Abstract
Plants grow in dense vegetations at the risk of being out-competed by neighbors. To increase their competitive power, plants display adaptive responses, such as rapid shoot elongation (shade avoidance) to consolidate light capture. These responses are induced upon detection of proximate neighbors through perception of the reduced ratio between red (R) and far-red (FR) light that is typical for dense vegetations. The plant hormone auxin is a central regulator of plant development and plasticity, but until now it has been unknown how auxin transport is controlled to regulate shade-avoidance responses. Here, we show that low R:FR detection changes the cellular location of the PIN-FORMED 3 (PIN3) protein, a regulator of auxin efflux, in Arabidopsis seedlings. As a result, auxin levels in the elongating hypocotyls are increased under low R:FR. Seedlings of the pin3-3 mutant lack this low R:FR-induced increase of endogenous auxin in the hypocotyl and, accordingly, have no elongation response to low R:FR. We hypothesize that low R:FR-induced stimulation of auxin biosynthesis drives the regulation of PIN3, thus allowing shade avoidance to occur. The adaptive significance of PIN3-mediated control of shade-avoidance is shown in plant competition studies. It was found that pin3 mutants are outcompeted by wild-type neighbors who suppress fitness of pin3-3 by 40%. We conclude that low R:FR modulates the auxin distribution by a change in the cellular location of PIN3, and that this control can be of great importance for plants growing in dense vegetations.
Keywords: phytochrome, phenotypic plasticity, plant–plant signaling
Plants often grow in dynamic environments, with severe competition for light with surrounding neighbors. As a result, light intensities can decrease drastically, but the light quality also changes. Because of selective absorption by chlorophyll, red light (R) is strongly depleted in dense canopies. Far-red light (FR), on the other hand, is mostly reflected, resulting in a steep decline of the R:FR ratio in a canopy, which can be sensed by the phytochrome photoreceptors of neighboring plants (1, 2). Once neighbors have been detected through this R:FR reduction, a suite of responses defined as the Shade-Avoidance Syndrome (SAS) are induced, including pronounced shoot elongation and upward leaf movement (hyponasty) to consolidate light capture (2, 3). Perception of the R:FR signal induces interactions with various hormones to control cell elongation, which underpins the elongation responses to neighbors (4–8).
It was proposed a decade ago that auxin would be an important regulator of the SAS, tentatively through enhanced lateral transport toward the epidermal cells of the elongating organs upon low R:FR perception (9). Accordingly, transcript levels of several auxin-related genes (e.g., AUX/IAA and PIN genes) are increased by low R:FR (7, 10, 11). Recently, robust evidence accumulated identifying auxin as a key regulator of shade avoidance. It was shown that enhanced auxin biosynthesis, intact auxin signaling, and auxin transport are essential to low R:FR-induced elongation (7, 12). Low R:FR regulates the expression of TRYPTOPHAN AMINOTRANSFERASE OF ARABIDOPSIS1 (TAA1), which facilitates auxin biosynthesis through a dedicated tryptophan-dependent pathway (12, 13). Despite these recent novel insights, a key question remains unanswered: which auxin transporters regulate shade-avoidance responses to low R:FR, an early warning signal of upcoming competition?
The regulation of auxin fluxes in plants has been well-studied, particularly in meristematic tissues. PIN-FORMED (PIN) proteins, a family of eight members in Arabidopsis, control the direction and rate of cellular auxin efflux (14, 15). Little is known about light regulation of PIN action, with the exception of phototropic hypocotyl bending toward blue light (16, 17). The cellular localization of PIN1 and PIN3 proteins in dark-grown seedlings changed from basal to lateral upon exposure to directional blue light. As a result, an auxin gradient was established to control differential cell expansion on the illuminated versus the nonilluminated side of the hypocotyl.
It is unknown if PINs play a role in the process of shade avoidance and if their abundance and localization are controlled by phytochrome-mediated detection of R:FR. Here we show that low R:FR detection enhances endogenous indole-3-acetic acid (IAA) levels of the hypocotyl in a PIN3-dependent manner. Low R:FR stimulates PIN3 abundance and induces a lateral cellular reorientation of PIN3. The adaptive significance of this PIN3-mediated regulation is shown in plant-competition experiments at high plant densities, where fitness of pin3-3 mutant is suppressed when competing against wild-type neighbors.
