Abstract
Objective:
To identify and describe correlates of medication adherence in a large, national sample of outpatients with bipolar disorder.
Method:
Data were collected via a self-report, Web-based survey in January and February of 2008 from US patients aged 18–65 years who reported a diagnosis of bipolar disorder and current use of psychotropic medication. Patients with a Composite International Diagnostic Interview-bipolar disorder (CIDI-bipolar disorder) score ≥ 7, indicating a high risk of bipolar disorder, were included in the analyses. Medication adherence was assessed via the Morisky Medication Adherence Scale, with scores ≥ 2 being considered nonadherent. The primary analysis was a multivariate binomial logistic regression with adherence as the dependent variable. Covariates included patient demographics, physical health measures including Medical Outcomes Study 8-item Short-Form Health Survey physical summary score, number of manic and depressive episodes, 24-item Behavior and Symptom Identification Scale (BASIS-24), Liverpool University Neuroleptic Side-Effect Rating Scale (LUNSERS), Satisfaction With Antipsychotic Medication scale (SWAM), and current psychiatric medication use.
Results:
Nearly half (49.5%) of the 1,052 bipolar patients in the analysis were classified as being nonadherent. Adherence was positively associated with college degree, higher SWAM total score, and monotherapy treatment. Adherence was negatively associated with female sex, alcohol use, BASIS-24 total score, and LUNSERS total score.
Conclusions:
Nonadherence is common among patients with bipolar disorder. By addressing tolerability issues and treatment satisfaction, which are both significant correlates of adherence, health care providers may be able to improve adherence and, ultimately, treatment outcomes.
Bipolar disorder is a chronic condition characterized by the recurrence of episodes of manic, hypomanic, depressive, and mixed states.1 The lifetime prevalence of bipolar disorder has been estimated to be 1%, although recent evidence suggests that the prevalence may be as high as 5% when applying broader definitions that encompass the entire bipolar spectrum.2,3 Worldwide, bipolar disorder is the sixth leading cause of medical disability among patients aged 15 to 44 years.1 The annual cost of bipolar disorder in the United States in 1991 was estimated to be $45 billion, $7 billion of which was direct costs and $38 billion was indirect costs, including lost productivity of patients and caregivers.1 Lost productivity alone may account for up to $14.1 billion among employed patients.3
Current pharmacologic interventions for bipolar disorder include lithium, anticonvulsant agents, and second-generation antipsychotic medications.4,5 As in many chronic disease states with intermittent symptoms, medication nonadherence is a common occurrence in bipolar disorder5 and is associated with poorer outcomes.6–10 One survey found that 34% of respondents with bipolar disorder reported missing at least 1 dose of psychotropic medication in a 10-day period.11 Studies of veterans with bipolar disorder found even higher rates of nonadherence at approximately 45.9% among those taking lithium or anticonvulsant agents12 and 48.1% among those taking antipsychotic medications.13 Nonadherent patients with bipolar disorder are at a greater risk of suicide,6,7 experience shorter duration between episodes, and are more likely to relapse,8,9 resulting in higher total medical expenditures and higher outpatient mental health care costs.10
Multiple sociodemographic factors have been associated with nonadherence in bipolar disorder, including gender,14 age,11 race,14,15 and education level.15 In addition, clinical factors such as alcohol dependence,11,14 comorbid affective disorders,11 manic episodes,14 and severity of depressive episodes15 are associated with poorer adherence. However, because medication nonadherence is a behavioral symptom related to specific decisions made by patients, patient-centered experiences should also be considered. Patient satisfaction with medication is a significant predictor of better adherence,14,15 whereas side effects such as cognitive impairment,15 extrapyramidal symptoms, sedation, sexual dysfunction, and weight gain are associated with poorer adherence.15,16
Clinical Points
♦ Nonadherence is common among patients with bipolar disorder.
♦ Several risk factors for nonadherence have been identified, including low medication satisfaction, a high number of medications, and psychic side effects.
♦ By assessing tolerability and side effects, physicians may help to assist patients to improve adherence and health outcomes.
To date, research on medication nonadherence in bipolar disorder has been limited to narrow subpopulations such as veterans or patients recently discharged from the hospital. The primary objective of this survey study was to identify and describe drivers of medication adherence in a large, national sample of outpatients with bipolar disorder.
METHOD
Study Design
This was a Web-based cross-sectional survey of a subpopulation within a probability sample of the US population. Data were collected for the Bipolar Evaluation of Satisfaction and Tolerability (BEST) study via a self-report, Web-based survey. The questionnaire took approximately 25 minutes to complete and was fielded in January and February 2008. The BEST study protocol and questionnaire were reviewed and approved by the Essex Institutional Review Board (Lebanon, New Jersey). Informed consent was obtained prior to entering the survey.
