Abstract
Tetrachloroethylene is a chlorinated solvent that is primarily used in dry cleaning and degreasing operations. Although the hepatotoxicity caused by tetrachloroethylene has been well documented in literature, it is rarely considered as a cause of acute liver failure. We report a case of a 39-yr-old man who was admitted to our hospital for acute liver failure due to tetrachloroethylene exposure. Histological examination of the liver revealed massive hepatic necrosis, prominently, in zone 3 of the hepatic lobules. The patient underwent supportive treatment along with 3 sessions of plasmapheresis, and consequently, he presented a favorable outcome. Repeat liver biopsy performed 6 months after the patient's discharge showed architectural distortion with postnecrotic cirrhosis. Physicians should be aware of the possibility of acute liver failure induced by tetrachloroethylene. Early plasmapheresis can be effective for individuals with sufficient capacity for hepatocyte regeneration.
Keywords: Drug-induced Liver Injury; Liver Failure, Acute; Plasma Exchange; Tetrachloroethylene
INTRODUCTION
Tetrachloroethylene or perchloroethylene (PCE), a common industrial solvent and environmental contaminant, is primarily used in the dry-cleaning industry, for metal degreasing operations, and in the textile industry (1, 2). It is also present in certain consumer products, including paint strippers, spot removers, silicone lubricants, and food (2). Studies have provided clear evidence that both acute and chronic exposure to PCE can cause numerous adverse effects. As in the cases of toxicity due to the exposure to other chlorinated solvents, the target organs for PCE toxicity in humans are the central nervous system, liver, and kidneys. The documented symptoms of toxicity due to the exposure to PCE include headache, dizziness, fatigue, nausea, vomiting, pulmonary edema, and signs of impaired hepatic or renal function, depending on the level of PCE exposure (2-4). Severe exposure to PCE can induce behavior alteration, coma, and death (2, 4). In addition, PCE causes irritation in the eyes, nose, and upper respiratory tract (4). Moreover, the International Agency for Research on Cancer classified PCE as a potential human carcinogen (5). Although hepatotoxic effects have been reported in humans after heavy exposure to PCE, acute liver failure has not been previously reported.
We report, for the first time, a case of successful treatment of PCE-induced acute liver failure with plasmapheresis. We also describe the pathological features of the condition on the basis of initial and repeat liver biopsies.
CASE DESCRIPTION
A 39-yr-old male patient was admitted to our liver intensive care unit (LICU) for acute liver failure on August 20, 2009. The patient worked at a leather-manufacturing plant, where he cleaned animal coats by using PCE. The patient informed us that he occasionally did not wear protective devices during this procedure and that the process room at the plant was poorly ventilated. After working for about 50 days, the patient suddenly began passing dark urine, but continued his regular work. During the next 4 days, the patient developed progressive jaundice with nausea, vomiting, loss of appetite, and weakness and was admitted to a local hospital. Blood chemistry revealed the following findings: total bilirubin (TBIL), 202.8 µM/L (normal range, 3.0-20.0 µM/L); alanine aminotransferase (ALT), 743 U/L (normal range, 5-40 U/L); alkaline phosphatase (ALP), 189 U/L (normal range, 15-130 U/L); and gamma glutamyl transferase (GGT), 99 U/L (normal range, 12-58 U/L). Serum concentration of PCE was 2.8 mg/L and urinary concentration of trichloroacetic acid (TCA)-an indirect biomarker for PCE poisoning-was 8.4 mg/L. Treatment with hepatoprotective agents failed to achieve any remission. Further, the patient developed coagulopathy associated with progressive deterioration of liver function, which in turn led to the development of grade II hepatic encephalopathy within 48 hr. Therefore, he was transferred to our hospital.
The patient had no notable exposure to toxic chemicals before this job. He denied smoking or alcohol or drug use before the onset of the symptoms. The patient had not received blood transfusion; not traveled recently; had no family history of liver disease; and none of his colleagues had similar manifestations. On the day of admission to the LICU, all his vital signs were within normal limits. Physical examination revealed grade II hepatic encephalopathy with confusion, slurred speech, asterixis, and disorientation for time and place. The patient had severe jaundice, but without peripheral edema. The patient developed ecchymoses at the therapeutic puncture sites. The liver, spleen, and kidneys were not palpable. Abdominal or costovertebral angle tenderness was not detected.
