Abstract
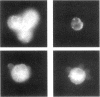
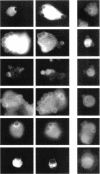
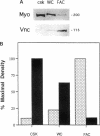
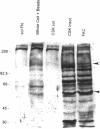
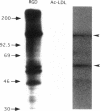
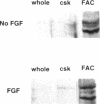
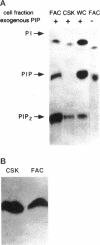
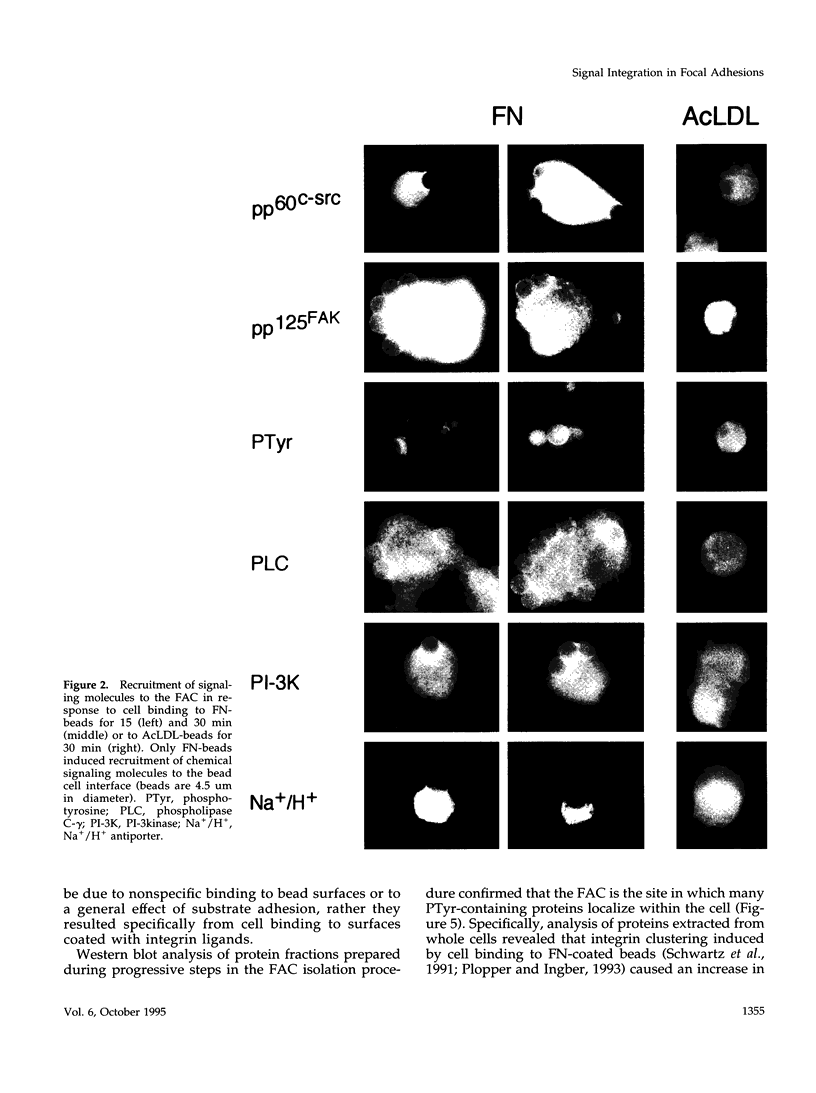
Extracellular matrix controls capillary endothelial cell sensitivity to soluble mitogens by binding integrin receptors and thereby activating a chemical signaling response that rapidly integrates with growth factor-induced signaling mechanisms. Here we report that in addition to integrins, growth factor receptors and multiple molecules that transduce signals conveyed by both types of receptors are immobilized on the cytoskeleton (CSK) and spatially integrated within the focal adhesion complex (FAC) at the site of integrin binding. FACs were rapidly induced in round cells and physically isolated from the remainder of the CSK after detergent-extraction using magnetic microbeads coated with fibronectin or a synthetic RGD-containing peptide. Immunofluorescence microscopy revealed that multiple signaling molecules (e.g., pp60c-src, pp125FAK, phosphatidylinositol-3-kinase, phospholipase C-gamma, and Na+/H+ antiporter) involved in both integrin and growth factor receptor signaling pathways became associated with the CSK framework of the FAC within 15 min after binding to beads coated with integrin ligands. Recruitment of tyrosine kinases to the FAC was also accompanied by a local increase in tyrosine phosphorylation, as indicated by enhanced phosphotyrosine staining at the site of integrin binding. In contrast, neither recruitment of signaling molecules nor increased phosphotyrosine staining was observed when cells bound to beads coated with a control ligand (acetylated low density lipoprotein) that ligates transmembrane scavenger receptors, but does not induce FAC formation. Western blot analysis confirmed that FACs isolated using RGD-beads were enriched for pp60c-src, pp125FAK, phospholipase C-gamma, and the Na+/H+ antiporter when compared with intact CSK or basal cell surface preparations that retained lipid bilayer. Isolated FACs were also greatly enriched for the high affinity fibroblast growth factor receptor flg. Most importantly, isolated FACs continued to exhibit multiple chemical signaling activities in vitro, including protein tyrosine kinase activities (pp60c-src and pp125FAK) as well as the ability to undergo multiple sequential steps in the inositol lipid synthesis cascade. These data suggest that many of the chemical signaling events that are induced by integrins and growth factor receptors in capillary cells may effectively function in a "solid-state" on insoluble CSK scaffolds within the FAC and that the FAC may represent a major site for signal integration between these two regulatory pathways. Future investigations into the biochemical and biophysical basis of signal transduction may be facilitated by this method, which results in isolation of FACs that retain the CSK framework as well as multiple associated chemical signaling activities.
