Abstract
Tissue and molecular heterogeneities are present in the developing secondary palate along the anteroposterior (AP) axis in mice. Here, we show that Wnt5a and its receptor Ror2 are expressed in a graded manner along the AP axis of the palate. Wnt5a deficiency leads to a complete cleft of the secondary palate, which exhibits distinct phenotypic alterations at histological, cellular and molecular levels in the anterior and posterior regions of the palate. We demonstrate that there is directional cell migration within the developing palate. In the absence of Wnt5a, this directional cell migration does not occur. Genetic studies and in vitro organ culture assays further demonstrate a role for Ror2 in mediating Wnt5a signaling in the regulation of cell proliferation and migration during palate development. Our results reveal distinct regulatory roles for Wnt5a in gene expression and cell proliferation along the AP axis of the developing palate, and an essential role for Wnt5a in the regulation of directional cell migration.
Keywords: Wnt5a, Ror2, Palatogenesis, Cleft palate, Non-canonical Wnt pathway, Cell migration
INTRODUCTION
Cleft palate is a congenital disorder arising from a failure in the highly regulated multiple-step process of the secondary palate development. In mice, the palatal shelves arise from the outgrowing maxillary prominences at embryonic day 11.5 (E11.5), and then grow vertically down on the side of the tongue (E12.5–E13.5), followed by rapid elevation to a horizontal position around E14.0. Subsequently, two palatal shelves meet at midline and fuse to establish the mesenchymal continuity of the definitive palate. Recent studies have demonstrated that the developing palate is not a homogeneous organ (Hilliard et al., 2005; Okano et al., 2006; Gritli-Linde, 2007). Many genes involved in palate development exhibit regional differential expression along the anteroposterior (AP) axis. The anterior and posterior palatal mesenchyme also shows different molecular and cellular responses to growth factors. This tissue and molecular heterogeneity could lead to differential cell behaviors, such as cell proliferation, production of different extracellular matrix and cell migration, in the palate along the AP axis. However, despite rapid progress in the field, the exact cellular and molecular mechanisms that are involved in morphological changes during palate development remain unknown.
The Wnt family genes encode a large and diverse group of signaling molecules in the patterning, proliferation and differentiation of a variety of organs and cell types (Cadigan and Nusse, 1997). Wnt ligand binds to its receptor Frizzled (Fz) and Lrp5/6 to activate the canonical Wnt/β-catenin signaling pathway, or functions through β-catenin-independent pathways, the noncanonical pathways, including the planar cell polarity (PCP) and Wnt/Ca2+ pathways (Veeman et al., 2003). Wnt ligands are typically classified into canonical and noncanonical Wnts by the pathways they work through. Wnt5a is a representative of the noncanonical Wnt class. One function of the noncanonical pathway is its inhibition of the canonical Wnt signaling. In Xenopus, overexpression of Wnt5a blocks secondary axis induction by Wnt8 (Torres et al., 1996). In the developing limbs, Wnt5a blocks canonical Wnt pathway by promoting degradation of β-catenin in a GSK3-independent way (Topol et al., 2003). Noncanonical Wnt signaling also regulates, via the PCP pathway, convergent extension movement, an essential cell migration process during vertebrate gastrulation (Mlodzik, 2002). Altered Wnt5a function leads to dysregulated convergent extension movement in vertebrates (Moon et al., 1993; Kilian et al., 2003). It was recently reported that an alternative Wnt receptor, Ror2, an orphan tyrosine kinase, mediates Wnt5a-initiated noncanonical signaling and is required for the Wnt5a-mediated inhibition of the canonical signaling (Oishi et al., 2003; Mikels and Nusse, 2006). Interestingly, Wnt5a is also capable of activating Wnt/β-catenin signaling in the presence of Fz4 and Lrp5 (Mikels and Nusse, 2006). In addition, the orphan receptor Ryk could also bind to Wnt5a besides to other Wnt ligands (Lu et al., 2004; Keeble et al., 2006).
Wnt signaling was initially implicated in palatogenesis based on the findings that WNT3 mutations in humans and Wnt9b targeted inactivation in mice cause a cleft palate phenotype with an incomplete penetrance (Niemann et al., 2004; Carroll et al., 2005). Mapping of the chromosomal translocation in individuals with non-syndromic cleft lip and palate identified a region containing WNT5A (Blanton et al., 2004). Although β-catenin expression was reported in the developing palate (Martinez-Alvarez et al., 2000; Tudela et al., 2002; Nawshad and Hay, 2003), specific ablation of β-catenin in the epithelial-derived tissues by a K14-Cre transgenic allele appeared not to affect palate formation (Huelsken et al., 2001).
We report here that Wnt5a and its receptors exhibit a gradient expression along the AP axis in the developing palate, consistent with a complete cleft palate phenotype in Wnt5a−/− mice. We analyzed the developing palate of Wnt5a mutants at the histological, cellular and molecular levels. We show for the first time the existence of active directional migration of mesenchymal cells in the palate, which is disrupted in the Wnt5a−/− palate. We further reveal a chemotactic role for Wnt5a in the palate. Genetic studies and in vitro assays provide evidence that Ror2 mediates Wnt5a signaling through a noncanonical pathway to regulate cell proliferation and cell migration during palatogenesis.
MATERIALS AND METHODS
Animal
Wnt5a+/− (Yamaguchi et al., 1999), TOPGAL (DasGupta and Fuchs, 1999) and Actinβ-Egfp transgenic mice were purchased from the Jackson laboratories. Generation of Ror2 mutant mice has been described previously (Takeuchi et al., 2000). The genotype of these mice were determined by PCR-based methods as described previously (DasGupta and Fuchs, 1999; Yamaguchi et al., 1999; Takeuchi et al., 2000). Actinβ-Egfp mice were identified by Egfp expression under UV light.
Histology, in situ hybridization, BrdU labeling and TUNEL assays
Mouse embryos were dissected in ice-cold PBS and fixed in 4% paraformaldehyde (PFA)/PBS overnight at 4°C before being dehydrated through graded ethanol and embedded in paraffin. Samples were sectioned at 10 µm for histological analysis and section in situ hybridization. Detection of cell proliferation was performed with the bromodeoxyuridine (BrdU) labeling and Detection Kit (Roche Diagnostics Corporation, Indianapolis), and the TUNEL assay was performed to detect cell apoptosis, as described previously (Zhang et al., 2002; Alappat et al., 2005). For in situ hybridization experiments, at least three samples were used for each gene. For BrdU labeling, nine sections from three samples were counted and applied for statistical calculation. BrdU-labeled cells were counted and presented as percentage of total nuclei within arbitrarily defined areas, and Student’s t-test was used to determine significance of differences.
In vitro organ culture, tissue grafting, DiI cell labeling, and bead implantation
Secondary palatal shelves were microdissected from E12.5 or E13.5 embryos, orientated with dorsum upwards, and placed on filter in Trowell type organ cultures, as described previously (Zhang et al., 2002). For in vitro palate fusion assay, paired palatal shelves from E13.5 embryos were placed in organ culture as described previously (Taya et al., 1999). Samples were harvested after 3 days in culture and processed for histological analysis. For tissue grafting, donor palatal mesenchyme from E12.5 Actinβ-Egfp embryos were carefully dissected with fine forceps, minced into small tissue pieces and implanted into the same position in explanted host palatal shelves from aged-matched embryos. Explants were cultured in α-MEM medium plus 10% FBS, monitored for cell migration under a fluorescence microscope at 24-hour intervals for 72 hours. For DiI labeling experiment, the lipophilic dye DiI (1,1-dioctadecyl-3,3,39,39-tetramethyl-indocarbocyanine perchlorate; Molecular Probes) was initially prepared at a concentration of 0.5% in 100% ethanol and then diluted 1:9 with 0.3 M sucrose containing 0.1% Nile Blue sulfate. The DiI solution was injected into explanted palatal shelves using a micropipette (Li and Muneoka, 1999). Protein-soaked beads were implanted to explanted palatal shelves, about 250–300 µm away from the DiI injection site. Migration of DiI-labeled cells was monitored in a similar way for monitoring Egfp-expressing cell migration. Bead preparation and implantation were performed as reported before (Chen et al., 1996; Zhang et al., 2002). Affi-Gel blue agarose beads (100–200 µm in diameter, from Bio-RAD, Hercules, CA) were used as carriers for Shh, Wnt5a and Bmp4, and heparin beads (Sigma, St Louis, MO) were used as carriers for Fgf10. All the proteins were purchased from R&D Systems (Minneapolis, MN).
RESULTS
Expression of Wnt5a and its receptors in the developing palate
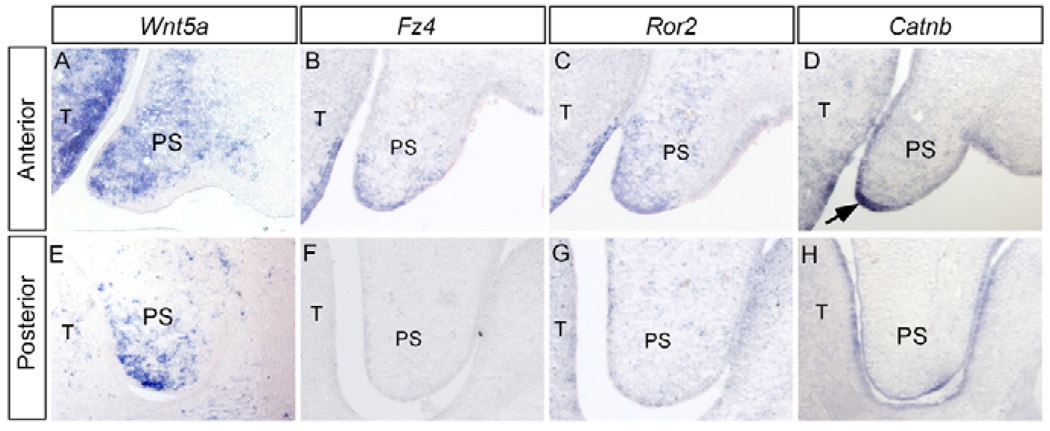
The developing secondary palate displays tissue heterogeneity as well as differential gene expression along the AP axis (Hilliard et al., 2005). In an effort to identify genes that exhibit such an expression pattern in the palate, we compared the gene expression profiles from the anterior (anterior to the first molar) versus posterior (posterior to the first molar) region of the E13.5 palatal shelves by Affimetrix chip microarray assays. Several genes that encode components of Wnt signaling pathway, including Wnt5a, frizzled 4 (Fzd4), Ror2 and β-catenin (Catnb), showed differential expression levels between the anterior and posterior palate, which was subsequently confirmed by in situ hybridization assays. Wnt5a expression was detected in the palatal mesenchyme, in a graded fashion along the AP axis (higher levels in the anterior region) (Fig. 1A,E). A gradient expression for Wnt5a along the nasal-oral axis was also observed (Fig. 1A,E). Fzd4 and Ror2 were also expressed, in a similar AP gradient fashion, in the epithelium and mesenchyme of the palate, with a restricted mesenchymal domain adjacent to the medial edge epithelium (MEE) (Fig. 1B,C,F,G). A graded expression of β-catenin, mainly in the MEE, was also detected along the AP axis of the palate (Fig. 1D,H).