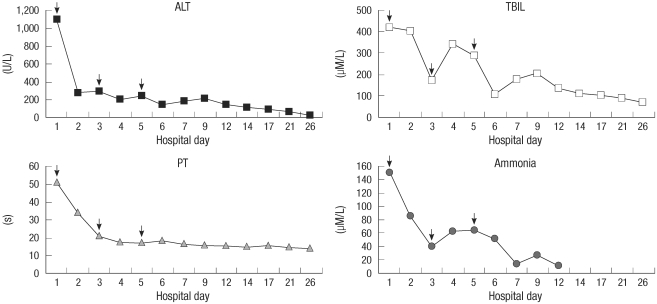
Laboratory examinations performed at the time of admission showed the following biochemical results: ALT, 1,100 U/L; aspartate aminotransferase (AST), 355 U/L (normal range, 10-40 U/L); TBIL, 421.8 µM/L; direct serum bilirubin (DBIL), 197.3 µM/L (normal range, 0.0-7.0 µM/L); ALP, 201 U/L; GGT, 139 U/L; prothrombin time (PT) 51.0 s (normal range, 9.0-12.8 s); international normalized ratio (INR), 3.32 (normal range, 0.8-1.4); ammonia, 151 µM/L (normal range, 9-33 µM/L); alfa-fetoprotein (AFP), 1,045 ng/mL (normal range, 0-9 ng/mL); and normal electrolyte, hematological, and renal functions. Peripheral T-lymphocyte subpopulations, analyzed by flow cytometry, showed CD4/CD8 ratio of 3.74 (normal range, 1.33-1.99). Serological studies for viral hepatitis (hepatitis A, B, C, D, and E viruses; human immunodeficiency virus; Epstein-Barr virus; cytomegalovirus) and autoimmunity markers (antinuclear, antimitochondrial, antineutrophil cytoplasmic, anti-smooth-muscle, and anti-liver-kidney microsome antibodies) were negative. Screening studies were performed to detect genetic and metabolic liver diseases and hematochromatosis, Wilson's disease, and alpha 1-antitrypsin deficiency were excluded. Doppler ultrasonography of the patient's abdomen revealed homogeneous echogenic liver that had decreased in size; had patent portal veins, hepatic artery, and hepatic veins; and no obstruction of the biliary tracts. The spleen appeared normal, and no ascites was observed.
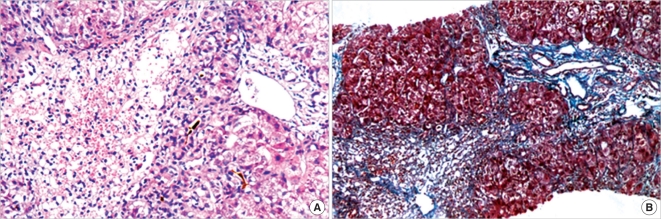
The patient was managed with general supportive care along with intravenous administration of dexamethasone (15 mg daily) for 5 consecutive days. He was also treated with liver-protective preparations, glutathione, ademetionine, hepatocyte growth-promoting factors, and prophylactic antibiotics. Furthermore, plasmapheresis was performed on days 1, 3, and 5, with 3,000 mL of plasma exchanged during each session. Gradually, the patient's mental status became normal, and liver function, coagulation profiles and other biochemical variables improved notably (Fig. 1). The maximum AFP level was 3,000 ng/mL. The patient underwent percutaneous liver biopsy on day 12; the biopsy revealed massive hepatocellular necrosis prominently in the centrilobular area with moderate inflammatory infiltration mainly of lymphocytes and neutrophils. We also detected diffuse swelling of the hepatocytes and hemorrhagic infiltration in some necrotic areas. Neither obvious expansion of portal vein nor increased deposition of fibrous tissue was detected. Bile ducts appeared normal except for cholangioles with bile plugs (Fig. 2). The pathological features indicated drug-induced acute liver injury. The patient responded well to treatment, and gradually, his general condition improved and all symptoms eventually resolved except for poor appetite and fatigue. The patient was discharged from the hospital on day 26 and advised to completely avoid exposure to PCE at his workplace.
Fig. 1.
Levels of biochemical parameters during hospitalization.
Fig. 2.
Initial liver biopsy performed on day 12 of hospitalization. (A) Massive hepatocellular necrosis was mainly located in zone 3 with moderately inflammatory infiltration (Hematoxylin and eosin staining × 200). (B) No obvious deposition of fibrous tissue observed within portal tracts (Masson's trichrome staining × 100).