Full text
PDF





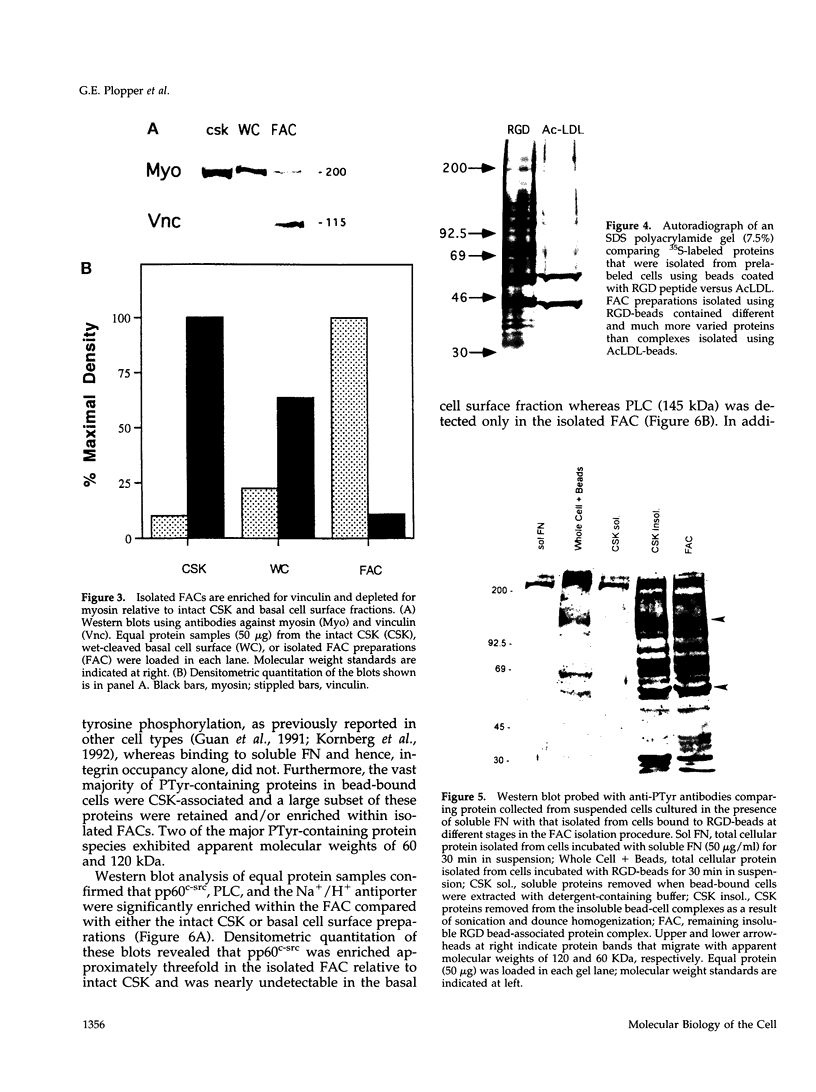
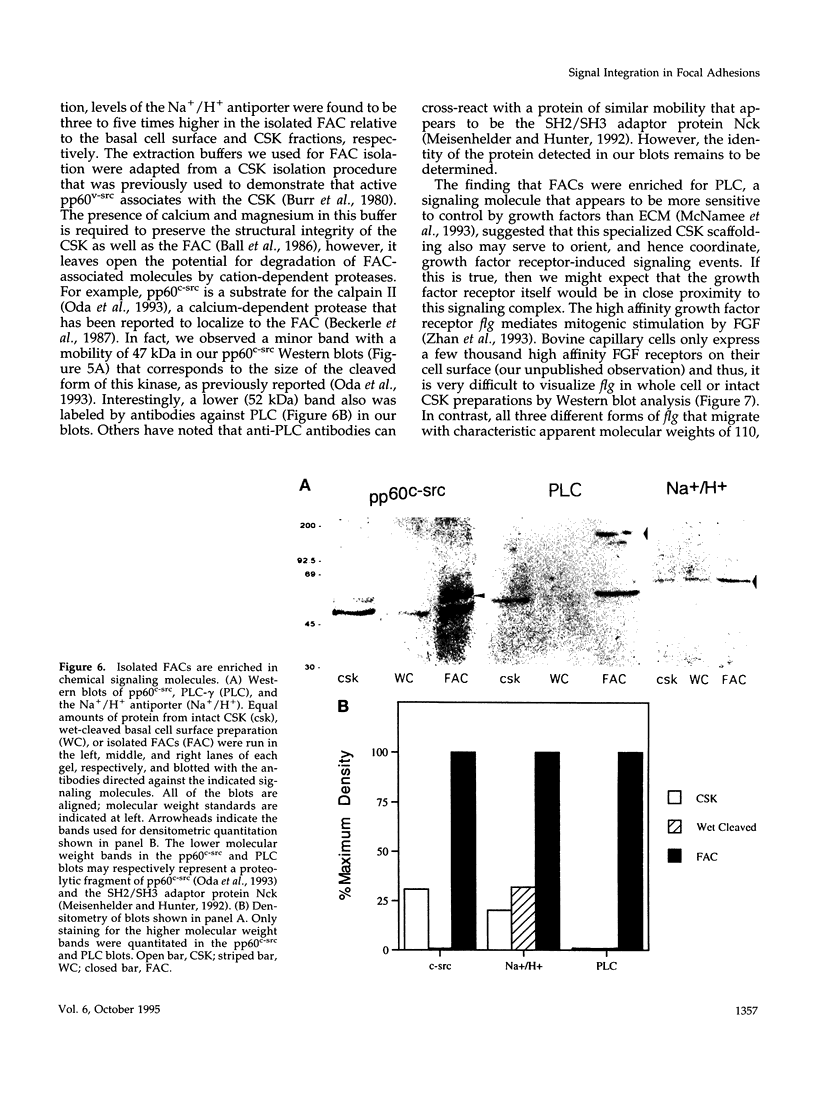
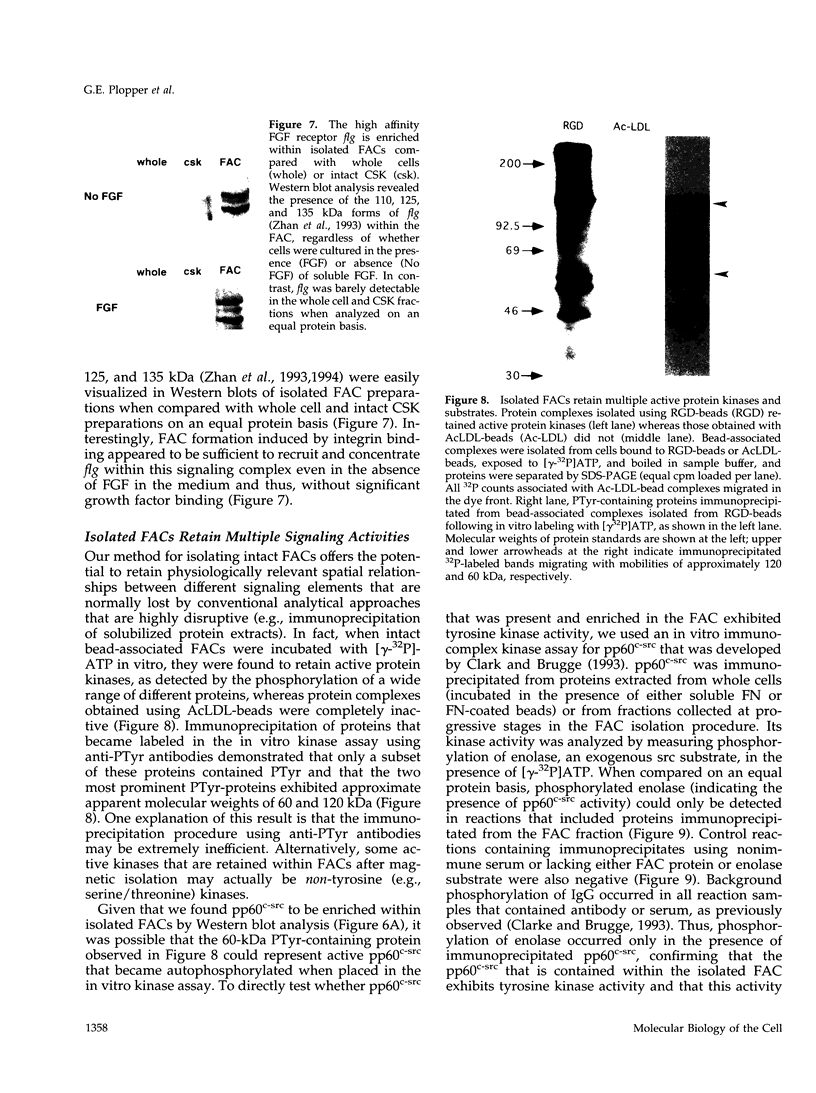
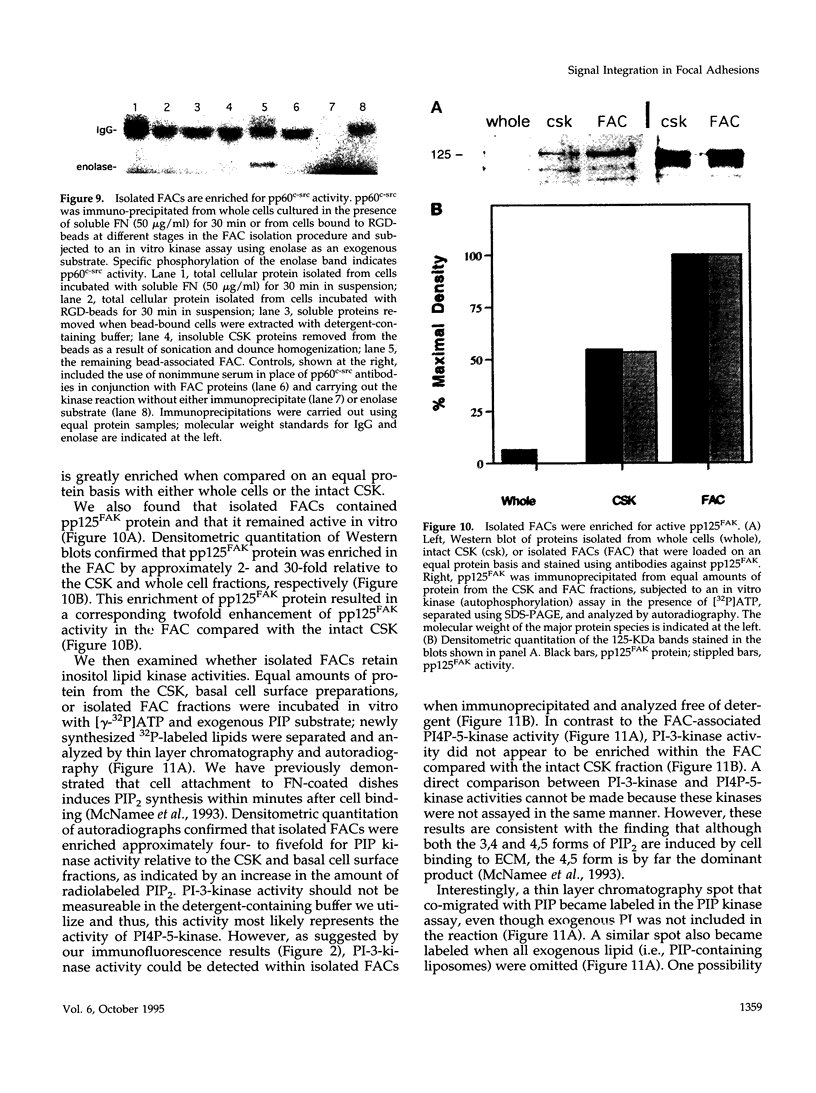






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ball E. H., Freitag C., Gurofsky S. Vinculin interaction with permeabilized cells: disruption and reconstitution of a binding site. J Cell Biol. 1986 Aug;103(2):641–648. doi: 10.1083/jcb.103.2.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banga H. S., Simons E. R., Brass L. F., Rittenhouse S. E. Activation of phospholipases A and C in human platelets exposed to epinephrine: role of glycoproteins IIb/IIIa and dual role of epinephrine. Proc Natl Acad Sci U S A. 1986 Dec;83(23):9197–9201. doi: 10.1073/pnas.83.23.9197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bar-Sagi D., Rotin D., Batzer A., Mandiyan V., Schlessinger J. SH3 domains direct cellular localization of signaling molecules. Cell. 1993 Jul 16;74(1):83–91. doi: 10.1016/0092-8674(93)90296-3. [DOI] [PubMed] [Google Scholar]
- Beckerle M. C., Burridge K., DeMartino G. N., Croall D. E. Colocalization of calcium-dependent protease II and one of its substrates at sites of cell adhesion. Cell. 1987 Nov 20;51(4):569–577. doi: 10.1016/0092-8674(87)90126-7. [DOI] [PubMed] [Google Scholar]
- Brands R., Feltkamp C. A. Wet cleaving of cells: a method to introduce macromolecules into the cytoplasm. Application for immunolocalization of cytosol-exposed antigens. Exp Cell Res. 1988 Jun;176(2):309–318. doi: 10.1016/0014-4827(88)90333-3. [DOI] [PubMed] [Google Scholar]
- Brooks P. C., Montgomery A. M., Rosenfeld M., Reisfeld R. A., Hu T., Klier G., Cheresh D. A. Integrin alpha v beta 3 antagonists promote tumor regression by inducing apoptosis of angiogenic blood vessels. Cell. 1994 Dec 30;79(7):1157–1164. doi: 10.1016/0092-8674(94)90007-8. [DOI] [PubMed] [Google Scholar]
- Burr J. G., Dreyfuss G., Penman S., Buchanan J. M. Association of the src gene product of Rous sarcoma virus with cytoskeletal structures of chicken embryo fibroblasts. Proc Natl Acad Sci U S A. 1980 Jun;77(6):3484–3488. doi: 10.1073/pnas.77.6.3484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burridge K., Fath K., Kelly T., Nuckolls G., Turner C. Focal adhesions: transmembrane junctions between the extracellular matrix and the cytoskeleton. Annu Rev Cell Biol. 1988;4:487–525. doi: 10.1146/annurev.cb.04.110188.002415. [DOI] [PubMed] [Google Scholar]
- Chen Q., Kinch M. S., Lin T. H., Burridge K., Juliano R. L. Integrin-mediated cell adhesion activates mitogen-activated protein kinases. J Biol Chem. 1994 Oct 28;269(43):26602–26605. [PubMed] [Google Scholar]
- Chun J. S., Jacobson B. S. Requirement for diacylglycerol and protein kinase C in HeLa cell-substratum adhesion and their feedback amplification of arachidonic acid production for optimum cell spreading. Mol Biol Cell. 1993 Mar;4(3):271–281. doi: 10.1091/mbc.4.3.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark E. A., Brugge J. S. Redistribution of activated pp60c-src to integrin-dependent cytoskeletal complexes in thrombin-stimulated platelets. Mol Cell Biol. 1993 Mar;13(3):1863–1871. doi: 10.1128/mcb.13.3.