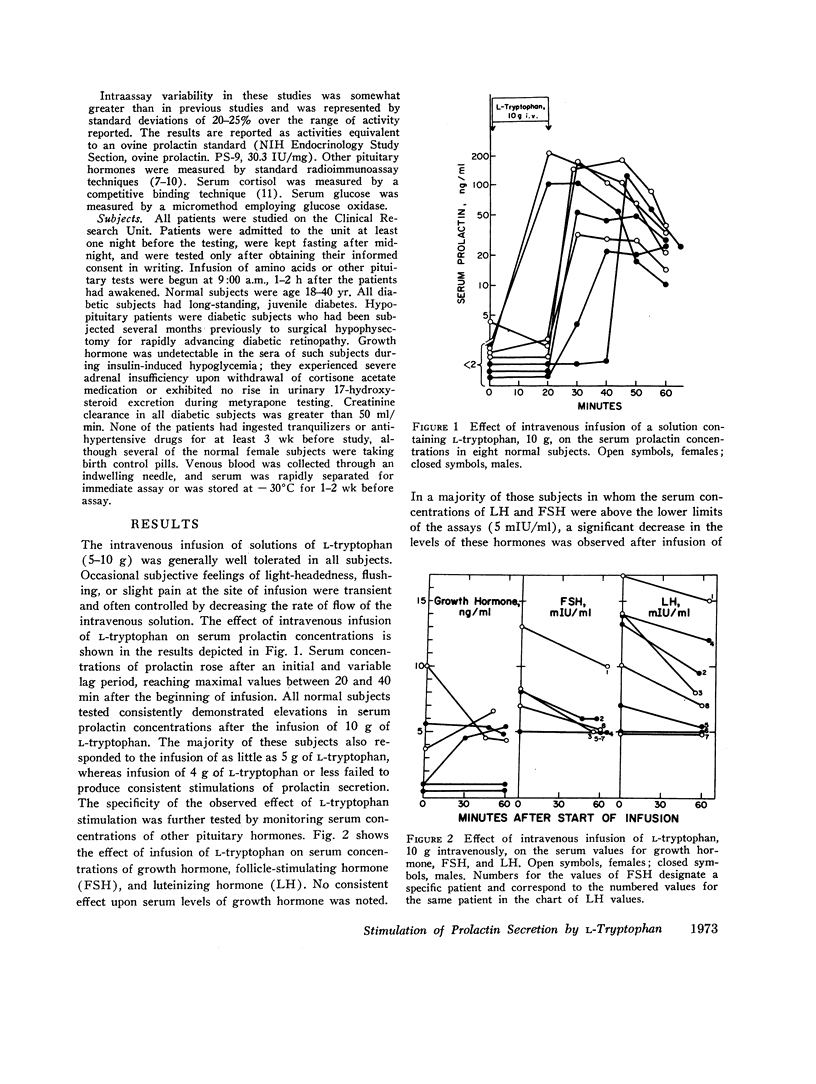
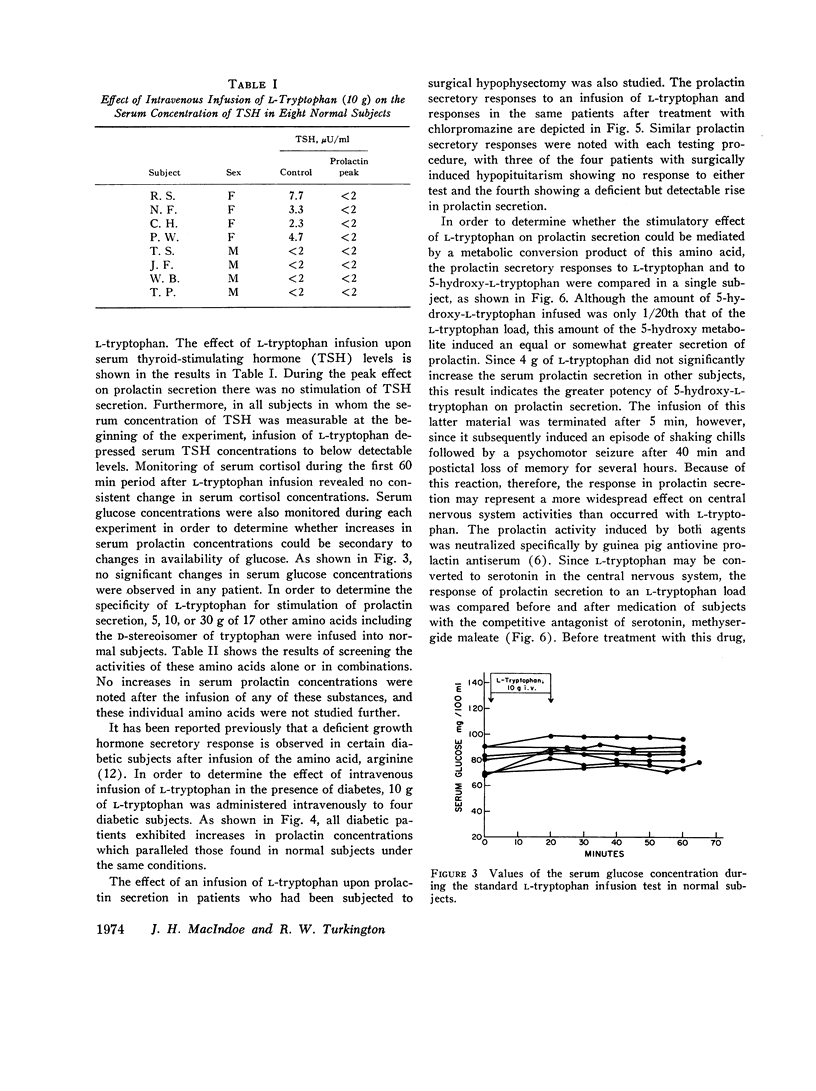
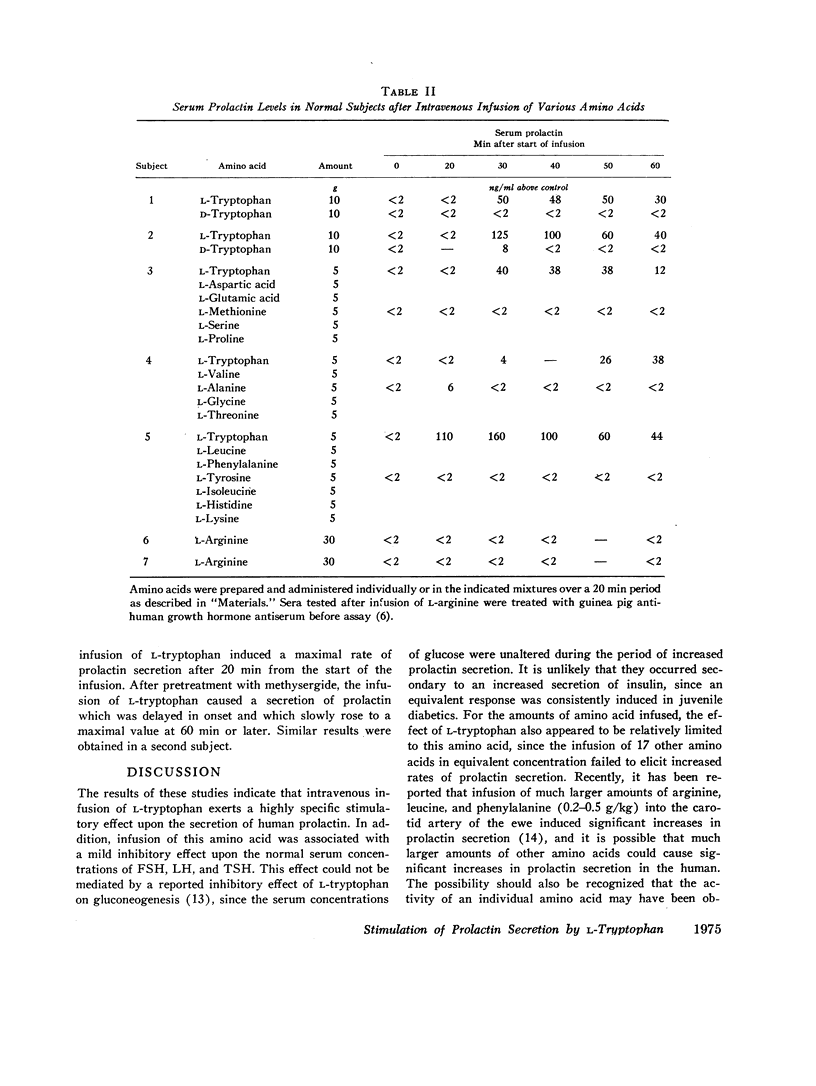
Abstract
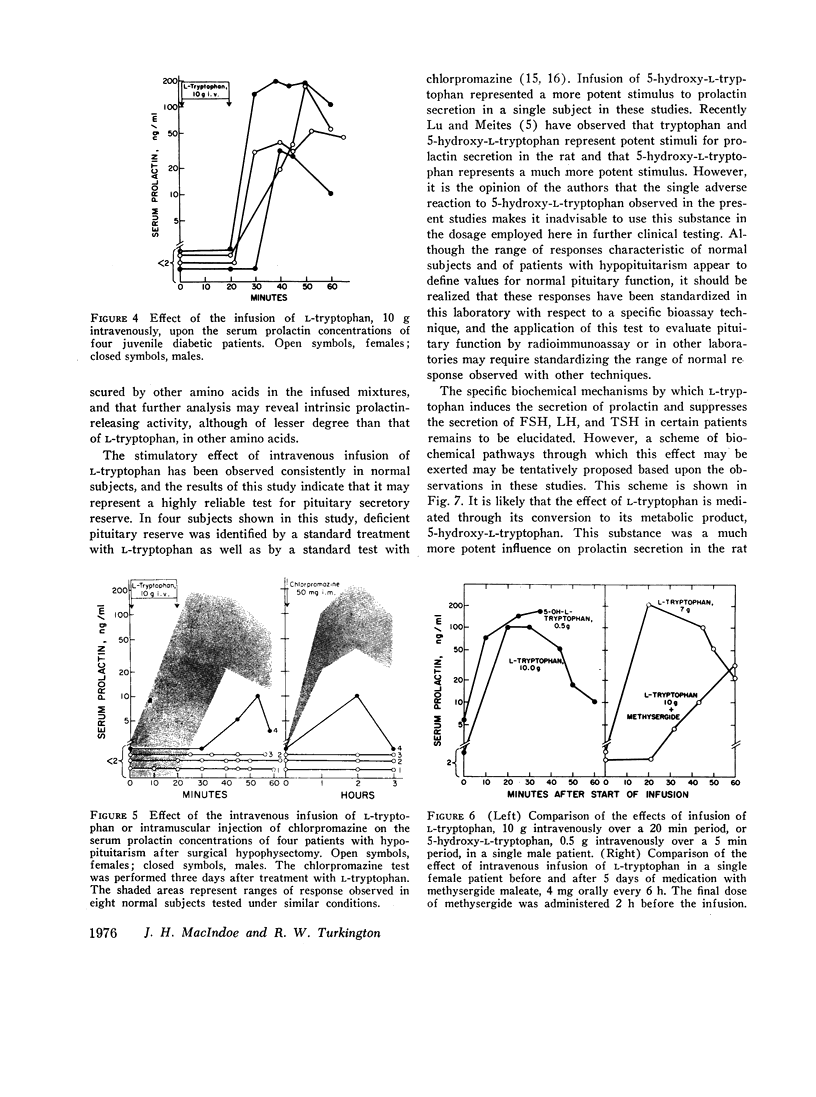
Previous studies have demonstrated that the secretion of human prolactin is regulated primarily by factors that influence catecholamines of the hypothalamus. In an effort to identify other factors that may regulate prolactin secretion, the amino acid L-tryptophan, a precursor in the synthesis of serotonin, was infused into normal human volunteers. Intravenous infusion of L-tryptophan, 5-10 g over a 20 min period, but not equivalent amounts of 17 other amino acids, induced marked increases in serum prolactin concentrations in eight normal human volunteers. Increases of 20-200 ng/ml above the control level were observed with peak values at 20-45 min after initiation of the infusion. In addition, infusion of L-tryptophan was associated with decreases in serum concentrations of follicle-stimulating hormone (FSH), luteinizing hormone (LH), and thyrotropin in those subjects in whom the base-line serum hormone concentration was above the lower limits of assay detectability. No consistent change was observed in serum concentrations of growth hormone, cortisol, or glucose. Four subjects with juvenile diabetes demonstrated increases in serum prolactin values comparable with those observed in healthy individuals in response to infusions of L-tryptophan. Serum prolactin