Abstract
Background
The aim of the present study was to examine how lipid profiles are associated with insulin resistance in Japanese community-dwelling adults.
Methods
This cross-sectional study included 614 men aged 58 ± 14 (mean ± standard deviation; range, 20-89) years and 779 women aged 60 ± 12 (range, 21-88) years. The study sample were 1,042 (74.8%) non-obese (BMI < 25.0 kg/m2) and 351 (25.2%) overweight (BMI ≥ 25 kg/m2) subjects. Insulin resistance was defined by homeostasis model assessment of insulin resistance (HOMA-IR) of at least 2.5. The areas under the curve (AUC) of the receiver operating characteristic curves (ROC) were used to compare the power of these serum markers.
Results
In non-obese subjects, the best marker of insulin resistance was low-density lipoprotein cholesterol (LDL-C)/high-density lipoprotein cholesterol (HDL-C) ratio of 0.74 (95% confidence interval (CI), 0.66-0.80). The HDL-C, triglyceride (TG)/HDL-C ratio, and non-HDL-C also discriminated insulin resistance, as the values for AUC were 0.31 (95% CI, 0.24-0.38), 0.69 (95% CI, 0.62-0.75) and 0.69 (95% CI, 0.62-0.75), respectively. In overweight subjects, the AUC for TG and TG/HDL-C ratio were 0.64 (0.58-0.71) and 0.64 (0.57-0.70), respectively. The optimal cut-off point to identifying insulin resistance for these markers yielded the following values: TG/HDL-C ratio of ≥1.50 and LDL-C/HDL-C ratio of ≥2.14 in non-obese subjects, and ≥2.20, ≥2.25 in overweight subjects. In non-obese subjects, the positive likelihood ratio was greatest for LDL-C/HDL-C ratio.
Conclusion
In non-obese Japanese adults, LDL-C/HDL-C ratio may be the best reliable marker of insulin resistance.
Background
Being overweight is also a major worldwide public health problem. In Japanese adults, it was demonstrated that 29.3% of men and 19.5% of women are overweight (body mass index (BMI); weight (kg)/height (m) 2, ≥25 kg/m2), with approximately 3.3% of men and 3.2% of women designated as obese (BMI ≥ 30 kg/m2) [1]. Several large-scale studies have shown that overweight people, defined on the basis of a high BMI, increases the risk of hypertension, dyslipidemia, diabetes and developing cardiovascular disease (CVD) such as stroke and myocardial infarction than subjects with normal BMI [2-4]. Moreover, it has been suggested that individuals exist who are not overweight, but who, like people with overt obesity, also have hyperinsulinemia, are insulin resistant, and are predisposed to hypertriglyceridemia, type 2 diabetes mellitus, and premature cardiovascular disease [5,6]. Much investigation has been focused on insulin resistance as the primary determinant of atherosclerotic risk factors, independent of BMI [7].
On the other hand, insulin resistant persons also have a characteristic dyslipidemia [8] and measuring these variables might help identify insulin resistance. Plasma triglycerides, high-density lipoprotein cholesterol (HDL-C) [9,10], and total cholesterol (T-C)/HDL-C are independently associated with insulin resistance and risk factors of CVD [11]. However, in Japanese community-dwelling persons, there are few studies to demonstrate a relationship between lipid profiles and insulin resistance, categorized by BMI.
We took advantage of the large representative sample of Japanese adults who participated at the time of their annual health examination. We investigated how lipid profiles were associated with insulin resistance in healthy Japanese adults. For this, we used cross-sectional data from community-dwelling participants without clinical diabetes.
Methods
Subjects
Participants were recruited at the time of their annual health examination in a rural town with a total population of 11,136 (as of April 2002) and located in Ehime prefecture, Japan, in 2002. Among the 9,133 adults (4,395 of them male) aged 19 to 90 years in this population, a random sample of 3,164 subjects (34.6%) was recruited. Information on medical history, present conditions, and drugs was obtained by interview. Other characteristics, e.g., smoking and alcohol habits, and medication, were investigated by individual interviews using a structured questionnaire. Subjects taking medications for hypertension, diabetes, or dyslipidemia were excluded. The final study sample included 614 men and 779 women. This study was approved by the ethics committee of Ehime University School of Medicine, and written informed consent was obtained from each subject.