Results and Discussion
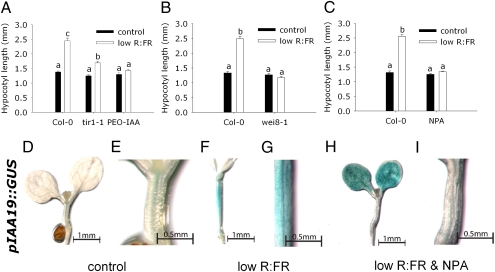
Fig. 1A shows that reduction of the R:FR, achieved by far-red enrichment of a standard white-light background (7), stimulates hypocotyl elongation in Arabidopsis. Seedlings of the auxin receptor mutant tir1-1 (18, 19) and seedlings exposed to 50 μM α-(phenylethyl-2-one)-IAA (PEO-IAA), an antagonist for the auxin receptor TIR1 and its homologs (20), displayed much reduced or absent hypocotyl elongation in low R:FR. The auxin biosynthesis mutant wei8-1 has a knock-out mutation for TAA1 (13) and shows no response to low R:FR (Fig. 1B). This finding confirms the requirement of elevated auxin biosynthesis for SAS (12). Blocking polar auxin transport (PAT) with naphthylphthalamic acid (NPA; 25 μM) (21) inhibited low R:FR-induced elongation (Fig. 1C). There is an overall up-regulation of IAA19 expression in low R:FR-exposed hypocotyls, as indicated by enhanced pIAA19::GUS staining (Fig. 1 D–G), which was confirmed by quantitative RT-PCR on IAA19 (Fig. S1), implying increased auxin activity (22). The pIAA19::GUS staining occurred in the more lateral hypocotyl regions under low R:FR, whereas under control conditions staining is restricted to the vascular tissue, which would suggest a change in directionality of auxin transport during low R:FR (Fig. 1 F and G). Although this staining pattern could also follow from differentially regulated increase of auxin responsiveness, this result is less likely the case because inhibition of PAT using NPA prevented the low R:FR-induced pIAA19::GUS staining pattern (Fig. 1 H and I). These data suggest that auxin biosynthesis, the established PAT, and auxin perception through TIR1 (and TIR1 homologs) are essential to low R:FR induced hypocotyl elongation.
Fig. 1.
Auxin involvement in low R:FR-induced hypocotyl elongation. Inhibition of auxin signaling (tir1-1 and 50 μM PEO-IAA) (A), biosynthesis (wei8-1) (B), or transport (25 μM NPA) (C), reduced the hypocotyl elongation response to 5-d low R:FR treatment. Auxin activity was visualized with the use of pIAA19::GUS, which indicated that the low R:FR treatment induced auxin activity all over the hypocotyl compared with control light conditions, and that this up-regulation is abolished when treated with NPA (D–I). E, G, and I are higher magnification pictures of the hypocotyls from D, F, and H. Data are means (n = 24–44) ± SE; the Genotype × Light interaction is significant in all panels (P < 0.05) and different letters indicate significant differences between means (P < 0.05) (A–C).
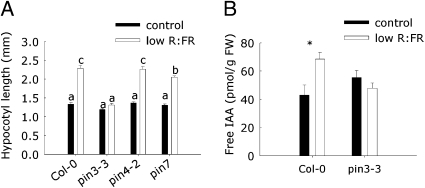
PIN proteins are known to be the key regulators of directional auxin transport and therefore several mutants for PINs that are known to be expressed in the hypocotyl were tested for the low R:FR-induced elongation response (Fig. 2A). We found that pin3-3 displayed an abolished response, similar to plants treated with the overall auxin efflux inhibitor NPA, whereas the other pin mutants tested showed a wild-type response. We, therefore, suggest that PIN3 is an important transport-associated protein to control low R:FR-induced elongation growth. We determined endogenous free IAA levels to confirm if auxin concentrations are enhanced during low R:FR and to study if this is PIN3-dependent. Care was taken to harvest separate hypocotyls, rather than whole seedlings, which was done previously (12). We found that free IAA levels were elevated in hypocotyls of low R:FR-exposed seedlings (Fig. 2B). Importantly, this low R:FR-induced increase of free IAA was completely abolished in pin3-3. Summarizing, inhibition of either low R:FR-induced auxin biosynthesis or of low R:FR-induced transport coordination of these increased IAA levels through PIN3 (Figs. 1 and 2) leads to complete inhibition of hypocotyl elongation during low R:FR conditions.
