Abstract
Mycobacterium ulcerans causes Buruli ulcer, a potentially disabling ulcerative skin disease. Only recently was antimicrobial therapy proven effective. Treatment for 2 months with rifampin plus streptomycin was first proposed after experiments in the mouse footpad model demonstrated bactericidal activity. This treatment is now considered the treatment of choice, although larger ulcers may require adjunctive surgery. Shorter, oral regimens are desired, but evaluating drug activity in mice is hampered by the very slow growth of M. ulcerans, which takes 3 months to produce countable colonies. We created a recombinant bioluminescent M. ulcerans strain expressing luxAB from Vibrio harveyi for real-time evaluation of antimicrobial effects in vivo. Mouse footpads were injected with wild-type M. ulcerans 1059 (WtMu) or the recombinant bioluminescent strain (rMu). Two weeks later, mice received rifampin plus streptomycin, kanamycin alone (to which rMu is resistant), or streptomycin alone for 4 weeks and were observed for footpad swelling (preventive model). Untreated controls and kanamycin-treated rMu-infected mice received rifampin plus streptomycin for 4 weeks after developing footpad swelling (curative model). Compared to WtMu, rMu exhibited similar growth and virulence in vivo and similar drug susceptibility. A good correlation was observed between luminescence (measured as relative light units) and number of viable bacteria (measured by CFU) in footpad homogenates. Proof of concept was also shown for serial real-time evaluation of drug activity in live mice. These results indicate the potential of bioluminescence as a real-time surrogate marker for viable bacteria in mouse footpads to accelerate the identification of new treatments for Buruli ulcer.
Buruli ulcer (BU) is an emerging, yet neglected, infectious disease occurring in certain humid tropical regions of the world (17, 22). The causative agent, Mycobacterium ulcerans, produces a mycolactone toxin which is responsible for subcutaneous necrosis leading to ulceration (8, 9). Traditionally, BU was managed by surgical excision with or without skin grafting. Antimicrobial therapy was thought to be ineffective until experiments in the mouse footpad model demonstrated the efficacy of combinations composed of an aminoglycoside and rifampin (RIF) (2, 6, 13). Subsequent clinical experience has confirmed that 2 months of streptomycin (STR) and RIF is the treatment of choice for all forms of BU, although adjunctive surgery may be necessary to heal large ulcers (4, 7, 12, 14). Although this regimen is now recommended for treating BU (22), it has significant disadvantages in that STR requires parenteral administration and may cause ototoxicity and nephrotoxicity, and the 2-month duration of therapy poses an obstacle to treatment completion. Therefore, a need exists to develop entirely oral regimens and/or regimens capable of treating BU in 1 month or less.
Efforts to develop new treatments for BU are hampered by the very slow growth of M. ulcerans, which requires 3 months of incubation to form colonies on solid media. We have recently demonstrated that recombinant bioluminescent reporter strains of M. ulcerans are useful for real-time evaluation of antimicrobial activity in vitro (23). In the present study, we sought to determine whether such bioluminescent strains are useful for real-time evaluation of antimicrobial effects in the mouse footpad model as well. To date, the endpoints used to measure efficacy in this model have included the median time to footpad swelling and the change in CFU counts in footpad homogenates. Experiments based on the latter outcome measure require 4 to 6 months for assessment of the drug effect. While the former outcome measure requires just a little longer than the time it takes for untreated footpads to swell (∼8 to 10 weeks) for active drugs to be distinguished from inactive drugs, this outcome measure requires a longer time for active regimens to be distinguished from one another (e.g., bacteriostatic from bactericidal regimens) as well as large numbers of mice for adequate statistical power for survival analyses.
Bioluminescent reporter strains offer the promise of real-time monitoring of bacterial viability during and after treatment, including the potential for noninvasive measurements using available imaging technology, such as sensitive charge-coupled-device (CCD) cameras capable of detecting low levels of light emission from luciferase reporters in vivo (10). In this study, we used a recombinant M. ulcerans strain (rMu) engineered to express the luxAB gene from Vibrio harveyi for proof-of-concept experiments to determine whether such bioluminescent reporter strains could be useful for such real-time evaluation of drug activity in the mouse footpad model of BU.
MATERIALS AND METHODS
Creation and selection of a recombinant bioluminescent strain.
Various bioluminescent constructs were introduced into 4 different strains of M. ulcerans (Cu001 [originally isolated from Côte d'Ivoire], Mu1059 [originally isolated from Ghana], Mu1615 [originally isolated from Malaysia and also known as ATCC 35840], and ATCC 19423 [originally isolated from Australia]) and evaluated for their stability, light production, and drug susceptibility, as previously described (23). The recombinant bioluminescent ATCC 19423, Mu1615, and Mu1059 strains and control strains containing empty plasmids were evaluated for virulence by injecting approximately 0.03 ml of a culture suspension containing approximately 3 × 104 CFU into the hind footpads of 4- to 6-week-old female BALB/c, C57BL/6, or BALB/c SCID mice (Charles River, Germantown, MD). The mice were observed over time for the development of footpad swelling.
