Abstract
Studies on rats have shown that lactic acid can improve excitability and function of depolarized muscles. The effect has been related to the ensuing reduction in intracellular pH causing inhibition of muscle fibre Cl− channels. However, since several carboxylic acids with structural similarities to lactate can inhibit muscle Cl− channels it is possible that lactate per se can increase muscle excitability by exerting a direct effect on these channels. We therefore examined the effects of lactate on the function of intact muscles and skinned fibres together with effects on pH and Cl− conductance (Gcl). In muscles where extracellular compound action potentials (M-waves) and tetanic force response to excitation were reduced by (mean ±s.e.m.) 82 ± 4% and 83 ± 2%, respectively, by depolarization with 11 mm extracellular K+, both M-waves and force exhibited an up to 4-fold increase when 20 mm lactate was added. This effect was present already at 5 mm and saturated at 15 mm lactate, and was associated with a 31% reduction in GCl. The effects of lactate were completely blocked by Cl− channel inhibition or use of Cl−-free solutions. Finally, both experiments where effects of lactate on intracellular pH in intact muscles were mimicked by increased CO2 tension and experiments with skinned fibres showed that the effects of lactate could not be related to reduced intracellular pH. It is concluded that addition of lactate can inhibit ClC-1 Cl− channels and increase the excitability and contractile function of depolarized rat muscles via mechanisms not related to a reduction in intracellular pH.
Introduction
Intense contractile activity leads to the accumulation of both lactate and H+ in the intra- and extracellular compartments of the active muscles. In addition, it is well established that muscle activity is associated with loss of K+ from the working muscle fibres causing extracellular K+ ([K+]o) to become markedly elevated. All three changes have been considered as important causative factors in muscle fatigue (Fitts, 1994; Sejersted & Sjøgaard, 2000; Clausen, 2003) although later studies have argued that the role of lactic acid is related to the concomitant acidification rather than to effects of the lactate per se (Fitts, 1994).
However, the experimental observations that suggest a depressing effect of acidification on muscle function have mostly been performed at temperatures below the physiologically relevant range for mammals. More recently it was found that the detrimental effects of lowering pH on muscle force were reduced considerably if the experimental temperature was increased to 25–37°C (Pate et al. 1995; Westerblad et al. 1997). Moreover, we have shown that when muscles are depolarized by exposure to elevated extracellular K+, thus mimicking the scenario of active muscle more closely, the addition of lactic acid significantly improves muscle excitability leading to increased contractile function (Nielsen et al. 2001). Based on these findings it has been argued that in mammals the detrimental effect of lactic acid on force production during intense exercise could be outweighed by protective effects on the excitability of the muscles (Lamb & Stephenson, 2006).
Since experiments on isolated muscles have shown that the improvement of excitability after addition of lactic acid to K+-depressed muscle could be mimicked by muscle acidification, it was concluded that the effect of lactic acid was related to the ensuing reduction in intracellular pH (pHi) (Nielsen et al. 2001). A similar role for an intracellular acidification was implicated by Kristensen et al. (2005) as an explanation for their finding that addition of lactate without external acidification could also recover contractile function in muscle exposed to elevated [K+]o. The mechanism for the effect of reduced pHi on excitability was demonstrated in other studies to be an inhibition of the abundant Cl− channels (ClC-1) in the muscle fibre membrane, which, by leading to a reduction in Cl− conductance (GCl), lowers the minimal Na+ current necessary to initiate an action potential (Pedersen et al. 2004, 2005). In depolarized muscle, where excitability is reduced due to inactivation of Na+ channels, reduced GCl can significantly recover muscle function (Pedersen et al. 2005).
Two observations suggest that the effect of lactate on muscle GCl and excitability cannot be simply explained by the lactate-dependent reduction in pHi. First, it has been demonstrated that the intracellular acidification is rather limited when muscles are exposed to lactate at normal pH (Wetzel et al. 2001). Second, it has been shown that several small carboxylates that are structurally similar to lactate (2-hydroxy-propionate), like propionate, can significantly reduce muscle GCl via direct interference with the ClC-1 channels (Rychkov et al. 2001). Together these observations indicate that lactate per se may improve muscle excitability by reducing GCl via pathways independent of changes in pHi. To test this hypothesis we examined in this project the effect of lactate on pHi and GCl, in intact and skinned muscle fibres, together with the effect of lactate on contractile function when the muscles were depolarized. The results show that addition of lactate can inhibit ClC-1 Cl− channels and markedly increase the excitability and the contractile function of depolarized rat muscles via a mechanism that is independent of pHi.
Methods
Animal care and whole muscle preparation
Experiments with intact muscles were performed using soleus or extensor digitorum longus (EDL) muscles from Wistar rats. For measurements of tetanic force production and compound action potentials (M-waves), muscles from 4-week-old rats (70–75 g) were used. Measurements of the electrical cable parameters of muscle fibres were done in muscles from adult 12-week-old female rats (230–260 g). Animals were fed ad libitum and were living in 12 h:12 h light/dark conditions at a thermostated temperature of 21°C. Rats were killed by cervical dislocation (4-week-old) or a halothane overdose followed by decapitation (12-week-old), and intact soleus or EDL muscles were dissected out.
No experiments were performed on live animals, and all handling and use of animals complied with Danish animal welfare legislation, including the animal housing and the killing of the rats that, in addition, was approved by the University Animal Welfare Officer.