Recruitment
The study sample was identified through the 2006 and 2007 US National Health and Wellness Survey (NHWS), an annual cross-sectional study of the disease status, health care attitudes, behaviors, and outcomes of the adult population aged 18 years and older, and the Chronic Ailment Panel of Lightspeed Research. In 2006 and 2007, the US NHWS was fielded to 60,000+ members of the General Panel of Lightspeed Research. Through panel identification numbers, respondents were identified and recontacted for this study. The Chronic Ailment Panel of Lightspeed Research is a subset of the general panel for which disease status has been provided by panelists. The General Panel of Lightspeed Research is an actively managed online consumer panel of registered panelists worldwide with a multisource recruiting methodology that includes opt-in e-mail, coregistration, e-newsletter campaigns, traditional banner placements, and internal and external affiliate networks.
Potential respondents were sent a general e-mail invitation from Lightspeed Research to participate in the study. Those interested in participation followed the Web link provided in the invitation to access the statement of informed consent. Those who provided their informed consent by agreeing to participate then entered the survey.
Study Population
Respondents were eligible for study inclusion if they were aged 18–65 years, could read and write English, had a self-reported diagnosis of bipolar disorder, were using a psychotropic medication to treat bipolar disorder, and resided in the United States. Patients were excluded from the study if they were themselves, or through an immediate family member, employed by a pharmaceutical company, a market research company, or media or advertising agency or were affiliated with a health care provider or company as a clinical investigator conducting clinical trials or providing consulting services. Additional eligibility requirements were established for inclusion in the analytic sample. Patients who reported ever receiving a diagnosis of schizophrenia were excluded from the analysis. The Composite International Diagnostic Interview-bipolar disorder (CIDI-bipolar disorder) was included in the study as a diagnostic confirmatory instrument. CIDI scores of 7 or 8 are classified as a high risk of bipolar disorder, and scores of 9 are classified as a very high risk of bipolar disorder.17 Only patients with a CIDI-bipolar disorder score of 7 or higher were included in the analytic sample.
Study Measures
Medication adherence. Medication adherence was assessed using the Morisky Medication Adherence Scale (MMAS).18 The MMAS consists of 4 yes/no questions that assess general adherence of using the prescribed medication. A score ≥ 2 has reportedly been associated with a high risk of medication nonadherence. It was determined (a priori) that respondents with a score ≥ 2 would be classified as being nonadherent and those with a score of 0 or 1 as being adherent.
Patient demographics and physical health measures. Patient demographic information available for analysis included gender, age, race, and educational status. Physical health measures included number of physical health conditions, body mass index (BMI), alcohol use, and physical health status. The following conditions were included in the count of physical health conditions: arthritis, asthma, cancer, congestive heart failure, diabetes, hypertension, high cholesterol, HIV/AIDS, and osteoporosis. BMI was calculated using self-reported height and weight. Alcohol use was assessed as a dichotomous variable of using alcohol versus not using alcohol.
Physical health status was assessed with the physical component summary score of the Medical Outcomes Study 8-item Short-Form Health Survey (SF-8).19 The SF-8 is a brief multipurpose generic measure of health status designed to assess physical functioning, role limitations due to physical health problems, bodily pain, general health, vitality, social functioning, role limitations due to emotional problems, and mental health. These 8 items directly correspond to the 8 subscales of the SF-36 and are scored accordingly. In addition to the subscales, a mental health summary score and a physical health summary score are computed. For both summary measures, the normative score for the US population is 50, with higher scores indicating better physical or mental well-being.19
Bipolar disorder characteristics. Patients were asked to provide the number of manic episodes and the number of depressive episodes experienced in the past year. Mental health status was assessed using the 24-item Behavior and Symptom Identification Scale (BASIS-24),20 a broad mental health status instrument designed to capture domains of psychiatric symptomatology. Using a weighted average algorithm, an overall score is computed, as well as scores for 6 subscales, which include depression and functioning, relationships, self-harm, emotional lability, psychosis, and substance abuse.20
Patient experience with medication. Side effect burden was assessed with the Liverpool University Neuroleptic Side-Effect Rating Scale (LUNSERS).21 The LUNSERS is a 51-item 5-point Likert scale, a self-report instrument developed from the 48-item Udvalg for Kliniske Undersøgelser (UKU) rating scale with 41 items from the 48 UKU items plus 10 red-herring items developed to detect malingering or hypochondriacal responding. The correlation for the total side effect scores on the LUNSERS and total scores on the UKU has been reported as 0.828 (P < .001).21 The instrument has also demonstrated sensitivity to discriminate between various types of side effects22 and chlorpromazine equivalent doses21 among patients with schizophrenia. The LUNSERS provides a total score with and without red-herring items (range, 0–164, excluding red herrings) and 8 subscale scores.
These subscales include extrapyramidal side effects (eg, muscle spasms and shakiness), other autonomic side effects (eg, dizziness and increased sweating), psychic side effects (eg, difficulty concentrating and lack of emotions), miscellaneous side effects (eg, headaches and losing weight), anticholinergic side effects (eg, dry mouth and constipation), allergic reactions (eg, rash and sensitivity to sun), hormonal side effects (eg, swollen or tender chest and increased sex drive), and red-herring items (eg, runny nose and hair loss).21–23 It should be noted that the use of the term psychic in this article refers to the specific name of the subscale in the LUNSERS instrument.