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cobb B. S., Schaller M. D., Leu T. H., Parsons J. T. Stable association of pp60src and pp59fyn with the focal adhesion-associated protein tyrosine kinase, pp125FAK. Mol Cell Biol. 1994 Jan;14(1):147–155. doi: 10.1128/mcb.14.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper J. A., Hunter T. Changes in protein phosphorylation in Rous sarcoma virus-transformed chicken embryo cells. Mol Cell Biol. 1981 Feb;1(2):165–178. doi: 10.1128/mcb.1.2.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cybulsky A. V., Carbonetto S., Cyr M. D., McTavish A. J., Huang Q. Extracellular matrix-stimulated phospholipase activation is mediated by beta 1-integrin. Am J Physiol. 1993 Feb;264(2 Pt 1):C323–C332. doi: 10.1152/ajpcell.1993.264.2.C323. [DOI] [PubMed] [Google Scholar]
- Damsky C. H., Werb Z. Signal transduction by integrin receptors for extracellular matrix: cooperative processing of extracellular information. Curr Opin Cell Biol. 1992 Oct;4(5):772–781. doi: 10.1016/0955-0674(92)90100-q. [DOI] [PubMed] [Google Scholar]
- Davies P. F., Robotewskyj A., Griem M. L. Quantitative studies of endothelial cell adhesion. Directional remodeling of focal adhesion sites in response to flow forces. J Clin Invest. 1994 May;93(5):2031–2038. doi: 10.1172/JCI117197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis C. M., Danehower S. C., Laurenza A., Molony J. L. Identification of a role of the vitronectin receptor and protein kinase C in the induction of endothelial cell vascular formation. J Cell Biochem. 1993 Feb;51(2):206–218. doi: 10.1002/jcb.240510213. [DOI] [PubMed] [Google Scholar]
- Davis S., Lu M. L., Lo S. H., Lin S., Butler J. A., Druker B. J., Roberts T. M., An Q., Chen L. B. Presence of an SH2 domain in the actin-binding protein tensin. Science. 1991 May 3;252(5006):712–715. doi: 10.1126/science.1708917. [DOI] [PubMed] [Google Scholar]
- Edidin M. Patches and fences: probing for plasma membrane domains. J Cell Sci Suppl. 1993;17:165–169. doi: 10.1242/jcs.1993.supplement_17.24. [DOI] [PubMed] [Google Scholar]
- Fey E. G., Wan K. M., Penman S. Epithelial cytoskeletal framework and nuclear matrix-intermediate filament scaffold: three-dimensional organization and protein composition. J Cell Biol. 1984 Jun;98(6):1973–1984. doi: 10.1083/jcb.98.6.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox J. E., Lipfert L., Clark E. A., Reynolds C. C., Austin C. D., Brugge J. S. On the role of the platelet membrane skeleton in mediating signal transduction. Association of GP IIb-IIIa, pp60c-src, pp62c-yes, and the p21ras GTPase-activating protein with the membrane skeleton. J Biol Chem. 1993 Dec 5;268(34):25973–25984. [PubMed] [Google Scholar]
- Goldschmidt-Clermont P. J., Machesky L. M., Baldassare J. J., Pollard T. D. The actin-binding protein profilin binds to PIP2 and inhibits its hydrolysis by phospholipase C. Science. 1990 Mar 30;247(4950):1575–1578. doi: 10.1126/science.2157283. [DOI] [PubMed] [Google Scholar]