Assessment of mycolactone production.
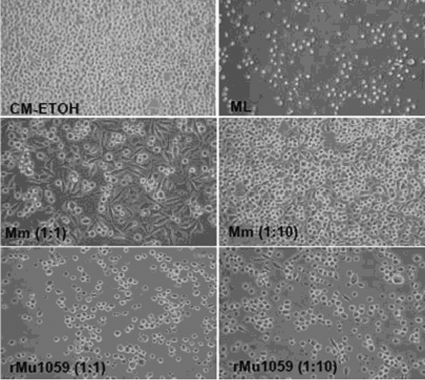
Purified mycolactone A/B and the mycolactone extraction method were generously provided by Pamela Small (University of Tennessee). Mycobacterium marinum M was a kind gift from Lian-Yong Gao (University of Maryland). In brief, M. ulcerans or M. marinum was grown on 7H11 agar, and colonies were scraped together. Colony material (73 mg of the recombinant Mu1059 strain expressing plasmid pTY60K and 167 mg of M. marinum M) was placed in a glass flask masked with aluminum foil. Ethanol (100%) was added to give a final volume of 10 ml. The mixture was stirred for 2 h and then centrifuged at 3,000 rpm for 10 min to spin down cellular debris. The supernatant was removed to another masked flask for dry down under N2 at no more than 60°C. Lipids were resuspended in 100 μl ethanol, and thin-layer chromatography (TLC) was performed as described by George et al. (8). A cytotoxicity assay was performed as previously described (9). Monolayers of L929 fibroblasts were set up in Dulbecco modified Eagle medium supplemented with 10% heat-inactivated fetal calf serum (Gibco BRL, Grand Island, NY) in 24-well plates with approximately 2 × 105 cells/well. Three microliters of ethanol extract was diluted 1:1 or 1:10 and added to the wells on the following day. Purified mycolactone was used as a positive control. Culture medium with or without ethanol was used as a negative control. Plates were incubated at 37°C with 5% CO2 for 72 h before cytotoxicity was assessed. Photomicrographs of the cells were taken with an optical microscope (catalog no. 12-560-45 [magnification, ×20]; Fisher Scientific).
Antibiotics.
RIF and STR were purchased from Sigma (St. Louis, MO). Kanamycin (KAN) was purchased from Invitrogen (Carlsbad, CA). The MICs of KAN against the wild-type Mu1059 strain (WtMu) and the recombinant Mu1059 strain expressing the bioluminescence plasmid pTY60K (rMu) are <10 μg/ml and >25 μg/ml, respectively, whereas the MICs of STR against both strains are 0.25 μg/ml (23). The MICs of RIF against WtMu and rMu were previously reported to be 0.13 μg/ml and 0.5 μg/ml, respectively, but the difference is likely to be a chance finding from a single experiment, as transformation of the ATCC 19423 strain using the same procedure did not affect the RIF MIC (23). All drugs were dissolved in distilled water and administered 5 days per week in 0.2 ml. RIF was administered by gavage. STR and KAN were administered by subcutaneous injection. The daily dosages were 10 mg/kg of body weight for RIF and 150 mg/kg for STR and KAN (6, 11).
Murine footpad infection models.
All procedures were approved by the institutional Animal Care and Use Committee of Johns Hopkins University. Four-week-old female BALB/c mice were injected in the left hind footpad with 0.03 ml of culture suspension of WtMu or rMu in phosphate-buffered saline (PBS). On the same day, the suspension was serially diluted and 500-μl aliquots were plated on Middlebrook 7H11 selective agar (21) to determine the CFU counts.
Three parameters were used to assess the severity of infection and the efficacy of treatment: (i) the footpad lesion index, (ii) footpad relative light unit (RLU) counts, and (iii) footpad CFU counts. The scoring of the lesion index has been described previously (6). Briefly, the presence and the degree of inflammatory swelling of the infected footpad are assessed weekly and scored from 0 (no swelling) to 4 (inflammatory swelling extending to the entire limb).
RLU counts in infected footpads were determined in 1 of 2 ways. In sacrificed mice, footpad tissue was removed aseptically, minced, and placed in 2 ml PBS. RLU counts were determined by adding 20 μl of 1% decanal (vol/vol in alcohol) to a 200-μl footpad sample in 1.5 ml polystyrene tubes (USA Scientific). Light production was measured for 4 s without delay using a TD20/20 luminometer (Turner BioSystems) and duplicate samples. Live mice were anesthetized with Avertin (Sigma) (protocol available online [http://herzenberg.stanford.edu/protocols/avertin.htm]) and injected in the footpad with ∼50 μl of 1% decanal, whereupon the mouse was placed in the luminometer and light production was measured for 4 s. For this study, we defined 1 RLU as 1 RLU in the 20/20n luminometer, which is equivalent to 0.001 units in the TD20/20 luminometer (20, 23).