Figure 1. Differential gene expression along the AP axis of E13.5 palatal shelf.
(A,E) Gradient Wnt5a expression is detected in palatal mesenchyme, higher in the anterior (A), and lower in the posterior (E) domain. (B,F) Fz4 expression is detected in the anterior palate (B), but not in the posterior region (F). (C,G) Ror2 exhibits a gradient expression, with higher level in the anterior palate (C) and lower in the posterior region (G). (D,H) Gradient Catnb expression is detected in the developing palate, higher in the anterior (D), while lower in the posterior (H) palate. There is strong Catnb expression in the MEE (arrow) of the anterior palate. T, tongue; PS, palatal shelf.
Wnt5a mutant mice exhibit complete cleft secondary palate
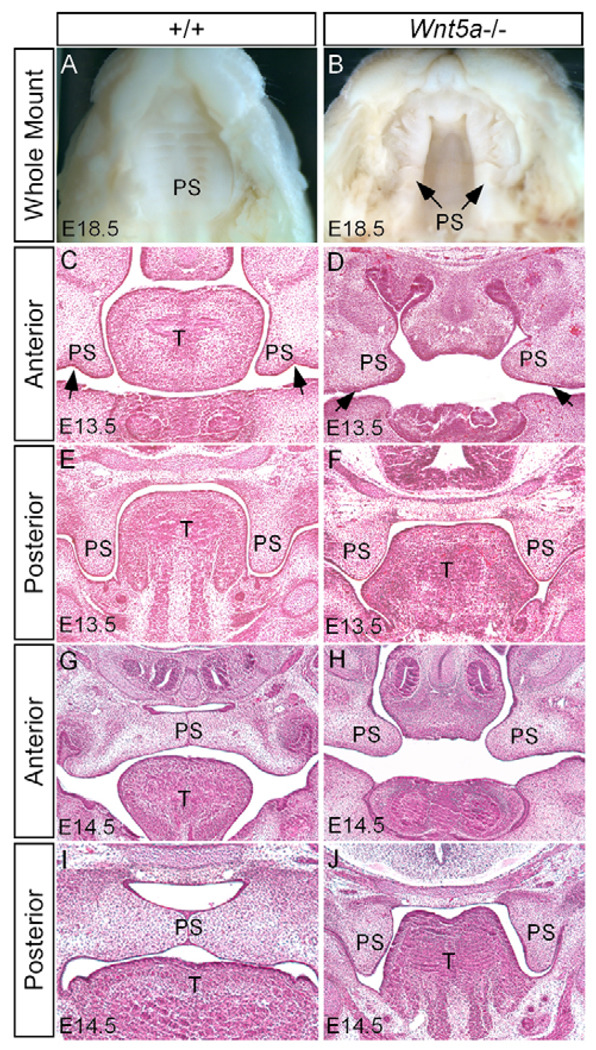
The expression of Wnt5a and its receptors Fzd4 and Ror2 suggests a role for these genes in palatogenesis. The fact that Ror2 knockout mice exhibit a cleft palate phenotype (Schwabe et al., 2004) and that individuals with non-syndromic cleft lip and palate have been linked to chromosomal translocation containing WNT5A locus (Blanton et al., 2004) promoted us to examine a cleft palate phenotype in Wnt5a−/− mice. Gross morphological and histological examinations revealed a complete cleft palate phenotype in Wnt5a−/− mice (Fig. 2A,B). In the mutants, the deformed palatal shelves were distinguishable as early as E12.5 but became pronounced by E13.5. At E13.5, when the palatal shelves in the wild type take the vertical position on both sides of the tongue, the anterior region of the mutant palatal shelves remained in the horizontal position (Fig. 2C,D). By contrast, the posterior portion of the mutant palatal shelves grew in a vertical growth position (Fig. 2E,F). Morphologically, the mutant anterior palatal shelf appeared round, shortened and blunt-ended, lacking the ventrolateral indention of the shelf (Fig. 2D), whereas the posterior shelf was shortened, broadened and wedge-shaped (Fig. 2F). At E14.5, the wild-type palatal shelves have elevated to above the tongue and fused at the midline (Fig. 2G,I). In Wnt5a mutant, the anterior region of the palatal shelves remained in horizontal level but was too short to make a contact (Fig. 2H). However, the posterior palatal shelves failed to elevate (Fig. 2J). The lack of contact between the palatal shelf and tongue/mandible suggests an intrinsic defect in elevation. The gradient expression of Wnt5a along the AP axis of the developing palate appears to account for the distinct morphological defects found in the anterior and posterior palate of the mutant. In addition, Wnt5a mutants also displayed a short and small tongue, and a deformed oral cavity, which may contribute, to some extent, to the cleft palate defect.
Figure 2. Cleft palate phenotype in Wnt5a mutant.
(A) An E18.5 wild-type mouse shows an intact palate. (B) An E18.5 Wnt5a mutant shows wide open cleft secondary palate. (C,E) Coronal sections of an E13.5 control embryo show the normal palatal shelves at the anterior (C) and posterior domain (E). Arrows in C indicate the ventrolateral indention. (D) A coronal section of an E13.5 Wnt5a mutant shows the palatal shelves in the horizontal level lacking the ventrolateral indention (arrows). (F) The posterior palatal shelves of an E13.5 mutant exhibit an abnormal shape. (G,I) An E14.5 control embryo shows closure of the palate at the anterior (G) and posterior region (I). (H) The anterior palatal shelves of an E14.5 Wnt5a mutant remain at the horizontal level but appear too short to make contact at the midline. (J) The posterior palate of an E14.5 mutant fails to elevate, remaining at the vertical position. T, tongue; PS, palatal shelf.
Wnt5a mutant exhibits an impaired palatal growth
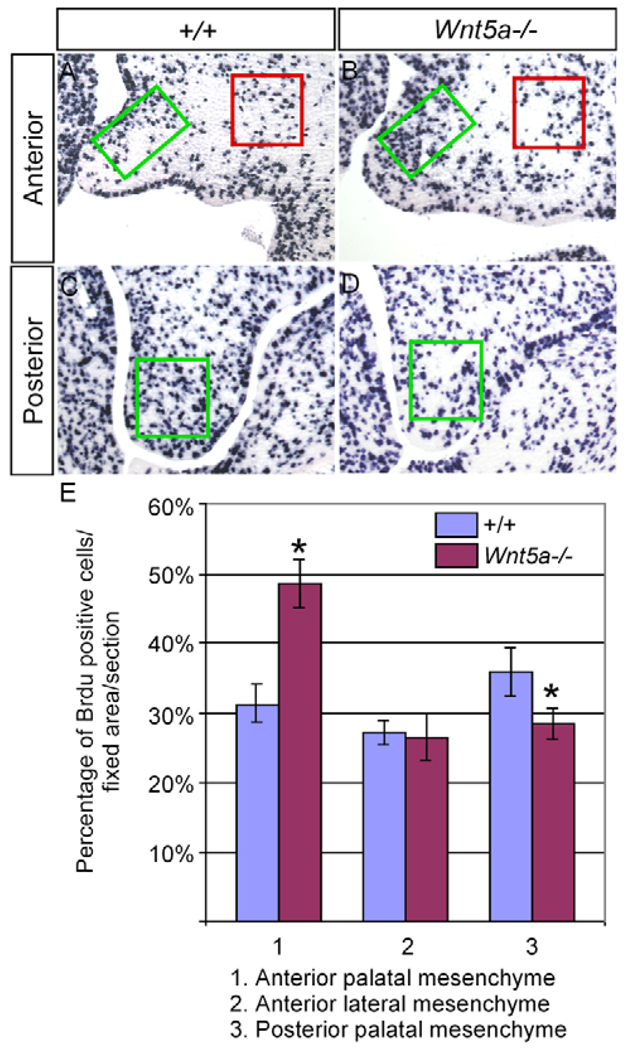
To reveal the cellular mechanisms responsible for the palate deformation in Wnt5a−/− mice, we carried out cell proliferation and apoptosis assays as well as in vitro palate fusion assay. We determined cell proliferation rate in the wild type and Wnt5a−/− palates at E13.5 by BrdU labeling. As controls, BrdU-positive cells were also counted in the maxillary mesenchyme lateral to the palate where Wnt5a is not expressed. An altered cell proliferation rate was found in the mutant palatal mesenchyme (Fig. 3). Intriguingly, a significantly elevated cell proliferation level was seen in the anterior palate, whereas a decreased rate occurred in the posterior palate. This differential alteration of cell proliferation rate in the Wnt5a−/− palate along the AP axis implicates dual roles for Wnt5a in the regulation of cell proliferation. It is interesting to note that, in the anterior palate of the mutant, the mesenchymal cells on the future nasal side were much more mitotic than cells on the future oral side. This dysregulated cell proliferation level in the future nasal aspect apparently accounts for, at least partially, the deformed shape of the mutant palatal shelves and a failure in change of palate growth direction from the horizontal to vertical position. However, TUNEL assays revealed a comparable level of cell apoptosis in the wild-type and Wnt5a−/− palatal shelves at E13.5 (data not shown). To determine whether the formation of a cleft palate in Wnt5a mutants could also be attributed to a failure in palate fusion, we employed an in vitro organ culture system to examine palate fusion. The results demonstrated that pairs of palatal shelves from E13.5 Wnt5a−/− embryos, when placed in contact with the MEE and facing one another, were able to fuse, in a way identically to the wild-type controls (data not shown). The dysregulated cell proliferation in the palatal mesenchyme therefore represents a major cellular process that causes a cleft palate formation in Wnt5a mutants.