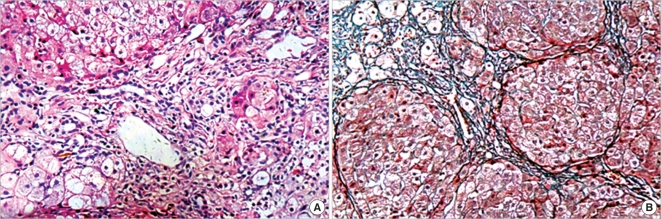
The patient underwent regular follow-up every month, and liver function and AFP level became normal after 2 months. Repeat liver biopsy performed 6 months after discharge showed fibrous expansion of the portal tracts caused by moderate inflammatory infiltration and nodular regeneration of the hepatic parenchyma delimited by thin fibrous bands and septa. The biopsy showed evidence of some swollen hepatocytes and scattered foci of hepatocellular necrosis within the liver lobules (Fig. 3). Currently, the patient is undergoing anti-liver fibrosis therapy using modern Chinese medicine (Fuzhenghuayu capsule) (6) and continues to receive follow-up.
Fig. 3.
Repeat liver biopsy performed 6 months after discharge. (A) Distortion of lobular structure of the liver accompanied by scattered foci of hepatocellular necrosis within lobules and lymphocytic piecemeal necrosis (Hematoxylin and eosin staining × 200). (B) Expanded portal tracts with fibrous septa indicative of postnecrotic hepatic cirrhosis (Masson's trichrome staining × 200).
DISCUSSION
PCE is mainly absorbed by mammals following inhalation and, to a lesser extent, by ingestion and dermal exposure (2, 4). PCE is highly soluble in adipose tissues. Most of the absorbed PCE (80%-90%) is excreted unchanged in expired air, regardless of the route of exposure, whereas only a small proportion (1%-3%) is metabolized to TCA, trichloroethanol, and trichloroacetyl chloride, which were subsequently eliminated in urine (2, 7). The serum half-life of PCE is estimated at 65 hr, and that of urinary TCA at 144 hr (8, 9). Biotransformation of PCE occurs predominantly in the liver via cytochrome P-450 (CYP) oxidation pathway, which accounts for the highest percentage of metabolites (2). Glutathione conjugation pathway, though a minor route, is activated when CYP oxidation is saturated (2, 10).
The mechanism of PCE-induced hepatotoxicity has not yet been well elucidated. There is evidence that TCA, the principal metabolite of PCE, derived from CYP pathway may mainly contribute to hepatotoxicity via several modes of action, including oxidative stress, mitochondrial dysfunction, and DNA damage (11). Few studies have indicated that PCE may exert suppressive effects on the immune system (12). However, our patient showed a remarkably high CD4/CD8 ratio at the initial presentation, indicating the possibility of a PCE-triggered cell-mediated immune response that might induce hepatocellular damage. In addition, the hepatic injury in our patient was more severe than those reported in patients in previous studies, and no abnormalities were detected in his colleagues who were exposed to the same conditions at work. We postulated that these differences may be attributable to genetic polymorphism in metabolizing enzymes, which might lead to variations among the level of susceptibility of individual to PCE toxicity (11). Other factors such as age, sex, pattern of activity, health status, and alcohol consumption also influence PCE metabolism and toxicity.
Several studies have reported PCE-induced hepatic abnormalities in patients. Meckler et al. (13) reported hepatocellular injury detected by liver biopsy in a woman with acute hepatitis after exposure to PCE. Bagnell et al. (14) reported a case of an infant presenting with obstructive jaundice and hepatomegaly due to PCE exposure via breast milk. Brodkin et al. (15) reported hepatic ultrasonography findings showing diffuse parenchymal changes in dry cleaners in comparison with ultrasonography findings in control subjects. Liver damage in the form of hepatomegaly, jaundice, and elevation in levels of several hepatic transaminases and bilirubin has also been reported. In most cases, hepatotoxicity was transient, and its effects were spontaneously resolved when exposure to PCE ended. Thus far, no study has reported acute liver failure associated with PCE exposure.
Initially, the patient's condition was similar to viral hepatitis and rapidly deteriorated, resulting in marked liver dysfunction, coagulopathy, and encephalopathy. On the basis of the patient's occupational history, clinical manifestations, laboratory data, and liver biopsy findings, acute liver failure was attributable to occupational exposure to PCE, after other liver diseases had been completely excluded. We also assessed the patient using Maria and Victorino's clinical diagnostic scale for drug-induced liver injury, and on the basis of his score 14, he was judged "probable" (16). Liver injuries caused by drugs are classified as three types: hepatocellular, cholestatic, and mixed injury (17). Our case presented with notably increased ALT level of 1,100 U/L and an ALT/ALP ratio more than 5, suggesting a hepatocellular injury pattern.