Fig. 2.
PIN3 regulates low R:FR-induced hypocotyl elongation and free IAA accumulation. (A) Different pin-like (pin) mutants were grown for 5 d under both control and low R:FR light conditions. The data shows that pin3-3 lacks the elongation response of the hypocotyl induced by low R:FR treatment, whereas pin4-2 and pin7 had a wild-type–like response. Data are mean (n = 25–41) ± SE; the Genotype * Light interaction is significant in all panels (P < 0.05) and different letters indicate significant differences between means (P < 0.05). (B) The levels of free IAA were measured on both Col-0 and pin3-3 hypocotyls after 2 d of low R:FR or control light treatment. Data are means (n = 4–5) ± SE; the Genotype × Light interaction is significant in all panels (P < 0.05). Different letters or asterisk (*) indicate significant differences between means (P < 0.05).
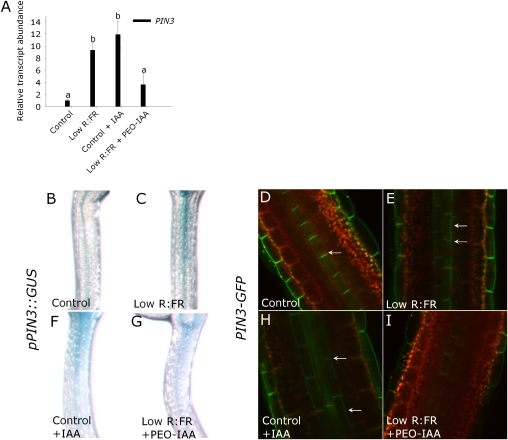
For PIN3 to regulate auxin transport in an R:FR-dependent manner, PIN3 itself would be expected to be regulated upon low R:FR detection. Staining of pPIN3::GUS indicated enhanced PIN3 expression in the elongating hypocotyl. This finding was confirmed by quantitative RT-PCR showing a ninefold up-regulation of PIN3 (Fig. 3 A–C). In addition to enhanced expression, the cellular localization of PIN3 protein appeared to be changed upon low R:FR exposure, as indicated by the PIN3-GFP fusion protein. In control light conditions, PIN3-GFP localized exclusively to the basal side of the endodermal cells in the hypocotyl (Fig. 3 D and E; see Fig. S2 for a Z-stack from epidermis into the vasculature). However, low R:FR changed this orientation to a predominantly lateral orientation. This new orientation would imply IAA to be transported from the inner cells toward the more lateral cell layers, which would be consistent with the increased auxin reporter activity (pIAA19::GUS) (Fig. 1 D–I) in those outer layers.
Fig. 3.
Low R:FR regulates PIN expression and PIN3-GFP localization in an auxin-dependent manner. (A) The relative transcript abundance of PIN3 in seedlings upon 2 d of low R:FR, IAA (10 μM), or the auxin receptor inhibitor PEO-IAA (50 μM). Data are means (n = 3–4) ± SE and different letters indicate significant differences between means (P < 0.05). (B–I) Using pPIN3::GUS, the localization of PIN3 expression was visualized (B, C, F, and G), whereas PIN3 cellular localization was studied using PIN3-GFP (D, E, H, and I). Expression and cellular localization were affected by 2 d of low R:FR (A–E), IAA (10 μM), or PEO-IAA (50 μM) (A and F–I). Arrows indicate the basal versus lateral orientation of PIN3-GFP.
