Abstract
Central chemoreception is the mechanism by which CO2/pH sensors regulate breathing in response to tissue pH changes. There is compelling evidence that pH-sensitive neurons in the retrotrapezoid nucleus (RTN) are important chemoreceptors. Evidence also indicates that CO2/H+-evoked adenosine 5′-triphosphate (ATP) release in the RTN, from pH-sensitive astrocytes, contributes to chemoreception. However, mechanism(s) by which RTN astrocytes sense pH is unknown and their contribution to chemoreception remains controversial. Here, we use the brain slice preparation and a combination of patch-clamp electrophysiology and immunohistochemistry to confirm that RTN astrocytes are pH sensitive and to determine mechanisms by which they sense pH. We show that pH-sensitive RTN glia are immunoreactive for aldehyde dehydrogenase 1L1, a marker of astrocytes. In HEPES buffer the pH-sensitive current expressed by RTN astrocytes reversed near EK+ (the equilibrium potential for K+) and was inhibited by Ba2+ and desipramine (blocker of Kir4.1-containing channels), characteristics most consistent with heteromeric Kir4.1–Kir5.1 channels. In bicarbonate buffer, the sodium/bicarbonate cotransporter also contributed to the CO2/H+-sensitive current in RTN astrocytes. To test the hypothesis that RTN astrocytes contribute to chemoreception by a purinergic mechanism, we used fluorocitrate to selectively depolarize astrocytes while measuring neuronal activity. We found that fluorocitrate increased baseline activity and pH sensitivity of RTN neurons by a P2-receptor–dependent mechanism, suggesting that astrocytes may release ATP to activate RTN chemoreceptors. We also found in bicarbonate but not HEPES buffer that P2-receptor antagonists decreased CO2 sensitivity of RTN neurons. We conclude that RTN astrocytes sense CO2/H+ in part by inhibition of a Kir4.1–Kir5.1-like current and may provide an excitatory purinergic drive to pH-sensitive neurons.
INTRODUCTION
A region of the caudal brain stem called the retrotrapezoid nucleus (RTN) contains a population of neurons that are highly pH sensitive in vivo and in vitro, are glutamatergic, and project to respiratory centers to influence breathing (Abbott et al. 2009; Mulkey et al. 2004). The mechanism by which these RTN chemoreceptors sense pH involves inhibition of an unidentified voltage-independent K+ conductance (Mulkey et al. 2007). The absence of an identified molecular pH sensor in RTN neurons has raised the possibility that multiple mechanisms contribute to chemoreception (Filosa and Putnam 2003; Pessia et al. 1996) and, despite the apparent intrinsic pH sensitivity of RTN neurons, it remains possible that local paracrine mechanisms contribute to chemoreception by the RTN.
There is increasing evidence that adenosine 5′-triphosphate (ATP), released by astrocytes, is an important mediator of chemoreception. Previous in vivo studies showed that hypercapnia evoked the discrete release of ATP within the RTN (Gourine et al. 2005). Further, application of ATP into the RTN stimulated respiratory output, whereas application of an ATP receptor antagonist (pyridoxal-phosphate-6-azophenyl-2′,4′-disulfonate [PPADS]) to the RTN lowered CO2 respiratory responses (Gourine et al. 2005). Recently, it was shown that RTN astrocytes respond to bath acidification with Ca2+-dependent, exocytosis-mediated, ATP release (Gourine et al. 2010). In addition, photostimulation of RTN astrocytes expressing channelrhodopsin-2 (ChR2) resulted in a robust increase in breathing (Gourine et al. 2010), strongly suggesting that astrocytes contribute to respiratory drive. This study also provided evidence suggesting that ATP release from astrocytes is a requisite signal for neuronal pH sensitivity (Gourine et al. 2010). At the cellular level, we have shown that purinergic signaling can activate pH-sensitive RTN neurons via P2Y receptors (Mulkey et al. 2006). However, we also found that the pH sensitivity of RTN neurons in HCO3−-free HEPES-buffered media was retained when ATP receptors were blocked with PPADS (100 μM) (Mulkey et al. 2004, 2006), indicating that ATP is not a requisite component of neuronal pH sensitivity. These results suggest that RTN neurons are intrinsically pH sensitive but that CO2-evoked release of ATP can modulate the activity of pH-sensitive neurons and contribute to the integrated output of the RTN (i.e., respiratory drive) during hypercapnia.
The goals of this study were to 1) confirm that RTN astrocytes are pH sensitive, 2) identify mechanism(s) by which these cells sense changes in pH, and 3) determine the extent to which they contribute to chemoreception. To make these determinations, we used the brain slice preparation and a combination of patch-clamp electrophysiology and immunohistochemistry to identity pH-sensitive glia and to determine the mechanism by which they sense changes in pH. To explore the possibility that pH-sensitive RTN astrocytes stimulate activity of pH-sensitive neurons by a purinergic mechanism, we determined the effect of fluorocitrate-mediated astrocyte activation on pH-sensitive neurons. We found that pH-sensitive RTN glia are protoplasmic astrocytes that sense H+, in part, by inhibition of a Kir4.1–Kir5.1-like current and may contribute to respiratory drive by increasing activity of pH-sensitive RTN neurons by a purinergic-dependent mechanism.
METHODS
Brain stem slices
Slices containing the RTN were prepared as previously described (Jiang et al. 2001; Kubo et al. 2005; Matthias et al. 2003). Briefly, neonatal rats (7–12 days postnatal) were decapitated under ketamine/xylazine anesthesia and transverse brain stem slices (300 μm) were cut using a microslicer (DSK 1500E; Dosaka, Kyoto, Japan) in ice-cold substituted Ringer solution containing (in mM): 260 sucrose, 3 KCl, 5 MgCl2, 1 CaCl2, 1.25 NaH2PO4, 26 NaHCO3, 10 glucose, and 1 kynurenic acid. Slices were incubated for about 30 min at 37°C and subsequently at room temperature in normal Ringer solution (in mM): 130 NaCl, 3 KCl, 2 MgCl2, 2 CaCl2, 1.25 NaH2PO4, 26 NaHCO3, and 10 glucose. Both substituted and normal Ringer solutions were bubbled with 95% O2-5% CO2.
Electrophysiology
Individual slices were transferred to a recording chamber mounted on a fixed-stage microscope (Zeiss Axioskop FS) and perfused continuously (∼2 ml min−1) with a bath solution composed of (in mM): 140 NaCl, 3 KCl, 2 MgCl2, 2 CaCl2, 10 N-2-hydroxyethylpiperazin-N′-2-ethanesulfonic acid (HEPES), 10 glucose; pH was adjusted between 6.9 and 7.5 by addition of HCl or NaOH. To test pH sensitivity of RTN astrocytes in bicarbonate-based buffer slices were perfused with normal Ringer solution bubbled with 95% O2-5% CO2 (bath pH = 7.45). The pH of the bicarbonate-based bath solution was decreased to a pH of about 6.8 by bubbling with 15% CO2 as previously described (Filosa and Putnam 2003).
All recordings were made with an Axopatch 200B patch-clamp amplifier, digitized with a Digidata 1322A A/D converter, and recorded using pCLAMP 10.0 software (Molecular Devices). Recordings were obtained at room temperature with patch electrodes pulled from borosilicate glass capillaries (Warner Instruments) on a two-stage puller (P89; Sutter Instrument) to a DC resistance of 4–7 MΩ when filled with an internal solution containing the following (in mM): 120 KCH3SO3, 4 NaCl, 1 MgCl2, 0.5 CaCl2, 10 HEPES, 10 EGTA, 3 Mg-ATP, 0.2% biocytin, and 0.3 GTP-Tris (pH 7.2); electrode tips were coated with Sylgard 184 (Dow Corning). Voltage-clamp recordings from glia were made at a holding potential of −80 mV and in the presence of tetrodotoxin (TTX, 0.1 μM) to block neuronal activity. Holding current, conductance, and current–voltage (I–V) relationships were determined using voltage steps between −40 and −150 mV. Neuronal firing rate histograms were generated by integrating action potential discharge in 10-s bins and plotted using Spike 5.0 software. It is important to recognize that in a whole cell recording circuit, recording cells with a low membrane resistance (e.g., passive astrocytes) in series with a large access resistance (Ra) can result in voltage-clamping error. In our experiments, we limited Ra to about 10 MΩ by preparing tissue from young animals; capacitance and Ra compensation (70%) were used to minimize voltage errors as previously described (Matthias et al. 2003). Recordings were discarded if Ra varied >10% during an experiment. A liquid junction potential of 10 mV was corrected off-line. Data were reported as means ± SE and were analyzed by the paired t-test or ANOVA and Dunnett's multiple comparison test when appropriate (P < 0.05).
