Abstract
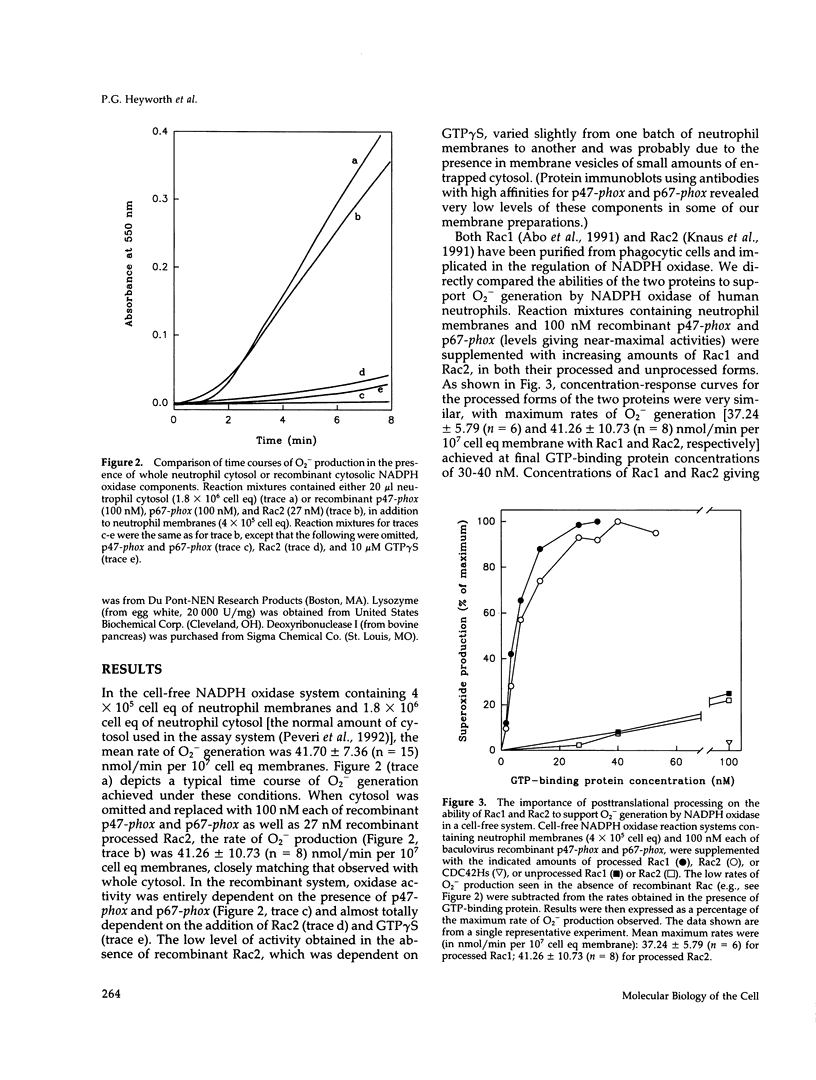
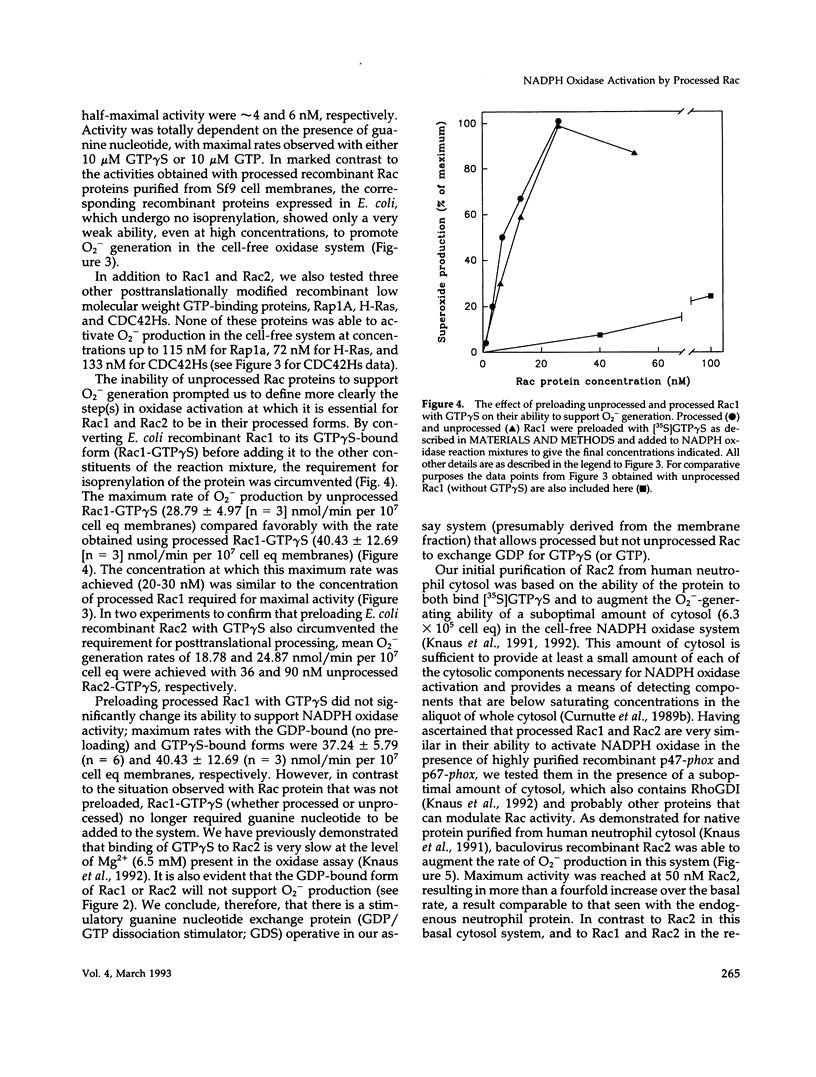
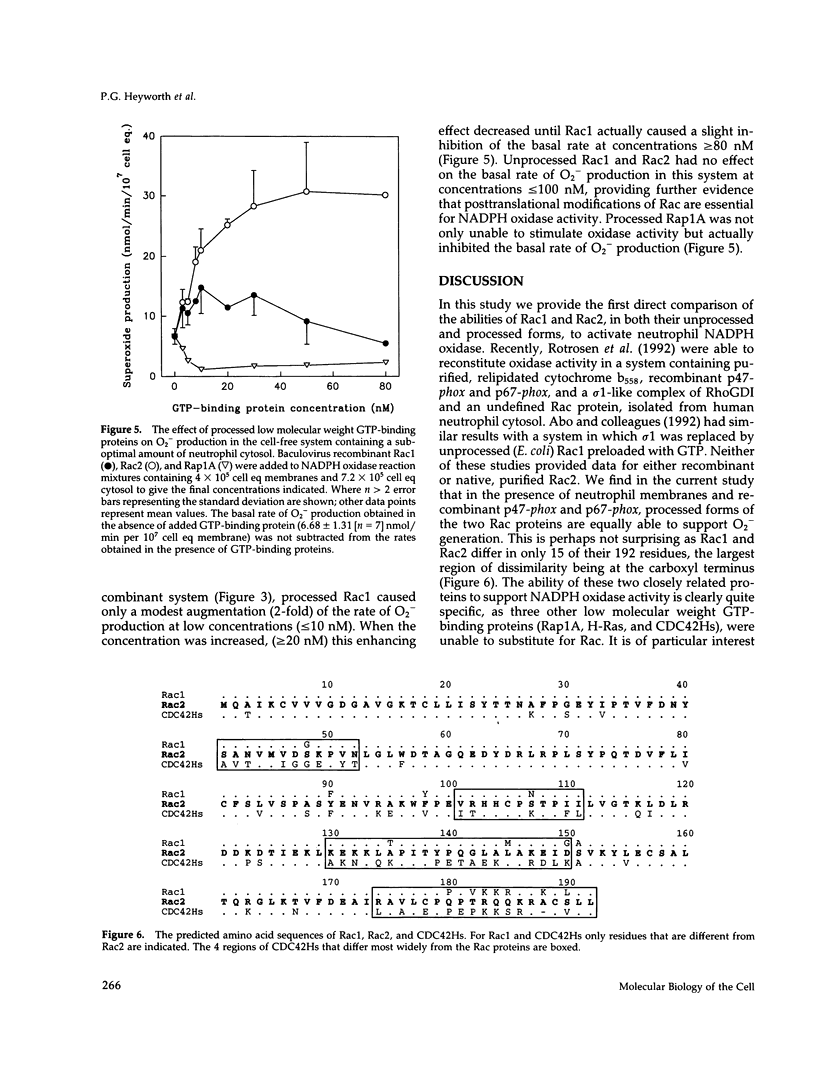
Rac1 and Rac2 are closely related, low molecular weight GTP-binding proteins that have both been implicated in regulation of phagocyte NADPH oxidase. This enzyme system is composed of multiple membrane-bound and cytosolic subunits and when activated catalyzes the one-electron reduction of oxygen to superoxide. Superoxide and its highly reactive derivatives are essential for killing microorganisms. Rac proteins undergo posttranslational processing, primarily the addition of an isoprenyl group to a carboxyl-terminal cysteine residue. We directly compared recombinant Rac1 and Rac2 in a human neutrophil cell-free NADPH oxidase system in which cytosol was replaced by purified recombinant cytosolic components (p47-phox and p67-phox). Processed Rac1 and Rac2 were both highly active in this system and supported comparable rates of superoxide production. Under different cell-free conditions, however, in which suboptimal amounts of cytosol were present in the assay mixture, processed Rac2 worked much better than Rac1 at all but the lowest concentrations. This suggests that a factor in the cytosol may suppress the activity of Rac1 but not of Rac2. Unprocessed Rac proteins were only weakly able to support superoxide generation in either system, but preloading of Rac1 or Rac2 with guanosine 5'-O-(3-thio-triphosphate) (GTP gamma S) restored activity. These results indicate that processing is required for nucleotide exchange but not for interaction with oxidase components.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abo A., Boyhan A., West I., Thrasher A. J., Segal A. W. Reconstitution of neutrophil NADPH oxidase activity in the cell-free system by four components: p67-phox, p47-phox, p21rac1, and cytochrome b-245. J Biol Chem. 1992 Aug 25;267(24):16767–16770. [PubMed] [Google Scholar]
- Abo A., Pick E., Hall A., Totty N., Teahan C. G., Segal A. W. Activation of the NADPH oxidase involves the small GTP-binding protein p21rac1. Nature. 1991 Oct 17;353(6345):668–670. doi: 10.1038/353668a0. [DOI] [PubMed] [Google Scholar]
- Ando S., Kaibuchi K., Sasaki T., Hiraoka K., Nishiyama T., Mizuno T., Asada M., Nunoi H., Matsuda I., Matsuura Y. Post-translational processing of rac p21s is important both for their interaction with the GDP/GTP exchange proteins and for their activation of NADPH oxidase. J Biol Chem. 1992 Dec 25;267(36):25709–25713. [PubMed] [Google Scholar]