Auxin has been shown to regulate its own efflux by affecting the expression and subcellular PIN localization (23–26). Therefore, we studied if elevated levels in low R:FR drives the change of PIN3 expression and PIN3 localization under low R:FR. Interestingly, the relative transcript abundance of PIN3 and pPIN3::GUS activity were enhanced by IAA and the induction under low R:FR was inhibited by the auxin signaling inhibitor PEO-IAA (Fig. 3 A, F, and G). PIN3-GFP abundance and localization was affected by these two treatments in much the same way (Fig. 3 H and I). These data are consistent with the hypothesis that elevated auxin levels in low R:FR itself regulates PIN3 gene expression and PIN3 protein abundance and localization, thus directing its own transport. It remains to be studied how PIN3 is directed toward the lateral side of the cell membrane, but one possibility is that this would occur through phosphorylation (27–29).
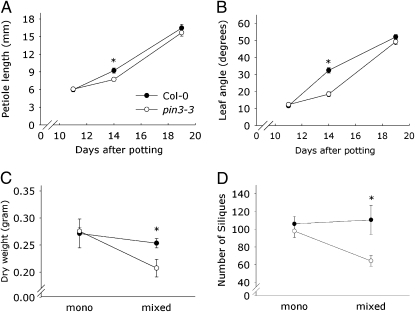
The above-mentioned data show how PIN3 controls auxin-driven shade avoidance in Arabidopsis seedlings exposed to low R:FR. Next, we studied if these PIN3-driven processes are adaptive to plants growing under intense competition for light in dense stands, which was done by comparing plants grown in high density (1,460 plants per square meter) monocultures of wild-type Col-0 and pin3-3. Both petiole length and leaf angles increased in response to proximate neighbors (classic shade-avoidance features; see also ref. 30) as the dense stands developed, but these responses were delayed in pin3-3 (Fig. 4 A and B). This reduced shade-avoidance response to real neighboring competitors is consistent with the lack of low R:FR-induced hypocotyl elongation (Fig. 2A) and with the reduced petiole elongation and leaf angle response to low R:FR in plants in the rosette stage (Fig. S3). To test if the delayed response to neighbors of pin3-3 has consequences for fitness, mixed stands of 1:1 = Col-0: pin3-3 were grown in a checkerboard design. Vegetative dry weight (Fig. 4C) was recorded as a measure for growth, whereas the number of siliques (Fig. 4D) was counted as a measure for reproductive output. Numbers of seeds per silique were counted for a number of subsamples and these were not significantly different between pin3-3 and Col-0 (Fig. S4A). Growth and reproductive output were similar between pin3-3 and Col-0 in their respective monocultures, but both were significantly suppressed in pin3-3 when competing with Col-0 neighbors in the mixtures. Reproductive output of pin3-3 in mixed stands was suppressed to only 60% of the reproductive output of neighboring Col-0. This finding also means that fitness of pin3-3 in competition with Col-0 is reduced to 60% of pin3-3 grown in monoculture. Consistent with the reduced reproductive output of pin3-3 in competition with Col-0, pin3-3 mutants had gained a subordinate position in the mixed canopy evidenced by their reduced final plant height (Fig. S4B).
Fig. 4.
Competitive ability is reduced in pin3-3. (A and B) Petiole length and leaf angle were measured over time on both Col-0 (●) and pin3-3 (○) grown in dense stands, revealing that pin3-3 has a delayed response to neighbor plants compared with Col-0. Data are means (n = 12) ± SE and an asterisk (*) indicates a significant difference in mean (P < 0.05). (C and D) Dry weight (C) and the number of siliques (D) were measured 67 d after potting on Col-0 and pin3-3 grown in mono or mixed culture dense stands. Data show that pin3-3 is outcompeted by Col-0 in the mixed stands. Data are means (n = 5) ± SE; * indicates a significant difference (P < 0.05).
Fig. 4 A and B show that pin3-3 is not completely irresponsive to neighbors and this mutant also retains a low R:FR response in leaves of 3-wk-old plants (Fig. S3), different from the seedling response to low R:FR, which is completely inhibited in pin3-3. This difference might be related to developmental stage or putative organ-specificity of PIN3 regulation. Furthermore, a real vegetation harbors a much greater signal complexity for neighbor detection. In addition to low R:FR, blue light fluence rates are also reduced in canopies (30) and elevated ethylene levels might possibly occur (31). These signals might act partly independent of PIN3, as has been suggested for ethylene-induced hyponasty (32).