- Grinstein S., Woodside M., Waddell T. K., Downey G. P., Orlowski J., Pouyssegur J., Wong D. C., Foskett J. K. Focal localization of the NHE-1 isoform of the Na+/H+ antiport: assessment of effects on intracellular pH. EMBO J. 1993 Dec 15;12(13):5209–5218. doi: 10.1002/j.1460-2075.1993.tb06216.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan J. L., Shalloway D. Regulation of focal adhesion-associated protein tyrosine kinase by both cellular adhesion and oncogenic transformation. Nature. 1992 Aug 20;358(6388):690–692. doi: 10.1038/358690a0. [DOI] [PubMed] [Google Scholar]
- Guan J. L., Trevithick J. E., Hynes R. O. Fibronectin/integrin interaction induces tyrosine phosphorylation of a 120-kDa protein. Cell Regul. 1991 Nov;2(11):951–964. doi: 10.1091/mbc.2.11.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen L. K., Mooney D. J., Vacanti J. P., Ingber D. E. Integrin binding and cell spreading on extracellular matrix act at different points in the cell cycle to promote hepatocyte growth. Mol Biol Cell. 1994 Sep;5(9):967–975. doi: 10.1091/mbc.5.9.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hildebrand J. D., Schaller M. D., Parsons J. T. Identification of sequences required for the efficient localization of the focal adhesion kinase, pp125FAK, to cellular focal adhesions. J Cell Biol. 1993 Nov;123(4):993–1005. doi: 10.1083/jcb.123.4.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. O. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992 Apr 3;69(1):11–25. doi: 10.1016/0092-8674(92)90115-s. [DOI] [PubMed] [Google Scholar]
- Ingber D. E. Extracellular matrix as a solid-state regulator in angiogenesis: identification of new targets for anti-cancer therapy. Semin Cancer Biol. 1992 Apr;3(2):57–63. [PubMed] [Google Scholar]
- Ingber D. E. Fibronectin controls capillary endothelial cell growth by modulating cell shape. Proc Natl Acad Sci U S A. 1990 May;87(9):3579–3583. doi: 10.1073/pnas.87.9.3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingber D. E., Folkman J. How does extracellular matrix control capillary morphogenesis? Cell. 1989 Sep 8;58(5):803–805. doi: 10.1016/0092-8674(89)90928-8. [DOI] [PubMed] [Google Scholar]
- Ingber D. E., Folkman J. Mechanochemical switching between growth and differentiation during fibroblast growth factor-stimulated angiogenesis in vitro: role of extracellular matrix. J Cell Biol. 1989 Jul;109(1):317–330. doi: 10.1083/jcb.109.1.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingber D. E., Madri J. A., Folkman J. A possible mechanism for inhibition of angiogenesis by angiostatic steroids: induction of capillary basement membrane dissolution. Endocrinology. 1986 Oct;119(4):1768–1775. doi: 10.1210/endo-119-4-1768. [DOI] [PubMed] [Google Scholar]
- Ingber D. E., Prusty D., Frangioni J. V., Cragoe E. J., Jr, Lechene C., Schwartz M. A. Control of intracellular pH and growth by fibronectin in capillary endothelial cells. J Cell Biol. 1990 May;110(5):1803–1811. doi: 10.1083/jcb.110.5.1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingber D. E. The riddle of morphogenesis: a question of solution chemistry or molecular cell engineering? Cell. 1993 Dec 31;75(7):1249–1252. doi: 10.1016/0092-8674(93)90612-t. [DOI] [PubMed] [Google Scholar]
