Abstract
Sex in mammals is genetically determined and is defined at the cellular level by sex chromosome complement (XY males and XX females). The Y chromosome–linked gene sex-determining region Y (SRY) is believed to be the master initiator of male sex determination in almost all eutherian and metatherian mammals, functioning to upregulate expression of its direct target gene Sry-related HMG box–containing gene 9 (SOX9). Data suggest that SRY evolved from SOX3, although there is no direct functional evidence to support this hypothesis. Indeed, loss-of-function mutations in SOX3 do not affect sex determination in mice or humans. To further investigate Sox3 function in vivo, we generated transgenic mice overexpressing Sox3. Here, we report that in one of these transgenic lines, Sox3 was ectopically expressed in the bipotential gonad and that this led to frequent complete XX male sex reversal. Further analysis indicated that Sox3 induced testis differentiation in this particular line of mice by upregulating expression of Sox9 via a similar mechanism to Sry. Importantly, we also identified genomic rearrangements within the SOX3 regulatory region in three patients with XX male sex reversal. Together, these data suggest that SOX3 and SRY are functionally interchangeable in sex determination and support the notion that SRY evolved from SOX3 via a regulatory mutation that led to its de novo expression in the early gonad.
Introduction
Sex in mammals is genetically determined and is defined at the cellular level by sex chromosome complement (XY males and XX females) and at the phenotypic level by the development of gender-specific anatomy, physiology, and behavior. Disorders of sexual development (DSDs) in humans are characterized by a complete or partial mismatch between genetic sex and phenotypic sex. Collectively, DSDs occur in at least 1 in 100 live births (1) and include relatively mild forms such as hypospadias (1 in 500 births) as well as more severe conditions such as ambiguous genitalia (1 in 4,500 births) and complete sex reversal (46, XY females and 46, XX males; 1 in 20,000 births). Although the etiology of many DSDs is not known, some cases of complete XX male and XY female sex reversal are associated with translocations or mutations of the Y-linked testis-determining gene SRY (sex-determining region Y) (2–8). SRY encodes a transcription factor that contains a highly conserved high-mobility group (HMG) DNA-binding domain and poorly conserved N- and C-terminal domains. Gain-of-function and loss-of-function studies in mice have demonstrated that Sry is necessary and sufficient for testis development (4, 7). Indeed, except in two species of the vole Ellobius (9) and the spiny rat (10), which lack the gene, SRY is believed to function as the master regulator of male sex determination in all therian (non-egg-laying) mammals (11).
The gonads develop from a band of mesoderm, the genital ridge (GR), that lies ventrally subjacent to the mesonephros, and together these constitute the urogenital ridge. The GR is considered to be bipotential, as it contains precursors that can differentiate into testis- or ovary-specific cell lineages (12). In mice, Sry is expressed in a transient center-to-pole wave in the XY GR from approximately 10.5 to 12.5 dpc (13, 14). Analysis of hypomorphic Sry alleles and inducible Sry transgenic mice indicate that the level of Sry activity must exceed a critical threshold within a developmental window of approximately 6 hours (12–15 tail somite [ts] stage) for irreversible commitment to a male fate (15–17). Despite its pivotal role in directing male differentiation, it appears that the sole critical function of Sry in sex determination is to upregulate its direct target gene Sox9 (Sry-related HMG box–containing gene 9), thereby initiating Sertoli cell differentiation (18–20). Sertoli cells subsequently undergo a period of rapid proliferation, aided by Sox9/Fgf9 and Sox9/Pgd2 positive feedback loops (21, 22), and migrate and surround germ cells within the gonad to form testis cords. Sertoli cells also drive the male-specific migration of cells from the underlying mesonephros into the gonad. These are mostly endothelial cells that give rise to the coelomic vessel and other testis-specific vasculature (23). By 12.5 dpc, steroidogenic Leydig cells have differentiated and, just 48 hours after Sry begins to initiate the male pathway, the early testis already has a highly organized stereotypical structure and is morphologically distinct from the ovary, which remains a mass of germ cells scattered within the gonadal mesenchyme.
Although female development has traditionally been considered by some to be a “default” pathway, it is now clear that sexual fate is determined by a balance of opposing signals within the gonad, in which Sry exerts a dominant masculinizing influence (22, 24). For example, loss-of-function mutations in Wnt4 and Rspo1 cause partial XX male sex reversal, indicating that canonical Wnt signaling predisposes the gonad to an ovarian fate (22, 25–29). Furthermore, gain-of-function mutations in Wnt4 and Ctnnb1 induce XY female sex reversal. While it is possible that high levels of pro-ovarian signals could block Sry activity, the evidence so far suggests that the effect of high levels of β-catenin is to prevent the maintenance of Sox9 expression (27, 29, 30). Unexplained cases of SRY-negative XX male sex reversal in humans may therefore arise from loss-of-function mutations in members of the pro-ovarian pathway (such as canonical Wnt signaling components) or gain-of-function mutations in genes that mimic SRY activity, of which members of the SOX gene family are good candidates.
SRY-related HMG box–containing gene 3 (SOX3) is a single-exon gene located in a highly conserved region of the X chromosome in therian mammals (31–33). Other vertebrate species also possess homologs of SOX3, but none of these have been found to be sex-linked, even in prototherian mammals, such as the echidna (34). Of the 20 SOX genes in the mammalian genome, SOX3 encodes a protein that is most similar to SRY, sharing 67% amino acid identity (and 90% similarity) across the DNA-binding HMG domain (31, 35). Comparative sequence data, coupled with molecular and cytogenetic studies of sex chromosomes, have led to the hypothesis that SRY arose during early mammalian evolution from a gain-of-function mutation in the proto-Y allele of SOX3. This mutation is thought to have activated gonadal expression of the proto-Y SOX3 allele, resulting in the emergence of a novel sex determination switch that is specific to therian mammals (32, 36). Consistent with this hypothesis, Sox3 expression is exceedingly low or absent in the developing gonads of mice and marsupials (33, 37). Sox3 is, however, widely expressed in the central nervous system of vertebrate embryos and is required for normal brain development and function in mice and humans, as well as in pituitary and craniofacial development (31, 38–44). Importantly, mice and humans with mutations in SOX3 do not show any defects in sex determination, although postnatal differentiation of some spermatogonia and the survival of some follicles are affected in Sox3-null mice (40, 45, 46). Thus, although SOX3 does not normally function in sex determination, the proposed evolutionary link between SOX3 and SRY raises the intriguing possibility that certain gain-of-function mutations in SOX3 may cause XX male sex reversal in mice and humans.
In addition to loss-of-function mutations of SOX3, duplications of the locus can also lead to pituitary hormone deficiencies in humans (38, 41, 47). This was assumed to be due to overexpression, and therefore, to investigate this further, transgenic mice were derived using a large mouse Sox3 genomic fragment. Some of these mice developed CNS abnormalities (P. Thomas, unpublished observations). However, here we describe a transgenic model of complete XX male sex reversal in which Sox3 is ectopically expressed in the developing XX gonads due to a position effect. Initiation of the testis pathway in XX hemizygous transgenic (Tg/+) gonads induced Sox9 upregulation, Sertoli cell differentiation, testis cord formation, and generation of a male-specific vasculature. Cotransfection assays using the recently identified Sox9 testis-specific enhancer element (TESCO; ref. 18) showed that SOX3, like SRY, had modest transactivation activity and functioned synergistically with steroidogenic factor–1 (SF1) in this context. Importantly, Sox3 failed to induce XX testis development in gonads that lacked Sox9. We also present 3 patients with XX male sex reversal, each of which has a unique genomic rearrangement of the SOX3 regulatory region. Together, these data suggest that Sox3 gain-of-function in the developing gonad induces testis development by functioning as a surrogate for Sry. Our findings also provide important functional evidence to support the long-standing hypothesis that Sox3 is the evolutionary precursor of Sry and suggest that rearrangements of SOX3 are a relatively frequent cause of XX male sex reversal in humans.
Results
Complete XX male sex reversal exhibited in a line of Sox3 transgenic mice.
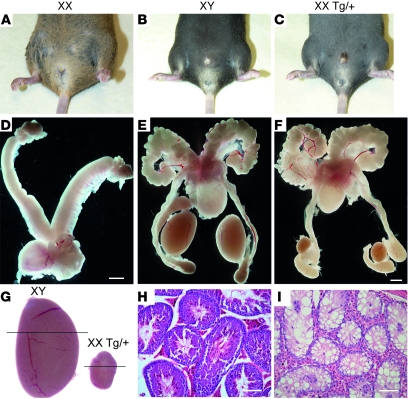
To investigate the developmental impact of Sox3 overexpression, we generated transgenic mice using a 37-kb murine Sox3 genomic fragment that included an IRES-EGFP reporter cassette. Intriguingly, 1 of 20 lines that we generated contained an excess of male progeny, suggesting XX male sex reversal. Comparison of genotype, chromosomal sex, and phenotypic sex of 670 weaned progeny revealed that XX male sex reversal had occurred in approximately 77% of XX hemizygous transgenic (Tg/+) animals. Accordingly, this transgenic line was named Sox3 transgene Sex reversed [Tg(Sox3)1Pqt, abbreviated here to Sr]. XX Tg/+ males exhibited normal male external genitalia (Figure 1, A–C), with completely masculinized reproductive tracts (Figure 1, D–F; n = 4), although their testes were significantly smaller than those of XY males and lacked sperm (Figure 1, G–I). Transgenic animals exhibited normal male mounting behavior.
Figure 1. XX Tg/+ adults develop as males.
(A–C) External genitalia of XX, XY, and XX Tg/+ individuals. Note the male phenotype of the XX Tg/+ animal. (D–F) XX, XY and XX Tg/+ internal reproductive tracts. Scale bars: 2 mm. (G) XY and XX Tg/+ testes. (H and I) Histological sections of XY and XX Tg/+ testes, respectively, showing the absence of sperm in the latter. Scale bars: 100 μm. All animals used for this analysis were between 20 and 24 weeks of age.
Sox3 Tg/+ XX gonads express Sox9 and activate the testis differentiation pathway.
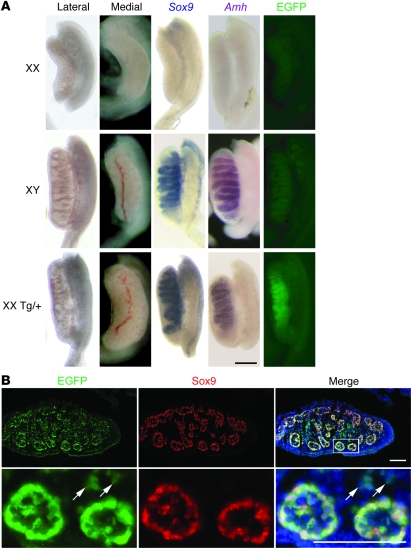
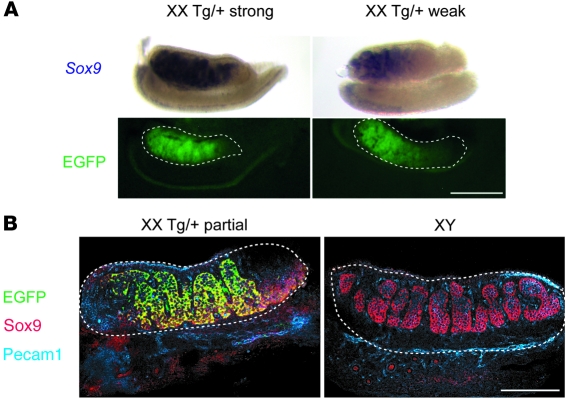
To investigate the mechanism of sex reversal in Sr mice, we analyzed gonad morphology and expression of male-specific marker genes at 13.5 dpc, when the outcome of sex determination is morphologically evident (Figure 2). Most XX Tg/+ gonads had a normal male appearance, including the presence of testis cords and a coelomic vessel extending across the ventral surface of the gonads (Figure 2A). Robust expression of Sox9 and its direct target gene Amh (48) was also clearly evident in the testis cords (Figure 2A). Confocal analysis revealed that EGFP (which completely overlapped with Sox3, Supplemental Figure 1; supplemental material available online with this article; doi: 10.1172/JCI42580DS1) was expressed in Sertoli cells (and some interstitial cells) but not germ cells (data not shown), consistent with a direct role for this Sox3 transgene in Sertoli cell differentiation (Figure 2B). Interestingly, some XX Tg/+ gonads exhibited regionally localized/restricted expression of the Sox3 transgene (Figure 3, A and B). In these cases, the location and intensity of expression from the transgene closely correlated with levels of Sox9 expression and the extent of testis cord formation. Moreover, this was always seen in the central and anterior regions of the gonad, consistent with other cases of ovotestis development and with these regions being the most sensitive to male-inducing signals (refs. 49, 50, and Figure 3). A small proportion (5 of 32) of 13.5-dpc XX Tg/+ gonads did not express detectable levels of the transgene, and this correlated with the absence of both Sox9 expression and testis cord formation (data not shown). Taken together, these data demonstrate a strong correlation among transgene expression, Sox9 activation, and testicular phenotype.