CFU counts in infected footpads were determined by plating 0.5-ml aliquots of serial dilutions of tissue homogenates in duplicate on selective 7H11 agar. Colonies were counted after 3 to 4 months of incubation at 32°C. Because only half of the total footpad homogenate was plated, the lower limit of detection was 2 CFU per footpad.
Preventive therapy model.
In this model, adapted from the kinetic method of Shepard (19), 125 mice were infected with WtMu and 160 mice were inoculated with rMu before random allocation to the following treatment groups: those receiving no treatment, STR-RIF, KAN alone, and STR alone (only rMu-infected mice were allocated to STR alone). Five mice per group were sacrificed the day after infection and weekly thereafter for determination of RLU and CFU counts from footpad homogenates (11, 13). Treatment began 2 weeks after infection, when the lesion index was still 0, and lasted for 4 weeks. After treatment, the remaining 15 mice in each group were monitored weekly for an additional 23 weeks to determine the time to footpad swelling, defined by a lesion index of 1 (noninflammatory swelling). Three mice per group were sacrificed for RLU and CFU detection after 10 weeks of follow-up to see if RLU counts can provide an early marker of treatment outcome before swelling occurs. All other mice were sacrificed at the end of the 23-week follow-up period and used for RLU and CFU counts.
Curative therapy model.
In this model, 25 mice from each of the untreated groups (WtMu and rMu infected) in the preventive therapy model were assigned to receive RIF-STR for 4 weeks after inflammatory swelling of the entire footpad had developed (i.e., lesion index of 3). No mice went untreated, as our animal care and use protocol requires euthanasia at this clinical stage unless treatment is performed. Mice were sacrificed at baseline and then weekly for RLU and CFU counts from footpad homogenates. An additional 15 rMu-infected mice which developed a similar degree of swelling despite treatment with KAN in the preventive therapy model were randomly allocated to 1 of 2 groups to assess the potential for serial monitoring of RLU counts in live mice. All mice received RIF-STR. In the first group of 9 mice, 3 mice were used each week to detect RLU under anesthesia and then sacrificed for determination of RLU and CFU counts from footpad homogenates. The second group of 6 mice was serially evaluated for RLU counts under anesthesia, except that 2 mice were sacrificed each week for RLU and CFU counts from footpad homogenates.
Data analysis.
RLU and CFU counts were log transformed before analysis. Data are presented as mean ± standard deviation throughout the study. Group means were compared by an unpaired t test or by one-way analysis of variance (ANOVA) with Dunnett's posttest when multiple comparisons were made. An alpha value of 0.05 was used to determine statistical significance. Time-to-swelling curves were compared using the log rank test. As 5 pairwise comparisons were made in the time-to-swelling analysis, an alpha value of 0.01 was used to determine statistical significance. All statistical tests were performed with Prism 4 software (GraphPad Software, Inc., San Diego, CA).
RESULTS
Selection of a virulent, stable recombinant bioluminescent strain.
Despite successful transformation of the ATCC 19423 strain of M. ulcerans with a variety of extrachromosomal plasmids (pNBV1 [luxAB-free control], p60LUX, pG13LUX, and pMOPLUX) and integrative plasmids (pCK0601 [luxAB-free control], pTY60H, and pTYG13H) (23), none of the transformants reliably caused footpad swelling after injection into BALB/c, C57BL/6, or even SCID mice (data not shown). However, both the Mu1059 and the Mu1615 strains transformed with the integrative plasmid pTY60K with a KAN resistance marker and luxAB under the Hsp60 promoter (23) produced reliable swelling which did not appear to differ from that produced by the wild-type strain (data not shown).
Comparing the mycolactones produced by the ATCC 19423 strain and the well-studied Mu1059 (16) and Mu1615 (8, 16) strains by TLC revealed that both the Mu1059 and the Mu1615 strains produce mycolactone A/B (15) but that the ATCC 19423 strain did not (data not shown). These results are consistent with a prior report that M. ulcerans strains from Australia do not produce mycolactone A/B (15). The mycolactone extracted from the Mu1059(pTY60K), or rMu, strain showed significant cytotoxicity at the 1:1 and 1:10 dilutions compared to the negative control and the extract from M. marinum M (Fig. 1). Based on these data, as well as evidence from a pilot experiment that the rMu strain caused footpad swelling similar to that caused by the wild type (data not shown), this strain was selected for subsequent experiments.
FIG. 1.
L929 murine fibroblasts were treated for 48 h with lipid extracts from mycobacteria. CM-ETOH, culture medium with ethanol; ML, purified mycolactone A/B (100 ng/ml); Mm, extract from M. marinum M; rMu1059, extract from recombinant Mu1059(pTY60K).
The recombinant bioluminescent strain (rMu) is virulent in the murine footpad model.