Evaluation of confounding factors
Information on demographic characteristics and risk factors was collected using clinical files. Body mass index was calculated by dividing weight (in kilograms) by the square of the height (in meters). We measured blood pressure in the right upper arm of participants in a sedentary position using an automatic oscillometric blood pressure recorder (BP-103i; Colin, Aichi, Japan) while the subjects were seated after having rested for at least 5 min. Smoking status was defined as the number of cigarette packs per day multiplied by the number of years smoked (pack·year), and the participants were classified into never smokers, past smokers, light smokers (<30 pack·year) and heavy smokers (≥30 pack·year). The daily alcohol consumption was measured using the Japanese liquor unit in which a unit corresponds to 22.9 g of ethanol, and the participants were classified into never drinkers, occasional drinkers (<1 unit/day), light drinkers (1-1.9 unit/day), and heavy drinkers (≥2 unit/day). T-C, TG, HDL-C, fasting blood glucose (FBG), creatinine (enzymatic method), uric acid, and immunoreactive insulin (IRI), and high molecular weight (HMW) adiponectin (FUJIREBIO, Tokyo, Japan) were measured during fasting low-density lipoprotein cholesterol (LDL-C) levels were calculated using the Friedewald formula [12]. Participants with TG levels ≥400 mg/dl were excluded. Homeostasis of minimal assessment of insulin resistance (HOMA-IR) was calculated from FBG and IRI levels using the following formula; {FBG (mg/dL) × IRI (mU/mL)}/405 [13], and a borderline level of Insulin resistance was defined as HOMA-IR ≥ 1.6 [13], and a definite level, HOMA-IR ≥ 2.5 [14].
Statistical analysis
Statistical analysis was performed using PASW Statistics 17.0 (Statistical Package for Social Science Japan, Inc., Tokyo, Japan). All values are expressed as mean ± standard deviation (SD), unless otherwise specified. Data for TG, FBG, HOMA-IR, and serum HMW adiponectin were skewed, and log-transformed for analysis. Subjects were divided into two groups based on BMI (non-obese, <25.0 kg/m2; overweight, ≥25.0 kg/m2), and differences between the two groups were determined by Student's t-test andχ2 test. In addition, areas under the receiver operating characteristic (ROC) curves were determined for each variable to identify the predictors of insulin resistance. Areas under the ROC curves are provided with standard errors. An ROC curve is a plot of the sensitivity (true positive) versus 1-specificity (false positive) for each potential marker tested. The area under the ROC curve is a summary of the overall diagnostic accuracy of the test. The best markers have ROC curves that are shifted to the left with areas under the curve near unity. Nondiagnostic markers are represented by diagonals with areas under the ROC curves close to 0.5. Likelihood ratios were calculated as the ratios of sensitivity - (1 - specificity) (positive likelihood ratio) and (1 - sensitivity) - specificity (negative likelihood ratio). A value of P < 0.05 was considered significant.
Results
Background factors of subjects categorized by BMI
Table 1 shows the value of each background factor categorized by BMI. The subjects comprised 614 men aged 58 ± 14 (mean ± standard deviation; range, 20-89) years and 779 women aged 60 ± 12 (range, 21-88) years. The mean BMI in the study sample was 23.1 ± 3.1 kg/m2, with 1,042 non-obese (BMI < 25.0 kg/m2) (74.8%) and 351 overweight (BMI ≥ 25 kg/m2) (25.2%). Alcohol consumption, systolic blood pressure (SBP), diastolic blood pressure (DBP), T-C, TG, LDL-C, TG/HDL-C ratio, non-HDL-C, LDL-C/HDL-C ratio, and uric acid were significantly higher in subjects with a BMI ≥ 25.0 kg/m2, but age, HDL-C and serum HMW adiponectin were significantly lower in that group. There were no inter-group differences in sex, smoking status, eGFR, and prevalence of CVD.