Figure 2. XX Tg/+ gonads have a normal male appearance and express the Sox3 transgene at 13.5 dpc.
(A) XX, XY, and XX Tg/+ 13.5-dpc gonads shown in lateral and medial views and analyzed for Sox9 and Amh expression and EGFP fluorescence in unfixed tissue. Scale bar: 250 μm. (B) XX Tg/+ 13.5-dpc gonad section stained with EGFP and Sox9 antibodies. Lower panel shows higher-magnification view of the boxed region. Arrows indicate EGFP-positive, Sox9-negative cells in the interstitium. Scale bars: 50 μm.
Figure 3. 13.5-dpc XX Tg/+ gonads show variable cord formation and Sox9 expression that coincides with the spatial localization of transgene (EGFP) expression.
(A) Sox9 expression and corresponding EGFP expression in representative XX Tg/+ (strong), and XX Tg/+ (weak) 13.5-dpc gonads. Scale bar: 250 μm. (B) Optical slices of whole mount 12.5-dpc XX Tg/+ and XY gonads stained with EGFP (green), Sox9 (red), and Pecam1 (blue). The dotted lines indicate the outline of the gonads. Scale bar: 150 μm.
Sox3 from the Sr transgene mimics Sry in its early expression and function.
Activation of Sox9 expression in supporting cell precursors by Sry is normally required for their differentiation into Sertoli cells and subsequent testis development. However, Sox9 itself can induce testis differentiation when it is ectopically expressed in the early gonad or even in cases where the locus has been duplicated (51–53). This implies that Sox9 is the only critical target of Sry but also raises the question as to whether Sox3, from the Sr transgene, acts like Sry or Sox9. The former would be consistent with the higher degree of similarity between the HMG box domains of Sry and Sox3, while the latter might reflect the presence of strong transactivation domains at the C-termini of both Sox9 and Sox3 (although with little homology), whereas no such domain is found in Sry (18, 33).
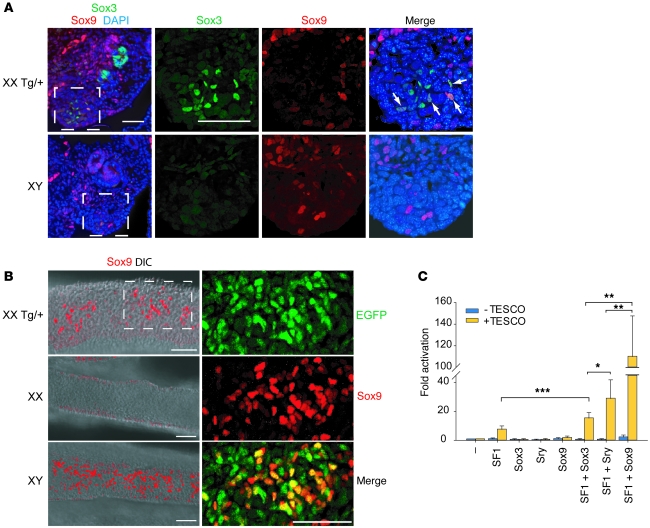
To address this, it was important to first look at details of expression from the Sox3 Sr transgene (Sox3-SrTg). Endogenous Sry can first be detected at approximately 12 ts, and is shortly followed by an upregulation of Sox9 in Sry-positive cells (19). Expression of the Sox3 transgene was also first detected at 12 ts (see below), and by 13 ts, several Sox3-positive cells were clearly present in the XX Tg/+ GR (Figure 4A). Some of these were also Sox9-positive (arrows), suggesting that Sox3 is directly involved in activating Sox9 expression in the XX Tg/+ GR. XY GRs had a similar small number of Sox9-positive cells, reflecting the initial upregulation of Sox9 by Sry (Figure 4A). Consistent with their differentiation along the testis pathway, and similar to control XY gonads, XX Tg/+ GRs exhibited robust Sox9 expression at 19 ts (approximately 11.5 dpc), and this regionally coincided with EGFP expression across the GR (Figure 4B and data not shown). High-magnification confocal analysis revealed coexpression of EGFP and Sox9 consistent with cell autonomous induction of Sox9 by Sox3. There was not an exact correlation, however, as EGFP-positive, Sox9-negative cells were also observed in 11.5-dpc and 12.5-dpc XX Tg/+ gonads (Figure 4B and data not shown). These may represent cells fated to be Sertoli cells that have yet to upregulate Sox9, or interstitial cell precursors. We also found some Sox9-positive, EGFP-negative cells in the XX Tg/+ genital ridges (Figure 4B). These probably represent supporting cell precursors recruited to the Sertoli lineage by preexisting Sertoli cells, which occurs in early XY gonads via non-cell-autonomous mechanisms (21, 22).
Figure 4. Mechanism of transgene-induced sex reversal and its activation in the gonads.
(A) 13 ts XX Tg/+ and XY transverse sections presented as a 6-μm stack stained with Sox3 and Sox9. Enlargements corresponding to the boxed region are shown on the right. Arrows indicate Sox3/Sox9-positive cells. Scale bars: 50 μm. (B) Single optical slices of whole mount 11.5-dpc XX, XY, and XX Tg/+ genital ridges stained with Sox9 and presented as an overlay with corresponding differential interference contrast (DIC) image. Higher-magnification view of the boxed region in the XX Tg/+ genital ridge is shown in the right panel, presented as a 60-μm stack of optical sections and highlighting the extent of colocalization between Sox3 and Sox9. Scale bars: 50 μm. In B, genital ridges are oriented so that anterior is to the left and the coelomic epithelium at the top. (C) Transactivation experiment using the mouse Sox9 TESCO enhancer reporter. Note that Sox3 and Sry exhibit similar synergistic activation of the reporter with SF1, but fail to activate in the absence of SF1. Negative control transfections using a reporter lacking the enhancer are also shown (–TESCO). Western blot analysis indicated that comparable levels of Sox3, Sry, and Sox9 protein were expressed (data not shown). Data are mean ± SEM. ***P < 0.001, **P < 0.01, *P < 0.05.
Sox3, like Sry, transactivates the Sox9 testis-specific enhancer element TESCO through synergistic interaction with Sf1.
Sry upregulates Sox9 transcription in the testis through binding and activation of a gonad-specific enhancer, testis-specific enhancer of Sox9 (TES), synergistically with Sf1 (18, 54). To further investigate functional similarity of Sox3 and Sry, we performed transactivation assays in vitro using the TES core element (TESCO) (18). Consistent with previously published data, Sry activated TESCO, but only when Sf1 was coexpressed (Figure 4C and ref. 18). In the absence of Sf1, Sox3 also failed to transactivate TESCO. However, cotransfection of Sox3 and Sf1 resulted in approximately 2-fold activation above Sf1 alone, indicating that Sox3, like Sry, synergistically activates the TESCO enhancer, albeit to a slightly lesser degree (ref. 18 and Figure 4C). In contrast, greater than 14-fold activation of the TESCO reporter was detected when Sox9 and Sf1 were coexpressed compared with Sf1 alone. These results are also consistent with Sox3 acting like Sry, and not like Sox9, in this context.
Sox3 from the Sr transgene requires endogenous Sox9 to induce testes.
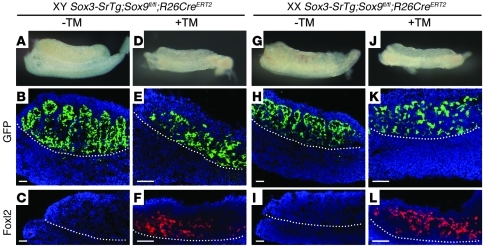
To further explore whether Sox3 is acting exclusively like Sry, or whether it also mimics Sox9 function, we designed a genetic experiment to test if the Sox3-SrTg was able to induce sex reversal in the absence of Sox9. If Sox3 is functionally equivalent to Sry, the Sox3-SrTg should be unable to rescue or induce testis development in either XY or XX embryos also null mutant for Sox9. Conversely, if Sox3 acts like Sox9, the Sox3-SrTg should be able to substitute for both Sry and Sox9 to promote testis cord formation. Sox3-SrTg;Sox9fl/fl;R26-CreERT2 stud males were bred to Sox9fl/fl females. Pregnant females were given tamoxifen at 9.5 and 10.5 dpc, a protocol found to give efficient Cre-mediated deletion of Sox9 and XY female sex reversal (data not shown), or ethanol as a vehicle control, and embryos harvested at 12.5 dpc to determine genotype and gonadal phenotype. In some experiments, a Tesco-cfp transgene was included, to allow a simple assay of Sertoli cell differentiation (18). When tamoxifen was omitted, the Sr transgene was able to give XX male development, despite the altered genetic background (see below). However, after tamoxifen administration, we obtained 2 XY Sox3-SrTg;Sox9fl/fl;R26-CreERT2 embryos, one with ovotestes and the other with morphologically normal ovaries, and a total of 6 XX Sox3-SrTg;Sox9fl/fl;R26-CreERT2 embryos (4 with Tesco-cfp, 2 without), all of which had gonads with the morphological appearance of ovaries (Figure 5). Marker analysis revealed robust expression of Foxl2, which is specific to ovarian cell types (Figure 5). A number of Sox9-positive cells were still present due to inefficient action of tamoxifen in the embryos (data not shown), and these are likely to be the same cells that show relatively high levels of EGFP expression from the Sox3-SrTg. However, these were not present in sufficient numbers to have organized into testis cords or to have induced the testis-specific vasculature (Figure 5 and data not shown). Collectively, these results provide strong evidence that Sox3 from the transgene is not able to compensate for the loss of Sox9, and therefore that it behaves like Sry and not like Sox9.
Figure 5. The Sox3 transgene is not able to mimic Sox9 in the gonad at 12.
dpc. (A–F) XY Sox3-SrTg;Sox9fl/fl;R26CreERT2 mice were either treated or not treated with tamoxifen (TM). In the absence of TM, the gonad developed into a normal testis containing testis cords (A–C). When Sox9 was ablated after TM treatment, testis cords were not formed (D and E), and Foxl2-positive cells were detected in the gonad (F). (G–L) Similar experiments were performed for XX Sox3-SrTg;Sox9fl/fl;R26CreERT2 mice. The mice developed XX sex-reversed testis without TM (G–I). In contrast, TM treatment resulted in ovarian morphology and the expression of Foxl2 (J–L). Scale bars: 0.5 mm (A, D, G, and J) and 50 μm (B, C, E, F, H, I, K, and L).
