C/EBPδ: friend or foe? A novel role for C/EBPδ in metastasis
This issue of The EMBO Journal features opposing functions of the assumed tumour suppressor C/EBPδ shown here to promote tumour metastasis. Mechanistically, C/EBPδ amplifies HIF-1α signalling, thus integrating hypoxic inflammation with tumour metastasis.
EMBO J 29 24, 4106–4117 (2010); published online November122010
Hypoxia contributes to major aspects of cancer biology such as angiogenesis, glucose/energy metabolism, invasion and metastasis. The activation of hypoxia-inducible factor 1α (HIF-1α) is a key step that promotes the transcriptional activation of target genes fundamental for these processes. In this issue of EMBO Journal, Balamurugan et al provide new insights on how hypoxia control integrates with inflammation and metastasis by describing an exciting role for the transcription factor C/EBPδ in the amplification of HIF-1α signalling. The authors show that C/EBPδ promotes the transcriptional repression of the tumour suppressor F-box and WD repeat-domain containing 7 protein (FBXW7), which in turn leads to mTOR protein stabilization and increased HIF-1α levels. Although C/EBPδ can act as a tumour suppressor gene at early stages of cancer development, it appears to have a positive role in progression and metastasis, thus adding to the ranks of genes that exert opposing effects at early and late stages of tumorigenesis.
An increasing body of evidence suggests that C/EBPδ functions as a tumour suppressor (Pawar et al, 2010; Wang et al, 2010). In this issue of EMBO Journal, Balamurugan et al provide in vivo evidence that germline deletion of this gene promotes increased tumour number in transgenic mice overexpressing the Neu/Her2/ERBB2 proto-oncogene in the mammary gland. Surprisingly, the authors report that though an increased number of primary tumours was observed in C/EBPδ-null mice, a paradoxical two-fold reduction in the incidence of metastasis was seen in the same animals. After some careful detective work, Balamurugan et al came to the conclusion that the mechanism involves an intricate interplay between C/EBPδ and HIF-1α. First, a link between hypoxia and C/EBPδ was established by the discovery of high immunoreactivity against C/EBPδ specifically in hypoxic areas of mammary tumours. Using isolated mammary tumour cells, the authors then showed that, while in WT cells HIF-1α is strongly induced under hypoxic culture conditions, cells derived from C/EBPδ-null tumours fail to show such induction. Fast growing tumours outpace the ability of vasculature to deliver enough oxygen and nutrients. HIF-1α activation is critical for adaptation of cells to hypoxic stress, in part, by promoting transcriptional changes leading to anaerobic metabolism (Semenza, 2010). C/EBPδ-null tumour cells fail to implement this adaptation as shown by decreased viability, glucose uptake and lactate production when subjected to hypoxia. These results seem to confirm a fundamental role of C/EBPδ in the establishment of a successful adjustment to low oxygen levels.
The serine-threonine protein kinase mTOR serves as a central regulator of cell metabolism, growth, proliferation and survival (Laplante and Sabatini, 2009). Oncogenic activation of this pathway by loss of function of several tumour suppressors (LKB1, PML, PTEN, TSC1/TSC2) or gain of function affecting protein tyrosine kinases, Ras and/or the downstream phosphotidylinositol-3-kinase/AKT pathway increases the rate of translation of HIF-1α mRNA into protein (Semenza, 2010). Using mouse embryonic fibroblasts, Balamurugan et al provide evidence to suggest that C/EBPδ increases HIF-1α induction by promoting its translation. Accordingly, the authors show that cells deficient in C/EBPδ have reduced activity of the phosphotidylinositol-3-kinase/AKT/mTOR pathway as shown by decreased basal and serum-induced AKT and S6K1 phosphorylation. These alterations are accompanied by decreased mTOR stability leading to defective HIF-1α activation, which can be rescued by exogenous C/EBPδ expression. Interestingly, in the absence of C/EBPδ, mTOR showed increased polyubiquitination and its reduced stability could be reversed by proteasome inhibition, leading to the conclusion that reduced mTOR levels in C/EBPδ-null cells are due to degradation by the ubiquitin-proteasome system.
