Abstract
In this study, we attempted to assess the interactions of resveratrol, a natural compound present in various plant species, with the purine analogues fludarabine and cladribine in terms of their effects on DNA damage and apoptosis in chronic lymphocytic leukemia (CLL) cells. The experiments were performed ex vivo using short-term cell cultures of blood and bone marrow cells from newly diagnosed untreated patients. We analyzed the expression of active caspase-3 and the BCL-2/BAX ratio as markers of apoptosis and the expression of phosphorylated histone H2AX (γH2AX) and activated ATM kinase, which are reporters of DNA damage. The results of our study revealed that resveratrol induced apoptosis in CLL cells in a tumor-specific manner but did not affect non-leukemic cells, and apoptosis was associated with a decreased BCL2/BAX ratio. Here, we report for the first time that both resveratrol + fludarabine and resveratrol + cladribine caused a higher rate of apoptosis in comparison to the rate caused by a single drug. The percentage of apoptotic cells induced by resveratrol alone was higher in the group of patients with better prognostic markers than in those with worse prognostic markers. However, the rates of apoptosis caused by resveratrol combined with purine analogues were independent of ZAP-70 and CD38 expression and the clinical state of the disease; they were only dependent on the presence of high-risk cytogenetic abnormalities. We also observed an increase in γH2AX expression together with a rise in activated ATM in most of the analyzed samples. The obtained results indicate that resveratrol might warrant further study as a new therapeutic option for CLL patients. This naturally occurring substance may be used as a single agent, especially in older persons for whom there are some limitations for the use of aggressive treatment. On the other hand, a lower purine analogue dose could potentially be used in combination with resveratrol because of their combined effect. One of the mechanisms of action of resveratrol is the induction of DNA damage, which ultimately leads to apoptosis.
Keywords: Apoptosis, CLL, DNA damage, Purine analogues, Resveratrol
Introduction
Chronic lymphocytic leukemia (CLL) is the most frequent type of adult leukemia in western countries and is characterized by the accumulation of monoclonal, mainly B, lymphocytes in blood, bone marrow, and lymphoid organs [1, 2]. Despite the recent introduction of many therapeutic regimens, CLL is still an incurable disorder. It can be effectively treated with purine analogues, glucocorticoids, alkylating agents, or monoclonal antibodies; however, some patients with relapsed or refractory disease have limited therapeutic options. Thus, there is an urgent need to discover novel, less toxic, and more effective drugs for CLL patients. Among other options, the use of immunomodulatory agents or plant-derived substances has been reported to improve the results of CLL treatment.
Resveratrol (trans-3,4′,5-trihyrdoxystilbene) is a natural compound that is present in various plant species and is relatively abundant in vines. It has many biological and pharmaceutical properties [3–5], and the most biologically active isomer is trans-resveratrol. Primary research on this substance began with the observation that a low incidence of cardiovascular diseases may co-exist with a high-fat diet and moderate consumption of red wine (it is known as the French paradox) [6]. The mechanisms of cardiovascular protection caused by resveratrol involve inhibition of platelet aggregation, arterial vasodilatation mediated by nitric oxide release, favorable changes in lipid metabolism, antioxidant effects, stimulation of angiogenesis, and induction of cardioprotective protein expression [6–8]. It has also been suggested that resveratrol has therapeutic potential for allergy and neurological disorders [9, 10]. The anticancer properties of this naturally occurring substance were first noticed when Jang et al. [11] demonstrated its cancer-chemopreventive and cytostatic properties via the initiation, promotion, and progression of carcinogenesis. Resveratrol was found to inhibit proliferation and induce apoptosis in several human cancer cells, including B-cell malignancies [12].
In the present study, we attempted to explore whether resveratrol could induce apoptosis in CLL leukemic cells and non-leukemic lymphocytes derived from peripheral blood and bone marrow and to assess the interactions of resveratrol with two purine analogues, fludarabine and cladribine, with respect to their effect on apoptosis in CLL cells. We also investigated whether resveratrol had an effect on DNA damage. Subsequently, apoptosis and DNA damage markers were correlated with CLL prognostic factors, such as ZAP-70 and CD38 expression, clinical states according to the Rai classification, and cytogenetic abnormalities.
The experiments were performed using short-term cell cultures of peripheral blood and bone marrow supplemented with resveratrol and peripheral blood supplemented with resveratrol in combination with purine analogues. We analyzed the active caspase-3 expression as a marker of apoptosis and the intracellular expression levels of BCL-2 and BAX as regulators of apoptosis. DNA damage was assessed by measuring the expression levels of phosphorylated histone H2AX (named γH2AX) and activated ATM kinase (ataxia telangiectasia mutated kinase).
Material and methods
Patients
Eighteen newly diagnosed, untreated CLL patients were enrolled in the study. The diagnosis of CLL was based on clinical examination and morphological and immunological criteria. Peripheral blood and bone marrow samples were obtained from the patients following the provision of informed consent. Peripheral blood samples from healthy donors were used as controls.
Cell isolation and culture
Peripheral blood and bone marrow mononuclear cells were isolated by density gradient centrifugation using Biocoll Separating Solution (Biochrom AG, Berlin, Germany). After two washes with phosphate-buffered saline (PBS), the cells were resuspended in culture medium consisting of RPMI 1640 with 2 mM l-glutamine, 100 units/ml penicillin, 100 μg/ml streptomycin, and 10% fetal calf serum (FCS) at a final density of 2 × 106 cells/ml. This culture medium was supplemented with 40 μM resveratrol, 1 μg/ml fludarabine, 1.4 μg/ml cladribine, or a mixture of resveratrol and fludarabine or resveratrol and cladribine. We selected a concentration of 40 μM resveratrol based on preliminary experiments in CLL cultures in which this concentration of resveratrol revealed a significant induction of apoptosis with spontaneous apoptosis that did not exceed 50% of the total cell culture. To obtain stock solutions, resveratrol was dissolved in 0.1% DMSO. All reagents were obtained from Sigma-Aldrich Chemie GmbH, Steinheim, Germany. The cells were cultured at 37°C in a 5% CO2 atmosphere. Cells were exposed to the drugs for 0, 2, and 24 h in culture, and cells treated for 2 h were analyzed for H2AX phosphorylation and ATM activation, whereas those treated for 24 h were assessed for the frequency of apoptosis. H2AX phosphorylation induced by external DNA damaging agents was observed early during the treatment (up to 2 h), whereas, at later time points (>3 h), H2AX phosphorylation was associated with apoptosis [13]. The respective cell samples incubated in absence of any drug or in the presence of the resveratrol solvent DMSO for periods of time equivalent to the drug-treated cells were considered negative controls.