Conclusion
Low R:FR-induced hypocotyl elongation requires intact auxin signaling and involves accumulation of free IAA in the hypocotyl. This accumulation requires auxin transport regulation through PIN3, which is likely to allow for increased auxin transport to the cortex and epidermal cells, corresponding with the enhanced activity of auxin reporters. To establish this auxin gradient, low R:FR induces increased PIN3 gene expression and PIN3 protein abundance, and induces a predominantly lateral cellular localization of PIN3 in the endodermis cells. IAA itself can induce and IAA perception is required for this PIN3 behavior to occur during low R:FR. We, therefore, hypothesize that low R:FR-induced auxin biosynthesis stimulates PIN3 abundance and lateral localization, thus inducing a change of auxin flow toward the epidermal cell layers where growth is likely controlled (33). The finding that fitness of pin3-3 mutants was severely impaired by competing wild-type neighbors indicates that PIN3-mediated control of shade avoidance is probably of great adaptive significance for shade-intolerant plants in nature.
Materials and Methods
Seedling Experiment.
Seeds were sterilized, stratified, and germinated on solid agar plates as in ref. 7, and 24 h after germination the plates were placed into low R:FR treatment or in control light conditions for 2 to 5 d, with both 130 μmol m-2 s-1 photosynthetically active radiation (16-h light/8-h dark). Low R:FR (R:FR = 0.3, compared with 1.2 under control light conditions) was created by supplemental far-red light, using 730-nm LEDs (for details and spectra see ref. 7). The following mutants, all in Columbia (Col-0) background, were used: tir1-1 (34), wei8-1 (13), pin3-3 and pin4-2 (16, 35), and pin7 (salk_048791).
Pharmacological Experiments.
Auxin activity was induced with indole-3-acetic acid (IAA; 10 μM), whereas auxin transport was inhibited with naphthylphthalamic acid (NPA; 25 μM) and auxin signaling was inhibited with α-(phenylethyl-2-one)-IAA (PEO-IAA; 50 μM) (20, 21). The chemicals were added to the medium at the start of light treatment.
GUS Assay.
To visualize auxin activity, low R:FR-treated hypocotyls, the transgenic pIAA19::GUS line expressing GUS driven by the IAA19 promoter was used (22). For the expression pattern of PIN3, the transgenic pPIN3::GUS line was used (16). The GUS assay was performed by overnight incubation of freshly harvested material in staining solution [1 mM X-Gluc (5-bromo-4-chloro-3-indolyl β-d-glucoronide)] in 100 mM NaPi buffer pH 7.0, 0.1 mM EDTA, 0.1% Triton X-100, 1 mM K4Fe(CN)6, 1 mM K3Fe(CN)6, 0.52 mg·mL−1 dimethyl formamide. Thereafter, seedlings were bleached in 70% ethanol for at least 1 d.
GFP Visualization.
To study PIN3 protein location, GFP fluorescence was studied in PIN3-GFP transgenic plants (36). PIN3-GFP fluorescence was visualized with confocal laser scanning microscopy (ZEISS LSM Pascal, 40× C-apochromat objective) using a 488-nm excitation wavelength, 505 to 530 band-path filter to separate GFP and a 560 long-pass filter to determine chlorophyll fluorescence.
Free IAA Determination.
After 2 d of low R:FR treatment, 50 mg hypocotyl tissue (fresh weight) was harvested, whereby the hypocotyls were cut from the cotyledons and roots and snap-frozen in liquid nitrogen. The plant material was then processed as explained in ref. 37. Free-IAA levels were then measured using a Varian Saturn 2000 GC-MS/MS system (Varian) (37).
Quantitative RT-PCR.