Figure 3. BrdU labeling of cells in the developing palate.
(A–D) Coronal sections through the anterior (A,B) and posterior (C,D) palates of an E13.5 wild-type control (A,C) and an E13.5 Wnt5a mutant (B,D) showing BrdU-labeled cells. Boxes in each panel indicate the area where the labeled cells were counted and compared. (E) Comparison of percentage of BrdU-labeled cells in the fixed areas of the palate in the wild-type control and mutant embryos. Standard deviation values are indicated as the error bars. *P<0.05.
Altered gene expression in the Wnt5a−/− palate
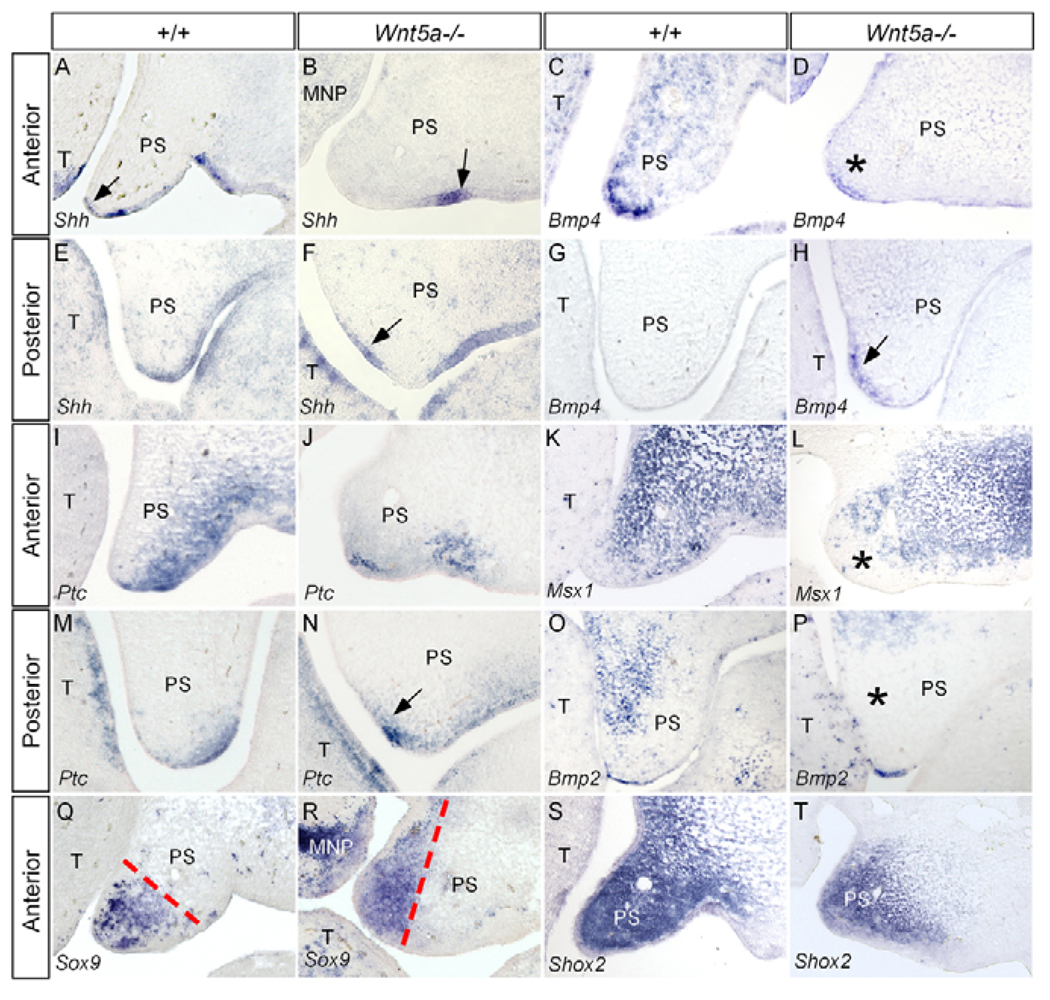
We next examined the expression of a number of genes in the palatal shelves of Wnt5a mutants. They are either potential Wnt5a downstream genes or genes that exhibit an overlapped expression domain in the palate with Wnt5a, including Shh, Ptch1, Bmp4, Bmp2, Msx1, Fgf10, Fgfr2, Shox2 and Sox9. In E13.5 Wnt5a mutants, Bmp4 was downregulated in the anterior palate (Fig. 4C,D), but displayed an ectopic expression domain in the posterior palatal mesenchyme on the nasal side, where Bmp4 is not normally expressed (Fig. 4G,H). Consistent with the fact that mesenchymally expressed Bmp4 positively regulates Shh in the palatal epithelium (Zhang et al., 2002), Shh in the MEE of the anterior palate was downregulated (Fig. 4A,B), whereas an ectopic Shh expression was observed in the nasal side epithelium of the posterior palate, immediately adjacent to the ectopically expressed Bmp4 domain in the palatal mesenchyme (Fig. 4F,H). Ptch1, a downstream target and receptor of Shh, displayed altered expression pattern accordingly along the AP axis (Fig. 4I,J,M,N). However, as the mutant palatal shelves are so malformed, the exact position where Shh is ectopically activated in the palate remains to be determined. Nevertheless, these observations place Wnt5a upstream of Shh and Bmp4, consistent with the previous report in the developing lung (Li et al., 2002; 2005). Msx1, a downstream target of Bmp4, was partially downregulated in the anterior palatal mesenchyme of the Wnt5a−/− palate (Fig. 4K, Fig. 5L). Although the expression of Bmp2, a downstream target of Shh that mediates the mitotic effect of Shh in the anterior palate (Zhang et al., 2002), remained unchanged in the anterior palate of the mutant (data not shown), a significant downregulation of Bmp2 was observed in the posterior palatal mesenchyme (Fig. 4O,P). It appears that other upstream factors besides Shh are also involved in regulating Bmp2 expression. Wnt5a and palatogenesis By contrast, Fgf10 and its receptor Fgfr2 exhibited comparable expression level and pattern in both wild-type and mutant palate (data not shown). These results demonstrate that Wnt5a acts differentially, in term of gene expression regulation, in the anterior and posterior palate.
Figure 4. Gene expression in E13.5 wild-type and Wnt5a−/− palate.
(A) The anterior palate of a wild-type embryo shows Shh expression in the MEE (arrow) and rugal epithelium. (B) Shh expression becomes undetectable in the MEE but remains in the rugal epithelium (arrow) in the anterior palate of a Wnt5a mutant. (C,D) Bmp4 expression is detected in the anterior palate of a wild-type control (C), but is significantly reduced in the mutant (asterisk, D). (E,F) In the posterior palate of the control, Shh expression is detected in the rugal epithelium at the ventral (oral side) (E), whereas in the mutant, an ectopic Shh expression is found in the dorsal (nasal) epithelium (arrow) of the palate (F). (G,H) Bmp4 expression is absent in the posterior palate of the control (G), but is found ectopically in the mesenchyme of the posterior palate on the future nasal side (arrow) in the mutant (H). (I) Ptch1 expression is seen in the anterior palate of the control, mainly in the ventral (oral) side. (J) A downregulated Ptch1 is seen in the anterior palate of a Wnt5a mutant. (K,L) Msx1 expression is detected in the anterior palatal mesenchyme of a wild-type control (K), but is downregulated in the palatal mesenchyme (asterisk) adjacent to the MEE at the similar level of a mutant palate (L). (M,N) Ectopic Ptch1 expression is found in the dorsal (nasal) aspect (arrow) of the palate in the posterior region in a Wnt5a mutant (N), when compared with its normal expression in the control (M). (O,P) A downregulation of Bmp2 expression is found in the posterior palate of Wnt5a mutant (P), when compared with its normal expression pattern in the control (O). (Q) Sox9 expression in the anterior palatal mesenchyme of a control embryo. (R) A shifted Sox9 expression domain is seen in the anterior palate of a Wnt5a mutant. Broken lines in Q,R indicate the expression boundary. (S,T) Shox2 expression remains in the anterior palate of the mutant (T), when compared with that in the control (S). T, tongue; PS, palatal shelf; MNP, medial nasal process.