Immunocytochemical detection of active caspase-3 and BCL-2 and BAX proteins
The blood and bone marrow samples that had been treated in culture with resveratrol, fludarabine, cladribine, or a mixture of these drugs (∼105 cells) were initially incubated for 15 min with anti-CD19 PE-Cy5 and anti-CD5 APC-conjugated monoclonal antibodies (mAb) (DAKO, Glostrup, Denmark) at room temperature. Subsequently, the cells were subjected to fixation and permeabilization procedures using an IntraPrep kit (Immunotech, Marseilles, France) according to the manufacturer’s instructions. The cells were then incubated with PE-conjugated anti-active caspase-3 mAb (Pharmingen, Franklin Lakes, NJ, USA), FITC-conjugated anti-intracellular BAX mAb (Santa Cruz Biotechnology, Santa Cruz, CA, USA), PE-conjugated anti-intracellular BCL-2 mAb (Pharmingen) or an isotype-matched negative control (DAKO) in the dark at room temperature for 15 min. Blood samples obtained from healthy donors that had been treated in culture with resveratrol were initially incubated with anti-CD19 and anti-CD3 mAb, followed by fixation, permeabilization, and staining with anti-active caspase-3 mAb.
Immunocytochemical detection of γH2AX and ATM activation
The samples of blood were initially incubated for 15 min with anti-CD19 PE-Cy5 and anti-CD5 APC-conjugated mAb (DAKO) at room temperature. Subsequently, the cells were subjected to fixation and permeabilization using the IntraPrep kit (Immunotech) following the manufacturer’s instructions. The cells were then incubated with Ser139-phosphorylated H2AX mAb conjugated to FITC (BioLegend, San Diego, CA, USA) or anti-phospho ATM (Ser-1981) Ab (Millipore, Billerica, MA, USA) labeled with the ZenonTM Alexa Fluor® 488 Mouse IgG1 Labeling Kit (Molecular Probes, Eugene, OR, USA).
Measurement of fluorescence
The fluorescence of the samples was measured using a FACSCalibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA). The CD19+/CD5+, CD19+/CD5−, or CD19−/CD5+ populations in the CLL samples or the CD19+ and CD3+ populations in healthy controls were gated and further analyzed. Ten thousand cells were measured per sample. To determine the frequency of apoptosis, the percentage of cells expressing active caspase-3 relative to the control cells was calculated. The mean BCL-2 and BAX immunofluorescence (IF) intensity was estimated for each sample. The BCL2/BAX IF ratio for CD19+/CD5+ cells was then calculated.
The mean γH2AX and activated ATM IF intensity was estimated for each sample. The data obtained for drug effects in CLL cells are presented as the drug-induced increase in the mean γH2AX IF or ATMP1981 IF over the respective values determined for the control cells, which were assessed in parallel.
Flow cytometric analysis of ZAP-70 and CD38 expression
A total of 1 × 106 peripheral blood cells were stained with the anti-CD19 PE-conjugated MoAb (BD Pharmingen, Franklin Lakes, NJ, USA), anti-CD5 CyChrome-conjugated MoAb (Caltag, Burlingame, CA, USA), or anti-CD3 PE-conjugated MoAb (BD Pharmingen). Following membrane staining, the cells were fixed with 1% paraformaldehyde solution in PBS for 15 min at room temperature and permeabilized with 70% ethanol for 1 h at −20°C. After washing with PBS, anti-ZAP-70 antibody (Biomol Research, Plymouth Meeting, PA, USA) labeled with the ZenonTM Alexa Fluor® 488 Mouse IgG2a Labeling Kit (Molecular Probes) was added to the sample tubes. The samples were incubated for 30 min, washed, and analyzed by flow cytometry Patients were considered positive for ZAP-70 when its expression was detected in 20% or more of the leukemic cells.
To assess CD38 expression, peripheral blood mononuclear cells were stained with anti-CD38 FITC, anti-CD19 PE, anti-CD5 CyChrome or IgG1 isotypic control mAbs for 20 min and analyzed by flow cytometry. Patients were considered positive for CD38 when its expression was detected in at least 20% of the leukemic cells.
Evaluation of the cooperative index
To evaluate the synergism between resveratrol and fludarabine or cladribine, a cooperative index (CI) based on the Chou-Talalay method was calculated [14–16]. The following formula was used: CI = sum of specific apoptosis caused by treatment with a single agent/specific apoptosis determined with combined treatment. The percentage of specific apoptosis was determined using the following formula: specific apoptosis = (drug-induced apoptosis − spontaneous apoptosis)/(100 − spontaneous apoptosis)100%. When CI < 1, CI = 1, and CI > 1, the effects were defined as synergistic, additive, and infra-additive, respectively [15, 16].
Fluorescence in situ hybridization
To analyze the chromosome alterations characteristic of CLL, locus-specific probes for 11q22.3 (LSI ATM), 17p13.1 (LSI TP53), 13q.14.3 (D13S319), and the chromosome 12 centromere (CEP12) (Abbott Diagnostics, Abbott Park, IL, USA) were used. Fluorescence in situ hybridization (FISH) was performed according to the manufacturer’s instructions. Probes were denatured at 73°C for 5 min and then applied to the designated areas of the slides. Following an overnight hybridization at 37°C, the slides were washed and air-dried in the dark. The slides were then stained with DAPI and stored at −20°C in the dark. The samples were analyzed using a BX51 fluorescence microscope (OLYMPUS), and images were captured with a CCD camera using the CytoVision image analysis system. At least 200 nuclei were analyzed for each probe, and the cutoff value for each probe was 20%.
Statistical analysis
Statistical analyses were performed using STATISTICA 8.0 software for Windows. The Mann–Whitney U test was employed for analyses of two independent groups. The Wilcoxon test was used for analyses of two dependent variables. p < 0.05 was considered to indicate statistical significance.