RNA extraction, cDNA synthesis, quantitative RT-PCR, and calculation of relative expression were performed as in ref. 7. Gene-specific primers: PIN3-F (At1g70940), 5′-GCGTCAATAAAAACCCGAAA-3′; PIN3-R, 5′- GGCGTCTTTTGGTCTCTCTG-3′, IAA19-F (At3g15540), 5′-GGCTTGAGATAACGGAGCTG-3′; IAA19-R, 5′-ACCATCTTTCAAGGCCACAC-3′. For this, UBIQUITIN was used as an internal standard to normalize for differences in cDNA concentration between samples: UBQ10-F, 5′ GGCCTTGTATAATCCCTGATGAATAAG-3′; UBQ10-R, 5′-AAAGAGATAACAGGAACGGAAACATAGT-3′.
Competition Experiments.
Seeds for canopy experiments were sown on soil with additional nutrients (7) and stratified for 4 d at 4 °C. Thereafter, the seeds were transferred to light. After 10 d (three leaf stages) seedlings were transplanted to a density of 1,460 plants per square meter. Each seedling was in one pot, preventing below-ground competition. Monoculture stands consisted of 49 plants (7 × 7) and the mixed stands of 64 plants (8 × 8, 32 Col-0 and 32 pin3-3 plants) in a checkerboard design (38, 39). The outer two rows of plants were not harvested to exclude possible edge-effects, leaving 9 (3 × 3) or 16 (4 × 4) plants per stand for measurements. Leaf angles and petiole lengths of the two most responsive leaves per individual were recorded at different times and the mean was taken for further calculations. At the end of the competition trial (after 67 d of competition) plants were harvested, plant height and number of siliques per inflorescence were determined, and shoots were dried for 4 d at 60 °C. The individual dry weight, silique number, and height were measured for every shoot and the mean of each stand was used for further calculations.
Statistical Analyses.
Data were analyzed with a two-way ANOVA followed by Tukey's-B post hoc test to allow for comparisons among all means or with Student's t test when two means were compared (SPSS v14).
Supplementary Material
Acknowledgments
We thank P. Düchting (Ruhr University, Bochum, Germany) for technical assistance, I. Ruberti (University of Rome, Rome) for discussions, and R. Sasidharan (Utrecht University, Utrecht, The Netherlands) for helpful comments on a draft of this manuscript. Seeds for mutants and reporters were kindly shared by J.M. Alonso (North Carolina State University, Raleigh, NC), I. Blilou (Utrecht University, Utrecht, The Netherlands), J. Friml (VIB, Ghent, Belgium), and K.T. Yamamoto (Hokkaido University, Sapporo, Japan) or obtained from the Nottingham Arabidopsis Stock Centre. The α-(phenylethyl-2-one)-indole-3-acetic acid was kindly provided by K.-I. Hayashi (Okayama University of Science, Okayama City, Japan). R.P. is funded by Veni Grant 86306001 from the Netherlands Organization for Scientific Research.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1013457108/-/DCSupplemental.
References
- 1.Ballaré CL, Scopel AL, Sánchez RA. Far-red radiation reflected from adjacent leaves: An early signal of competition in plant canopies. Science. 1990;247:329–332. doi: 10.1126/science.247.4940.329. [DOI] [PubMed] [Google Scholar]
- 2.Franklin KA. Shade avoidance. New Phytol. 2008;179:930–944. doi: 10.1111/j.1469-8137.2008.02507.x. [DOI] [PubMed] [Google Scholar]
- 3.Ballaré CL. Keeping up with the neighbours: Phytochrome sensing and other signalling mechanisms. Trends Plant Sci. 1999;4:97–102. doi: 10.1016/s1360-1385(99)01383-7. [DOI] [PubMed] [Google Scholar]
- 4.Keuskamp DH, Sasidharan R, Pierik R. Physiological regulation and functional significance of shade avoidance responses to neighbors. Plant Signal Behav. 2010;5:655–662. doi: 10.4161/psb.5.6.11401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Martinez-Garcia JF, et al. Regulatory components of shade avoidance syndrome. Adv Bot Res. 2010;53:65–116. [Google Scholar]