All experiments with whole muscle took place at 30°C. Muscles were incubated in standard Krebs–Ringer bicarbonate solution containing (mm): 122 NaCl, 25 NaHCO3, 2.8 KCl, 1.2 KH2PO4, 1.2 MgSO4, 1.3 CaCl2 and 5.0 d-glucose. If not otherwise noted, solutions were gassed with a mixture of 95% O2 and 5% CO2 (pH ∼7.4) throughout the experiment. In all experiments the muscles were allowed to rest for 30 min after being mounted in the experimental chambers at optimal length in the standard solution. After initial incubation, the standard solution was exchanged for a solution in which 20 mm NaCl was substituted with 20 mm sodium methanesulfonate (control solution). The introduction of 20 mm sodium methanesulfonate was done to allow a later addition of up to 20 mm of the l-lactate (in the rest of the paper referred to as lactate) in exchange for sodium methanesulfonate without altering extracellular concentrations of Na+ and Cl−. The introduction of 20 mm lactate was without effect on the pH of the solution. d-Lactate was used in pilot experiments with similar effect but data are not shown. It is well established that methanesulfonate and methylsulphate do not conduct through muscle Cl− channels and substitution of Cl− with these ions therefore causes a reduction in GCl (Pedersen et al. 2005, 2009a,b;). In the standard experimental condition employed in this study where only 20 mm Cl− was substituted with equimolar methanesulfonate or methylsulphate, GCl will, however, only be marginally reduced when compared to conditions with no Cl− substitution. In Cl−-free solution, methanesulfonate salts replaced NaCl and KCl, and Ca(NO3)2 replaced CaCl2. In a few experiments, where methylsulphate was used instead of methanesulfonate, similar experimental observations were obtained (data not shown).
Isometric force measurements, electrical stimulation and muscle M-wave recordings
Muscles were mounted vertically on force transducers for measurements of isometric force production (Grass FT03) and their lengths were adjusted to give maximum force production. In general, contractions were evoked via field stimulation using constant voltage pulses applied through two platinum wire electrodes running perpendicular to the muscles. If not otherwise noted, pulses of 0.2 ms duration and supramaximal voltage (25–30 V cm−1) were used. M-waves were measured during contractions evoked via nerve stimulation as has been previously described (Overgaard & Nielsen, 2001).
Na+–K+ pump activity and intracellular Na+ content
Na+–K+ pump activity was determined as the ouabain-suppressible K+ uptake using 86Rb+ as a tracer for K+ as previously described (Buchanan et al. 2002). Briefly, after pre-incubation in standard solution, muscles were incubated for 20 min in solution with or without 10−3m ouabain followed by another 10 min of incubation in ouabain-containing solution with 0.1 μCi 86Rb+ ml−1 (specific activity 0.5 mCi mmol−1). Since, the ouabain specifically blocks the activity of the Na+–K+ pump (Clausen, 2003), the ouabain-suppressible 86Rb+ uptake reflects the activity of the Na+–K+ pump. This was calculated by subtraction of the ouabain-non-suppressible 86Rb+ uptake from the total 86Rb+ uptake. Intracellular Na+ content was determined by flame photometry as previously described (Everts & Clausen, 1992).
Intracellular pH measurements
Measurements of pHi were done using an epiflorescence system as previously described (Nielsen et al. 2001). In brief, muscles were loaded with 20 mm 2′,7′-bis(2-carboxyethyl)-5(6)-carboxyfluorescein for ∼30 min and then washed. The emission ratio was calibrated to pHi by the K+–nigericin technique (Thomas et al. 1979) in which the calibration curve was fitted by linear regression (r2 > 0.96).
Electrical cable parameters of muscle fibres
Recordings of cable parameters were done using a two-electrode constant-current technique as previously described (Pedersen et al. 2005). One electrode was used to inject constant current pulses and the other electrode was used to measure the membrane potential. The microelectrodes were filled with 2 m potassium citrate and had a resistance of 10–30 MΩ. In muscles at 9 mm K+, measurements were started after 65 min incubation in the solution and continued for 25 min. For measurements in the presence of lactate, the same time course was followed but 20 mm lactate was added after 60 min incubation at 9 mm K+. Composite conductance of K+ (GK) was taken as the membrane conductance in Cl−-free solution disregarding the contribution of the conductance of other ions. GCl was calculated by subtracting GK from the total resting membrane conductance (Gm) in solutions with normal Cl− concentration (Gm=GCl+GK). All fibres in which the resting membrane potential was below −50 mV or depolarized more than 5 mV while electrodes were inserted were disregarded.
Experiments with mechanically skinned muscle fibres
Experiments with mechanically skinned muscle fibres were performed at room temperature (20–22°C) using EDL muscles from 6- to 8-month-old male Sprague–Dawley rats. After dissection the muscles were blotted and placed under paraffin oil where the skinning procedure was performed as described in detail elsewhere (Lamb & Stephenson, 1994; Nielsen et al. 2009). Briefly, single fibres were isolated from the intact muscle. Using jeweller's forceps the surface of the fibre was gently pulled back causing the sarcolemma to roll back. The skinned fibre segment (3–4 mm long) was then mounted to a force transducer (AME875, SensoNor, Norway), stretched to 120% of resting length and immersed in a standard solution mimicking the cytosol (see solutions below). In this preparation, also known as the mechanically skinned fibre (Stephenson & Williams, 1981), the transverse tubular (T-) system reseals (Lamb et al. 1995; Launikonis & Stephenson, 2001) and it becomes normally polarized when the preparation is placed in a solution mimicking the cytosolic environment of the intact cell (Lamb & Stephenson, 1994; Lamb et al. 1995). The sealed T-system could then be electrically stimulated to produce action potentials leading to contraction as in intact muscles (Posterino et al. 2000). Thus, this preparation allows for the determination of excitability and force production while at the same time offering direct access to the intracellular environment, making it possible to obtain simultaneous control of pHi, the intracellular lactate concentration and T-system membrane potential.
Fibre excitation was achieved by electrical field stimulation (2 ms pulses at 75 V cm−1) using two platinum wire electrodes running parallel to the fibre. When the effects of depolarizing solutions were determined, fibres were subjected to the solution from a fully polarized state and stayed in the solution for exactly 15 s before the force response to excitation was determined and the fibre was then transferred back to the fully polarizing solution. In case there was a slight decrease in force when the fibre was repolarized, the force response was bracketed by determinations of force responses in the control solution. All force recordings were sampled at 1 kHz using a custom-made LabVIEW program (LabVIEW 8.0, National Instruments, Austin, TX, USA).
