Abstract
Although the benefits of adoptive T-cell therapy can be increased by prior lymphodepletion of the recipient, this process usually requires chemotherapy or radiation. Vaccination with antigens to which the transferred T cells respond should be a less toxic means of enhancing their activity, but to date such vaccines have not been effective. We, therefore, determined which characteristics an adenoviral vaccine has to fulfill to optimally activate and expand adoptively transferred antigen-specific T cells in vivo. We evaluated (i) antigen, (ii) flagellin, a Toll-like receptor (TLR) 5 ligand, and (iii) an inhibitor of the antigen-presenting attenuator A20. Vaccination of mice before T-cell transfer with a vaccine that contained all three components dramatically enhanced the effector function of ovalbumin (OVA)-specific T cells as judged by the regression of established B16-OVA tumors compared to one- and two-component vaccines. Immunization with the three-component vaccine induced a strong Th1 environment, which was critical for the observed synergy and proved as effective as cytoxan-induced lymphodepletion in enhancing in vivo T-cell expansion. Thus, the combination of our vaccine with T-cell therapy has the potential to enhance and broaden adoptive cellular immunotherapy.
Introduction
Adoptive immunotherapy with antigen-specific cytotoxic T cells has shown promise in the treatment of viral disease and malignancy.1,2,3,4,5 One of the primary obstacles to the broader effectiveness of the approach is the lack of expansion and persistence of T cells with sustained cytotoxic activity in the peripheral blood following infusion. Instead, infused T cells may become anergic, switch to a Th2 functional phenotype, or simply disappear. Lymphodepletion with chemotherapy or irradiation followed by administration of exogenous lymphostimulating cytokines is currently one of the most promising strategies for enhancing expansion and efficacy,1,6 but may not always maintain a Th1 phenotype and, by producing nonspecific destruction of the immune system, can be lethal.
Vaccines have the potential to boost both endogenous and adoptive T-cell therapies without such adverse effects. However, the results of most clinical cancer vaccine studies have been disappointing: even when expansion is obtained it may still be at the cost of losing the desired proinflammatory/cytotoxic (Th1) polarity of the cellular response.7,8 The use of adenoviral vectors encoding vaccine antigens has been particularly problematic in this regard.9
Our goal was to develop a means of successfully boosting the in vivo expansion of adoptively transferred antigen-specific T cells, while retaining their cytotoxic properties. We sought to enhance the immunostimulatory capacity of resident host dendritic cells (DCs) by including in our adenoviral vaccine both a Toll-like receptor (TLR) ligand as a DC stimulator, and an antagonist of A20, a ubiquitin-modifying enzyme that downregulates TLR-induced responses in these DCs.10,11 Our results show that such a compound vaccine creates and sustains a strong Th1 environment, which efficiently enhances the expansion of adoptively transferred T cells and sustains their cytotoxic activity.
Results
Ad-shAF induces DC maturation and activation in vivo
To determine whether a single adenoviral vector administered in vivo could both activate TLR and silence A20 in DC, we generated a recombinant adenoviral vector, which coexpresses an A20-specific short-hairpin RNA (shA) and a secretory form of flagellin (F) that binds TLR5 (Ad-shAF; Supplementary Figure S1). Flagellin12,13 was chosen because TLR5 is expressed in vivo on the cell surface of DCs isolated from lymph nodes, and flagellin-induced DC activation further upregulates TLR5 expression, whereas silencing of A20 did not (Supplementary Figure S2). To confirm silencing of A20 and flagellin expression in vivo, DCs were isolated from draining lymph nodes 24 hours after intramuscular injection of Ad-empty or Ad-shAF. Reverse transcription-PCR amplification of RNA from DCs from Ad-shAF-injected mice demonstrated around 60% silencing of A20 (P < 0.01) and expression of flagellin, whereas DCs from control or Ad-empty-injected mice showed the converse pattern—expression of A20 but absence of flagellin (Figure 1a,b).
Figure 1.
Ad-shAF induces dendritic cell (DC) maturation and activation in vivo. Mice were immunized intramuscularly (i.m.) with 1 × 1010 virus particles of Ad-shAF (shAF), Ad-empty (Ad) in 50 µl sterile PBS or PBS control. CD11c+ DCs were isolated 24 hours postinjection and (a) flagellin expression was detected by reverse transcription-PCR (RT-PCR) and (b) silencing of A20 by quantitative RT-PCR. (c) Mice were immunized i.m. as described above with Ad-shAF (shAF), Ad-shA (shA), Ad-F (F), or Ad-shGFP (shG) or PBS control. Inguinal draining lymph node cell suspensions were prepared 24 hours later. Surface expression of co-stimulatory and MHC class II molecules on CD11c+ gated DCs as determined by fluorescence-activated cell-sorting analysis. Experiments were repeated three times with similar results. MFI, mean fluorescence intensity. (d) Cytokine production determined by enzyme-linked immunosorbent assay of draining lymph node purified CD11c+ DCs (5 × 105 cells/ml) after in vitro culture for 24 hours without stimulation. Ad, adenovirus; Ad-shAF, adenoviral vector coexpressing an A20-specific short-hairpin RNA and a secretory form of flagellin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; IL, interleukin; PBS, phosphate-buffered saline; TNF, tumor necrosis factor.