Integration of Sr transgene upstream of the embryonic testis gene Aldh1a1.
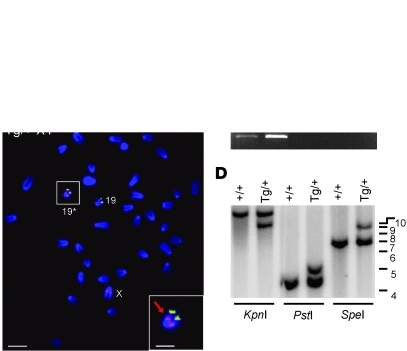
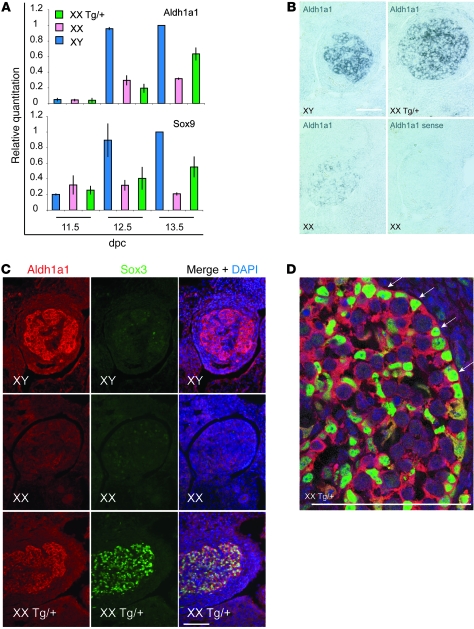
Sex ratios are unaffected in Sox3-null mice, indicating that Sox3 is not required for sex determination (40, 45). Furthermore, although very low levels of Sox3 transcripts in 11.5-dpc gonads have been reported (33), we were unable to detect Sox3 protein in wild-type GRs during sex determination (data not shown). We therefore postulated that Sox3 was ectopically upregulated in the gonads of Sr Tg/+ embryos due to a position effect. This was supported by analysis of a second independent Sox3 transgenic line in which transgene expression was not detected in the gonads by immunohistochemistry (11.5 dpc and 13.5 dpc) or quantitative RT-PCR (qRT-PCR) (11.5 dpc) despite comparable CNS expression (data not shown). Furthermore, none of 8 XX Sox3 transgenic founders that were generated exhibited XX male sex reversal (data not shown). We initially characterized the Sr integration site using FISH analysis of metaphase chromosomes, which revealed a single transgene insertion site on chromosome 19 in the B/C1 region (Figure 6B). GenomeWalker, PCR, and Southern blot analyses confirmed these data and revealed that 2 copies of the transgene construct had integrated upstream of the retinaldehyde dehydrogenase A1 (Aldh1a1, also known as Raldh1) gene (Figure 6, A, C, and D, and data not shown). The Aldh1a1 gene product Aldh1a1 catalyzes the conversion of retinaldehyde into all-trans-retinoic acid, albeit with less efficiency than the related enzymes Aldh1a2 and Aldh1a3 (55, 56). Although not required for sex determination (57, 58), Aldh1a1 was previously identified as a testis-enriched gene in a subtractive hybridization screen (59) and is strongly expressed in Sertoli cells and a subset of interstitial cells from 11.5 dpc onward (60). To assess whether Aldh1a1 is upregulated during differentiation of XX Tg/+ gonads into testes, we compared Aldh1a1 and Sox9 expression during gonad differentiation by qRT-PCR (Figure 7A). At 13.5 dpc, Aldh1a1 and Sox9 expression was considerably higher in XX Tg/+ gonads compared with XX controls. However, expression of both genes was even higher in XY controls, at 13.5 dpc and 12.5 dpc, which suggests that activation of the male pathway is less efficient in the XX Tg/+ gonads, consistent with occasional ovotestis or ovary development (Figures 2 and 3). In situ hybridization and immunohistochemistry on 13.5-dpc gonads confirmed that Aldh1a1 was strongly expressed in XX Tg/+ gonads, where it was localized to Sertoli and some interstitial cells in an identical pattern to XY controls (Figure 7, B–D, and data not shown).
Figure 6. The Sr transgene has integrated upstream of Aldh1a1 on chromosome 19.
(A) Schematic representation of transgene insertion site on chromosome 19 illustrating PCR primer position (black arrows), Southern probe (red), and relevant restriction sites. (B) FISH analysis of Tg/+ XY metaphase chromosomes. Sox3 transgene (modified RP23-174O19 BAC) and chromosome 19 (RP23-142D22 BAC) signals are shown in red and green, respectively. The inset shows a magnified view of chromosome 19 into which the transgene has integrated (19*). Scale bars: 2.5 μm; 0.5 μm in inset. (C) PCR assay of genomic DNA using primers that flank the transgene integration site. Product is amplified from Tg/+ DNA and not from +/+ DNA. All genomic DNA samples were shown to be amplifiable using Gapdh primers (data not shown). H2O indicates negative control reaction. (D) Southern blot analysis comparing +/+ and Tg/+ DNA digested with KpnI, PstI, and SpeI. Marker sizes (kb) are shown on the right. Note the additional bands of expected size in each of the Tg/+ tracts. K, KpnI; P, PstI; S, SpeI.
Figure 7. Expression profile of Aldh1a1/Aldh1a1 and Sox3 in XY and XX Tg/+ gonads from 11.5 to 13.5 dpc.
(A) qRT-PCR analysis of 11.5- to 13.5-dpc gonads. Normalized expression levels of each gene are shown relative to β-actin. Two cDNA series were analyzed twice each, and error bars represent SD of the mean of the two series. (B) In situ hybridization showing Aldh1a1 expression in transverse sections of 13.5-dpc XY, XX Tg/+, and XX gonads. No signal was detected using an Aldh1a1 sense probe. (C) Transverse sections of XY, XX Tg/+, and XX 13.5-dpc gonads stained with Aldh1a1, Sox3, and DAPI. Protein expression levels are consistent with the transcript expression analysis shown in B. (D) Confocal micrograph of 13.5-dpc XX Tg/+ gonad showing the extensive overlap in Sox3 and Aldh1a1 expression. Arrows indicate Sox3/Aldh1a1-positive cells lining the testis cords. Scale bars: 100 μm.
Coexpression of Sox3 transgene and Aldh1a1 in Sertoli cells in XX Tg/+ gonads.
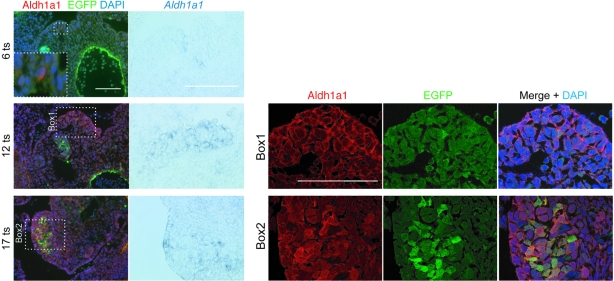
To investigate whether proximity of the transgene to Aldh1a1 could influence its expression, we compared the distribution of their gene products in XX Tg/+ gonads. At 13.5 dpc, we observed extensive overlap of Sox3 and Aldh1a1 throughout the gonad (Figure 7, C and D), including the Sertoli cells lining the testis cords, suggesting that regulatory elements from Aldh1a1 may be important for ectopic transgene expression in the testis (arrows, Figure 7D). To further investigate the relationship between Aldh1a1 and transgene expression during sex determination in XX Tg/+ gonads, we compared the distribution of Aldh1a1 and EGFP from 6 to 17 ts (Figure 8). Initially, Aldh1a1 was detected in the GR at very low levels, and no EGFP was present. By 12 ts, Aldh1a1-positive cells, detectable by in situ hybridization and immunohistochemistry, contained the initial population of EGFP-positive cells (Figure 8, box 1). By 17 ts, the number of EGFP-positive cells increased markedly, and most, if not all, of these cells also expressed Aldh1a1, albeit at low levels in some cases (Figure 8, box 2). The lack of clear singly positive EGFP cells suggests that Aldh1a1 expression may provide a permissive regulatory environment for transgene activation. Once Sox3 transgene expression is initiated in uncommitted XX gonads and Sox9 expression is upregulated, a positive feedback loop may arise, resulting in robust coexpression of the transgene and Aldh1a1 in Sertoli cells (60).
Figure 8. Expression of Aldh1a1 and EGFP in the gonadal ridge of XX Tg/+ gonads at 6–17 ts.
Immunohistochemistry and in situ hybridization of serial transverse sections showing EGFP and Aldh1a1 distribution in 6–17 ts embryos. Given the low level of Aldh1a1 protein in the XX Tg/+ GR during these early stages of sex determination, the expression of Aldh1a1 in adjacent sections is included at each stage for comparison. Aldh1a1/Aldh1a1 is first observed at 6 ts (magnified in box inset). Aldh1a1/Aldh1a1 is detected in a diffuse pattern at 12 ts. A rare EGFP-positive cell is found within a population of Aldh1a1-positive cells in the GR at this stage (magnified in box 1). At 17 ts, Aldh1a1/Aldh1a1 becomes more restricted, while the abundance of EGFP within the GR increases (magnified in box 2). Scale bars: 100 μm.
Rearrangements of the SOX3 regulatory region are associated with XX male sex reversal in humans.
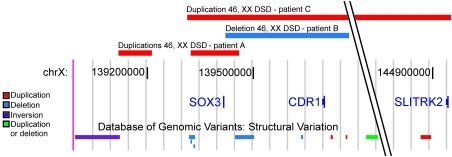
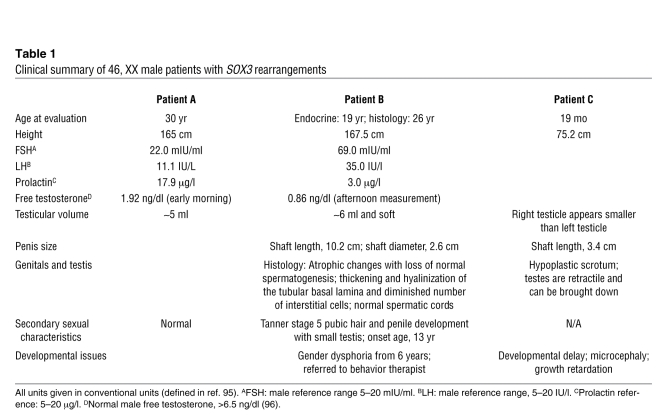
Given that SOX3 function in the CNS is broadly conserved in mice and humans and that Sox3 is able to induce sex reversal when ectopically expressed in the developing murine GR, it is possible that rearrangements at the SOX3 locus are associated with human XX male sex reversal. We therefore screened a cohort of 16 SRY-negative 46, XX male patients for copy number variation using Affymetrix 6.0 whole genome SNP (7 patients) and Illumina 1M (9 patients) microarrays. Rearrangements covering or in close proximity to SOX3 were identified in 3 patients (Figure 9, Supplemental Figures 2 and 3, and Table 1). Patient A contained two microduplications of approximately 123 kb and 85 kb, the former of which spanned the entire SOX3 gene. Previously published examples of SOX3 duplications in 46, XY males have been associated with pituitary hormone deficiencies and cognitive defects, whereas 46, XX female carriers have been unaffected (38, 47). Consistent with these data, there was no evidence of hypopituitarism or intellectual disability in this patient. X-inactivation studies showed no evidence for skewed inactivation in DNA derived from lymphocytes (data not shown). FISH analysis using a BAC clone covering the SOX3 gene consistently gave a stronger signal on one of the X chromosomes, indicative of tandem duplication (Supplemental Figure 3). The centromeric duplication is approximately 70 kb downstream from the SOX3 gene, very close to a previously described deletion-insertion breakpoint in individuals with X-linked hypoparathyroidism (61), which was postulated to have a positional effect on SOX3 expression. Patient B contained a single 343-kb microdeletion immediately upstream of SOX3. Unlike in patient A, the coding sequence of SOX3 is not affected in this patient, suggesting that altered regulation (and not increased dosage) of SOX3 is the cause of XX male sex reversal. Patient C has a more complex phenotype that also includes a scrotal hypoplasia, microcephaly, developmental delay, and growth retardation. This patient has a large (approximately 6-Mb) duplication that encompasses SOX3 and at least 18 additional distally located genes, overexpression of which probably contributes to the phenotypic complexity. Notably, the proximal breakpoint falls within the SOX3 regulatory region (62) and is close to the proximal SOX3 duplication breakpoint in patient A (Figure 9).
