI] [PubMed] [Google Scholar]
- Wallace E.L., Churchill W.H., Surgenor D.M., An J., Cho G., McGurk S., Murphy L. 1995. Collection and transfusion of blood and blood components in the United States, 1992. Transfusion. 35:802–812 10.1046/j.1537-2995.1995.351096026360.x [DOI] [PubMed] [Google Scholar]
- Yoshida Y., Yamanaka S. 2010. iPS cells: A source of cardiac regeneration. J. Mol. Cell. Cardiol. 10.1016/j.yjmcc.2010.20.026 [DOI] [PubMed] [Google Scholar]