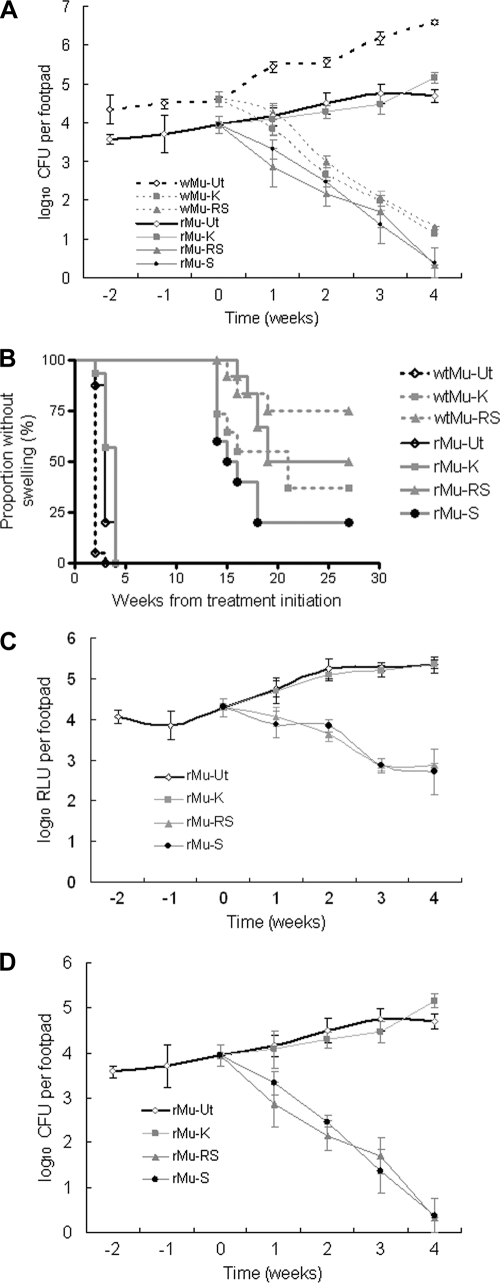
The bacterial titers of the broth culture used to infect mouse footpads were 5.78 log10 CFU/ml for WtMu and 4.45 log10 CFU/ml for rMu. The CFU counts obtained over time are shown in Fig. 2A. On the day after injection, the mean footpad CFU counts in rMu-infected mice (3.58 ± 0.12 log10) were 0.77 log10 lower than those in WtMu-infected mice (4.35 ± 0.36 log10). Although the levels of multiplication of the 2 strains were similar over the first 2 weeks prior to treatment, the WtMu strain experienced a greater increase in CFU than the rMu strain in untreated mice thereafter. However, this difference may have been due to the difference in the infectious dose. The median times to swelling in untreated WtMu- and rMu-infected mice were 4 and 5 weeks, respectively, after infection (P < 0.0001) (Fig. 2B). All untreated mice developed swelling by the 6th week of infection. Considering the difference in the number of CFU injected and its expected impact on the time-to-swelling results (1), the rMu strain appears similar in virulence to its parent strain.
FIG. 2.
Results for CFU counts, time to footpad swelling, and RLU counts in the preventive therapy model. WtMu, wild-type Mu1059 strain; rMu, recombinant bioluminescent Mu1059(pTY60K) strain. Ut, untreated; K, KAN treated; RS, RIF-STR treated; S, STR treated. (A) mean CFU counts for all groups; (B) time-to-footpad-swelling curves for all groups; (C) mean RLU counts for rMu-infected groups; (D) mean CFU counts for rMu-infected groups (for comparison with the corresponding RLU results in panel C). Week 0 is the point of treatment initiation.
WtMu and rMu strains have similar susceptibilities to RIF-STR but not to KAN.
On the basis of footpad CFU counts (Fig. 2A), the standard RIF-STR regimen had similar bactericidal activities against both strains, resulting in roughly 3-log10 reductions in footpad CFU counts over 4 weeks of treatment. Treatment with KAN was as effective as that of RIF-STR against WtMu but was ineffective against rMu, confirming the presence of the resistance marker. On the other hand, STR alone was as effective as RIF-STR against rMu.
On the basis of time to footpad swelling (Fig. 2B), all treatments were more effective than no treatment against either strain. Even KAN modestly delayed the time to swelling in mice infected with rMu (P = 0.0055), extending the median time to swelling from 3 to 4 weeks after treatment, perhaps because maximal serum concentrations of KAN exceeded the MIC for a fraction of the dosing interval. Treatment with RIF-STR produced similar time-to-swelling curves in mice infected with either strain (P = 0.2701). In mice infected with WtMu, swelling occurred earlier and more often after KAN treatment than after RIF-STR treatment (P = 0.0392). Similarly, in mice infected with rMu, time to swelling occurred earlier and more often after STR treatment than after RIF-STR treatment (P = 0.0131). These differences between the effect of RIF-STR and that of an aminoglycoside alone were statistically significant before, but not after, adjustment for multiple comparisons.
Taken together, these results demonstrate that (i) the WtMu and rMu strains respond similarly to treatment with non-KAN-containing regimens, (ii) the KAN resistance cassette in rMu does not confer cross-resistance to STR, and (iii) although RIF does not contribute to the ability of the RIF-STR regimen to reduce footpad CFU counts, it may play a role in delaying and/or preventing footpad swelling after treatment.
RLU counts constitute an effective real-time surrogate marker for CFU counts.