Solutions for the mechanically skinned fibre preparation
The intracellular solution (intracellular control solution) contained 126 mm K+, 40 mm Na+, 17 mm methylsulphate, 3 mm Cl−, 90 mm Hepes, 40 mm hexamethylene-diamine-tetraacetate (HDTA2−), 8 mm ATP, 10 mm CrP, 8.45 mm total Mg2+ (1 mm free Mg2+), 0.05 mm total EGTA. In depolarizing solutions with reduced [K+] (75 mm or 60 mm K+), K+ was replaced with NH4+, and methylsulphate was replaced with Cl− to maintain a constant [K+] and [Cl−] product ([K+][Cl−] of 378 mm2 (Pedersen et al. 2004). To produce solutions with lactate, 10 mm methylsulphate was exchanged for 10 mm lactate and total Mg2+ was increased to 8.54 mm in order to keep free Mg2+ at 1 mm. In Cl−-free solutions, Cl− was exchanged for methylsulphate. The osmolality of all solutions was 291 ± 6 mosmol kg−1, determined by vapour pressure osmometry (Wescor 5500, Logan, UT, USA), and pH was 7.10 ± 0.01. The pCa (–log10[Ca2+]) in the intracellular control solutions was determined to be 7.0 ± 0.4 using a Ca2+-sensitive electrode (Orion Research, Boston, MA, USA). Determinations of the Ca2+ sensitivity and the maximum Ca2+-activated force of the contractile apparatus was done in skinned fibres treated with 1% Triton X-100 for 3 min in order to lyse all membranes, washed twice, and exposed to a series of solutions where the free [Ca2+] was progressively increased from pCa 6.7 to 4.5 as described by Fink et al. (1986). The data were fitted to a Hill curve using GraphPad Prism 4 (San Diego, CA, USA) and the pCa required to elicit 50% of the maximum force and Hill coefficients were obtained for all conditions.
Statistics
All data are expressed as means ±s.e.m. The statistical significance of any difference between groups was ascertained using Student's one- or two-tailed t test for paired or non-paired observations.
Results
Effects of lactate on action potential activity, GCl and contractile function in depolarized muscles
Figure 1A–D shows the effect of adding 20 mm lactate on the force and action potential activity in response to electrical stimulation in soleus muscles depolarized by prolonged exposure to a control solution containing 11 mm K+. The depolarization produced by the elevation in extracellular K+ drastically depressed the action potential activity as evidenced by reduction of M-wave amplitude, a prolongation of the time from the stimulus at time zero to the peak of the M-wave (Fig. 1Aversus 1B) and an 83% reduction in M-wave area (Fig. 1D). Simultaneously, the force production was reduced to 17% of the force at 4 mm K+. When 20 mm lactate was added subsequently, a significant recovery of both M-wave area and force was observed (Fig. 1D). Figure 1E shows that the effect of lactate was dose dependent, being already present at 5 mm and saturated at 15 mm. Similar effects of lactate were found in EDL muscles. Thus, when the maximal tetanic force, obtained during 0.5 s trains of 90 Hz stimulation, was reduced in these muscles to 35 ± 6% (n= 4) of the control level by exposure to 13 mm K+, subsequent addition of 20 mm lactate led to a 2.6 ± 0.8-fold increase in the force production.
Figure 1. Effect of lactate on M-wave and contractile properties in K+-depressed soleus muscles.
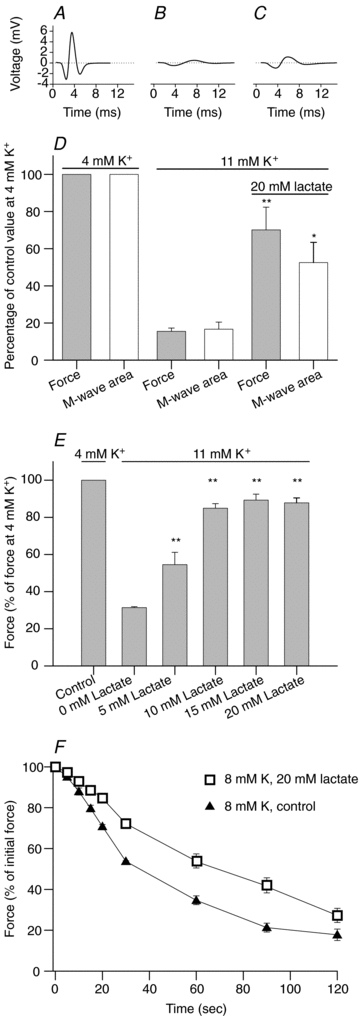
A–E, after determination of control tetanic force at 4 mm K+, the intact soleus muscles were incubated for 90 min in control solution with 11 mm K+ and then for 30 min in solution with 11 mm K+ and 20 mm lactate. Tetanic contractions were elicited every 10 min using 60 Hz pulse trains of 2 s duration. Representative M-wave traces from a single soleus muscle recorded during contractions at the end of the incubation period at 4 mm K+ (A), 11 mm K+ (B) and 11 mm K+ with 20 mm lactate (C). D, tetanic force and M-wave area at end of the incubation at 4 mm K+, 11 mm K+, and 11 mm K+ with 20 mm lactate, as indicated by bars (n= 6). E, effect of lactate concentration on tetanic force in soleus muscles at 11 mm K+ (n= 6–29). F, force production in muscles pre-equilibrated at 8 mm K+ for 60 min after which a prolonged contraction was induced by 120 s of 60 Hz continuous stimulation in the presence (open squares) or absence (filled triangles) of 20 mm lactate (n= 4). In all panels the data show means and s.e.m. Significantly different from the corresponding value at 4 mm K+: *P < 0.05, **P < 0.01.
To explore the role of lactate in muscle function during intensive contractile activity, soleus muscles were pre-incubated at 8 mm K+ to mimic the conditions of working muscle while still retaining excitability, and then stimulated continuously at 60 Hz for 2 min to induce fatigue. Before fatigue, there was no difference in the maximal force at 4 and 8 mm K+ either in the presence or in the absence of lactate. However, Fig. 1F shows that the force decline during 60 Hz stimulation was reduced in muscle that had been pre-incubated with lactate. Thus, in control muscles without lactate, force declined to 50% of the initial force at 8 mm K+ in ∼40 s while in muscles with lactate it lasted ∼75 s for the force to decline by 50%.