Table 1.
Characteristics of subjects categorized by body mass index
| Total | Non-obese | Overweight | ||
|---|---|---|---|---|
| Body mass index† Characteristics |
All N = 1,393 |
<25.0 kg/m2 N = 1,042 |
≥25.0 kg/m2 N = 351 |
P -value* |
| Male sex, % | 44.1 | 43.3 | 46.4 | 0.320 |
| Age (years) | 59 ± 13 | 60 ± 13 | 57 ± 12 | 0.006 |
| Body mass index (kg/m2) | 23.1 ± 3.1 | 21.7 ± 2.0 | 27.1 ± 2.2 | <0.001 |
| Smoking status {never/ex/light/heavy (%)} | 71.0/9.6/9.0/10.4 | 72.3/8.4/8.8/10.5 | 67.2/13.1/9.4/10.3 | 0.073 |
| Alcohol consumption {never/light/moderate/heavy(%)} | 39.0/31.7/18.6/10.8 | 40.7/30.3/19.0/10.0 | 33.9/35.6/17.4/13.1 | 0.043 |
| Systolic blood pressure (mmHg) | 133 ± 21 | 132 ± 21 | 139 ± 19 | <0.001 |
| Diastolic blood pressure (mmHg) | 80 ± 11 | 79 ± 11 | 83 ± 11 | <0.001 |
| Total cholesterol (mg/dL) | 201 ± 35 | 200 ± 34 | 204 ± 35 | 0.031 |
| Triglycerides (mg/dL) | 90 (68-127) | 85 (64-117) | 108 (81-153) | <0.001 |
| HDL cholesterol (mg/dL) | 62 ± 15 | 64 ± 16 | 57 ± 13 | <0.001 |
| LDL cholesterol (mg/dL) | 118 ± 31 | 116 ± 31 | 123 ± 32 | <0.001 |
| Triglycerides/HDL cholesterol ratio | 1.5 (1.0-2.3) | 1.3 (0.9-2.0) | 1.9 (1.3-3.2) | 0.001 |
| Non-HDL cholesterol (mg/dL) | 139 ± 34 | 135 ± 34 | 148 ± 34 | <0.001 |
| LDL-cholesterol/HDL-cholesterol ratio | 2.01 ± 0.78 | 1.92 ± 0.74 | 2.30 ± 0.82 | <0.001 |
| Uric acid (mg/dL) | 5.0 ± 1.4 | 4.8 ± 1.4 | 5.5 ± 1.4 | <0.001 |
| eGFR | 83.3 ± 16.4 | 83.4 ± 16.1 | 82.9 ± 17.2 | 0.659 |
| Serum HMW adiponectin (μg/mL) | 3.0 (4.9-8.0) | 5.4 (3.2-8.7) | 3.9 (2.2-5.8) | <0.001 |
| Cardiovascular disease, % | 4.7 | 4.8 | 4.3 | 0.771 |
HDL, high-density lipoprotein; LDL, low-density lipoprotein; eGFR, estimated glomerular filtration rate; HMW, high molecular weight. Data presented are mean ± standard deviation. Data for triglycerides, TG/HDL-C ratio, and serum adiponectin were skewed, and are presented as median (interquartile range). Data for triglycerides, TG/HDL-C ratio, and serum adiponectin were log-transformed for analysis. * Student's t-test or χ2 test.
Insulin resistance of subjects categorized by BMI
Fasting blood glucose, IRI, and HOMA-IR were significantly higher in overweight subjects (Table 2), and prevalence of insulin resistance (HOMA-IR > 1.6 or >2.5) was significantly higher in overweight subjects than in non-obese subjects.
Table 2.