Comparing RLU counts (Fig. 2C) and CFU counts (Fig. 2D) from footpad homogenates of rMu-infected mice demonstrates that RLU counts correlated very well with CFU counts for each treatment group until the RLU counts in mice receiving RIF-STR and STR alone approached the background luminescence level between the third and fourth weeks of treatment. Based on the CFU counts obtained at this time point, the lower limit of bioluminescent bacteria which could be discriminated from background level was between 2.16 and 1.70 log10 CFU per footpad. After 1 week of treatment, the mean RLU count in RIF-STR-treated mice was significantly lower than the mean RLU count at day 0 (P < 0.01), whereas the mean RLU count in untreated mice was significantly higher (P < 0.01). The mean RLU count in STR-treated mice was lower than the day 0 count, but the difference was not statistically significant. After 2 weeks of treatment, both RIF-STR- and STR-treated mice had significantly lower mean footpad RLU counts than did day 0 mice, whereas untreated mice still had significantly higher mean footpad RLU counts.
Conclusions based on CFU counts were similar. Compared to those in day 0 mice, mean footpad CFU counts were significantly reduced after 1 week of treatment with RIF-STR (P < 0.01) or STR alone (P < 0.05). CFU counts in untreated mice were not statistically significantly higher than baseline CFU counts. After 2 weeks of treatment, both RIF-STR and STR-treated mice had significantly lower CFU counts than day 0 mice (P < 0.01), whereas untreated mice had significantly higher footpad CFU counts than day 0 mice (P < 0.05). These results indicate that RLU counts provided an accurate and sensitive real-time surrogate marker for CFU counts which may permit discrimination of bacteriostatic (e.g., if RLU counts are lower than those in untreated controls but no lower than those at day 0) and bactericidal (e.g., if RLU counts are lower than day 0 counts) drugs and regimens 3 months before CFU counts are available.
Three mice without swelling in each treated group were sacrificed after 10 weeks of follow-up without treatment to determine if they were RLU and/or CFU positive to assess whether bioluminescence could be used to assess treatment response before footpad swelling occurs. However, no RLU were detected in any footpad homogenate. Among the rMu-infected mice, CFU counts revealed that 2 mice in the STR alone group were culture negative but that the other 4 (1 from the STR alone group and 3 from the RIF-STR group) were culture positive, with 0.3 to 2.36 log10 CFU/footpad, indicating that (i) light produced by ≤2.36 log10 CFU per footpad was not readily differentiated from the background luminescence level and (ii) detectable RLU may be an earlier indicator of failure of bacterial eradication than swelling.
luxAB construct and gene expression are stable in rMu.
Mice that developed swollen footpads after treatment were euthanized at 10, 15, 19, and 23 weeks after treatment completion. RLU counts were over 4 log10 RLU/footpad in all rMu-infected mice with swollen footpads. Furthermore, all 272 randomly selected colonies from rMu-treated mice at week 19 (176 colonies from 6 mice) and at week 27 (96 colonies from 6 mice) were luminescent, indicating that the luxAB construct is very stable in vivo even after antibiotic therapy.
Utility of RLU counts as a surrogate marker for CFU counts in the curative therapy model.
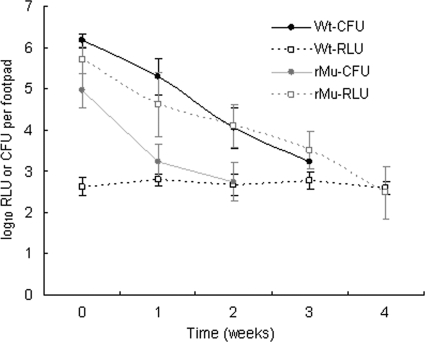
As expected, RIF-STR had strong bactericidal activity in this model of established disease, reducing the footpad CFU counts by approximately 2 log10 in both WtMu- and rMu-infected mice over the first 2 weeks of treatment (Fig. 3). RLU counts in WtMu-infected mice identified a background luminescence level of just under 3 log10 RLU in swollen footpads, the same level at which RLU counts ceased to decline in Fig. 2C. In rMu-infected mice, RLU counts correlated very well with CFU counts up to the week 2 time point, when the CFU count was 2.74 ± 0.47 log10 per footpad. Unfortunately, the CFU counts in rMu-infected mice at weeks 3 and 4 could not be determined, due to contaminated culture plates (as was the case for WtMu-infected mice at week 4). However, RLU counts in rMu-infected mice continued to fall from week 2 to week 4, paralleling the fall in CFU in WtMu-infected mice, and only reached the background level at week 4. This suggests that RLU counts remained above the background level even as the CFU count in rMu-infected mice declined well below 2.5 log10 CFU. These data are again consistent with the prior observations that the lower limit of RLU detectable above the baseline level is between 100 and 200 CFU. The complication of contamination further demonstrates the potential utility of RLU counts, which, in addition to being available in real-time, are not subject to the risk of contamination.
FIG. 3.