Immunohistochemistry
Recorded slices were fixed in 4% paraformaldehyde in 0.1 M sodium phosphate buffer (pH 7.4) for 2 h at 4°C and rinsed in phosphate-buffered saline (PBS). Slices were blocked and permeabilized in PBS containing 5% normal goat serum and 0.4% Triton X-100 for 1 h at room temperature. Biocytin was detected by incubation in Alexa 488–conjugated strepavidin (1:1,000; Invitrogen) for 1 h at room temperature. After three PBS washes, the slices were incubated in primary antibodies overnight at 4°C, washed, and then incubated in secondary antibodies for 1 h at room temperature. Slices were washed in PBS and mounted in Vectashield. Images were collected on a Leica TCS SP2 confocal microscope equipped with 488-, 543-, and 633-nm laser lines and tunable emission wavelength detection. For each labeled slice, a biocytin-positive cell was identified and confocal z-stacks were collected sequentially for the other two channels to detect glial antigens. A cell was scored as immunopositive if the immunoreactivity for one or more glial markers was detected in the biocytin-positive cell body and processes. To determine background fluorescence of the tissue and nonspecific binding of secondary antibodies, we tested secondary antibodies without preincubation in primary antibody. These methods have been optimized for 300-μm slices sequentially detecting biocytin and glial antigens.
Drugs
Tetrodotoxin was purchased from Alomone Labs (Bethlehem, Israel); all other chemicals were obtained from Sigma (St. Louis, MO). Fluorocitrate was prepared as previously described (Holleran et al. 2001). Briefly, the Ba2+ salt of fluorocitric acid was dissolved in 0.1 M HCl and the Ba2+ was precipitated by the addition of Na2SO4 (0.1 M). This solution was centrifuged at 800 g for 10 min and the supernatant removed and diluted in HEPES-buffered medium to a final concentration of 100 μM (pH 7.3 by addition of 1 N NaOH). To confirm that Ba2+ was adequately removed during the preparation of fluorocitrate, we also prepared a solution of equal Ba2+ concentration minus fluorocitric acid and subjected it to the same precipitation protocol described earlier.
RESULTS
This study consists of two series of experiments. First, we made whole cell recordings to identify pH-sensitive RTN glia and to characterize the mechanism by which they sense pH. We initially identified glia by their small size, hyperpolarized membrane potential, and inability to form action potentials during depolarizing current injection; cellular identity was later confirmed using immunohistochemistry. In a second series of experiments, we made loose-patch recordings from pH-sensitive RTN neurons to determine the effects of fluorocitrate-mediated astrocyte activation on baseline activity and pH sensitivity of RTN neurons and to test effects of P2-receptor antagonists on neuronal CO2 sensitivity.
Astrocyte chemoreceptors
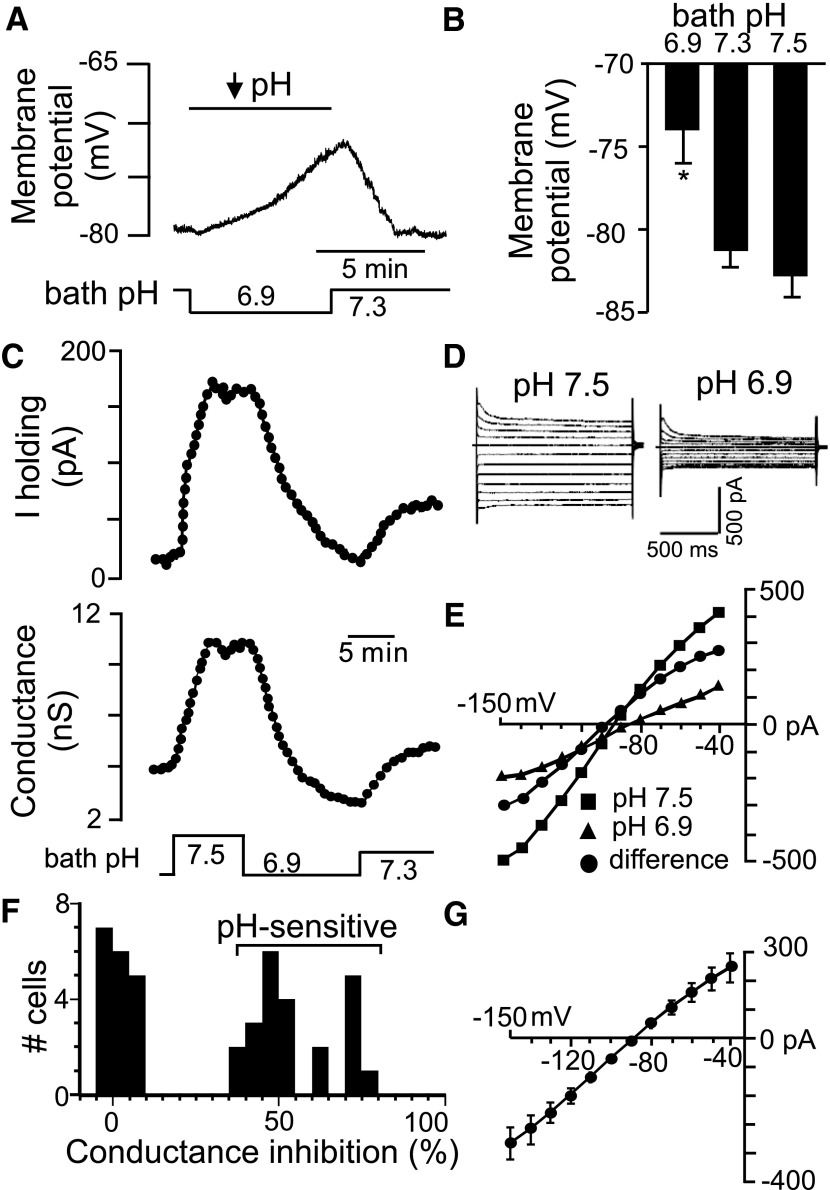
Previous sharp electrode recordings showed that ≤44% of RTN glia respond to extracellular acidification with membrane depolarization (Fukuda et al. 1978). In the whole cell configuration and HCO3−-free HEPES-buffered solution we found that roughly 20% of RTN glia were pH sensitive; cells were considered pH sensitive if they showed >10 mV/pH unit change in membrane potential, ≥40 pA change in holding current, or 35% change in conductance when cycling bath pH between 7.5 and 6.9. Based on these criteria, we found a bimodal distribution of pH-sensitive and -insensitive glial cells (Fig. 1F). In current-clamp, pH-sensitive RTN glia have a resting membrane potential of −80 ± 1 mV (n = 8) and respond to acidification from pH 7.5 to 6.9, with a depolarization of 8.8 ± 1.4 mV (n = 8) that occurs within about 6 min (Fig. 1, A and B). We hypothesize that the slow kinetics of pH-sensitive astrocytes is reflective of their modulatory role and likely helps maintain stable chemoreceptor output despite fast breath-by-breath oscillations in tissue pH (Millhorn et al. 1984). In contrast, pH-insensitive glia have a resting membrane potential of −74 ± 3 mV (n = 12) that did not vary over the same pH range.
Fig. 1.
Characteristics of pH-sensitive retrotrapezoid nucleus (RTN) glia in HEPES buffer. A: traces of membrane potential and bath pH show that acidification from 7.3 to 6.9 causes a reversible depolarization. B: summary of H+ effects on membrane potential of pH-sensitive astrocytes (n = 5, *P < 0.05). C: traces of holding current (top) and conductance (bottom) during changes in bath pH show that alkalization increases and acidification decreases outward current and conductance. D and E: current responses to voltage steps from −80 mV to between −40 and −150 mV (E) and corresponding current–voltage (I–V) relationships under alkaline and acidic conditions (D). To isolate the pH-sensitive I–V (♣), we subtract I–V relationships obtained at pH 6.9 (▴) from those recorded at pH 7.5 (■). F: percentage change in conductance during acidification from pH 7.5 to 6.9 observed from all glial cells recorded in the RTN. G: average I–V relationship (n = 5) of pH-sensitive current reverses near the equilibrium potential for K+ (EK+, arrow).
