Abstract
Purpose
Oxidative damage induced by H2O2 treatment can irreversibly damage the lens epithelium, resulting in cell death and cataract. Grape seed extract (GSE) is a widely consumed dietary supplement that has the capability to scavenge oxidants and free radicals. GSE contain 70%–95% standardized proanthocyanidins. The study described herein investigated the protective effect of Grape seed proanthocyanidin extract (GSPE) on H2O2-induced oxidative stress in human lens epithelial B-3 (HLEB-3) cells and the possible molecular mechanism involved.
Methods
HLE-B3 cells exposed to different doses of H2O2 were cultured with various concentrations of GSPE and subsequently monitored for cell viability by the 4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT) assay. The apoptosis rate and ROS generation were detected by flow cytometric analysis. Expression of NF-кB/P65 and mitogen activated protein kinase (MAPK) proteins were measured by western blot.
Results
GSPE clearly reduced H2O2 induced cell apoptosis and reactive oxygen species (ROS) generation and protected HLEB-3 cells from H2O2 induced oxidative damage. GSPE depressed H2O2-induced activation and translocation of NF-кB/p65. GSPE also depressed H2O2-induced phosphorylation of the p38 and c-Jun N-terminal kinase (JNK) proteins of the MAPK family at various time points studied.
Conclusions
GSPE could be useful in attenuation of H2O2-induced oxidative stress and the activation of NF-кB and MAPK signaling in HLE-B3 cells, which suggests that GSPE has a potential protective effect against cataractogenesis.
Introduction
Cataract remains the leading cause of visual disability and blindness worldwide. At present, the only remedy is surgical removal of the cataractous lens and substituting it with a lens made of synthetic polymers. However, the incidence is so large that the available surgical facilities are unable to cope up with the problem. In addition to these, postoperative complications can occur such as posterior capsular opacification, endophthalmitis, and uncorrected residual refractive error. Therefore, it is so necessary to search for pharmacological intervention that will maintain the transparency of the lens.
Loss of transparency during human cataract formation results from a variety of complex metabolic and physiologic mechanisms. In the cells, reactive oxygen species (ROS) may initiate a surge of toxic biochemical reactions such as peroxidation of membrane lipids and extensive damage to proteins causing intracellular protein aggregation and precipitation and eventually leading to lens opacification [1,2]. Several studies have shown that exposure of lens epithelial cells to H2O2 increases ROS production and oxidative stress [3,4].
Proanthocyanidins are natural compounds found in high concentrations in fruits, vegetables, wine, tea, nuts, and seeds [5,6]. They are a class of phenolic compounds that take the form of oligomers or polymers of polyhydroxy flavan-3-ol units, such as (+)-catechin and (−)-epicatechin. The seeds of the grape are particularly rich source of proanthocyanidins. The grape seed proanthocyanidins extract (GSPE) are mainly dimers, trimers, and highly polymerized oligomers of monomeric catechins [7,8]. Proanthocyanidins possess a wide array of pharmacological and biochemical actions including anti-inflammatory, anti-carcinogenic activity, and cardioprotective biologic effects [5,6,9,10]. In addition to these, GSPE have been shown to be potent antioxidants and free radical scavengers, being more effective than either ascorbic acid or vitamin E [5,9]. In vivo experiments, it has been reported that GSPE can prevent cataract formation in rats predisposed to hereditary cataracts [11]. Besides, some experiments have given evidences that GSPE can prevent selenite cataract development in rats [12,13]. GSPE is marketed as a dietary supplement in the United States, due to their powerful antioxidant activity, low toxicity and no genotoxic potential [14]. However, until now, there is no study about the effect of GSPE on H2O2-induced oxidative stress and the precise mechanism of signal transduction in human lens epithelial (HLE) cells.
Dietary modulation of the mitogen-activated protein kinases (MAP K) pathway and nuclear factor kappa-B (NF-кB) has emerged as a potential target of dietary antioxidants. Members of the two pathways, as well as protein38 (p38), c-jun N-terminal kinase (JNK), and NF-кB/protein65 (p65), are involved in the regulation of cellular differentiation, migration, proliferation and survival [15,16].