Correspondence of CFU and RLU counts in the curative therapy model. Wt, wild-type Mu1059 strain; rMu, recombinant bioluminescent Mu1059(pTY60K) strain.
Feasibility of serial real-time assessment of the response to therapy in live mice.
We also used RIF-STR treatment of rMu-infected mice from the same curative model experiment to compare serial RLU counts measured in the intact footpads of living, anesthetized mice to RLU counts measured in homogenized footpads from mice sacrificed during treatment. As expected, RLU counts were higher in footpad homogenates than in intact footpads (Fig. 4). The mean RLU counts in footpad homogenates paralleled the results displayed in Fig. 3, falling from 5.67 ± 0.54 to 5.16 ± 0.13 log10 during the first week of treatment with RIF-STR (P = 0.0687). After the second week of treatment, the mean RLU count was 4.61 ± 0.85 (P = 0.0238). Although RLU counts measured in intact footpads were lower, similar results were obtained. The difference between the mean RLU count before treatment (3.12 ± 0.40) and that observed after 1 week of treatment with RIF-STR (3.03 ± 0.42) was not significant, but the mean RLU count after 2 weeks of treatment with RIF-STR (2.07 ± 0.39) was significantly lower than the initial day 0 value (P = 0.0018). These results indicate the potential utility of bioluminescent reporter strains of M. ulcerans for serial, real-time monitoring of drug effects in live mice. Based on the week 2 rMu CFU results shown in Fig. 3 and the fact that the RLU count in intact footpads of RIF-STR-treated live mice continued to fall from week 2 to week 3, the dynamic range of the RLU assay in intact footpads extends below 2.74 log10 CFU.
FIG. 4.
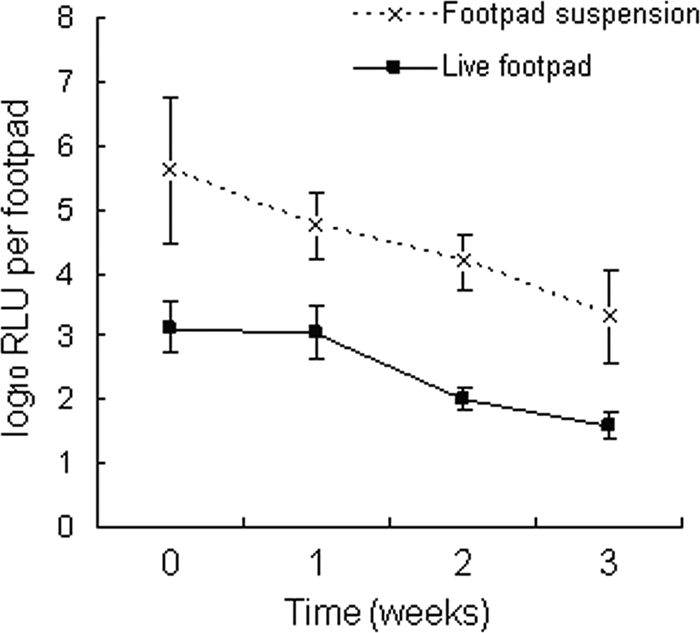
Results for serial RLU counts in live, anesthetized mice compared to those in footpad homogenates. WtMu, wild-type Mu1059 strain; rMu, recombinant bioluminescent Mu1059(pTY60K) strain. The background RLU counts in WtMu-infected mice are approximately 2.7 log10 RLU/footpad with the use of the footpad suspension method and approximately 1.5 log10 RLU/footpad with the use of live mice.
DISCUSSION
The slow growth of M. ulcerans is an obstacle for assessing new drugs against this pathogen. It necessitates prolonged cultivation in vitro to record CFU counts, which is not only time-consuming but prone to contamination. Measuring time to swelling after treatment of infected mouse footpads in a preventive therapy model may avoid the complications of CFU enumeration and may discriminate active from inactive drugs in as little as 8 to 10 weeks but requires at least 12 to 14 weeks to determine whether a bactericidal effect has occurred and even longer observation periods to gauge the extent of bactericidal activity. Here, we demonstrate that a recombinant bioluminescent M. ulcerans strain expressing luxAB from V. harveyi enables real-time monitoring of the viable bacterial population in mouse footpads during experimental chemotherapy and may significantly shorten the time needed to evaluate new drug candidates in vivo. Moreover, we demonstrate proof of concept that one can serially monitor drug activity using bioluminescence in live mice and therefore reduce the numbers of mice required and improve the efficiency of experimentation by stopping or otherwise modifying experiments on the basis of real-time information.
We found the Mu1059 strain transformed with pTY60K to be essentially as virulent as the wild-type parent strain. In addition, it has the same drug susceptibility profile as the parent, with the exception of the KAN resistance marker, and the RLU counts generated by this strain were previously shown to be a good real-time marker of CFU counts after exposure to a variety of antibiotics in vitro (23). Measuring luminescence (as RLU counts) may be a very useful surrogate marker for CFU counts in either the preventive or the curative murine footpad model, because RLU results correlated well with CFU counts in both models, predicted which treatment groups would go on to develop swelling before the others, and, most importantly, were available in real time 3 months earlier than CFU counts. We did not observe a significant light-quenching effect from footpad tissue, as has been described previously for samples from internal organs using different genetic constructs (18, 20).