Insulin resistance of subjects categorized by body mass index
| Total | Non-obese | Overweight | ||
|---|---|---|---|---|
| Body mass index Characteristics |
All N = 1,393 |
<25.0 kg/m2 N = 1,042 |
≥25.0 kg/m2 N = 351 |
P -value* |
| Fasting blood glucose (mg/dL) | 92 (87-99) | 91 (86-98) | 95 (89-103) | <0.001 |
| Immunoreactive insulin (mU/mL) | 5.1 (3.4-7.6) | 4.5 (3.0-6.5) | 8.0 (5.3-11.2) | <0.001 |
| HOMA-IR§ | 1.19 (0.75-1.83) | 1.02 (0.65-1.52) | 1.91 (1.23-2.81) | <0.001 |
| HOMA-IR§ ≤ 1.6, % | 68.1 | 77.9 | 39.0 | |
| HOMA-IR§ > 1.6 and ≤2.5, % | 19.8 | 16.1 | 30.8 | <0.001 |
| HOMA-IR§ > 2.5, % | 12.1 | 6.0 | 30.2 | <0.001 |
HOMA-IR, homeostasis of minimal assessment of insulin resistance. Data for fasting blood glucose, fasting insulin, and HOMA-IR were skewed, and are presented as median (interquartile range). §HOMA-IR was calculated using the following formula; {fasting blood glucose (FBG) (mg/dL) X fasting Immunoreactive insulin (mU/mL)}/405. Data for fasting blood glucose, fasting insulin, and HOMA-IR were log-transformed for analysis. *Student's t test or χ2 test.
Comparison of areas under ROC curves (95% CI) for potential markers of insulin resistance of subjects categorized by BMI
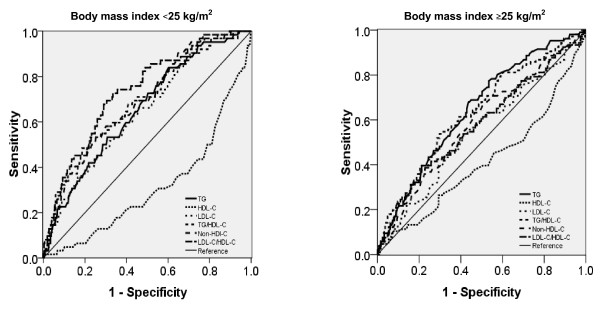
In non-obese subjects, the ROC curve analyses showed that the best marker of insulin resistance was LDL-C/HDL-C ratio, with an area under the ROC curve of 0.74 (0.66-0.80) (Table 3; Figure 1). The TG/HDL-C ratio, HDL-C, and non-HDL-C also discriminated insulin resistance, as they had areas under the ROC curve of 0.31 (0.24-0.38), 0.69 (0.62-0.75) and 0.69 (0.62-0.75), respectively. In overweight subjects, TG and TG/HDL-C ratio were more effective.
Table 3.
Comparison of areas under the ROC curves (95% CI) for potential markers of insulin resistance (HOMA-IR§ > 2.5) of subjects categorized by body mass index
| AUC (95% CI) | ||||||
|---|---|---|---|---|---|---|
| Total | Non-obese | Overweight | ||||
| Body mass index Characteristics |
All N = 1,393 |
P-value |
<25.0 kg/m2 N = 1,042 |
P-value |
>25.0 kg/m2 N = 351 |
P-value |
| Triglycerides (mg/dL) | 0.69 (0.45-0.73) | <0.001 | 0.66 (0.59-0.72) | <0.001 | 0.64 (0.58-0.71) | <0.001 |
| HDL cholesterol (mg/dL) | 0.33 (0.28-0.37) | <0.001 | 0.31 (0.24-0.38) | <0.001 | 0.41 (0.35-0.48) | 0.009 |
| LDL cholesterol (mg/dL) | 0.61 (0.56-0.65) | <0.001 | 0.65 (0.58-0.72) | <0.001 | 0.54 (0.48-0.61) | 0.188 |
| Triglycerides/HDL cholesterol ratio | 0.70 (0.66-0.75) | <0.001 | 0.69 (0.62-0.75) | <0.001 | 0.64 (0.57-0.70) | <0.001 |
| Non-HDL cholesterol (mg/dL) | 0.66 (0.61-0.70) | <0.001 | 0.69 (0.62-0.75) | <0.001 | 0.59 (0.52-0.65) | 0.011 |
| LDL-cholesterol/HDL-cholesterol ratio | 0.69 (0.65-0.73) | <0.001 | 0.74 (0.66-0.80) | <0.001 | 0.58 (0.51-0.65) | 0.021 |
ROC, receiver operating characteristics; HOMA-IR, homeostasis of minimal assessment of insulin resistance; CI, confidence interval; AUC, area under ROC curve; HDL, high-density lipoprotein; LDL, low-density lipoprotein. §HOMA-IR was calculated using the following formula; {fasting blood glucose (mg/dL) X immuno-reactive insulin (mU/mL)}/405.