- Babior B. M., Kuver R., Curnutte J. T. Kinetics of activation of the respiratory burst oxidase in a fully soluble system from human neutrophils. J Biol Chem. 1988 Feb 5;263(4):1713–1718. [PubMed] [Google Scholar]
- Badwey J. A., Curnutte J. T., Berde C. B., Karnovsky M. L. Cytochalasin E diminishes the lag phase in the release of superoxide by human neutrophils. Biochem Biophys Res Commun. 1982 May 14;106(1):170–174. doi: 10.1016/0006-291x(82)92073-3. [DOI] [PubMed] [Google Scholar]
- Bokoch G. M., Prossnitz V. Isoprenoid metabolism is required for stimulation of the respiratory burst oxidase of HL-60 cells. J Clin Invest. 1992 Feb;89(2):402–408. doi: 10.1172/JCI115599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bokoch G. M., Quilliam L. A., Bohl B. P., Jesaitis A. J., Quinn M. T. Inhibition of Rap1A binding to cytochrome b558 of NADPH oxidase by phosphorylation of Rap1A. Science. 1991 Dec 20;254(5039):1794–1796. doi: 10.1126/science.1763330. [DOI] [PubMed] [Google Scholar]
- Bordier C. Phase separation of integral membrane proteins in Triton X-114 solution. J Biol Chem. 1981 Feb 25;256(4):1604–1607. [PubMed] [Google Scholar]
- Borregaard N., Heiple J. M., Simons E. R., Clark R. A. Subcellular localization of the b-cytochrome component of the human neutrophil microbicidal oxidase: translocation during activation. J Cell Biol. 1983 Jul;97(1):52–61. doi: 10.1083/jcb.97.1.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bromberg Y., Pick E. Unsaturated fatty acids stimulate NADPH-dependent superoxide production by cell-free system derived from macrophages. Cell Immunol. 1984 Oct 1;88(1):213–221. doi: 10.1016/0008-8749(84)90066-2. [DOI] [PubMed] [Google Scholar]
- Chavrier P., Gorvel J. P., Stelzer E., Simons K., Gruenberg J., Zerial M. Hypervariable C-terminal domain of rab proteins acts as a targeting signal. Nature. 1991 Oct 24;353(6346):769–772. doi: 10.1038/353769a0. [DOI] [PubMed] [Google Scholar]
- Chuang T. H., Xu X., Knaus U. G., Hart M. J., Bokoch G. M. GDP dissociation inhibitor prevents intrinsic and GTPase activating protein-stimulated GTP hydrolysis by the Rac GTP-binding protein. J Biol Chem. 1993 Jan 15;268(2):775–778. [PubMed] [Google Scholar]