To determine whether simultaneously silencing A20 and activating DCs through TLR5 is superior to A20 silencing or TLR5 activation alone, we compared the ability of Ad-shA, Ad-F, and Ad-shAF vaccines to activate and mature DCs in vivo. C57BL/6 mice were immunized by intramuscular injection into the hind leg with 1 × 1010 virus particles of Ad-shA, Ad-F, Ad-shAF, Ad-shGFP, or phosphate-buffered saline. At 24 hours after vaccination, CD11c-positive DCs were purified from draining inguinal lymph nodes by magnetic beads. Cell surface expression of CD40, CD80, and CD86, and MHC class II antigen was determined by fluorescence-activated cell-sorting analysis. Vaccination with Ad-F, Ad-shA, and Ad-shAF induced CD40 and CD86 expression. Upregulation of CD80 and MHC class II expression, however, was only observed in recipients of adenoviral vaccines encoding flagellin (Ad-F and Ad-shAF; Figure 1c and Supplementary Figure S3a). In addition, the Ad-shAF vaccine only induced DC activation in draining and not in systemic lymph nodes (Supplementary Figure S3b). DC production of proinflammatory cytokines [interleukin-12p70 (IL-12p70), IL-6, and tumor necrosis factor-α] 24 hours postvaccination (Figure 1d) showed that DCs from Ad-shAF-immunized mice produced significantly higher levels (P < 0.05) of IL-12p70 and IL-6 in comparison to all other vaccines or phosphate-buffered saline control. Ad-shAF also induced significantly higher levels of tumor necrosis factor-α in comparison to Ad-shA, Ad-shGFP, and phosphate-buffered saline.
Ad-shAF/Ad-OVA vaccination enhances the effector function of adoptively transferred OT-I T cells
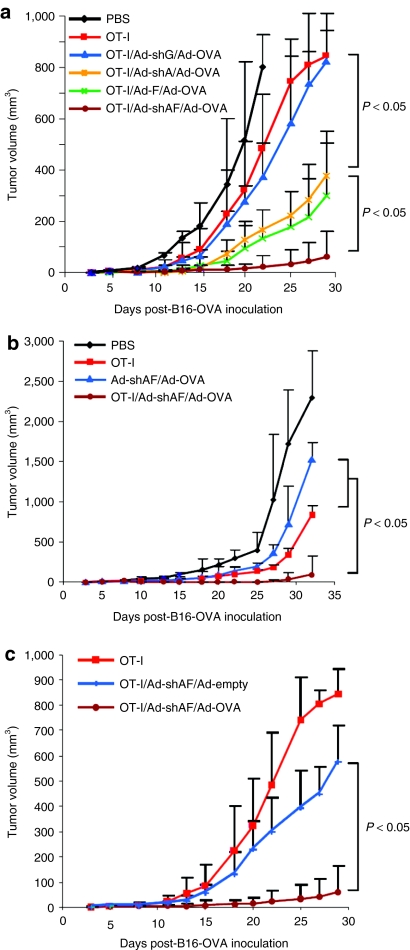
Because Ad-shAF induces superior DC maturation and activation in vivo compared to Ad-shA and Ad-F, we next examined whether vaccinating mice with Ad-shAF in combination with an adenovirus encoding ovalbumin (Ad-OVA) enhanced the effector function of adoptively transferred OT-I-specific T cells. We injected B-16/OVA tumor cells subcutaneously into mice, and on day 5 we vaccinated the animals with a single dose of Ad-shAF/Ad-OVA; control groups included Ad-shA/Ad-OVA, Ad-F/Ad-OVA, Ad-OVA, or no vaccine. On day 7, the mice received a single intravenous injection of ex vivo activated OT-I-specific T cells. Subsequent tumor growth was followed by standard caliper measurements. OT-I T-cell transfer in combination with Ad-shAF/Ad-OVA significantly reduced tumor growth compared to all other experimental groups tested. In particular, OT-I T-cell transfer alone or in combination with Ad-OVA vaccination only marginally inhibited tumor growth. Although Ad-shA/Ad-OVA or Ad-F/Ad-OVA vaccination enhanced the antitumor effects of OT-I T cells, the benefit was significantly less than in Ad-shAF/Ad-OVA-vaccinated mice (Figure 2a). Ad-shAF/Ad-OVA vaccination alone had a marginal effect on tumor growth, so that the maximal therapeutic effect required both the vaccine and the adoptively transferred T cells (Figure 2b). In addition, Ad-shAF vaccination before OT-I T-cell transfer resulted in only a marginal enhancement of antitumor effects, indicating that the provision of antigen (Ad-OVA) is important (Figure 2c). Hence, a vaccine that provides antigen, TLR5 stimulation, and silencing of A20 affords superior enhancement of the antitumor effects of adoptively transferred OT-I T cells than a vaccine encoding just two of these three components.
Figure 2.