Treatment for PCE-induced hepatotoxicity requires supportive care with close monitoring to prevent associated toxicities to other systemic organs. Our patient developed acute liver failure, which was a rare medical condition associated with high mortality. On the basis of Model for End-Stage Liver Disease (MELD) score of 30, the patient was a candidate for living-related orthotropic liver transplantation (18). However, the operation was postponed owing to shortage of donors. Plasmapheresis has been successfully used in management of acute or chronic liver failure, and it serves a treatment modality to achieve recovery of native liver function or until an appropriate donor is available. Although plasmapheresis has no established effects on elimination of serum PCE due to its relatively small molecular weight and short half time, it is an effective approach for removing various endogenous toxins and immune complexes from systemic circulation, decreasing serum aminotransferase level, and improving the encephalopathy stage and coagulation profiles (19). All these effects of plasmapheresis may create a better internal environment for native hepatocyte regeneration and change the final outcomes in liver failure. Our patient may have benefited from plasmapheresis, as was evident by the notable improvement of clinical status and liver function. Due to the adjuvant evidence of increase in the CD4/CD8 ratio, steroids were administered to control immune-mediated liver damage. Indeed, his survival can be mainly attributed to the sufficient residual hepatic capacity for regeneration, as evidenced by the short-term dramatically high levels of serum AFP, with maximum value being more than 150 times greater than the normal range.
Initial biopsy revealed massive hepatic necrosis, primarily, in zone 3, which was consistent with the fact that hepatocytes in zone 3 were probably more susceptible to chemical-induced hepatotoxicity owing to the high concentration of CYP in these cells. Furthermore, although his liver function test revealed persistently normal results for approximately 4 months, repeat biopsy revealed foci of hepatocellular necrosis and lymphocytic piecemeal necrosis with inflammatory infiltration. Therefore, since liver function test cannot completely reflect the severity of liver injury and may give a falsely optimistic suggestion, liver biopsy is a better approach to assess the prognosis of liver disease. Our patient's liver histology examination also showed typical postnecrotic cirrhosis, which was a result of active repair following massive hepatic necrosis.
Liver cirrhosis, in fact, is a rare sequel to drug-induced acute hepatitis, but it may occur if liver disease progresses to active chronic hepatitis, in which long-term liver lesions maintain. Moreover, a single episode of sufficiently severe injury, such as massive/submassive hepatic necrosis, can also cause liver cirrhosis due to collapse of lobular architecture and irregularly scarred repair. A rapid progression from severe hepatitis to cirrhosis, which is similar to that seen in our patient, has been documented in ebrotidine-induced liver injury with serial biopsy confirmation (20).
In conclusion, physicians should enhance their awareness of the possibility of acute liver failure induced by PCE exposure. Increased awareness and early diagnosis of the exposure to toxic substances are essential for performing prompt management, improving clinical outcomes. In addition, early plasmapheresis can serve as a useful treatment approach for patients with PCE-induced acute liver failure, particularly for those with sufficient hepatic capacity for hepatocyte regeneration.
References
- 1.Gold LS, De Roos AJ, Waters M, Stewart P. Systematic literature review of uses and levels of occupational exposure to tetrachloroethylene. J Occup Environ Hyg. 2008;5:807–839. doi: 10.1080/15459620802510866. [DOI] [PubMed] [Google Scholar]
- 2.Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological profile for tetrachloroethylene. Atlanta: U.S.: Department of Health and Human Services, Public Health Services; 1997. [Google Scholar]
- 3.Azimi Pirsaraei SR, Khavanin A, Asilian H, Soleimanian A. Occupational exposure to perchloroethylene in dry-cleaning shops in Tehran, Iran. Ind Health. 2009;47:155–159. doi: 10.2486/indhealth.47.155. [DOI] [PubMed] [Google Scholar]