Based on these recent studies, we hypothesized that GSPE would protect HLE cells from oxidative stress by influencing several signaling pathways and thus would be beneficial in the treatment of cataract. The study was designed to determine whether GSPE could reduce H2O2-induced cell apoptosis and cell death in cultured HLEB-3 cells. We also detected if GSPE can scavenge ROS accumulation. At last, we investigated the mechanism of GSPE in protecting the HLEB-3 cells from oxidative damages.
Methods
Materials
GSPE, constituting of 95% (wt/wt) proanthocyanidins, was provided by JF-NATURAL Corp. (Tian Jin, China) and was dissolved in deionized water. The same lot of GSPE was used for all experiments. HLE-B3 cells, a human lens epithelial cell line immortalized by SV-40 viral transformation [17], were purchased from the ATCC (American Type Culture Collection; Rockville, MD). Fetal bovine serum (FBS) and Dulbecco’s modified Eagle’s medium (DMEM) were obtained from Gibco (Grand Island, NY). Anti-JNK, anti-phosphorylation of JNK (p-JNK), anti-p38 and anti-phosphorylation of p38 (p-p38) were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA).
Cell culture
HLE-B3 cells were maintained at 37 °C in DMEM supplemented with 20% FBS and 1% antimicrobial solution (Sigma Chemical Co., St. Louis, MO) in an environment composed of 5% CO2/95% O2. When grown to 75%–80% confluence, the cells were treated with the indicated concentration of H2O2 for the required time or pretreated with GSPE for different time before the H2O2 treatment. At the indicated time points, the cells were collected for different assays.
Measurement of cell viability
The 4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT) assay was used to verify the viability of HLEB-3 cells. Cells were plated at a density of 1×104 cells/well in 96-well microplates. After a 24 h incubation, cells were pretreated with 2.5, 5.0, 10.0, and 20.0 mg/l GSPE for 12 h, 24 h, and 48 h. After incubation for the indicated time, cells were treated with 50, 100, and 200 μmol/l H2O2 in combination for 24 h. Cells were incubated with 20 μl of MTT solution (0.5 mg/ml) for 4 h at 37 °C, and the solution was then replaced with 200 μl DMSO after the incubation. The absorbance was measured at 490 nm by a microplate reader (ELx800; BioTek, Winooski, VT).
Apoptosis assay
For the quantification of apoptotic death, Cells were grown on a six-well plate at 1×105 cells per plate and pretreated with or without different concentration of GSPE for 12 h before 100 μmol/l H2O2 treatment for 24 h. Thereafter, cells were collected and stained with Annexin V and PI using Vybrant Apoptosis Assay Kit 2 (Molecular Probes, Eugene, OR) essentially following the instructions of the manufacturer. Stained cells were analyzed using Cell-Quest software.
ROS detection
The production of ROS was monitored using flow cytometry. ROS production was quantified by the 2',7'-dichlorofluorescin-diacetate (DCFH-DA) method based on the ROS-dependent oxidation of DCFH-DA to DCF. Cells were incubated with 10.0 μM DCFH-DA (Molecular Probes, Eugene, OR) for 30 min. They were then washed and incubated with complete medium for 2 h. ROS generation was determined using FAC Scan flow cytometry and results were analyzed using Cell-Quest software (Becton, Dickinson and Company, Franklin Lakes, NJ).
Western blotting
After treatments, cultured cells were washed with cold PBS and then lysed in a buffer containing 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1 mM Na2EDTA, 1 mM EDTA, 1% Triton-X100, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 mM NaF, 1 μg/ml leupeptin, and 1 mM phenylmethanesulfonyl fluoride (PMSF). Nuclear extracts were collected according to the instruction of the nuclear extract kit (Active Motif, Carlsbad, CA). Cell lysates were collected and their protein concentration was evaluated using a protein assay (Bio-Rad, Melville, NY). After boiling at 100 °C for 5 min, the proteins were resolved by electrophoresis on 10%–15% SDS-polyacrylamide gels (20 μg proteins per lane) and transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, Milford, MA). Membranes was incubated in 5% fat-free milk powder in Tris-buffered saline solution–Tween-20 (TBST) for 1 h at room temperature and then exposed to the primary antibodies at 4 °C overnight. After rinsing in TBST (3 times for 5 min each time), the membranes were incubated with HRP-conjugated secondary antibodies for 1 h at room temperature. Membranes were then detected by enhanced chemiluminescence (ECL) western detection reagents (Amersham, Pittsburgh, PA).