Patient satisfaction with medication was assessed using the Satisfaction With Antipsychotic Medication (SWAM) scale. The SWAM is a 33-item, 5-point Likert scale, a self-report instrument (range, 0–10). The SWAM provides a total score plus subscale scores for treatment acceptability and medication insight.24 The SWAM total score was included in the primary analysis.
Patients reported their current use of prescription medications to treat their bipolar disorder from a prompted list. Medications were categorized into the following categories: second-generation antipsychotics, first-generation antipsychotics, mood stabilizers (lithium and anticonvulsant agents), selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, dopamine-norepinephrine reuptake inhibitors, other antidepressants, and sedative hypnotics. Patients were also categorized as currently using monotherapy (ie, a single psychotropic medication from any of the above-mentioned class of drugs) versus combination therapy (ie, more than 1 psychotropic medication).
Functional measures. The Work Productivity and Activity Impairment (WPAI) questionnaire was used to assess productivity loss and activity impairment for the past 7 days. The WPAI yields 4 metrics of impairment due to health: absenteeism or percent work time missed; presenteeism or percent of impairment while working; overall work productivity impairment, which considers both absenteeism and presenteeism; and impairment in daily activities.25
Economic measures. Self-reported health resource utilization within the previous 6 months consisted of questions regarding number of visits to an emergency room, having been hospitalized, and total number of days hospitalized.
Statistical Analyses
Descriptive analyses were conducted in the total study sample for all variables of interest, as well as MMAS total scores and individual MMAS items. Adherent and nonadherent samples were compared through bivariate analyses using χ2 for categorical variables and t tests for continuous variables. A correlation matrix was developed to assess the degree of association between all potential predictors. When 2 variables were highly correlated, the most clinically relevant was chosen for inclusion in analysis to reduce multicollinearity.
The primary analysis was a multivariate binomial logistic regression with adherence (ie, MMAS) as the dependent variable. An additional model was developed using each of the 4 MMAS items as dependent variables. Continuous scale scores were dichotomized into high (mean and greater) and low (less than mean). Post hoc regression models were also developed using the BASIS-24 subscales and the LUNSERS subscales as explanatory variables.
RESULTS
Sample Characteristics
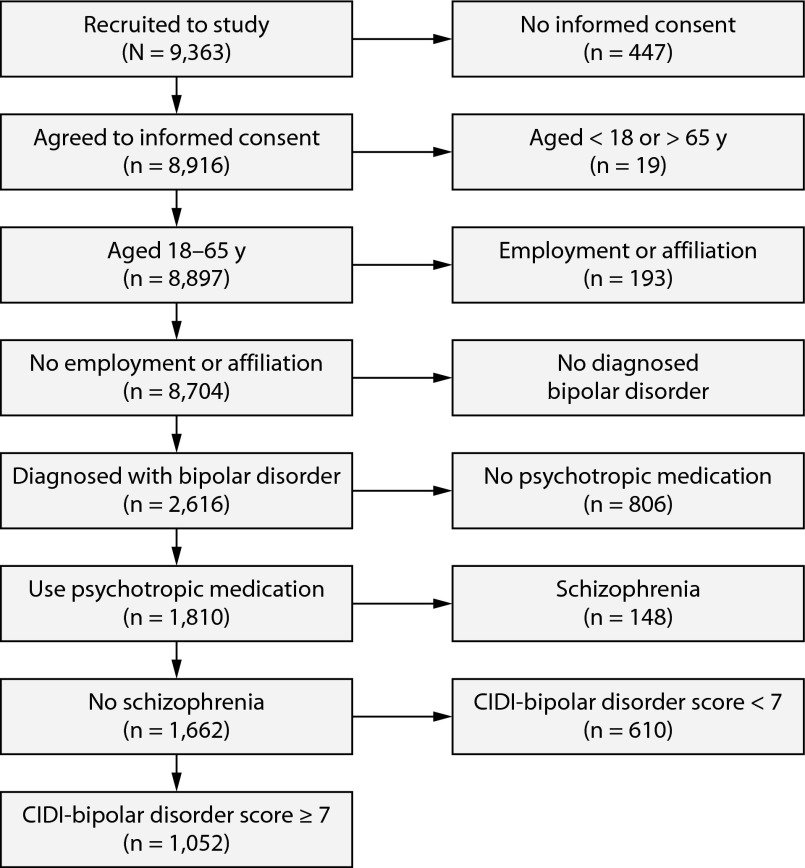
Recruitment of 9,363 subjects yielded 1,810 (19.3%) subjects who met the full inclusion criteria for the study. Of these, 1,052 (52.6%) met the additional eligibility requirements for analytic inclusion of no schizophrenia and a CIDI-bipolar disorder score of ≥7. Figure 1 illustrates the flow of patients through eligibility criteria for the study and analysis.
Figure 1.
Flowchart of Sample Sizes by Study Eligibility
Abbreviation: CIDI = Composite International Diagnostic Interview.