Figure 1.
Receiver operating characteristics (ROC) curves. Sensitivity represents the true-positive results and 1-specificity, the false-positive results. The best markers have ROC curves that are shifted to the left with areas under the curve near unity. Nondiagnostic markers are represented by diagonals with areas under the ROC curves close to 0.5.
Optimal cut-off point of TG/HDL-C ratio and LDL-C/HDL-C ratio for predicting insulin resistance of subjects categorized by BMI
Table 4 shows the cut-off points of TG/HDL-C ratio and LDL-C/HDL-C ratio for identifying insulin resistance. The optimal cut-off point to identifying insulin resistance for these markers yielded the following values: TG/HDL-C ratio of ≥1.50 and LDL-C/HDL-C ratio of ≥2.14 in non-obese subjects, and ≥2.20, ≥2.25 in overweight. In non-obese subjects, the positive likelihood ratio value indicates that the odds of insulin resistance increased by 2.30-fold if the LDL-C/HDL-C ratio was positive (the value ≥2.14). This ratio was greater for LDL-C/HDL-C ratio than TG/HDL-C ratio. The negative likelihood ratios indicate the extent to which the odds of insulin resistance decrease if the test is negative. These odds also decreased more so for LDL-C/HDL-C ratio. In overweight subjects, these values were similar.
Table 4.
Comparison of triglyceride/HDL cholesterol ratio and LDL cholesterol/ HDL cholesterol ratio for predicting of insulin resistance (HOMA-IR§ > 2.5) of subjects categorized by body mass index
| HOMA-IR§ | |||||||
|---|---|---|---|---|---|---|---|
| Cut-off point Characteristics | ≤2.5 | >2.5 | Sensitivity | Specificity | Positive LR | Negative LR | Accuracy |
| Body mass index† < 25.0 kg/m2 | N = 980 | N = 62 | % | ||||
| Triglycerides/HDL cholesterol ratio <1.50 | 580 | 20 | 0.67 | 0.59 | 1.63 | 0.56 | 59.7 |
| Triglycerides/HDL cholesterol ratio ≥1.50 | 400 | 42 | |||||
| LDL cholesterol/HDL cholesterol ratio <2.14 | 682 | 19 | 0.69 | 0.70 | 2.30 | 0.44 | 69.6 |
| LDL cholesterol/HDL cholesterol ratio ≥2.14 | 298 | 43 | |||||
| Body mass index† ≥ 25.0 kg/m2 | N = 245 | N = 106 | |||||
| Triglycerides/HDL cholesterol ratio <2.20 | 157 | 45 | 0.57 | 0.64 | 1.58 | 0.67 | 62.1 |
| Triglycerides/HDL cholesterol ratio ≥2.20 | 88 | 61 | |||||
| LDL cholesterol/HDL cholesterol ratio <2.25 | 143 | 49 | 0.59 | 0.58 | 1.40 | 0.71 | 57.0 |
| LDL cholesterol/HDL cholesterol ratio ≥2.25 | 102 | 57 | |||||
HOMA-IR, homeostasis of minimal assessment of insulin resistance; LR, likelihood ratio; HDL, high-density lipoprotein; LDL, low-density lipoprotein. §HOMA-IR was calculated using the following formula; (fasting blood glucose (mg/dL) X immunoreactive insulin (mU/mL))/405.