Statistical analysis
All experiments were performed at least three times and the results presented are from representative experiments. Data were compared by one-way ANOVA. For all experiments, data were reported as mean±SD as indicated, and p values <0.05 were considered significant.
Results
GSPE increases the viability of HLEB-3 cells treated by H2O2
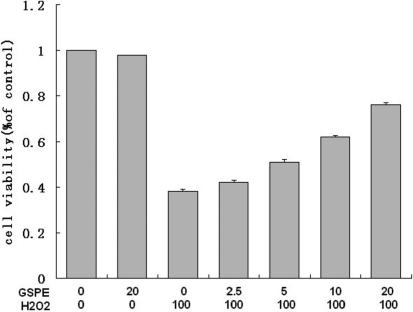
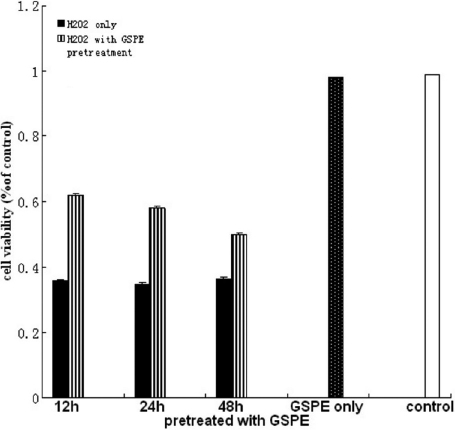
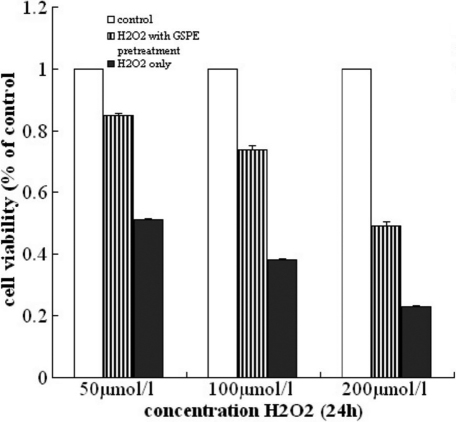
First we studied the effect of GSPE on the viability of HLEB-3 cells treated by H2O2. After HLEB-3 cells were treated with GSPE at the concentrations ranging from 0 to 20 mg/l for 12 h, these HLEB-3 cells were incubated with 100 μmol/l H2O2 for 24 h. The results showed a concentration- dependent increase in the viability of cells measured in terms of absorbance of color formed by reduction of MTT dye by live cells (Figure 1).Thus 20 mg/l was determined as the optimal dose for GSPE pretreatment. Then we investigated whether the protective effect of GSPE vary with incubation time of GSPE. Increase in treatment times to 24 and 48 h reduced the viability. Figure 2 showed that pretreatment with GSPE for 12 h was the optimal time compared with others. Finally, we selected a 12 h pretreatment with 20 mg/l GSPE before exposure to different concentrations of H2O2 and found that the protecting effect of GSPE was inverse ratio to H2O2 concentration, as seen in Figure 3. From these results, it could be concluded that GSPE had a protective role against H2O2 induced cell damage and the optimal incubation time for GSPE pretreatment is 12 h.
Figure 1.
GAPE increased the viability of cells in concentration-dependent manner. The cell viability of HLEB-3 cells pretreated with GSPE at different concentrations (0, 2.5, 5.0, 10.0, and 20.0 mg/l) for 12 h before H2O2 (100 μmol/l) treatment for 24 h was estimated by using MTT. The results showed a concentration- dependent increase of GSPE in the viability of cells.
Figure 2.
The pretreatment with GSPE for 12 h was the optimal time. The cells were incubated with 20.0 mg/l GSPE for different time (12, 24, 48 h), then treated with 100 μmol/l H2O2 for 24 h. The cell viability was measured by MTT assay. The results showed that pretreatment with GSPE for 12 h was the optimal time.