Ad-shAF/Ad-OVA vaccination enhances the antitumor activity of adoptively transferred T cells. C57BL/6 mice were inoculated subcutaneously with B16-OVA tumor cells (5 × 105) and 5 days later were immunized i.m. with 1 × 1010 virus particles of adenoviral vaccine or PBS control. On day 7 mice received 2 × 106 activated OT-I T cells by tail-vein injection. (a) Comparison of antitumor activity of OT-I/Ad-OVA/Ad-shAF, OT-I/Ad-OVA/Ad-shA, OT-I/Ad-OVA/Ad-F, OT-I/Ad-OVA/Ad-shGFP, OT-I, or PBS. (b) Comparison of antitumor activity of OT-I/Ad-OVA/Ad-shAF, OT-I, Ad-OVA/Ad-shAF, or PBS. (c) Comparison of antitumor activity of OT-I/Ad-OVA/Ad-shAF, OT-I/Ad-shAF/Ad-empty, or OT-I. Tumor growth curves (n = 5 mice/group) represent one of two repeated experiments. Ad-shAF, adenoviral vector coexpressing an A20-specific short-hairpin RNA and a secretory form of flagellin; OVA, ovalbumin; PBS, phosphate-buffered saline.
Ad-shAF/Ad-OVA vaccine recruits adoptively transferred OT-I T cells to the vaccine site and induces their expansion
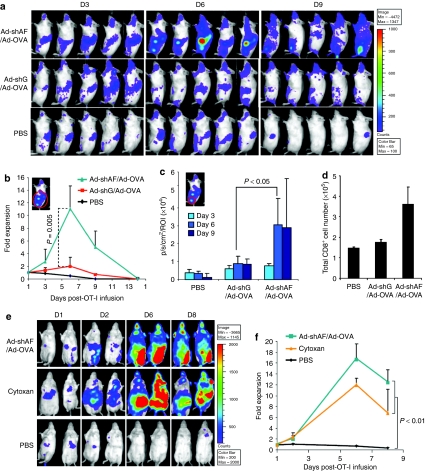
To investigate whether the Ad-shAF/Ad-OVA vaccine induces the expansion of OT-I T cells in vivo, we transduced these cells with a retroviral vector encoding enhanced firefly luciferase (efluc) and tracked them in vivo using bioluminescence imaging. Albino C57BL/6 mice were injected intravenously with efluc-OT-I T cells 2 days after vaccination with Ad-OVA or Ad-shAF/Ad-OVA. Ad-shAF/Ad-OVA vaccination recruited adoptively transferred OT-I T cells to the vaccination site, whereas Ad-OVA did not (Figure 3a,c). More importantly, OT-I T cells expanded 11-fold in vivo after Ad-shAF/Ad-OVA vaccination, which was significantly higher than after Ad-OVA vaccination (twofold; Figure 3a,b). To determine vaccine-induced local expansion of T cells with alternative specificities, we determined absolute CD8-positive T-cell numbers in draining lymph nodes postvaccination. There was a twofold increase in the number of CD8-positive T cells 6 days postvaccinations in mice receiving Ad-shAF/Ad-OVA versus control vaccine (Ad-shG/Ad-OVA; Figure 3d). To compare Ad-shAF/Ad-OVA-induced T-cell expansion to T-cell expansion induced by lymphodepletion, mice were either vaccinated with Ad-shAF/Ad-OVA or treated with a lymphodepleting dose of cytoxan 2 days before OT-I T-cell infusion (Figure 3e,f). In both groups of mice, OT-I T-cell expansion was similar indicating that OT-I T-cell expansion in vivo can be achieved without the unwanted side effects of chemotherapeutic agents.
Figure 3.
Ad-shAF/Ad-OVA recruits adoptively transferred OT-I T cells and induces their expansion in vivo. (a–c) In vivo expansion of adoptively transferred efluc-OT-I T cells: Albino C57BL/6 mice were immunized with 1 × 1010 virus particles of Ad-shAF/Ad-OVA, Ad-OVA/Ad-shGFP, or PBS control followed by intravenous (i.v.) injection of efluc expressing OT-I T cells 2 days later. The expansion of efluc-OT-I T cells was determined by in vivo imaging (a). Quantification of (b) total body and (c) vaccine site bioluminescence signal. (d) To determine the expansion of T cells with alternative specificity, the absolute numbers of CD8-positive T cells was determined in draining lymph nodes 6 days postvaccinations with Ad-shAF/Ad-OVA or Ad-shG/Ad-OVA. (e and f) Comparison of boosting effects of Ad-shAF/Ad-OVA with cytoxan: C57BL/6 mice were immunized with 1 × 1010 virus particles of Ad-shAF/Ad-OVA (intramuscular) or 250 mg/kg cytoxan (intraperitoneal) followed by i.v. injection of 2 × 106 efluc-OT-I T cells 2 days later. Values shown are mean ± SEM. Ad-shAF, adenoviral vector coexpressing an A20-specific short-hairpin RNA and a secretory form of flagellin; OVA, ovalbumin; PBS, phosphate-buffered saline.