- 6.de Lucas M, et al. A molecular framework for light and gibberellin control of cell elongation. Nature. 2008;451:480–484. doi: 10.1038/nature06520. [DOI] [PubMed] [Google Scholar]
- 7.Pierik R, Djakovic-Petrovic T, Keuskamp DH, de Wit M, Voesenek LACJ. Auxin and ethylene regulate elongation responses to neighbor proximity signals independent of gibberellin and della proteins in Arabidopsis. Plant Physiol. 2009;149:1701–1712. doi: 10.1104/pp.108.133496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vandenbussche F, Pierik R, Millenaar FF, Voesenek LACJ, Van Der Straeten D. Reaching out of the shade. Curr Opin Plant Biol. 2005;8:462–468. doi: 10.1016/j.pbi.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 9.Morelli G, Ruberti I. Shade avoidance responses. Driving auxin along lateral routes. Plant Physiol. 2000;122:621–626. doi: 10.1104/pp.122.3.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Devlin PF, Yanovsky MJ, Kay SA. A genomic analysis of the shade avoidance response in Arabidopsis. Plant Physiol. 2003;133:1617–1629. doi: 10.1104/pp.103.034397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Salisbury FJ, Hall A, Grierson CS, Halliday KJ. Phytochrome coordinates Arabidopsis shoot and root development. Plant J. 2007;50:429–438. doi: 10.1111/j.1365-313X.2007.03059.x. [DOI] [PubMed] [Google Scholar]
- 12.Tao Y, et al. Rapid synthesis of auxin via a new tryptophan-dependent pathway is required for shade avoidance in plants. Cell. 2008;133:164–176. doi: 10.1016/j.cell.2008.01.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stepanova AN, et al. TAA1-mediated auxin biosynthesis is essential for hormone crosstalk and plant development. Cell. 2008;133:177–191. doi: 10.1016/j.cell.2008.01.047. [DOI] [PubMed] [Google Scholar]
- 14.Blilou I, et al. The PIN auxin efflux facilitator network controls growth and patterning in Arabidopsis roots. Nature. 2005;433:39–44. doi: 10.1038/nature03184. [DOI] [PubMed] [Google Scholar]
- 15.Wisniewska J, et al. Polar PIN localization directs auxin flow in plants. Science. 2006;312 doi: 10.1126/science.1121356. [DOI] [PubMed] [Google Scholar]
- 16.Friml J, Wiśniewska J, Benková E, Mendgen K, Palme K. Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature. 2002;415:806–809. doi: 10.1038/415806a. [DOI] [PubMed] [Google Scholar]
- 17.Blakeslee JJ, Bandyopadhyay A, Peer WA, Makam SN, Murphy AS. Relocalization of the PIN1 auxin efflux facilitator plays a role in phototropic responses. Plant Physiol. 2004;134:28–31. doi: 10.1104/pp.103.031690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dharmasiri N, Dharmasiri S, Estelle M. The F-box protein TIR1 is an auxin receptor. Nature. 2005;435:441–445. doi: 10.1038/nature03543. [DOI] [PubMed] [Google Scholar]
- 19.Kepinski S, Leyser O. The Arabidopsis F-box protein TIR1 is an auxin receptor. Nature. 2005;435:446–451. doi: 10.1038/nature03542. [DOI] [PubMed] [Google Scholar]
- 20.Hayashi K, et al. Small-molecule agonists and antagonists of F-box protein-substrate interactions in auxin perception and signaling. Proc Natl Acad Sci USA. 2008;105:5632–5637. doi: 10.1073/pnas.0711146105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Petrásek J, et al. Do phytotropins inhibit auxin efflux by impairing vesicle traffic? Plant Physiol. 2003;131:254–263. doi: 10.1104/pp.012740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tatematsu K, et al. MASSUGU2 encodes Aux/IAA19, an auxin-regulated protein that functions together with the transcriptional activator NPH4/ARF7 to regulate differential growth responses of hypocotyl and formation of lateral roots in Arabidopsis thaliana. Plant Cell. 2004;16:379–393. doi: 10.1105/tpc.018630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Friml J, et al. A PINOID-dependent binary switch in apical-basal PIN polar targeting directs auxin efflux. Science. 2004;306:862–865. doi: 10.1126/science.1100618. [DOI] [PubMed] [Google Scholar]