- Ingber D. Integrins as mechanochemical transducers. Curr Opin Cell Biol. 1991 Oct;3(5):841–848. doi: 10.1016/0955-0674(91)90058-7. [DOI] [PubMed] [Google Scholar]
- Jaken S., Leach K., Klauck T. Association of type 3 protein kinase C with focal contacts in rat embryo fibroblasts. J Cell Biol. 1989 Aug;109(2):697–704. doi: 10.1083/jcb.109.2.697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janmey P. A., Stossel T. P. Modulation of gelsolin function by phosphatidylinositol 4,5-bisphosphate. Nature. 1987 Jan 22;325(6102):362–364. doi: 10.1038/325362a0. [DOI] [PubMed] [Google Scholar]
- Kapron-Bras C., Fitz-Gibbon L., Jeevaratnam P., Wilkins J., Dedhar S. Stimulation of tyrosine phosphorylation and accumulation of GTP-bound p21ras upon antibody-mediated alpha 2 beta 1 integrin activation in T-lymphoblastic cells. J Biol Chem. 1993 Oct 5;268(28):20701–20704. [PubMed] [Google Scholar]
- Kellie S., Patel B., Mitchell A., Critchley D. R., Wigglesworth N. M., Wyke J. A. Comparison of the relative importance of tyrosine-specific vinculin phosphorylation and the loss of surface-associated fibronectin in the morphology of cells transformed by Rous sarcoma virus. J Cell Sci. 1986 Jun;82:129–142. doi: 10.1242/jcs.82.1.129. [DOI] [PubMed] [Google Scholar]
- Kellie S., Patel B., Wigglesworth N. M., Critchley D. R., Wyke J. A. The use of Rous sarcoma virus transformation mutants with differing tyrosine kinase activities to study the relationships between vinculin phosphorylation, pp60v-src location and adhesion plaque integrity. Exp Cell Res. 1986 Jul;165(1):216–228. doi: 10.1016/0014-4827(86)90546-x. [DOI] [PubMed] [Google Scholar]
- Koch C. A., Anderson D., Moran M. F., Ellis C., Pawson T. SH2 and SH3 domains: elements that control interactions of cytoplasmic signaling proteins. Science. 1991 May 3;252(5006):668–674. doi: 10.1126/science.1708916. [DOI] [PubMed] [Google Scholar]
- Kornberg L., Earp H. S., Parsons J. T., Schaller M., Juliano R. L. Cell adhesion or integrin clustering increases phosphorylation of a focal adhesion-associated tyrosine kinase. J Biol Chem. 1992 Nov 25;267(33):23439–23442. [PubMed] [Google Scholar]
- Kume N., Arai H., Kawai C., Kita T. Receptors for modified low-density lipoproteins on human endothelial cells: different recognition for acetylated low-density lipoprotein and oxidized low-density lipoprotein. Biochim Biophys Acta. 1991 Jan 10;1091(1):63–67. doi: 10.1016/0167-4889(91)90223-k. [DOI] [PubMed] [Google Scholar]
- Lewis J. M., Schwartz M. A. Mapping in vivo associations of cytoplasmic proteins with integrin beta 1 cytoplasmic domain mutants. Mol Biol Cell. 1995 Feb;6(2):151–160. doi: 10.1091/mbc.6.2.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling L. E., Schulz J. T., Cantley L. C. Characterization and purification of membrane-associated phosphatidylinositol-4-phosphate kinase from human red blood cells. J Biol Chem. 1989 Mar 25;264(9):5080–5088. [PubMed] [Google Scholar]