values in patients with surgically induced hypopituitarism were undetectable or deficient after infusion of 10 g of L-tryptophan. In this respect, infusion of L-tryptophan was equally effective in these subjects as the standard chlorpromazine stimulation test in identifying patients with hypopituitarism, indicating that the infusion of L-tryptophan may serve as a sensitive and reliable clinical test of prolactin secretory reserve. Further studies relating to the possible mechanism of action of L-tryptophan indicated that infusion of 5-hydroxytryptophan represents a much more potent stimulus for the secretion of prolactin and that premedication with the serotonin antagonist, methysergide maleate, serves to blunt the effect of L-tryptophan on prolactin secretion. These results support the concept that the effect of L-tryptophan on the secretion of human prolactin is mediated through its conversion to serotonin and are consistent with reported experimental observations that serotonin may participate in the reciprocal regulation of prolactin and gonadotropins.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amoss M., Burgus R., Blackwell R., Vale W., Fellows R., Guillemin R. Purification, amino acid composition and N-terminus of the hypothalamic luteinizing hormone releasing factor (LRF) of ovine origin. Biochem Biophys Res Commun. 1971 Jul 2;44(1):205–210. doi: 10.1016/s0006-291x(71)80179-1. [DOI] [PubMed] [Google Scholar]
- Bowers C. Y., Friesen H. G., Hwang P., Guyda H. J., Folkers K. Prolactin and thyrotropin release in man by synthetic pyroglutamyl-histidyl-prolinamide. Biochem Biophys Res Commun. 1971 Nov;45(4):1033–1041. doi: 10.1016/0006-291x(71)90441-4. [DOI] [PubMed] [Google Scholar]
- Cotton G. E., Gorman C. A., Mayberry W. E. Suppression of thyrotropin (h-TSH) in serums of patients with myxedema of varying etiology treated with thyroid hormones. N Engl J Med. 1971 Sep 2;285(10):529–533. doi: 10.1056/NEJM197109022851001. [DOI] [PubMed] [Google Scholar]
- Currie B. L., Johansson N. G., Folkers K. On the chemical existence and partial purification of the hypothalamic follicle stimulating hormone releasing hormone. Biochem Biophys Res Commun. 1973 Jan 4;50(1):14–19. doi: 10.1016/0006-291x(73)91056-5. [DOI] [PubMed] [Google Scholar]
- Davis S. L. Plasma levels of prolactin, growth hormone and insulin in sheep following the infusion of arginine, leucine and phenylalanine. Endocrinology. 1972 Aug;91(2):549–555. doi: 10.1210/endo-91-2-549. [DOI] [PubMed] [Google Scholar]
- Fernstrom J. D., Wurtman R. J. Brain serotonin content: physiological dependence on plasma tryptophan levels. Science. 1971 Jul 9;173(3992):149–152. doi: 10.1126/science.173.3992.149. [DOI] [PubMed] [Google Scholar]
- Fernstrom J. D., Wurtman R. J. Brain serotonin content: physiological regulation by plasma neutral amino acids. Science. 1972 Oct 27;178(4059):414–416. doi: 10.1126/science.178.4059.414. [DOI] [PubMed] [Google Scholar]