- Clark R. A., Volpp B. D., Leidal K. G., Nauseef W. M. Two cytosolic components of the human neutrophil respiratory burst oxidase translocate to the plasma membrane during cell activation. J Clin Invest. 1990 Mar;85(3):714–721. doi: 10.1172/JCI114496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curnutte J. T. Activation of human neutrophil nicotinamide adenine dinucleotide phosphate, reduced (triphosphopyridine nucleotide, reduced) oxidase by arachidonic acid in a cell-free system. J Clin Invest. 1985 May;75(5):1740–1743. doi: 10.1172/JCI111885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curnutte J. T., Kuver R., Babior B. M. Activation of the respiratory burst oxidase in a fully soluble system from human neutrophils. J Biol Chem. 1987 May 15;262(14):6450–6452. [PubMed] [Google Scholar]
- Curnutte J. T., Scott P. J., Babior B. M. Functional defect in neutrophil cytosols from two patients with autosomal recessive cytochrome-positive chronic granulomatous disease. J Clin Invest. 1989 Apr;83(4):1236–1240. doi: 10.1172/JCI114006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curnutte J. T., Scott P. J., Mayo L. A. Cytosolic components of the respiratory burst oxidase: resolution of four components, two of which are missing in complementing types of chronic granulomatous disease. Proc Natl Acad Sci U S A. 1989 Feb;86(3):825–829. doi: 10.1073/pnas.86.3.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Didsbury J. R., Uhing R. J., Snyderman R. Isoprenylation of the low molecular mass GTP-binding proteins rac 1 and rac 2: possible role in membrane localization. Biochem Biophys Res Commun. 1990 Sep 14;171(2):804–812. doi: 10.1016/0006-291x(90)91217-g. [DOI] [PubMed] [Google Scholar]
- Didsbury J., Weber R. F., Bokoch G. M., Evans T., Snyderman R. rac, a novel ras-related family of proteins that are botulinum toxin substrates. J Biol Chem. 1989 Oct 5;264(28):16378–16382. [PubMed] [Google Scholar]
- Dorseuil O., Vazquez A., Lang P., Bertoglio J., Gacon G., Leca G. Inhibition of superoxide production in B lymphocytes by rac antisense oligonucleotides. J Biol Chem. 1992 Oct 15;267(29):20540–20542. [PubMed] [Google Scholar]
- Gabig T. G., English D., Akard L. P., Schell M. J. Regulation of neutrophil NADPH oxidase activation in a cell-free system by guanine nucleotides and fluoride. Evidence for participation of a pertussis and cholera toxin-insensitive G protein. J Biol Chem. 1987 Feb 5;262(4):1685–1690. [PubMed] [Google Scholar]
- Grussenmeyer T., Scheidtmann K. H., Hutchinson M. A., Eckhart W., Walter G. Complexes of polyoma virus medium T antigen and cellular proteins. Proc Natl Acad Sci U S A. 1985 Dec;82(23):7952–7954. doi: 10.1073/pnas.82.23.7952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hart M. J., Shinjo K., Hall A., Evans T., Cerione R. A. Identification of the human platelet GTPase activating protein for the CDC42Hs protein. J Biol Chem. 1991 Nov 5;266(31):20840–20848. [PubMed] [Google Scholar]