- Lipfert L., Haimovich B., Schaller M. D., Cobb B. S., Parsons J. T., Brugge J. S. Integrin-dependent phosphorylation and activation of the protein tyrosine kinase pp125FAK in platelets. J Cell Biol. 1992 Nov;119(4):905–912. doi: 10.1083/jcb.119.4.905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maher P. A., Pasquale E. B., Wang J. Y., Singer S. J. Phosphotyrosine-containing proteins are concentrated in focal adhesions and intercellular junctions in normal cells. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6576–6580. doi: 10.1073/pnas.82.19.6576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchisio P. C., Di Renzo M. F., Comoglio P. M. Immunofluorescence localization of phosphotyrosine containing proteins in RSV-transformed mouse fibroblasts. Exp Cell Res. 1984 Sep;154(1):112–124. doi: 10.1016/0014-4827(84)90672-4. [DOI] [PubMed] [Google Scholar]
- Masters D. B., Griggs C. T., Berde C. B. High sensitivity quantification of RNA from gels and autoradiograms with affordable optical scanning. Biotechniques. 1992 Jun;12(6):902-6, 908-11. [PubMed] [Google Scholar]
- McNamee H. P., Ingber D. E., Schwartz M. A. Adhesion to fibronectin stimulates inositol lipid synthesis and enhances PDGF-induced inositol lipid breakdown. J Cell Biol. 1993 May;121(3):673–678. doi: 10.1083/jcb.121.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meisenhelder J., Hunter T. The SH2/SH3 domain-containing protein Nck is recognized by certain anti-phospholipase C-gamma 1 monoclonal antibodies, and its phosphorylation on tyrosine is stimulated by platelet-derived growth factor and epidermal growth factor treatment. Mol Cell Biol. 1992 Dec;12(12):5843–5856. doi: 10.1128/mcb.12.12.5843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto S., Akiyama S. K., Yamada K. M. Synergistic roles for receptor occupancy and aggregation in integrin transmembrane function. Science. 1995 Feb 10;267(5199):883–885. doi: 10.1126/science.7846531. [DOI] [PubMed] [Google Scholar]
- Niederreiter M., Gimona M., Streichsbier F., Celis J. E., Small J. V. Complex protein composition of isolated focal adhesions: a two-dimensional gel and database analysis. Electrophoresis. 1994 Mar-Apr;15(3-4):511–519. doi: 10.1002/elps.1150150169. [DOI] [PubMed] [Google Scholar]
- Oda A., Druker B. J., Ariyoshi H., Smith M., Salzman E. W. pp60src is an endogenous substrate for calpain in human blood platelets. J Biol Chem. 1993 Jun 15;268(17):12603–12608. [PubMed] [Google Scholar]
- Pelletier A. J., Bodary S. C., Levinson A. D. Signal transduction by the platelet integrin alpha IIb beta 3: induction of calcium oscillations required for protein-tyrosine phosphorylation and ligand-induced spreading of stably transfected cells. Mol Biol Cell. 1992 Sep;3(9):989–998. doi: 10.1091/mbc.3.9.989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plopper G., Ingber D. E. Rapid induction and isolation of focal adhesion complexes. Biochem Biophys Res Commun. 1993 Jun 15;193(2):571–578. doi: 10.1006/bbrc.1993.1662. [DOI] [PubMed] [Google Scholar]
- Rahilly M. A., Fleming S. A tumour promoter induces alterations in vinculin and actin distribution in human renal epithelium. J Pathol. 1992 Mar;166(3):283–288. doi: 10.1002/path.1711660311. [DOI] [PubMed] [Google Scholar]