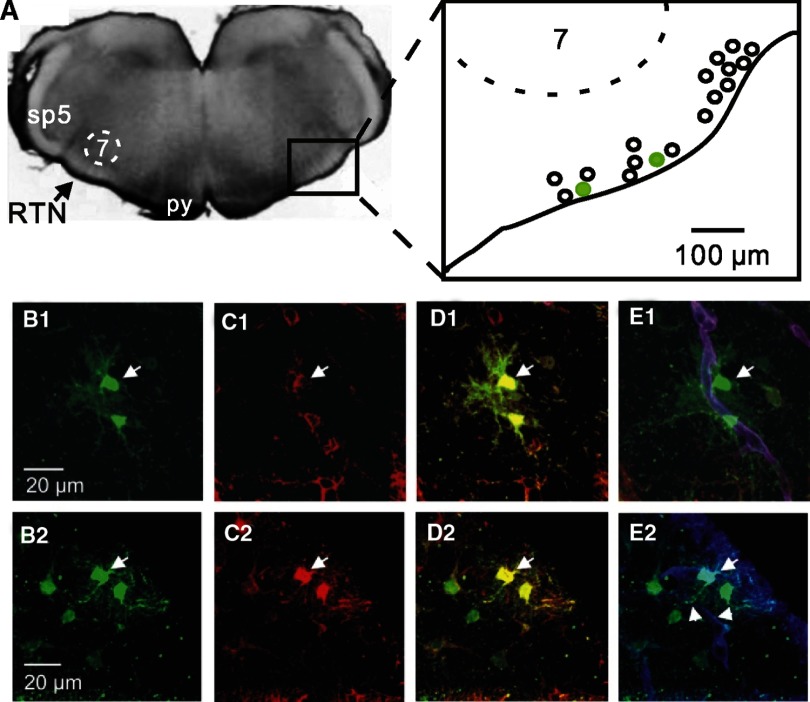
After recording, cells were filled with biocytin to characterize morphology and immunohistochemical phenotype. We found that 18 of 20 (90%) pH-sensitive glia were immunoreactive for aldehyde dehydrogenase 1L1 (Aldh1L1) (Fig. 2, A–C), an antigen that is expressed by protoplasmic astrocytes (Cahoy et al. 2008). In addition, pH-sensitive glia typically extended numerous fine radial projections near blood vessels (Fig. 2D) and to be dye-coupled (evidence of gap junctions) to other astrocytes (Fig. 2, A–C). These results support recent evidence that GFAP-expressing cells near the ventral surface are CO2 sensitive (Gourine et al. 2010) and indicate that pH-sensitive glia are protoplasmic astrocytes.
Fig. 2.
pH-sensitive RTN glia are gap junction coupled protoplasmic astrocytes. A, left: a transverse slice between bregma −11.6 to −11.2 mm that contains the RTN. Right: map showing relative locations of 18 pH-sensitive RTN astrocytes (green circles correspond to the cells shown in the bottom panels). B1 and B2: biocytin filled pH-sensitive RTN glial cells (green, arrows) and dye-coupled cells (other green cells). C1 and C2: aldehyde dehydrogenase 1L1-labeled (Aldh1L1, red) astrocytes. D1 and D2: merged images of the same cells in B and C show double-labeling for biocytin (green) and Aldh1L1 (red); the pH-sensitive astrocytes (arrows) and dye-coupled partners colocalize with Aldh1L1 labeling. E1 and E2: merged images of the same cells in B and C show double-labeling for biocytin (green) and laminin (blood vessels, blue). Note that pH-sensitive RTN astrocytes have cell bodies positioned near blood vessels (E1) and extend processes to blood vessels (truncated arrows in E2). Each image is the average projection of a confocal stack; around 30 images taken every 0.26 μm in the Z plane.
To further characterize electrical properties of pH-sensitive astrocytes and establish their role as intrinsic pH sensors, we made voltage-clamp recordings of whole cell currents in HEPES buffer at control pH 7.3 and during exposure to alkaline and acidic conditions. Note that these experiments were performed in TTX (0.1 μM) to block neuronal activity. Under control conditions, pH-sensitive astrocytes display high K+ conductance and exhibit fairly time- and voltage-independent responses to voltage steps. These properties are consistent with those of passive astrocytes described in the hippocampus (Matthias et al. 2003; Zhou et al. 2006) and brain stem (Grass et al. 2004). Astrocytes in the RTN respond to acidification from pH 7.5 to 6.9, with a decrease in holding current of 91.8 ± 10 pA (n = 23) and a decrease in conductance of 6.8 ± 0.8 nS (n = 23) (Fig. 1C). This corresponds with a decrease in whole cell current of about 53% at +40 mV. By comparison, RTN astrocytes are more pH sensitive than astrocytes in a nonrespiratory region like the hippocampus; passive hippocampal astrocytes respond to acidification from 7.4 to 6.0 with a decrease in whole cell current at +20 mV of only about 28% (Zhou et al. 2009). To confirm that the pH sensitivity of RTN astrocytes is determined by intrinsic membrane properties, rather than intercellular current spread through gap junctions, we tested the effects of carbenoxolone (a nonspecific gap junction blocker) on astrocyte pH sensitivity. Carbenoxolone (100 μM) increased the pH-sensitive current by roughly 18% (Supplemental Fig. S1B), suggesting that RTN astrocytes are intrinsically pH sensitive.1
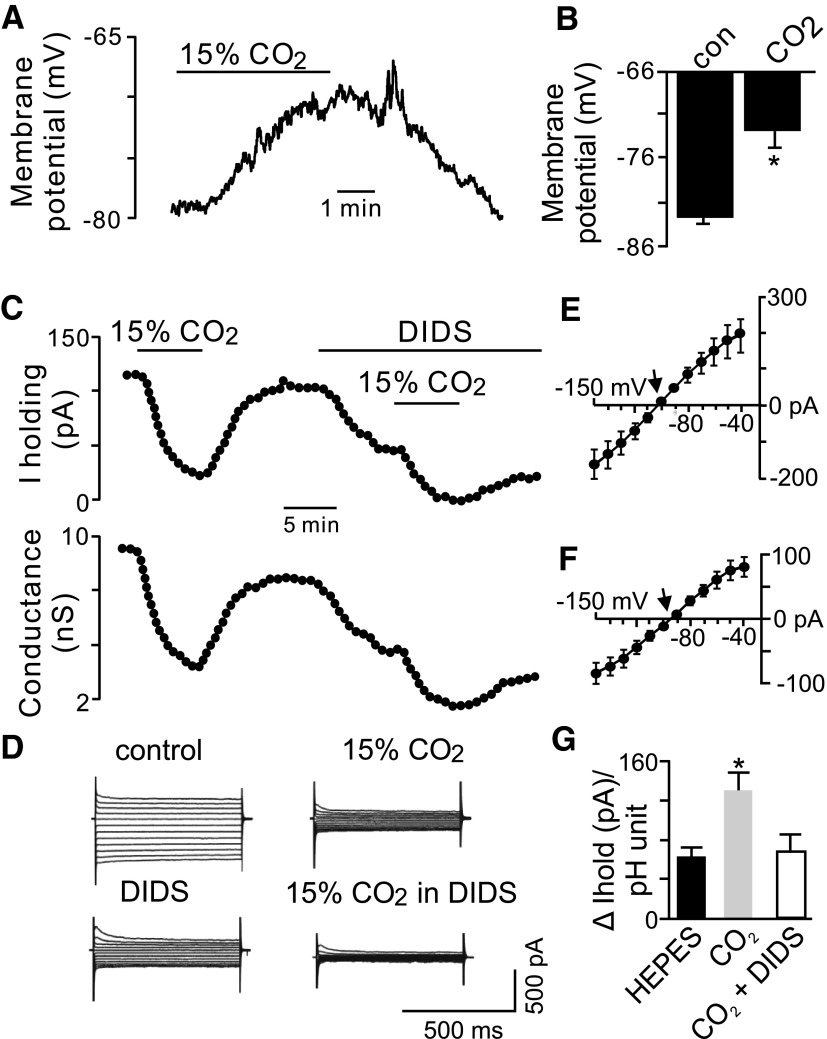
Astrocytes are known to express high levels of the electrogenic Na+/HCO3− cotransporter (NBC), which can generate an inward depolarizing current (i.e., HCO3− efflux) during bath acidification (Deitmer 1991; Munsch and Deitmer 1994). To determine whether activity of the NBC contributes to the pH-sensitive current in RTN astrocytes, we characterized CO2/H+ sensitivity of RTN astrocytes in normal Ringer solution (26 mM HCO3−, 5% CO2). In current-clamp, pH-sensitive RTN astrocytes had a membrane potential of −82 ± 1.2 mV (n = 3), which is about 2 mV hyperpolarized to resting potential of astrocytes in HCO3−-free HEPES buffer, and responded to hypercapnia (15% CO2, pHo = 6.8) with a membrane depolarization of 9 ± 2.3 mV (Fig. 3, A and B). In voltage-clamp, exposure to 15% CO2 decreased holding current from 139.8 ± 20 to 54.9 ± 8.9 pA (n = 8), which corresponded with a change in holding current of 130 ± 17 pA/pH unit, whereas in HEPES buffer acidification from 7.3 to 6.9 changed in the holding current 62.7 ± 6 pA/pH unit (Fig. 3G). In addition, the CO2/H+-sensitive current had a reversal potential of −105 mV (Fig. 3E), which is more negative than the reversal potential of the pH-sensitive current in HCO3−-free HEPES buffer, suggesting that activity of one or more HCO3− transporters contributes to pH sensitivity of RTN astrocytes. The most likely candidate is the electrogenic NBC, which can generate a depolarizing inward current (i.e., HCO3− efflux) during bath acidification (Chesler 2003; Deitmer 1991). Therefore we used diisothiocyanatostilbene-2,2′-disulfonic acid (DIDS, an NBC blocker) to isolate the contribution of this transporter to pH sensitivity of RTN astrocytes. At control pH = 7.4, exposure to DIDS (0.5 mM) decreased holding current by 41.6 ± 8 pA (n = 3) (Fig. 3C). In the continued presence of DIDS, exposure to 15% CO2 decreased holding current by 45.2 ± 13 pA (n = 3) (Fig. 3C), which corresponds with a change in holding current of 69.5 ± 20 pA/pH unit (Fig. 3G). The CO2/H+-sensitive current in DIDS reversed at −94 mV (i.e., near the equilibrium potential for K+ [EK+]) (Fig. 3, D and F), as expected for a current dominated by K+ conductance.