Figure 9. SOX3 rearrangements in 46, XX males.
An image derived from the UCSC genome browser ( http://genome.ucsc.edu) showing the location of the rearrangements identified in patients A–C in relation to the SOX3 gene. Also shown are rearrangements previously identified in healthy individuals (as recorded in the Database of Genomic Variants; http://projects.tcag.ca/variation), including CNVs and an inversion. It is noteworthy that duplication breakpoints in patients A and C are in close proximity to other known CNV/inversion breakpoints, suggesting structural or sequence motifs that may be responsible for recurrent rearrangements. Numbering corresponds to nucleotide position on the X chromosome (chrX), based on reference sequence hg18.
Table 1 .
Clinical summary of 46, XX male patients with SOX3 rearrangements
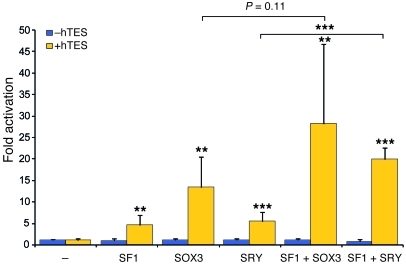
To investigate the possible mechanism of sex reversal in these patients, we performed transactivation assays using a human SOX9 testis enhancer sequence (hTES) that is homologous to the mouse TESCO element (18, 63). hSOX3 and hSRY activated hTES approximately 10- and 5-fold, respectively. Activation levels by these proteins were further enhanced in the presence of exogenous SF1, suggesting synergistic activation, similar to the murine system (Figure 10). Together, these data suggest that SOX3 gain of function in humans can cause XX male sex reversal.
Figure 10. SOX3 activates the hTES.
Human SOX3 (100 ng) activates hTES-luc (1.6 μg) (10–fold) compared with human SRY (100 ng) (5-fold) in CHO cells. In the presence of added SF1 (20 ng), further transactivation occurs with both SOX3 (25-fold) and SRY (20-fold) (n = 2–3). Error bars represent SD. Unless otherwise indicated, statistical comparisons were made between fold activations of expression vector and control empty vector. ***P < 0.001, **P < 0.01.
Discussion
Through characterization of a unique transgenic mouse line, we show that ectopic expression of Sox3 in uncommitted XX gonads is sufficient to divert the program of ovarian development toward testis formation, leading to XX males. Our data indicate that Sox3 functions as a molecular switch, activating the testis differentiation pathway via a mechanism that is functionally analogous to Sry. Furthermore, we provide the first evidence to our knowledge that SOX3 gain of function in humans can also lead to complete XX male sex reversal. Together, these data provide insight into the genetic mechanism of mammalian sex determination, the evolution of Sry, and the molecular pathology of disorders of sexual development.
Sox3 can function as an analog of Sry.
Molecular genetic studies carried out mostly in mice and humans, but supported by work in other species, provide strong evidence that mammalian testis differentiation is critically dependent on a threshold of SOX9 activity in supporting cell precursors that, once exceeded, suppresses the pro-ovarian differentiation program and promotes and then reinforces Sertoli cell and subsequent testis differentiation (24, 53, 64). As shown in mice, Sry along with its cofactor Sf1 bind the TESCO enhancer element upstream of Sox9 to mediate its initial upregulation (18). Based on the high sequence similarity of Sox3 and Sry across the HMG domain and the proposed common evolutionary ancestry of their cognate genes (32), we hypothesized that Sox3 causes XX male sex reversal in Sox3-SrTg mice by functioning as a surrogate for Sry. Our data provide several lines of evidence to support this view. First, adult Sr XX males exhibit complete sex reversal and in this respect are phenotypically identical to XX Sry transgenic adult males (7). Second, both mouse and human SOX3 exhibit in vitro transactivation properties very similar to those of Sry on TESCO and its human equivalent, including a requirement for Sf1. These are rather weak in contrast to the strong activation shown by Sox9. While a strong transactivation domain at the C-terminus of Sox3 can be demonstrated in other contexts (38), this appears not to operate in the cotransfection assays reported here. Presumably this is because any interaction with Sf1, which could be via the HMG box of Sox3 (and Sry), is not sufficient to elicit further recruitment of transcriptional coactivators to the complex bound to TESCO. If there are no specific requirements for features other than the HMG box, then this would provide an explanation for the rapid drift in the non-box sequences of SRY during evolution (see below and ref. 64). Third, there is a close correlation between expression of the Sox3 transgene and endogenous Sox9 in the developing XX Sox3-SrTg/+ gonad, suggesting that Sox3 may activate Sox9 expression in vivo. Although a small proportion of Sox9-positive cells do not express the transgene during early XX Tg/+ gonadogenesis, this is reminiscent of Sox9-positive/Sry-negative cells that have been identified in XY gonads at the same stage, which arise through prostaglandin D2 and Fgf signaling (21, 22), suggesting that non-cell-autonomous recruitment of cells to the Sertoli lineage also occurs in XX Tg/+ gonads. Indeed, this might be expected if the Sr transgene corresponds to a slightly weak allele of Sry. Fourth, the Sox3 transgene is expressed in the central gonad from 12 ts (Figures 4 and 8) and is therefore in the correct spatiotemporal location to influence the decision of the supporting cell precursors to adopt a male fate (16, 19, 65). Finally, we find that the Sr transgene also mimics Sry in vivo, because it depends on the presence of Sox9 to trigger testis development. This again supports the notion that Sox3 from the transgene acts like Sry and not Sox9.
In contrast to Sry, transgene expression in the gonads of the Sr line is maintained in Sertoli cells beyond the critical window of sex determination. This is not surprising given that Sry is switched off once Sox9 reaches the critical threshold to trigger Sertoli cell differentiation, and it is unlikely that Sox3 would respond in the same way (66, 67). Instead, Sox3 expression may be maintained by virtue of its integration next to Aldh1a1, which itself is activated directly or indirectly by Sox9 (see below). The functional consequence of persistent transgene expression is not clear. Sox3 could activate multiple male-specific target genes, in a manner that is analogous to the promotion of male development by Sox9, although this possibility is not supported by the failure of the Sr transgene to elicit testis development in the absence of Sox9. Alternatively, by virtue of its relatively poor transactivation activity in this context (compared with Sox9), Sox3 could interfere with Sox9 action through competitive binding to its target sites, including to the TESCO enhancer. However, although Sry expression is normally turned off, this is not essential, as shown with a constitutively active Sry transgene, and it naturally stays on for an extended period in some species (65, 68). The continuous expression of Sox3 may therefore have little or no impact on subsequent testis development.
Delayed testis development and incompletely penetrant sex reversal in Sr transgenic XX gonads.
While most Sr XX transgenic adults showed complete female-to-male sex reversal (77%), the gonadal development of XX Tg/+ embryos was often delayed compared with XY controls, as evidenced by lower Sox9 and Aldh1a1 expression, fewer Sox9-positive cells, and incomplete testis cord formation especially at the posterior poles (Figure 3). This phenomenon has also been observed with weak Sry alleles such as Y-POS, in which normal onset but lower levels of Sry expression and, in some cases, reduced efficacy of Sry isoforms leads to reduced Sox9 expression, a delay in testis cord development, and XY sex reversal on a C57BL/6 (B6) background (17, 69–72). qRT-PCR analysis of Sry and Sox3 transcript levels in 11.5-dpc XY and XX Tg/+ gonads, respectively, indicated that these genes are expressed at similar levels (data not shown), suggesting that expression level per se is not the primary cause of the developmental delay. A more likely explanation is that Sox3 is a less efficient activator of Sox9, given its weaker transactivation activity on the Sox9 enhancer element TESCO in the presence of Sf1 (Figure 4). We also noticed that increased contribution of the pro-ovarian B6 genetic background (73) reduced the penetrance of sex reversal in the Sr line, resulting in XX Tg/+ gonads that lacked transgene expression completely (Supplemental Figure 4). One explanation for this might be the earlier and more robust pro-ovarian program in B6 gonads preventing the reinforcement of transgene expression and the male pathway (30, 70, 73). Thus, Sox3, like Sry, may function in a narrow window of competence so that any genetic or environmental conditions that reduce its capacity to reach the critical threshold required for Sertoli cell differentiation lead to delayed development of testes, to ovotestis, or even to ovaries.
Aldh1a1 and transgene regulation in the gonads.
We were unable to detect Sox3 protein in the GR of wild-type XY or XX embryos, indicating that Sox3 is ectopically expressed in Sr gonads. Furthermore, EGFP was not expressed in the GRs of an independent Sox3 transgenic line (which does not exhibit XX male sex reversal), suggesting that regulatory elements flanking the Sr integration site control transgene expression in the gonad. Mapping studies indicated that the Sr transgene had inserted upstream of Aldh1a1, a gene with sexually dimorphic expression. Aldh1a1 is expressed at much higher levels in embryonic testis in comparison with ovaries at 12.5 dpc and 13.5 dpc (ref. 60 and Figure 7), and in the former is restricted to Sertoli cells and an undefined population of interstitial cells. The overlap of Sox3 and Aldh1a1 expression in XX transgenic gonads at these time points suggests that regulatory elements associated with testis-specific expression of Aldh1a1 may also control the Sr transgene. However, as discussed above, irreversible commitment to a male fate would almost certainly require transgene expression in the GR between 12 and 15 ts, prior to Aldh1a1 expression becoming sexually dimorphic. Intriguingly, Aldh1a1 and transgene expression overlap in XX Tg/+ gonads at 12–18 ts, although coexpression appears to be restricted to a subpopulation of Aldh1a1-expressing cells. These data suggest that transcriptional activity from Aldh1a1 in the context of the bipotential XX Tg/+ gonad may create a permissive chromatin environment that is necessary but not sufficient for transgene activation. Perhaps the latter occurs only in cells expressing Sf1 or another factor restricted to supporting cell precursors. However, after transgenic XX gonads switch to a testis differentiation program, transgene expression (unlike Sry) is upregulated in Sertoli cells. While the regulatory element(s) that direct Aldh1a1 expression are not known, a recent study indicates that Aldh1a1 is genetically downstream of Sox9 (60). This may have implications for the amplification and/or maintenance of Sox3 expression in Sr gonads and provides a direct link to Sox9, not only in establishing the male pathway but also in regulating transgene expression. Identification of Aldh1a1 gonadal enhancers may provide useful tools for transgenic analysis of gonad development and would likely lead to a better understanding of transgene regulation in Sr transgenic embryos.
Sox3, Sry, and the evolution of the mammalian sex determination switch.
