Abstract
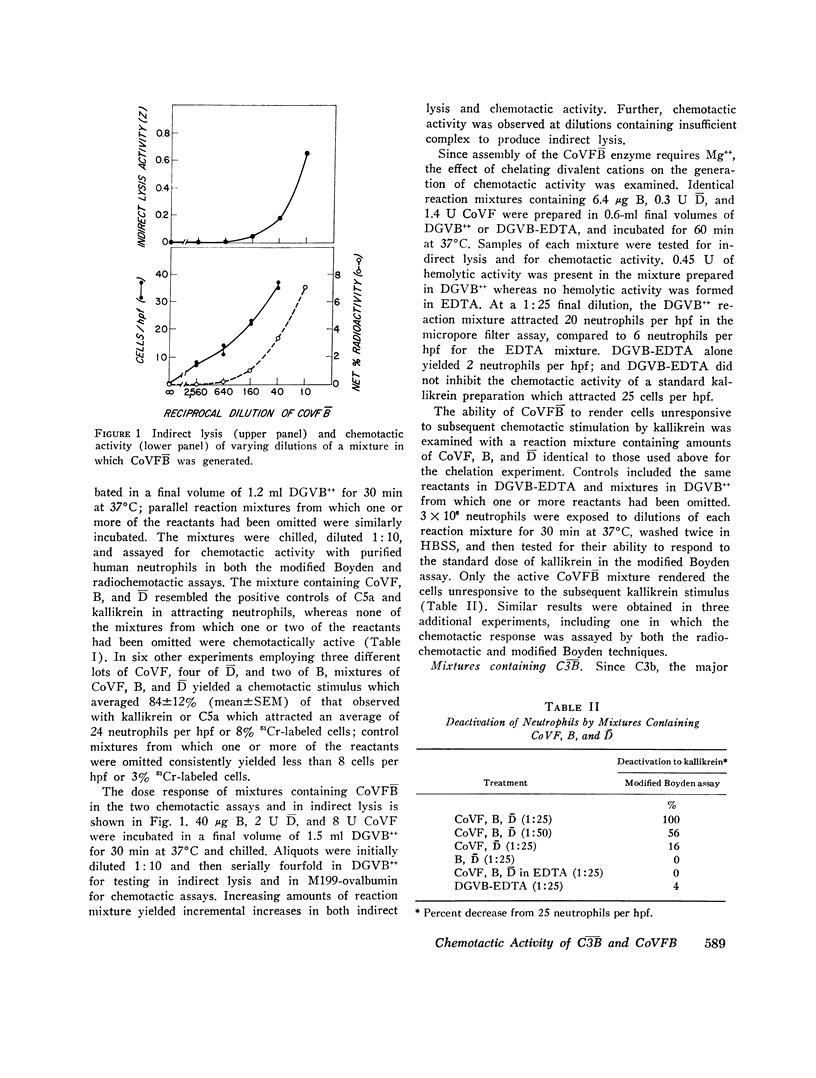
Interaction of D (the activated form of D) and B, factors of the properdin pathway, with C3b (the major cleavage fragment of C3) generates a convertase, C3B, which cleaves C3 and initiates the terminal complement sequence C5-C9. A functionally analogous more stable C3 convertase, CoVFB, ir formed by substituting cobra venom factor (CoVF) for C3b. Mixtures of highly purified CoVF, B, and D were chemotactic for human neutrophil polymorphonuclear leukocytes as assessed in Boyden chambers either by microscopic enumeration of migrating cells or by counting of 51Cr-labeled cells. Control mixtures containing CoVF, B, and D, reacted in the absence of Mg++, were hemolytically inactive and devoid of chemotactic activity. Over a range of doses, the chemotactic activity of mixtures yielding CoVFB correlated with their hemolytic activity. Pretreatment of neutrophils with mixtures containing CoVFB rendered them unresponsive to subsequent chemotactic stimulation by kallikrein of C5a, indicating cross-deactivation to other chemotactic factors. Similar neutrophil deactivation occurred after exposure to a mixture of C3b, B, and D in which C3B was formed; with short incubation times and high cell concentration C3B also exhibited some chemotactic activity. The chemotactic activity of C3B and CoVFB is an example of a biologic function arising from interactions among factors of the properdin pathway per se, as distinguished from the capacity of this pathway to activate C3 and the terminal complement sequence.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ballow M., Cochrane C. G. Two anticomplementary factors in cobra venom: hemolysis of guinea pig erythrocytes by one of them. J Immunol. 1969 Nov;103(5):944–952. [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Formation of a hemolytically active cellular intermediate by the interaction between properdin factors B and D and the activated third component of complement. J Exp Med. 1973 Dec 1;138(6):1305–1313. doi: 10.1084/jem.138.6.1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Properdin factor D: characterization of its active site and isolation of the precursor form. J Exp Med. 1974 Feb 1;139(2):355–366. doi: 10.1084/jem.139.2.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Serum proteins involved in decay and regeneration of cobra venom factor-dependent complement activation. J Immunol. 1973 Dec;111(6):1730–1736. [PubMed] [Google Scholar]
- Fearon D. T., Ruddy S., Knostman J. D., Carpenter C. B., Austen K. F. The functional significance of complement. Adv Nephrol Necker Hosp. 1974;4:15–35. [PubMed] [Google Scholar]
- Goetzl E. J., Austen K. F. A method for assessing the in vitro chemotactic response of neutrophils utilizing 51cr-labeled human leukocytes. Immunol Commun. 1972;1(5):421–430. doi: 10.3109/08820137209022954. [DOI] [PubMed] [Google Scholar]
- Goetzl E. J., Austen K. F. A neutrophil-immobilizing factor derived from human leukocytes. I. Generation and partial characterization. J Exp Med. 1972 Dec 1;136(6):1564–1580. doi: 10.1084/jem.136.6.1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goetzl E. J., Austen K. F. A neutrophil-immobilizing factor derived from human leukocytes. I. Generation and partial characterization. J Exp Med. 1972 Dec 1;136(6):1564–1580. doi: 10.1084/jem.136.6.1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Götze O., Müller-Eberhard H. J. The C3-activator system: an alternate pathway of complement activation. J Exp Med. 1971 Sep 1;134(3 Pt 2):90s–108s. [PubMed] [Google Scholar]
- Hunsicker L. G., Ruddy S., Austen K. F. Alternate complement pathway: factors involved in cobra venom factor (CoVF) activation of the third component of complement (C3). J Immunol. 1973 Jan;110(1):128–138. [PubMed] [Google Scholar]