Fig. 3.
Characteristics of pH-sensitive RTN glia in bicarbonate buffer. A: traces of membrane potential shows that exposure to 15% CO2 causes a reversible membrane depolarization. B: summary of the effects of 15% CO2 on membrane potential pH-sensitive astrocytes (n = 3). C: trace of holding current shows that 15% CO2 decreases outward current and conductance. Under control conditions (i.e., 5% CO2, pHo = 7.45), exposure to diisothiocyanatostilbene-2,2′-disulfonic acid (DIDS, 0.5 mM) also decreased holding current close to 100 pA. In the continued presence of DIDS, a second exposure to 15% CO2 decreased holding current also by about 100 pA. D and E: current responses to voltage steps from −80 mV to between −40 and −150 mV during 5% and 15% CO2 exposure in bicarbonate buffer alone or in the presence of DIDS (D) and corresponding I–V relationships of the average CO2/H+-sensitive current (i.e., difference current) (n = 3) in bicarbonate buffer alone (E) or bicarbonate buffer with DIDS (F). Note that under control conditions the reversal potential of the CO2/H+-sensitive current is hyperpolarized to EK+ (arrow in E), but when activity of the electrogenic Na+/HCO3− cotransporter (NBC) is blocked with DIDS, the CO2/H+-sensitive current reverses near EK+ (arrow in F). G: summary data comparing effects of CO2/H+ on holding current (at a holding potential of −80 mV) in HEPES and bicarbonate-buffered media. Activity of the NBC significantly increased acid-induced changes in holding current compared with HCO3−-free HEPES solution or in the presence of DIDS (*P < 0.05).
The DIDS-sensitive current exhibited a reversal potential of −104 mV, which is slightly hyperpolarized to the predicted reversal potential for the NBC (ENBC). For example, the ENBC was calculated with the following equation (Newman 1991): ENBC = RT/F(n − 1) ln [Na+]i[HCO3−]in/[Na+]o[HCO3−]on, where n is the 2HCO3−:1Na+ stoichiometry and R (gas constant), T (temperature), and F (Faraday constant) have their usual meanings. For a temperature of 22°C, [Na+]o = 157 mM, [Na+]i = 4 mM, [HCO3−]o = 26 mM, and [HCO3−]i was estimated to be 22 mM using the Henderson–Hasselbalch equation as previously described (Gross et al. 2001); we calculated ENBC to be −99 mV. It is not clear why the DIDS-sensitive current reversed about 5 mV negative to ENBC, although DIDS has been shown to have nonspecific effects on anion exchangers and Cl− channels in glia (Munsch and Deitmer 1994), which may account for the discrepancy. To confirm that Cl− channels do not contribute to CO2 sensitivity of RTN astrocytes, we tested CO2 sensitivity in low chloride (50 mM) buffer.
The low Cl− solution was made isoosmotic by replacing 80 mM NaCl with an equal amount of Na-gluconate. We found the CO2/H+-sensitive current was not affected by chloride reduction. For example, there was no difference between the CO2/H+-induced change in holding current under control conditions (92.3 ± 38 pA; n = 3) and in low Cl− buffer (91.3 ± 37 pA; n = 3) (not shown). Likewise, amplitude and reversal potential of the CO2/H+-sensitive current were similar in control and low Cl− conditions (not shown).
In the absence of NBC activity (HCO3−-free HEPES buffer or Ringer solution with DIDS), the CO2/H+-sensitive current expressed by RTN astrocytes reversed near the equilibrium potential for K+ (EK+ = −97 mV), suggesting that one or more pH-sensitive K+ channels contribute to the pH sensitivity of RTN astrocytes. Astrocytes have been shown to express several types of pH-sensitive K+ channels, including background TASK channels and heteromeric Kir4.1–Kir5.1 channels. In particular, heteromeric Kir4.1–Kir5.1 channels are exquisitely sensitive to pH in the physiological range (Casamassima et al. 2003; Xu et al. 2000a), are preferentially expressed by astrocytes (Hibino et al. 2004; Tang et al. 2009), and in situ hybridization shows detectable levels of both Kir4.1 and Kir5.1 mRNA in the RTN (Wu et al. 2004). Therefore we consider heteromeric Kir4.1–Kir5.1 channels the most likely molecular pH sensor in RTN astrocytes.
Heteromeric Kir4.1–Kir5.1 channels confer pH sensitivity to RTN astrocytes
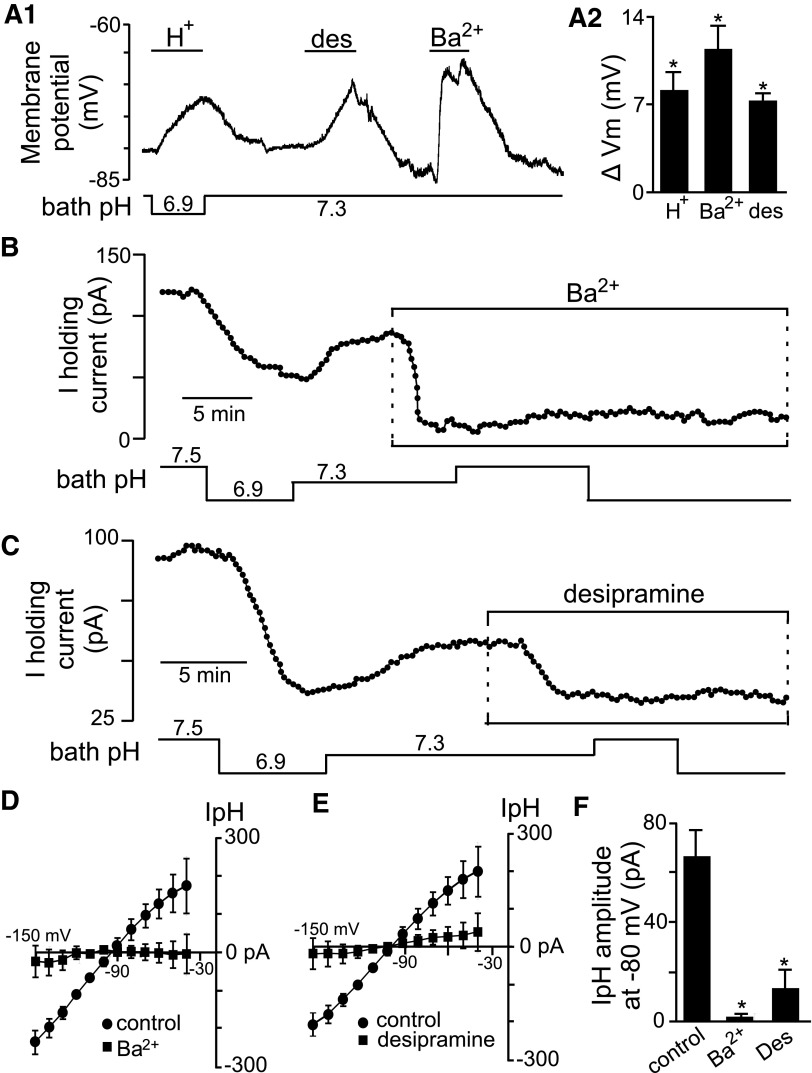
The possible contribution of Kir channels can easily be distinguished from background K+ channels based on Ba2+ sensitivity (Olsen and Sontheimer 2008); most Kir channels, but not background K+ channels, are fully inhibited by micromolar concentrations of Ba2+ (for review of background and Kir channel pharmacology see Goldstein et al. 2005; Kubo et al. 2005; Patel and Honore 2001). Therefore to determine whether heteromeric Kir4.1–Kir5.1 channels contribute to pH sensitivity in RTN astrocytes, we first determine effects of Kir channel inhibition by Ba2+ (100 μM) on membrane potential and the pH-sensitive current. In current-clamp, Ba2+ depolarized the membrane potential by 16.7 ± 3.0 mV (n = 4) (Fig. 4A). In voltage-clamp (at −80 mV, TTX), Ba2+ decreased outward current by 86.5 ± 5 pA (n = 4) (Fig. 4B) and blocked the pH-sensitive current by 96.5 ± 3.5% (n = 4) (Fig. 4, B, D, and F). Note that if a more Ba2+-sensitive background channel like TWIK (Ba2+ half-maximal inhibitory concentration [IC50] = 100 μM; Patel and Honore 2001) or TASK-1 (Ba2+ IC50 = 350 μM; Patel and Honore 2001) contributed to the pH-sensitive current in RTN astrocytes, we would expect that only a portion (at best ∼50%) of the pH-sensitive current would be blocked by 100 mM Ba2+. However, we show that this concentration of Ba2+ virtually eliminated the pH-sensitive current, suggesting that background K+ channels do not confer pH sensitivity to RTN astrocytes. Inhibition of other voltage-gated K+ channels with tetraethylammonium (TEA, 10 mM) or 4-aminopyridine (4-AP, 50 μM) had no effect on the pH-sensitive current in RTN astrocytes (Supplemental Fig. S1A). These results indicate that one or more Kir channels contribute to the pH-sensitive current in RTN astrocytes.