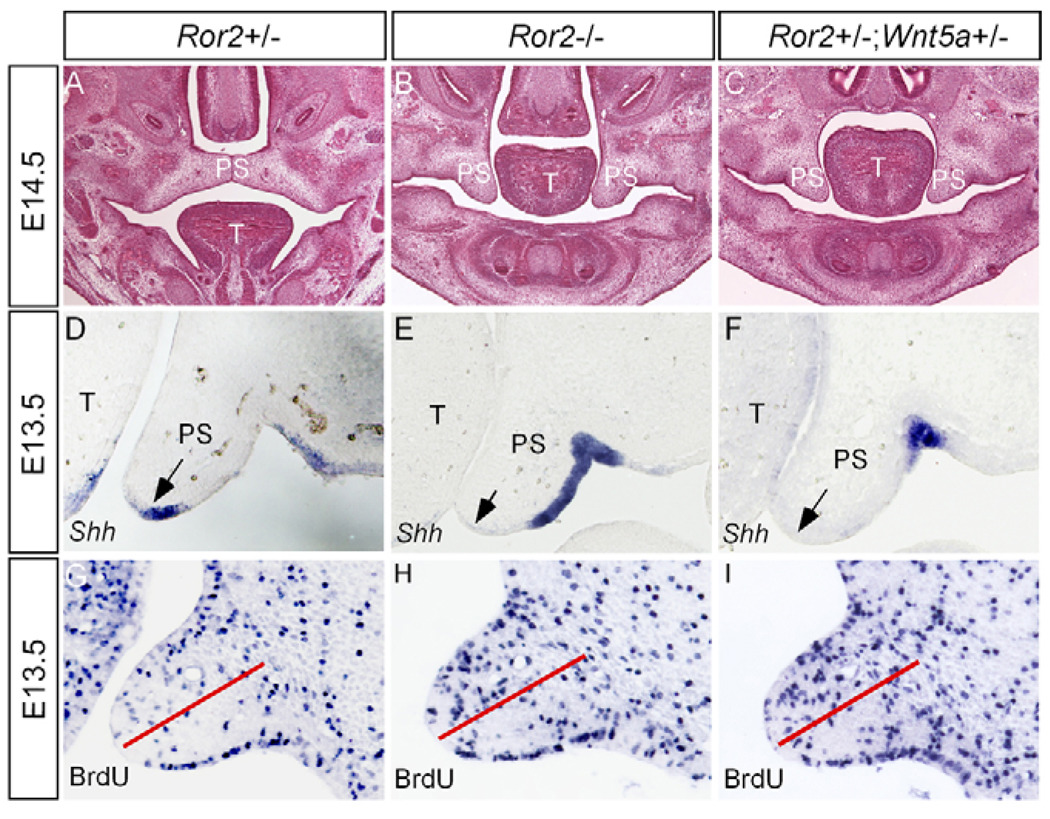
Figure 5. Wnt5a and Ror2 have an epistatic effect on palate development.
(A) An E14.5 wild-type embryo shows the closure of the palate. (B) An E14.5 Ror2−/− embryo shows a failed palatal elevation. (C) Failed palatal elevation is observed in an E14.5 Ror2+/−;Wnt5a+/− embryo. (D–F) Sections through the anterior palate of E13.5 wild-type control (D), Ror2 mutant (E) and Ror2+/−;Wnt5a+/− embryo (F) show Shh expression in the MEE of the control palate, but a lack of Shh expression in the MEE of the Ror2−/− and Ror2+/−;Wnt5a+/− palate. Arrows indicate the MEE. (G) The anterior palate of an E13.5 control embryo shows BrdU-labeled cells. (H,I) The level of cell proliferation is similarly increased in the anterior palate of Ror2−/− (H) and Ror2+/−;Wnt5a+/− (I) embryos. The red lines in G–I separate the dorsal and ventral halves of the palate. T, tongue; PS, palatal shelf.
Wnt5a and Ror2 exert an epistatic effect on palate development
As cell proliferation was increased in the anterior palatal mesenchyme lacking Wnt5a and as the canonical Wnt signaling is known to control proliferation, it is possible that Wnt5a normally antagonizes the canonical pathway in the developing palate. To determine this possibility, we examined the expression of TOPGAL, a transgenic reporter of activation of the canonical Wnt signaling pathway, in the wild type and Wnt5a−/− background (DasGupta and Fuchs, 1999). We failed to detect TOPGAL expression in the developing palate of both wild type and Wnt5a mutants (data not shown), suggesting that the canonical Wnt signaling pathway may not be involved in palate development. The fact that mice lacking Fz4, the receptor that is expressed in the developing palate and mediates the canonical signaling of Wnt5a, survive to adulthood without a defective palate (Hsieh et al., 2005), implies an involvement of noncanonical pathway. The fact that both Ror2−/− and Wnt5a−/− mice exhibit a cleft palate phenotype strongly implicates an overlapping role for these two molecules. We therefore examined the cleft palate defect in Ror2−/− embryos. Similar to Wnt5a mutant, we found a downregulated Shh in the MEE and an enhanced cell proliferation rate in the anterior palatal mesenchyme of Ror2−/− embryos (Fig. 5). We subsequently tested a genetic interaction between Wnt5a and Ror2 by compounding Wnt5a+/− and Ror2+/− to generate double heterozygous mice. Wnt5a+/−;Ror2+/− mice indeed exhibited cleft palate defects that phenocopy that of Ror2−/− mutants, including downregulated Shh expression in the MEE and upregulated cell proliferation rate in the mesenchyme of the anterior palate, particularly in the nasal aspect (Fig. 5). These results demonstrate an epistatic effect of these two genes on palate development. However, the posterior palatal shelves of Ror2−/− and Wnt5a+/−;Ror2+/− mice did not show an altered cell proliferation rate, consistent with an extremely low level of Ror2 expression in this region. As the palatal phenotypes in Ror2−/− and Ror2+/−;Wnt5a+/− mice appear milder when compared with that of Wnt5a mutants, Ror2 is not the only receptor mediating the function of Wnt5a during palatogenesis.
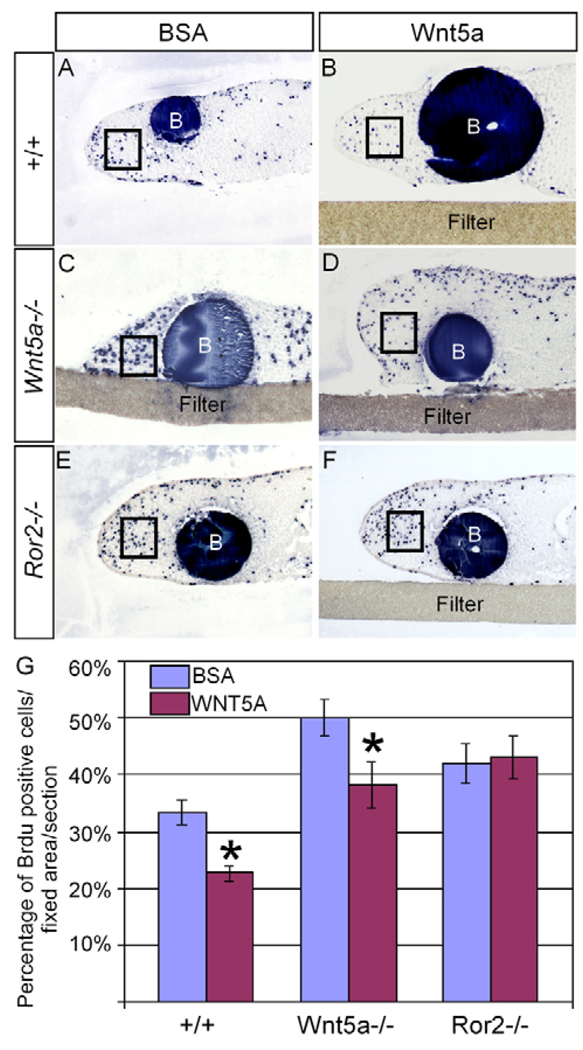
We further confirm a role for Ror2 in mediating Wnt5a signaling in the regulation of cell proliferation by bead implantation experiments. Beads soaked with BSA (1 mg/ml) or Wnt5a (0.5 mg/ml) were implanted, respectively, onto explanted anterior palatal tissues from E13.5 wild-type, Wnt5a−/− and Ror2−/− embryos. Samples were cultured for 24 hours and pulsed with BrdU prior to fixation. Consistent with the results by in vivo BrdU labeling assays, elevated cell proliferation levels were observed in the Wnt5a−/− and Ror2−/− palatal tissues implanted with BSA beads, when compared with that in the wild-type controls (Fig. 6A,C,E). A significantly decreased rate of cell proliferation was seen in the wild-type palatal tissues implanted with Wnt5a beads (Fig. 6B, 6G), indicating that overdosed exogenous Wnt5a protein represses cell proliferation. Furthermore, a dramatically reduced level of cell proliferation was found in the Wnt5a−/− palate implanted with Wnt5a bead (Fig. 6B,D,G), demonstrating a rescue of the altered cell proliferation phenotype in the mutants. By contrast, Wnt5a beads failed to rescue cell proliferation defect in the Ror2−/− palatal tissues (Fig. 6F,G).
Figure 6. Regulation of cell proliferation in palatal mesenchyme by Wnt5a is mediated by Ror2.
(A) An explanted wild-type palatal shelf implanted with BSA bead shows BrdU-labeled cells in the distal (the MEE side) mesenchyme. (B) An explanted wild-type palatal shelf implanted with a Wnt5a-soaked protein shows a reduced rate of cell proliferation in the distal mesenchyme. (C) A higher cell proliferation rate is seen in an explanted Wnt5a−/− palatal shelf implanted with a BSA bead. (D) Cell proliferation rate is rescued to about the normal level in an explanted Wnt5a−/− palate by a Wnt5a-soaked bead. (E,F) Elevated cell proliferation rate is seen in an explanted Ror2−/− palatal shelf implanted with a BSA bead (E), and remains in an explanted Ror2−/− palatal shelf implanted with a Wnt5a bead (F). (G) Comparison of percentage of BrdU-labeled cells in the fixed area (box) of the palate of different genotypes implanted with BSA- or Wnt5a-soaked beads. Standard deviation values were indicated as the error bars. B, bead. In all images, the MEE is towards the left. *P<0.01.