Discussion
In the present study, we examined whether lipid profiles (i.e., TG, HDL-C, LDL-C, TG/HDL-C ratio, LDL-C/HDL-C ratio and non-HDL-C) were associated with insulin resistance in Japanese adults, categorized by body mass index. Most fundamental is the fact that not all overweight or obese persons are insulin resistant. In non-obese subjects, 6.0% of them were insulin resistant, and the best marker of insulin resistance was LDL-C/HDL-C ratio, but HDL-C, TG/HDL-C ratio and non-HDL-C also discriminated insulin resistance. In overweight subjects, the areas of TG and TG/HDL-C ratio were greater than those of the other parameters. The optimal cut-off point to identifying insulin resistance for these markers yielded the following values: TG/HDL-C ratio of ≥1.50 and LDL-C/HDL-C ratio of ≥2.14 in non-obese subjects, and ≥2.20 and ≥2.25, respectively in overweight. The positive likelihood ratio was greatest for LDL-C/HDL-C ratio in non-obese subjects. Lipid ratio of LDL-C/HDL-C might be used as an integrated and simple lipid measure to evaluate insulin resistance in non-obese subjects.
Resistance to insulin-mediated glucose disposal is distributed continuously through the general population [15], and we have no criterion with which to identify a participant as being insulin resistance or insulin sensitive. However, we classified a participant as insulin resistant if he or she was in HOMA-IR > 2.5 [14]. Previous studies have shown that HOMAIR-based insulin resistance scores strongly correlate with glucose clamp-assessed insulin resistance [13,15]. However, the validation was carried out in only a few subjects, and HOMA-IR is less accurate and precise than the glucose clamp in measuring insulin resistance, but this limitation is mitigated when the number of subjects examined is large, as in our study [16].
Hypertriglyceridemia and low HDL-C almost never occurred as isolated disorders, and were nearly always associated with insulin resistance because insulin affects very low-density lipoprotein and HDL-C metabolism [17]. In previous studies, several lipid ratios have been proposed as simple and useful clinical indicators of insulin resistance. The TG/HDL-C, the T-C/HDL-C, and the LDL-C/HDL-C ratio have shown similar potential for insulin resistance, though the reports are not entirely consistent. In 50 white Americans, both TG and TG/HDL-C ratio were acceptable markers for insulin resistance, with area under the ROC curve of 0.763 and 0.770, respectively, but poor predictors in 99 African Americans, with the values at 0.625 and 0.639, respectively [18]. It was demonstrated that the relationship between TG and TG/HDL-C with insulin resistance differs by ethnicity. In 3,014 patients (mean age 54 years; 55% women), TG/HDL-C ratio and T-C/HDL-C ratio were related to insulin resistance assessed by the top quartile of the HOMA-IR, and the area under the ROC curves for predicting insulin resistance with TG/HDL-C ratio and T-C/HDL-C ratio were 0.745 and 0.707, respectively [19]. LDL-C/HDL-C ratio [20] as well as TG/HDL-C ratio [21] have advantages for standardization and identification of patients with an atherogenic lipoprotein profile as a surrogate maker of insulin resistance. Also in our study, both LDL-C/HDL-C and TG/HDL-C ratio were useful makers of insulin resistance, especially in all subjects or non-obese subjects. However, these makers were weaker in overweight subjects. Kimm et al. [21] demonstrated that the lipid ratios of TC/HDL-C, LDL-C/HDL-C and TG/HDL-C, as well as TG and HDL-C, were consistently associated with metabolic syndrome and insulin resistance in participants without metabolic syndrome, though these relations were weaker in participants with metabolic syndrome. Lipid ratios that include information on at least two measures might have a more integrated explanation than single lipid measures such as TG or HDL-C [21].
Some limitations of this study must be considered. First, the response rate was as low as 35% that is usually the casein other conventional community studies in Japan. However, the relatively large sample size enabled the assessment of an extensive array of insulin resistances in relation to lipid profiles. Second, the cross-sectional study design is limited in its ability to eliminate causal relationships between lipid profiles and HOMA-IR. Third, our definition of HOMA-IR is based on a single assessment of FBS and IRI, which may introduce misclassification bias. Therefore the demographics and referral source may limit generalizability.