Since ClC-1 channels are inhibited by small carboxylic acids similar to lactate and a reduced GCl previously has been shown to recover excitability and contractile function in K+-depressed muscles we next examined whether the lactate-induced improvement of muscle excitability was related to an inhibition of Cl− channels. Figure 2 shows that when GCl was determined in soleus muscles using a conventional two-electrode constant current technique, 20 mm lactate reduced GCl by 31%. Such a reduction in GCl has previously been shown to significantly increase the tolerance of muscles to elevated extracellular K+ (Pedersen et al. 2005). In contrast to the change in GCl, GK was unaffected by lactate (Fig. 2).
Figure 2. Effect of lactate on the specific membrane conductance for Cl− and K+ in soleus muscles.
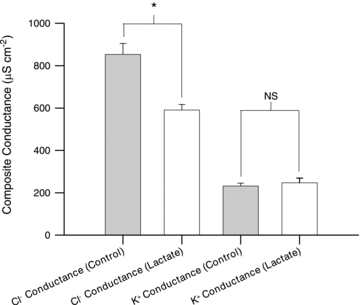
Muscles were incubated in 9 mm K+ with or without 20 mm lactate and with or without Cl− for 60 min before the membrane conductance was determined. K+ conductance is the membrane conductance in the absence of Cl−. Cl− conductance is calculated by subtracting the membrane conductance in the absence of Cl− from the total membrane conductance determined in soleus fibres incubated in control solution. Data are shown as means with s.e.m. (n= 16–33 muscle fibres). *Significant difference between values obtained with and without lactate, P < 0.05. NS, not significantly different.
Role of reduced intracellular pH in the effect of lactate on the function of depolarized muscle
To clarify the role of pHi in the observed effect of lactate on muscle excitability (Fig. 1), we first determined the magnitude of the reduction in pHi after addition of 20 mm lactate to soleus muscles. Secondly, we tested the effect of such a reduction in pHi on the contractile force in muscles depressed by elevated K+ using an increase in CO2 in the gas mixture that resulted in a pHi reduction of similar magnitude to the lactate addition. Figure 3A shows a representative pHi trace demonstrating that the addition of 20 mm lactate led to a fast reduction in pHi followed by a slow recovery. As shown in Fig. 3C, the average maximal reduction in pHi was 0.13 pH units. To evaluate the effect of this reduction in pHi on the force response in muscles at elevated K+, in another series of experiments this pHi reduction was mimicked by increasing CO2 in the gas mixture, in equilibrium with the incubation solution, from 5 to 7%. Figure 3B and C shows that this increase in CO2 induced a slightly larger and more stable reduction in pHi (P= 0.003 when compared to addition of lactate). Despite this, the recovery of force induced by the increase in CO2 was substantially smaller than the force recovery observed after addition of 20 mm lactate (Fig. 3D), indicating that reduced pHi was of little importance for the force recovery induced by lactate in K+-depressed muscles.
Figure 3. Effect of lactate and increased CO2 on pHi and force in soleus muscles.
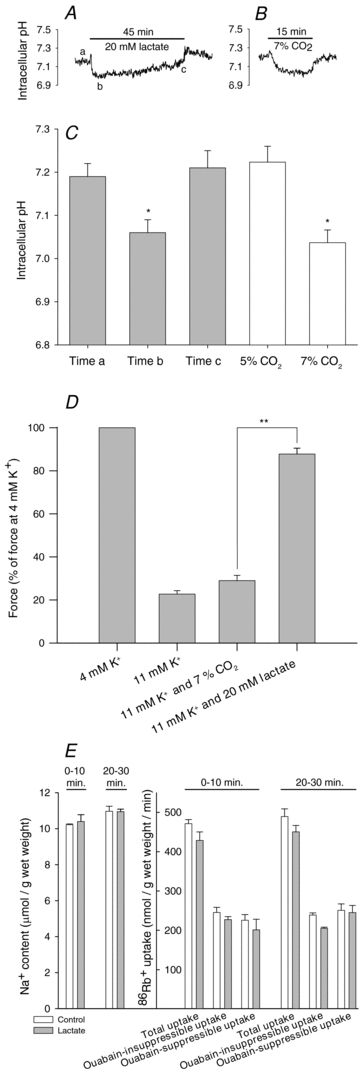
A and B, representative traces showing pHi in muscles exposed to 20 mm lactate or increased CO2 (from 5% to 7%): before exposure to 20 mm lactate (a), the lowest pHi after exposure to lactate (b), and pHi after 45 min of exposure to lactate (c). C, summarized values for pHi in soleus muscles exposed to 20 mm lactate or increased CO2. Data show means and s.e.m. (n= 4–6). *Significantly different from pHi at the beginning of experiments. D, effect of 7% CO2 and 20 mm lactate on tetanic force in soleus muscles depolarized by incubation at 11 mm K+. Data show means and s.e.m. (n= 7–8). *P < 0.05 and **P < 0.01, significantly different from force in soleus muscles incubated in control solution with 11 mm K+. E, effect of 20 mm lactate on intracellular Na+ content and 86Rb+ uptake. Two groups of soleus muscles were pre-incubated in control solution with 11 mm K+. After 30 min 20 mm lactate was added to one group of soleus muscles and Na+ content and 86Rb+ uptake was assessed from 0 to 10 min and from 20 to 30 min of the subsequent incubation. Data show means and s.e.m. (n= 6).
Effects of lactate on intracellular Na+ content and Na+–K+ pump activity
In a previous study (Kristensen et al. 2005), it was suggested that addition of lactate to soleus muscles leads to an increase in intracellular Na+ that stimulates the Na+–K+ pump. To investigate such an effect, the intracellular Na+ content and the ouabain-suppressible 86Rb+ uptake was determined after 10 and 30 min exposure of soleus muscles to 20 mm lactate (Fig. 3E). These experiments showed that lactate did not affect either the intracellular Na+ content (P= 0.64 and 0.96 after 10 and 30 min, respectively) or the total ouabain-suppressible 86Rb+ uptake (P= 0.44 and 0.82 after 10 and 30 min, respectively).