Wnt5a-deficiency leads to altered cell migration in the palatal mesenchyme
In the assays for gene expression, we made an interesting observation about Sox9 expression, a HMG box gene whose mutation also leads to cleft palate formation (Bi et al., 1999; Mori-Akiyama et al., 2003). Sox9 is expressed and regulated by Shox2 in the anterior palatal mesenchyme (Fig. 4Q) (Yu et al., 2005; Gu et al., 2008). Consistent with an unaltered Shox2 expression (Fig. 4S,T), Sox9 expression was maintained in the anterior palatal mesenchyme of Wnt5a mutant (Fig. 4R). However, a close examination identified a shifted expression domain of Sox9. In the controls, Sox9 expression was restricted to the distal region (the MEE side) of the anterior palate (Fig. 4Q). In the mutant, however, Sox9 expression domain was shifted to the future nasal side (Fig. 4R), which could reflect a positional change of the Sox9-expressing cells in the palate. Based on this assumption, we Wnt5a and palatogenesis reasoned that a constant cell migration exists in the developing palatal mesenchyme. Aberrant cell migration could lead to a change in position of the mesenchymal cells within the Wnt5a−/− palate.
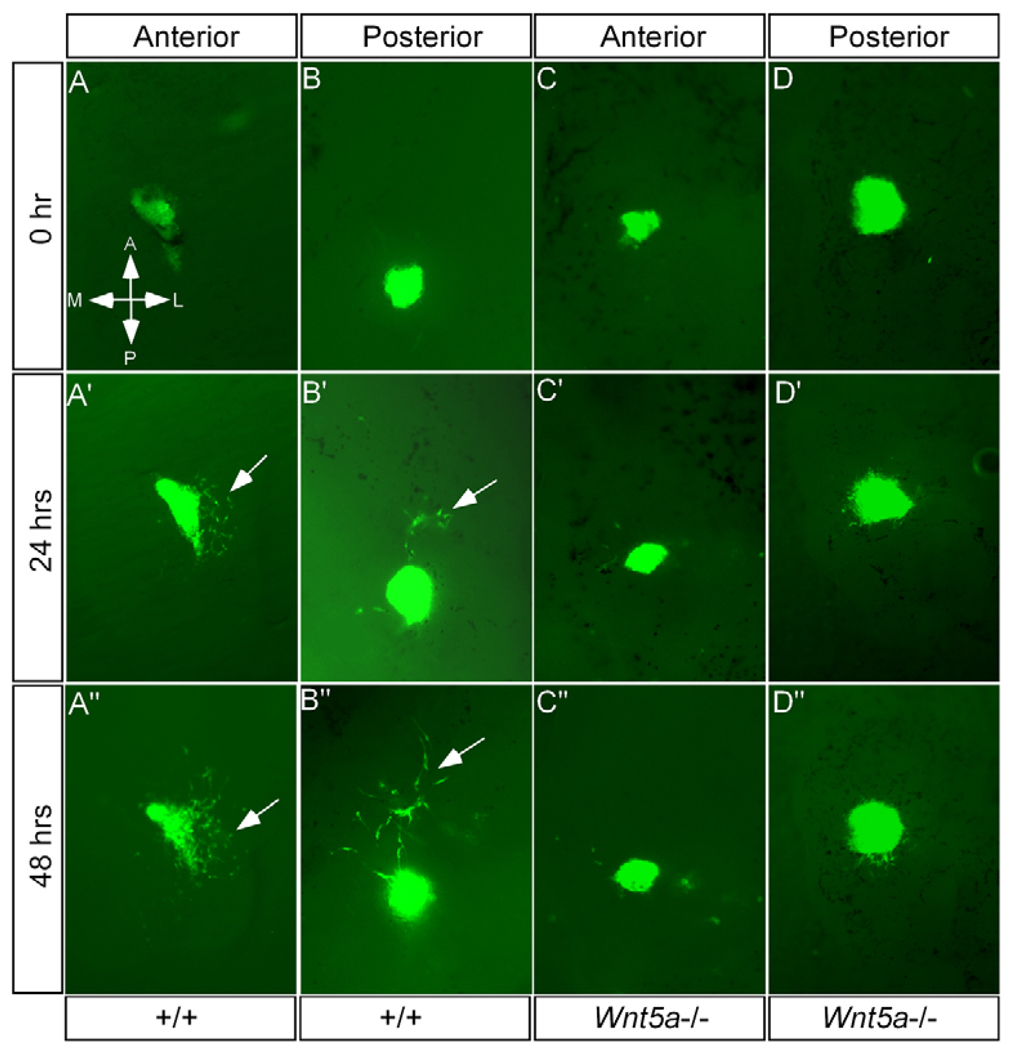
To test these hypotheses, we first determined if constant cell migration does exist in the developing palate. E12.5 palatal shelves were isolated and placed with the AP axis marked in organ culture. The anterior and posterior palatal mesenchymal tissues from E12.5 Actinβ-Egfp mice were grafted into the explanted palatal shelves in their original positions, respectively. Cell migration was subsequently monitored at 24-hour intervals. As shown in Fig. 7, the Egfp-expressing cells underwent an active cell migration within the host tissue environment. Surprisingly, the cell migration was not random but directional, indicating the existence of a chemoattractant gradient. When the Egfp-expressing posterior palatal mesenchyme was grafted into the posterior region of the host explants, they displayed a directional migration towards the anterior side in 77% of grafts (Fig. 7B; Table 1). This posterior to anterior cell migration pattern coincides with the gradient Wnt5a expression along the AP axis. However, in the anterior palate, the grafted anterior palatal cells exhibited a directional migration towards the lateral side in the majority of the cases (54%) (Fig. 7A; Table 1). In about 13% of cases, directional cell migration towards the medial side was recorded. Nevertheless, these results demonstrate the existence of active directional cell movement in the developing palate.
Figure 7. Aberrant cell migration in the Wnt5a−/− palate.
(A-A″) Egfp-expressing mesenchymal cells from the anterior palate, when grafted to the anterior region of an explanted wild-type palatal shelf, exhibit a directional migration towards to the lateral side after 24 hours. Arrows indicate the migrating cells. (B-B″) Egfp-expressing mesenchymal cells from the posterior palate, when grafted to the posterior region of an explanted wild-type palatal shelf, show a directional migration towards the anterior end after 24 hours. (C-C″,D-D″) Directional migration by the Egfp-expressing cells is not observed when these cells are grafted to either the anterior (C-C″) or the posterior (D-D″) palatals shelves from Wnt5a mutants.
Table 1.
Mesenchymal cell migration direction in the developing palate after EGFP graft
| Directional migration (sample/total number) | Undirectional migration | |||||
|---|---|---|---|---|---|---|
| Host palate | Anterior | Posterior | Lateral | Medial | Random | No migration |
| Wild-type anterior | 2/70 | 38/70 | 9/70 | 13/70 | 8/70 | |
| Wild-type posterior | 23/30 | 2/30 | 1/30 | 4/30 | ||
| Wnt5a−/− anterior | 2/10 | 1/10 | 3/10 | 5/10 | ||
| Wnt5a−/− posterior | 2/12 | 4/12 | 6/12 | |||
We next asked whether the directional cell migration could become altered in the palatal mesenchyme lacking Wnt5a. In a similar assay using E12.5, Wnt5a−/− palatal shelves as hosts, we did not observe a constant directional cell migration in both the anterior and posterior palatal regions (Table 1). In most cases (more than 80%), the grafted cells either migrated slightly but randomly, or never moved (Fig. 7C,D), indicating a defective cell movement in the Wnt5a−/− palate.
Ror2 mediates the chemotactic activity of Wnt5a in the developing palate
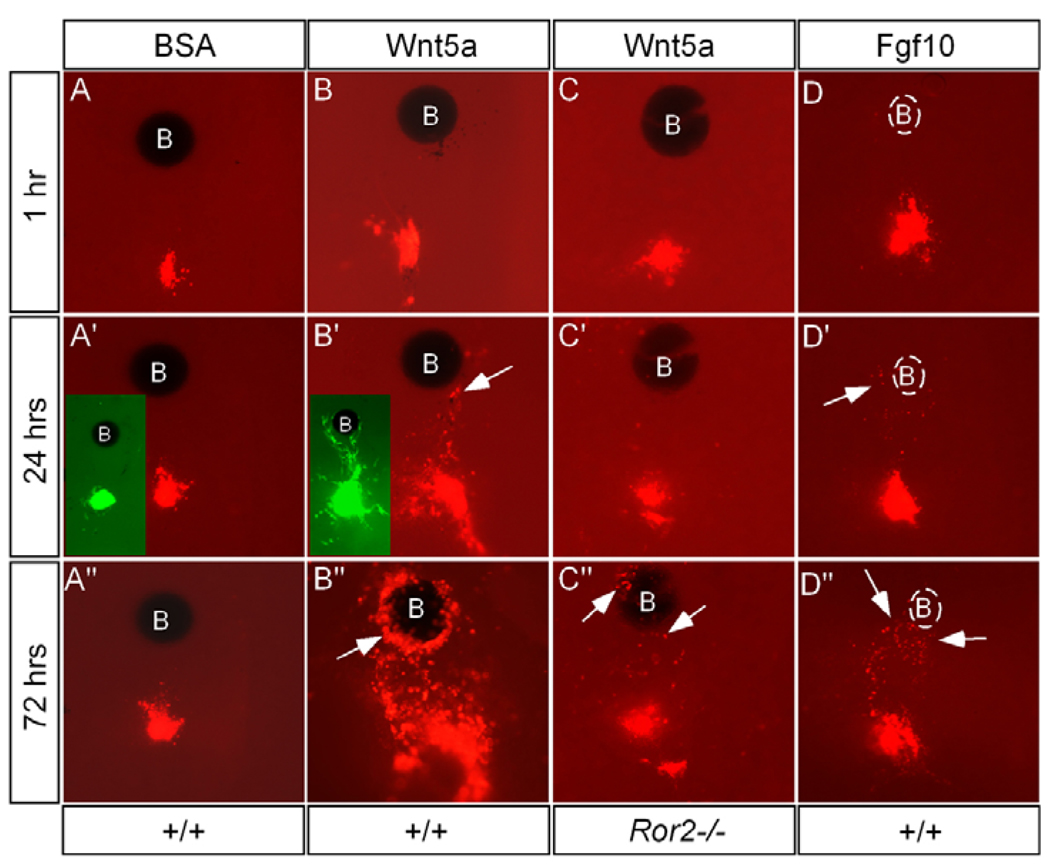
We tried to determine whether Wnt5a acts as a chemoattractant for mesenchymal cells of the palate. We coupled DiI-cell labeling/tissue grafting with bead implantation in explanted palatal shelves. As shown in Fig. 8, BSA-soaked beads (1 mg/ml) had no influence on migration direction of DiI-labeled cells or grafted Egfp-expressing cells (26/28). By contrast, DiI-labeled cells/grafted Egfp-expressing cells exhibited a directional migration towards Wnt5a-soaked beads (0.5 mg/ml) in 24 hours (Fig. 8B). This chemotactic effect of Wnt5a was seen in 69% of cases (25/36). We also found that cells responded to the chemotactic action of Wnt5a identically, regardless where the beads were implanted, implying that exogenous Wnt5a has overcome the endogenous gradient. To further test whether Wnt5a is essential and sufficient to drive AP cell movement in the palate, we applied Wnt5a beads onto the anterior side of E12.5 Wnt5a−/− palatal shelves in culture and monitored the movement of DiI-labeled cells in the posterior palate. A constant cell migration towards the anterior aspect (exogenous Wnt5a source) was observed (6/10) (data not shown).