- 4.Garnier R, Bédouin J, Pépin G, Gaillard Y. Coin-operated dry cleaning machines may be responsible for acute tetrachloroethylene poisoning: report of 26 cases including one death. J Toxicol Clin Toxicol. 1996;34:191–197. doi: 10.3109/15563659609013769. [DOI] [PubMed] [Google Scholar]
- 5.International Agency for Research on Cancer (IARC) IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Volume 63. Lyon, France: International Agency for Research on Cancer; 1995. Dry cleaning, some chlorinated solvents and other industrial compounds. [Google Scholar]
- 6.Liu P, Hu YY, Liu C, Xu LM, Liu CH, Sun KW, Hu DC, Yin YK, Zhou XQ, Wan MB, Cai X, Zhang ZQ, Ye J, Zhou RX, He J, Tang BZ. Multicenter clinical study on Fuzhenghuayu capsule against liver fibrosis due to chronic hepatitis B. World J Gastroenterol. 2005;11:2892–2899. doi: 10.3748/wjg.v11.i19.2892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Birner G, Richling C, Henschler D, Anders MW, Dekant W. Metabolism of tetrachloroethene in rats: identification of N epsilon-(dichloroacetyl)-L-lysine and N epsilon-(trichloroacetyl)-L-lysine as protein adducts. Chem Res Toxicol. 1994;7:724–732. doi: 10.1021/tx00042a003. [DOI] [PubMed] [Google Scholar]
- 8.Ikeda M. Metabolism of trichloroethylene and tetrachloroethylene in human subjects. Environ Health Perspect. 1977;21:239–245. doi: 10.1289/ehp.7721239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stewart RD, Baretta ED, Dodd HC, Torkelson TR. Experimental human exposure to tetrachloroethylene. Arch Environ Health. 1970;20:225–229. doi: 10.1080/00039896.1970.10665579. [DOI] [PubMed] [Google Scholar]
- 10.Dehon B, Humbert L, Devisme L, Stievenart M, Mathieu D, Houdret N, Lhermitte M. Tetrachloroethylene and trichloroethylene fatality: case report and simple headspace SPME-capillary gas chromatographic determination in tissues. J Anal Toxicol. 2000;24:22–26. doi: 10.1093/jat/24.1.22. [DOI] [PubMed] [Google Scholar]
- 11.Lash LH, Parker JC. Hepatic and renal toxicities associated with perchloroethylene. Pharmacol Rev. 2001;53:177–208. [PubMed] [Google Scholar]
- 12.Veraldi A, Costantini AS, Bolejack V, Miligi L, Vineis P, van Loveren H. Immunotoxic effects of chemicals: a matrix for occupational and environmental epidemiological studies. Am J Ind Med. 2006;49:1046–1055. doi: 10.1002/ajim.20364. [DOI] [PubMed] [Google Scholar]
- 13.Meckler LC, Phelps DK. Liver disease secondary to tetrachloroethylene exposure. A case report. JAMA. 1966;197:662–663. [PubMed] [Google Scholar]
- 14.Bagnell PC, Ellenberger HA. Obstructive jaundice due to a chlorinated hydrocarbon in breast milk. Can Med Assoc J. 1977;117:1047–1048. [PMC free article] [PubMed] [Google Scholar]
- 15.Brodkin CA, Daniell W, Checkoway H, Echeverria D, Johnson J, Wang K, Sohaey R, Green D, Redlich C, Gretch D. Hepatic ultrasonic changes in workers exposed to perchloroethylene. Occup Environ Med. 1995;52:679–685. doi: 10.1136/oem.52.10.679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maria VA, Victorino RM. Development and validation of a clinical scale for the diagnosis of drug-induced hepatitis. Hepatology. 1997;26:664–669. doi: 10.1002/hep.510260319. [DOI] [PubMed] [Google Scholar]
- 17.Yang HN, Kim DJ, Kim YM, Kim BH, Sohn KM, Choi MJ, Choi YH. Aloe-induced toxic hepatitis. J Korean Med Sci. 2010;25:492–495. doi: 10.3346/jkms.2010.25.3.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D'Amico G, Dickson ER, Kim WR. A model to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464–470. doi: 10.1053/jhep.2001.22172. [DOI] [PubMed] [Google Scholar]
- 19.Bektas M, Idilman R, Soykan I, Soydan E, Arat M, Cinar K, Coban S, Tuzun A, Bozkaya H, Ormeci N, Ozden A. Adjuvant therapeutic plasma exchange in liver failure: assessments of clinical and laboratory parameters. J Clin Gastroenterol. 2008;42:517–521. doi: 10.1097/MCG.0b013e31815878ff. [DOI] [PubMed] [Google Scholar]
- 20.Pineda JA, Larrauri J, Macias J, Hernandez A, Guijarro J, Sayago M, Gavilan F, Aguilar J, Lissen E. Rapid progression to liver cirrhosis of toxic hepatitis due to ebrotidine. J Hepatol. 1999;31:777–778. doi: 10.1016/s0168-8278(99)80362-6. [DOI] [PubMed] [Google Scholar]