Abstract
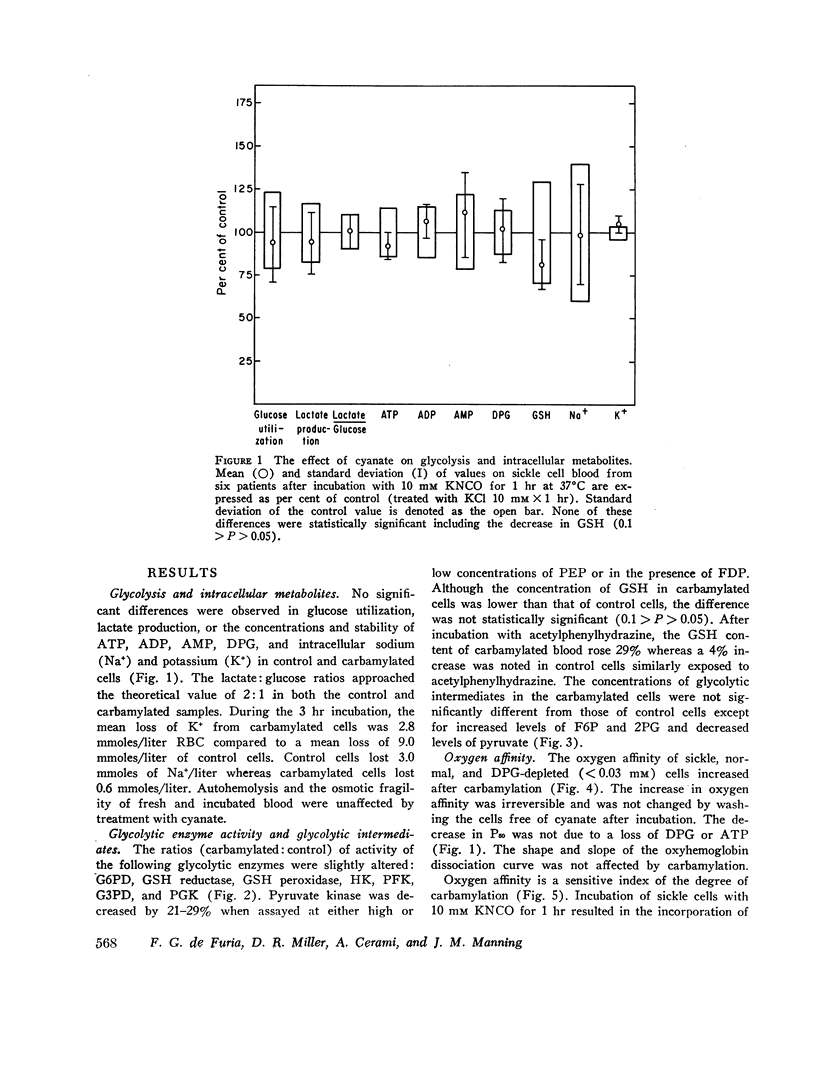
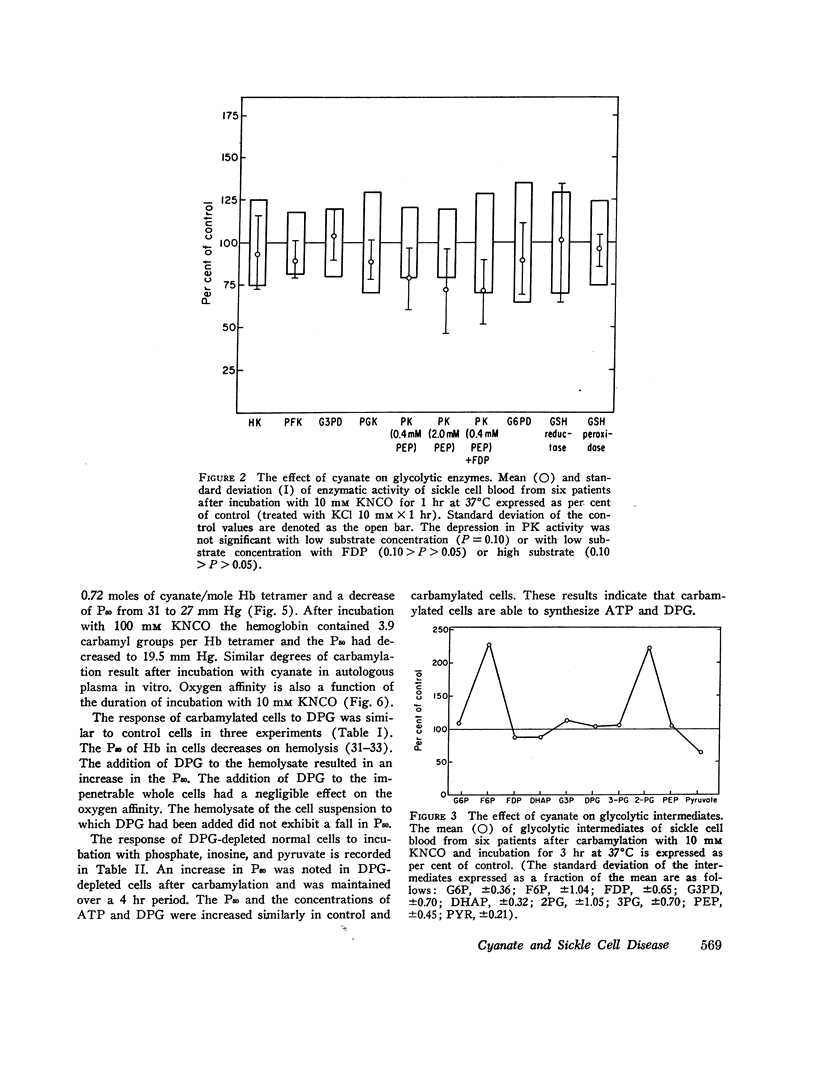
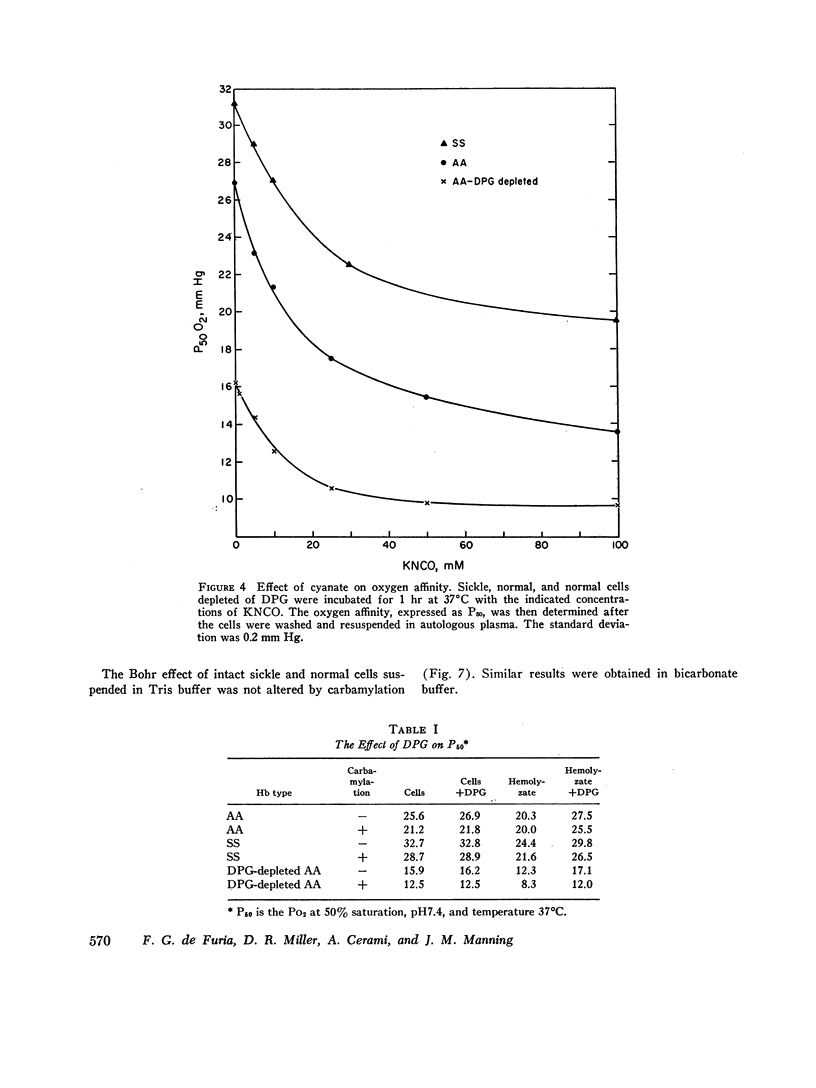
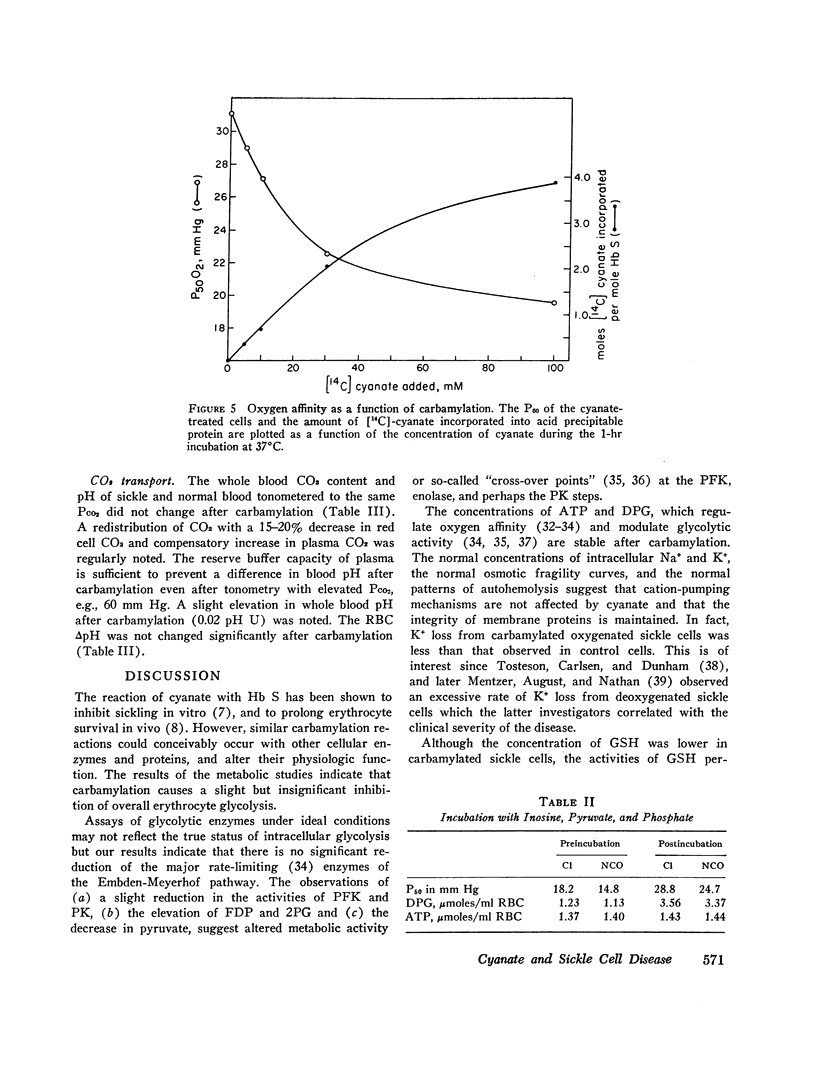
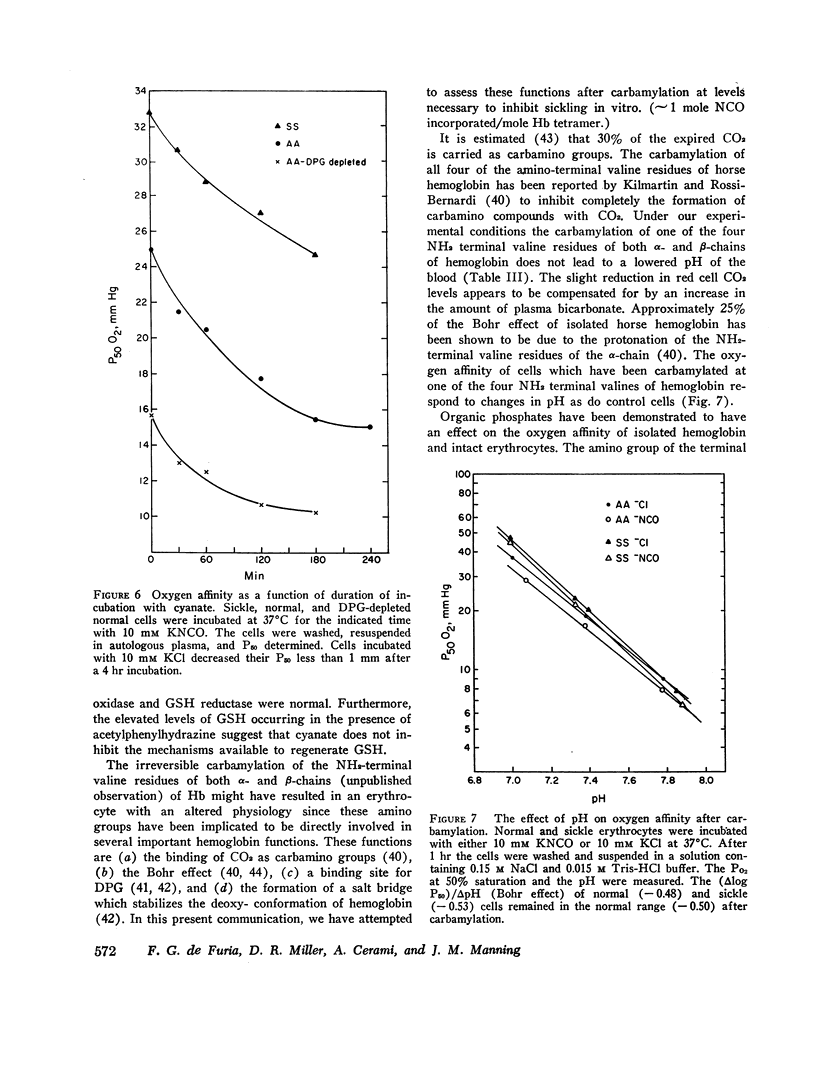
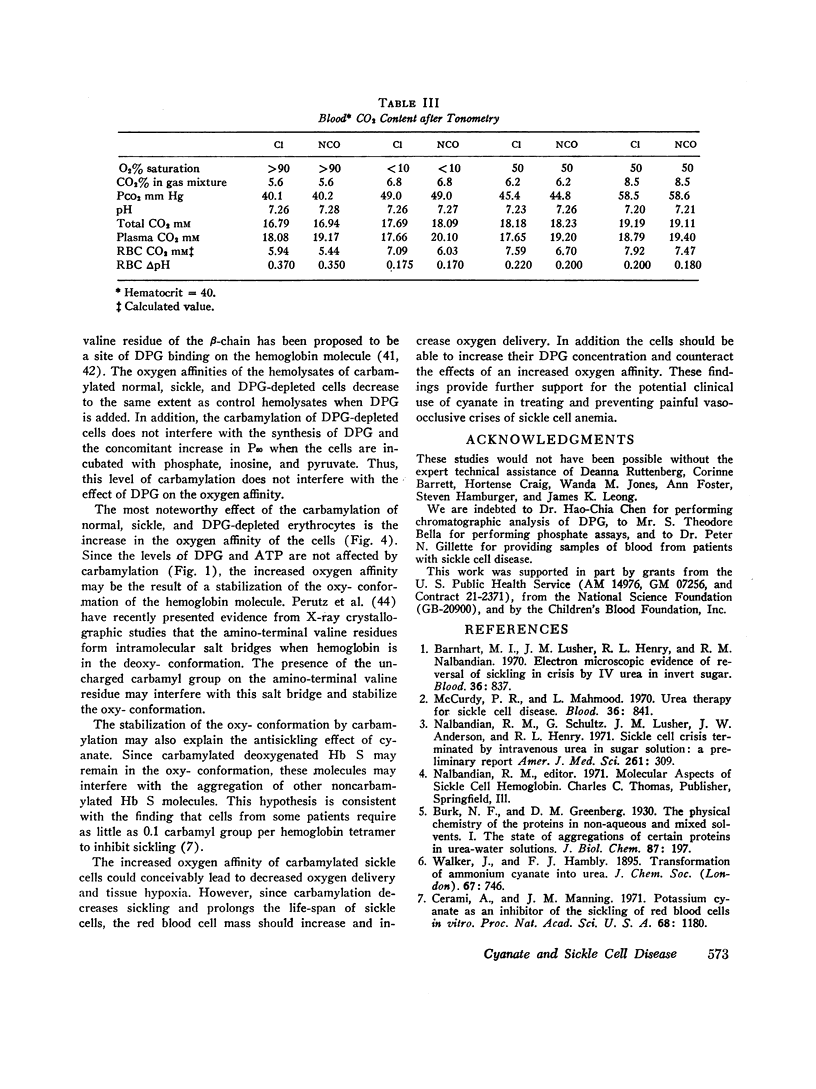
Cyanate, which is in equilibrium with urea, combines with the α-amino group of the aminoterminal valine of hemoglobin in an irreversible, specific carbamylation reaction. Partial carbamylation (0.72 residues/hemoglobin tetramer) as determined by cyanate-14C incorporation or hydantoin analysis diminishes the in vitro sickling phenomenon. Since cyanate may react not only with hemoglobin but also with functional groups of other red blood cell proteins, the in vitro effect of cyanate was studied on sickle cells. Cells were incubated with 10 mM KCl (control) or 10 mM KNCO (carbamylated) for 1 hr, washed, and resuspended in autologous plasma. Glycolysis, ATP and 2,3-diphosphoglyceric acid (DPG) stability, autohemolysis, and osmotic fragility were not affected by carbamylation. Potassium loss in carbamylated cells (2.8 mmol/liter) was less than in control cells (9.0 mmol/liter). Pyruvate kinase activity of carbamylated cells was decreased (∼25%) but the activities of other glycolytic enzymes were similar to those of control cells. Oxygen affinity of carbamylated sickle, normal, and