In conclusion, the present study demonstrated that special lipid profiles are associated with insulin resistance according to BMI in a general population. The ability to identify who is non-obese or overweight and who are insulin resistant could help health care professionals in bringing about lifestyle interventions. In that context, use of the cutoff-points of LDL-C/HDL-C ratio or TG/HDL-C ratio described in this report is simple and useful. The present data documented that insulin resistance was present even in subjects with BMI within the normal range. Further prospective population-based studies are needed to investigate the changes in lipid metabolism by lifestyle interventions.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
RK, YT, and KK participated in the design of the study, performed the statistical analysis and drafted the manuscript. NO, TaK, and ToK contributed to acquisition of data and its interpretation. ST and MA contributed to conception and design of the statistical analysis. TM conceived of the study, participated in its design, coordination and helped to draft the manuscript. All authors read and approved the manuscript.
Contributor Information
Ryuichi Kawamoto, Email: rykawamo@yahoo.co.jp.
Yasuharu Tabara, Email: tabara@m.ehime-u.ac.jp.
Katsuhiko Kohara, Email: koharak@m.ehime-u.ac.jp.
Tetsuro Miki, Email: tmiki@m.ehime-u.ac.jp.
Tomo Kusunoki, Email: rykawamo@yahoo.co.jp.
Shuzo Takayama, Email: spya6yq9@rhythm.ocn.ne.jp.
Masanori Abe, Email: masaben@m.ehime-u.ac.jp.
Tateaki Katoh, Email: rykawamo@yahoo.co.jp.
Nobuyuki Ohtsuka, Email: ohtsukan@hotmail.com.
Acknowledgements
This work was supported in part by a grant-in-aid for Scientific Research from the Foundation for Development of Community (2009).
References
- Organization of the Ministry of Health, Labour and Welfare. 2006. pp. 173–175.http://www.mhlw.go.jp/bunya/kenkou/eiyou09/dl/01-03.pdf
- Lee IM, Manson JE, Hennekens CH, Paffenbarger RS Jr. Body weight and mortality. A 27-year follow-up of middle-aged men. JAMA. 1993;270(23):2823–2828. doi: 10.1001/jama.270.23.2823. [DOI] [PubMed] [Google Scholar]
- Manson JE, Willett WC, Stampfer MJ, Colditz GA, Hunter DJ, Hankinson SE. Body weight and mortality among women. N Engl J Med. 1995;333(11):677–685. doi: 10.1056/NEJM199509143331101. [DOI] [PubMed] [Google Scholar]
- Calle EE, Thun MJ, Petrelli JM, Rodriguez C, Heath CW. Body-mass index and mortality in a prospective cohort of U.S. adults. N Engl J Med. 1999;341(15):1097–1105. doi: 10.1056/NEJM199910073411501. [DOI] [PubMed] [Google Scholar]
- Ruderman NB, Schneider SH, Berchtold P. The "metabolically-obese," normal-weight individual. Am J Clin Nutr. 1981;34(8):1617–1621. doi: 10.1093/ajcn/34.8.1617. [DOI] [PubMed] [Google Scholar]
- Kawamoto R, Ohtsuka N, Ninomiya D, Nakamura S. Carotid atherosclerosis in normal-weight metabolic syndrome. Intern Med. 2007;46(21):1771–1777. doi: 10.2169/internalmedicine.46.0261. [DOI] [PubMed] [Google Scholar]
- Trirogoff ML, Shintani A, Himmelfarb J, Ikizler TA. Body mass index and fat mass are the primary correlates of insulin resistance in nondiabetic stage 3-4 chronic kidney disease patients. Am J Clin Nutr. 2007;86(6):1642–1648. doi: 10.1093/ajcn/86.5.1642. [DOI] [PubMed] [Google Scholar]
- Reaven GM. Insulin resistance, the insulin resistance syndrome, and cardiovascular disease. Panminerva Med. 2005;47(4):201–210. [PubMed] [Google Scholar]