By serially measuring RLU from the footpads of live, anesthetized mice during treatment and in parallel with mice sacrificed for footpad RLU and CFU counts, we have shown proof of concept for real-time, noninvasive monitoring of drug effects. Our method, which relied on a tabletop luminometer, has obvious advantages over other live-imaging methods, which measure radioactivity (5) or require more-expensive CCD cameras (3). In addition, it measures bioluminescence as a continuous value which is more objective and robust for statistical analyses than the occurrence of swelling. Hence, real-time bioluminescent reporter strains monitored with this system have the potential to significantly reduce both the amount of time and the number of animals required for evaluation of the effects of new drugs or drug regimens against M. ulcerans in mice. As a quantitative measure of viable bacteria, the system should also be useful for the study of vaccine efficacy if the effect of the vaccine is to limit M. ulcerans growth.
Current limitations of the system include the need to provide exogenous substrate for the luciferase reaction, which hampers serial noninvasive monitoring due to the need for repeated footpad injections, and the reduced sensitivity of measuring luminescence from whole footpads, which may result from poor diffusion of substrate in the footpad after injection. Efforts are under way to engineer strains which constitutively produce both luciferase and its substrate in hope of addressing both of these limitations. Additionally, the luminescence of the rMu strain cannot be differentiated from the background luminescence of infected footpads when the bacterial population falls much below ∼200 organisms per footpad homogenate. This may limit the ability to detect differences in activity between highly active compounds or regimens. However, a sufficient window remains for discrimination of active compounds from inactive compounds and bacteriostatic compounds from bactericidal compounds as well as discrimination of weak bactericidal effects from strong bactericidal effects. Additional studies with classes of drugs which act by different mechanisms of action will be required before this system can be considered to be qualified for routine use. However, in vitro studies with the same bioluminescent strain have suggested that RLU and CFU counts correlate well regardless of the drug class tested (23).
Acknowledgments
We acknowledge the financial support of the National Institute of Allergy and Infectious Diseases (R01 AI082612) and the Raoul Follereau foundation.
We also thank Richard L. Friedman at the University of Arizona for providing the p60LUX plasmid as a gift. We acknowledge the support of Pamela Small at the University of Tennessee and her kind gifts of the 1059 and 1615 strains, purified mycolactone A/B, and the protocol for extracting mycolactone from M. ulcerans.
Footnotes
Published ahead of print on 15 November 2010.
REFERENCES
- 1.Addo, P., E. Owusu, B. Adu-Addai, M. Quartey, M. Abbas, A. Dodoo, and D. Ofori-Adjei. 2005. Findings from a buruli ulcer mouse model study. Ghana Med. J. 39:86-93. [PMC free article] [PubMed] [Google Scholar]
- 2.Bentoucha, A., J. Robert, H. Dega, N. Lounis, V. Jarlier, and J. Grosset. 2001. Activities of new macrolides and fluoroquinolones against Mycobacterium ulcerans infection in mice. Antimicrob. Agents Chemother. 45:3109-3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berger, F., R. Paulmurugan, S. Bhaumik, and S. S. Gambhir. 2008. Uptake kinetics and biodistribution of 14C-D-luciferin—a radiolabeled substrate for the firefly luciferase catalyzed bioluminescence reaction: impact on bioluminescence based reporter gene imaging. Eur. J. Nucl. Med. Mol. Imaging 35:2275-2285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chauty, A., M. F. Ardant, A. Adeye, H. Euverte, A. Guèdèenon, C. Johnson, J. Aubry, E. Nuermberger, and J. Grosset. 2007. Promising clinical efficacy of streptomycin-rifampin combination for treatment of buruli ulcer (Mycobacterium ulcerans disease). Antimicrob. Agents Chemother. 51:4029-4035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Davis, S. L., N. A. Be, G. Lamichhane, S. Nimmagadda, M. G. Pomper, W. R. Bishai, and S. K. Jain. 2009. Bacterial thymidine kinase as a non-invasive imaging reporter for Mycobacterium tuberculosis in live animals. PLoS One 4:e6297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dega, H., J. Robert, P. Bonnafous, V. Jarlier, and J. Grosset. 2000. Activities of several antimicrobials against Mycobacterium ulcerans infection in mice. Antimicrob. Agents Chemother. 44:2367-2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Etuaful, S., B. Carbonnelle, J. Grosset, S. Lucas, C. Horsfield, R. Phillips, M. Evans, D. Ofori-Adjei, E. Klustse, J. Owusu-Boateng, G. K. Amedofu, P. Awuah, E. Ampadu, G. Amofah, K. Asiedu, and M. Wansbrough-Jones. 2005. Efficacy of the combination rifampin-streptomycin in preventing growth of Mycobacterium ulcerans in early lesions of Buruli ulcer in humans. Antimicrob. Agents Chemother. 49:3182-3186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.George, K. M., D. Chatterjee, G. Gunawardana, D. Welty, J. Hayman, R. Lee, and P. L. Small. 1999. Mycolactone: a polyketide toxin from Mycobacterium ulcerans required for virulence. Science 283:854-857. [DOI] [PubMed] [Google Scholar]