Figure 8. Ror2 mediates chemotactic action of Wnt5a in the developing palate.
(A-A″) BSA bead fails to exert chemotactic effect on DiI-labeled mesenchymal cells and grafted Egfp-expressing cells (insert in A′). (B-B″) Wnt5a acts as a potent chemoattractant to attract DiI-labeled mesenchymal cells and grafted Egfp-expressing cells (inserts in B′, 48 hours in culture). Arrows indicate DiI-labeled cells attracted to Wnt5a bead. (C-C″) Few DiI-labeled cells (arrows) are attracted to Wnt5a bead in an explanted palatal shelf from Ror2−/− embryo. (D-D″) Fgf10 acts as a chemoattractant for palatal mesenchymal cells. Heparin beads were used for Fgf10 soaking, which is invisible under UV light, and are outlined by the broken line. B, bead.
It has previously been reported that Ror2 is required for Wnt5ainduced cell migration (Nishita et al., 2006; Yamamoto et al., 2007). To determine whether Ror2 also mediates the chemotactic activity of Wnt5a in the developing palate, we performed DiI labeling and bead implantation on the explanted palatal shelves from E12.5 Ror2−/− embryos. As expected, Wnt5a beads did not exhibit a chemotactic effect on the DiI-labeled cells (9/10). Occasionally, very weak chemotactic response was observed. A few labeled cells could be found around the Wnt5a bead after an extended culture period (Fig. 8C).
Several other growth factors are expressed in the anterior palate, including Bmp4, Fgf10 and Shh (Zhang et al., 2002; Rice et al., 2004; Alappat et al., 2005). We wondered whether any of these factors are also implicated in regulating cell migration. We found that Shh (0.5 mg/ml) and Bmp4 (0.4 mg/ml) did not act either as a chemoattractant or a chemorepellent in the palate (data not shown). By contrast, Fgf10, a known chemoattractant for several types of cells (Park et al., 1998; Ceccarelli et al., 2007; Natanson-Yaron et al., 2007), exhibited a chemotactic potency for the palatal mesenchymal cells (Fig. 8D), which was observed in 63% of total cases (10/16).
DISCUSSION
Cell movement is an essential process during embryogenesis and the formation of many organs. In the developing limbs, cell migration is crucial for pattern formation and outgrowth. Abnormal cell migration or change of migration direction could cause dramatic changes in limb bud shape and outgrowth inhibition (Li and Muneoka, 1999). During palate development, palatal shelves undergo rapid outgrowth and significant changes in palate shape and position. Cell proliferation and extracellular matrix production and deposition within the palatal mesenchyme contribute to these processes significantly (Ferguson, 1988; Gritli-Linde, 2007). The morphological movement of the palatal shelf has been considered an active process of shelf elevation. However, an active directional cell migration by the mesenchymal cells during palate development has not been reported. In this paper, we demonstrate the existence of two streams of active directional cell migration: in the posterior palate, cells migrate towards to anterior aspect, whereas in the anterior palate, cells move towards the lateral aspect. Though its biological significance in palate formation is yet to be elucidated, the directional cell migration should have a crucial role in palate patterning (shaping) and elevation. The Wnt5a−/− deformed palate could be partially attributed to the aberrant cell migration. During palate shelf elevation, different means (rotation versus remodeling) are thought to be used in different regions of the palate along the AP axis (Okano et al., 2006). The distinct directions of cell migration in the anterior and posterior palate may represent a cellular process underlying these two different elevation mechanisms. Computer modeling studies will be required to model cell migration directionality associated with palate elevation.
Wnt5a has been implicated in cell migration in developing vertebrate embryos and in invasive cancer cells (Weeraratna et al., 2002; Jopling and den Hertog, 2005; Kim et al., 2005; Witze et al., 2008). Here, we show a defective cell migration in the Wnt5a−/− palate. Our results prove that Wnt5a is a potent and specific chemoattractant for the palatal mesenchymal cells. The gradient of Wnt5a expression along the AP axis therefore sets a chemotactic base for the posterior-to-anterior migration of the mesenchymal cells in the palate. Such graded expression was also observed for Wnt5a in those organs and tissues that undergo outgrowth, including the limb and tailbud (Yamaguchi et al., 1999). Consistent with the graded fashion of Wnt5a expression in the distal limb mesenchyme, active proximal-distal cell migration has been reported in developing chick limb buds (Li and Muneoka, 1999). Although defective cell migration has not yet been reported in the limbs of mice lacking Wnt5a, a reduced Wnt5a dose indeed impairs cell migration in fish fins, the homolog of the tetrapod limbs (Sakahuchi et al., 2006). In addition, impaired cell migration has been reported in the developing pancreas of Wnt5a−/− embryo (Kim et al., 2005). In the developing palate, Ror2 appears to be essential for the chemotactic action of Wnt5a, as cells lacking Ror2 failed to respond to Wnt5a. It has been shown that Ror2 is required for Wnt5a-induced migration of cultured cells by mediating filopodia formation (Nishita et al., 2006). By contrast, it has recently been shown that Wnt5a can also act as a chemorepellent for neuronal cells (Keeble et al., 2006). This chemorepulsive action of Wnt5a is mediated by Ryk. Wnt5a thus has dual roles in the regulation of cell migration, depending on the presence of specific receptor. As Ryk is also expressed in the developing palate in a punctuate pattern (Halford et al., 2000), the Ryk-expressing cells could move in a direction away from the source of Wnt5a. This potential chemorepulsive action of Wnt5a may account partially for the lateral directionality of mesenchymal cells migration in the anterior palate, which warrants future investigation.
Many growth factors and receptors are expressed in a specific temporal and spatial manner in the developing palate (Gritli-Linde, 2007). Of them, some are proven chemoattractants. We demonstrate in this study that, besides Wnt5a, Fgf10 is also a potent chemoattractant for mesenchymal cells of the palate. Fgf10 is expressed in the lateral region of the anterior palate at E12.5 and E13.5 (Alappat et al., 2005), which could at least partially account for the lateral migration of mesenchymal cells. As the primary Fgf10 receptor Fgfr2b is only expressed in the palatal epithelium (Rice et al., 2004), this chemotactic action of Fgf10 is probably mediated by other Fgf receptor(s) in the mesenchyme. This idea is supported by the fact that exogenous Fgf10 inhibits cell proliferation in the anterior palatal mesenchyme (Yu et al., 2005). As multiple chemoattractants and chemorepellents are likely to be present in the developing palate, the directionality of cell migration is probably controlled by a combinatorial effect of multiple factors.
The gradient expression of Wnt5a and its receptors Fz4 and Ror2 correlates with the tissue heterogeneity along the AP axis of the developing palate. We observed in the Wnt5a−/− palate altered expression of several genes crucial for palate development. However, a closer examination of gene expression reveals distinct or even opposite responses of the anterior and posterior palatal tissues to the absence of Wnt5a. For example, Shh and Bmp4 were downregulated in the anterior palate, but were ectopically activated in the posterior region. In addition, opposite cellular responses (cell proliferation) were found in the anterior and posterior palatal mesenchyme lacking Wnt5a, with an increased level of cell proliferation in the anterior but a decreased level in the posterior palate. Shh has been shown to positively regulate cell proliferation in the palatal mesenchyme (Zhang et al., 2002). However, the altered Shh expression patterns do not correlate with the changes in cell proliferation rate in the Wnt5a−/− palate. One would expect a decreased cell proliferation rate in the anterior region of the Wnt5a−/− palate where Shh is downregulated. However, a hyperproliferation of mesenchymal cells was instead observed, suggesting an involvement of multiple signaling pathways in cell proliferation regulation in the anterior palate where Wnt5a-mediated signaling acts as a negative regulator. By contrast, the proliferation rate in the posterior palate of Wnt5a mutant is significantly decreased when compared with the controls, revealing a positive role for Wnt5a in cell proliferation regulating in the posterior palate. As the number of migrating mesenchymal cells in the palate appears small, the altered expression of these crucial morphogenetic genes and the aberrant cell proliferation appear to have major impact on cleft palate formation in Wnt5a mutants.