DPG-depleted normal cells increased, and was a sensitive index of the degree and duration of reaction with cyanate. The reactivity of carbamylated cells to DPG was similar to control cells. DPG-depleted carbamylated cells regenerated DPG and increased the P50 when incubated with pyruvate, inosine, and phosphate. The Bohr effect of normal and of sickle cells was not affected (Δlog P50/Δ pH=-0.48 and -0.53, respectively) after carbamylation. The reserve buffering capacity of plasma offset the slightly diminished (∼15%) CO2 capacity of carbamylated cells so that whole blood CO2 capacity, pH, and PCO2 were normal. These studies provide further support for the potential clinical use of cyanate in treating and preventing the anemia and painful crises of sickle cell disease.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALTMAN K. I., IZZO M. J., SWISHER S. N., YOUNG L. E. Studies on spontaneous in vitro autohemolysis in hemolytic disorders. Blood. 1956 Nov;11(11):977–997. [PubMed] [Google Scholar]
- BATTAGLIA F. C., BEHRMAN R. E., HELLEGERS A. E., BATTAGLIA J. D. INTRACELLULAR HYDROGEN ION CONCENTRATION CHANGES DURING ACUTE RESPIRATORY ACIDOSIS AND ALKALOSIS. J Pediatr. 1965 Apr;66:737–742. doi: 10.1016/s0022-3476(65)80008-7. [DOI] [PubMed] [Google Scholar]