Results
Resveratrol induces apoptosis in CLL leukemic cells derived from peripheral blood and bone marrow
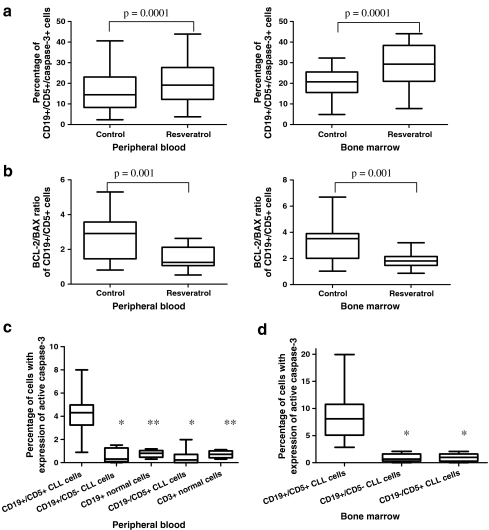
Data showing the induction of apoptosis in CLL cells treated with resveratrol ex vivo are presented in Fig. 1a, b. We measured the expression of active caspase-3 in CD19+/CD5+ cells and the changes in BCL-2/BAX ratio in leukemic cells. The percentage of cells demonstrating caspase-3 expression in cultures treated with resveratrol for 24 h was significantly higher than that detected in 24-h parallel control cultures. Such an increased frequency of resveratrol-induced apoptosis was observed in blood and bone marrow cultures, and significantly higher levels of apoptosis were detected in bone marrow compared to peripheral blood (Fig. 1a). The BCL-2/BAX ratio in CD19+/CD5+ cells was significantly lower in resveratrol-induced cultures after 24 h of treatment in comparison to 0-h cultures (Fig. 1b).
Fig. 1.
a Percentage of apoptotic leukemic cells in 24-h cultures with resveratrol in samples of peripheral blood and bone marrow. Statistically significant differences are indicated. b BCL-2/BAX ratio of CD19+/CD5+ cells in cultures treated with resveratrol for 24 h compared to the control 0-h culture. A decreased ratio was detected in resveratrol-induced cultures after 24 h. Statistically significant differences are indicated. c Percentage of apoptotic cells in populations of non-leukemic B (CD19+/CD5−) and T (CD19−/CD5+) cells from the chosen CLL patients (n = 9) and healthy donors (n = 5) in comparison to a population of leukemic CD19+/CD5+ cells in ex vivo peripheral blood cultures treated with resveratrol. Data represent the drug-induced increase in the percentage of apoptotic cells above the respective values observed in control cultures of the same cell population. Single and double asterisks indicate statistically significant differences of p = 0.0001 and p = 0.002, respectively, in comparison to the CD19+/CD5+ population. d Percentage of apoptotic cells in populations of non-leukemic B and T cells from the chosen CLL patients compared to the leukemic CD19+/CD5+ cell population in ex vivo bone marrow cultures with resveratrol. Data represent the drug-induced increase in the percentage of apoptotic cells above the respective values observed in control cultures of the same cell population. Asterisk indicates statistically significant differences of p = 0.0001 in comparison to the CD19+/CD5+ population. Graphs represent the median ± minimum/maximum
Resveratrol induces apoptosis in leukemic cells in a tumor-specific manner
To assess the effects of resveratrol on non-leukemic cells, we measured the percentage of apoptotic cells that expressed active caspase-3 within a population of CD19+/CD5− and CD19−/CD5+ cells of CLL patients, which represented non-leukemic B and T cells, respectively, and within populations of CD3+ and CD19+ cells of healthy blood donors cultured with resveratrol. The results are shown in Fig. 1c, d. In the group of CLL patients, this analysis was performed in nine out of 15 patients for whom the percentage of non-leukemic cells in both peripheral blood and bone marrow was greater than 10%. As shown in Fig. 1c, d, no significant apoptotic effect was detected in the population of non-leukemic lymphocytes cultured with resveratrol, or in samples of peripheral blood or bone marrow. Statistical analyses of the data revealed a significantly lower percentage of apoptotic cells in the group of non-leukemic cells compared to the group of leukemic cells, as well as in the healthy donor group compared to the leukemic cell group.
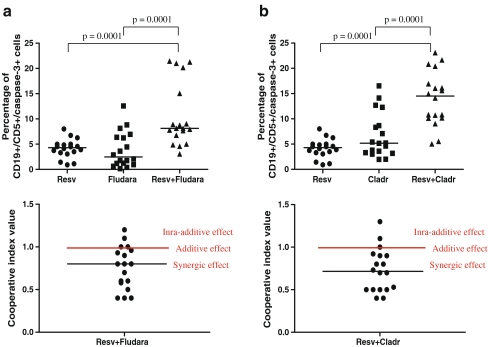
Rates of apoptosis elicited by resveratrol in combination with fludarabine and cladribine are higher than those caused by a single agent
To assess the rate of apoptosis induced by resveratrol in combination with purine analogues, the expression of active caspase-3 in blood cells cultured with resveratrol + fludarabine or resveratrol + cladribine was measured. As shown in Fig. 2a, the frequency of apoptosis in resveratrol + fludarabine-treated cultures was significantly greater than those in resveratrol-treated and fludarabine-treated cultures. Similarly, a significant increase in apoptosis was observed in resveratrol + cladribine-treated cultures compared to resveratrol cultures and to cladribine cultures (Fig. 2a). To evaluate the effect of the combined action of resveratrol and purine analogues, the CI for each sample was evaluated and is presented in Fig. 2b. In 14 out of 18 resveratrol + fludarabine combinations and in 15 out of 18 resveratrol + cladribine combinations, a synergic effect was observed.