On the basis of the MMAS, nearly half (49.5%) of bipolar patients in the sample were classified as being nonadherent. Of the sample, 57.8% forgot to take medication, 42.2% were careless at times about taking medicine, 34.2% sometimes stopped taking medication when feeling better, and 31.5% sometimes stopped taking medication when feeling worse. Table 1 shows the characteristics of the total patient sample and the comparison of adherent and nonadherent patients. Over half (52.2%) of the patients were taking only 1 medication; 30.9% were taking 2, and 12.9% were taking 3 medications (Table 2).
Table 1.
Sample Characteristics and Comparison of Adherent and Nonadherent Patientsa
| Variable | Total (N = 1,052) | Adherent (n = 531) | Nonadherent (n = 521) | P Value |
| Patient demographics | ||||
| Female | 77.6 | 75.7 | 79.5 | .144 |
| Age, y | 42.08 (10.63) | 43.17 (10.49) | 40.98 (10.66) | < .001 |
| White | 85.8 | 86.1 | 85.6 | .831 |
| College graduate | 24.1 | 29.2 | 19.0 | < .001 |
| Physical health measures | ||||
| Alcohol use | 55.1 | 47.3 | 63.2 | < .001 |
| Body mass index, kg/m2 | 31.76 (8.93) | 32.02 (8.94) | 31.50 (8.91) | .353 |
| Comorbid count | ||||
| None | 19.0 | 16.8 | 21.3 | .060 |
| 1 | 23.3 | 23.2 | 23.4 | .923 |
| 2 | 22.0 | 21.7 | 22.3 | .812 |
| 3+ | 35.7 | 38.4 | 33.0 | .067 |
| SF-8 physical score | 40.57 (9.95) | 39.90 (10.38) | 41.25 (9.46) | .028 |
| Bipolar disease characteristics | ||||
| No. of depressive episodes | 8.39 (11.08) | 7.41 (8.70) | 9.39 (13.0) | .004 |
| No. of manic episodes | 6.54 (9.30) | 5.62 (8.24) | 7.47 (10.19) | .001 |
| BASIS-24 total score | 1.82 (0.75) | 1.63 (0.76) | 2.01 (0.70) | < .001 |
| Depression/functioning | 2.14 (0.97) | 1.94 (0.98) | 2.34 (0.91) | < .001 |
| Interpersonal relationships | 1.79 (0.94) | 1.63 (0.95) | 1.95 (0.91) | < .001 |
| Self-harm | 0.83 (1.0) | 0.67 (0.93) | 0.99 (1.04) | < .001 |
| Emotional lability | 2.36 (1.0) | 2.13 (1.04) | 2.60 (0.90) | < .001 |
| Psychosis | 1.00 (1.00) | 0.81 (0.92) | 1.20 (1.03) | < .001 |
| Substance abuse | 0.69 (0.92) | 0.54 (0.86) | 0.85 (0.96) | < .001 |
| Bipolar treatment characteristics | ||||
| LUNSERS score | ||||
| Total (excluding red herrings) | 55.58 (28.13) | 51.36 (28.39) | 59.88 (27.21) | < .001 |
| Extrapyramidal side effects | 9.00 (6.13) | 8.33 (6.23) | 9.67 (5.97) | < .001 |
| Other autonomic side effects | 5.99 (4.33) | 5.42 (4.29) | 6.57 (4.30) | < .001 |
| Psychic side effects | 20.17 (8.84) | 18.50 (8.96) | 21.88 (8.38) | < .001 |
| Miscellaneous side effects | 5.33 (3.13) | 4.99 (3.14) | 5.66 (3.09) | < .001 |
| Anticholinergic side effects | 6.04 (4.07) | 5.70 (3.90) | 6.38 (4.22) | .007 |
| Allergic reaction | 3.16 (3.21) | 3.06 (3.23) | 3.27 (3.19) | .289 |
| Hormonal side effects | 5.90 (4.24) | 5.36 (4.13) | 6.45 (4.28) | < .001 |
| Red herrings | 9.90 (7.10) | 9.60 (7.19) | 10.21 (7.00) | .166 |
| Satisfaction With Antipsychotic Medication scale score | 7.71 (1.36) | 8.09 (1.31) | 7.31 (1.29) | < .001 |
| Second-generation antipsychotics | 47.0 | 46.7 | 47.2 | .868 |
| First-generation antipsychotics | 1.7 | 0.9 | 2.5 | .052 |
| Mood stabilizers | 83.3 | 84.1 | 82.3 | .424 |
| Selective serotonin reuptake inhibitors | 16.8 | 15.8 | 17.8 | .379 |
| Serotonin-norepinephrine reuptake inhibitors | 6.2 | 5.8 | 6.5 | .643 |
| Dopamine-norepinephrine reuptake inhibitors | 7.2 | 7.5 | 6.9 | .696 |
| Other antidepressants | 3.6 | 4.9 | 2.3 | .024 |
| Sedative hypnotics | 5.9 | 5.5 | 6.3 | .548 |
| Monotherapy use | 52.2 | 54.0 | 50.3 | .222 |
| Economic outcomes | ||||
| WPAI absenteeism | 15.47 (25.59) | 15.74 (27.05) | 15.23 (24.30) | .882 |
| WPAI presenteeism | 41.49 (27.17) | 36.96 (27.24) | 45.58 (26.56) | .020 |
| WPAI overall work impairment | 34.75 (22.98) | 30.28 (22.65) | 38.78 (22.6) | .006 |
| WPAI activity impairment | 46.16 (29.91) | 39.91 (29.23) | 51.83 (29.51) | .002 |
| Visited the emergency room | 15.87 | 11.68 | 20.15 | < .001 |
| No. of emergency room visits | 0.31 (0.93) | 0.20 (0.77) | 0.42 (1.06) | < .001 |
| Hospitalized | 11.69 | 8.10 | 15.36 | < .001 |
| Days hospitalized | 0.98 (5.25) | 0.87 (6.22) | 1.09 (4.04) | .504 |
Categorical variables are shown as percentages, with χ2 used to assess significance. Continuous variables are shown as mean (SD), with t tests used to assess significance.
