Abstract
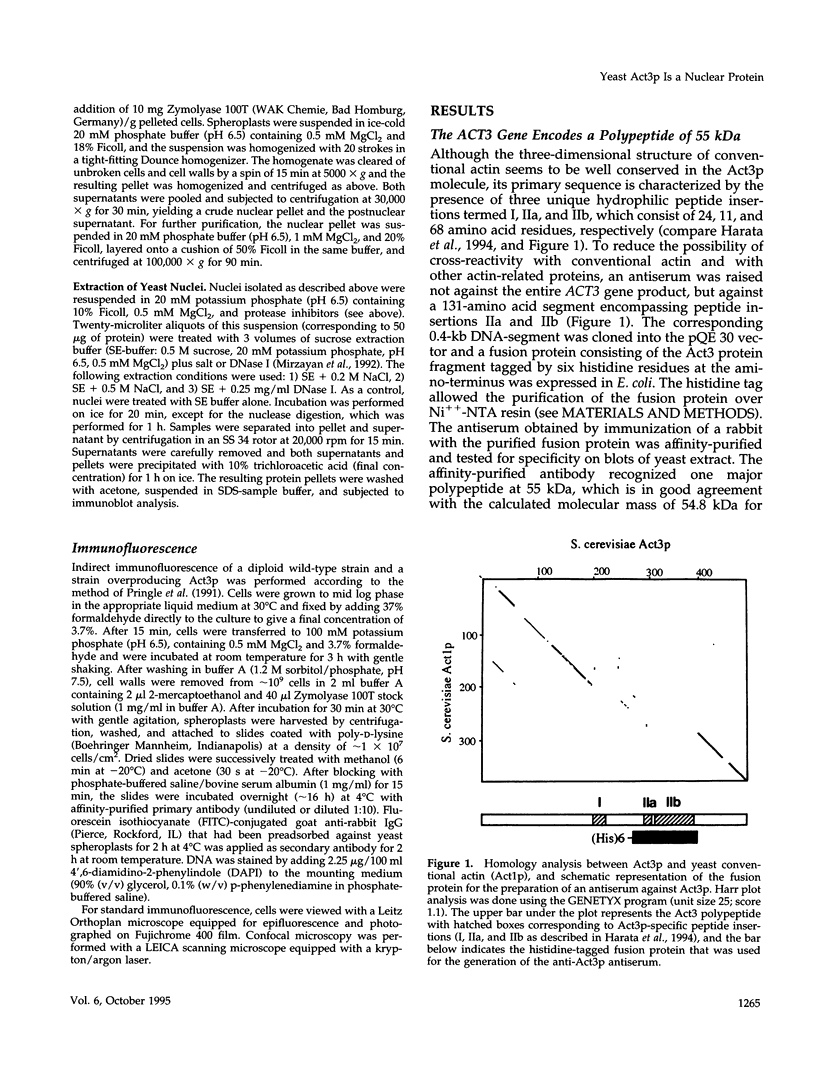
Actin-related proteins, a group of protein families that exhibit about 50% sequence identity among each other and to conventional actin, have been found in a variety of eukaryotic organisms. In the budding yeast Saccharomyces cerevisiae, genes for one conventional actin (ACT1) and for three actin-related proteins (ACT2, ACT3, and ACT5) are known. ACT3, which we recently discovered, is an essential gene coding for a polypeptide of 489 amino acids (Act3p), with a calculated molecular mass of 54.8 kDa. Besides its homology to conventional actin, Act3p possesses a domain exhibiting weak similarity to the chromosomal protein HMG-14 as well as a potential nuclear localization signal. An antiserum prepared against a specific segment of the ACT3 gene product recognizes a polypeptide band of approximately 55 kDa in yeast extract. Indirect immunofluorescence experiments with this antiserum revealed that Act3p is located in the nucleus. Nuclear staining was observed in all cells regardless of the stage of the cell cycle. Independently, immunoblotting experiments with subcellular fractions showed that Act3p is indeed highly enriched in the nuclear fraction. We suggest that Act3p is an essential constituent of yeast chromatin.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alfonso P. J., Crippa M. P., Hayes J. J., Bustin M. The footprint of chromosomal proteins HMG-14 and HMG-17 on chromatin subunits. J Mol Biol. 1994 Feb 11;236(1):189–198. doi: 10.1006/jmbi.1994.1128. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Bretscher A., Drees B., Harsay E., Schott D., Wang T. What are the basic functions of microfilaments? Insights from studies in budding yeast. J Cell Biol. 1994 Aug;126(4):821–825. doi: 10.1083/jcb.126.4.821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. W., Meyer D. I. ACT3: a putative centractin homologue in S. cerevisiae is required for proper orientation of the mitotic spindle. J Cell Biol. 1994 Oct;127(1):129–138. doi: 10.1083/jcb.127.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark S. W., Meyer D. I. Centractin is an actin homologue associated with the centrosome. Nature. 1992 Sep 17;359(6392):246–250. doi: 10.1038/359246a0. [DOI] [PubMed] [Google Scholar]
- Ding H. F., Rimsky S., Batson S. C., Bustin M., Hansen U. Stimulation of RNA polymerase II elongation by chromosomal protein HMG-14. Science. 1994 Aug 5;265(5173):796–799. doi: 10.1126/science.8047885. [DOI] [PubMed] [Google Scholar]
- Dingwall C., Laskey R. A. Nuclear targeting sequences--a consensus? Trends Biochem Sci. 1991 Dec;16(12):478–481. doi: 10.1016/0968-0004(91)90184-w. [DOI] [PubMed] [Google Scholar]
- Frankel S., Heintzelman M. B., Artavanis-Tsakonas S., Mooseker M. S. Identification of a divergent actin-related protein in Drosophila. J Mol Biol. 1994 Jan 28;235(4):1351–1356. doi: 10.1006/jmbi.1994.1090. [DOI] [PubMed] [Google Scholar]
