Abstract
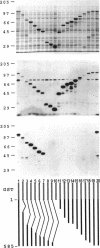
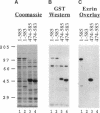
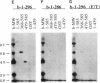
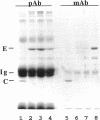
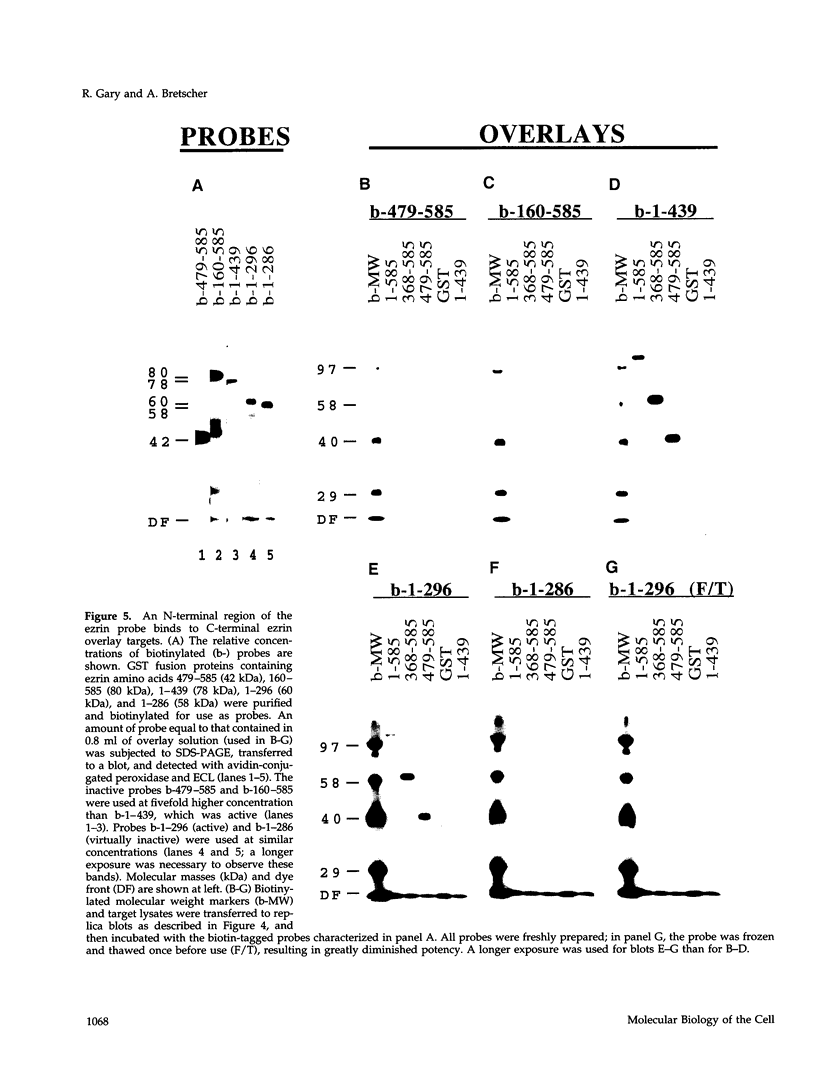
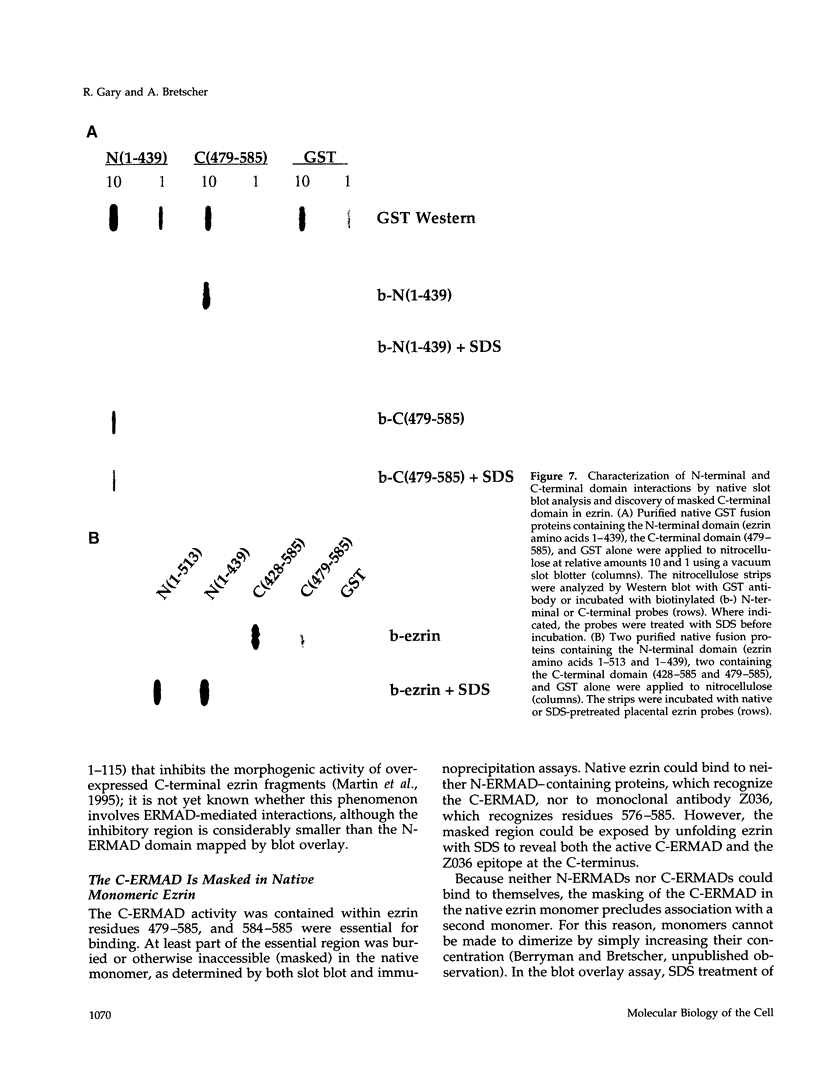
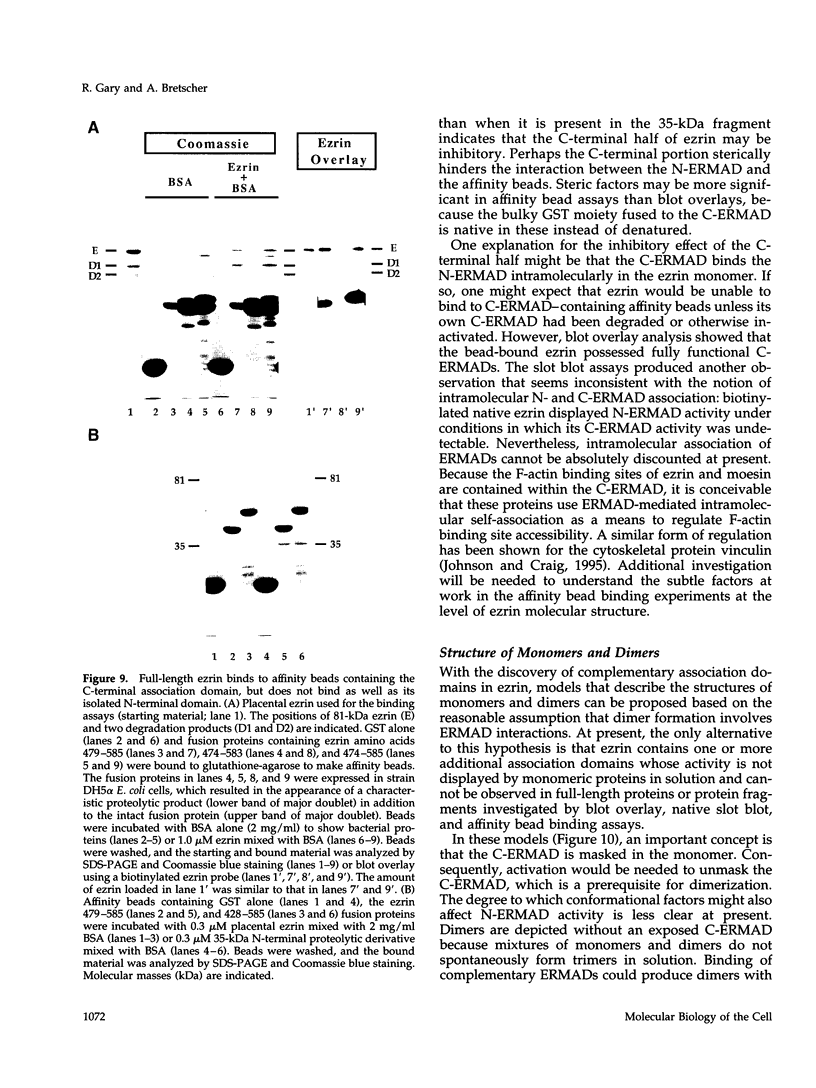
Ezrin is a membrane-cytoskeletal linking protein that is concentrated in actin-rich surface structures. It is closely related to the microvillar proteins radixin and moesin and to the tumor suppressor merlin/schwannomin. Cell extracts contain ezrin dimers and ezrin-moesin heterodimers in addition to monomers. Truncated ezrin fusion proteins were assayed by blot overlay to determine which regions mediate self-association. Here we report that ezrin self-association occurs by head-to-tail joining of distinct N-terminal and C-terminal domains. It is likely that these domains, termed N- and C-ERMADs (ezrin-radixin-moesin association domain), are responsible for homotypic and heterotypic associations among ERM family members. The N-ERMAD of ezrin resided within amino acids 1-296; deletion of 10 additional residues resulted in loss of activity. The C-ERMAD was mapped to the last 107 amino acids of ezrin, residues 479-585. The two residues at the C-terminus were required for activity, and the region from 530-585 was insufficient. The C-ERMAD was masked in the native monomer. Exposure of this domain required unfolding ezrin with sodium dodecyl sulfate or expressing the domain as part of a truncated protein. Intermolecular association could not occur unless the C-ERMAD had been made accessible to its N-terminal partner. It can be inferred that dimerization in vivo requires an activation step that exposes this masked domain. The conformationally inaccessible C-terminal region included the F-actin binding site, suggesting that this activity is likewise regulated by masking.