Abbreviations: BASIS-24 = 24-item Behavior and Symptom Identification Scale, LUNSERS = Liverpool University Neuroleptic Side-Effect Rating Scale, SF-8 = Medical Outcomes Study 8-item Short-Form Health Survey, WPAI = Work Productivity and Activity Impairment.
Table 2.
Number of Medications Patients Were Taking at Time of Survey
| No. of Medications | n | % |
| 1 | 549 | 52.19 |
| 2 | 325 | 30.89 |
| 3 | 136 | 12.93 |
| 4 | 34 | 3.23 |
| 5 | 5 | 0.48 |
| 7 | 1 | 0.1 |
| 9 | 1 | 0.1 |
| 10 | 1 | 0.1 |
In the study sample, 78% were female, 86% were white, and 24% were college graduates with a mean age of 42 years. Adherent patients were significantly older and were more likely to be college graduates than nonadherent patients. Adherent patients were also less likely to use alcohol but had poorer physical functioning than nonadherent patients.
In bivariate analyses, bipolar disease severity and treatment characteristics varied by treatment adherence. Adherent patients reported significantly fewer depressive and manic episodes in the previous year and lower (ie, less pathological) scores on the BASIS-24. Adherent patients experienced fewer extrapyramidal, other autonomic, psychic, miscellaneous, anticholinergic, and hormonal side effects and rated treatment satisfaction higher than nonadherent patients.
Among employed patients, those who were adherent had less productivity impairment while working and less overall work impairment than nonadherent patients. Adherent patients had less activity impairment and were also less likely to report a visit to the emergency room or a hospitalization in the prior 6 months than were nonadherent patients.
Multivariate Analyses
In the analysis that dichotomized continuous scales into high versus low categories (Table 3), sex, physical health status, and red-herring scores were not significantly associated with adherence. Ethnicity, BMI, number of manic and depressive episodes, number of medical comorbidities, and psychotropic medication classes used were not significantly associated with adherence. Patients displaying greater pathology on the BASIS-24 were only 54.2% as likely to be adherent as those with less pathological scores, whereas patients endorsing more side effects on the LUNSERS were only 59.7% as likely to be adherent as those with fewer side effects.
Table 3.
Correlates of Adherence (scale scores entered as dichotomous variables)a
| Variable | Odds Ratio | 95% CI | P Value | |
| Lower Limit | Upper Limit | |||
| Female | 0.752 | 0.542 | 1.044 | .089 |
| Age | 0.999 | 0.985 | 1.014 | .937 |
| White | 0.998 | 0.683 | 1.459 | .993 |
| College graduate* | 1.652 | 1.202 | 2.270 | .002 |
| Alcohol use** | 0.521 | 0.396 | 0.686 | < .001 |
| Body mass index | 1.001 | 0.985 | 1.017 | .922 |
| Comorbid count | 1.033 | 0.930 | 1.149 | .541 |
| SF-8 physical score | 0.847 | 0.631 | 1.138 | .272 |
| No. of depressive episodes | 1.001 | 0.986 | 1.016 | .879 |
| No. of manic episodes | 0.993 | 0.976 | 1.011 | .454 |
| BASIS-24 total score** | 0.542 | 0.403 | 0.728 | < .001 |
| LUNSERS | ||||
| Total (excluding red herrings)** | 0.597 | 0.412 | 0.865 | .006 |
| Red herrings | 1.364 | 0.951 | 1.957 | .092 |
| Satisfaction With Antipsychotic Medication scale* | 2.401 | 1.817 | 3.172 | < .001 |
| Second-generation antipsychotics | 1.124 | 0.836 | 1.512 | .439 |
| Traditional antipsychotics | 0.414 | 0.137 | 1.247 | .117 |
| Mood stabilizers | 1.188 | 0.763 | 1.849 | .447 |
| Selective serotonin reuptake inhibitors | 0.953 | 0.628 | 1.448 | .823 |
| Serotonin-norepinephrine reuptake inhibitors | 0.834 | 0.462 | 1.504 | .546 |
| Dopamine-norepinephrine reuptake inhibitors | 1.216 | 0.711 | 2.081 | .475 |
| Other antidepressants | 1.784 | 0.847 | 3.757 | .128 |
| Sedative hypnotics | 0.930 | 0.522 | 1.657 | .805 |
| Monotherapy use* | 1.418 | 1.025 | 1.962 | .035 |
Method: logistic regression analysis.