Figure 3.
The protecting effect of GSPE was inverse ratio to H2O2 concentration. The cells were pretreated with 20.0 mg/l GSPE for 12 h, then exposed to different concentration of H2O2 (50, 100, and 200 μmol/l) for 24 h. The cell viability was measured by MTT assay. The results showed the protecting effect of GSPE was inverse ratio to H2O2 concentration.
GSPE reduced apoptotic death in HLEB-3 cells treated by H2O2
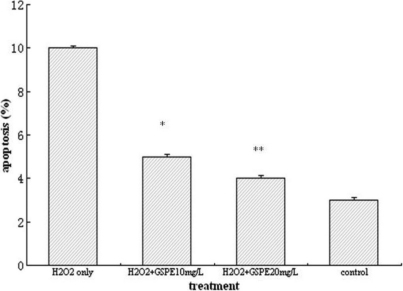
Since we observed that GSPE treatment causes significant protection in HLEB-3 cells, we next examined if the protective effects induced by GSPE involve reduction of apoptosis. Based on the results from MTT assay, the HLEB-3 cells were incubated with 10 mg/l and 20 mg/l GSPE for 12 h then treated with 100 μmol/l H2O2 for 24 h. Flow cytometric analysis of annexin V-PI stained cells revealed that indeed GSPE clearly decreased H2O2 induced apoptosis. As shown in Figure 4 the results were displayed as histograms.
Figure 4.
GSPE significantly decreased the apoptosis rate of HLEB-3 cells. The HLEB-3 cells were incubated with 10 mg/l and 20 mg/l GSPE for 12 h then treated with 100 μmol/l H2O2 for 24 h. Flow cytometric analysis of annexin V-PI stained cells showed GSPE significantly decreased the apoptosis rate of HLEB-3 cells. **p<0.01; *p<0.05.
GSPE reduces the generation of ROS induced by H2O2 in HLEB-3 cells
DCF-DA was used to carry out the generation of ROS. As shown in Figure 5, treatment with 100 μmol/l H2O2 significantly enhanced the generation of ROS. Pretreatment with 20 mg/l GSPE distinctly reduced the generation of ROS. Three repeated experiments showed GSPE pretreatment inhibited ROS generation induced by H2O2, which had statistical significance.
Figure 5.
Effect of GSPE on H2O2-induced generation of reactive oxygen species in HLEB-3 cells. HLEB-3 cells were pretreated with 20 mg/l GSPE for 12 h followed by treatment with 100 μmol/l H2O2. The ROS production was measured by DCF flow cytometry as described in Methods. The result showed GSPE pretreatment reduced ROS generation induced by H2O2.
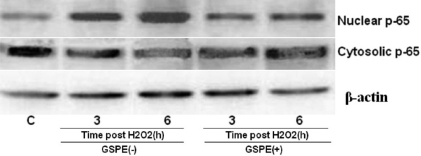
GSPE reduces H2O2-induced activation and translocation of NF-кB in HLEB-3 cells
Next, we examined protein level of transcription factor—NF-κB. HLEB-3 cells were incubated with 20 mg/l GSPE for 12 h. Cells were harvested at 3 h and 6 h time points after treated with 100 μmol/l H2O2, and cell lysates were prepared to determine the activation of NF-кB . Western blotting indicated that treatment with GSPE before H2O2 markedly abrogated H2O2-induced activation of NF-кB/p65 compared with control cells. Activation of NF-кB was based on the detection of its translocation into cell nuclei from its initial location in the cytoplasm where it exists in an inactive form. Cells treated by H2O2 exhibited an enhancement of nuclear NF-кB/p65 and a reduction of cytosolic NF-кB /p65 at 3h, and this became more evident at 6 h (Figure 6). Western blot indicated that treatment with GSPE before H2O2 markedly abrogated H2O2-induced activation of NF-кB/p65.
Figure 6.
Effect of GSPE on H2O2-induced activation of NF-кB in HLEB-3 cells. LEB-3 cells were incubated with 20 mg/l GSPE for 12 h. Cells were harvested at 3 h and 6 h time points after treated with 100 μmol/l H2O2. Western blot indicated that treatment with GSPE before H2O2 markedly decreased H2O2-induced activation of NF-кB/p65.