Ad-shAF/Ad-OVA vaccine augments the effector function of OT-I T cells in a CD4-dependent manner
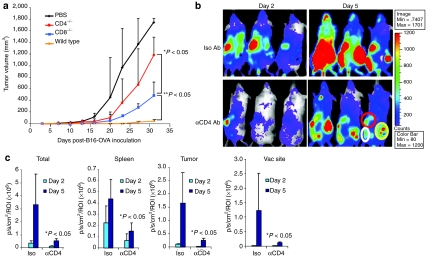
To investigate whether endogenous CD4- or CD8-positive T cells are critical for enhancing the antitumor effects of infused OT-I T cells, tumor-bearing CD4−/−, CD8−/−, or wild-type C57BL/6 mice were vaccinated with Ad-shAF/Ad-OVA 2 days before transfer of OT-I T cells. In both CD4−/− and CD8−/− mice, the antitumor activity of the Ad-shAF/Ad-OVA vaccine and OT-I T cells was reduced, but the impact was much greater in CD4−/− mice (CD4−/− versus CD8−/−: P < 0.05; Figure 4a). To further investigate the contribution of CD4-positive T cells to the expansion of adoptively transferred OT-I T cells, we vaccinated tumor-bearing albino C57BL/6 mice with Ad-shAF/Ad-OVA and injected them with a murine CD4–depleting antibody. Two days later, efluc-OT-I T cells were injected intravenously and tracked by bioluminescence imaging. At all sites measured (spleen, tumor, and vaccination site), OT-I T-cell expansion was significantly reduced in CD4-depleted mice compared to recipients of control antibodies (Figure 4b,c).
Figure 4.
Ad-shAF/Ad-OVA boosts adoptive OT-I T-cell therapy in CD4-dependent manner. (a) Comparison of antitumor activity: wild-type C57BL/6 mice, CD8−/− mice, or CD4−/− mice were inoculated subcutaneously with B16-OVA tumor cells (5 × 105) and 5 days later were immunized intramuscularly with 1 × 1010 virus particles of Ad-shAF/Ad-OVA followed by injection of activated OT-I T cells (2 × 106) 2 days later. (b) Comparison of in vivo expansion: albino C57BL/6 mice were immunized with 1 × 1010 virus particles of Ad-OVA/Ad-shAF followed by intraperitoneal injection of anti-CD4 antibody or isotype; efluc OT-I T cells were injected 2 days later. The expansion of efluc-OT-I T cells was determined by in vivo bioluminescence imaging. (c) Quantification of spleen (red circle), tumor (yellow circle), and vaccination site (white circle) bioluminescence signal (values shown are mean ± SEM; WT versus CD4 depletion: P < 0.05). Ad-shAF, adenoviral vector coexpressing an A20-specific short-hairpin RNA and a secretory form of flagellin; OVA, ovalbumin; PBS, phosphate-buffered saline; ROI, region of interest.
Ad-shAF/Ad-OVA vaccine induces robust Th1, Th2, and Th17 responses in B16-OVA-bearing mice
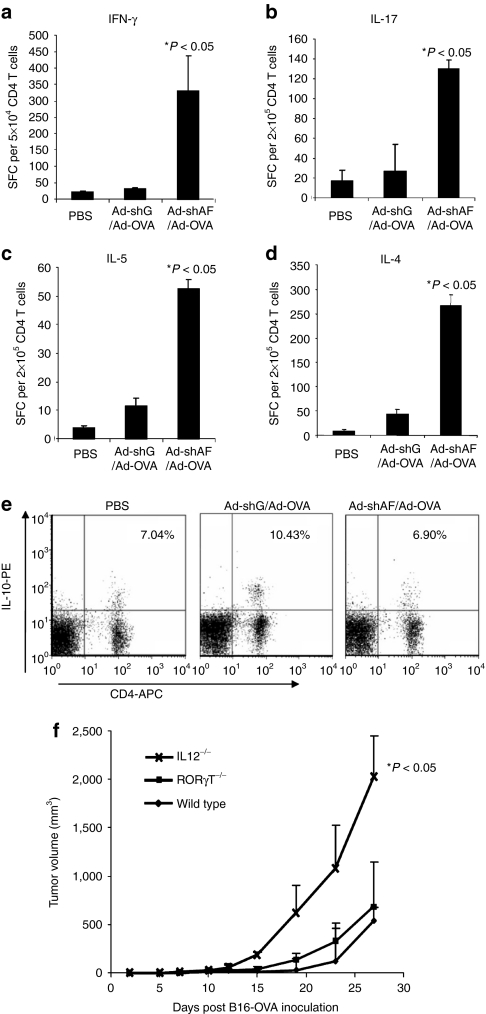
As CD4-positive T cells make a critical contribution to the enhanced antitumor effects of OT-I T cells in Ad-shAF/Ad-OVA immunized mice, we investigated which CD4-positive T-cell subset is activated by the Ad-shAF/Ad-OVA vaccine. C57BL/6 mice-bearing large B16-OVA tumors (1 cm in diameter) were vaccinated with Ad-shAF/Ad-OVA or Ad-shGFP/Ad-OVA. Seven days after vaccination, isolated splenocytes were stimulated with OT-II peptide pulsed bone marrow DCs and Th1, Th2, and Th17 CD4-positive T cells enumerated using interferon-γ, IL-4, IL-5, or IL-17 enzyme-linked immunosorbent spot (ELISPOT) assays. Ad-shAF/Ad-OVA vaccination increased the number of interferon-γ, IL-4, IL-5, and IL-17 secreting CD4-positive T cells compared to mice vaccinated with Ad-OVA (Figure5a–d). Following vaccination, we also used fluorescence-activated cell-sorting analysis of lymphocytes from draining lymph nodes to determine the number of CD4-positive T cells with a Treg phenotype and which also produced IL-10. Although all groups of mice had a similar frequency of FoxP3-positive Tregs (8–9%; data not shown), those receiving Ad-OVA had an increased frequency of OTII-specific, IL-10-producing CD4-positive T cells, whereas Ad-shAF/Ad-OVA-vaccinated mice did not (Figure 5e). Thus, Ad-shAF/Ad-OVA vaccine induces robust Th1, Th2, and Th17 responses, but prevents the activation of inhibitory, IL-10-secreting T cells in B16-OVA-bearing mice.