- Heyworth P. G., Curnutte J. T., Nauseef W. M., Volpp B. D., Pearson D. W., Rosen H., Clark R. A. Neutrophil nicotinamide adenine dinucleotide phosphate oxidase assembly. Translocation of p47-phox and p67-phox requires interaction between p47-phox and cytochrome b558. J Clin Invest. 1991 Jan;87(1):352–356. doi: 10.1172/JCI114993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heyworth P. G., Shrimpton C. F., Segal A. W. Localization of the 47 kDa phosphoprotein involved in the respiratory-burst NADPH oxidase of phagocytic cells. Biochem J. 1989 May 15;260(1):243–248. doi: 10.1042/bj2600243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiroyoshi M., Kaibuchi K., Kawamura S., Hata Y., Takai Y. Role of the C-terminal region of smg p21, a ras p21-like small GTP-binding protein, in membrane and smg p21 GDP/GTP exchange protein interactions. J Biol Chem. 1991 Feb 15;266(5):2962–2969. [PubMed] [Google Scholar]
- Itoh H., Gilman A. G. Expression and analysis of Gs alpha mutants with decreased ability to activate adenylylcyclase. J Biol Chem. 1991 Aug 25;266(24):16226–16231. [PubMed] [Google Scholar]
- Kinsella B. T., Erdman R. A., Maltese W. A. Carboxyl-terminal isoprenylation of ras-related GTP-binding proteins encoded by rac1, rac2, and ralA. J Biol Chem. 1991 May 25;266(15):9786–9794. [PubMed] [Google Scholar]
- Knaus U. G., Heyworth P. G., Evans T., Curnutte J. T., Bokoch G. M. Regulation of phagocyte oxygen radical production by the GTP-binding protein Rac 2. Science. 1991 Dec 6;254(5037):1512–1515. doi: 10.1126/science.1660188. [DOI] [PubMed] [Google Scholar]
- Knaus U. G., Heyworth P. G., Kinsella B. T., Curnutte J. T., Bokoch G. M. Purification and characterization of Rac 2. A cytosolic GTP-binding protein that regulates human neutrophil NADPH oxidase. J Biol Chem. 1992 Nov 25;267(33):23575–23582. [PubMed] [Google Scholar]
- Marshall M. S., Davis L. J., Keys R. D., Mosser S. D., Hill W. S., Scolnick E. M., Gibbs J. B. Identification of amino acid residues required for Ras p21 target activation. Mol Cell Biol. 1991 Aug;11(8):3997–4004. doi: 10.1128/mcb.11.8.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McPhail L. C., Shirley P. S., Clayton C. C., Snyderman R. Activation of the respiratory burst enzyme from human neutrophils in a cell-free system. Evidence for a soluble cofactor. J Clin Invest. 