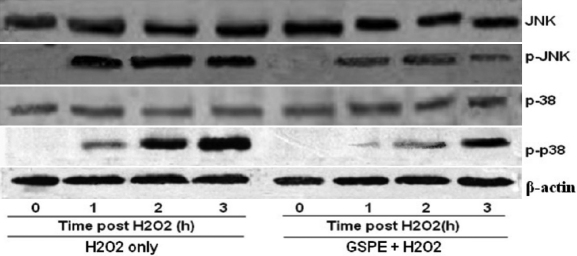
GSPE reduces H2O2-induced phosphorylation of the MAPK pathway
Whole cell extracts were prepared and analyzed using antibodies against the active phosphorylated forms of JNK and p38. As shown in Figure 7, phosphorylation of p38 and JNK started 1 h after H2O2 treatment. Western blotting and subsequent measurement of the intensity of the bands relative to the total amount of p38 and JNK phosphorylation indicated that treatment with GSPE markedly depressed H2O2 induced phosphorylation of p38 and JNK at each time point studied. Further, the total amount of JNK and p38 remained unchanged at each time point studied. These results suggested that GSPE pretreatment suppressed JNK and p38 phosphorylation.
Figure 7.
Effect of GSPE on H2O2-induced phosphorylation of the MAPK pathway. HLEB-3 cells were incubated with 20 mg/l GSPE for 12 h. Cells were harvested at 1, 2, and 3 h time points after treated with 100 μmol/l H2O2, and p38 and JNK activities were evaluated by western blotting. GSPE markedly reduced H2O2induced phosphorylation of p38 and JNK at each time point studied. The total amount of JNK and p38 remained unchanged at each time point studied.
Discussion
Cataract formation is associated with cell signaling, cell migration, and inflammation [18,19]. It has been found that GSPE attenuates these processes and finally cause slowing or inhibition of cataract formation [11-13]. Furthermore, GSPE has been found to be nontoxic in other studies [14]. Therefore, the study reported here was to evaluate the effects of GSPE on factors that contribute to cataract formation, including ROS production and cell signaling. The results suggest that GSPE is a potent inhibitor of H2O2 -induced oxidative stress and the activation of NF-кB and MAPK signaling in human lens epithelial cells. Thus it has a potential therapeutic role in the prevention of cataract. Our experiment is the first to show GSPE protective function in HLE cells and its mechanism against oxidative stress.
In our experiment, H2O2 was used as the oxidant model of classical oxidative stress. H2O2 treated cells showed an increased production of intracellular ROS and a high apoptosis rate. However GSPE pretreatment can significantly depress the ROS production in H2O2 treated HLEB-3 cells, which suggested a primary role of GSPE in protection against ROS. Moreover, flow cytometry analysis using PI and annexin V showed that H2O2-induced cell apoptosis in HLEB-3 cells were significantly reduced by GSPE pretreatment. These results indicated that GSPE can not only reduce the production of ROS, but also prevent HLEB-3 cells from apoptosis caused by H2O2.
To investigate the protective effect of GSPE against oxidative stress in HLEB-3 cells, we looked into the potential pathway involved. NF-кB is one of the most ubiquitous transcription factors. The genes regulated by NF-κB include apoptosis, cell adhesion, proliferation, inflammation, and cellular-stress response [20,21]. In unstimulated cells, NF-кB resides in the cytoplasm in an inactive complex with inhibitor kappa B. Pathogenic stimuli induce phosphorylation and the subsequent release of inhibitor kappa B, resulting in NF-кB translocation to the nucleus where it binds to DNA control elements and thus influences the transcription of certain specific genes [22,23]. Many studies indicate that NF-κB is a stress sensitive transcription factor and its activation is regulated by reactive oxygen species. In our study, NF-кB is activated by H2O2 and subsequently translocated into the nucleus, which is consistent with these studies [24,25]. However, the activation and translocation were effectively decreased by pretreatment with GSPE. Therefore, this observation provides a possible mechanism for the effect of GSPE.