- BEUTLER E., BALUDA M. C. METHEMOGLOBIN REDUCTION. STUDIES OF THE INTERACTION BETWEEN CELL POPULATIONS AND OF THE ROLE OF METHYLENE BLUE. Blood. 1963 Sep;22:323–333. [PubMed] [Google Scholar]
- BEUTLER E., DURON O., KELLY B. M. Improved method for the determination of blood glutathione. J Lab Clin Med. 1963 May;61:882–888. [PubMed] [Google Scholar]
- Benesch R., Benesch R. E., Yu C. I. Reciprocal binding of oxygen and diphosphoglycerate by human hemoglobin. Proc Natl Acad Sci U S A. 1968 Feb;59(2):526–532. doi: 10.1073/pnas.59.2.526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bocchini V., Alioto M. R., Najjar V. A. The activation of phosphoglucomutase by denaturing agents, urea, guanidine hydrochloride, and heat. Biochemistry. 1967 Oct;6(10):3242–3249. doi: 10.1021/bi00862a033. [DOI] [PubMed] [Google Scholar]
- Brewer G. J., Eaton J. W. Erythrocyte metabolism: interaction with oxygen transport. Science. 1971 Mar 26;171(3977):1205–1211. doi: 10.1126/science.171.3977.1205. [DOI] [PubMed] [Google Scholar]
- Bunn H. F., Briehl R. W. The interaction of 2,3-diphosphoglycerate with various human hemoglobins. J Clin Invest. 1970 Jun;49(6):1088–1095. doi: 10.1172/JCI106324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carey M. J., Butterworth P. J. The action of cyanate on human and pig kidney alkaline phosphatases. Biochem J. 1969 Mar;111(5):745–748. doi: 