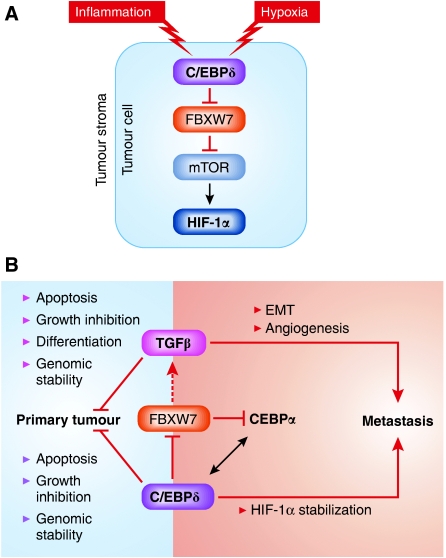
A further piece of this complex puzzle was the observation that C/EBPδ directly inhibits transcription of the FBXW7 gene. FBXW7 is a member of the F-box protein family and functions as a component of the SCF (SKP1-cullin-F-box) ubiquitin protein ligase complex, and a potent haploinsufficient tumour suppressor (Mao et al, 2004). Among the important oncoproteins controlled by FBXW7 at the level of protein stability are cMyc, Notch, cJun, and, importantly for this discussion, mTor (Mao et al, 2008; Welcker and Clurman, 2008). Balamurugan et al conclude that C/EBPδ, by inhibiting FBXW7 and causing increased stability of mTOR in the tumour cells, leads to an increase in HIF-1α protein levels, contributing to tumour progression and metastasis (Figure 1A).
Figure 1.
(A) C/EBPδ expression is restricted to hypoxic tumour areas and increases mTOR stability through repression of FBXW7. C/EBPδ acts as a tumour suppressor through various mechanisms leading to decreased primary tumour formation. Balamurugan et al now propose that C/EBPδ acts through HIF-1α stabilization to promote metastasis formation. This effect is dependent on repression of FBXW7, which leads to increased mTOR and HIF-1α. (B) C/EBPδ parallels TGFβ effects on tumorigenesis. C/EBPδ has also been shown by Bengoechea-Alonso et al to influence TGFβ signalling, although in the latter case the directionality of the effect in this context is unclear. FBXW7 also controls levels of CEBPα, and may mediate crosstalk between these transcription factors.
As with many interesting studies, however, the conclusions raise additional questions that remain to be addressed. Although the authors favour a cell-autonomous action of C/EBPδ in the mammary tumours, non-cell-autonomous effects in the stromal cells and tumour microenvironment cannot be ruled out, particularly as they used mice carrying a germline deletion of C/EBPδ. As the biological activity of FBXW7 is strongly dosage dependent, subtle changes in levels caused by loss of CEBPδ in multiple cell types may well contribute to the phenotypes observed. It is also difficult at this stage to reconcile the results of Balamurugan et al with published data on the significance of C/EBPδ in human oncogenesis. Chiefly, its inclusion in a 70-gene signature predicting better survival of breast cancer patients (Naderi et al, 2007) does not readily fit with its proposed pro-metastatic function. Again, the use of a global knockout strategy might obscure the effects of somatic loss of C/EBPδ during tumour progression and metastasis. In this regard, further studies using tissue specific and/or temporally controlled targeting could provide new and valuable insights. The finding that an early inflammatory response gene such as C/EBPδ amplifies HIF-1α induction begs the question of what is the role of this mechanism in immune-mediated responses to tumour cells. Recent data (Corzo et al, 2010; Doedens et al, 2010) suggesting a prominent role for HIF-1α in tumour-associated macrophage biology raise exciting questions about the potential role of C/EBPδ in these processes.
In the light of these new data, C/EBPδ joins the ranks of other genes with a dual role in tumorigenesis, the paradigmatic example being TGFβ. Indeed, the parallels between these biological pathways are quite striking (Figure 1B). The tumour suppressor activity of both proteins has been ascribed to their ability to cause growth arrest, induce apoptosis and control genomic stability. Both have suppressor effects on early stage tumour development, but can promote progression and metastasis. The mechanisms used by TGFβ to promote metastasis have been intensively investigated and include both cell-autonomous effects in induction of the epithelial mesenchymal transition leading to an invasive metastatic phenotype, as well as non-cell-autonomous effects on the immune system and tumour microenvironment (Barcellos-Hoff and Akhurst, 2009). It remains to be seen whether such a disparate range of mechanisms is also employed by C/EBPδ to influence tumour progression. Tantalizing links between FBXW7 and both TGFβ and CEBPα signalling (Bengoechea-Alonso and Ericsson, 2010a, 2010b) provide a hint of the complexity of the interacting networks that endow cancer cells with their seemingly endless flexibility in discovering new routes to progression.