Figure 5.
Ad-shAF/Ad-OVA induces robust Th1, Th2, and Th17 responses in vivo. C57BL/6 mice were immunized intramuscularly (i.m.) with 1 × 1010 virus particles of Ad-shAF/Ad-OVA or Ad-shGFP/Ad-OVA in 50 µl sterile PBS or PBS control. Isolated CD4+ T cells from spleen were subjected to (a) IFN-γ, (b) IL-17, (c) IL-5, or (d) IL-4 ELISPOT assays. (e) Lymph node cells were prepared and subjected for IL-10 intracellular staining (Ad-shAF/Ad-OVA versus Ad-shGFP/Ad-OVA: P < 0.05). (f) Comparison of antitumor activity: wild-type C57BL/6 mice, CD4−/− mice, IL12−/− mice, or RORγT−/− mice were inoculated subcutaneously with B16-OVA tumor cells (5 × 105) and 8 days later were immunized i.m. with 1 × 1010 virus particles of Ad-shAF/Ad-OVA followed by intravenous injection of activated OT-I T cells (2 × 106) through tail vein at day 10 (P < 0.05, wild-type mice versus IL-12−/− mice). Ad-shAF, adenoviral vector coexpressing an A20-specific short-hairpin RNA and a secretory form of flagellin; IFN, interferon; IL, interleukin; OVA, ovalbumin; PBS, phosphate-buffered saline; SFC, spot-forming cell.
To determine which of the Th1, Th2, and Th17 responses induced by Ad-shAF/Ad-OVA vaccine was most important for enhancing the effector function of adoptively transferred T cells, we used IL-12−/− and RORγT−/− mice. Although IL-12−/− mice are deficient in Th1 responses, RORγT−/− mice lack Th17 T cells. The beneficial effect of Ad-shAF/Ad-OVA vaccination was lost in IL-12−/− mice, in contrast full activity was seen in RORγT−/− mice (Figure 5f).
Discussion
Our results demonstrate that an adenoviral vaccine that provides antigen, stimulates TLR5, and inhibits the antigen-presenting attenuator A20, can efficiently enhance the effector function of adoptively transferred T cells by creating a strong Th1 environment.
Although the use of adoptive T-cell therapies to treat viral disease and malignancies has had some clinical success, the broader application of this therapeutic approach is hampered by limited in vivo expansion of adoptively transferred T cells in patients, who do not receive lymphodepleting chemotherapy and/or radiation before T-cell transfer. Vaccines are one promising approach to overcome this limitation, however, current therapeutic vaccines do not activate all arms of the immune system, which has proven critical for successful protective vaccines.9,14
One strategy to enhance therapeutic vaccines is the use of TLR ligands, including lipopolysaccharide, double-stranded RNA (poly I:C), single-stranded RNA (imiquimod), bacterial DNA (CpG ODN), peptidoglycan, and flagellin.15,16 Of these, flagellin is the only TLR ligand, which is a protein and therefore ideally suited to be included in adenoviral vaccines. Here, we show that incorporation of flagellin into an adenoviral vaccine induces in vivo DC maturation and production of proinflammatory cytokines. Flagellin not only activated DCs, but also induced the expression of its ligand, TLR5, which might further amplify flagellin-induced DC activation. Vaccinating mice intramuscularly with Ad-F/Ad-OVA before the infusion of OT-I T cells significantly enhanced the effector function of these cells, but with only transient therapeutic effects. This finding is in agreement with a recent report in which flagellin had limited adjuvant activity in therapeutic vaccines.17
To further enhance TLR-mediated DC activation in vivo, we combined TLR activation with inhibition of the ubiquitin ligase A20, an antigen-presenting attenuator that induces the degradation of key molecules of the TLR-signaling cascade.10,11 The Ad-shAF vaccine significantly increased the production of immunostimulatory cytokines by DCs in comparison to Ad-F or Ad-shA. Combining TLR activation and A20 inhibition in a single in vivo vaccine is simpler and less toxic than the alternative of preparing and silencing DCs ex vivo before injecting them systemically with TLR agonists.18 We found that in addition to TLR activation and A20 inhibition, the provision of antigen is necessary for optimal therapeutic effect. We vaccinated mice 2 days before T-cell infusion, to provide antigen that was present outside the locally immunosuppressive tumor microenvironment, and vaccine-induced Th1 cytokines at the time of T-cell infusion. This approach should help prevent the immediate induction of anergy of infused T cells; although, it is possible that vaccinating mice immediately after T-cell transfer would be equally as efficient.19