1985 May;75(5):1735–1739. doi: 10.1172/JCI111884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizuno T., Kaibuchi K., Ando S., Musha T., Hiraoka K., Takaishi K., Asada M., Nunoi H., Matsuda I., Takai Y. Regulation of the superoxide-generating NADPH oxidase by a small GTP-binding protein and its stimulatory and inhibitory GDP/GTP exchange proteins. J Biol Chem. 1992 May 25;267(15):10215–10218. [PubMed] [Google Scholar]
- Morel F., Doussiere J., Vignais P. V. The superoxide-generating oxidase of phagocytic cells. Physiological, molecular and pathological aspects. Eur J Biochem. 1991 Nov 1;201(3):523–546. doi: 10.1111/j.1432-1033.1991.tb16312.x. [DOI] [PubMed] [Google Scholar]
- Nauseef W. M., Volpp B. D., McCormick S., Leidal K. G., Clark R. A. Assembly of the neutrophil respiratory burst oxidase. Protein kinase C promotes cytoskeletal and membrane association of cytosolic oxidase components. J Biol Chem. 1991 Mar 25;266(9):5911–5917. [PubMed] [Google Scholar]
- Nunoi H., Rotrosen D., Gallin J. I., Malech H. L. Two forms of autosomal chronic granulomatous disease lack distinct neutrophil cytosol factors. Science. 1988 Dec 2;242(4883):1298–1301. doi: 10.1126/science.2848319. [DOI] [PubMed] [Google Scholar]
- Nur-E-Kamal M. S., Sizeland A., D'Abaco G., Maruta H. Asparagine 26, glutamic acid 31, valine 45, and tyrosine 64 of Ras proteins are required for their oncogenicity. J Biol Chem. 1992 Jan 25;267(3):1415–1418. [PubMed] [Google Scholar]
- Parkos C. A., Allen R. A., Cochrane C. G., Jesaitis A. J. Purified cytochrome b from human granulocyte plasma membrane is comprised of two polypeptides with relative molecular weights of 91,000 and 22,000. J Clin Invest. 1987 Sep;80(3):732–742. doi: 10.1172/JCI113128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peveri P., Heyworth P. G., Curnutte J. T. Absolute requirement for GTP in activation of human neutrophil NADPH oxidase in a cell-free system: role of ATP in regenerating GTP. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2494–2498. doi: 10.1073/pnas.89.6.2494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quilliam L. A., Der C. J., Clark R., O'Rourke E. C., Zhang K., McCormick F., Bokoch G. M. Biochemical characterization of baculovirus-expressed rap1A/Krev-1 and its regulation by GTPase-activating proteins. Mol Cell Biol. 1990 Jun;10(6):2901–2908. doi: 10.1128/mcb.10.6.2901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quilliam L. A., Mueller H., Bohl B. P., Prossnitz V., Sklar L. A., Der C. J., Bokoch G. M. Rap1A is a substrate for cyclic AMP-dependent protein kinase in human neutrophils. J Immunol. 1991 Sep 1;147(5):1628–1635. [PubMed] [Google Scholar]