Fig. 4.
pH sensitivity of RTN glia involves heteromeric Kir4.1 channels. A1: membrane potential trace shows that inhibition of Kir channels mimics the effects of H+; Ba2+ (100 μM) and desipramine (des, 100 μM) depolarize membrane potential by about 18 and 9 mV, respectively. A2: average depolarization of RTN pH-sensitive astrocyte when exposed to either H+ (acidification from 7.3 to 6.9, n = 5), 100 μM Ba2+ (n = 4), or 100 μM desipramine (n = 5). B: trace of holding current shows that H+ decreases outward current. At pH 7.3, Ba2+ decreases holding current and blocks effects of pH on holding current. C: trace of holding current from a pH-sensitive astrocyte shows desipramine decreases holding current and blocks effects of pH on holding current. D: average (n = 4) I–V relationship of the pH-sensitive current under control conditions and in the presence of 100 μM Ba2+ shows negligible pH-sensitive current in Ba2+. E: average (n = 4) I–V relationship of the pH-sensitive current under control conditions and in the presence of desipramine shows negligible pH-sensitive current in desipramine. F: average pH-sensitive current amplitude (at −80 mV) recorded from RTN astrocytes during acidification from pH 7.5 to 6.9, exposure to Ba2+ (n = 3), or desipramine (n = 4) (*P < 0.05).
To differentiate the contribution of Kir4.1–Kir5.1 channels from other Kir channels, we inhibited Kir4.1-containing channels (i.e., homomeric and heteromeric channels) with desipramine (100 μM). At this concentration, desipramine inhibits channels containing Kir4.1 subunits, with minimal effects on other pH-sensitive Kir channels (Su et al. 2007; Tang et al. 2009). Our results indicate that desipramine mimics effects of acidification; in current-clamp exposure to desipramine depolarized membrane potential by 8.4 ± 3.0 mV (n = 4) (Fig. 4A) and in voltage-clamp it decreased holding current by 35.0 ± 13 pA (n = 4) (Fig. 4C) and the pH-sensitive current by 72.7 ± 21.3% (n = 4) (Fig. 4, C, E, and F). It should be noted that desipramine is also a tricyclic antidepressant and may have nonspecific effects on neuronal activity. However, RTN astrocytes are not responsive to norepinephrine or serotonin (DK Mulkey, unpublished observations), suggesting the effects of desipramine on these cells is mediated by inhibition of Kir4.1-containing channels. These results suggest that heteromeric Kir4.1–Kir5.1 channels confer pH sensitivity to RTN astrocytes.
RTN astrocytes activate pH-sensitive neurons by a purinergic mechanism
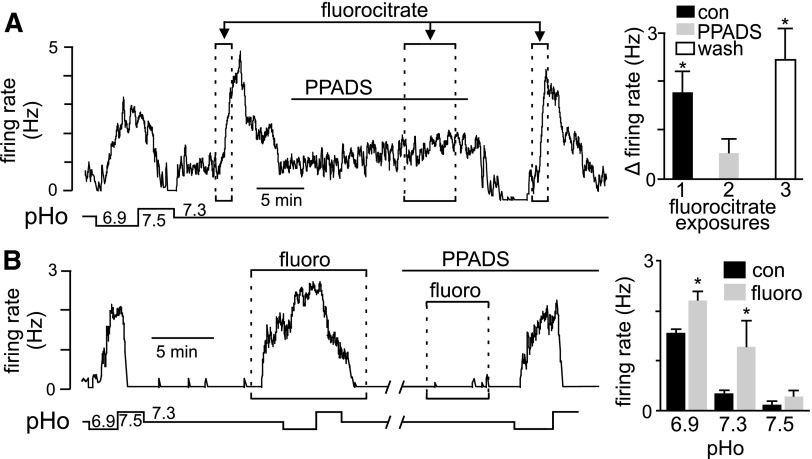
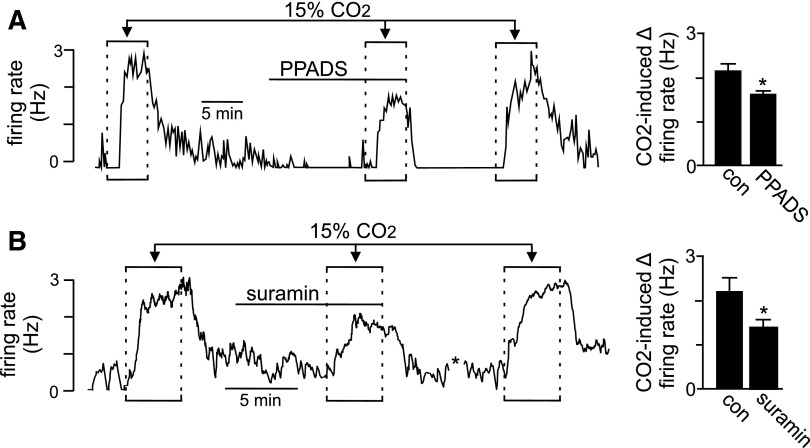
Recent evidence suggests that a subset of RTN astrocytes contribute to the mechanism of chemoreception by sensing CO2/H+ and releasing ATP to activate pH-sensitive neurons and increase respiratory drive (Gourine et al. 2010). These results are supported by our evidence that roughly 20% of RTN astrocytes are intrinsically pH sensitive. To test the possibility that pH-sensitive astrocytes modulate activity of pH-sensitive neurons, we paralleled previous in vivo experiments by using fluorocitrate to selectively depolarize astrocytes (Erlichman et al. 1998; Holleran et al. 2001) while recording activity of pH-sensitive neurons. Fluorocitrate is a metabolic toxin that is selectively taken up by astrocytes, where it can initiate membrane depolarization (Erlichman and Leiter 2010), presumably by energy depletion and loss of cytoplasmic K+. At subtoxic doses, fluorocitrate has been shown to reversibly depolarize RTN glia and increase ventilatory sensitivity to CO2 (Erlichman et al. 1998; Holleran et al. 2001), by yet unknown mechanisms. We confirmed that fluorocitrate (50 μM) can reversibly depolarize pH-sensitive astrocytes (Supplemental Fig. S2). We also confirmed the effects of fluorocitrate are not due to residual Ba2+ by determining that Ba2+ precipitation control solution, prepared in a manner similar to fluorocitrate, had no effect on neuronal or glial membrane potential. We found that fluorocitrate evoked a large and reversible neuronal activation (Fig. 5, A and B). Importantly, a second exposure to fluorocitrate, this time in the presence of an ATP receptor antagonist (PPADS, 100 μM), had no effect on neuronal activity (Fig. 5A). In addition, fluorocitrate also increased neuronal pH sensitivity; in the continued presence of fluorocitrate, responsiveness to acidification increased by 0.7 ± 0.2 Hz (n = 3), compared with acid alone (Fig. 5B). These results also confirm previous results that P2 receptors are not required for pH sensitivity of RTN neurons in slices incubated in HEPES buffer (Mulkey et al. 2004, 2006); pH sensitivity of RTN neurons was wholly retained in the presence of PPADS (Fig. 5B). It has been suggested that CO2 and not H+ per se is the stimulus required for ATP release from RTN astrocytes (Spyer 2009). Therefore we tested the effect of PPADS and another P2-receptor antagonist (suramin, 100 μM) on CO2/H+ sensitivity in bicarbonate-buffered medium. As shown in Fig. 6, exposure to 15% CO2 alone increased neuronal activity 2.2 ± 0.1 Hz (n = 12). After washing to 5% CO2 exposure to PPADS (100 μM) significantly decreased baseline activity from 0.13 ± 0.05 to 0.02 ± 0.01 Hz (n = 8). Likewise, suramin (100 μM) decreased baseline activity 0.50 ± 0.1 Hz to 0.3 ± 0.1 Hz (n = 4). These results suggest that in bicarbonate buffer (pH 7.3) RTN chemoreceptors receive tonic excitatory purinergic input. Prior to testing CO2/H+ sensitivity in PPADS or suramin, a depolarizing current (∼1 nA) was delivered to the neuron to approximate the control level of activity. In the continued presence of PPADS (with baseline activity adjusted by DC current injection to near control levels), a second exposure to 15% CO2 increased neuronal activity by 1.6 ± 0.1 Hz (n = 8); this response was significantly less than the CO2 response under control conditions (initial response or in wash) (Fig. 6A). In the presence of suramin (with baseline activity adjusted to near control levels), a second exposure to 15% CO2 increased neuronal activity by 1.4 ± 0.1 Hz; this response was significantly less than the CO2 response under control conditions (Fig. 6B). These results indicate that P2-receptor antagonists PPADS and suramin decreased CO2/H+ sensitivity. To the extent that fluorocitrate selectively affects astrocytes (Erlichman and Leiter 2010), these results suggest that astrocytes can release ATP and activate pH-sensitive neurons, presumably by a P2Y-dependent mechanism (Mulkey et al. 2006), to increase chemoreceptor output. These results also lend support to the hypothesis that pH-sensitive astrocytes contribute to chemoreception by increasing activity of pH-sensitive neurons.