Comparative cytogenetic and sequence analyses indicate that the mammalian sex chromosomes evolved from a pair of autosomes, termed the proto-X/Y (36). It has been proposed that Sry was generated by a dominant mutation of the proto-Y Sox3 allele in a mammalian ancestor approximately 200 million years ago, prior to divergence of the therian lineage (32, 36). The functional interchangeability of Sox3 and Sry demonstrated here indicates that their sole conserved domain, the HMG box, may be all that is critical for sex determination. This is in agreement with the (exclusive) conservation of the HMG domain in SRY genes, transgenic domain swap experiments in mice, and the predominance of SRY HMG mutations in XY females (35, 74, 75). The finding that Sox3 can promote male development when expressed in the genital ridge, in a manner that is comparable to Sry, adds further evidence against the hypothesis that Sox3 is required for ovary development or that Sry is a dominant negative version of Sox3, repressing the latter’s activity (or expression) (76). Instead it implies that the evolution of Sry involved a mutation event that led to expression of one allele of Sox3 (the proto-Sry) within the supporting cell lineage of the genital ridge. This mutation may have been subtle, with the proto-Sry retaining the conserved pattern of Sox3 expression within the CNS and elsewhere, or it could have involved a deletion or translocation, where most of the latter was lost. In several species examined, including mouse, rat, and human, Sry is expressed in parts of the developing and adult CNS (77–79), although given the rapid evolution of Sry flanking sequences, it will be a challenge to prove whether this reflects conservation of specific regulatory elements or the acquisition of new ones. Concomitant with the change in proto-Sry expression, Sox9 must have acquired the ability to respond to it. If, however, Sox9 was already involved in regulating its own expression within the Sertoli cell lineage of the therian ancestor, as shown in the mouse (18), then this capacity to respond to the proto-Sry may have already existed. To this end, recent evolutionary analyses have demonstrated that conserved elements within TESCO are present in all tetrapods, suggesting that mechanisms of Sox9 regulation are conserved and precede the evolution of Sry (63).
SOX3 rearrangement is associated with XX male sex reversal in humans.
Cases of SRY-negative XX male sex reversal are particularly intriguing, and their genetic basis has been debated extensively in the literature since SRY was identified almost 20 years ago. To date, loss of only one gene, RSPO1, has been shown to be associated with a clear XX male sex reversal condition, albeit in an unusual syndromic form of this disorder involving skin abnormalities (palmoplantar hyperkeratosis and predisposition to squamous cell carcinoma; ref. 26). Genomic rearrangements and mutations of SOX9, SOX10, and SF1 have also been detected in rare cases of XX male and XY female sex reversal (80–82), although the molecular etiology of most cases is unknown. In this study, we have identified SOX3 rearrangements in 3 patients with XX male sex reversal. Furthermore, we show that hSOX3 transactivates a human TES-like enhancer sequence, suggesting that human SOX3, like its murine counterpart, can functionally compensate for SRY. In contrast to the murine TESCO analysis, transactivation experiments using hTES were performed in CHO cells in which endogenous SF1 is already present. This is likely to function in synergy with hSOX3 and hSRY to give the observed limited transactivation in the absence of exogenous SF1 (82). Taken together, these data raise two possible mechanisms by which SOX3 could activate the male differentiation pathway in these patients: increased dosage or ectopic expression. Based on previous reports of SOX3 duplication in humans (which is associated with hypopituitarism and variable mild intellectual disability; refs. 38, 47) and the absence of Sox3 protein in murine bipotential gonads, it is unlikely that increased dosage of SOX3 in these 3 patients is the mechanism responsible for the sex reversal. It is more probable that rearrangement of the SOX3 locus, through duplication or deletion, leads to ectopic gonadal expression and subsequent activation of the male pathway at a critical stage of sex determination. As conserved elements that direct regional SOX3 expression are located across at least a 500-kb interval flanking the gene, it is plausible that these rearrangements activate SOX3 expression in the gonad, perhaps by generating a gonadal enhancer or ablation of a repression element (62). Indeed, the centromeric duplication breakpoint in patients A and C is in close proximity to a previously described deletion-insertion breakpoint in individuals with X-linked hypoparathyroidism, which was proposed to induce ectopic SOX3 expression (61). While it would be very interesting to assess the impact of SOX3 rearrangements on embryonic gonadal expression in humans, this is obviously not possible. However, based on our analysis of the Sr transgenic mice, these are exactly the kind of mutations that could reasonably be expected to occur in patients with XX male sex reversal. Notably, SOX3 expression was not detected in a lymphoblast cell line from patient B (the only patient from whom a cell line was available; data not shown), suggesting that the rearrangement has not caused widespread ectopic SOX3 expression, which instead may be relatively restricted and include the developing gonad.
Taken together, these data provide a molecular entry point into the etiology of XX male sex reversal in humans. It is worth noting the high frequency of SOX3 rearrangements in 46, XX male patients (19%), suggesting that this may be a significant cause of this disorder. Indeed, this may be an underestimate given that balanced rearrangements/translocations would not have been detected by our analysis. Further chromosomal and molecular analysis of DSD patient cohorts is therefore required to determine the relative contribution of SOX3 rearrangements to the underlying pathology of this poorly understood group of conditions.
Methods
Generation of Sr transgenic mice.
The transgene construct was derived from a modified BAC clone (RP23-174O19) containing an internal ribosome entry site–EGFP (IRES-EGFP) reporter cassette inserted into the 3′UTR of Sox3 using homologous recombination (83). The PCR product for recombineering contained a 5′ Sox3 homology arm, IRES-EGFP reporter, FRT site, kanamycin resistance cassette, FRT site, and 3′ Sox3 homology arm and was generated using Expand High Fidelity PCR System (Roche) amplification of a modified pIRES2-EGFP plasmid (BD Biosciences — Clontech; forward primer: CAGCTCTTCGCCCCCACCCCGCCCCACACCCCCCACGTTGGGACGCCTTGGCCCCTCTCCCTCCCCCCCCCCTAA; Reverse primer: CGCACTTCAAATTTCGGACCCGCAAAATTATCAGGACTCCAGCATATTGGGAAGTTCCTATTCTCTAGAAAGTATAGGAACTTCATTTATTCTGTCTTTTTATTGCCGTC (Sox3 homology sequence is in bold and FRT site in italics). The kanamycin resistance cassette was removed in bacteria prior to transgene preparation using an FLP recombinase expression vector. The 37-kb transgene spanned positions 58,136,727–58,172,890 of the X chromosome (July 2007 [NCBI 39/mm9] assembly) and contained approximately 26 kb and 8 kb of 5′ and 3′ Sox3 genomic sequence, respectively. It was generated by PmeI/AhdI digestion, purified by agarose gel electrophoresis and microdialysis, and injected into the pronucleus of C57BL/6 × CBA F1 zygotes. The Sr line was established from an XY founder animal and was maintained on a C57BL/6 × CBA mixed genetic background. Routine genotyping was performed by PCR amplification of genomic DNA derived from tail biopsies (weanlings) or embryonic/yolk sac tissue (embryos) using PCR (Sry primers: CACTGGCCTTTTCTCCTACC and CATGGCATGCTGTATTGACC; Gapdh primers: CTTGCTCAGTGTCCTTGCTG and ACCCAGAAGACTGTGGATGG; Egfp: ATGGTGAGCAAGGGCGAGGAGCTGTT and CTGGGTGCTCAGGTAGTGGTTGTC; cycle conditions: denaturation 95°C 2 minutes; amplification 95°C 30 seconds, 60°C 30 seconds, 72°C 3 minutes 30 seconds (35 cycles); final extension 72°C 7 minutes, 25°C 5 minutes). For the Sox9 rescue experiment, previously published lines that contain a homozygous floxed allele of Sox9 (Sox9fl/fl) (84) and a tamoxifen-inducible CreERT2 knocked into the Rosa26 locus (R26-CreERT2) (85) maintained on a F1 (C57BL/10 × CBA) genetic background were used. Tamoxifen (Sigma-Aldrich) was dissolved in ethanol at 100 mg/ml. To activate Cre-ERT2, the tamoxifen solution was diluted with sunflower oil to the final concentration of 10 mg/ml, and 0.3 ml of the diluted solution was administered by gavage to pregnant mice at 9.5 and 10.5 dpc. All animal experimentation was performed in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes and was approved by the University of Adelaide Animal Ethics Committee or by the UK Home Office.
FISH of chromosomes.
For mouse studies, FISH probes were directly labeled with spectrum orange/spectrum green using commercial kits (Vysis). Hybridization to metaphase spreads and fluorescence detection were performed on metaphase preparations as previously described (86). For human studies, FISH using BAC RP11-51C14 was carried out on metaphase spreads obtained from the 46, XX male patient according to previously described methods (87).
Cloning the Sr integration site.
The 5′ integration site was identified by GenomeWalker (BD Biosciences — Clontech) amplification of Tg/+ genomic DNA using nested Sox3 transgene primers (Sox3GSP1: ACAGCCTTGTGAGTAGGTATGCTCTTG; Sox3GSP2: CTTCTTTGTGCGGACCCTTATTCCAAG). The sequence immediately 5′ to the transgene integration site was identified as a long interspersed nuclear element (LINE) repeat using RepeatMasker ( http://www.repeatmasker.org/) and could not be unequivocally assigned to a genomic location using BLAST ( http://www.ncbi.nlm.nih.gov/) or BLAT (http://genome.ucsc.edu/) analysis. However, a LINE repeat that perfectly matched this transgene-flanking sequence was identified by megaBLAST analysis of the mouse genome sequence trace archive ( http://www.ncbi.nlm.nih.gov/blast/mmtrace.shtml), and a contig was assembled through reiterative interrogation. PCR amplification using unique flanking sequence and transgene primers generated a product from Tg/+ genomic DNA and not +/+ genomic DNA (primers: ACAGCCTTGTGAGTAGGTATGCTCTTG and TGGCTGTGGTAACCATTCATAAGGTAG; cycle conditions: denaturation 95°C 2 minutes; amplification 95°C 30 seconds, 60°C 30 seconds, 72°C 3 minutes [35 cycles]; final extension 72°C 7 minutes, 25°C 5 minutes). Sequencing confirmed the authenticity of this PCR product. To further confirm the integration site of the Sr transgene, standard Southern blot analysis was performed using a 32P-labeled chromosome 19–specific probe that spanned positions 20547495–20548089 (July 2007 assembly).
Immunohistochemistry, histology, and imaging.
Mouse embryos were dissected in PBS, fixed in 4% PFA/PBS, cryoprotected in 30% sucrose, and embedded in OCT medium. Sections (10 μm) were prepared using a Leica CM1900 cryostat. Blocking in PBS/0.1% Triton X-100/1% BSA/10% heat-inactivated horse serum was performed for 30 minutes, followed by overnight incubation at 4°C in the same solution containing diluted primary antibody. Sections were washed 3 times for 10 minutes in PBS and incubated in diluted secondary antibody at 4°C for 6 hours. After washing 3 times for 10 minutes each in PBS, slides were mounted in Prolong Gold Antifade (Molecular Probes, Invitrogen) and imaged using a Zeiss Axioplan 2 microscope and AxioCam MRm with Axiovision software. Antibodies and their corresponding dilutions were: rabbit anti-ALDH1A1 (Abcam Ab24343; 1:200), goat anti-SOX3 (R&D Systems; 1:100), rabbit anti-EGFP (Abcam; 1:2,000), goat anti-EGFP (Rockland; 1:400), rabbit anti-SOX9 (19) (1:1,000), goat anti-SOX9 (R&D Systems; 1:300), rabbit anti-FOXL2 (gift from Dagmar Wilhelm, Institute for Molecular Bioscience, The University of Queensland, Brisbane, Queensland, Australia; 1:1,000), Cy3–donkey anti-rabbit (Jackson ImmunoResearch Laboratories Inc.; 1:400), Cy5–donkey anti-goat (Jackson ImmunoResearch Laboratories Inc.; 1:400), Cy3–donkey anti-rabbit (Rockland; 1:1,000), Cy3–donkey anti-goat (Jackson ImmunoResearch Laboratories Inc.; 1:400) and Cy5–donkey anti-rabbit (Jackson ImmunoResearch Laboratories Inc.; 1:400) and Alexa Fluor 488–donkey anti-goat (Molecular Probes, Invitrogen; 1:400). Adult testis were fixed in Bouin’s fixative and stained using hematoxylin and eosin. Whole mount images were captured using a Nikon SMZ1000 GFP dissecting microscope with AnalySIS software and processed using Adobe Photoshop CS. Confocal images were captured on a Leica Sp5 Spectral scanning confocal microscope and processed using Leica Application Suite — Advanced Fluorescence 1.8.0 build 1346. Select images were subject to median measurements using a radius of 3 with 4 iterations to decrease graininess from the Cy5 fluorophore.