- Fyrberg C., Ryan L., Kenton M., Fyrberg E. Genes encoding actin-related proteins of Drosophila melanogaster. J Mol Biol. 1994 Aug 19;241(3):498–503. doi: 10.1006/jmbi.1994.1526. [DOI] [PubMed] [Google Scholar]
- Gallwitz D., Sures I. Structure of a split yeast gene: complete nucleotide sequence of the actin gene in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1980 May;77(5):2546–2550. doi: 10.1073/pnas.77.5.2546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Bustos J., Heitman J., Hall M. N. Nuclear protein localization. Biochim Biophys Acta. 1991 Mar 7;1071(1):83–101. doi: 10.1016/0304-4157(91)90013-m. [DOI] [PubMed] [Google Scholar]
- Grosschedl R., Giese K., Pagel J. HMG domain proteins: architectural elements in the assembly of nucleoprotein structures. Trends Genet. 1994 Mar;10(3):94–100. doi: 10.1016/0168-9525(94)90232-1. [DOI] [PubMed] [Google Scholar]
- Harata M., Karwan A., Wintersberger U. An essential gene of Saccharomyces cerevisiae coding for an actin-related protein. Proc Natl Acad Sci U S A. 1994 Aug 16;91(17):8258–8262. doi: 10.1073/pnas.91.17.8258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman I. M. Actin isoforms. Curr Opin Cell Biol. 1993 Feb;5(1):48–55. doi: 10.1016/s0955-0674(05)80007-9. [DOI] [PubMed] [Google Scholar]
- Inoue H., Nojima H., Okayama H. High efficiency transformation of Escherichia coli with plasmids. Gene. 1990 Nov 30;96(1):23–28. doi: 10.1016/0378-1119(90)90336-p. [DOI] [PubMed] [Google Scholar]
- Kabsch W., Mannherz H. G., Suck D., Pai E. F., Holmes K. C. Atomic structure of the actin:DNase I complex. Nature. 1990 Sep 6;347(6288):37–44. doi: 10.1038/347037a0. [DOI] [PubMed] [Google Scholar]
- Kabsch W., Vandekerckhove J. Structure and function of actin. Annu Rev Biophys Biomol Struct. 1992;21:49–76. doi: 10.1146/annurev.bb.21.060192.000405. [DOI] [PubMed] [Google Scholar]
- Lees-Miller J. P., Helfman D. M., Schroer T. A. A vertebrate actin-related protein is a component of a multisubunit complex involved in microtubule-based vesicle motility. Nature. 1992 Sep 17;359(6392):244–246. doi: 10.1038/359244a0. [DOI] [PubMed] [Google Scholar]
- Lees-Miller J. P., Henry G., Helfman D. M. Identification of act2, an essential gene in the fission yeast Schizosaccharomyces pombe that encodes a protein related to actin. Proc Natl Acad Sci U S A. 1992 Jan 1;89(1):80–83. doi: 10.1073/pnas.89.1.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lillie S. H., Brown S. S. Artifactual immunofluorescent labelling in yeast, demonstrated by affinity purification of antibody. Yeast. 1987 Jun;3(2):63–70. doi: 10.1002/yea.320030202. [DOI] [PubMed] [Google Scholar]
- Mann K., Mecke D. Isolation and characterization of nuclei and nuclear membranes from Saccharomyces cerevisiae protoplasts. FEBS Lett. 1980 Dec 15;122(1):95–99. doi: 10.1016/0014-5793(80)80410-8. [DOI] [PubMed] [Google Scholar]