The Ad-shAF/Ad-OVA vaccine induced strong Th1, Th2, and Th17 responses without increasing the frequency of Tregs. Although the results of our IL-12−/− mice experiments suggest that the induction of Th1 cells are critical for boosting adoptively transferred T cells in vivo, we cannot completely exclude a contribution from Th2 and Th17 cells. In addition, TLR5-mediated activation of the innate immune system may also contribute to the observed effects. Overall, our results are in agreement with findings from protective vaccine studies, which have highlighted the critical role of Th1 responses to confer protection.20
One of the most successful strategies to enhance T-cell therapy is to lymphodeplete patients with chemotherapy and/or irradiation before adoptive T-cell transfer.6 However, cytotoxic agents have unwanted adverse effects. Indeed, cytoxan induced a significant decrease in the total number of peripheral blood leukocytes and an increase in proinflammatory cytokine levels in the peripheral blood of treated mice, whereas, our vaccine did not (Supplementary Figure S4a,b). Moreover, cytotoxic agents also induce the nonspecific destruction of the immune system including Th1 T cells, which we found to be critical for the expansion and effector function of infused T cells. Direct comparison revealed that the vaccine-induced Th1 environment is as efficient as cytoxan-induced lymphodepletion in enhancing the expansion of adoptively transferred T cells. Whether lymphodepletion or the creation of a Th1 environment is an optimal strategy for enhancing T-cell expansion may depend on the clinical situation. If debulking of a tumor mass is required, cytotoxic agents have obvious advantages, whereas for patients with minimal residual disease or nonmalignant diseases, in whom therapeutic T-cell products are to be used, a less toxic approach using a Th1-inducing vaccine would be preferable.
In summary, the vaccine we describe may improve the effectiveness of adoptive T-cell immunotherapies, and the combination of such vaccination with T-cell transfer should allow the ex vivo generation and reinfusion of high-affinity T cells whose function can be sustained in the Th1-supporting environment the vaccine induces.
Materials and Methods
Mice and cell lines. C57BL/6J, Albino C57BL/6J-Tyr-2J/J, H-2Kb/OT-I-TCR(OT-I), CD4−/−, CD8−/−, RORγt−/−, and IL12−/− (IL12btm1Jm) mice were purchased from Jackson Laboratories (Bar Harbor, ME) and maintained in a pathogen-free mouse facility at Baylor College of Medicine according to institutional guidelines. This study was approved by the Institutional Animal Care and Use Committees of Baylor College of Medicine. The GP+E-86 producer cell line was purchased from ATCC (Manassas, VA).
Generation of recombinant replication-defective adenoviruses. An Ad-Easy system (E1 and E3 deleted; Quantum Biotechnologies, Palo Alto, CA) was used to construct and generate replication-defective adenoviruses.21 The recombinant replication-competent Ad-shAF virus containing the A20 short-hairpin RNA under H1 RNA promoter and flagellin under control of the cytomegalovirus promoter was constructed by PCR cloning. Briefly, a modified flagellin gene with a signal leader sequence from human tyrosinase was amplified by PCR using Salmonella enterica serovar Typhimurium DNA (ATCC) as a template. Correct insertion of shA20 and flagellin into the adenoviral backbone was determined by sequencing. Expression of flagellin and shA20 was confirmed by reverse transcription-PCR.
Adoptive transfer and vaccination experiments. For adoptive transfer experiments, splenocytes from OT-I TCR transgenic mice were activated with 10 µg/ml OT-I peptide and 300 U IL-2/ml for 7 days in vitro. At the time of transfer, >95% of cells were CD3- and CD8-positive. For in vivo experiments, 5 × 105 B16-OVA tumor cells were injected subcutaneously into the right flank of syngeneic mice. On day 5 after tumor inoculation, the mice were randomly divided into groups (n = 5/group) and vaccinated intramuscularly into the left leg with 1 × 1010 virus particles of adenoviral vaccine. At 2 days after vaccination, mice received 2 × 106 OT-I T cells by tail-vein injection. Tumor volumes were measured every 2–3 days with an electronic caliper.