Whole mount and section in situ hybridization.
Whole mount in situ hybridization and in situ hybridization of 10-μm cryosections were performed as described previously (88, 89). The Aldh1a1 plasmid was a gift from Peter Koopman (Institute for Molecular Bioscience, The University of Queensland). The Sox9 and Amh plasmids were published previously (19, 90).
RNA extraction and qRT-PCR analysis.
RNA extractions were performed on GR explants and contained mesonephros unless stated otherwise. For 10.5- and 11.5-dpc embryos, the two genital ridges were pooled from each embryo, while individual gonads and associated mesonephros were isolated from 12.5-dpc and 13.5-dpc embryos. Samples were homogenized by brief sonication prior to being passed over QIAshredder columns (QIAGEN). RNA was isolated using the RNeasy Plus kit (QIAGEN) and analyzed for RNA integrity and concentration on a Bioanalyzer (Agilent). Fifty nanograms of each sample was reverse transcribed using the High Capacity RNA-to-cDNA kit (Applied Biosystems). qRT-PCR was performed using Fast SYBR Green Master Mix (Applied Biosystems) and run on an ABI 7500 StepOnePlus system. Gene-specific primer pairs were designed to cross intron-exon boundaries. The sequences and lengths of amplified products were: Aldh1a1 (158 bp) 5′-CCTTGCATTGTGTTTGCAGATG-3′ and 5′-GCTCGCTCAACACTCCTTTTC-3′; Sox9 (106 bp) 5′-GCAAGCTGGCAAAGTTGATCT-3′ and 5′-GCTGCTCAGTTCACCGATG-3′; EGFP (95 bp) 5′-ACGACGGCAACTACAAGACC-3′ and 5′-GTCCTCCTTGAAGTCGATGC-3′; and β-actin (89 bp) 5′-CTGCCTGACGGCCAGG-3′ and 5′-GATTCCATACCCAAGAAGGAAGG-3′. Efficiency of the amplification was verified by analysis of standard curves. No template and minus-RT controls were performed to ensure signal was cDNA-specific.
Cotransfection assays.
Transactivation assays using the mouse TESCO reporter were performed in COS7 cells as described previously (18). DNA encoding human SRY, SF1, and mouse and human SOX3 were contained within the pcDNA3 mammalian expression plasmid (Clontech). The hTES sequence was amplified from the BAC contig clone RP11-84E24 (GenBank AC007461), located on chromosome 17, by PCR using a forward oligonucleotide containing an XhoI restriction site (GATCATCCGCTCGAGCGGTGTTGAGAAGTGAACTGT) and a reverse oligonucleotide containing an AccI restriction site (GATGGCCGGTCGACCGGCCACTTGGCTCAAATCTCAC). The resultant PCR product was cloned into the multiple cloning site of the E1b-luc reporter construct (91). hTES cotransfection assays were conducted in the CHO cell line cultured in Dulbecco’s medium supplemented with 10% fetal bovine serum and l-glutamine in an atmosphere of 5% CO2. Briefly, cells were seeded at a density of 2.3 × 105 cells per well in a 6-well plate 24 hours prior to transfection. Forty-eight hours after transfection, the culture media was removed, cell lysate collected, and luciferase reporter activity measured according to the manufacturer’s instructions (Promega). Reporter activity was normalized to β-galactosidase as an internal control (Promega). Empty reporter, E1b-luc, transfection data were subtracted from each transfection condition to standardize data. Fold activations were determined by dividing by vector-alone transfection data.
Microarray analysis.
Genomic DNA from patients A and B was hybridized to an Affymetrix Genome-Wide Human SNP 6.0 array at the Australian Genome Research Facility according to the manufacturer’s instructions. Data analysis was performed with An R Object-oriented Microarray Analysis (AROMA) algorithm (92). Multiplex ligation–dependent probe amplification (MLPA) analysis to confirm the duplication was performed as previously described (93). Genomic DNA from patient C was hybridized to an Illumina Human1M-Duo DNA Analysis BeadChip at the Southern California Genotyping Consortium according to the manufacturer’s instructions. Data analysis was performed using CNV partition v2.3.4 on Illumina Genome Studio (94).
Clinical data.
Clinical data for patients A, B, and C are summarized in Table 1. These studies were approved by the Human Research Ethics Committee of the Royal Children’s Hospital, Melbourne, and the UCLA Institutional Review Board. Written informed consent was obtained from participants or their parents/guardians.
Patient A is a male of European descent who presented at 30 years of age for infertility. He had no significant past medical history and was healthy at time of presentation. His height was 165 cm, and he weighed 64 kg, with no abnormal symptoms. He underwent puberty between 14 and 15 years of age. Family history was not significant, and there was no consanguinity. Infertility was indicated by two spermograms, which confirmed azoospermia. Testicular biopsy was not performed. The patient was SRY-negative (data not shown). Parental DNA was not available.
Patient B is a 35-year-old of European descent. Gender dysphoria was reported from around 6 years, leading ultimately to gender reassignment surgery at age 26 years. Puberty commenced around age 14. At age 19, height was 167.5 cm, weight 73.5 kg, with no medical problems apart from ongoing gender identity issues. The external genitalia were normal male, apart from small, soft 6-ml testes (by orchidometer), subsequently measured at less than 4 ml (by orchidometer) at age 25 years. Shaving was infrequent, and there was little body hair, but axillary and pubic hair was normal (minimal abdominal extension). Primary hypogonadism was evident at age 19 years, with follicle-stimulating hormone (FSH) and luteinizing hormone (LH) both elevated, at 69 mIU/ml and 35 mIU/ml, respectively, and random free testosterone level being low, at 8.7 pg/ml. Serum prolactin was normal. Histological examination following gender reassignment surgery showed atrophic changes in the testes, with loss of normal spermatogenesis, thickening and hyalinization of the tubular basal lamina, and diminished number of interstitial cells. Spermatic cords were histologically normal, as was the penis. The patient was SRY-negative in both peripheral blood and testicular tissue. The SOX3 rearrangement was not present in his mother. DNA was not available for the father.
Patient C is a boy of Mexican origin who presented at 19 months with failure to thrive and developmental delay (height 75.2 cm [<5th percentile]). There were no significant problems during pregnancy or the newborn period, and cytogenetic analysis showed that this child was 46, XX, SRY-negative. Family history was significant only for learning disabilities. There was no consanguinity. No endocrine evaluation or parental DNA was available.
Statistics.
Statistical analysis for transactivation assays was conducted using a 2-tailed, paired Student’s t test, and P < 0.05 was considered significant. Error bars indicate the mean ± SEM in Figure 4 and SD in Figure 10.
Supplementary Material
Acknowledgments
The authors thank Peter Koopman and Josephine Bowles for sharing unpublished data and useful discussions, Terry Speed for assistance with microarray analysis, and Frank Grutzner for critical reading of the manuscript. We also thank Sandra Piltz, Mizuho Mamiya, Rhonda Hutchinson, Julie Scott, and the staff of the University of Adelaide Laboratory Animal Services for technical assistance. The authors also thank Tiago Rocha for patient A’s clinical evaluation and the staff of Unidade de Citogenética of Instituto Nacional de Saúde Ricardo Jorge for the chromosome analysis, as well as the contribution of the patients involved in the study. We gratefully acknowledge funding support from the Australian Research Council (P. Thomas), the Australian National Health and Medical Research Council (P. Thomas, S. White, A. Sinclair, and V. Harley), the UK Medical Research Council (U117512772) (R. Lovell-Badge), the Helen Macpherson Smith Trust (A. Sinclair), the Fundação para a Ciência e a Tecnologia (FCT; POCTI/SAU/97/2001) and the Centro de Investigação em Genética Molecular Humana da Universidade Nova de Lisboa (J. Goncalves), the Doris Duke Charitable Foundation (E. Vilain), and the Louis Jeantet Foundation (R. Lovell-Badge). Paul Thomas is a Pfizer Australia Research Fellow.
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2011;121(1):328–341. doi:10.1172/JCI42580.