Effects of lactate and reduced GCl on contractile function of depolarized skinned fibres
To further control for the role of a change in the intracellular environment in the effect of lactate on muscle excitability, experiments were conducted using a mechanically skinned muscle fibre preparation where the intracellular environment could be directly controlled via the solution used for incubation. In this preparation, the sarcolemma membrane was removed by micro-dissection while the transverse tubular (T-) system seals off and becomes normally polarized when the preparation is incubated in cytosolic-like solution. This preparation can, therefore, be field stimulated to generate action potentials in the T-tubular system that triggers the fibres to contract. Figure 4A shows a representative trace of twitch and tetanic force responses in such a mechanically skinned fibre obtained from EDL muscles. When normally polarized (incubated in intracellular control solution with 126 mm K+) the twitch and tetanic forces were about 35 and 90% of the maximal Ca2+-activated force that the fibre could generate, respectively. This maximal Ca2+-activated force of the fibre was measured by incubation in a high Ca2+ solution (see trace at far right). When depolarizing the fibre, by lowering the K+ concentration in the solution to either 75 or 60 mm K+ (corresponding to a membrane potential of roughly −67 mV and −62 mV, respectively) there was a clear and reversible reduction in both twitch and tetanic force. However, when lactate was included in the depolarizing solutions the drop in force was reduced when compared to observations in depolarizing solutions without lactate. As shown in Fig. 4B, this enhanced force production in depolarized fibres when exposed to lactate was a general finding for both twitch and tetanic force with the effect of lactate tending to be more marked in the most depolarized conditions (60 compared to 75 mm K+). Contrary to this effect of lactate, the trace in Fig. 4A illustrates that when the fibres were normally polarized (i.e. incubated in control buffer with 126 mm K+) exposure to lactate had no effect on the twitch and tetanic force responses. Since the experiments on intact muscle had implicated a reduced GCl in the lactate-induced recovery of excitability and force in K+-depressed muscle, a series of experiments with skinned fibre was next conducted in which GCl was eliminated by either using Cl−-free solution or by inhibition of Cl− channels with 9-Anthracene-carboxylic acid (9-AC). For both approaches of eliminating GCl, two important observations were made (Fig. 4C and D). Firstly, in the absence of lactate, the force production in depolarized fibres was markedly enhanced when compared to conditions with normal GCl in the T-system. Secondly, the enhancement of force with lactate in depolarized fibres disappeared. To test for a possible effect of lactate and lowered K+ on the Ca2+ sensitivity and the maximal force of the contractile apparatus, the force–pCa2+ relationship was determined in five fibres in control solution, in control solution with 10 mm lactate and in depolarizing solutions with reduced K+ (60 and 75 mm). These experiments showed that lactate and reduced K+ had no effect on the maximal Ca2+-activated force, averaging 0.88 ± 0.08 mN (n= 5). Likewise, the Ca2+ sensitivity of the fibres was unaffected with no significant differences in [Ca2+] required to elicit 50% of the maximum force or the slope of the force–pCa2+ curves, averaging 5.77 ± 0.03 and 3.9 ± 0.1, respectively.
Figure 4. Effect of lactate and T-system depolarization on the force response of skinned fibres from EDL muscles.
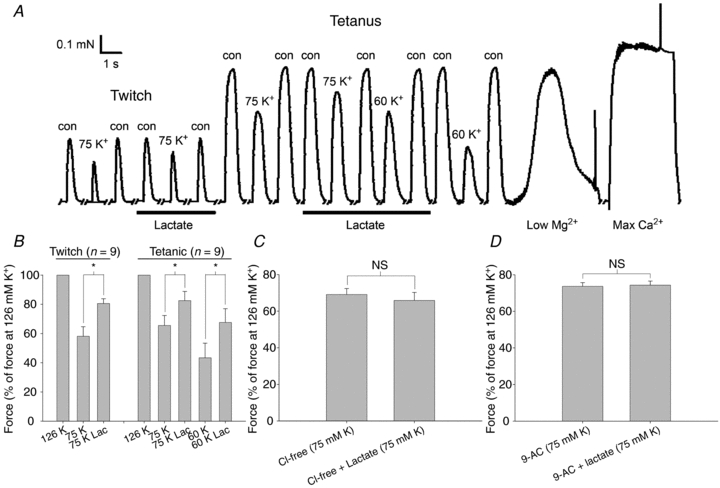
A, representative trace showing the effect of 10 mm lactate (indicated by bars) on twitch and tetanic force in a skinned muscle fibre when incubated in the intracellular control solution (con) and when depolarized by incubation in solutions with 75 and 60 mm K+. Tetani were elicited by 25 Hz pulse trains of 2 s duration. At the end of the protocol a prolonged contraction was elicited by exposure of the fibre to maximally Ca2+-activating solution. B, summarized data showing the effect of 10 mm lactate on maximal twitch and tetanic force in skinned muscle fibres incubated in the intracellular control solution or depolarized by incubation in solutions with 75 or 60 mm K+. *Force in the presence of lactate is significantly higher than without lactate, P < 0.05. C and D, summarized data showing the effect of 10 mm lactate on skinned fibres depolarized by incubation at 75 mm K+ in a Cl−-free solution (C) or in the presence of 100 μm 9-AC (D). Data show means and s.e.m. (n= 3–9). NS, not significant.
Discussion
The major finding of this study was that addition of lactate at concentrations of 5–20 mm can inhibit ClC-1 Cl− channels and markedly increase the excitability and the contractile function of both fast-twitch and slow-twitch muscles from the rat.