The MAPK signaling pathway is an important upstream regulator of transcriptional factor activities and their signaling affects a wide variety of extracellular stimuli into intracellular events and thus control the activities of downstream transcription factors [26,27]. Several investigations reported that GSPE reduced the oxidative stress of various cells via inhibition of the MAPK signaling cascade [28,29]. It is generally accepted that ERK activation is essential for cell survival, whereas activation of JNK and p38 is thought to play an important role in cell death. Therefore, we investigated whether the p38 and JNK pathways were inhibited by treatment with GSPE. Our data showed that H2O2-induced p38 and JNK activation was significantly reduced when cells were pretreated with GSPE. These results suggested that GSPE reduced H2O2-induced intracellular ROS accumulation, decreased the intracellular oxidative level, indirectly prevented HLEB-3 cells from p38 and JNK signaling pathway, and further proved that GSPE plays an important role in protecting HLEB-3 cells against oxidative stress.
Though the exact mechanism of the reduction of H2O2-induced phosphorylation of MAPK proteins by GSPE is not clear based on the present data, previous studies have shown proteins of the MAPK family to be involved in the activation of NF-кB [30-32]. In this experiment, it appears that the antioxidant property of GSPE contributed to the reduction of the H2O2-induced phosphorylation of MAPKs through both a modulation of ROS and prevention of downstream events such as NF-кB activation. Therefore, the reduction of the MAPK and NF-кB signaling pathways could potentially be used by GSPE to activate certain antioxidant-responsive genes to protect against the H2O2-induced oxidative stress in HLE cells.
Some researches have demonstrated that the contributions of the various receptors in human lens epithelial cells differ in primary and immortalized cells [33]. This is may be one of reasons why our results are slightly different from those of experiments in which primary cells were used.
In conclusion, GSPE protects HLE cells from H2O2-induced oxidative stress by reducing the generation of ROS and modulating the activation of NF-кB and MAPK pathways. GSPE possesses a potential pharmacological application in attenuating H2O2-induced oxidative stress, suggesting a protective effect against cataractogenesis. On the basis of the data reported here, there is strong evidence that GSPE could potentially protect HLEB-3 cells from the damaging effects of oxidative stress. Delaying cataract formation would be beneficial because it has been estimated that a delay of 10 years for cataract formation in humans would decrease sight-threatening cataracts by 45% [34]. Therefore, even a slight delay in the progression of cataract formation could provide increases in visual acuity.
Acknowledgments
The authors thank Pucheng Chen of Harbin Veterinary Research Institute (HVRC) for use of the flow cytometric analysis; Jinxiong Liu (HVRC) for a critical reading of the manuscript; Dongdong Cai (HVRC) and Fumei Wang (HVRC) for data processing.
References
- 1.Williams DL. Oxidation, antioxidants and cataract formation: a literature review. Vet Ophthalmol. 2006;9:292–8. doi: 10.1111/j.1463-5224.2006.00498.x. [DOI] [PubMed] [Google Scholar]
- 2.Colitz CM, Bomser JA, Kusewitt DF. The endogenous and exogenous mechanisms for protection from ultraviolet irradiation in the lens. Int Ophthalmol Clin. 2005;45:141–55. [PubMed] [Google Scholar]
- 3.Spector A. Oxidative stress-induced cataract: mechanism of action. FASEB J. 1995;9:1173–82. [PubMed] [Google Scholar]