Fig. 5.
RTN astrocytes increase baseline activity and pH sensitivity of RTN chemoreceptors by a P2-receptor–dependent mechanism. A: traces of pH and firing rate (Hz) from a pH-sensitive neuron shows a characteristic response to H+: increasing pH to 7.5 inhibited activity, whereas acidification to pH 6.9 increased firing rate >2 Hz. At a control pH of 7.3, exposure to fluorocitrate (fluoro, 50 μM) evoked a large and reversible increase in neuronal activity. A second exposure to fluorocitrate, this time in the presence of a P2-receptor antagonist pyridoxal-phosphate-6-azophenyl-2′,4′-disulfonate (PPADS), 100 μM, blocked the effects of fluorocitrate. Fluorocitrate responsiveness returned in wash. B: traces of pH and firing rate from a pH-sensitive RTN neuron exposed to acidic and alkaline conditions in the presence and absence of fluorocitrate (fluoro, 50 μM) or PPADS (100 μM). Fluorocitrate alone stimulated neuronal activity. In the continued presence of fluorocitrate, acidification further increased firing rate by about 1 Hz, whereas alkalization decreased activity. Note that the effects of pH but not fluorocitrate were retained in the presence of PPADS. Bar graphs summarize the effects of fluorocitrate in the presence of PPADS (top, n = 5) and in acidic and alkaline conditions (bottom, n = 3) (*P < 0.05).
Fig. 6.
In bicarbonate-buffered solution astrocytes contribute to chemosensitivity by a purinergic mechanism. A: trace of firing rate shows the response of an RTN neuron to increases in CO2 from 5 to 15% in bicarbonate buffer alone or in the presence of PPADS. An initial exposure to 15% CO2 increased the firing rate about 2.8 Hz. After returning to 5% CO2, exposure to PPADS (100 μM) decreased baseline firing rate, suggesting that in bicarbonate buffer RTN chemoreceptors receive tonic excitatory purinergic input. A depolarizing current (∼1 nA) was delivered to the neuron to increase firing rate to near control levels. In the continued presence of PPADS (with baseline activity adjusted by DC current injection to near control levels), a second exposure to 15% CO2 increased the firing rate about 1.5 Hz. After washing, PPADS CO2 sensitivity returned to initial levels. Bar graph on the right summarizes the firing rate response of RTN neurons (n = 9) to 15% CO2 in control and PPADS (*P < 0.05). B: firing rate trace shows the response of a RTN chemoreceptor to 15% CO2 in bicarbonate-buffered solution alone and in the presence of suramin (100 μM). An initial exposure to 15% CO2 increased the firing rate about 2.7 Hz. As was observed with PPADS, exposure to suramin (100 μM) in 5% CO2 decreased baseline firing rate. A depolarizing current (∼1 nA) was delivered to the neuron to increase firing rate to near control levels. In the continued presence of suramin (with baseline activity adjusted by DC current injection to near control levels), a second exposure to 15% CO2 increased firing rate about 1.7 Hz. After washing, suramin CO2 sensitivity returned to initial levels. The asterisk designates a 5-min break. Bar graph on the right summarizes the firing rate response of RTN neurons (n = 5) to 15% CO2 in control and suramin (*P < 0.05).
DISCUSSION
Neuron–glia communication has emerged in recent years as a fundamental process in the brain that can be an important determinant of neuronal activity. However, by most accounts the activity of astrocytes is thought to be under the strict control of neuronal input (Fellin et al. 2006). Here we show that RTN astrocytes sense H+ in part by inhibition of heteromeric Kir4.1–Kir5.1 channels and stimulate activity of RTN chemoreceptors by a P2-receptor–dependent mechanism. Our evidence also indicates that purinergic input is not required for RTN chemoreceptor function. These results identify the most likely mechanism by which RTN astrocytes sense pH and build on recent evidence that astrocytes contribute to respiratory drive by a purinergic mechanism (Gourine et al. 2010).
Molecular mechanism underlying pH sensing by RTN astrocytes
Fukuda et al. (1978) showed >30 yr ago that the RTN contains a population of pH-sensitive nonspiking cells and more recent evidence shows these pH sensors express the NBC (Ritucci et al. 2005), thus narrowing the scope of candidates to either astrocytes or oligodendrocytes. Our data indicate that pH-sensitive RTN glia are protoplasmic astrocytes; specifically, these cells have a hyperpolarized membrane potential; high K+ conductance; exhibit an electrically passive profile; and express Aldh1L1, a marker of protoplasmic astrocytes (Cahoy et al. 2008).
Several pH-sensitive channels have been proposed to contribute to the mechanism of chemoreception, including Ca+2-activated K+ channels (KCa) (Wellner-Kienitz et al. 1998), voltage-sensitive K+ channels (e.g., transient and delayed rectifying K+ channels) (Filosa and Putnam 2003; Putnam et al. 2004), background TASK channels (Bayliss et al. 2001; Mulkey et al. 2004), and Kir channels (Schultz et al. 2003; Xu et al. 2000a; Zhu et al. 2000). All of these channels are expressed in various types of glial cells and conceivably contribute to the pH sensitivity of RTN astrocytes. Our results show that the pH-sensitive current in RTN astrocytes can be blocked with 100 μM Ba2+, which will fully block most Kir channels, with only modest effects on background K+ channels (e.g., TASK channels) (Goldstein et al. 2005; Kubo et al. 2005; Patel and Honore 2001). In addition, the pH-sensitive current in RTN astrocytes was not blocked by TEA (10 mM) or 4-AP (50 μM), which can inhibit other voltage- and Ca2+-dependent K+ channels, thus narrowing the scope of candidate molecular sensors to one or more Kir channels.
The Kir family of channels consists of 16 different channel subunits grouped into seven subfamilies (Kir1.x–Kir7.x) based on sequence homology (Olsen and Sontheimer 2008). All Kir channels are composed of four α-subunits assembled as homomeric or heteromeric channels that show varying degrees of rectification, single-channel conductance, and pharmacology. A common feature of most Kir channels is high sensitivity to low micromolar concentrations of Ba2+ (Kubo et al. 2005; Mulkey et al. 2004), a property that is frequently used to differentiate contributions of Kir channels from other K+ channels, including background K+ channels (Mulkey et al. 2004). Only certain Kir channels are pH sensitive, e.g., Kir1.1 (pK = 6.7) (Xu et al. 2000b), Kir2.3 (pK = 6.8) (Putnam et al. 2004), homomeric Kir4.1 (pK = 6.1) (Pessia et al. 2001), and the heterodimeric Kir4.1–Kir5.1 channels (pK = 7.4) (Casamassima et al. 2003; Jiang et al. 2001; Xu et al. 2000b). Note that Kir5.1 subunits do not form functional homomeric channels, but preferentially heteromerize with Kir4.1 subunits in astrocytes (Casamassima et al. 2003; Pessia et al. 1996). Of all pH-sensitive Kir channels, only heteromeric Kir4.1–Kir5.1 channels are exquisitely sensitive to physiologically relevant pH changes. Further, in situ hybridization found that the RTN expresses high levels of Kir4.1 and Kir5.1 mRNA (Wu et al. 2004). Here, we show that RTN astrocytes respond to physiologically relevant pH changes. The pH-sensitive current expressed by these cells appears relatively voltage independent between −150 and −30 mV and was almost completely blocked by micromolar concentrations of Ba2+ and desipramine (a blocker of Kir4.1-containing channels; Su et al. 2007; Tang et al. 2009). This combination of features (i.e., inhibition by physiologically relevant changes in pH and micromolar concentrations of Ba2+ and desipramine) is most consistent with heteromeric Kir4.1–Kir5.1 channels. Therefore we consider heteromeric Kir4.1–Kir5.1 channels to be excellent candidate pH sensors of RTN astrocytes.