Wnt5a has been previously shown to exert distinct effects on cell proliferation in different developing organs. Inactivation of Wnt5a results in reduced proliferation rate of the progenitor cells in the developing limbs, but leads to an increased level of cell proliferation during distal lung morphogenesis and mammary tissues (Yamaguchi et al., 1999; Li et al., 2002; Li et al., 2005; Roarty and Serra, 2007). Wnt5a has also been shown to inhibit B cell proliferation and functions as a tumor suppressor in hematopoietic tissue (Liang et al., 2003). However, in the developing palate, Wnt5a appears to exert opposite roles in the regulation of gene expression and cell proliferation in different regions. The differential expression of Wnt5a receptors (such as Ror2 and Fz4) in the palatal shelf along the AP axis could account for the distinct functions of Wnt5a. It is possible that other unidentified Wnt5a receptors are expressed and involved in mediating Wnt5a signaling in the palate. However, several signaling pathways initiated by other growth factors, including BMP, FGF and TGFβ, have been proven to differentially operate and exert their distinct effects on cell proliferation regulation in different domains of the developing palate along the AP axis. A combinatorial effect of, and a finely tuned balance between, these signaling pathways appear essential for the precise control of normal palate growth. The absence of Wnt5a could disrupt the closely linked signaling network, leading to dysregulated cell proliferation rate.
Wnt5a has dual roles in signaling: activating either the Wnt canonical or noncanonical signaling pathways. At least two known Wnt5a receptors, Fz4, which mediates the activation of the canonical pathway by Wnt5a, and Ror2, which mediates the noncanonical signaling, are expressed in an overlapping pattern with Wnt5a in the developing palate. However, activities of the canonical Wnt signaling were not detected in the developing palate in wild-type and Wnt5a−/− mice, excluding an involvement of the canonical Wnt signaling in palate development. However, we cannot rule out the possibility that the TOPGAL transgenic allele is not sensitive enough to detect a low signaling level. Wnt5a appears to regulate palatogenesis via a noncanonical Wnt pathway that does not antagonize the canonical signaling. The fact that Fz4-null mice do not exhibit a cleft palate defect and that the inactivation of both Lef1 and Tcf4, the essential nuclear mediators of the canonical Wnt signaling, does not cause an abnormal palate formation, further supports this conclusion (Hsieh et al., 2005; Brugmann et al., 2007). Several lines of evidence support the conclusion that Ror2 mediates the function of Wnt5a in regulating palatogenesis: (1) Wnt5a and Ror2 are co-expressed in the anterior palatal mesenchyme; (2) Wnt5a and Ror2 exert an epistatic effect in palate development; (3) the similar cleft palate phenotype, including elevated cell proliferation rate and altered expression pattern of downstream genes, are seen in both Wnt5a and Ror2 mutants; (4) exogenous Wnt5a protein fails to rescue (reduce) cell proliferation rate in Ror2−/− palatal mesenchyme; (5) exogenous Wnt5a protein fails to exert chemotactic effect on the palatal cells lacking Ror2. In fact, Ror2-mediated Wnt5a signaling can active the Wnt-JNK pathway and/or inhibit Wnt canonical signaling cascade (Oishi et al., 2003; Mikels and Nusse, 2006; Schambony and Wedlich, 2007). Although the activity of Wnt/β-catenin canonical signaling is not detected or elevated in Wnt5a−/− palate, it still remains unknown whether Wnt5a/Ror2 acts through the JNK pathway to regulate palatogenesis.
Acknowledgments
The authors thank Drs Manjong Han and Ken Muneoka for providing DiI reagent and protocols, and Dr Philippa Francis-West for her critical reading and suggestions on the manuscript. This work was supported by NIH grants R01DE12329 and R01 DE14044 to Y.P.C., and by a subcontract of P50 DE16215 to J. Murray (University of Iowa), which supported Y.P.C.
References
- Alappat SR, Zhang Z, Suzuki K, Zhang X, Liu H, Jiang R, Yamada G, Chen Y. The cellular and molecular etiology of the cleft secondary palate in Fgf10 mutant mice. Dev. Biol. 2005;277:102–113. doi: 10.1016/j.ydbio.2004.09.010. [DOI] [PubMed] [Google Scholar]
- Bi W, Deng JM, Zhang Z, Behringer RR, de Crombrugghe B. Sox9 is required for cartilage formation. Nat. Genet. 1999;22:85–89. doi: 10.1038/8792. [DOI] [PubMed] [Google Scholar]
- Blanton SH, Bertin T, Serna M, Stal S, Mulliken JB, Hecht JT. Association of chromosomal regions 3p21.2, 10p13, and 16p13.3 with nonsyndromic cleft lip and palate. Am. J. Med. Genet. 2004;125A:23–27. doi: 10.1002/ajmg.a.20426. [DOI] [PubMed] [Google Scholar]
- Brugmann SA, Goodnough LH, Gregorieff A, Leucht P, ten Berge D, Fuerer C, Clevers H, Nusse R, Helms JA. Wnt signaling mediates regional specification in the vertebrate face. Development. 2007;134:3283–3295. doi: 10.1242/dev.005132. [DOI] [PubMed] [Google Scholar]
- Cadigan KM, Nusse R. Wnt signaling: a common theme in animal development. Genes Dev. 1997;11:3286–3305. doi: 10.1101/gad.11.24.3286. [DOI] [PubMed] [Google Scholar]
- Carroll TJ, Park J-S, Hayashi S, Majumdar A, McMahon AP. Wnt9b plays a central role in the regulation of mesenchymal to epithelial transitions underlying organogenesis of the mammalian urogenital system. Dev. Cell. 2005;9:283–292. doi: 10.1016/j.devcel.2005.05.016. [DOI] [PubMed] [Google Scholar]
- Ceccarelli S, Cardinali G, Aspite N, Picardo M, Marchese C, Torrisi MR, Mancini P. Cortactin involvement in the keratinocyte growth factor and fibroblast growth factor 10 promotion of migration and cortical actin assembly in human keratinocytes. Exp. Cell Res. 2007;313:1758–1777. doi: 10.1016/j.yexcr.2007.03.013. [DOI] [PubMed] [Google Scholar]
- Chen Y, Bei M, Woo I, Satokata I, Maas R. Msx1 controls inductive signaling in mammalian tooth morphogenesis. Development. 1996;122:3035–3044. doi: 10.1242/dev.122.10.3035. [DOI] [PubMed] [Google Scholar]
- Dasgupta R, Fuchs E. Multiple roles for activated LEF/TCF transcription complexes during hair follicle development and differentiation. Development. 1999;126:4557–4568. doi: 10.1242/dev.126.20.4557. [DOI] [PubMed] [Google Scholar]
- Ferguson MWJ. Palate development. Development. 1988;103:41–60. doi: 10.1242/dev.103.Supplement.41. [DOI] [PubMed] [Google Scholar]
- Gritli-Linde A. Molecular control of secondary palate development. Dev. Biol. 2007;301:309–326. doi: 10.1016/j.ydbio.2006.07.042. [DOI] [PubMed] [Google Scholar]
- Gu S, Wei N, Yu XY, Jiang Y, Fei J, Chen YP. Mice with an anterior cleft of the palate survive neonatal lethality. Dev. Dyn. 2008;237:1509–1516. doi: 10.1002/dvdy.21534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halford MM, Armes J, Buchert M, Meskenaite V, Grail D, Hibbs ML, Wilks AF, Farlie PG, Newgreen DF, Hovens CM, et al. Ryk-deficient mice exhibit craniofacial defects associated with perturbed Eph receptor crosstalk. Nat. Genet. 2000;25:414–418. doi: 10.1038/78099. [DOI] [PubMed] [Google Scholar]
- Hilliard SA, Yu L, Gu S, Zhang Z, Chen YP. Regional regulation of palatal growth and patterning along the anterior-posterior axis in mice. J. Anat. 2005;207:655–667. doi: 10.1111/j.1469-7580.2005.00474.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh M, Boerboom D, Shimada M, Lo Y, Parlow AF, Luhmann UFO, Berger W, Richards JS. Mice null for Frizzled4 (Fzd4−/−) are infertile and exhibit impaired corpora lutea formation and function. Biol. Reprod. 2005;73:1135–1146. doi: 10.1095/biolreprod.105.042739. [DOI] [PubMed] [Google Scholar]
- Huelsken J, Vogel R, Erdmann B, Cotsarelis G, Birchmeier W. β-catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell. 2001;105:533–545. doi: 10.1016/s0092-8674(01)00336-1. [DOI] [PubMed] [Google Scholar]
- Jopling C, den Hertog J. Fyn/Yes and non-canonical Wnt signaling converge on RhoA in vertebrate gastrulation cell movements. EMBO Rep. 2005;6:426–431. doi: 10.1038/sj.embor.7400386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keeble TR, Halford MM, Seaman C, Kee N, Macheda M, Anderson RB, Stacker SA, Cooper HM. The Wnt receptor Ryk is required for Wnt5a-mediated axon guidance on the contralateral side of the corpus callosum. J. Neurosci. 2006;26:5840–5848. doi: 10.1523/JNEUROSCI.1175-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilian B, Mansukoski H, Barbosa FC, Ulrich F, Tada M, Heisenberg CP. The role of Ppt/Wnt5a in regulating cell shape and movement during zebrafish gastrulation. Mech. Dev. 2003;120:467–476. doi: 10.1016/s0925-4773(03)00004-2. [DOI] [PubMed] [Google Scholar]