Full text
PDF



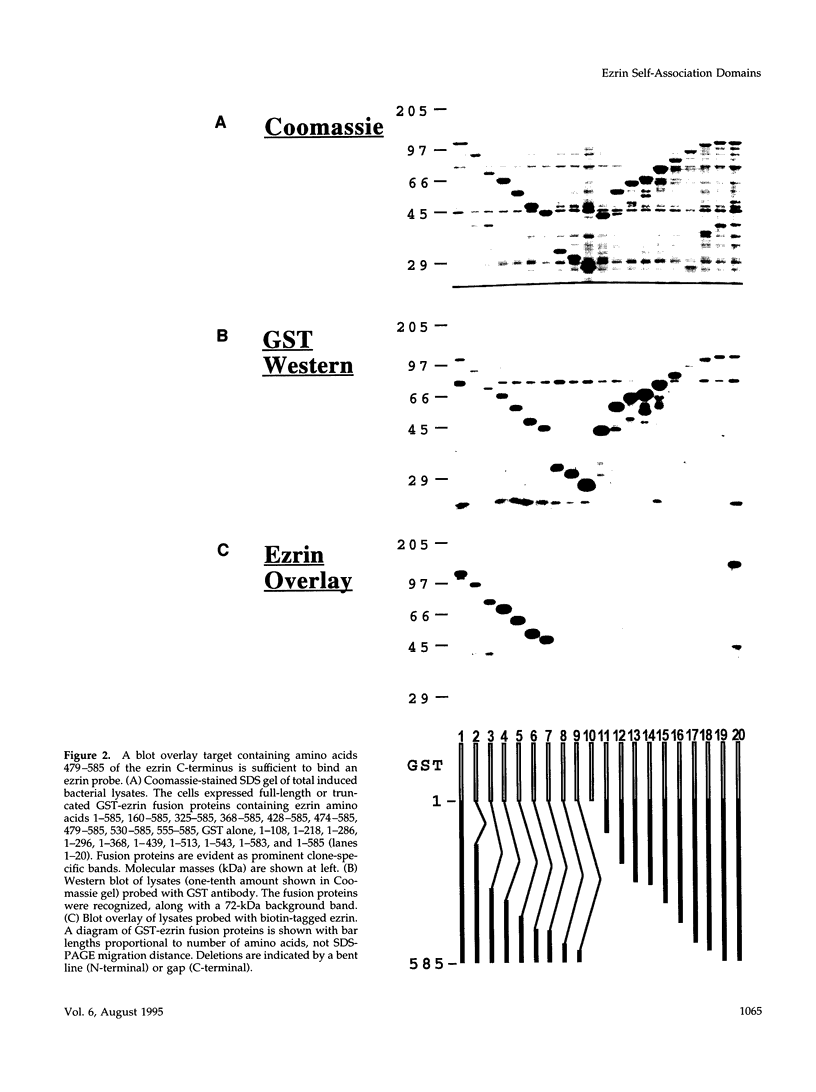
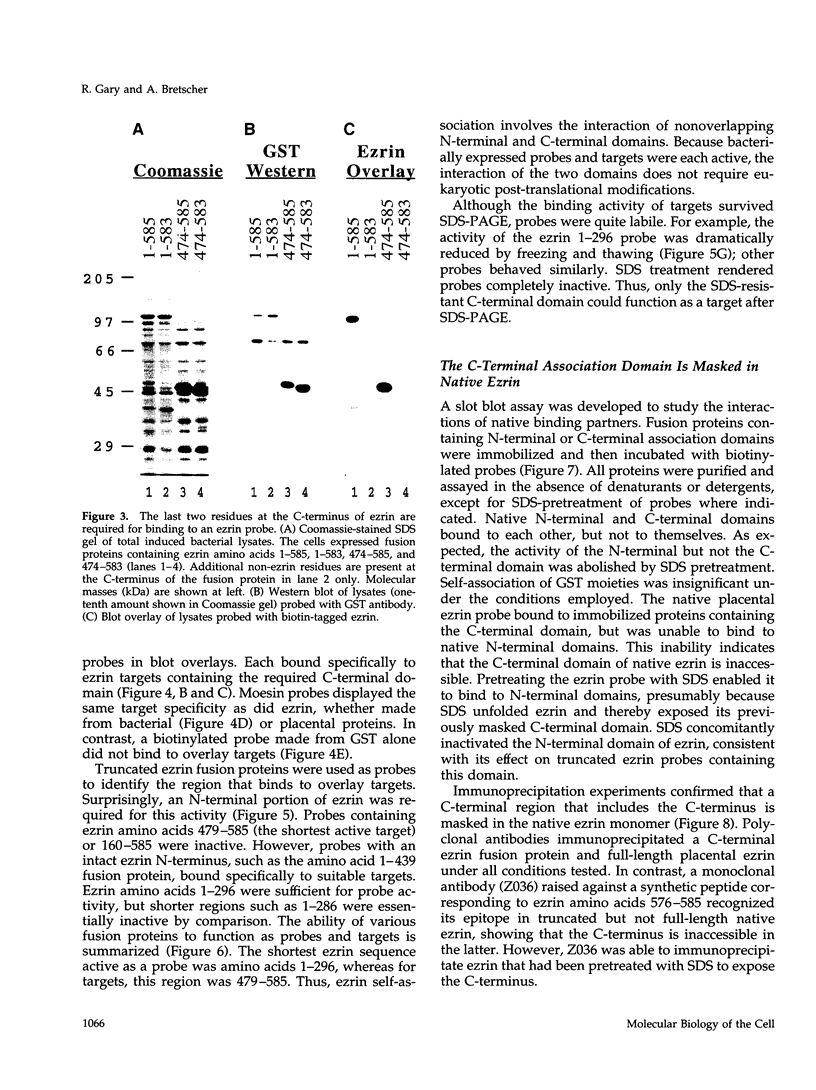
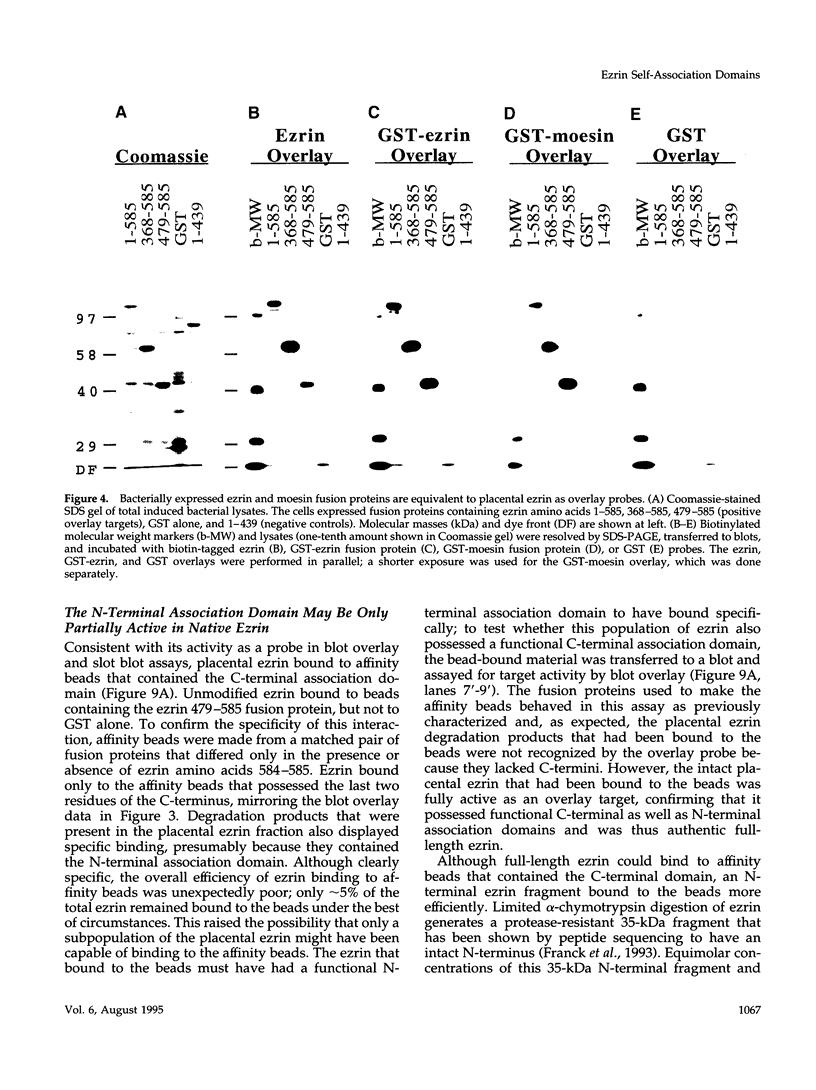





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Algrain M., Turunen O., Vaheri A., Louvard D., Arpin M. Ezrin contains cytoskeleton and membrane binding domains accounting for its proposed role as a membrane-cytoskeletal linker. J Cell Biol. 1993 Jan;120(1):129–139. doi: 10.1083/jcb.120.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amieva M. R., Wilgenbus K. K., Furthmayr H. Radixin is a component of hepatocyte microvilli in situ. Exp Cell Res. 1994 Jan;210(1):140–144. doi: 10.1006/excr.1994.1021. [DOI] [PubMed] [Google Scholar]
- Andréoli C., Martin M., Le Borgne R., Reggio H., Mangeat P. Ezrin has properties to self-associate at the plasma membrane. J Cell Sci. 1994 Sep;107(Pt 9):2509–2521. doi: 10.1242/jcs.107.9.2509. [DOI] [PubMed] [Google Scholar]
- Berryman M., Franck Z., Bretscher A. Ezrin is concentrated in the apical microvilli of a wide variety of epithelial cells whereas moesin is found primarily in endothelial cells. J Cell Sci. 1993 Aug;105(Pt 4):1025–1043. doi: 10.1242/jcs.105.4.1025. [DOI] [PubMed] [Google Scholar]
- Bianchi A. B., Hara T., Ramesh V., Gao J., Klein-Szanto A. J., Morin F., Menon A. G., Trofatter J. A., Gusella J. F., Seizinger B. R. Mutations in transcript isoforms of the neurofibromatosis 2 gene in multiple human tumour types. Nat Genet. 1994 Feb;6(2):185–192. doi: 10.1038/ng0294-185. [DOI] [PubMed] [Google Scholar]