Acknowledgments
FGP is partially supported by a postdoctoral fellowship from the Pew Latin American Fellows Program. AB acknowledges support from the NCI Mouse Models of Human Cancer Consortium and the Barbara Bass Bakar Chair of Cancer Genetics.
Footnotes
The authors declare that they have no conflict of interest.
References
- Balamurugan K, Wang J-M, Tsai H-H, Sharan S, Anver M, Leighty R, Sterneck E (2010) The tumour suppressor C/EBPδ inhibits FBXW7 expression and promotes mammary tumour metastasis. EMBO J 29: 4106–4117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barcellos-Hoff MH, Akhurst RJ (2009) Transforming growth factor-beta in breast cancer: too much, too late. Breast Cancer Res 11: 202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bengoechea-Alonso MT, Ericsson J (2010a) Tumor suppressor Fbxw7 regulates TGFbeta signaling by targeting TGIF1 for degradation. Oncogene 29: 5322–5328 [DOI] [PubMed] [Google Scholar]
- Bengoechea-Alonso MT, Ericsson J (2010b) The ubiquitin ligase Fbxw7 controls adipocyte differentiation by targeting C/EBPalpha for degradation. Proc Natl Acad Sci U S A 107: 11817–11822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P, Cho HI, Celis E, Quiceno DG, Padhya T, McCaffrey TV, McCaffrey JC, Gabrilovich DI (2010) HIF-1alpha regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J Exp Med 207: 2439–2453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doedens AL, Stockmann C, Rubinstein MP, Liao D, Zhang N, DeNardo DG, Coussens LM, Karin M, Goldrath AW, Johnson RS (2010) Macrophage expression of hypoxia-inducible factor-1 alpha suppresses T-cell function and promotes tumor progression. Cancer Res 70: 7465–7475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laplante M, Sabatini DM (2009) mTOR signaling at a glance. J Cell Sci 122: 3589–3594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao JH, Kim IJ, Wu D, Climent J, Kang HC, DelRosario R, Balmain A (2008) FBXW7 targets mTOR for degradation and cooperates with PTEN in tumor suppression. Science 321: 1499–1502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao JH, Perez-Losada J, Wu D, Delrosario R, Tsunematsu R, Nakayama KI, Brown K, Bryson S, Balmain A (2004) Fbxw7/Cdc4 is a p53-dependent, haploinsufficient tumour suppressor gene. Nature 432: 775–779 [DOI] [PubMed] [Google Scholar]
- Naderi A, Teschendorff AE, Barbosa-Morais NL, Pinder SE, Green AR, Powe DG, Robertson JF, Aparicio S, Ellis IO, Brenton JD, Caldas C (2007) A gene-expression signature to predict survival in breast cancer across independent data sets. Oncogene 26: 1507–1516 [DOI] [PubMed] [Google Scholar]
- Pawar SA, Sarkar TR, Balamurugan K, Sharan S, Wang J, Zhang Y, Dowdy SF, Huang AM, Sterneck E (2010) C/EBP{delta} targets cyclin D1 for proteasome-mediated degradation via induction of CDC27/AP. Proc Natl Acad Sci USA 107: 9210–9215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semenza GL (2010) HIF-1: upstream and downstream of cancer metabolism. Curr Opin Genet Dev 20: 51–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Sarkar TR, Zhou M, Sharan S, Ritt DA, Veenstra TD, Morrison DK, Huang AM, Sterneck E (2010) CCAAT/enhancer binding protein delta (C/EBPdelta, CEBPD)-mediated nuclear import of FANCD2 by IPO4 augments cellular response to DNA damage. Proc Natl Acad Sci U S A 107: 16131–16136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welcker M, Clurman BE (2008) FBW7 ubiquitin ligase: a tumour suppressor at the crossroads of cell division, growth and differentiation. Nat Rev Cancer 8: 83–93 [DOI] [PubMed] [Google Scholar]