*Indicates a positive correlation.
** Indicates a negative correlation.
Abbreviations: BASIS-24 = 24-item Behavior and Symptom Identification Scale, LUNSERS = Liverpool University Neuroleptic Side-Effect Rating Scale, SF-8 = Medical Outcomes Study 8-item Short-Form Health Survey, WPAI = Work Productivity and Activity Impairment.
Post hoc analysis of subscales found significant negative correlations between the BASIS-24 emotional lability subscale (OR = 0.812, 95% CI, 0.665–0.992) and the LUNSERS psychic side effects subscale (OR = 0.962, 95% CI, 0.937–0.987) on adherence. Psychic side effects consisted of sleeping difficulties, cognitive difficulties, lack of emotions, and depression.
Greater medication satisfaction, as measured by the SWAM, was positively associated with total MMAS score (ie, adherence) and each of the 4 MMAS items. Patients who reported being satisfied with their medication were 2.4 times as likely to be adherent as patients who were not satisfied. In the primary model, patients using a single psychotropic medication to treat bipolar disorder were 1.4 times as likely to be adherent as those using more than 1 psychotropic medication.
DISCUSSION
Nonadherence with prescription medication is common among patients with bipolar disorder.5 In this cross-sectional Web-based survey, nearly half of patients were classified as nonadherent, which is consistent with rates reported in other studies with bipolar patients.12,13 Correlates of adherence in this study included male gender, college graduate, no alcohol use, poorer physical well-being, fewer psychiatric symptoms, fewer side effects, greater treatment satisfaction, and monotherapy. It is unclear why males were more adherent, in contrast to prior evidence that females are more adherent in this population.14 However, the Copeland et al14 study was conducted in a veterans’ population and may be substantially different from ours. The association of higher education with adherence, and alcohol use with nonadherence, is consistent with prior studies.11,14,15 The finding that poorer physical health is associated with higher adherence was unexpected; however, this finding was not consistent between the primary and secondary models. Moreover, the effect size was rather small and of questionable clinical significance in light of the other statistically significant factors. Initial unadjusted analyses indicated that nonadherent patients experienced both more depressive and manic episodes. However, when adjusting for covariates in logistic regression models, prior manic and depressive episodes were not associated with nonadherence in contrast with reports of previous investigators.14,15 We did however find a significant association between greater symptom burden and nonadherence, which probably represents a worsening of illness due to a lack of medication adherence.
Our finding that tolerability and satisfaction with medication are strong mediators of adherence is consistent with other studies.14–16,26 The post hoc analysis found that psychic side effects (sleeping difficulties, cognitive difficulties, lack of emotions, and depression) was the only subscale associated with nonadherence. The role of monotherapy, defined as using a single psychotropic medication, in treatment adherence is unclear. Although it may be possible that taking fewer medications may be associated with fewer side effects and lower overall pill burden, this may also reflect a less clinically complex patient population.
This study was a cross-sectional, patient-reported, Internet-based study and has several limitations. The cross-sectional design prevents causal inferences from being made. All data were self-reported by patients and were not confirmed by clinicians, clinical records, or claims data. Although validated measures were used, the lack of longitudinal observation increases the overall variance within the data, which may be subject to unobserved transient or situational confounding.
The use of an Internet-based survey approach may limit the generalizability of the results due to sample selection bias. These patients were functioning well enough to respond to a 25-minute online survey. In addition, it is not clear what proportion of patients with bipolar disorder has Internet access, although according to the US Census Bureau, approximately 70% of adults in the United States had Internet access in 2006.27
The results are also limited by the fact that the sample in this analysis was largely female. In the NHWS, over 56% of those diagnosed with bipolar disorder were women. In the BEST study, this percentage increased to 78%. We are not certain why this occurred. It is possible that the Chronic Ailment Panel recruited a disproportionate number of women or that women were just more likely to participate.
Pill burden among patients may be another limitation in this study's generalizability. In this study, 83.1% of patients were taking 1 or 2 medications, 16.9% took 3 or more medications, and only 4.0% were taking 4 or more medications. In the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD) study, 60% of patients took 1 or 2 medications, 40% took 3 or more, and 18% took 4 or more medications. In both cases, the majority of patients took 1 or 2 medications; however, the STEP-BD study had a much higher percentage of patients taking the greatest number of medications (4 or more medications).28 However, as our study did not examine pill burden, nor was it discussed in the article by Goldberg et al,28 its affect on adherence is not clear.