- Rankin S., Rozengurt E. Platelet-derived growth factor modulation of focal adhesion kinase (p125FAK) and paxillin tyrosine phosphorylation in Swiss 3T3 cells. Bell-shaped dose response and cross-talk with bombesin. J Biol Chem. 1994 Jan 7;269(1):704–710. [PubMed] [Google Scholar]
- Ridley A. J., Hall A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell. 1992 Aug 7;70(3):389–399. doi: 10.1016/0092-8674(92)90163-7. [DOI] [PubMed] [Google Scholar]
- Rohrschneider L., Rosok M., Shriver K. Mechanism of transformation by Rous sarcoma virus: events within adhesion plaques. Cold Spring Harb Symp Quant Biol. 1982;46(Pt 2):953–965. doi: 10.1101/sqb.1982.046.01.089. [DOI] [PubMed] [Google Scholar]
- Ross W., Bertrand W., Morrison A. A photoactivatable probe for the Na+/H+ exchanger cross-links a 66-kDa renal brush border membrane protein. J Biol Chem. 1990 Apr 5;265(10):5341–5344. [PubMed] [Google Scholar]
- Sardet C., Counillon L., Franchi A., Pouysségur J. Growth factors induce phosphorylation of the Na+/H+ antiporter, glycoprotein of 110 kD. Science. 1990 Feb 9;247(4943):723–726. doi: 10.1126/science.2154036. [DOI] [PubMed] [Google Scholar]
- Schaller M. D., Borgman C. A., Cobb B. S., Vines R. R., Reynolds A. B., Parsons J. T. pp125FAK a structurally distinctive protein-tyrosine kinase associated with focal adhesions. Proc Natl Acad Sci U S A. 1992 Jun 1;89(11):5192–5196. doi: 10.1073/pnas.89.11.5192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer D. D., Hanks S. K., Hunter T., van der Geer P. Integrin-mediated signal transduction linked to Ras pathway by GRB2 binding to focal adhesion kinase. Nature. 1994 Dec 22;372(6508):786–791. doi: 10.1038/372786a0. [DOI] [PubMed] [Google Scholar]
- Schwartz M. A., Ingber D. E. Integrating with integrins. Mol Biol Cell. 1994 Apr;5(4):389–393. doi: 10.1091/mbc.5.4.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz M. A., Lechene C., Ingber D. E. Insoluble fibronectin activates the Na/H antiporter by clustering and immobilizing integrin alpha 5 beta 1, independent of cell shape. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7849–7853. doi: 10.1073/pnas.88.17.7849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz M. A. Spreading of human endothelial cells on fibronectin or vitronectin triggers elevation of intracellular free calcium. J Cell Biol. 1993 Feb;120(4):1003–1010. doi: 10.1083/jcb.120.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shattil S. J., Brugge J. S. Protein tyrosine phosphorylation and the adhesive functions of platelets. Curr Opin Cell Biol. 1991 Oct;3(5):869–879. doi: 10.1016/0955-0674(91)90062-4. [DOI] [PubMed] [Google Scholar]
- Sorisky A., King W. G., Rittenhouse S. E. Accumulation of PtdIns(3,4)P2 and PtdIns(3,4,5)P3 in thrombin-stimulated platelets. Different sensitivities to Ca2+ or functional integrin. Biochem J. 1992 Sep 1;286(Pt 2):581–584. doi: 10.1042/bj2860581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Symington B. E. Fibronectin receptor modulates cyclin-dependent kinase activity. J Biol Chem. 1992 Dec 25;267(36):25744–25747. [PubMed] [Google Scholar]