- 24.Paciorek T, et al. Auxin inhibits endocytosis and promotes its own efflux from cells. Nature. 2005;435:1251–1256. doi: 10.1038/nature03633. [DOI] [PubMed] [Google Scholar]
- 25.Sauer M, et al. Canalization of auxin flow by Aux/IAA-ARF-dependent feedback regulation of PIN polarity. Genes Dev. 2006;20:2902–2911. doi: 10.1101/gad.390806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vieten A, et al. Functional redundancy of PIN proteins is accompanied by auxin-dependent cross-regulation of PIN expression. Development. 2005;132:4521–4531. doi: 10.1242/dev.02027. [DOI] [PubMed] [Google Scholar]
- 27.Kleine-Vehn J, et al. PIN auxin efflux carrier polarity is regulated by PINOID kinase-mediated recruitment into GNOM-independent trafficking in Arabidopsis. Plant Cell. 2009;21:3839–3849. doi: 10.1105/tpc.109.071639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang F, et al. Phosphorylation of conserved PIN motifs directs Arabidopsis PIN1 polarity and auxin transport. Plant Cell. 2010;22:1129–1142. doi: 10.1105/tpc.109.072678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang J, Nodzynski T, Pencík A, Rolcík J, Friml J. PIN phosphorylation is sufficient to mediate PIN polarity and direct auxin transport. Proc Natl Acad Sci USA. 2010;107:918–922. doi: 10.1073/pnas.0909460107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Djakovic-Petrovic T, de Wit M, Voesenek LACJ, Pierik R. DELLA protein function in growth responses to canopy signals. Plant J. 2007;51:117–126. doi: 10.1111/j.1365-313X.2007.03122.x. [DOI] [PubMed] [Google Scholar]
- 31.Pierik R, Whitelam GC, Voesenek LACJ, de Kroon H, Visser EJW. Canopy studies on ethylene-insensitive tobacco identify ethylene as a novel element in blue light and plant-plant signalling. Plant J. 2004;38:310–319. doi: 10.1111/j.1365-313X.2004.02044.x. [DOI] [PubMed] [Google Scholar]
- 32.van Zanten M, et al. Auxin perception and polar auxin transport are not always a prerequisite for differential growth. Plant Signal Behav. 2009;4:899–901. doi: 10.4161/psb.4.9.9528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Savaldi-Goldstein S, Peto C, Chory J. The epidermis both drives and restricts plant shoot growth. Nature. 2007;446:199–202. doi: 10.1038/nature05618. [DOI] [PubMed] [Google Scholar]
- 34.Ruegger M, et al. The TIR1 protein of Arabidopsis functions in auxin response and is related to human SKP2 and yeast grr1p. Genes Dev. 1998;12:198–207. doi: 10.1101/gad.12.2.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Friml J, et al. AtPIN4 mediates sink-driven auxin gradients and root patterning in Arabidopsis. Cell. 2002;108:661–673. doi: 10.1016/s0092-8674(02)00656-6. [DOI] [PubMed] [Google Scholar]
- 36.Zádníková P, et al. Role of PIN-mediated auxin efflux in apical hook development of Arabidopsis thaliana. Development. 2010;137:607–617. doi: 10.1242/dev.041277. [DOI] [PubMed] [Google Scholar]
- 37.Müller A, Düchting P, Weiler EW. A multiplex GC-MS/MS technique for the sensitive and quantitative single-run analysis of acidic phytohormones and related compounds, and its application to Arabidopsis thaliana. Planta. 2002;216:44–56. doi: 10.1007/s00425-002-0866-6. [DOI] [PubMed] [Google Scholar]
- 38.Pierik R, Visser EJW, de Kroon H, Voesenek LACJ. Ethylene is required in tobacco to successfully compete with proximate neighbours. Plant Cell Environ. 2003;26:1229–1234. [Google Scholar]
- 39.Schmitt J, McCormac AC, Smith H. A test of the adaptive plasticity hypothesis using transgenic and mutant plants disabled in phytochrome-mediated elongation responses to neighbors. Am Nat. 1995;146:937–953. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.