Effects of lactate on excitability and contractile function in depolarized muscles
Although several studies have failed to demonstrate a depressing effect of lactate on the function of skeletal muscle (Andrews et al. 1996; Dutka & Lamb, 2000; Posterino & Fryer, 2000; Posterino et al. 2001; Kristensen et al. 2005) it is still a widespread belief that lactate accumulation is an important cause of muscle fatigue. However, since it is likely that muscle fatigue can arise from multiple causes, it is relevant to search for possible interacting effects of the cellular changes in active muscle that have been proposed to contribute to fatigue in order to further understand the physiological significance of such changes. Here we explore for possible interacting effects of the lactate and depolarization because it is well established that the repetitive action potential firing in active muscle leads to a rise in extracellular K+ that could contribute to fatigue by depolarizing the muscle fibres and thereby reduce their excitability. This report describes three series of experiments which demonstrate that, in contrast to the notion of lactate as a fatiguing agent, the lactate actually counteracts loss of muscle function when muscles are depolarized: Firstly, when muscles were exposed to elevated extracellular K+, where loss of excitability caused a marked depression in tetanic force, the addition of low concentrations of lactate caused a pronounced recovery of excitability and force. Secondly, in experiments where intact muscles were exposed to only moderately elevated extracellular K+, thereby ensuring excitability was maintained at the onset of continuous stimulation, lactate markedly reduced the decline in force during stimulation. Thirdly, of key importance in the evaluation of the possible loss of muscle excitability during activity is the ability of the T-system to maintain its excitability when muscles are depolarized. To expand on this question we used the mechanically skinned muscle fibre, which allows the excitability of the T-system to be evaluated specifically (Pedersen et al. 2004). Also in these experiments lactate improved force when the fibres were depolarized. During intense exercise, lactate accumulation in the active muscles starts within a few seconds (Jacobs et al. 1983). Our results suggest, therefore, that the production of lactate provides an early protection of the excitability of active muscles against the depressing effects of the work-induced elevation in extracellular K+.
The present experiments were done at 30°C (whole muscle) or room temperature (skinned fibres). The reason for this was that the muscle preparations used are unable to maintain viability for prolonged periods at physiological temperatures (Segal & Faulkner, 1985; Lännergren & Westerblad, 1987; Lamb & Stephenson, 1994). However, in a study on isolated whole muscle it was demonstrated that, qualitatively, the loss of force induced by depolarization and the subsequent recovery of force after addition of lactic acid was the same at 30 and 37°C (Nielsen et al. 2001).
Role of GCl in the effect of lactate on the function of depolarized muscles
Based on previous studies showing that small carboxylic acids can inhibit the muscular ClC-1 Cl− channels we next proceeded to investigate whether the lactate, which is a carboxylic acid, was reducing GCl and thereby recovered excitability in depolarized muscle. Several findings indeed argue for such a role of the ClC-1 channels in the effect of lactate. Firstly, addition of 20 mm lactate caused a ∼30% reduction in the GCl of soleus muscles. In other studies a reduction in GCl of similar magnitude has been shown to significantly increase muscle excitability and recover function after K+-induced depolarizations (Pedersen et al. 2005). Secondly, the effect of lactate was absent in Cl−-free solutions. Thirdly, the effect of lactate was prevented by 9-AC which inhibits ClC-1 channels.
The recovery of pHi after addition of lactate (Fig. 3A) most likely signifies an increase in the activity of transporters involved in the cellular pH regulation (Juel, 1997). It could be envisaged, therefore, that an increased influx of Na+ via the Na+/H+ exchanger after the addition of lactate led to accumulation of intracellular Na+. Since such an increase in intracellular Na+ would increase the activity of the Na+–K+ pumps, which has been shown to improve the excitability of depolarized muscles per se (Clausen, 2003), it was possible that elevated Na+–K+ pump activity contributed to the recovery of force and excitability after addition of lactate to muscles at elevated K+. Arguing against such a role for intracellular Na+ are the facts that the effect of lactate on force was also seen in skinned fibres where lactate was unlikely to exert an effect on the cytosolic Na+ concentration and that the addition of lactate to intact muscles had no effect on either the intracellular Na+ content or the total or ouabain-suppressible 86Rb+ uptake. Although in contrast to observations by Kristensen et al. (2005), who in almost identical experiments found an around 23% increase in the total 86Rb+ uptake after addition of 20 mm lactate, this indicates that the Na+–K+ pumps were not involved in the lactate-induced increase in excitability in the present study.
Evidence for a pH-independent effect of lactate on the function of depolarized muscles
In previous studies, the effects of extracellular added lactate and lactic acid on muscle excitability and GCl have been suggested to be caused by an intracellular acidification. Although there is evidence that intracellular acidification certainly can produce these effects, the observation that extracellular lactate at neutral pH only leads to a minor reduction in intracellular pH (Wetzel et al. 2001) indicates that additional mechanisms play a role in the effect of lactate and lactic acid on muscle excitability. To evaluate the functional significance of pHi in the observed effect of lactate, we therefore measured pHi before and after addition of a high concentration of lactate to the extracellular solution of soleus muscles. These experiments showed that the lactate only caused a very moderate intracellular acidification and that when this acidification was mimicked by slight elevation of CO2 there was only a negligible effect of muscle force. Further support for a direct, pH-independent effect of lactate comes from the experiment with skinned fibres from EDL where the effect of lactate was maintained despite pHi and metabolic status was kept constant by the incubation solution. In summary, our experiments suggest that lactate caused a direct inhibition of ClC-1 channels that improved the excitability of depolarized muscle.
Importance of lactate for maintenance of muscle excitability during exercise
During exercise, active muscles lose K+ leading to an extracellular accumulation of the ion that can potentially jeopardize their excitability. Our results suggest, however, that simultaneous production of lactate can provide early protection of the excitability of active muscles against the depressing effects of elevated extracellular K+, which together with other protective mechanisms (Nielsen & de Paoli, 2007) may reduce the importance of elevated extracellular K+ for the development of fatigue. Since fast-twitch fibres are likely to have a larger release of K+ (Clausen et al. 2004) and a larger production of lactate than slow-twitch fibres, this effect may be most relevant for the fast-twitch fibres. However, since K+ lost from active muscles is exported to the bloodstream, the resting or less active muscles can also experience an elevation of extracellular K+ of up to more than 8 mm K+ during intense exercise (Sejersted & Sjøgaard, 2000), which is sufficient to severely reduce their contractile endurance if later engaged in contractile activity (Cairns et al. 1997; Clausen & Nielsen, 2007). Importantly, the finding that the effects of lactate in the present study were seen at extracellular additions of less than 10 mm indicates that lactate released to the bloodstream from active muscles during exercise will also lead to increases in the excitability of the less active or less lactate-producing muscle fibres in the body. Thus, lactate accumulating in the bloodstream could serve as a systemic signal that during exercise leads to improvement in the tolerance of muscles to elevated K+ everywhere in the body irrespective of their activity level or histochemical phenotype. In this context, the present findings add to the emerging picture of lactate being important for maintaining the function of several tissues with roles for lactate as varied as being an important energy substrate in skeletal muscle, heart and brain, a major gluconeogenic precursor, an autocrine signalling molecule with effects on gene expression associated with exercise adaptation (Cairns, 2004; Gladden, 2004; Philp et al. 2005; Brooks, 2007, 2009) and, as shown in the present study, a metabolite that reinforces muscle excitability.