Retroviral gene transfer of mouse spleen T lymphocytes. The construction of pMSCV-ffFLuc-pIRES-Thy1.1 was previously described.22 Vesicular stomatitis virus-G pseudotyped retroviral particles were used to generate a GP+E-86 ecotropic packaging cell line. Splenocytes from OT-I TCR transgenic mice were activated with 1 µg/ml concanavalin A (Sigma, St Louis, MO) in RPMI (Invitrogen, Carlsbad, CA) with 10% fetal calf serum (HyClone, Logan, UT), 2 mmol/l GlutaMAX-I (complete RPMI; Invitrogen). After 24 hours, retrovirus was added in the presence of 5 µg/ml of polybrene (Sigma) and spinfected at 1,000g for 1.5 hours. The following day, cells were washed and expanded in fresh complete RPMI and 300 U IL-2/ml. Cells were stained 3–4 days after transduction with anti-mouse Thy1.1 PE (BD Biosciences, Franklin Lakes, NJ). Greater than 97% of Thy1.1-positive cells were CD3/CD8-positive. Transduced T cells were washed in phosphate-buffered saline and injected to albino C57BL/6 mice via tail vein. T-cell expansion was determined by in vivo bioluminescence imaging.
In vivo bioluminescence imaging. Isoflurane-anesthetized animals were imaged using an IVIS system (Caliper Life Sciences, Hopkinton, MA) 10 minutes after intraperitoneal injection of 150 mg/kg -luciferin (Xenogen, Alameda, CA). Living image software was used to analyze the data. A constant region of interest was drawn over the tumor regions and the intensity of the signal measured as total photon/second/cm2/region of interest as previously described.23 Mice were killed according to preset criteria, in accordance with Baylor College of Medicine's Center for Comparative Medicine guidelines.
Flow cytometric analysis. Flow cytometric analysis of DCs and T cells were performed as previously described.18,24 Stained cells were analyzed on a FACScalibur instrument (BD, Becton Dickinson, Mountain View, CA) using CellQuest software (BD) for all flow cytometric analyses.
Enzyme-linked immunosorbent assays. Cytokine concentrations in DC cultures were measured by commercially available two-site sandwich enzyme-linked immunosorbent assays from R&D (IL-12p70; Minneapolis, MN) and BD Bioscience (IL-6 and tumor necrosis factor-α) according to manufacturer's instructions.18,24
ELISPOT assays. ELISPOT assays of isolated CD4-positive T cells were performed as described previously.18,24 The results were evaluated in a blinded fashion by ZellNet Consulting (New York, NY) with an automated ELISPOT reader system, using KS ELISPOT 4.3 software.
Statistical analysis. For statistical analysis, we used Student's t-test with a 95% confidence limit, defined as P < 0.05. Results are typically presented as means ± SEM For the bioluminescence experiments, intensity signals were log-transformed and summarized using mean ± SD at baseline and multiple subsequent time points for each group of mice. Changes in intensity of signal from baseline at each time point were calculated and compared using paired t-tests or Wilcoxon signed-ranks test.
SUPPLEMENTARY MATERIAL Figure S1. Scheme of adenoviral constructs. Figure S2. TLR5 expression is upregulated in vivo after Ad-shAF vaccination. Figure S3. Ad-shAF induces DC maturation and activation in vivo. Figure S4. Systemic side effects of Ad-shAF and Cytoxan.
Acknowledgments
We thank Malcolm K. Brenner for the helpful discussion and advice, Aaron Foster and Adham Bear for advice and technical assistance. This work was supported by grants from the National Institutes of Health (P50 CA126752), and a SCOR grant from the Leukemia and Lymphoma Society (R7016-09).
Supplementary Material
Scheme of adenoviral constructs.
TLR5 expression is upregulated in vivo after Ad-shAF vaccination.
Ad-shAF induces DC maturation and activation in vivo.
Systemic side effects of Ad-shAF and Cytoxan.
REFERENCES
- Rosenberg SA, Restifo NP, Yang JC, Morgan RA., and, Dudley ME. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat Rev Cancer. 2008;8:299–308. doi: 10.1038/nrc2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bollard CM, Gottschalk S, Leen AM, Weiss H, Straathof KC, Carrum G, et al. Complete responses of relapsed lymphoma following genetic modification of tumor-antigen presenting cells and T-lymphocyte transfer. Blood. 2007;110:2838–2845. doi: 10.1182/blood-2007-05-091280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudley ME., and, Rosenberg SA. Adoptive-cell-transfer therapy for the treatment of patients with cancer. Nat Rev Cancer. 2003;3:666–675. doi: 10.1038/nrc1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunder NN, Wallen H, Cao J, Hendricks DW, Reilly JZ, Rodmyre R, et al. Treatment of metastatic melanoma with autologous CD4+ T cells against NY-ESO-1. N Engl J Med. 2008;358:2698–2703. doi: 10.1056/NEJMoa0800251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pule MA, Savoldo B, Myers GD, Rossig C, Russell HV, Dotti G, et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat Med. 2008;14:1264–1270. doi: 10.1038/nm.1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudley ME, Yang JC, Sherry R, Hughes MS, Royal R, Kammula U, et al. Adoptive cell therapy for patients with metastatic melanoma: evaluation of intensive myeloablative chemoradiation preparative regimens. J Clin Oncol. 2008;26:5233–5239. doi: 10.1200/JCO.2008.16.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridgway D. The first 1000 dendritic cell vaccinees. Cancer Invest. 2003;21:873–886. doi: 10.1081/cnv-120025091. [DOI] [PubMed] [Google Scholar]