Fig. 2.
a Percentage of apoptotic leukemic cells in cultures treated with resveratrol, fludarabine, and resveratrol combined with fludarabine for 24 h and the CI calculated for each sample. b Percentage of apoptotic leukemic cells in cultures treated with resveratrol, cladribine, and resveratrol combined with cladribine for 24 h and the CI calculated for each sample. Data represent the drug-induced increase in the percentage of apoptotic cells compared to the respective values observed in parallel control cultures. CI = (sum of specific apoptosis of single agent treatment/specific apoptosis of combined treatment). The percentage of specific apoptosis was determined using the following formula: specific apoptosis = (drug induced apoptosis − spontaneous apoptosis)/(100 − spontaneous apoptosis) × 100%. When CI < 1, CI = 1, and CI > 1, the effects were defined as synergistic, additive, and infra-additive, respectively
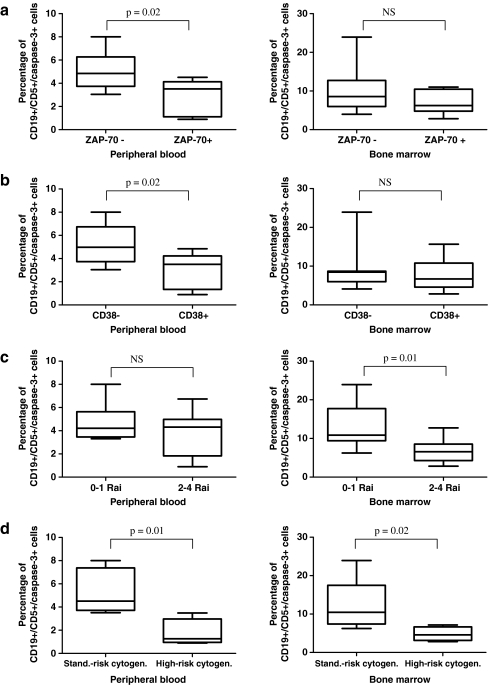
Resveratrol-induced apoptosis in CLL cells differs in patients with better prognostic markers vs those with worse prognostic markers
To assess whether the rate of apoptosis caused by resveratrol is associated with the prognosis of CLL, the percentages of apoptotic cells were compared between patients with worse prognostic markers (ZAP-70+, CD38+, advanced 2–4 stages according to the Rai classification, 17p13.1 or 11q22.3 genetic abnormalities) and those with better prognostic markers (ZAP-70−, CD38−, early 0–1 stages according to the Rai classification, 13q14.3 or trisomy 12 genetic abnormalities, or an absence of genetic changes). We detected a higher percentage of apoptotic caspase-3-positive cells in resveratrol-induced cultures from patients with low-risk markers in comparison to those in the higher-risk group, and the results are shown in Fig. 3. The rates of apoptosis in cultures treated with resveratrol together with purine analogues differed only between the group of patient with low-risk cytogenetic changes and that with high-risk abnormalities, with significantly higher values detected in the former group. With respect to cultures treated with resveratrol together with purine analogues, we did not detect significant differences in the rate of apoptosis between the ZAP-70− and the ZAP-70+ group, the CD38− and the CD38+ group or the 0–1 Rai and the 2–4 Rai group.
Fig. 3.
a Percentage of apoptotic leukemic cells in cultures treated with resveratrol for 24 h in the group of ZAP-70− patients (n = 11) and in the group of ZAP-70+ patients (n = 7). b Percentage of apoptotic leukemic cells in cultures treated with resveratrol in the group of CD38− patients (n = 7) and in the group of CD38+ patients (n = 10). c Percentage of apoptotic leukemic cells in cultures treated with resveratrol in the group of patients with 0–1 stadium (n = 6) and in the group patients with 2–4 stadium (n = 12) according to the Rai classification. d Percentage of apoptotic leukemic cells in cultures treated with resveratrol in the group of patients with standard-risk cytogenetic changes (13q14.3, trisomy 12 or no changes detected, n = 5) and in the group of patients with high-risk cytogenetic changes (17p13.1 or 11q22.3, n = 4). The percentage of apoptotic leukemic cells was calculated above the level in control cultures. Statistically significant differences are indicated in the figure. Graphs represent means ± standard deviations. NS, not statistically significant
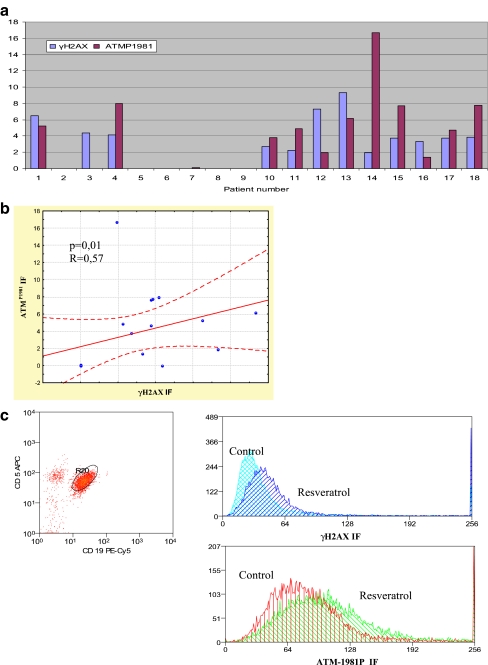
Resveratrol induces phosphorylation of H2AX and activation of ATM in CLL malignant cells
To assess whether resveratrol causes DNA damage, the expression of phosphorylated H2AX and activated ATM in CD19+/CD5+ cells was measured. In most cases, an increase in γH2AX expression concurrent with increased expression of activated ATM was detected after a 2-h incubation with resveratrol, as compared to the expression levels of these phosphoproteins in control cultures. We detected a statistically significant correlation between γH2AX expression and activated ATM expression (R = 0.57, p = 0.01) (Fig. 4b). Data showing the increase in γH2AX and activated ATM expression compared to their expression levels in control cultures of cells from individual patients, as well as representative histograms showing the expressions, are presented in Fig. 4. As presented in Fig. 4a, however, the cells from a few patients remained unaffected. The expression of γH2AX was also significantly higher in the ZAP-70−, 0–1 Rai stadium and better cytogenetic abnormalities groups in comparison to the ZAP-70+, 2–4 Rai stadium and worse cytogenetic changes groups, respectively. We also compared the rate of apoptosis in response to resveratrol in the group of patients in which an increase in γH2AX expression above its “constitutive” level was observed (“γH2AX positive”, number of patients = 12) and in the group that demonstrated no increase in γH2AX expression (“γH2AX negative”, number of patients = 6). The rate of apoptosis was higher in the “γH2AX positive” group than in the “γH2AX negative” group (4.7 ± 1.7 vs 2.9 ± 1.4, p = 0.003, and 10.5 ± 5.1 vs 5.2 ± 2.1, p = 0.004, for peripheral blood and bone marrow, respectively).