- Bretscher A. Microfilament structure and function in the cortical cytoskeleton. Annu Rev Cell Biol. 1991;7:337–374. doi: 10.1146/annurev.cb.07.110191.002005. [DOI] [PubMed] [Google Scholar]
- Bretscher A. Purification of an 80,000-dalton protein that is a component of the isolated microvillus cytoskeleton, and its localization in nonmuscle cells. J Cell Biol. 1983 Aug;97(2):425–432. doi: 10.1083/jcb.97.2.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bretscher A. Rapid phosphorylation and reorganization of ezrin and spectrin accompany morphological changes induced in A-431 cells by epidermal growth factor. J Cell Biol. 1989 Mar;108(3):921–930. doi: 10.1083/jcb.108.3.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egerton M., Burgess W. H., Chen D., Druker B. J., Bretscher A., Samelson L. E. Identification of ezrin as an 81-kDa tyrosine-phosphorylated protein in T cells. J Immunol. 1992 Sep 15;149(6):1847–1852. [PubMed] [Google Scholar]
- Fabian J. R., Vojtek A. B., Cooper J. A., Morrison D. K. A single amino acid change in Raf-1 inhibits Ras binding and alters Raf-1 function. Proc Natl Acad Sci U S A. 1994 Jun 21;91(13):5982–5986. doi: 10.1073/pnas.91.13.5982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fazioli F., Wong W. T., Ullrich S. J., Sakaguchi K., Appella E., Di Fiore P. P. The ezrin-like family of tyrosine kinase substrates: receptor-specific pattern of tyrosine phosphorylation and relationship to malignant transformation. Oncogene. 1993 May;8(5):1335–1345. [PubMed] [Google Scholar]