- 4.Wolf N, Penn P, Pendergrass W, Van Remmen H, Bartke A, Rabinovitch P, Martin GM. Age-related cataract progression in five mouse models for anti-oxidant protection or hormonal influence. Exp Eye Res. 2005;81:276–85. doi: 10.1016/j.exer.2005.01.024. [DOI] [PubMed] [Google Scholar]
- 5.Bagchi D, Garg A, Krohn RL, Bagchi M, Tran MX, Stohs SJ. Oxygen free radical scavenging abilities of vitamins C and E, and a grape seed proanthocyanidin extract in vitro. Res Commun Mol Pathol Pharmacol. 1997;95:179–89. [PubMed] [Google Scholar]
- 6.Bagchi D, Sen CK, Ray SD, Das DK, Bagchi M, Preuss HG, Vinson JA. Molecular mechanisms of cardioprotection by a novel grape seed proanthocyanidin extract. Mutat Res. 2003;523–524:87–97. doi: 10.1016/s0027-5107(02)00324-x. [DOI] [PubMed] [Google Scholar]
- 7.Prasain JK, Peng N, Dai Y, Moore R, Arabshahi A, Wilson L, Barnes S, Michael Wyss J, Kim H, Watts RL. Liquid chromatography tandem mass spectrometry identification of proanthocyanidins in rat plasma after oral administration of grape seed extract. Phytomedicine. 2009;16:233–43. doi: 10.1016/j.phymed.2008.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nassiri-Asl M, Hosseinzadeh H. Review of the pharmacological effects of Vitis vinifera (Grape) and its bioactive compounds. Phytother Res. 2009;23:1197–204. doi: 10.1002/ptr.2761. [DOI] [PubMed] [Google Scholar]
- 9.Bagchi D, Bagchi M, Stohs SJ, Das DK, Ray SD, Kuszynski CA, Joshi SS, Pruess HG. Free radicals and grape seed proanthocyanidin extract: importance in human health and disease prevention. Toxicology. 2000;148:187–97. doi: 10.1016/s0300-483x(00)00210-9. [DOI] [PubMed] [Google Scholar]
- 10.Middleton E, Jr, Kandaswami C, Theoharides TC. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol Rev. 2000;52:673–751. [PubMed] [Google Scholar]
- 11.Yamakoshi J, Saito M, Kataoka S, Tokutake S. Procyanidin-rich extract from grape seeds prevents cataract formation in hereditary cataractous (ICR/f) rats. J Agric Food Chem. 2002;50:4983–8. doi: 10.1021/jf0201632. [DOI] [PubMed] [Google Scholar]
- 12.Cheng X, Cheng WB, Yan H, Zhang SC, Wang Y. Study on preventing effects of procyanidins on selenite cataract development in rats. Zhongguo Zhong Yao Za Zhi. 2008;33:300–2. [PubMed] [Google Scholar]
- 13.Durukan AH, Evereklioglu C, Hurmeric V, Kerimoglu H, Erdurman C, Bayraktar MZ, Mumcuoglu T. Ingestion of IH636 grape seed proanthocyanidin extract to prevent selenite-induced oxidative stress in experimental cataract. J Cataract Refract Surg. 2006;32:1041–5. doi: 10.1016/j.jcrs.2006.02.041. [DOI] [PubMed] [Google Scholar]
- 14.Ray S, Bagchi D, Lim PM, Bagchi M, Gross SM, Kothari SC, Preuss HG, Stohs SJ. Acute and long-term safety evaluation of a novel IH636 grape seed proanthocyanidin extract. Res Commun Mol Pathol Pharmacol. 2001;109:165–97. [PubMed] [Google Scholar]
- 15.Torres M. Mitogen-activated protein kinase pathways in redox signaling. Front Biosci. 2003;8:d369–91. doi: 10.2741/999. [DOI] [PubMed] [Google Scholar]
- 16.Papa S, Bubici C, Zazzeroni F, Pham CG, Kuntzen C, Knabb JR, Dean K, Franzoso G. The NF-kappaB-mediated control of the JNK cascade in the antagonism of programmed cell death in health and disease. Cell Death Differ. 2006;13:712–29. doi: 10.1038/sj.cdd.4401865. [DOI] [PubMed] [Google Scholar]
- 17.Andley UP, Rhim JS, Chylack LT, Jr, Fleming TP. Propagation and immortalization of human lens epithelial cells in culture. Invest Ophthalmol Vis Sci. 1994;35:3094–102. [PubMed] [Google Scholar]
- 18.Truscott RJ. Age-related nuclear cataract-oxidation is the key. Exp Eye Res. 2005;80:709–25. doi: 10.1016/j.exer.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 19.Spector A. Review: Oxidative stress and disease. J Ocul Pharmacol Ther. 2000;16:193–201. doi: 10.1089/jop.2000.16.193. [DOI] [PubMed] [Google Scholar]