- Jacobs L. S., Snyder P. J., Wilber J. F., Utiger R. D., Daughaday W. H. Increased serum prolactin after administration of synthetic thyrotropin releasing hormone (TRH) in man. J Clin Endocrinol Metab. 1971 Dec;33(6):996–998. doi: 10.1210/jcem-33-6-996. [DOI] [PubMed] [Google Scholar]
- Kamberi I. A., Mical R. S., Porter J. C. Effects of melatonin and serotonin on the release of FSH and prolactin. Endocrinology. 1971 Jun;88(6):1288–1293. doi: 10.1210/endo-88-6-1288. [DOI] [PubMed] [Google Scholar]
- Lardy H. A. The role of tryptophan metabolites in regulating gluconeogenesis. Am J Clin Nutr. 1971 Jul;24(7):764–765. doi: 10.1093/ajcn/24.7.764. [DOI] [PubMed] [Google Scholar]
- Lovenberg W., Jequier E., Sjoerdsma A. Tryptophan hydroxylation in mammalian systems. Adv Pharmacol. 1968;6(Pt A):21–36. doi: 10.1016/s1054-3589(08)61153-9. [DOI] [PubMed] [Google Scholar]
- Malarkey W. B., Jacobs L. S., Daughaday W. H. Levodopa suppression of prolactin in nonpuerperal galactorrhea. N Engl J Med. 1971 Nov 18;285(21):1160–1163. doi: 10.1056/NEJM197111182852102. [DOI] [PubMed] [Google Scholar]
- Merimee T. J., Burgess J. A., Rabinowitz D. Arginine infusion in maturity-onset diabetes mellitus. Defective output of insulin and of growth hormone. Lancet. 1966 Jun 11;1(7450):1300–1301. doi: 10.1016/s0140-6736(66)91204-9. [DOI] [PubMed] [Google Scholar]