- Franck Z., Gary R., Bretscher A. Moesin, like ezrin, colocalizes with actin in the cortical cytoskeleton in cultured cells, but its expression is more variable. J Cell Sci. 1993 May;105(Pt 1):219–231. doi: 10.1242/jcs.105.1.219. [DOI] [PubMed] [Google Scholar]
- Frosch P. M., Frosch M., Pfister T., Schaad V., Bitter-Suermann D. Cloning and characterisation of an immunodominant major surface antigen of Echinococcus multilocularis. Mol Biochem Parasitol. 1991 Oct;48(2):121–130. doi: 10.1016/0166-6851(91)90108-i. [DOI] [PubMed] [Google Scholar]
- Funayama N., Nagafuchi A., Sato N., Tsukita S., Tsukita S. Radixin is a novel member of the band 4.1 family. J Cell Biol. 1991 Nov;115(4):1039–1048. doi: 10.1083/jcb.115.4.1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gary R., Bretscher A. Heterotypic and homotypic associations between ezrin and moesin, two putative membrane-cytoskeletal linking proteins. Proc Natl Acad Sci U S A. 1993 Nov 15;90(22):10846–10850. doi: 10.1073/pnas.90.22.10846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goslin K., Birgbauer E., Banker G., Solomon F. The role of cytoskeleton in organizing growth cones: a microfilament-associated growth cone component depends upon microtubules for its localization. J Cell Biol. 1989 Oct;109(4 Pt 1):1621–1631. doi: 10.1083/jcb.109.4.1621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gould K. L., Bretscher A., Esch F. S., Hunter T. cDNA cloning and sequencing of the protein-tyrosine kinase substrate, ezrin, reveals homology to band 4.1. EMBO J. 1989 Dec 20;8(13):4133–4142. doi: 10.1002/j.1460-2075.1989.tb08598.