- Kim HJ, Schleiffarth JR, Jessurun J, Sumanas S, Lin S, Ekker SC. Wnt5a signaling in vertebrate pancreas development. BMC Biol. 2005;3:23. doi: 10.1186/1741-7007-3-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S, Muneoka K. Cell migration and chick limb development: Chemotactic action of FGF4 and the AER. Dev. Biol. 1999;211:335–347. doi: 10.1006/dbio.1999.9317. [DOI] [PubMed] [Google Scholar]
- Li C, Xiao J, Hormi K, Borok Z, Minoo P. Wnt5a participates in distal lung morphogenesis. Dev. Biol. 2002;248:68–81. doi: 10.1006/dbio.2002.0729. [DOI] [PubMed] [Google Scholar]
- Li C, Hu L, Xiao J, Chen H, Li JT, Bellusci S, Delanghe S, Minoo P. Wnt5a regulates Shh and Fgf10 signaling during lung development. Dev. Biol. 2005;287:86–97. doi: 10.1016/j.ydbio.2005.08.035. [DOI] [PubMed] [Google Scholar]
- Liang H, Chen Q, Coles AH, Anderson SJ, Pihan G, Bradley A, Gerstein R, Jurecic R, Jones SN. Wnt5a inhibits B cell proliferation and functions as a tumor suppressor in hematopoietic tissue. Cancer Cell. 2003;4:349–360. doi: 10.1016/s1535-6108(03)00268-x. [DOI] [PubMed] [Google Scholar]
- Lu W, Yamamoto V, Ortega B, Baltimore D. Mammalian Ryk is a Wnt coreceptor required for stimulation of neurite outgrowth. Cell. 2004;119:97–108. doi: 10.1016/j.cell.2004.09.019. [DOI] [PubMed] [Google Scholar]
- Martinez-Alvarez C, Tudela C, Perez-Miguelsanz J, O’Kane S, Puerta J, Ferguson MWJ. Medial edge epithelial cell fate during palatal fusion. Dev. Biol. 2000;220:343–357. doi: 10.1006/dbio.2000.9644. [DOI] [PubMed] [Google Scholar]
- Mikels AJ, Nusse R. Purified Wnt5a protein activates or inhibits beta-catenin/TCF signaling depending on receptor context. PLoS Biol. 2006;4:e115. doi: 10.1371/journal.pbio.0040115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mlodzik M. Planar cell polarization: do the same mechanisms regulate Drosophila tissue polarity and vertebrate gastrulation? Trends Genet. 2002;18:564–571. doi: 10.1016/s0168-9525(02)02770-1. [DOI] [PubMed] [Google Scholar]
- Moon RT, Campbell RM, Christian JL, McGrew LL, Shih J, Fraser S. Xwnt-5A: a maternal Wnt that affects morphogenetic movements after overexpression in embryos of Xenopus laevis. Development. 1993;119:97–111. doi: 10.1242/dev.119.1.97. [DOI] [PubMed] [Google Scholar]
- Mori-Akiyama Y, Akiyama H, Rowitch DH, de Crombrugghe B. Sox9 is required for determination of the chondrogenic cell lineage in the cranial neural crest. Proc. Natl. Acad. Sci. USA. 2003;100:9360–9365. doi: 10.1073/pnas.1631288100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natanson-Yaron S, Anteby EY, Greenfield C, Goldman-Wohl D, Hamani Y, Hochner-Celnikier D, Yagel S. FGF10 and Sprouty 2 modulate trophoblast invasion and branching morphogenesis. Mol. Hum. Reprod. 2007;13:511–519. doi: 10.1093/molehr/gam034. [DOI] [PubMed] [Google Scholar]
- Nawshad A, Hay ED. TGFβ3 signaling activates transcription of the LEF1 gene to induce epithelial mesenchymal transformation during mouse palate development. J. Cell Biol. 2003;163:1291–1301. doi: 10.1083/jcb.200306024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niemann S, Zhao C, Pascu F, Stahl U, Aulepp U, Niswander L, Weber J, Muller U. Homozygous WNT3 mutation causes Tetra-Amelia in a large consanguineous family. Am. J. Hum. Genet. 2004;74:558–563. doi: 10.1086/382196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishita M, Yoo SK, Nomachi A, Kani S, Sougawa N, Ohta Y, Takada S, Kikuchi A, Minami Y. Filopodia formation mediated by receptor tyrosine kinase Ror2 is required for Wnt5a-induced cell migration. J. Cell Biol. 2006;175:555–562. doi: 10.1083/jcb.200607127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oishi I, Suzuki H, Onishi N, Takada R, Kani S, Ohkawara B, Koshida I, Suzuki K, Yamada G, Schwabe GC, et al. The receptor tyrosine kinase Ror2 is involved in non-canonical Wnt5a/JNK signalling pathway. Genes Cells. 2003;8:645–654. doi: 10.1046/j.1365-2443.2003.00662.x. [DOI] [PubMed] [Google Scholar]
- Okano J, Suzuki S, Shiota K. Regional heterogeneity in the developing palate: morphological and molecular evidence for normal and abnormal palatogenesis. Congenit. Anom. 2006;46:49–54. doi: 10.1111/j.1741-4520.2006.00103.x. [DOI] [PubMed] [Google Scholar]
- Park WY, Miranda B, Lebeche D, Hashimoto G, Cardoso WV. FGF-10 is a chemotactic factor for distal epithelial buds during lung development. Dev. Biol. 1998;201:125–134. doi: 10.1006/dbio.1998.8994. [DOI] [PubMed] [Google Scholar]
- Rice R, Spencer-Dene B, Connor EC, Gritli-Linde A, McMahon AP, Dickson C, Thesleff I, Rice DPC. Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate. J. Clin. Invest. 2004;113:1692–1900. doi: 10.1172/JCI20384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roarty K, Serra R. Wnt5a is required for proper mammary gland development and TGF-β-mediated inhibition of ductal growth. Development. 2007;134:3929–3939. doi: 10.1242/dev.008250. [DOI] [PubMed] [Google Scholar]
- Sakaguchi S, Nakatani Y, Takamatsu N, Hori H, Kawakami A, Inohaya K, Kudo A. Medaka unextended-fin mutants suggest a role for Hox8c in cell migration and osteoblast differentiation during appendage formation. Dev. Biol. 2006;293:426–438. doi: 10.1016/j.ydbio.2006.02.017. [DOI] [PubMed] [Google Scholar]
- Schambony A, Wedlich D. Wnt-5A/Ror2 regulate expression of XPAPC through an alternative noncanonical signaling pathway. Dev. Cell. 2007;12:779–792. doi: 10.1016/j.devcel.2007.02.016. [DOI] [PubMed] [Google Scholar]
- Schwabe GC, Trepczik B, Suring K, Brieske N, Tucker AS, Sharpe PT, Minami Y, Mundlos S. Ror2 knockout mouse as a model for the developmental pathology of autosomal recessive Robinow syndrome. Dev. Dyn. 2004;229:400–410. doi: 10.1002/dvdy.10466. [DOI] [PubMed] [Google Scholar]
- Takeuchi S, Takeda K, Oishi I, Nomi M, Ikeya M, Itoh K, Tamura S, Ueda T, Hatta T, Otani H, et al. Mouse Ror2 receptor tyrosine kinase is required for the heart development and limb formation. Genes Cells. 2000;5:71–78. doi: 10.1046/j.1365-2443.2000.00300.x. [DOI] [PubMed] [Google Scholar]
- Taya Y, Okane S, Ferguson MWJ. Pathogenesis of cleft palate in TGF-β3 knockout mice. Development. 1999;126:3869–3879. doi: 10.1242/dev.126.17.3869. [DOI] [PubMed] [Google Scholar]
- Topol L, Jiang X, Choi H, Garrett-Beal L, Carolan PJ, Yang Y. Wnt-5a inhibits the canonical Wnt pathway by promoting GSK-3-independent {beta}-catenin degradation. J. Cell Biol. 2003;162:899–908. doi: 10.1083/jcb.200303158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres MA, Yang-Snyder JA, Purcell SM, DeMarais AA, McGrew LL, Moon RT. Activities of the Wnt-1 class of secreted signaling factors are antagonized by the Wnt-5A class and by a dominant negative cadherin in early Xenopus development. J. Cell Biol. 1996;133:1123–1137. doi: 10.1083/jcb.133.5.1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tudela C, Formoso M-A, Martínez T, Pérez R, Aparicio M, Maestro C, Río AD, Martínez E, Ferguson MW, Martínez-Alvarez C. TGF-β3 is required for the adhesion and intercalation of medial edge epithelial cells during palate fusion. Int. J. Dev. Biol. 2002;46:333–336. [PubMed] [Google Scholar]
- Veeman MT, Axelrod JD, Moon RT. A second canon: functions and mechanisms of β-catenin-independent Wnt signaling. Dev. Cell. 2003;5:367–377. doi: 10.1016/s1534-5807(03)00266-1. [DOI] [PubMed] [Google Scholar]
- Weeraratna AT, Jiang Y, Hostetter G, Rosenblatt K, Duray P, Bittner M, Trent JM. Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer Cell. 2002;3:279–288. doi: 10.1016/s1535-6108(02)00045-4. [DOI] [PubMed] [Google Scholar]
- Witze E, Litman ES, Argast GM, Moon RT, Ahn NG. Wnt5a control of cell polarity and directional movement by polarized redistribution of adhesion receptors. Science. 2008;320:365–369. doi: 10.1126/science.1151250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi TP, Bradley A, McMahon AP, Jones S. A Wnt5a pathway underlies outgrowth of multiple structures in the vertebrate embryo. Development. 1999;126:1211–1223. doi: 10.1242/dev.126.6.1211. [DOI] [PubMed] [Google Scholar]
- Yamamoto H, Yoo SK, Nishita M, Kikuchi A, Minami Y. Wnt5a modulates glycogen synthase kinase 3 to induce phosphorylation of receptor tyrosine kinase Ror2. Genes Cells. 2007;12:1215–1223. doi: 10.1111/j.1365-2443.2007.01128.x. [DOI] [PubMed] [Google Scholar]
- Yu L, Gu S, Alappat S, Song Y, Yan M, Zhang X, Zhang G, Jiang Y, Zhang Z, Zhang Y, Chen YP. Shox2-deficient mice exhibit a rare type of incomplete clefting of the secondary palate. Development. 2005;132:4397–4406. doi: 10.1242/dev.02013. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Song Y, Zhao X, Zhang X, Fermin C, Chen YP. Rescue of cleft palate in Msx1-deficient mice by transgenic Bmp4 reveals a network of BMP and Shh signaling in the regulation of mammalian palatogenesis. Development. 2002;129:4135–4146. doi: 10.1242/dev.129.17.4135. [DOI] [PubMed] [Google Scholar]