- Nencioni A, Grünebach F, Schmidt SM, Müller MR, Boy D, Patrone F, et al. The use of dendritic cells in cancer immunotherapy. Crit Rev Oncol Hematol. 2008;65:191–199. doi: 10.1016/j.critrevonc.2007.10.002. [DOI] [PubMed] [Google Scholar]
- Sekaly RP. The failed HIV Merck vaccine study: a step back or a launching point for future vaccine development. J Exp Med. 2008;205:7–12. doi: 10.1084/jem.20072681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee EG, Boone DL, Chai S, Libby SL, Chien M, Lodolce JP, et al. Failure to regulate TNF-induced NF-κB and cell death responses in A20-deficient mice. Science. 2000;289:2350–2354. doi: 10.1126/science.289.5488.2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boone DL, Turer EE, Lee EG, Ahmad RC, Wheeler MT, Tsui C, et al. The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat Immunol. 2004;5:1052–1060. doi: 10.1038/ni1110. [DOI] [PubMed] [Google Scholar]
- Hayashi F, Smith KD, Ozinsky A, Hawn TR, Yi EC, Goodlett DR, et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature. 2001;410:1099–1103. doi: 10.1038/35074106. [DOI] [PubMed] [Google Scholar]
- Honko AN., and, Mizel SB. Effects of flagellin on innate and adaptive immunity. Immunol Res. 2005;33:83–101. doi: 10.1385/IR:33:1:083. [DOI] [PubMed] [Google Scholar]
- Gaucher D, Therrien R, Kettaf N, Angermann BR, Boucher G, Filali-Mouhim A, et al. Yellow fever vaccine induces integrated multilineage and polyfunctional immune responses. J Exp Med. 2008;205:3119–3131. doi: 10.1084/jem.20082292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lahiri A, Das P., and, Chakravortty D. Engagement of TLR signaling as adjuvant: towards smarter vaccine and beyond. Vaccine. 2008;26:6777–6783. doi: 10.1016/j.vaccine.2008.09.045. [DOI] [PubMed] [Google Scholar]
- van Duin D, Medzhitov R., and, Shaw AC. Triggering TLR signaling in vaccination. Trends Immunol. 2006;27:49–55. doi: 10.1016/j.it.2005.11.005. [DOI] [PubMed] [Google Scholar]
- Tosch C, Geist M, Ledoux C, Ziller-Remi C, Paul S, Erbs P, et al. Adenovirus-mediated gene transfer of pathogen-associated molecular patterns for cancer immunotherapy. Cancer Gene Ther. 2009;16:310–319. doi: 10.1038/cgt.2008.85. [DOI] [PubMed] [Google Scholar]
- Song XT, Evel-Kabler K, Shen L, Rollins L, Huang XF., and, Chen SY. A20 is an antigen presentation attenuator, and its inhibition overcomes regulatory T cell-mediated suppression. Nat Med. 2008;14:258–265. doi: 10.1038/nm1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park MY, Kim CH, Sohn HJ, Oh ST, Kim SG., and, Kim TG. The optimal interval for dendritic cell vaccination following adoptive T cell transfer is important for boosting potent anti-tumor immunity. Vaccine. 2007;25:7322–7330. doi: 10.1016/j.vaccine.2007.08.037. [DOI] [PubMed] [Google Scholar]
- Darrah PA, Patel DT, De Luca PM, Lindsay RW, Davey DF, Flynn BJ, et al. Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major. Nat Med. 2007;13:843–850. doi: 10.1038/nm1592. [DOI] [PubMed] [Google Scholar]
- Ren W, Strube R, Zhang X, Chen SY., and, Huang XF. Potent tumor-specific immunity induced by an in vivo heat shock protein-suicide gene-based tumor vaccine. Cancer Res. 2004;64:6645–6651. doi: 10.1158/0008-5472.CAN-04-1084. [DOI] [PubMed] [Google Scholar]
- Rabinovich BA, Ye Y, Etto T, Chen JQ, Levitsky HI, Overwijk WW, et al. Visualizing fewer than 10 mouse T cells with an enhanced firefly luciferase in immunocompetent mouse models of cancer. Proc Natl Acad Sci USA. 2008;105:14342–14346. doi: 10.1073/pnas.0804105105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed N, Salsman VS, Yvon E, Louis CU, Perlaky L, Wels WS, et al. Immunotherapy for osteosarcoma: genetic modification of T cells overcomes low levels of tumor antigen expression. Mol Ther. 2009;17:1779–1787. doi: 10.1038/mt.2009.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song XT, Evel-Kabler K, Rollins L, Aldrich M, Gao F, Huang XF, et al. An alternative and effective HIV vaccination approach based on inhibition of antigen presentation attenuators in dendritic cells. PLoS Med. 2006;3:e11. doi: 10.1371/journal.pmed.0030011. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Scheme of adenoviral constructs.
TLR5 expression is upregulated in vivo after Ad-shAF vaccination.
Ad-shAF induces DC maturation and activation in vivo.
Systemic side effects of Ad-shAF and Cytoxan.