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanzel D., Reggio H., Bretscher A., Forte J. G., Mangeat P. The secretion-stimulated 80K phosphoprotein of parietal cells is ezrin, and has properties of a membrane cytoskeletal linker in the induced apical microvilli. EMBO J. 1991 Sep;10(9):2363–2373. doi: 10.1002/j.1460-2075.1991.tb07775.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson R. P., Craig S. W. F-actin binding site masked by the intramolecular association of vinculin head and tail domains. Nature. 1995 Jan 19;373(6511):261–264. doi: 10.1038/373261a0. [DOI] [PubMed] [Google Scholar]
- Jooss K. U., Müller R. Deregulation of genes encoding microfilament-associated proteins during Fos-induced morphological transformation. Oncogene. 1995 Feb 2;10(3):603–608. [PubMed] [Google Scholar]
- Kamps M. P., Buss J. E., Sefton B. M. Rous sarcoma virus transforming protein lacking myristic acid phosphorylates known polypeptide substrates without inducing transformation. Cell. 1986 Apr 11;45(1):105–112. doi: 10.1016/0092-8674(86)90542-8. [DOI] [PubMed] [Google Scholar]
- Lankes W. T., Furthmayr H. Moesin: a member of the protein 4.1-talin-ezrin family of proteins. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8297–8301. doi: 10.1073/pnas.88.19.8297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leevers S. J., Paterson H. F., Marshall C. J. Requirement for Ras in Raf activation is overcome by targeting Raf to the plasma membrane. Nature. 1994 Jun 2;369(6479):411–414. doi: 10.1038/369411a0. [DOI] [PubMed] [Google Scholar]
- Leto T. L., Marchesi V. T. A structural model of human erythrocyte protein 4.1. J Biol Chem. 1984 Apr 10;259(7):4603–4608. [PubMed] [Google Scholar]