Our results also indicate that the NBC contributes to the pH-sensitive current in RTN astrocytes. It is well established in various types of astrocytes that activity of the NBC generates a hyperpolarizing outward current (i.e., net HCO3− influx) at membrane potentials depolarized to ENBC (Chesler 2003). In our experimental conditions we calculated the ENBC to be about −99 mV. As expected, at a holding potential of −80 mV in bicarbonate-buffered solution we observed a DIDS-sensitive outward current. This outward current will likely increase during depolarization and so potentially blunt H+-mediated inhibition of Kir4.1–Kir5.1 and the corresponding membrane potential. However, the NBC is also modulated by changes in extracellular pH; bath acidification can reverse Na+-HCO3− transport to produce an inward depolarizing current (i.e., net HCO3− efflux) (Chesler 2003; Deitmer 1991). In voltage-clamp (at a holding potential of −80 mV), we show that inward current produced by the NBC increased amplitude of the CO2/H+-sensitive current and caused a negative shift in its reversal potential. When activity of the NBC was blocked with DIDS, we observed a CO2/H+-sensitive current that was similar in amplitude to that of the pH-sensitive current in HCO3−-free HEPES solution and reversed near EK+. A contribution of the NBC to central chemoreception has yet to be established and considering that H+-induced depolarization and extracellular acidification have offsetting effects on directionality of Na+/HCO3− transport by the NBC, further experiments are required to define potential roles of this transporter in RTN chemoreception.
Possible role of astrocytes in central chemoreception
There are two mechanisms by which astrocytes are thought to influence chemoreception. First, a well-known function of astrocytes is to help regulate extracellular K+ by taking up extracellular K+ ions during increased neuronal activity (Olsen and Sontheimer 2008). This process effectively buffers changes in extracellular K+; however, it can also depolarize membrane potential and increase Na+/HCO3− cotransport into glia (Chesler 2003), which would enhance the CO2-induced fall in extracellular pH and potentiate activation of chemoreceptors. This possibility is supported by a series of in vivo experiments in which fluorocitrate microinjected into the RTN caused extracellular acidification followed by increased respiratory frequency and ventilatory sensitivity to CO2 (Erlichman and Leiter 2010; Holleran et al. 2001). Note that fluorocitrate is a toxin that inhibits metabolic activity; at low concentrations it has been shown to be selectively taken up by astrocytes but not neurons, where it initiates membrane depolarization (Erlichman and Leiter 2010), presumably by loss of cytoplasmic K+. However, it is also possible that the fluorocitrate-mediated effects described in these experiments resulted from the release of excitatory gliotransmitters acting on pH-sensitive neurons. Our data support this latter possibility. For example, we found that exposure to fluorocitrate activated pH-sensitive neurons under conditions in which the NBC is minimally active (i.e., in HEPES buffer). In addition, we were able to block effects of fluorocitrate with a P2-receptor antagonist. Therefore the most likely mechanism by which astrocytes contribute to chemoreception involves release of ATP and activation of pH-sensitive neurons. Our data also indicate that fluorocitrate caused an upward parallel shift in the pH sensitivity of RTN neurons, suggesting that fluorocitrate-mediated ATP release from RTN astrocytes contributes to chemoreception by increasing excitability of RTN neurons. These results are consistent with evidence in other brain regions that astrocytes serve as a source of excitatory purinergic drive able to rapidly modulate neural activity (Gordon et al. 2009). However, it should be noted that these experiments are limited by a dearth of pharmacological tools available to selectively manipulate astrocytes and the possibility that fluorocitrate may have nonspecific effects on other cell types, including neurons. Therefore additional experiments that use genetic approaches to selectively manipulate astrocytes are required to definitively establish a role of pH-sensitive astrocytes in chemoreception. It is important to note that our results are entirely consistent with a recent study that used the adenoviral system to target expression of ChR2 to RTN astrocytes (Gourine et al. 2010). This study showed that photostimulation of RTN astrocytes increased activity of RTN neurons and stimulated breathing by a purinergic-dependent mechanism. These results are similar to the observed effects of fluorocitrate, thus adding to our confidence that fluorocitrate effects are mediated by astrocytes.
Based on the results described earlier, it is reasonable to expect P2-receptor antagonists to decrease pH sensitivity of RTN neurons. However, previous experiments performed in HEPES-buffered media (Mulkey et al. 2004, 2006) indicate that PPADS had no effect on pH sensitivity of RTN neurons. A possible explanation for this apparent discrepancy is that CO2, not H+, is the effector responsible for hypercapnic-induced ATP release (Spyer 2009). Therefore in a parallel series of experiments, we tested the effects of two P2-receptor antagonists (PPADS and suramin) on CO2/H+ sensitivity of RTN neurons in bicarbonate-buffered media. Interestingly, we found that under these conditions both P2-receptor antagonists blunted neuronal CO2/H+ sensitivity. These results are consistent with the possibility that CO2 is the required stimulus for ATP release in the RTN.
In summary, we demonstrate that pH-sensitive RTN glia are protoplasmic astrocytes that sense H+ by inhibition of heteromeric Kir4.1–Kir5.1-like conductance and contribute to respiratory drive by increasing activity of pH-sensitive neurons by a purinergic-dependent mechanism. Our results also suggest that the NBC may contribute to the pH-sensitive current in RTN astrocytes. Thus we conclude that RTN astrocytes contribute to the mechanism of chemoreception and influence integrated output of the RTN.
GRANTS
This work was supported by the National Institutes of Health Grants HL-104101 to D. K. Mulkey and NS-049267 to A. Nishiyama and a University of Connecticut Research Foundation grant to D. K. Mulkey.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
Supplementary Material
Footnotes
The online version of this article contains supplemental data.
REFERENCES
- Abbott et al., 2009. Abbott SB, Stornetta RL, Fortuna MG, DePuy SD, West GH, Harris TE, Guyenet PG. Photostimulation of retrotrapezoid nucleus phox2b-expressing neurons in vivo produces long-lasting activation of breathing in rats. J Neurosci 29: 5806–5819, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayliss et al., 2001. Bayliss DA, Talley EM, Sirois JE, Lei Q. TASK-1 is a highly modulated pH-sensitive “leak” K(+) channel expressed in brainstem respiratory neurons. Respir Physiol 129: 159–174, 2001 [DOI] [PubMed] [Google Scholar]
- Cahoy et al., 2008. Cahoy JD, Emery B, Kaushal A, Foo LC, Zamanian JL, Christopherson KS, Xing Y, Lubischer JL, Krieg PA, Krupenko SA, Thompson WJ, Barres BA. A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. J Neurosci 28: 264–278, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casamassima et al., 2003. Casamassima M, D'Adamo MC, Pessia M, Tucker SJ. Identification of a heteromeric interaction that influences the rectification, gating, and pH sensitivity of Kir4.1/Kir5.1 potassium channels. J Biol Chem 278: 43533–43540, 2003 [DOI] [PubMed] [Google Scholar]
- Chesler, 2003. Chesler M. Regulation and modulation of pH in the brain. Physiol Rev 83: 1183–1221, 2003 [DOI] [PubMed] [Google Scholar]
- Deitmer, 1991. Deitmer JW. Electrogenic sodium-dependent bicarbonate secretion by glial cells of the leech central nervous system. J Gen Physiol 98: 637–655, 1991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erlichman and Leiter, 2010. Erlichman JS, Leiter JC. Glia modulation of extracellular milieu as a factor in central CO2 chemoreception and respiratory control. J Appl Physiol 108: 1803–1811, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erlichman et al., 1998. Erlichman JS, Li A, Nattie EE. Ventilatory effects of glial dysfunction in a rat brain stem chemoreceptor region. J Appl Physiol 85: 1599–1604, 1998 [DOI] [PubMed] [Google Scholar]
- Fellin et al., 2006. Fellin T, Pascual O, Haydon PG. Astrocytes coordinate synaptic networks: balanced excitation and inhibition. Physiology 21: 208–215, 2006 [DOI] [PubMed] [Google Scholar]