References
- 1.Blackless M, et al. How sexually dimorphic are we? Review and synthesis. Am J Hum Biol. 2000;12(2):151–166. doi: 10.1002/(SICI)1520-6300(200003/04)12:2<151::AID-AJHB1>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 2.Temel SG, et al. Extended pedigree with multiple cases of XX sex reversal in the absence of SRY and of a mutation at the SOX9 locus. Sex Dev. 2007;1(1):24–34. doi: 10.1159/000096236. [DOI] [PubMed] [Google Scholar]
- 3.Sinclair AH, et al. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature. 1990;346(6281):240–244. doi: 10.1038/346240a0. [DOI] [PubMed] [Google Scholar]
- 4.Lovell-Badge R, Robertson E. XY female mice resulting from a heritable mutation in the primary testis-determining gene, Tdy. Development. 1990;109(3):635–646. doi: 10.1242/dev.109.3.635. [DOI] [PubMed] [Google Scholar]
- 5.Lim HN, Berkovitz GD, Hughes IA, Hawkins JR. Mutation analysis of subjects with 46, XX sex reversal and 46, XY gonadal dysgenesis does not support the involvement of SOX3 in testis determination. Hum Genet. 2000;107(6):650–652. doi: 10.1007/s004390000428. [DOI] [PubMed] [Google Scholar]
- 6.Koopman P, Munsterberg A, Capel B, Vivian N, Lovell-Badge R. Expression of a candidate sex-determining gene during mouse testis differentiation. Nature. 1990;348(6300):450–452. doi: 10.1038/348450a0. [DOI] [PubMed] [Google Scholar]
- 7.Koopman P, Gubbay J, Vivian N, Goodfellow P, Lovell-Badge R. Male development of chromosomally female mice transgenic for Sry. Nature. 1991;351(6322):117–121. doi: 10.1038/351117a0. [DOI] [PubMed] [Google Scholar]
- 8.Gubbay J, et al. A gene mapping to the sex-determining region of the mouse Y chromosome is a member of a novel family of embryonically expressed genes. Nature. 1990;346(6281):245–250. doi: 10.1038/346245a0. [DOI] [PubMed] [Google Scholar]
- 9.Just W, et al. Absence of Sry in species of the vole Ellobius. Nat Genet. 1995;11(2):117–118. doi: 10.1038/ng1095-117. [DOI] [PubMed] [Google Scholar]
- 10.Soullier S, Hanni C, Catzeflis F, Berta P, Laudet V. Male sex determination in the spiny rat Tokudaia osimensis (Rodentia: Muridae) is not Sry dependent. Mamm Genome. 1998;9(7):590–592. doi: 10.1007/s003359900823. [DOI] [PubMed] [Google Scholar]
- 11.Waters PD, Wallis MC, Marshall Graves JA. Mammalian sex — origin and evolution of the Y chromosome and SRY. Semin Cell Dev Biol. 2007;18(3):389–400. doi: 10.1016/j.semcdb.2007.02.007. [DOI] [PubMed] [Google Scholar]
- 12.Albrecht KH, Eicher EM. Evidence that Sry is expressed in pre-Sertoli cells and Sertoli and granulosa cells have a common precursor. Dev Biol. 2001;240(1):92–107. doi: 10.1006/dbio.2001.0438. [DOI] [PubMed] [Google Scholar]
- 13.Hacker A, Capel B, Goodfellow P, Lovell-Badge R. Expression of Sry, the mouse sex determining gene. Development. 1995;121(6):1603–1614. doi: 10.1242/dev.121.6.1603. [DOI] [PubMed] [Google Scholar]
- 14.Bullejos M, Koopman P. Spatially dynamic expression of Sry in mouse genital ridges. Dev Dyn. 2001;221(2):201–205. doi: 10.1002/dvdy.1134. [DOI] [PubMed] [Google Scholar]
- 15.Nagamine CM, Morohashi K, Carlisle C, Chang DK. Sex reversal caused by Mus musculus domesticus Y chromosomes linked to variant expression of the testis-determining gene Sry. Dev Biol. 1999;216(1):182–194. doi: 10.1006/dbio.1999.9436. [DOI] [PubMed] [Google Scholar]
- 16.Hiramatsu R, et al. A critical time window of Sry action in gonadal sex determination in mice. Development. 2009;136(1):129–138. doi: 10.1242/dev.029587. [DOI] [PubMed] [Google Scholar]
- 17.Bullejos M, Koopman P. Delayed Sry and Sox9 expression in developing mouse gonads underlies B6-Y(DOM) sex reversal. Dev Biol. 2005;278(2):473–481. doi: 10.1016/j.ydbio.2004.11.030. [DOI] [PubMed] [Google Scholar]
- 18.Sekido R, Lovell-Badge R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature. 2008;453(7197):930–934. doi: 10.1038/nature06944. [DOI] [PubMed] [Google Scholar]
- 19.Sekido R, Bar I, Narvaez V, Penny G, Lovell-Badge R. SOX9 is up-regulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev Biol. 2004;274(2):271–279. doi: 10.1016/j.ydbio.2004.07.011. [DOI] [PubMed] [Google Scholar]
- 20.Palmer SJ, Burgoyne PS. In situ analysis of fetal, prepuberal and adult XX----XY chimaeric mouse testes: Sertoli cells are predominantly, but not exclusively, XY. Development. 1991;112(1):265–268. doi: 10.1242/dev.112.1.265. [DOI] [PubMed] [Google Scholar]
- 21.Wilhelm D, et al. Sertoli cell differentiation is induced both cell-autonomously and through prostaglandin signaling during mammalian sex determination. Dev Biol. 2005;287(1):111–124. doi: 10.1016/j.ydbio.2005.08.039. [DOI] [PubMed] [Google Scholar]
- 22.Kim Y, et al. Fgf9 and Wnt4 act as antagonistic signals to regulate mammalian sex determination. PLoS Biol. 2006;4(6):e187. doi: 10.1371/journal.pbio.0040187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Capel B, Albrecht KH, Washburn LL, Eicher EM. Migration of mesonephric cells into the mammalian gonad depends on Sry. Mech Dev. 1999;84(1–2):127–131. doi: 10.1016/s0925-4773(99)00047-7. [DOI] [PubMed] [Google Scholar]
- 24.DiNapoli L, Capel B. SRY and the standoff in sex determination. Mol Endocrinol. 2008;22(1):1–9. doi: 10.1210/me.2007-0250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tomizuka K, et al. R-spondin1 plays an essential role in ovarian development through positively regulating Wnt-4 signaling. Hum Mol Genet. 2008;17(9):1278–1291. doi: 10.1093/hmg/ddn036. [DOI] [PubMed] [Google Scholar]
- 26.Parma P, et al. R-spondin1 is essential in sex determination, skin differentiation and malignancy. Nat Genet. 2006;38(11):1304–1309. doi: 10.1038/ng1907. [DOI] [PubMed] [Google Scholar]
- 27.Maatouk DM, DiNapoli L, Alvers A, Parker KL, Taketo MM, Capel B. Stabilization of beta-catenin in XY gonads causes male-to-female sex-reversal. Hum Mol Genet. 2008;17(19):2949–2955. doi: 10.1093/hmg/ddn193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jeays-Ward K, et al. Endothelial and steroidogenic cell migration are regulated by WNT4 in the developing mammalian gonad. Development. 2003;130(16):3663–3670. doi: 10.1242/dev.00591. [DOI] [PubMed] [Google Scholar]
- 29.Chang H, Gao F, Guillou F, Taketo MM, Huff V, Behringer RR. Wt1 negatively regulates beta-catenin signaling during testis development. Development. 2008;135(10):1875–1885. doi: 10.1242/dev.018572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jordan BK, Shen JH, Olaso R, Ingraham HA, Vilain E. Wnt4 overexpression disrupts normal testicular vasculature and inhibits testosterone synthesis by repressing steroidogenic factor 1/beta-catenin synergy. Proc Natl Acad Sci U S A. 2003;100(19):10866–10871. doi: 10.1073/pnas.1834480100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stevanovic M, Lovell-Badge R, Collignon J, Goodfellow PN. SOX3 is an X-linked gene related to SRY. Hum Mol Genet. 1993;2(12):2013–2018. doi: 10.1093/hmg/2.12.2013. [DOI] [PubMed] [Google Scholar]
- 32.Foster JW, Graves JA. An SRY-related sequence on the marsupial X chromosome: implications for the evolution of the mammalian testis-determining gene. Proc Natl Acad Sci U S A. 1994;91(5):1927–1931. doi: 10.1073/pnas.91.5.1927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Collignon J, et al. A comparison of the properties of Sox-3 with Sry and two related genes, Sox-1 and Sox-2. Development. 1996;122(2):509–520. doi: 10.1242/dev.122.2.509. [DOI] [PubMed] [Google Scholar]
- 34.Wallis MC, Waters PD, Graves JA. Sex determination in mammals — before and after the evolution of SRY. Cell Mol Life Sci. 2008;65(20):3182–3195. doi: 10.1007/s00018-008-8109-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bowles J, Schepers G, Koopman P. Phylogeny of the SOX family of developmental transcription factors based on sequence and structural indicators. Dev Biol. 2000;227(2):239–255. doi: 10.1006/dbio.2000.9883. [DOI] [PubMed] [Google Scholar]
- 36.Graves JA. Sex chromosome specialization and degeneration in mammals. Cell. 2006;124(5):901–914. doi: 10.1016/j.cell.2006.02.024. [DOI] [PubMed] [Google Scholar]
- 37.Pask AJ, Harry JL, Renfree MB, Marshall Graves JA. Absence of SOX3 in the developing marsupial gonad is not consistent with a conserved role in mammalian sex determination. Genesis. 2000;27(4):145–152. doi: 10.1002/1526-968X(200008)27:4<145::AID-GENE30>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 38.Woods KS, et al. Over- and underdosage of SOX3 is associated with infundibular hypoplasia and hypopituitarism. Am J Hum Genet. 2005;76(5):833–849. doi: 10.1086/430134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Solomon NM, et al. Array comparative genomic hybridisation analysis of boys with X linked hypopituitarism identifies a 3.9 Mb duplicated critical region at Xq27 containing SOX3. J Med Genet. 2004;41(9):669–678. doi: 10.1136/jmg.2003.016949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rizzoti K, Brunelli S, Carmignac D, Thomas PQ, Robinson IC, Lovell-Badge R. SOX3 is required during the formation of the hypothalamo-pituitary axis. Nat Genet. 2004;36(3):247–255. doi: 10.1038/ng1309. [DOI] [PubMed] [Google Scholar]
- 41.Laumonnier F, et al. Transcription factor SOX3 is involved in X-linked mental retardation with growth hormone deficiency. Am J Hum Genet. 2002;71(6):1450–1455. doi: 10.1086/344661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dee CT, Hirst CS, Shih YH, Tripathi VB, Patient RK, Scotting PJ. Sox3 regulates both neural fate and differentiation in the zebrafish ectoderm. Dev Biol. 2008;320(1):289–301. doi: 10.1016/j.ydbio.2008.05.542. [DOI] [PubMed] [Google Scholar]
- 43.Bylund M, Andersson E, Novitch BG, Muhr J. Vertebrate neurogenesis is counteracted by Sox1-3 activity. Nat Neurosci. 2003;6(11):1162–1168. doi: 10.1038/nn1131. [DOI] [PubMed] [Google Scholar]
- 44.Rizzoti K, Lovell-Badge R. SOX3 activity during pharyngeal segmentation is required for craniofacial morphogenesis. Development. 2007;134(19):3437–3448. doi: 10.1242/dev.007906. [DOI] [PubMed] [Google Scholar]
- 45.Weiss J, Meeks JJ, Hurley L, Raverot G, Frassetto A, Jameson JL. Sox3 is required for gonadal function, but not sex determination, in males and females. Mol Cell Biol. 2003;23(22):8084–8091. doi: 10.1128/MCB.23.22.8084-8091.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Raverot G, Weiss J, Park SY, Hurley L, Jameson JL. Sox3 expression in undifferentiated spermatogonia is required for the progression of spermatogenesis. Dev Biol. 2005;283(1):215–225. doi: 10.1016/j.ydbio.2005.04.013. [DOI] [PubMed] [Google Scholar]
- 47.Solomon NM, Nouri S, Warne GL, Lagerstrom-Fermer M, Forrest SM, Thomas PQ. Increased gene dosage at Xq26-q27 is associated with X-linked hypopituitarism. Genomics. 2002;79(4):553–559. doi: 10.1006/geno.2002.6741. [DOI] [PubMed] [Google Scholar]
- 48.De Santa Barbara P, et al. Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Mullerian hormone gene. Mol Cell Biol. 1998;18(11):6653–6665. doi: 10.1128/mcb.18.11.6653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hiramatsu R, et al. Regionally distinct potencies of mouse XY genital ridge to initiate testis differentiation dependent on anteroposterior axis. Dev Dyn. 2003;228(2):247–253. doi: 10.1002/dvdy.10379. [DOI] [PubMed] [Google Scholar]