- Marshall C. J. Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell. 1995 Jan 27;80(2):179–185. doi: 10.1016/0092-8674(95)90401-8. [DOI] [PubMed] [Google Scholar]
- Martin M., Andréoli C., Sahuquet A., Montcourrier P., Algrain M., Mangeat P. Ezrin NH2-terminal domain inhibits the cell extension activity of the COOH-terminal domain. J Cell Biol. 1995 Mar;128(6):1081–1093. doi: 10.1083/jcb.128.6.1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick F. Raf: the holy grail of Ras biology? Trends Cell Biol. 1994 Oct;4(10):347–350. doi: 10.1016/0962-8924(94)90075-2. [DOI] [PubMed] [Google Scholar]
- Okayama H., Berg P. High-efficiency cloning of full-length cDNA. Mol Cell Biol. 1982 Feb;2(2):161–170. doi: 10.1128/mcb.2.2.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pakkanen R., Hedman K., Turunen O., Wahlström T., Vaheri A. Microvillus-specific Mr 75,000 plasma membrane protein of human choriocarcinoma cells. J Histochem Cytochem. 1987 Aug;35(8):809–816. doi: 10.1177/35.8.3298422. [DOI] [PubMed] [Google Scholar]
- Pakkanen R., Vaheri A. Cytovillin and other microvillar proteins of human choriocarcinoma cells. J Cell Biochem. 1989 Sep;41(1):1–12. doi: 10.1002/jcb.240410102. [DOI] [PubMed] [Google Scholar]
- Pestonjamasp K., Amieva M. R., Strassel C. P., Nauseef W. M., Furthmayr H., Luna E. J. Moesin, ezrin, and p205 are actin-binding proteins associated with neutrophil plasma membranes. Mol Biol Cell. 1995 Mar;6(3):247–259. doi: 10.1091/mbc.6.3.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rouleau G. A., Merel P., Lutchman M., Sanson M., Zucman J., Marineau C., Hoang-Xuan K., Demczuk S., Desmaze C., Plougastel B. Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature. 1993 Jun 10;363(6429):515–521. doi: 10.1038/363515a0. [DOI] [PubMed] [Google Scholar]