Acknowledgments
We thank T. L. Andersen, V. Uhre and M. S. Johansen for technical assistance and Dr Christian Aalkjær for letting us use his equipment for intracellular pH measurements. This work was supported by The Danish Medical Research Council (T.H.P. and O.B.N.), The Lundbeck Foundation (O.B.N.) and the Faculty of Health Science, University of Aarhus (T.H.P. and F.V.deP.).
Glossary
Abbreviations
- EDL
extensor digitorum longus
Author contributions
Conception and design of the experiments: F.V.deP., R.J., N.Ø., T.H.P. and O.B.N. Collection, analysis and interpretation of data: F.V.deP., R.J., N.Ø., T.H.P. and O.B.N. Drafting the article or revising it critically for important intellectual content: F.V.deP., N.Ø., T.H.P. and O.B.N. The experiments were performed at the Institute of Physiology and Biophysics, University of Aarhus, DK-8000 C, Denmark (intact muscles) and The Institute of Sports Science and Clinical Biomechanics, University of Southern Denmark, DK-5230 M, Denmark (skinned fibres).
References
- Andrews MA, Godt RE, Nosek TM. Influence of physiological l(+)-lactate concentrations on contractility of skinned striated muscle fibers of rabbit. J Appl Physiol. 1996;80:2060–2065. doi: 10.1152/jappl.1996.80.6.2060. [DOI] [PubMed] [Google Scholar]
- Brooks GA. Lactate: link between glycolytic and oxidative metabolism. Sports Med. 2007;37:341–343. doi: 10.2165/00007256-200737040-00017. [DOI] [PubMed] [Google Scholar]
- Brooks GA. Cell–cell and intracellular lactate shuttles. J Physiol. 2009;587:5591–5600. doi: 10.1113/jphysiol.2009.178350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchanan R, Nielsen OB, Clausen T. Excitation- and β2-agonist-induced activation of the Na+–K+ pump in rat soleus muscle. J Physiol. 2002;545:229–240. doi: 10.1113/jphysiol.2002.023325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cairns SP. Lactic acid and exercise performance: culprit or friend? Sports Med. 2004;36:279–291. doi: 10.2165/00007256-200636040-00001. [DOI] [PubMed] [Google Scholar]
- Cairns SP, Hing WA, Slack JR, Mills RG, Loiselle DS. Different effects of raised [K+]o on membrane potential and contraction in mouse fast- and slow-twitch muscle. Am J Physiol Cell Physiol. 1997;273:C598–C611. doi: 10.1152/ajpcell.1997.273.2.C598. [DOI] [PubMed] [Google Scholar]
- Clausen T. Na+-K+ pump regulation and skeletal muscle contractility. Physiol Rev. 2003;83:1269–1324. doi: 10.1152/physrev.00011.2003. [DOI] [PubMed] [Google Scholar]
- Clausen T, Nielsen OB. Potassium, Na+, K+-pumps and fatigue in rat muscle. J Physiol. 2007;584:295–304. doi: 10.1113/jphysiol.2007.136044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clausen T, Overgaard K, Nielsen OB. Evidence that the Na+-K+ leak/pump ratio contributes to the difference in endurance between fast- and slow-twitch muscles. Acta Physiol Scand. 2004;180:209–216. doi: 10.1111/j.0001-6772.2003.01251.x. [DOI] [PubMed] [Google Scholar]
- Dutka TL, Lamb GD. Effect of lactate on depolarization-induced Ca2+ release in mechanically skinned skeletal muscle fibres. Am J Physiol Cell Physiol. 2000;278:C517–C525. doi: 10.1152/ajpcell.2000.278.3.C517. [DOI] [PubMed] [Google Scholar]
- Everts ME, Clausen T. Activation of the Na-K pump by intracellular Na in rat slow- and fast-twitch muscle. Acta Physiol Scand. 1992;145:353–362. doi: 10.1111/j.1748-1716.1992.tb09375.x. [DOI] [PubMed] [Google Scholar]
- Fink RH, Stephenson DG, Williams DA. Calcium and strontium activation of single skinned muscle fibres of normal and dystrophic mice. J Physiol. 1986;373:513–525. doi: 10.1113/jphysiol.1986.sp016060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fitts RH. Cellular mechanisms of muscle fatigue. Physiol Rev. 1994;74:49–94. doi: 10.1152/physrev.1994.74.1.49. [DOI] [PubMed] [Google Scholar]
- Gladden LB. Lactate metabolism: a new paradigm for the third millennium. J Physiol. 2004;558:5–30. doi: 10.1113/jphysiol.2003.058701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs I, Tesch PA, Bar-Or O, Karlsson J, Dotan R. Lactate in human skeletal muscle after 10 and 30 s of supramaximal exercise. J Appl Physiol. 1983;55:365–367. doi: 10.1152/jappl.1983.55.2.365. [DOI] [PubMed] [Google Scholar]
- Juel C. Lactate-proton transport in skeletal muscle. Physiol Rev. 1997;77:321–358. doi: 10.1152/physrev.1997.77.2.321. [DOI] [PubMed] [Google Scholar]
- Kristensen M, Albertsen J, Rentsch M, Juel C. Lactate and force production in skeletal muscle. J Physiol. 2005;562:521–526. doi: 10.1113/jphysiol.2004.078014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb GD, Junankar PR, Stephenson DG. Raised intracellular [Ca2+] abolishes excitation–contraction coupling in skeletal muscle fibres of rat and toad. J Physiol. 1995;489:349–362. doi: 10.1113/jphysiol.1995.sp021056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb GD, Stephenson DG. Effects of intracellular pH and [Mg2+] on excitation–contraction coupling in skeletal muscle fibres of the rat. J Physiol. 1994;478:331–339. doi: 10.1113/jphysiol.1994.sp020253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb GD, Stephenson DG. Point: lactic acid accumulation is an advantage during muscle activity. J Appl Physiol. 2006;100:1410–1412. doi: 10.1152/japplphysiol.00023.2006. [DOI] [PubMed] [Google Scholar]