- Quinn M. T., Mullen M. L., Jesaitis A. J., Linner J. G. Subcellular distribution of the Rap1A protein in human neutrophils: colocalization and cotranslocation with cytochrome b559. Blood. 1992 Mar 15;79(6):1563–1573. [PubMed] [Google Scholar]
- Quinn M. T., Parkos C. A., Jesaitis A. J. The lateral organization of components of the membrane skeleton and superoxide generation in the plasma membrane of stimulated human neutrophils. Biochim Biophys Acta. 1989 Dec 11;987(1):83–94. doi: 10.1016/0005-2736(89)90458-6. [DOI] [PubMed] [Google Scholar]
- Quinn M. T., Parkos C. A., Walker L., Orkin S. H., Dinauer M. C., Jesaitis A. J. Association of a Ras-related protein with cytochrome b of human neutrophils. Nature. 1989 Nov 9;342(6246):198–200. doi: 10.1038/342198a0. [DOI] [PubMed] [Google Scholar]
- Reibel L., Dorseuil O., Stancou R., Bertoglio J., Gacon G. A hemopoietic specific gene encoding a small GTP binding protein is overexpressed during T cell activation. Biochem Biophys Res Commun. 1991 Mar 15;175(2):451–458. doi: 10.1016/0006-291x(91)91585-z. [DOI] [PubMed] [Google Scholar]
- Ridley A. J., Hall A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell. 1992 Aug 7;70(3):389–399. doi: 10.1016/0092-8674(92)90163-7. [DOI] [PubMed] [Google Scholar]
- Ridley A. J., Paterson H. F., Johnston C. L., Diekmann D., Hall A. The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell. 1992 Aug 7;70(3):401–410. doi: 10.1016/0092-8674(92)90164-8. [DOI] [PubMed] [Google Scholar]
- Rotrosen D., Yeung C. L., Leto T. L., Malech H. L., Kwong C. H. Cytochrome b558: the flavin-binding component of the phagocyte NADPH oxidase. Science. 1992 Jun 5;256(5062):1459–1462. doi: 10.1126/science.1318579. [DOI] [PubMed] [Google Scholar]
- Schaber M. D., Garsky V. M., Boylan D., Hill W. S., Scolnick E. M., Marshall M. S., Sigal I. S., Gibbs J. B. Ras interaction with the GTPase-activating protein (GAP). Proteins. 1989;6(3):306–315. doi: 10.1002/prot.340060313. [DOI] [PubMed] [Google Scholar]
- Segal A. W. Absence of both cytochrome b-245 subunits from neutrophils in X-linked chronic granulomatous disease. Nature. 1987 Mar 5;326(6108):88–91. doi: 10.1038/326088a0. [DOI] [PubMed] [Google Scholar]