- Laws A, Reaven GM. Evidence for an independent relationship between insulin resistance and fasting plasma HDL-cholesterol, triglyceride and insulin concentrations. J Intern Med. 1992;231(1):25–30. doi: 10.1111/j.1365-2796.1992.tb00494.x. [DOI] [PubMed] [Google Scholar]
- McLaughlin T, Abbasi F, Cheal K, Chu J, Lamendola C, Reaven G. Use of metabolic markers to identify overweight individuals who are insulin resistant. Ann Intern Med. 2003;139(10):802–809. doi: 10.7326/0003-4819-139-10-200311180-00007. [DOI] [PubMed] [Google Scholar]
- Jeppesen J, Facchini FS, Reaven GM. Individuals with high total cholesterol/HDL cholesterol ratios are insulin resistant. J Intern Med. 1998;243(4):293–298. doi: 10.1046/j.1365-2796.1998.00301.x. [DOI] [PubMed] [Google Scholar]
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502. [PubMed] [Google Scholar]
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- Taniguchi A, Fukushima M, Sakai M, Kataoka K, Miwa K, Nagata I, Doi K, Tokuyama K, Nakai Y. Insulin-sensitive and insulin-resistant variants in nonobese Japanese type 2 diabetic patients. The role of triglycerides in insulin resistance. Diabetes Care. 1999;22(12):2100–2101. doi: 10.2337/diacare.22.12.2100. [DOI] [PubMed] [Google Scholar]
- Yeni-Komshian H, Carantoni M, Abbasi F, Reaven GM. Relationship between several surrogate estimates of insulin resistance and quantification of insulin-mediated glucose disposal in 490 healthy nondiabetic volunteers. Diabetes Care. 2000;23(2):171–175. doi: 10.2337/diacare.23.2.171. [DOI] [PubMed] [Google Scholar]
- Bonora E, Kiechl S, Willeit J, Oberhollenzer F, Egger G, Targher G, Alberiche M, Bonadonna RC, Muggeo M. Prevalence of insulin resistance in metabolic disorders: the Bruneck Study. Diabetes. 1998;47(10):1643–1649. doi: 10.2337/diabetes.47.10.1643. [DOI] [PubMed] [Google Scholar]
- Lewis GF, Uffelman KD, Szeto LW, Steiner G. Effects of acute hyperinsulinemia on VLDL triglyceride and VLDL apoB production in normal weight and obese individuals. Diabetes. 1993;42(6):833–842. doi: 10.2337/diabetes.42.6.833. [DOI] [PubMed] [Google Scholar]
- Kim-Dorner SJ, Deuster PA, Zeno SA, Remaley AT, Poth M. Should triglycerides and the triglycerides to high-density lipoprotein cholesterol ratio be used as surrogates for insulin resistance? Metabolism. 2010;59(2):299–304. doi: 10.1016/j.metabol.2009.07.027. [DOI] [PubMed] [Google Scholar]
- Kannel WB, Vasan RS, Keyes MJ, Sullivan LM, Robins SJ. Usefulness of the triglyceride-high-density lipoprotein versus the cholesterol-high-density lipoprotein ratio for predicting insulin resistance and cardiometabolic risk (from the Framingham Offspring Cohort) Am J Cardiol. 2008;101(4):497–501. doi: 10.1016/j.amjcard.2007.09.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamada M, Makita S, Abiko A, Naganuma Y, Nagai M, Nakamura M. Low-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio as a useful marker for early-stage carotid atherosclerosis. Metabolism. 2010;59(5):653–657. doi: 10.1016/j.metabol.2009.09.009. [DOI] [PubMed] [Google Scholar]
- Kimm H, Lee SW, Lee HS, Shim KW, Cho CY, Yun JE, Jee SH. Associations between lipid measures and metabolic syndrome, insulin resistance and adiponectin. - Usefulness of lipid ratios in Korean men and women - Circ J. 2010;74(5):931–937. doi: 10.1253/circj.CJ-09-0571. [DOI] [PubMed] [Google Scholar]