- Lännergren J, Westerblad H. The temperature dependence of isometric contractions of single, intact fibres dissected from a mouse foot muscle. J Physiol. 1987;390:285–293. doi: 10.1113/jphysiol.1987.sp016700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Launikonis BS, Stephenson DG. Effects of membrane cholesterol manipulation on excitation–contraction coupling in skeletal muscle of the toad. J Physiol. 2001;534:71–85. doi: 10.1111/j.1469-7793.2001.00071.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen J, Schrøder HD, Rix CG, Ortenblad N. Distinct effects of subcellular glycogen localization on tetanic relaxation time and endurance in mechanically skinned rat skeletal muscle fibres. J Physiol. 2009;587:3679–3690. doi: 10.1113/jphysiol.2009.174862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen OB, de Paoli FV. Regulation of Na+-K+ homeostasis and excitability in contracting muscles: implications for fatigue. Appl Physiol Nutr Metab. 2007;32:974–984. doi: 10.1139/H07-099. [DOI] [PubMed] [Google Scholar]
- Nielsen OB, de Paoli F, Overgaard K. Protective effects of lactic acid on force production in rat skeletal muscle. J Physiol. 2001;536:161–166. doi: 10.1111/j.1469-7793.2001.t01-1-00161.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overgaard K, Nielsen OB. Activity-induced recovery of excitability in K+-depressed rat soleus muscle. Am J Physiol Regul Integr Comp Physiol. 2001;280:R48–R55. doi: 10.1152/ajpregu.2001.280.1.R48. [DOI] [PubMed] [Google Scholar]
- Pate E, Bhimani M, Franks-Skiba K, Cooke R. Reduced effect of pH on skinned rabbit psoas muscle mechanics at high temperatures: implications for fatigue. J Physiol. 1995;486:689–694. doi: 10.1113/jphysiol.1995.sp020844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen TH, de Paoli F, Nielsen OB. Increased excitability of acidified skeletal muscle: role of chloride conductance. J Gen Physiol. 2005;125:237–246. doi: 10.1085/jgp.200409173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen TH, de Paoli FV, Flatman JA, Nielsen OB. Regulation of ClC-1 and KATP channels in action potential-firing fast-twitch muscle fibers. J Gen Physiol. 2009a;134:309–322. doi: 10.1085/jgp.200910290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen TH, Nielsen OB, Lamb GD, Stephenson DG. Intracellular acidosis enhances the excitability of working muscle. Science. 2004;305:1144–1147. doi: 10.1126/science.1101141. [DOI] [PubMed] [Google Scholar]
- Pedersen TH, Macdonald WA, de Paoli FV, Gurung IS, Nielsen OB. Comparison of regulated passive membrane conductance in action potential firing fast and slow-twitch muscle. J Gen Physiol. 2009b;134:323–337. doi: 10.1085/jgp.200910291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philp A, Macdonald AL, Watt PW. Lactate – a signal coordinating cell and systemic function. J Exp Biol. 2005;208:4561–4575. doi: 10.1242/jeb.01961. [DOI] [PubMed] [Google Scholar]
- Posterino GS, Dutka TL, Lamb GD. L(+)-lactate does not affect twitch and titanic responses in mechanically skinned mammalian muscle fibres. Pflügers Arch. 2001;442:197–203. doi: 10.1007/s004240100528. [DOI] [PubMed] [Google Scholar]
- Posterino GS, Fryer MW. Effects of high myoplasmaic L-lactate concentration on E-C coupling in mammalian skeletal muscle. J Appl Physiol. 2000;89:517–528. doi: 10.1152/jappl.2000.89.2.517. [DOI] [PubMed] [Google Scholar]
- Posterino GS, Lamb GD, Stephenson DG. Twitch and tetanic force responses and longitudinal propagation of action potentials in skinned skeletal muscle fibres of the rat. J Physiol. 2000;527:131. doi: 10.1111/j.1469-7793.2000.t01-2-00131.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rychkov GY, Pusch M, Roberts ML, Bretag AH. Interaction of hydrophobic anions with the rat skeletal muscle chloride channel ClC-1: effects on permeation and gating. J Physiol. 2001;530:379–393. doi: 10.1111/j.1469-7793.2001.0379k.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segal SS, Faulkner JA. Temperature-dependent physiological stability of rat skeletal muscle in vitro. Am J Physiol Cell Physiol. 1985;248:C265–C270. doi: 10.1152/ajpcell.1985.248.3.C265. [DOI] [PubMed] [Google Scholar]
- Sejersted OM, Sjøgaard G. Dynamics and consequences of potassium shifts in skeletal muscle and heart during exercise. Physiol Rev. 2000;80:1411–1481. doi: 10.1152/physrev.2000.80.4.1411. [DOI] [PubMed] [Google Scholar]
- Stephenson DG, Williams DA. Calcium-activated force responses in fast- and slow-twitch skinned muscle fibres of the rat at different temperature. J Physiol. 1981;317:281–302. doi: 10.1113/jphysiol.1981.sp013825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas JA, Buchsbaum RN, Zimniak A, Racker E. Intracellular pH measurements in Ehrlich ascites tumor-cells utilizing spectroscopic probes generated in situ. Biochemistry. 1979;18:2210–2218. doi: 10.1021/bi00578a012. [DOI] [PubMed] [Google Scholar]
- Westerblad H, Bruton JD, Lännergren J. The effect of intracellular pH on contractile function of intact, single fibres of mouse muscle declines with increasing temperature. J Physiol. 1997;500:193–204. doi: 10.1113/jphysiol.1997.sp022009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wetzel P, Hasse A, Papadopoulos S, Voipio J, Kaila K, Gros G. Extracellular carbonic anhydrase activity facilitates lactic acid transport in rat skeletal muscle fibres. J Physiol. 2001;531:743–756. doi: 10.1111/j.1469-7793.2001.0743h.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