- Segal A. W., West I., Wientjes F., Nugent J. H., Chavan A. J., Haley B., Garcia R. C., Rosen H., Scrace G. Cytochrome b-245 is a flavocytochrome containing FAD and the NADPH-binding site of the microbicidal oxidase of phagocytes. Biochem J. 1992 Jun 15;284(Pt 3):781–788. doi: 10.1042/bj2840781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seifert R., Rosenthal W., Schultz G. Guanine nucleotides stimulate NADPH oxidase in membranes of human neutrophils. FEBS Lett. 1986 Sep 1;205(1):161–165. doi: 10.1016/0014-5793(86)80886-9. [DOI] [PubMed] [Google Scholar]
- Uhlinger D. J., Burnham D. N., Lambeth J. D. Nucleoside triphosphate requirements for superoxide generation and phosphorylation in a cell-free system from human neutrophils. Sodium dodecyl sulfate and diacylglycerol activate independently of protein kinase C. J Biol Chem. 1991 Nov 5;266(31):20990–20997. [PubMed] [Google Scholar]
- Uhlinger D. J., Inge K. L., Kreck M. L., Tyagi S. R., Neckelmann N., Lambeth J. D. Reconstitution and characterization of the human neutrophil respiratory burst oxidase using recombinant p47-phox, p67-phox and plasma membrane. Biochem Biophys Res Commun. 1992 Jul 15;186(1):509–516. doi: 10.1016/s0006-291x(05)80837-x. [DOI] [PubMed] [Google Scholar]
- Volpp B. D., Nauseef W. M., Clark R. A. Two cytosolic neutrophil oxidase components absent in autosomal chronic granulomatous disease. Science. 1988 Dec 2;242(4883):1295–1297. doi: 10.1126/science.2848318. [DOI] [PubMed] [Google Scholar]
- Willumsen B. M., Vass W. C., Velu T. J., Papageorge A. G., Schiller J. T., Lowy D. R. The bovine papillomavirus E5 oncogene can cooperate with ras: identification of p21 amino acids critical for transformation by c-rasH but not v-rasH. Mol Cell Biol. 1991 Dec;11(12):6026–6033. doi: 10.1128/mcb.11.12.6026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodman R. C., Ruedi J. M., Jesaitis A. J., Okamura N., Quinn M. T., Smith R. M., Curnutte J. T., Babior B. M. Respiratory burst oxidase and three of four oxidase-related polypeptides are associated with the cytoskeleton of human neutrophils. J Clin Invest. 1991 Apr;87(4):1345–1351. doi: 10.1172/JCI115138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang K., Noda M., Vass W. C., Papageorge A. G., Lowy D. R. Identification of small clusters of divergent amino acids that mediate the opposing effects of ras and Krev-1. Science. 1990 Jul 13;249(4965):162–165. doi: 10.1126/science.2115210. [DOI] [PubMed] [Google Scholar]