A German study29 (N = 312) found that patients took a mean ± SD of 3.1 ± 1.6 medications daily; however, there was no significant association between either the daily number of pills taken or the daily number of medications taken and adherence. In addition, a survey of experts examining adherence in patients with bipolar disease did not find pill burden to be a risk factor for nonadherence.30
Because medication adherence is such a critical factor in ensuring favorable treatment outcomes, efforts to optimize the acceptability of treatment regimens are important, particularly given the wide range of options for bipolar disorder. By utilizing treatment strategies, which address tolerability and treatment satisfaction, health care providers may be able to assist patients to improve adherence and, ultimately, treatment outcomes.6–10
Drug names: lithium (Lithobid and others).
Potential conflicts of interest: Dr Bates is an employee of and stock shareholder in Bristol-Myers Squibb. Dr Kim is a former employee of Bristol-Myers Squibb. Dr Bolge is a former employee of Consumer Health Sciences/Kantar Health. Mr Whitehead has no personal affiliation or financial relationship with any commercial interest to disclose relevant to this article.
Funding/support: The Bipolar Evaluation of Satisfaction and Tolerability (BEST) study was funded by Bristol-Myers Squibb and conducted by Consumer Health Sciences/Kantar Health. The study, analysis, and manuscript preparation were carried out by Consumer Health Sciences/Kantar Health and funded by Bristol-Myers Squibb.
Previous presentation: Results were presented at the Academy of Managed Care Pharmacy 2008 Educational Conference; October 15–18, 2008; Kansas City, Missouri.
Acknowledgments
The authors would like to thank Jonathan Chapnick, BA, who managed the study implementation, and Shaloo Gupta, MS, who performed data analysis for the study. Both Mr Chapnick and Ms Gupta are employees of Consumer Health Sciences/Kantar Health, Princeton, New Jersey; neither individual reports any other conflict of interest related to this study.
REFERENCES
- 1.Sajatovic M. Bipolar disorder: disease burden. Am J Manag Care. 2005;11(suppl 3):S80–S84. [PubMed] [Google Scholar]
- 2.Hirschfeld RM, Vornik LA. Bipolar disorder: costs and comorbidity. Am J Manag Care. 2005;11(suppl 3):S85–S90. [PubMed] [Google Scholar]
- 3.Kessler RC, Akiskal HS, Ames M, et al. Prevalence and effects of mood disorders on work performance in a nationally representative sample of US workers. Am J Psychiatry. 2006;163(9):1561–1568. doi: 10.1176/appi.ajp.163.9.1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Buckley PF.Update on the treatment and management of schizophrenia and bipolar disorder CNS Spectr 200813suppl 11–10.,quiz 11–12 [DOI] [PubMed] [Google Scholar]
- 5.Miklowitz DJ, Johnson SL. The psychopathology and treatment of bipolar disorder. Annu Rev Clin Psychol. 2006;2(1):199–235. doi: 10.1146/annurev.clinpsy.2.022305.095332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gonzalez-Pinto A, Mosquera F, Alonso M, et al. Suicidal risk in bipolar I disorder patients and adherence to long-term lithium treatment. Bipolar Disord. 2006;8(5, pt 2):618–624. doi: 10.1111/j.1399-5618.2006.00368.x. [DOI] [PubMed] [Google Scholar]
- 7.Baldessarini RJ, Tondo L, Davis P, et al. Decreased risk of suicides and attempts during long-term lithium treatment: a meta-analytic review. Bipolar Disord. 2006;8(5, pt 2):625–639. doi: 10.1111/j.1399-5618.2006.00344.x. [DOI] [PubMed] [Google Scholar]
- 8.Keller MB. Improving the course of illness and promoting continuation of treatment of bipolar disorder. J Clin Psychiatry. 2004;65(suppl 15):10–14. [PubMed] [Google Scholar]
- 9.Altman S, Haeri S, Cohen LJ, et al. Predictors of relapse in bipolar disorder: a review. J Psychiatr Pract. 2006;12(5):269–282. doi: 10.1097/00131746-200609000-00002. [DOI] [PubMed] [Google Scholar]
- 10.Gianfrancesco FD, Sajatovic M, Rajagopalan K, et al. Antipsychotic treatment adherence and associated mental health care use among individuals with bipolar disorder. Clin Ther. 2008;30(7):1358–1374. doi: 10.1016/s0149-2918(08)80062-8. [DOI] [PubMed] [Google Scholar]
- 11.Baldessarini RJ, Perry R, Pike J. Factors associated with treatment nonadherence among US bipolar disorder patients. Hum Psychopharmacol. 2008;23(2):95–105. doi: 10.1002/hup.908. [DOI] [PubMed] [Google Scholar]