10.1042/bj1110745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerami A., Manning J. M. Potassium cyanate as an inhibitor of the sickling of erythrocytes in vitro. Proc Natl Acad Sci U S A. 1971 Jun;68(6):1180–1183. doi: 10.1073/pnas.68.6.1180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chanutin A., Curnish R. R. Effect of organic and inorganic phosphates on the oxygen equilibrium of human erythrocytes. Arch Biochem Biophys. 1967 Jul;121(1):96–102. doi: 10.1016/0003-9861(67)90013-6. [DOI] [PubMed] [Google Scholar]
- DULBECCO R., VOGT M. Plaque formation and isolation of pure lines with poliomyelitis viruses. J Exp Med. 1954 Feb;99(2):167–182. doi: 10.1084/jem.99.2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards M. J., Martin R. J. Mixing technique for the oxygen-hemoglobin equilibrium and Bohr effect. J Appl Physiol. 1966 Nov;21(6):1898–1902. doi: 10.1152/jappl.1966.21.6.1898. [DOI] [PubMed] [Google Scholar]
- GOMEZ D. M. Considerations of oxygen-hemoglobin equilibrium in the physiological state. Am J Physiol. 1961 Jan;200:135–142. doi: 10.1152/ajplegacy.1961.200.1.135. [DOI] [PubMed] [Google Scholar]
- Gillette P. N., Manning J. M., Cerami A. Increased survival of sickle-cell erythrocytes after treatment in vitro with sodium cyanate. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2791–2793. doi: 10.1073/pnas.68.11.2791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HILPERT P., FLEISCHMANN R. G., KEMPE D., BARTELS H. THE BOHR EFFECT RELATED TO BLOOD AND ERYTHROCYTE PH. Am J Physiol. 1963 Aug;205:337–340. doi: 10.1152/ajplegacy.1963.205.2.337. [DOI] [PubMed] [Google Scholar]
- HOLADAY D. A., VEROSKY M. Improved micromanometric methods for the analysis of respiratory gases in plasma and whole blood. J Lab Clin Med. 1956 Apr;47(4):634–644. [PubMed] [Google Scholar]
- Kilmartin J. V., Rossi-Bernardi L. Inhibition of CO2 combination and reduction of the Bohr effect in haemoglobin chemically modified at its alpha-amino groups. Nature. 1969 Jun 28;222(5200):1243–1246. doi: 10.1038/2221243a0. [DOI] [PubMed] [Google Scholar]
- Lichtman M. A., Miller D. R. Erythrocyte glycolysis, 2,3-diphosphoglycerate and adenosine triphosphate concentration in uremic subjects: relationship to extracellular phosphate concentration. J Lab Clin Med. 1970 Aug;76(2):267–279. [PubMed] [Google Scholar]
- Minakami S., Suzuki C., Saito T., Yoshikawa H. Studies on erythrocyte glycolysis. I. Determination of the glycolytic intermediates in human erythrocytes. J Biochem. 1965 Dec;58(6):543–550. doi: 10.1093/oxfordjournals.jbchem.a128240. [DOI] [PubMed] [Google Scholar]