- Michaille J. J., Gouy M., Blanchet S., Duret L. Isolation and characterization of a cDNA encoding a chicken actin-like protein. Gene. 1995 Mar 10;154(2):205–209. doi: 10.1016/0378-1119(94)00865-p. [DOI] [PubMed] [Google Scholar]
- Mirzayan C., Copeland C. S., Snyder M. The NUF1 gene encodes an essential coiled-coil related protein that is a potential component of the yeast nucleoskeleton. J Cell Biol. 1992 Mar;116(6):1319–1332. doi: 10.1083/jcb.116.6.1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muhua L., Karpova T. S., Cooper J. A. A yeast actin-related protein homologous to that in vertebrate dynactin complex is important for spindle orientation and nuclear migration. Cell. 1994 Aug 26;78(4):669–679. doi: 10.1016/0092-8674(94)90531-2. [DOI] [PubMed] [Google Scholar]
- Murgia I., Maciver S. K., Morandini P. An actin-related protein from Dictyostelium discoideum is developmentally regulated and associated with mitochondria. FEBS Lett. 1995 Mar 6;360(3):235–241. doi: 10.1016/0014-5793(95)00111-l. [DOI] [PubMed] [Google Scholar]
- Osborne M. A., Schlenstedt G., Jinks T., Silver P. A. Nuf2, a spindle pole body-associated protein required for nuclear division in yeast. J Cell Biol. 1994 May;125(4):853–866. doi: 10.1083/jcb.125.4.853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pringle J. R., Adams A. E., Drubin D. G., Haarer B. K. Immunofluorescence methods for yeast. Methods Enzymol. 1991;194:565–602. doi: 10.1016/0076-6879(91)94043-c. [DOI] [PubMed] [Google Scholar]
- Schafer D. A., Gill S. R., Cooper J. A., Heuser J. E., Schroer T. A. Ultrastructural analysis of the dynactin complex: an actin-related protein is a component of a filament that resembles F-actin. J Cell Biol. 1994 Jul;126(2):403–412. doi: 10.1083/jcb.126.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroer T. A., Fyrberg E., Cooper J. A., Waterston R. H., Helfman D., Pollard T. D., Meyer D. I. Actin-related protein nomenclature and classification. J Cell Biol. 1994 Dec;127(6 Pt 2):1777–1778. doi: 10.1083/jcb.127.6.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwob E., Martin R. P. New yeast actin-like gene required late in the cell cycle. Nature. 1992 Jan 9;355(6356):179–182. doi: 10.1038/355179a0. [DOI] [PubMed] [Google Scholar]
- Walker J. M., Goodwin G. H., Johns E. W. The primary structure of the nucleosome-associated chromosomal protein HMG 14. FEBS Lett. 1979 Apr 15;100(2):394–398. doi: 10.1016/0014-5793(79)80378-6. [DOI] [PubMed] [Google Scholar]
- Wintersberger U., Kühne C., Karwan A. Scp160p, a new yeast protein associated with the nuclear membrane and the endoplasmic reticulum, is necessary for maintenance of exact ploidy. Yeast. 1995 Aug;11(10):929–944. doi: 10.1002/yea.320111004. [DOI] [PubMed] [Google Scholar]