- 50.Bagheri-Fam S, et al. Loss of Fgfr2 leads to partial XY sex reversal. Dev Biol. 2008;314(1):71–83. doi: 10.1016/j.ydbio.2007.11.010. [DOI] [PubMed] [Google Scholar]
- 51.Huang B, Wang S, Ning Y, Lamb AN, Bartley J. Autosomal XX sex reversal caused by duplication of SOX9. Am J Med Genet. 1999;87(4):349–353. doi: 10.1002/(SICI)1096-8628(19991203)87:4<349::AID-AJMG13>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- 52.Qin Y, Kong LK, Poirier C, Truong C, Overbeek PA, Bishop CE. Long-range activation of Sox9 in Odd Sex (Ods) mice. Hum Mol Genet. 2004;13(12):1213–1218. doi: 10.1093/hmg/ddh141. [DOI] [PubMed] [Google Scholar]
- 53.Sekido R, Lovell-Badge R. Sex determination and SRY: down to a wink and a nudge? Trends Genet. 2009;25(1):19–29. doi: 10.1016/j.tig.2008.10.008. [DOI] [PubMed] [Google Scholar]
- 54.Canning CA, Lovell-Badge R. Sry and sex determination: how lazy can it be? Trends Genet. 2002;18(3):111–113. doi: 10.1016/s0168-9525(01)02615-4. [DOI] [PubMed] [Google Scholar]
- 55.Vasiliou V, Pappa A, Estey T. Role of human aldehyde dehydrogenases in endobiotic and xenobiotic metabolism. Drug Metab Rev. 2004;36(2):279–299. doi: 10.1081/DMR-120034001. [DOI] [PubMed] [Google Scholar]
- 56.Duester G, Mic FA, Molotkov A. Cytosolic retinoid dehydrogenases govern ubiquitous metabolism of retinol to retinaldehyde followed by tissue–specific metabolism to retinoic acid. Chem Biol Interact. 2003;143–144:201–210. doi: 10.1016/s0009-2797(02)00204-1. [DOI] [PubMed] [Google Scholar]
- 57.Fan X, et al. Targeted disruption of Aldh1a1 (Raldh1) provides evidence for a complex mechanism of retinoic acid synthesis in the developing retina. Mol Cell Biol. 2003;23(13):4637–4648. doi: 10.1128/MCB.23.13.4637-4648.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Matt N, et al. Retinoic acid-dependent eye morphogenesis is orchestrated by neural crest cells. Development. 2005;132(21):4789–4800. doi: 10.1242/dev.02031. [DOI] [PubMed] [Google Scholar]
- 59.McClive PJ, Hurley TM, Sarraj MA, van den Bergen JA, Sinclair AH. Subtractive hybridisation screen identifies sexually dimorphic gene expression in the embryonic mouse gonad. Genesis. 2003;37(2):84–90. doi: 10.1002/gene.10231. [DOI] [PubMed] [Google Scholar]
- 60.Bowles J, et al. Male-specific expression of Aldh1a1 in mouse and chicken fetal testes: implications for retinoid balance in gonad development. Dev Dyn. 2009;238(8):2073–2080. doi: 10.1002/dvdy.22024. [DOI] [PubMed] [Google Scholar]
- 61.Bowl MR, et al. An interstitial deletion-insertion involving chromosomes 2p25.3 and Xq27.1, near SOX3, causes X-linked recessive hypoparathyroidism. J Clin Invest. 2005;115(10):2822–2831. doi: 10.1172/JCI24156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Navratilova P, Fredman D, Hawkins TA, Turner K, Lenhard B, Becker TS. Systematic human/zebrafish comparative identification of cis-regulatory activity around vertebrate developmental transcription factor genes. Dev Biol. 2009;327(2):526–540. doi: 10.1016/j.ydbio.2008.10.044. [DOI] [PubMed] [Google Scholar]
- 63.Bagheri-Fam S, Sinclair AH, Koopman P, Harley VR. Conserved regulatory modules in the Sox9 testis-specific enhancer predict roles for SOX, TCF/LEF, Forkhead, DMRT, and GATA proteins in vertebrate sex determination. Int J Biochem Cell Biol. 2010;42(3):472–477. doi: 10.1016/j.biocel.2009.07.001. [DOI] [PubMed] [Google Scholar]
- 64.Polanco JC, Koopman P. Sry and the hesitant beginnings of male development. Dev Biol. 2007;302(1):13–24. doi: 10.1016/j.ydbio.2006.08.049. [DOI] [PubMed] [Google Scholar]
- 65.Kidokoro T, et al. Influence on spatiotemporal patterns of a male-specific Sox9 activation by ectopic Sry expression during early phases of testis differentiation in mice. Dev Biol. 2005;278(2):511–525. doi: 10.1016/j.ydbio.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 66.Lee CH, Taketo T. Normal onset, but prolonged expression, of Sry gene in the B6.YDOM sex-reversed mouse gonad. Dev Biol. 1994;165(2):442–452. doi: 10.1006/dbio.1994.1266. [DOI] [PubMed] [Google Scholar]
- 67.Chaboissier MC, et al. Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development. 2004;131(9):1891–1901. doi: 10.1242/dev.01087. [DOI] [PubMed] [Google Scholar]
- 68.Hanley NA, et al. SRY, SOX9, and DAX1 expression patterns during human sex determination and gonadal development. Mech Dev. 2000;91(1–2):403–407. doi: 10.1016/S0925-4773(99)00307-X. [DOI] [PubMed] [Google Scholar]
- 69.Taketo T, Lee CH, Zhang J, Li Y, Lee CY, Lau YF. Expression of SRY proteins in both normal and sex-reversed XY fetal mouse gonads. Dev Dyn. 2005;233(2):612–622. doi: 10.1002/dvdy.20352. [DOI] [PubMed] [Google Scholar]
- 70.Palmer SJ, Burgoyne PS. The Mus musculus domesticus Tdy allele acts later than the Mus musculus musculus Tdy allele: a basis for XY sex-reversal in C57BL/6-YPOS mice. Development. 1991;113(2):709–714. doi: 10.1242/dev.113.2.709. [DOI] [PubMed] [Google Scholar]
- 71.Lee CH, Taketo T. Low levels of Sry transcripts cannot be the sole cause of B6-Y(TIR) sex reversal. Genesis. 2001;30(1):7–11. doi: 10.1002/gene.1026. [DOI] [PubMed] [Google Scholar]
- 72.Albrecht KH, Young M, Washburn LL, Eicher EM. Sry expression level and protein isoform differences play a role in abnormal testis development in C57BL/6J mice carrying certain Sry alleles. Genetics. 2003;164(1):277–288. doi: 10.1093/genetics/164.1.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Munger SC, Aylor DL, Syed HA, Magwene PM, Threadgill DW, Capel B. Elucidation of the transcription network governing mammalian sex determination by exploiting strain-specific susceptibility to sex reversal. Genes Dev. 2009;23(21):2521–2536. doi: 10.1101/gad.1835809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bergstrom DE, Young M, Albrecht KH, Eicher EM. Related function of mouse SOX3, SOX9, and SRY HMG domains assayed by male sex determination. Genesis. 2000;28(3–4):111–124. doi: 10.1002/1526-968X(200011/12)28:3/4<111::AID-GENE40>3.0.CO;2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lovell-Badge R, Canning C, Sekido R. Sex–determining genes in mice: building pathways. Novartis Found Symp. 2002;244:4–18; discussion 18–22, 35–42, and 253–257. [PubMed] [Google Scholar]
- 76.Graves JA. Interactions between SRY and SOX genes in mammalian sex determination. Bioessays. 1998;20(3):264–269. doi: 10.1002/(SICI)1521-1878(199803)20:3<264::AID-BIES10>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 77.Dewing P, et al. Direct regulation of adult brain function by the male-specific factor SRY. Curr Biol. 2006;16(4):415–420. doi: 10.1016/j.cub.2006.01.017. [DOI] [PubMed] [Google Scholar]
- 78.Mayer A, Lahr G, Swaab DF, Pilgrim C, Reisert I. The Y-chromosomal genes SRY and ZFY are transcribed in adult human brain. Neurogenetics. 1998;1(4):281–288. doi: 10.1007/s100480050042. [DOI] [PubMed] [Google Scholar]
- 79.Milsted A, Serova L, Sabban EL, Dunphy G, Turner ME, Ely DL. Regulation of tyrosine hydroxylase gene transcription by Sry. Neurosci Lett. 2004;369(3):203–207. doi: 10.1016/j.neulet.2004.07.052. [DOI] [PubMed] [Google Scholar]
- 80.Vidal VP, Chaboissier MC, de Rooij DG, Schedl A. Sox9 induces testis development in XX transgenic mice. Nat Genet. 2001;28(3):216–217. doi: 10.1038/90046. [DOI] [PubMed] [Google Scholar]
- 81.Polanco JC, Wilhelm D, Davidson TL, Knight D, Koopman P. Sox10 gain-of-function causes XX sex reversal in mice: implications for human 22q-linked disorders of sex development. Hum Mol Genet. 2010;19(3):506–516. doi: 10.1093/hmg/ddp520. [DOI] [PubMed] [Google Scholar]
- 82.Lin L, et al. Heterozygous missense mutations in steroidogenic factor 1 (SF1/Ad4BP, NR5A1) are associated with 46,XY disorders of sex development with normal adrenal function. J Clin Endocrinol Metab. 2007;92(3):991–999. doi: 10.1210/jc.2006-1672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lee EC, et al. A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics. 2001;73(1):56–65. doi: 10.1006/geno.2000.6451. [DOI] [PubMed] [Google Scholar]
- 84.Akiyama H, Chaboissier M C, Martin J F, Schedl A, de Crombrugghe B. The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev. 2002;16(21):2813–2828. doi: 10.1101/gad.1017802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Seibler J, et al. Rapid generation of inducible mouse mutants. Nucleic Acids Res. 2003;31(4):e12. doi: 10.1093/nar/gng012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Baker E, et al. Study of 250 children with idiopathic mental retardation reveals nine cryptic and diverse subtelomeric chromosome anomalies. Am J Med Genet. 2002;107(4):285–293. doi: 10.1002/ajmg.10159. [DOI] [PubMed] [Google Scholar]
- 87.Bruno DL, et al. Detection of cryptic pathogenic copy number variations and constitutional loss of heterozygosity using high resolution SNP microarray analysis in 117 patients referred for cytogenetic analysis and impact on clinical practice. J Med Genet. 2009;46(2):123–131. doi: 10.1136/jmg.2008.062604. [DOI] [PubMed] [Google Scholar]
- 88.Thomas PQ, Brown A, Beddington RS. Hex: a homeobox gene revealing peri-implantation asymmetry in the mouse embryo and an early transient marker of endothelial cell precursors. Development. 1998;125(1):85–94. doi: 10.1242/dev.125.1.85. [DOI] [PubMed] [Google Scholar]
- 89.Wilson LD, Ross SA, Lepore DA, Wada T, Penninger JM, Thomas PQ. Developmentally regulated expression of the regulator of G-protein signaling gene 2 (Rgs2) in the embryonic mouse pituitary. Gene Expr Patterns. 2005;5(3):305–311. doi: 10.1016/j.modgep.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 90.Swain A, Narvaez V, Burgoyne P, Camerino G, Lovell-Badge R. Dax1 antagonizes Sry action in mammalian sex determination. Nature. 1998;391(6669):761–767. doi: 10.1038/35799. [DOI] [PubMed] [Google Scholar]
- 91.Bernard P, Fleming A, Lacombe A, Harley VR, Vilain E. Wnt4 inhibits beta-catenin/TCF signalling by redirecting beta-catenin to the cell membrane. Biol Cell. 2008;100(3):167–177. doi: 10.1042/BC20070072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bengtsson H, Irizarry R, Carvalho B, Speed TP. Estimation and assessment of raw copy numbers at the single locus level. Bioinformatics. 2008;24(6):759–767. doi: 10.1093/bioinformatics/btn016. [DOI] [PubMed] [Google Scholar]
- 93.White SJ, et al. Two-color multiplex ligation-dependent probe amplification: detecting genomic rearrangements in hereditary multiple exostoses. Hum Mutat. 2004;24(1):86–92. doi: 10.1002/humu.20054. [DOI] [PubMed] [Google Scholar]
- 94.Yau C, Holmes CC. CNV discovery using SNP genotyping arrays. Cytogenet Genome Res. 2008;123(1–4):307–312. doi: 10.1159/000184722. [DOI] [PubMed] [Google Scholar]
- 95. Journal of the American Medical Association. Système International (SI) Conversion Factors for Selected Laboratory Components. http://jama.ama.assn.org/content/vol295/issue1/images/data/103/DC6/JAMA_auinst_si.dtl#dual . Updated September 28, 2001. [Google Scholar]
- 96.Rosner W, Auchus RJ, Azziz R, Sluss PM, Raff H. Position statement: utility, limitations, and pitfalls in measuring testosterone: an Endocrine Society position statement. J Clin Endocrinol Metab. 2007;92(2):405–413. doi: 10.1210/jc.2006-1864. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.