- Minakami S., Yoshikawa H. Studies on erythrocyte glycolysis. II. Free energy changes and rate limitings steps in erythrocyte glycolysis. J Biochem. 1966 Feb;59(2):139–144. doi: 10.1093/oxfordjournals.jbchem.a128274. [DOI] [PubMed] [Google Scholar]
- Munro G. F., Miller D. R. Mechanism of fructose diphosphate activation of a mutant pyruvate kinase from human red cells. Biochim Biophys Acta. 1970 Apr 22;206(1):87–97. doi: 10.1016/0005-2744(70)90085-9. [DOI] [PubMed] [Google Scholar]
- Nalbandian R. M., Shultz G., Lusher J. M., Anderson J. W., Henry R. L. Sickle cell crisis terminated by intravenous urea in sugar solutions--a preliminary report. Am J Med Sci. 1971 Jun;261(6):309–324. doi: 10.1097/00000441-197106000-00002. [DOI] [PubMed] [Google Scholar]
- Paglia D. E., Valentine W. N., Baughan M. A., Miller D. R., Reed C. F., McIntyre O. R. An inherited molecular lesion of erythrocyte pyruvate kinase. Identification of a kinetically aberrant isozyme associated with premature hemolysis. J Clin Invest. 1968 Aug;47(8):1929–1946. doi: 10.1172/JCI105883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perutz M. F., Muirhead H., Mazzarella L., Crowther R. A., Greer J., Kilmartin J. V. Identification of residues responsible for the alkaline Bohr effect in haemoglobin. Nature. 1969 Jun 28;222(5200):1240–1243. doi: 10.1038/2221240a0. [DOI] [PubMed] [Google Scholar]