Fig. 4.
a Ex vivo induction of γH2AX and phosphorylated ATM (ATMP1981) in CD5+/CD19+ cells treated with resveratrol for 2 h. Data represent the drug-induced increase in expression of γH2AX (γH2AX IF) and activated ATM (ATMP1981 IF) compared to the respective values observed in parallel control cultures. No increase in γH2AX expression after resveratrol treatment was detected in leukemic cells of patient nos. 2, 5, 6, 7, 8, and 9. No increase in expression of ATMP1981 after resveratrol treatment was detected in leukemic cells of patient nos. 2, 3, 5, 6, 8, and 9. In patient no. 7, a slight increase in ATMP1981 expression was observed, but no increase in ATMP1981 expression was detected in patient no. 3. b Diagram showing the correlation between γH2AX and ATMP1981 expression. c Representative flow cytometric histograms showing the expression of γH2AX and ATMP1981 in CD5+/CD19+ cells treated with resveratrol for 2 h
Discussion
One of the most important biological activities of resveratrol is its cancer-chemopreventive and anti-cancer properties [17]. Resveratrol has been shown to suppress the in vitro proliferation of a variety of tumor cells [5, 18]. Its anti-proliferative activity is associated with the perturbed cell transit through G0-1 phase of the cell cycle and with the induction of apoptosis. It has also been reported that resveratrol inhibits the activity of ribonucleotide reductase, the enzyme that provides proliferating cells with the deoxyribonucleotides required for DNA synthesis during early S phase of the cell cycle, which leads to an inhibition of DNA synthesis [19, 20]. Resveratrol affects other critical events associated with tumor development, such as the up-regulation of p53 and p21 levels, induction of nitric oxide, inhibition of cyclooxygenase, protection against reactive oxygen intermediates, down-regulation of survival factors, and down-regulation of proteinases [21, 22].
These properties of resveratrol led us to assess its effects on leukemic cells of CLL patients. Our results indicated that resveratrol caused apoptosis in leukemic CD19+/CD5+ cells in both the peripheral blood and bone marrow in a tumor-specific manner. Non-leukemic cells were not significantly affected. We also showed that apoptosis caused by resveratrol was associated with the regulation of BCL-2 and BAX protein expression, resulting in a decreased BCL-2/BAX ratio. Resveratrol-induced apoptosis of CLL cells was first described by Roman et al. [23]. They showed that resveratrol caused apoptosis of leukemic CLL cells and that this effect could be mediated by the down-regulation of two anti-apoptotic proteins, inducible nitric oxide synthase (iNOS) and BCL-2, whereas apoptosis of normal lymphocytes was not observed [23]. The authors continued their research by assessing other polyphenols and flavones with respect to their influence on CLL and normal lymphocytes and confirmed the role of these substances as inducers of CLL tumor-specific apoptosis [24, 25]. In the present study, the anti-apoptotic effects of resveratrol were analyzed in suspension cultures, which have been commonly used by other researchers in in vitro experiments to assess the effects of drugs. However, it has been recently reported that CLL cells are protected against the activity of drugs in the tissue microenvironment, such as the bone marrow and secondary lymphoid organs. It could be associated with drug resistance or could pave the way to disease relapse [26, 27]. Thus, further studies in the presence of microenvironment cells, such as bone marrow stromal cells, are required to assess the precise mechanism of resveratrol action. The results of the present study, however, undoubtedly confirm the anti-apoptotic effects of resveratrol on leukemic cells in suspension and its potential to reduce the number of leukemic cells in peripheral blood in vivo. Taking these findings into account, resveratrol may be considered a good therapeutic option, especially in older CLL patients for whom there are some limitations to the use of aggressive treatment, and a reduction of lymphocytosis is often an acceptable effect of therapy.
In the next part of our study, we analyzed the effects of resveratrol in combination with the purine analogues fludarabine and cladribine on the induction of apoptosis. This is the first report to describe a rate of apoptosis caused by resveratrol in combination with purine analogues that is higher than that caused by a single drug. Based on the CI, we assessed the effect of the combined treatment, which was synergistic in most of the analyzed cases.
Purine analogues are widely used for the treatment of CLL, and it has been reported that higher remission rates can be obtained when they are employed as a first-line therapy [28, 29]. Two purine analogues—fludarabine and cladribine—have been proven to possess antitumor activity, as evidenced by a direct interference with DNA and RNA synthesis, interference with DNA repair mechanisms, induction of apoptosis, and regulation of the cell cycle and signal transduction pathways [30, 31]. However, some patients are refractory to these drugs. Other problems associated with the use of purine analogues are associated with their toxicities, for example, immunosuppression with a decrease in the CD4+/CD8+ ratio, which leads to the development of opportunistic infections, myelosuppression, and gastrointestinal toxicities, including nausea, vomiting and hepatic lesions [16, 32, 33]. Thus, the reduction of purine analogue-associated toxicity achieved with a lower dose may be of great importance. Because of the combined effects of resveratrol and both fludarabine and cladribine, lower purine analogue doses in combination with resveratrol are possible. Such a combination of purine analogues with other drugs like thalidomide or valproate has been reported in CLL patients based on in vitro experiments and clinical trials [16, 34–36].
CLL is a heterogeneous disease with a variable clinical course and prognosis. Thus, the roles of prognostic factors in CLL are very important, especially in distinguishing the group of patients that require intensive treatment from those who will benefit from milder forms of therapy. A number of prognostic markers have been identified in CLL patients, and among them, markers related to the biology of the disease, such as genomic aberrations, gene abnormalities, the mutation status of the variable segment of immunoglobulin heavy chain genes (IgVH), and surrogate markers like ZAP-70 and CD38 expression have been described as the most powerful ones [37–40].
In the present study, we tried to assess whether the rate of apoptosis caused by resveratrol differed between patients with better and worse prognostic markers. We detected differences in the percentage of resveratrol-induced apoptotic rates between the group of patients with ZAP-70−, CD38−, early clinical stages, and standard-risk cytogenetic changes in comparison to the group with ZAP-70+, CD38+, advanced clinical stages, and high-risk cytogenetic abnormalities, which indicated better pro-apoptotic effects in the former group. We can suppose that treatment with resveratrol may be most effective in patients with better prognostic factors and in early stages of the disease. Interestingly, there were no significant differences between both groups with respect to resveratrol in combination with purine analogues, excluding the group with standard-risk cytogenetic changes compared to that with high-risk cytogenetic changes. Thus, resveratrol combined with purine analogues can be equally effective in CLL patients, independently of ZAP-70 expression, CD38 expression, and clinical stadium according to the Rai classification.