- 20.Hayden MS, Ghosh S. Signaling to NF-kappaB. Genes Dev. 2004;18:2195–224. doi: 10.1101/gad.1228704. [DOI] [PubMed] [Google Scholar]
- 21.Pasparakis M, Luedde T, Schmidt-Supprian M. Dissection of the NF-kappaB signalling cascade in transgenic and knockout mice. Cell Death Differ. 2006;13:861–72. doi: 10.1038/sj.cdd.4401870. [DOI] [PubMed] [Google Scholar]
- 22.Schreck R, Albermann K, Baeuerle PA. Nuclear factor kappa B: an oxidative stress-responsive transcription factor of eukaryotic cells (a review). Free Radic Res Commun. 1992;17:221–37. doi: 10.3109/10715769209079515. [DOI] [PubMed] [Google Scholar]
- 23.Boileau TW, Bray TM, Bomser JA. Ultraviolet radiation modulates nuclear factor kappa B activation in human lens epithelial cells. J Biochem Mol Toxicol. 2003;17:108–13. doi: 10.1002/jbt.10067. [DOI] [PubMed] [Google Scholar]
- 24.Jin XH, Ohgami K, Shiratori K, Koyama Y, Yoshida K, Kase S, Ohno S. Inhibition of nuclear factor-kappa B activation attenuates hydrogen peroxide-induced cytotoxicity in human lens epithelial cells. Br J Ophthalmol. 2007;91:369–71. doi: 10.1136/bjo.2006.107037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dudek EJ, Shang F, Taylor AH. (2)O(2)-mediated oxidative stress activates NF-kappa B in lens epithelial cells. Free Radic Biol Med. 2001;31:651–8. doi: 10.1016/s0891-5849(01)00634-7. [DOI] [PubMed] [Google Scholar]
- 26.Roux PP, Blenis J. ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev. 2004;68:320–44. doi: 10.1128/MMBR.68.2.320-344.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen Z, Gibson TB, Robinson F, Silvestro L, Pearson G, Xu B, Wright A, Vanderbilt C, Cobb MH. MAP kinases. Chem Rev. 2001;101:2449–76. doi: 10.1021/cr000241p. [DOI] [PubMed] [Google Scholar]
- 28.Barden CA, Chandler HL, Lu P, Bomser JA, Colitz CM. Effect of grape polyphenols on oxidative stress in canine lens epithelial cells. Am J Vet Res. 2008;69:94–100. doi: 10.2460/ajvr.69.1.94. [DOI] [PubMed] [Google Scholar]
- 29.Cho ES, Lee KW, Lee HJ. Cocoa procyanidins protect PC12 cells from hydrogen-peroxide-induced apoptosis by inhibiting activation of p38 MAPK and JNK. Mutat Res. 2008;640:123–30. doi: 10.1016/j.mrfmmm.2007.12.012. [DOI] [PubMed] [Google Scholar]
- 30.Vayalil PK, Mittal A, Katiyar SK. Proanthocyanidins from grape seeds inhibit expression of matrix metalloproteinases in human prostate carcinoma cells, which is associated with the inhibition of activation of MAPK and NF kappa B. Carcinogenesis. 2004;25:987–95. doi: 10.1093/carcin/bgh095. [DOI] [PubMed] [Google Scholar]
- 31.Bonvin C, Guillon A, van Bemmelen MX, Gerwins P, Johnson GL, Widmann C. Role of the N-terminal domains of MEKKs in the activation of NF kappa B and MAPK pathways and in the regulation of cell proliferation and apoptosis. Cell Signal. 2002;14:123–31. doi: 10.1016/s0898-6568(01)00219-4. [DOI] [PubMed] [Google Scholar]
- 32.Wang D, Richmond A. Nuclear factor-kappa B activation by the CXC chemokine melanoma growth-stimulatory activity/growth-regulated protein involves the MEKK1/p38 mitogen-activated protein kinase pathway. J Biol Chem. 2001;276:3650–9. doi: 10.1074/jbc.M006115200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Collison DJ, Coleman RA, James RS, Carey J, Duncan G. Characterization of muscarinic receptors in human lens cells by pharmacologic and molecular techniques. Invest Ophthalmol Vis Sci. 2000;41:2633–41. [PubMed] [Google Scholar]
- 34.Kupfer C. Bowman lecture. The conquest of cataract: a global challenge. Trans Ophthalmol Soc U K. 1985;104:1–10. [PubMed] [Google Scholar]