- Perutz M. F. Stereochemistry of cooperative effects in haemoglobin. Nature. 1970 Nov 21;228(5273):726–739. doi: 10.1038/228726a0. [DOI] [PubMed] [Google Scholar]
- Rose Z. B., Liebowitz J. Direct determination of 2,3-diphosphoglycerate. Anal Biochem. 1970 May;35(1):177–180. doi: 10.1016/0003-2697(70)90023-0. [DOI] [PubMed] [Google Scholar]
- Rossi-Bernardi L., Roughton F. J. The specific influence of carbon dioxide and carbamate compounds on the buffer power and Bohr effects in human haemoglobin solutions. J Physiol. 1967 Mar;189(1):1–29. doi: 10.1113/jphysiol.1967.sp008152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SEVERINGHAUS J. W. Oxyhemoglobin dissociation curve correction for temperature and pH variation in human blood. J Appl Physiol. 1958 May;12(3):485–486. doi: 10.1152/jappl.1958.12.3.485. [DOI] [PubMed] [Google Scholar]
- TOSTESON D. C., CARLSEN E., DUNHAM E. T. The effects of sickling on ion transport. I. Effect of sickling on potassium transport. J Gen Physiol. 1955 Sep 20;39(1):31–53. doi: 10.1085/jgp.39.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weed R. I., Bowdler A. J. Metabolic dependence of the critical hemolytic volume of human erythrocytes: relationship to osmotic fragility and autohemolysis in hereditary spherocytosis and normal red cells. J Clin Invest. 1966 Jul;45(7):1137–1149. doi: 10.1172/JCI105420. [DOI] [PMC free article] [PubMed] [Google Scholar]