- Sato N., Funayama N., Nagafuchi A., Yonemura S., Tsukita S., Tsukita S. A gene family consisting of ezrin, radixin and moesin. Its specific localization at actin filament/plasma membrane association sites. J Cell Sci. 1992 Sep;103(Pt 1):131–143. doi: 10.1242/jcs.103.1.131. [DOI] [PubMed] [Google Scholar]
- Shuster C. B., Herman I. M. Indirect association of ezrin with F-actin: isoform specificity and calcium sensitivity. J Cell Biol. 1995 Mar;128(5):837–848. doi: 10.1083/jcb.128.5.837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi K., Kawashima A., Nagafuchi A., Tsukita S. Structural diversity of band 4.1 superfamily members. J Cell Sci. 1994 Jul;107(Pt 7):1921–1928. doi: 10.1242/jcs.107.7.1921. [DOI] [PubMed] [Google Scholar]
- Takeuchi K., Sato N., Kasahara H., Funayama N., Nagafuchi A., Yonemura S., Tsukita S., Tsukita S. Perturbation of cell adhesion and microvilli formation by antisense oligonucleotides to ERM family members. J Cell Biol. 1994 Jun;125(6):1371–1384. doi: 10.1083/jcb.125.6.1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thuillier L., Hivroz C., Fagard R., Andreoli C., Mangeat P. Ligation of CD4 surface antigen induces rapid tyrosine phosphorylation of the cytoskeletal protein ezrin. Cell Immunol. 1994 Jul;156(2):322–331. doi: 10.1006/cimm.1994.1178. [DOI] [PubMed] [Google Scholar]
- Tikoo A., Varga M., Ramesh V., Gusella J., Maruta H. An anti-Ras function of neurofibromatosis type 2 gene product (NF2/Merlin). J Biol Chem. 1994 Sep 23;269(38):23387–23390. [PubMed] [Google Scholar]