- Tanaka A., Gibbs C. P., Arthur R. R., Anderson S. K., Kung H. J., Fujita D. J. DNA sequence encoding the amino-terminal region of the human c-src protein: implications of sequence divergence among src-type kinase oncogenes. Mol Cell Biol. 1987 May;7(5):1978–1983. doi: 10.1128/mcb.7.5.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tapley P., Horwitz A., Buck C., Duggan K., Rohrschneider L. Integrins isolated from Rous sarcoma virus-transformed chicken embryo fibroblasts. Oncogene. 1989 Mar;4(3):325–333. [PubMed] [Google Scholar]
- Torti M., Sinigaglia F., Ramaschi G., Balduini C. Platelet glycoprotein IIb-IIIa is associated with 21-kDa GTP-binding protein. Biochim Biophys Acta. 1991 Nov 18;1070(1):20–26. doi: 10.1016/0005-2736(91)90141-t. [DOI] [PubMed] [Google Scholar]
- Truskey G. A., Burmeister J. S., Grapa E., Reichert W. M. Total internal reflection fluorescence microscopy (TIRFM). II. Topographical mapping of relative cell/substratum separation distances. J Cell Sci. 1992 Oct;103(Pt 2):491–499. doi: 10.1242/jcs.103.2.491. [DOI] [PubMed] [Google Scholar]
- Turner C. E., Pavalko F. M., Burridge K. The role of phosphorylation and limited proteolytic cleavage of talin and vinculin in the disruption of focal adhesion integrity. J Biol Chem. 1989 Jul 15;264(20):11938–11944. [PubMed] [Google Scholar]
- Vuori K., Ruoslahti E. Activation of protein kinase C precedes alpha 5 beta 1 integrin-mediated cell spreading on fibronectin. J Biol Chem. 1993 Oct 15;268(29):21459–21462. [PubMed] [Google Scholar]
- Wang N., Butler J. P., Ingber D. E. Mechanotransduction across the cell surface and through the cytoskeleton. Science. 1993 May 21;260(5111):1124–1127. doi: 10.1126/science.7684161. [DOI] [PubMed] [Google Scholar]
- Wang N., Ingber D. E. Control of cytoskeletal mechanics by extracellular matrix, cell shape, and mechanical tension. Biophys J. 1994 Jun;66(6):2181–2189. doi: 10.1016/S0006-3495(94)81014-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White M. F., Kahn C. R. The insulin signaling system. J Biol Chem. 1994 Jan 7;269(1):1–4. [PubMed] [Google Scholar]
- Woods A., Couchman J. R. Protein kinase C involvement in focal adhesion formation. J Cell Sci. 1992 Feb;101(Pt 2):277–290. doi: 10.1242/jcs.101.2.277. [DOI] [PubMed] [Google Scholar]
- Wu H., Reynolds A. B., Kanner S. B., Vines R. R., Parsons J. T. Identification and characterization of a novel cytoskeleton-associated pp60src substrate. Mol Cell Biol. 1991 Oct;11(10):5113–5124. doi: 10.1128/mcb.11.10.5113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing Z., Chen H. C., Nowlen J. K., Taylor S. J., Shalloway D., Guan J. L. Direct interaction of v-Src with the focal adhesion kinase mediated by the Src SH2 domain. Mol Biol Cell. 1994 Apr;5(4):413–421. doi: 10.1091/mbc.5.4.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zachary I., Rozengurt E. Focal adhesion kinase (p125FAK): a point of convergence in the action of neuropeptides, integrins, and oncogenes. Cell. 1992 Dec 11;71(6):891–894. doi: 10.1016/0092-8674(92)90385-p. [DOI] [PubMed] [Google Scholar]
- Zhan X., Hu X., Friesel R., Maciag T. Long term growth factor exposure and differential tyrosine phosphorylation are required for DNA synthesis in BALB/c 3T3 cells. J Biol Chem. 1993 May 5;268(13):9611–9620. [PubMed] [Google Scholar]