- Hunsicker L. G., Ruddy S., Carpenter C. B., Schur P. H., Merrill J. P., Müller-Eberhard H. J., Austen K. F. Metabolism of third complement component (C3) in nephritis. Involvement of the classic and alternate (properdin) pathways for complement activation. N Engl J Med. 1972 Oct 26;287(17):835–840. doi: 10.1056/NEJM197210262871701. [DOI] [PubMed] [Google Scholar]
- Kaplan A. P., Kay A. B., Austen K. F. A prealbumin activator of prekallikrein. 3. Appearance of chemotactic activity for human neutrophils by the conversion of human prekallikrein to kallikrein. J Exp Med. 1972 Jan;135(1):81–97. doi: 10.1084/jem.135.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Götze O. C3 proactivator convertase and its mode of action. J Exp Med. 1972 Apr 1;135(4):1003–1008. doi: 10.1084/jem.135.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Polley M. J., Calcott M. A. Formation and functional significance of a molecular complex derived from the second and the fourth component of human complement. J Exp Med. 1967 Feb 1;125(2):359–380. doi: 10.1084/jem.125.2.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NILSSON U. R., MUELLER-EBERHARD H. J. ISOLATION OF BETA IF-GLOBULIN FROM HUMAN SERUM AND ITS CHARACTERIZATION AS THE FIFTH COMPONENT OF COMPLEMENT. J Exp Med. 1965 Aug 1;122:277–298. doi: 10.1084/jem.122.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson R. A., Jr, Jensen J., Gigli I., Tamura N. Methods for the separation, purification and measurement of nine components of hemolytic complement in guinea-pig serum. Immunochemistry. 1966 Mar;3(2):111–135. doi: 10.1016/0019-2791(66)90292-8. [DOI] [PubMed] [Google Scholar]
- Polley M. J., Müller-Eberhard H. J. Enharncement of the hemolytic activity of the second component of human complement by oxidation. J Exp Med. 1967 Dec 1;126(6):1013–1025. doi: 10.1084/jem.126.6.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruddy S., Austen K. F. A stoichiometric assay for the fourth component of complement in whole human serum using EAC'la-gp and functionally pure human second component. J Immunol. 1967 Dec;99(6):1162–1172. [PubMed] [Google Scholar]
- Ruddy S., Austen K. F. C3 inactivator of man. I. Hemolytic measurement by the inactivation of cell-bound C3. J Immunol. 1969 Mar;102(3):533–543. [PubMed] [Google Scholar]
- Ruddy S., Klemperer M. R., Rosen F. S., Austen K. F., Kumate J. Hereditary deficiency of the second component of complement (C2) in man: correlation of C2 haemolytic activity with immunochemical measurements of C2 protein. Immunology. 1970 Jun;18(6):943–954. [PMC free article] [PubMed] [Google Scholar]
- Theofilopoulos A. N., Bokisch V. A., Dixon F. J. Receptor for soluble C3 and C3b on human lymphoblastoid (RAJI) cells. Properties and biologocal significance. J Exp Med. 1974 Mar 1;139(3):696–711. doi: 10.1084/jem.139.3.696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WARD P. A., COCHRANE C. G., MUELLER-EBERHARD H. J. THE ROLE OF SERUM COMPLEMENT IN CHEMOTAXIS OF LEUKOCYTES IN VITRO. J Exp Med. 1965 Aug 1;122:327–346. doi: 10.1084/jem.122.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward P. A. A plasmin-split fragment of C'3 as a new chemotactic factor. J Exp Med. 1967 Aug 1;126(2):189–206. doi: 10.1084/jem.126.2.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward P. A., Becker E. L. The deactivation of rabbit neutrophils by chemotactic factor and the nature of the activatable esterase. J Exp Med. 1968 Apr 1;127(4):693–709. doi: 10.1084/jem.127.4.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward P. A., Newman L. J. A neutrophil chemotactic factor from human C'5. J Immunol. 1969 Jan;102(1):93–99. [PubMed] [Google Scholar]