- Filosa and Putnam, 2003. Filosa JA, Putnam RW. Multiple targets of chemosensitive signaling in locus coeruleus neurons: role of K+ and Ca2+ channels. Am J Physiol Cell Physiol 284: C145–C155, 2003 [DOI] [PubMed] [Google Scholar]
- Fukuda et al., 1978. Fukuda Y, Honda Y, Schlafke ME, Loeschcke HH. Effect of H+ on the membrane potential of silent cells in the ventral and dorsal surface layers of the rat medulla in vitro. Pflügers Arch 376: 229–235, 1978 [DOI] [PubMed] [Google Scholar]
- Goldstein et al., 2005. Goldstein SAN, Bayliss DA, Kim D, Lesage F, Plant LD, Rajan S. International Union of Pharmacology. LV. Nomenclature and molecular relationships of two-P potassium channels. Pharmacol Rev 57: 527–540, 2005 [DOI] [PubMed] [Google Scholar]
- Gordon et al., 2009. Gordon GR, Iremonger KJ, Kantevari S, Ellis-Davies GC, MacVicar BA, Bains JS. Astrocyte-mediated distributed plasticity at hypothalamic glutamate synapses. Neuron 64: 391–403, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gourine et al., 2010. Gourine AV, Kasymov V, Marina N, Tang F, Figueiredo MF, Lane S, Teschemacher AG, Spyer MK, Deisseroth K, Kasparov S. Astrocytes control breathing through pH-dependent release of ATP. Science 329: 571–575, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gourine et al., 2005. Gourine AV, Llaudet E, Dale N, Spyer KM. ATP is a mediator of chemosensory transduction in the central nervous system. Nature 436: 108–111, 2005 [DOI] [PubMed] [Google Scholar]
- Grass et al., 2004. Grass D, Pawlowski PG, Hirrlinger J, Papadopoulos N, Richter DW, Kirchhoff F, Hulsmann S. Diversity of functional astroglial properties in the respiratory network. J Neurosci 24: 1358–1365, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross et al., 2001. Gross E, Abuladze N, Pushkin A, Kurtz I, Cotton CU. The stoichiometry of the electrogenic sodium bicarbonate cotransporter pNBC1 in mouse pancreatic duct cells is 2 HCO3−:1 Na(+). J Physiol 531: 375–382, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hibino et al., 2004. Hibino H, Fujita A, Iwai K, Yamada M, Kurachi Y. Differential assembly of inwardly rectifying K+ channel subunits, Kir4.1 and Kir5.1, in brain astrocytes. J Biol Chem 279: 44065–44073, 2004 [DOI] [PubMed] [Google Scholar]
- Holleran et al., 2001. Holleran J, Babbie M, Erlichman JS. Ventilatory effects of impaired glial function in a brain stem chemoreceptor region in the conscious rat. J Appl Physiol 90: 1539–1547, 2001 [DOI] [PubMed] [Google Scholar]
- Jiang et al., 2001. Jiang C, Xu H, Cui N, Wu J. An alternative approach to the identification of respiratory central chemoreceptors in the brainstem. Respir Physiol 129: 141–157, 2001 [DOI] [PubMed] [Google Scholar]
- Kubo et al., 2005. Kubo Y, Adelman JP, Clapham DE, Jan LY, Karschin A, Kurachi Y, Lazdunski M, Nichols CG, Seino S, Vandenberg CA. International Union of Pharmacology. LIV. Nomenclature and molecular relationship of inwardly rectifying potassium channels. Pharmacol Rev 57: 509–526, 2005 [DOI] [PubMed] [Google Scholar]
- Matthias et al., 2003. Matthias K, Kirchhoff F, Seifert G, Huttmann K, Matyash M, Kettenmann H, Steinhauser C. Segregated expression of AMPA-type glutamate receptors and glutamate transporters defines distinct astrocyte populations in the mouse hippocampus. J Neurosci 23: 1750–1758, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millhorn et al., 1984. Millhorn DE, Eldridge FL, Kiley JP. Oscillations of medullary extracellular fluid pH caused by breathing. Respir Physiol 55: 193–203, 1984 [DOI] [PubMed] [Google Scholar]
- Mulkey et al., 2006. Mulkey DK, Mistry AM, Guyenet PG, Bayliss DA. Purinergic P2 receptors modulate excitability but do not mediate pH sensitivity of RTN respiratory chemoreceptors. J Neurosci 26: 7230–7233, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulkey et al., 2004. Mulkey DK, Stornetta RL, Weston MC, Simmons JR, Parker A, Bayliss DA, Guyenet PG. Respiratory control by ventral surface chemoreceptor neurons in rats. Nat Neurosci 7: 1360–1369, 2004 [DOI] [PubMed] [Google Scholar]
- Mulkey et al., 2007. Mulkey DK, Talley EM, Stornetta RL, Siegel AR, West GH, Chen X, Sen N, Mistry AM, Guyenet PG, Bayliss DA. TASK channels determine pH sensitivity in select respiratory neurons but do not contribute to central respiratory chemosensitivity. J Neurosci 27: 14049–14058, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munsch and Deitmer, 1994. Munsch T, Deitmer JW. Sodium-bicarbonate cotransport current in identified leech glial cells. J Physiol 474: 43–53, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman, 1991. Newman EA. Sodium-bicarbonate cotransport in retinal Muller (glial) cells of the salamander. J Neurosci 11: 3972–3983, 1991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen and Sontheimer, 2008. Olsen ML, Sontheimer H. Functional implications for Kir4.1 channels in glial biology: from K+ buffering to cell differentiation. J Neurochem 107: 589–601, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel and Honore, 2001. Patel AJ, Honore E. Properties and modulation of mammalian 2P domain K+ channels. Trends Neurosci 24: 339–346, 2001 [DOI] [PubMed] [Google Scholar]
- Pessia et al., 2001. Pessia M, Imbrici P, D'Adamo MC, Salvatore L, Tucker SJ. Differential pH sensitivity of Kir4.1 and Kir4.2 potassium channels and their modulation by heteropolymerisation with Kir5.1. J Physiol 532: 359–367, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pessia et al., 1996. Pessia M, Tucker SJ, Lee K, Bond CT, Adelman JP. Subunit positional effects revealed by novel heteromeric inwardly rectifying K+ channels. EMBO J 15: 2980–2987, 1996 [PMC free article] [PubMed] [Google Scholar]
- Putnam et al., 2004. Putnam RW, Filosa JA, Ritucci NA. Cellular mechanisms involved in CO2 and acid signaling in chemosensitive neurons. Am J Physiol Cell Physiol 287: C1493–C1526, 2004 [DOI] [PubMed] [Google Scholar]
- Ritucci et al., 2005. Ritucci NA, Erlichman JS, Leiter JC, Putnam RW. Response of membrane potential and intracellular pH to hypercapnia in neurons and astrocytes from rat retrotrapezoid nucleus. Am J Physiol Regul Integr Comp Physiol 289: R851–R861, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz et al., 2003. Schultz JH, Czachurski J, Volk T, Ehmke H, Seller H. Central sympathetic chemosensitivity and Kir1 potassium channels in the cat. Brain Res 963: 113–120, 2003 [DOI] [PubMed] [Google Scholar]
- Spyer, 2009. Spyer KM. To breathe or not to breathe? That is the question. Exp Physiol 94: 1–10, 2009 [DOI] [PubMed] [Google Scholar]
- Su et al., 2007. Su S, Ohno Y, Lossin C, Hibino H, Inanobe A, Kurachi Y. Inhibition of astroglial inwardly rectifying Kir4.1 channels by a tricyclic antidepressant, nortriptyline. J Pharmacol Exp Ther 320: 573–580, 2007 [DOI] [PubMed] [Google Scholar]
- Tang et al., 2009. Tang X, Taniguchi K, Kofuji P. Heterogeneity of Kir4.1 channel expression in glia revealed by mouse transgenesis. Glia 57: 1706–1715, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wellner-Kienitz et al., 1998. Wellner-Kienitz MC, Shams H, Scheid P. Contribution of Ca2+-activated K+ channels to central chemosensitivity in cultivated neurons of fetal rat medulla. J Neurophysiol 79: 2885–2894, 1998 [DOI] [PubMed] [Google Scholar]
- Wu et al., 2004. Wu J, Xu H, Shen W, Jiang C. Expression and coexpression of CO2-sensitive Kir channels in brainstem neurons of rats. J Membr Biol 197: 179–191, 2004 [DOI] [PubMed] [Google Scholar]
- Xu et al., 2000. Xu H, Cui N, Yang Z, Qu Z, Jiang C. Modulation of Kir4.1 and Kir5.1 by hypercapnia and intracellular acidosis. J Physiol 524: 725–735, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu et al., 2000. Xu H, Yang Z, Cui N, Giwa LR, Abdulkadir L, Patel M, Sharma P, Shan G, Shen W, Jiang C. Molecular determinants for the distinct pH sensitivity of Kir1.1 and Kir4.1 channels. Am J Physiol Cell Physiol 279: C1464–C1471, 2000 [DOI] [PubMed] [Google Scholar]
- Zhou et al., 2006. Zhou M, Schools GP, Kimelberg HK. Development of GLAST(+) astrocytes and NG2(+) glia in rat hippocampus CA1: mature astrocytes are electrophysiologically passive. J Neurophysiol 95: 134–143, 2006 [DOI] [PubMed] [Google Scholar]
- Zhou et al., 2009. Zhou M, Xu G, Xie M, Zhang X, Schools GP, Ma L, Kimelberg HK, Chen H. TWIK-1 and TREK-1 are potassium channels contributing significantly to astrocyte passive conductance in rat hippocampal slices. J Neurosci 29: 8551–8564, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu et al., 2000. Zhu G, Liu C, Qu Z, Chanchevalap S, Xu H, Jiang C. CO2 inhibits specific inward rectifier K(+) channels by decreases in intra- and extracellular pH. J Cell Physiol 183: 53–64, 2000 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.