Among the different activities of resveratrol, its antioxidant potential has generated some controversy. Resveratrol is indicated as an agent that possesses protective effects against oxidative stress. It is both a free radical scavenger and a potent antioxidant based on its ability to promote the activities of a variety of antioxidant enzymes [6, 41]. However, subsequent research has provided new, interesting insight into the effect of resveratrol on the intracellular redox state. These results seem to support both anti- and pro-oxidant activities of resveratrol, depending on the concentration of resveratrol and the cell type [6]. It has been proposed that such a pro-oxidant action of resveratrol could be an important mechanism of its anticancer and apoptotic-inducing effects [6]. Ahmad et al. [42] reported that resveratrol elicited pro-oxidant properties, as evidenced by an increase in the intracellular concentration of superoxide. DNA damage induced by resveratrol in the presence of copper ions has been reported by Zheng et al. [43]. Similarly, DNA degradation by resveratrol alone or in the presence of copper has been demonstrated in human peripheral blood lymphocytes by Azmi et al. [44, 45].
The phosphorylated histone H2AX (γH2AX) is a marker of double-stranded DNA breaks, whereas ATM kinase is considered a major physiological mediator of H2AX phosphorylation. ATM activation occurs via its autophosphorylation at Ser1981 [46, 47]. A few studies have examined the influence of resveratrol on H2AX phosphorylation. Tyagi et al. [48] reported that resveratrol treatment increased the level of phosphorylated γH2AX via ATM/ATR kinases. Rusin et al. [49] examined the influence of resveratrol on the growth of human cancer cell lines and its modulation of the expression of key proteins involved in DNA repair and cell cycle regulation. Their results showed that resveratrol induced DNA damage, as evidenced by the phosphorylation of histone H2AX and p53 protein.
In the present study, the expression of both phosphorylated H2AX and activated ATM in CD19+/CD5+ cells were examined using flow cytometry. We observed an increase in γH2AX expression together with an elevation of activated ATM in the same samples. Interestingly, this increase was observed in most but not all cases. The analysis of γH2AX expression in the group with worse vs better prognostic markers revealed an association with prognostic factors, such as ZAP-70 and CD38 expression, clinical stadium of disease, and cytogenetic abnormalities. A significantly higher expression level of γH2AX was detected in patients with better prognostic markers. Thus, it can be concluded that more aggressive cases of CLL associated with an unmutated IgVH region or cytogenetic abnormalities, which involve proteins that play a role in the DNA damage response, such as p53 or ATM kinase, are more resistant to DNA damage caused by resveratrol. Furthermore, in the group of patients exhibiting H2AX phosphorylation, a significantly higher percentage of apoptosis caused by resveratrol was observed, as compared to the group with no detectable H2AX phosphorylation. Thus, phosphorylation of H2AX makes the cells more prone to apoptosis and might be used as a predictor of apoptosis sensitivity.
In conclusion, the obtained results indicate that resveratrol alone and in combination with purine analogues warrants further study as a new therapeutic option for CLL patients. This naturally occurring substance may be especially useful in older patients for whom there are some limitations on the use of aggressive treatments. On the other hand, there is a possibility of using lower doses of purine analogues in combination with resveratrol because of their combined effect. The mechanisms of resveratrol action appear to be complex and remain to be precisely established. One of mechanisms in question is the induction of DNA damage, which ultimately leads to apoptosis.
Acknowledgments
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Calligaris-Cappio F, Hamblin TJ. B-cell chronic lymphocytic leukemia: a bird of a different feather. J Clin Oncol. 1999;17:399–408. doi: 10.1200/JCO.1999.17.1.399. [DOI] [PubMed] [Google Scholar]
- 2.Hamblin TJ, Oscier DG. Chronic lymphocytic leukemia: the nature of the leukemic cells. Blood Rev. 1997;11:119–128. doi: 10.1016/S0268-960X(97)90007-2. [DOI] [PubMed] [Google Scholar]
- 3.Gu X, Chu Q, O’Dwyer M, Zeece M. Analysis of resveratrol in wine by capillary electrophoresis. J Chromatogr A. 2000;881:471–481. doi: 10.1016/S0021-9673(00)00211-9. [DOI] [PubMed] [Google Scholar]
- 4.Clément MV, Hirpara JL, Chawdhury SH, Pervaiz S. Chemopreventive agent resveratrol, a natural product derived from grapes, triggers CD95 signaling-dependent apoptosis in human tumor cells. Blood. 1998;92:996–1002. [PubMed] [Google Scholar]
- 5.Bhat KPL, Kosmeder JW, Pezzuto JM. Biological effects of resveratrol. Antioxid Redox Signal. 2001;3:1041–1064. doi: 10.1089/152308601317203567. [DOI] [PubMed] [Google Scholar]
- 6.de la Lastra CA, Villegas I. Resveratrol as an antioxidant and pro-oxidant agent: mechanisms and clinical implications. Biochem Soc Trans. 2007;35:1156–1160. doi: 10.1042/BST0351156. [DOI] [PubMed] [Google Scholar]
- 7.Soleas GJ, Diamandis EP, Goldberg DM. The world of resveratrol. Adv Exp Med Biol. 2001;492:159–182. doi: 10.1007/978-1-4615-1283-7_13. [DOI] [PubMed] [Google Scholar]