- Trofatter J. A., MacCollin M. M., Rutter J. L., Murrell J. R., Duyao M. P., Parry D. M., Eldridge R., Kley N., Menon A. G., Pulaski K. A novel moesin-, ezrin-, radixin-like gene is a candidate for the neurofibromatosis 2 tumor suppressor. Cell. 1993 Mar 12;72(5):791–800. doi: 10.1016/0092-8674(93)90406-g. [DOI] [PubMed] [Google Scholar]
- Tsukita S., Oishi K., Sato N., Sagara J., Kawai A., Tsukita S. ERM family members as molecular linkers between the cell surface glycoprotein CD44 and actin-based cytoskeletons. J Cell Biol. 1994 Jul;126(2):391–401. doi: 10.1083/jcb.126.2.391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turunen O., Wahlström T., Vaheri A. Ezrin has a COOH-terminal actin-binding site that is conserved in the ezrin protein family. J Cell Biol. 1994 Sep;126(6):1445–1453. doi: 10.1083/jcb.126.6.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turunen O., Winqvist R., Pakkanen R., Grzeschik K. H., Wahlström T., Vaheri A. Cytovillin, a microvillar Mr 75,000 protein. cDNA sequence, prokaryotic expression, and chromosomal localization. J Biol Chem. 1989 Oct 5;264(28):16727–16732. [PubMed] [Google Scholar]
- Ullrich S. J., Robinson E. A., Appella E. Characterization of a chemically homogeneous tumor antigen from a methylcholanthrene-induced sarcoma, Meth A. Mol Immunol. 1986 May;23(5):545–555. doi: 10.1016/0161-5890(86)90118-5. [DOI] [PubMed] [Google Scholar]
- Williams N. G., Roberts T. M., Li P. Both p21ras and pp60v-src are required, but neither alone is sufficient, to activate the Raf-1 kinase. Proc Natl Acad Sci U S A. 1992 Apr 1;89(7):2922–2926. doi: 10.1073/pnas.89.7.2922. [DOI] [PMC free article] [PubMed] [Google Scholar]