- 9.George, K. M., L. Pascopella, D. M. Welty, and P. L. Small. 2000. A Mycobacterium ulcerans toxin, mycolactone, causes apoptosis in guinea pig ulcers and tissue culture cells. Infect. Immun. 68:877-883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Heuts, F., B. Carow, H. Wigzell, and M. E. Rottenberg. 2009. Use of non-invasive bioluminescent imaging to assess mycobacterial dissemination in mice, treatment with bactericidal drugs and protective immunity. Microbes Infect. 11:1114-1121. [DOI] [PubMed] [Google Scholar]
- 11.Ji, B., A. Chauffour, J. Robert, and V. Jarlier. 2008. Bactericidal and sterilizing activities of several orally administered combined regimens against Mycobacterium ulcerans in mice. Antimicrob. Agents Chemother. 52:1912-1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kibadi, K., M. Boelaert, A. G. Fraga, M. Kayinua, A. Longatto-Filho, J. B. Minuku, J. B. Mputu-Yamba, J. J. Muyembe-Tamfum, J. Pedrosa, J. J. Roux, W. M. Meyers, and F. Portaels. 2010. Response to treatment in a prospective cohort of patients with large ulcerated lesions suspected to be Buruli ulcer (Mycobacterium ulcerans disease). PLoS Negl. Trop. Dis. 4:e736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lefrançois, S., J. Robert., A. Chauffour, B. Ji, and V. Jarlier. 2007. Curing Mycobacterium ulcerans infection in mice with a combination of rifampin-streptomycin or rifampin-amikacin. Antimicrob. Agents Chemother. 51:645-650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nienhuis, W. A., Y. Stienstra, W. A. Thompson, P. C. Awuah, K. M. Abass, W. Tuah, N. Y Awua-Boateng, E. O. Ampadu, V. Siegmund, J. P. Schouten, O. Adjei, G. Bretzel, and T. S. van der Werf. 2010. Antimicrobial treatment for early, limited Mycobacterium ulcerans infection: a randomised controlled trial. Lancet 375:664-672. [DOI] [PubMed] [Google Scholar]
- 15.Pidot, S. J., H. Hong, T. Seemann, J. L. Porter, M. J. Yip, A. Men, M. Johnson, P. Wilson, J. K. Davies, P. F. Leadlay, and T. P. Stinear. 2008. Deciphering the genetic basis for polyketide variation among mycobacteria producing mycolactones. BMC Genomics 9:462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Phillips, R., F. S. Sarfo, L. Guenin-Macé, J. Decalf, M. Wansbrough-Jones, M. L. Albert, and C. Demangel. 2009. Immunosuppressive signature of cutaneous Mycobacterium ulcerans infection in the peripheral blood of patients with buruli ulcer disease. J. Infect. Dis. 200:1675-1684. [DOI] [PubMed] [Google Scholar]
- 17.Portaels, F., M. T. Silva, and W. M. Meyers. 2009. Buruli ulcer. Clin. Dermatol. 27:291-305. [DOI] [PubMed] [Google Scholar]
- 18.Rosseels, V., V. Roupie, D. Zinniel, R. G. Barletta, and K. Huygen. 2006. Development of luminescent Mycobacterium avium subsp. paratuberculosis for rapid screening of vaccine candidates in mice. Infect. Immun. 74:3684-3686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shepard, C. C. 1969. Further experience with the kinetic method for the study of drugs against Mycobacterium leprae in mice. Activities of DDS, DFD, ethionamide, capreomycin and PAM 1392. Int. J. Lepr. Other Mycobact. Dis. 37:389-397. [PubMed] [Google Scholar]
- 20.Snewin, V. A., M. P. Gares, P. O. Gaora, Z. Hasan, I. N. Brown, and D. B. Young. 1999. Assessment of immunity to mycobacterial infection with luciferase reporter constructs. Infect. Immun. 67:4586-4593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tyagi, S., E. Nuermberger, T. Yoshimatsu, K. Williams, I. Rosenthal, N. Lounis, W. Bishai, and J. Grosset. 2005. Bactericidal activity of the nitroimidazopyran PA-824 in a murine model of tuberculosis. Antimicrob. Agents Chemother. 49:2289-2293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.World Health Organization. 2007. Global plan to combat neglected tropical diseases, 2008-2015. http://whqlibdoc.who.int/hq/2007/WHO_CDS_NTD_2007.3_eng.pdf.
- 23.Zhang, T., W. R. Bishai, J. H. Grosset, and E. L. Nuermberger. 2010. Rapid assessment of antibacterial activity against Mycobacterium ulcerans using recombinant luminescent strains. Antimicrob. Agents Chemother. 54:2806-2813. [DOI] [PMC free article] [PubMed] [Google Scholar]