- 8.Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992;8808:1523–1526. doi: 10.1016/0140-6736(92)91277-F. [DOI] [PubMed] [Google Scholar]
- 9.Pervaiz S, Holme AL. Resveratrol: its biologic targets and functional activity. Antioxid Redox Signal. 2009;11:2851–1897. doi: 10.1089/ars.2008.2412. [DOI] [PubMed] [Google Scholar]
- 10.Nicolini G, Rigolio R, Miloso M, Bertelli AA, Tredici G. Anti-apoptotic effect of trans-resveratrol on paclitaxel-induced apoptosis in the human neuroblastoma SH-SY5Y cell line. Neurosci Lett. 2001;302:41–44. doi: 10.1016/S0304-3940(01)01654-8. [DOI] [PubMed] [Google Scholar]
- 11.Jang M, Cai L, Udeani GO, Slowing KV, Thomas CF, Beecher CW, et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science. 1997;275:218–220. doi: 10.1126/science.275.5297.218. [DOI] [PubMed] [Google Scholar]
- 12.Jazirehi AR, Bonavida B. Resveratrol modifies the expression of apoptotic regulatory proteins and sensitizes non-Hodgkin’s lymphoma and multiple myeloma cell lines to paclitaxel-induced apoptosis. Mol Cancer Ther. 2004;3:71–84. [PubMed] [Google Scholar]
- 13.Huang X, Darzynkiewicz Z. Cytometric assessment of histone H2AX phosphorylation: a reporter of DNA damage. Meth Mol Biol. 2006;314:73–80. doi: 10.1385/1-59259-973-7:073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70:440–446. doi: 10.1158/0008-5472.CAN-09-1947. [DOI] [PubMed] [Google Scholar]
- 15.ten Cate B, Samplonius DF, Bijma T, de Leij LF, Helfrich W, Bremer E. The histone deacetylase inhibitor valproic acid potently augments gemtuzumab ozogamicin-induced apoptosis in acute myeloid leukemic cells. Leukemia. 2007;21:248–252. doi: 10.1038/sj.leu.2404477. [DOI] [PubMed] [Google Scholar]
- 16.Bouzar AB, Boxus M, Defoiche J, Berchem G, Macallan D, Pettengell R, et al. Valproate synergizes with purine nucleoside analogues to induce apoptosis of B-chronic lymphocytic leukaemia cells. Br J Haematol. 2009;144:41–52. doi: 10.1111/j.1365-2141.2008.07426.x. [DOI] [PubMed] [Google Scholar]
- 17.Aziz MH, Kumar R, Ahmad N. Cancer chemoprevention by resveratrol: in vitro and in vivo studies and the underlying mechanisms. Int J Oncol. 2003;23:17–28. [PubMed] [Google Scholar]
- 18.Gusman J, Malonne H, Atassi G. A reappraisal of the potential chemopreventive and chemotherapeutic properties of resveratrol. Carcinogenesis. 2001;22:1111–1117. doi: 10.1093/carcin/22.8.1111. [DOI] [PubMed] [Google Scholar]
- 19.Fontecave M, Lepoivre M, Elleingand E, Gerez C, Guittet O. Resveratrol, a remarkable inhibitor of ribonucleotide reductase. FEBS Lett. 1998;42:277–279. doi: 10.1016/S0014-5793(97)01572-X. [DOI] [PubMed] [Google Scholar]
- 20.Joe AK, Liu H, Suzui M, Vural ME, Xiao D, Weinstein IB. Resveratrol induces growth inhibition, S-phase arrest, apoptosis, and changes in biomarker expression in several human cancer cell lines. Clin Cancer Res. 2002;3:893–903. [PubMed] [Google Scholar]
- 21.Dong Z. Molecular mechanism of the chemopreventive effect of resveratrol. Mutat Res. 2003;523:145–150. doi: 10.1016/s0027-5107(02)00330-5. [DOI] [PubMed] [Google Scholar]
- 22.Pervaiz S. Resveratrol: from grapevines to mammalian biology. FASEB J. 2003;17:1975–1985. doi: 10.1096/fj.03-0168rev. [DOI] [PubMed] [Google Scholar]
- 23.Roman V, Billard C, Kern C, Ferry-Dumazet H, Izard JC, Mohammad R, et al. Analysis of resveratrol-induced apoptosis in human B-cell chronic leukaemia. Br J Haematol. 2002;117:842–851. doi: 10.1046/j.1365-2141.2002.03520.x. [DOI] [PubMed] [Google Scholar]
- 24.Billard C, Izard JC, Roman V, Kern C, Mathiot C, Mentz F, Kolb JP. Comparative antiproliferative and apoptotic effects of resveratrol, epsilon-viniferin and vine-shots derived polyphenols (vineatrols) on chronic B lymphocytic leukemia cells and normal human lymphocytes. Leuk Lymphoma. 2002;43:1991–2002. doi: 10.1080/1042819021000015952. [DOI] [PubMed] [Google Scholar]
- 25.Kurtova AV, Balakrishnan K, Chen R, Ding W, Schnabl S, Quiroga MP, Sivina M, Wierda WG, Estrov Z, Keating MJ, Shehata M, Jäger U, Gandhi V, Kay NE, Plunkett W, Burger JA. Diverse marrow stromal cells protect CLL cells from spontaneous and drug-induced apoptosis: development of a reliable and reproducible system to assess stromal cell adhesion-mediated drug resistance. Blood. 2009;114:4441–4450. doi: 10.1182/blood-2009-07-233718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shehata M, Schnabl S, Demirtas D, Hilgarth M, Hubmann R, Ponath E, Badrnya S, Lehner C, Hoelbl A, Duechler M, Gaiger A, Zielinski C, Schwarzmeier JD, Jaeger U (2010) Reconstitution of PTEN activity by CK2 inhibitors and interference with the PI3-K/Akt cascade counteract the anti-apoptotic effect of human stromal cells in chronic lymphocytic leukemia. Blood. Epub ahead of print [DOI] [PubMed]
- 27.Quiney C, Dauzonne D, Kern C, Fourneron JD, Izard JC, Mohammad RM, Kolb JP, Billard C. Flavones and polyphenols inhibit the NO pathway during apoptosis of leukemia B-cells. Leuk Res. 2004;28:851–861. doi: 10.1016/j.leukres.2003.12.003. [DOI] [PubMed] [Google Scholar]
- 28.Nabhan C, Gartenhaus RB, Tallman MS. Purine nucleoside analogues and combination therapies in B-cell chronic lymphocytic leukemia: dawn of a new era. Leuk Res. 2004;28:429–442. doi: 10.1016/j.leukres.2003.08.017. [DOI] [PubMed] [Google Scholar]
- 29.Van den Neste E, Cardoen S, Offner F, Bontemps F. Old and new insights into the mechanisms of action of two nucleoside analogs active in lymphoid malignancies: fludarabine and cladribine. Int J Oncol. 2005;27:1113–1124. [PubMed] [Google Scholar]