- Murphy B. E. Some studies of the protein-binding of steroids and their application to the routine micro and ultramicro measurement of various steroids in body fluids by competitive protein-binding radioassay. J Clin Endocrinol Metab. 1967 Jul;27(7):973–990. doi: 10.1210/jcem-27-7-973. [DOI] [PubMed] [Google Scholar]
- Odell W. D., Parlow A. F., Cargille C. M., Ross G. T. Radioimmunoassay for human follicle-stimulating hormone: physiological studies. J Clin Invest. 1968 Dec;47(12):2551–2562. doi: 10.1172/JCI105937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCHALCH D. S., PARKER M. L. A SENSITIVE DOUBLE ANTIBODY IMMUNOASSAY FOR HUMAN GROWTH HORMONE IN PLASMA. Nature. 1964 Sep 12;203:1141–1142. doi: 10.1038/2031141a0. [DOI] [PubMed] [Google Scholar]
- Schalch D. S., Parlow A. F., Boon R. C., Reichlin S. Measurement of human luteinizing hormone in plasma by radioimmunoassay. J Clin Invest. 1968 Mar;47(3):665–678. doi: 10.1172/JCI105762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turkington R. W. Measurement of prolactin activity in human serum by the induction of specific milk proteins in mammary gland in vitro. J Clin Endocrinol Metab. 1971 Aug;33(2):210–216. doi: 10.1210/jcem-33-2-210. [DOI] [PubMed] [Google Scholar]
- Turkington R. W., Underwood L. E., Van Wyk J. J. Elevated serum prolactin levels after pituitary-stalk section in man. N Engl J Med. 1971 Sep 23;285(13):707–710. doi: 10.1056/NEJM197109232851302. [DOI] [PubMed] [Google Scholar]