- 12.Sajatovic M, Valenstein M, Blow F, et al. Treatment adherence with lithium and anticonvulsant medications among patients with bipolar disorder. Psychiatr Serv. 2007;58(6):855–863. doi: 10.1176/ps.2007.58.6.855. [DOI] [PubMed] [Google Scholar]
- 13.Sajatovic M, Valenstein M, Blow FC, et al. Treatment adherence with antipsychotic medications in bipolar disorder. Bipolar Disord. 2006;8(3):232–241. doi: 10.1111/j.1399-5618.2006.00314.x. [DOI] [PubMed] [Google Scholar]
- 14.Copeland LA, Zeber JE, Salloum I.M., et al. Treatment adherence and illness insight in veterans with bipolar disorder. J Nerv Ment Dis. 2008;196(1):16–21. doi: 10.1097/NMD.0b013e318160ea00. [DOI] [PubMed] [Google Scholar]
- 15.Johnson FR, Ozdemir S, Manjunath R, et al. Factors that affect adherence to bipolar disorder treatments: a stated-preference approach. Med Care. 2007;45(6):545–552. doi: 10.1097/MLR.0b013e318040ad90. [DOI] [PubMed] [Google Scholar]
- 16.Zarate CA., Jr.Antipsychotic drug side effect issues in bipolar manic patients J Clin Psychiatry 200061suppl 852–61,.discussion 62–63 [PubMed] [Google Scholar]
- 17.Kessler RC, Akiskal HS, Angst J., et al. Validity of the assessment of bipolar spectrum disorders in the WHO CIDI 3.0. J Affect Disord. 2006;96(3):259–269. doi: 10.1016/j.jad.2006.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Morisky DE, Green L.W., Levine DM. Concurrent and predictive validity of a self-reported measure of medication adherence. Med Care. 1986;24(1):67–74. doi: 10.1097/00005650-198601000-00007. [DOI] [PubMed] [Google Scholar]
- 19.Ware JE, Kosinski M, Dewey JE, et al. A Manual for Users of the SF-8 Health Survey. Lincoln, RI: QualityMetric Incorporated; 2001. [Google Scholar]
- 20.Eisen SV, Gerena M, Ranganathan G, et al. Reliability and validity of the BASIS-24 mental health survey for whites, African-Americans, and Latinos. J Behav Health Serv Res. 2006;33(3):304–323. doi: 10.1007/s11414-006-9025-3. [DOI] [PubMed] [Google Scholar]
- 21.Day JC, Wood G, Dewey M, et al. A self-rating scale for measuring neuroleptic side-effects: validation in a group of schizophrenic patients. Br J Psychiatry. 1995;166(5):650–653. doi: 10.1192/bjp.166.5.650. [DOI] [PubMed] [Google Scholar]
- 22.Lambert TJ, Cock N, Alcock SJ, et al. Measurement of antipsychotic-induced side effects: support for the validity of a self-report (LUNSERS) versus structured interview (UKU) approach to measurement. Hum Psychopharmacol. 2003;18(5):405–411. doi: 10.1002/hup.495. [DOI] [PubMed] [Google Scholar]
- 23.Morrison P, Gaskill D, Meehan T, et al. The use of the Liverpool University Neuroleptic Side-Effect Rating Scale (LUNSERS) in clinical practice. Aust N Z J Ment Health Nurs. 2000;9(4):166–176. doi: 10.1046/j.1440-0979.2000.00181.x. [DOI] [PubMed] [Google Scholar]
- 24.Rofail D, Gray R, Gournay K. The development and internal consistency of the Satisfaction With Antipsychotic Medication Scale. Psychol Med. 2005;35(7):1063–1072. doi: 10.1017/s0033291705004526. [DOI] [PubMed] [Google Scholar]
- 25.Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–365. doi: 10.2165/00019053-199304050-00006. [DOI] [PubMed] [Google Scholar]
- 26.Fleck DE, Keck PE, Jr., Corey KB, et al. Factors associated with medication adherence in African American and white patients with bipolar disorder. J Clin Psychiatry. 2005;66(5):646–652. doi: 10.4088/jcp.v66n0517. [DOI] [PubMed] [Google Scholar]
- 27.US Census Bureau. 127th ed. Washington, DC: Government Printing Office; 2008. Table 1128 adult computer and adult Internet users, by selected characteristics 1995 to 2006. Statistical Abstracts of the United States: 2008. 719. [Google Scholar]
- 28.Goldberg JF, Brooks JO, 3rd, Kurita K, et al. Depressive illness burden associated with complex polypharmacy in patients with bipolar disorder: findings from the STEP-BD. J Clin Psychiatry. 2009;70(2):155–162. doi: 10.4088/jcp.08m04301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bauer M, Glenn T, Grof P, et al. The association between concurrent psychotropic medications and self-reported adherence with taking a mood stabilizer in bipolar disorder. Hum Psychopharmacol. 2010;25(1):47–54. doi: 10.1002/hup.1077. [DOI] [PubMed] [Google Scholar]
- 30.Velligan DI, Weiden PJ, Sajatovic M, et al. The Expert Consensus Guideline Series: Adherence Problems in Patients With Serious and Persistent Mental Illness. J Clin Psychiatry. 2009;70(suppl 4):1–46,. quiz 47–48. [PubMed] [Google Scholar]