- 30.Robak T. Therapy of chronic lymphocytic leukemia with purine analogs and monoclonal antibodies. Transfus Apher Sci. 2005;32:33–44. doi: 10.1016/j.transci.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 31.Kobylinska A, Bednarek J, Blonski JZ, Hanausek M, Walaszek Z, Robak T. In vitro sensitivity of B-cell chronic lymphocytic leukemia to cladribine and its combinations with mafosfamide and/or mitoxantrone. Oncol Rep. 2006;16:1389–1395. [PubMed] [Google Scholar]
- 32.Morrison VA. Infectious complications in patients with chronic lymphocytic leukemia: pathogenesis, spectrum of infection, and approaches to prophylaxis. Clin Lymphoma Myeloma. 2009;9:365–370. doi: 10.3816/CLM.2009.n.071. [DOI] [PubMed] [Google Scholar]
- 33.Robak T, Błoński JZ, Urbańska-Ryś H, Błasińska-Morawiec M, Skotnicki AB. 2-Chlorodeoxyadenosine (Cladribine) in the treatment of patients with chronic lymphocytic leukemia 55 years old and younger. Leukemia. 1999;13:518–523. doi: 10.1038/sj/leu/2401368. [DOI] [PubMed] [Google Scholar]
- 34.Podhorecka M, Halicka HD, Klimek P, Kowal M, Dmoszynska A. Thalidomide induces phosphorylation of histone H2AX and increases rate of apoptosis caused by fludarabine in malignant lymphocytes of chronic lymphocytic leukemia in short-term cell cultures. Leuk Res. 2009;33:997–1000. doi: 10.1016/j.leukres.2008.09.023. [DOI] [PubMed] [Google Scholar]
- 35.Giannopoulos K, Dmoszynska A, Kowal M, Wasik-Szczepanek E, Bojarska-Junak A, Rolinski J, et al. Thalidomide exerts distinct molecular antileukemic effects and combined thalidomide/fludarabine therapy is clinically effective in high-risk chronic lymphocytic leukemia. Leukemia. 2009;23:1771–1778. doi: 10.1038/leu.2009.98. [DOI] [PubMed] [Google Scholar]
- 36.Chanan-Khan AA, Miller KC, Takeshita K, Koryzyna A, DonohueK BZP, et al. Results of a phase 1 clinical trial of thalidomide in combination with fludarabine as initial therapy for patients with treatment-requiring chronic lymphocytic leukemia (CLL) Blood. 2005;106:3348–3352. doi: 10.1182/blood-2005-02-0669. [DOI] [PubMed] [Google Scholar]
- 37.Stilgenbauer S. Chromic lymphocytic leukemia:genetics for predicting outcome. Hematology. 2006;2:185–190. [Google Scholar]
- 38.Crespo M, Bosch F, Villamor N, Bellosillo B, Colomer D, Rozman M, et al. ZAP-70 expression as a surrogate for immunoglobulin-variable-region mutations in chronic lymphocytic leukemia. N Engl J Med. 2003;348:1764–1775. doi: 10.1056/NEJMoa023143. [DOI] [PubMed] [Google Scholar]
- 39.Hamblin TJ, Davis Z, Gardiner A, Oscier DG, Stevenson FK. Unmutated Ig V(H) genes are associated with a more aggressive form of chronic lymphocytic leukemia. Blood. 1999;94:1848–1854. [PubMed] [Google Scholar]
- 40.Oscier DG, Thompsett A, Zhu D, Stevenson FK. Differential rates of somatic hypermutation in V(H) genes among subsets of chronic lymphocytic leukemia defined by chromosomal abnormalities. Blood. 1997;89:4153–4160. [PubMed] [Google Scholar]
- 41.De Salvia R, Festa F, Ricordy R, Perticone P, Cozzi R. Resveratrol affects in a different way primary versus fixed DNA damage induced by H(2)O(2) in mammalian cells in vitro. Toxicol Lett. 2002;135:1–9. doi: 10.1016/S0378-4274(02)00151-0. [DOI] [PubMed] [Google Scholar]
- 42.Ahmad KA, Clement MV, Pervaiz S. Pro-oxidant activity of low doses of resveratrol inhibits hydrogen peroxide-induced apoptosis. Ann NY Acad Sci. 2003;1010:365–373. doi: 10.1196/annals.1299.067. [DOI] [PubMed] [Google Scholar]
- 43.Zheng LF, Wei QY, Cai YJ, Fang JG, Zhou B, Yang L, et al. DNA damage induced by resveratrol and its synthetic analogues in the presence of Cu (II) ions: mechanism and structure-activity relationship. Free Radic Biol Med. 2006;4:1807–1816. doi: 10.1016/j.freeradbiomed.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 44.Azmi AS, Bhat SH, Hadi SM. Resveratrol-Cu(II) induced DNA breakage in human peripheral lymphocytes: implications for anticancer properties. FEBS Lett. 2005;579:3131–3135. doi: 10.1016/j.febslet.2005.04.077. [DOI] [PubMed] [Google Scholar]
- 45.Azmi AS, Bhat SH, Hanif S, Hadi SM. Plant polyphenols mobilize endogenous copper in human peripheral lymphocytes leading to oxidative DNA breakage: a putative mechanism for anticancer properties. FEBS Lett. 2006;580:533–538. doi: 10.1016/j.febslet.2005.12.059. [DOI] [PubMed] [Google Scholar]
- 46.Tanaka T, Halicka HD, Huang X, Traganos F, Darzynkiewicz Z. Constitutive histone H2AX phosphorylation and ATM activation, the reporters of DNA damage by endogenous oxidants. Cell Cycle. 2006;17:1940–1945. doi: 10.4161/cc.5.17.3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Darzynkiewicz Z, Huang X, Okafuji M. Detection of DNA strand breaks by flow and laser scanning cytometry in studies of apoptosis and cell proliferation (DNA replication) Meth Mol Biol. 2006;314:81–93. doi: 10.1385/1-59259-973-7:081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tyagi A, Singh RP, Agarwal C, Siriwardana S, Sclafani RA, Agarwal R. Resveratrol causes Cdc2-tyr15 phosphorylation via ATM/ATR-Chk1/2-Cdc25C pathway as a central mechanism for S phase arrest in human ovarian carcinoma Ovcar-3 cells. Carcinogenesis. 2005;26:1978–1987. doi: 10.1093/carcin/bgi165. [DOI] [PubMed] [Google Scholar]
- 49.Rusin M, Zajkowicz A, Butkiewicz D. Resveratrol induces senescence–like growth inhibition of U-2 OS cells associated with the instability of telomeric DNA and upregulation of BRCA1. Mech Ageing Dev. 2009;130:528–537. doi: 10.1016/j.mad.2009.06.005. [DOI] [PubMed] [Google Scholar]