Abstract
Previous studies have suggested that the susceptibility of newborns to infections is linked to the immaturity of their immune system, but very few data are available on the early stages of maturation of the immune response. Therefore, we decided to investigate the evolution of the interferon (IFN)-α and interleukin (IL)-10 responses in neonatal mononuclear cells. To this end, mononuclear cells isolated from cord blood and peripheral blood of 2-, 6- and 18-month-old children and adults were stimulated with unmethylated cytosine-phosphate-guanosine oligodeoxynucleotide (CpG-ODN) 2216 (IFN-α response) or lipopolysaccharide (LPS) (IL-10 response) for 24 h. The production of IFN-α and IL-10 was then measured in culture supernatants using enzyme-linked immunosorbent assay (ELISA) or a 6-plex cytokine array, respectively. Compared to adults, we found a significant impairment in both the IFN-α and IL-10 responses of neonatal mononuclear cells. Interestingly, both responses had increased significantly after 2 months, but remained lower than the adult responses throughout the first 18 months of life. This study shows that although the immune response of neonates tends to mature fairly quickly, it remains different when compared to the adult immune response throughout the first 18 months of life. This could have important consequences on children's ability to mount an appropriate immune response to various challenges and to establish tolerance and immune homeostasis.
Keywords: cord blood, LPS, newborns, oligodeoxynucleotide, peripheral blood mononuclear cell
Introduction
Newborns have been shown to have a higher susceptibility to infection and a lower response to vaccination than adults [1,2]. This is thought to be due to the immaturity of both their innate and adaptive immune system [3,4]. This immaturity could be related to impaired T cell responses [5] and to deficient functions of antigen-presenting cells (APC). Indeed, impaired responses of either myeloid or plasmacytoid dendritic cells (DC), the major inducers of effector T cell responses or tolerance [6], to various Toll-like receptor (TLR) ligands have been described extensively in human newborns [7–14]. However, most studies are performed with whole blood, rather than isolated mononuclear cells, potentially highlighting the effects of factors contained in the blood itself, and are restricted to one or a few time-points in early childhood. In this study, we wanted to investigate the differences between mononuclear cells of neonates and adults and follow the evolution of the immune response over the first 18 months of life.
Interferon (IFN)-α is a cytokine belonging to the type 1 IFN family that is fundamental for the anti-viral responses [15], but may also mediate immunoregulatory functions [16]. Indeed, type 1 IFNs promote a T helper type 1 (Th1)-like immune response with the production of IFN-γ and interleukin (IL)-10 in humans [17–20]. Virtually all nucleated cells are able to produce IFN-α, but among peripheral blood mononuclear cells (PBMC), plasmacytoid DC (pDC) are the major source of IFN-α because these cells are able to produce large amounts of IFN-α within the first 24 h in response to viral DNA and RNA or bacterial DNA [16,18,21–24]. TLRs are involved mainly in the control of innate immunity in response to pathogens. Among these, TLR-3, -7, -8 and -9 mediate immune responses to viral proteins or nucleic acids with the production of type 1 IFN (reviewed in [25]).
Synthetic TLR-9 ligands, such as the unmethylated cytosine-phosphate-guanosine oligodeoxynucleotide (CpG-ODN) 2216, have been shown to trigger in vitro the production of high level of IFN-α by pDC [7,8]. Such TLR-9 ligands are therefore proposed as adjuvants for vaccines against intracellular pathogens, cancer or atopic disorders (reviewed in [26] and [27]).
Interleukin (IL)-10 is a type II cytokine produced by a wide array of immune and non-immune cells (reviewed in [28]), including Th2 cells, regulatory T cells and DCs. Its potent regulatory and anti-inflammatory properties make it a key component of the immune system. Indeed, IL-10 production prevents tissue damage by limiting the inflammatory response [28]. In addition, IL-10 can inhibit both Th1 and Th2 responses and plays an essential role in immune homeostasis [28,29]. Finally, IL-10 contributes to the development of tolerance and is therefore believed to play a role in various disorders, including allergy [29].
In order to shed light on the maturation of the immune response of neonates, we followed the production of IFN-α and IL-10 by mononuclear cells over the first 18 months of life and compared it to adult responses. These results could have an application in vaccination strategy in early life.
Materials and methods
Cells isolation and culture
Human subjects
The present study was approved by the University and Hospital ethical committee and written informed consent was obtained from each individual or parents before blood sampling. Umbilical cord blood samples were collected from healthy full-term neonates. Children were included in a prospective cohort with regular clinical follow-up.
Cord blood mononuclear cells (CBMC)/PBMC isolation
CBMC/PBMC from healthy children/adults were isolated by density gradient centrifugation using Ficoll-Paque (GE Healthcare, Diegem, Belgium) on heparinized blood. CBMC/PBMC were collected, washed three times with Hanks' balanced salt solution (HBSS) (Gibco BRL, Merelbeke, Belgium) and resuspended in RPMI-1640 (GibcoBRL) supplemented with 1% of penicillin/streptomycin (GibcoBRL), 2 mM l-glutamine (GibcoBRL), 1% non-essential amino acids (GibcoBRL) and 10% fetal calf serum (FCS) (AEScientific, Marcq, Belgium). Cell viability estimated by blue trypan exclusion was >95%.
CBMC/PBMC stimulation
To study the IFN-α response, 5 × 105 CBMC/PBMC were cultured for 24 h in a volume of 500 µl in a 48-well plate, in the presence of 20 µg/ml of either unmethylated CpG-ODN (CpG 2216) or non-stimulatory CpG-ODN (CpG-CTRL). The CpG-ODN were purchased from Tib Molbiol (Berlin, Germany): CpG 2216, 5′-ggGGGACGATCGTCgggggG-3′; CpG-CTRL, 5′-TgCTgCTTTTgTgCTTTTgTgCTT-3′. The CpG-ODN concentration used in this assay was selected according to previous dose–response studies performed in a whole-blood model (15; 28). To study the IL-10 response, 5 × 105 CBMC/PBMC were cultured for 24 h, in a volume of 500 µl in a 48-well plate, in the presence or absence of lipopolysaccharide (LPS) (1 µg/ml). After 24 h of stimulation, the supernatants were collected and stored at −20°C until use.
Cytokine quantification
The IFN-α produced in response to CpG stimulation were measured in the culture supernatants by enzyme-linked immunosorbent assay (ELISA) from PBL Biomedical Laboratories (detection limit: 6·8 pg/ml) (Invitrogen, Merelbeke, Belgium) according to the manufacturer's instructions and the data were analysed with a Multiskan EX absorbance reader (Thermo, Aalst, Belgium). IL-10 produced in response to LPS stimulation was measured in the culture supernatants using the a 6-plex bead array (detection limit: 0·45 pg/ml) (Luminex Technology, Biorad Laboratories, Eke, Belgium) according to the manufacturer's instructions and the data were analysed using the Bio-plex manager software. The results, obtained by subtracting the control values from the stimulated values, are expressed in pg/ml.
Statistical analysis
Data were compared using the non-parametric Mann-Whitney unpaired t-test or the non-parametric Wilcoxon's paired t-test. P-values ≤ 0·05 were considered significant; P-values ≤ 0·01 were considered very significant; and P-values ≤ 0·001 were considered highly significant; n.s.: not significant.
Results
Deficiency in the IFN-α response of mononuclear blood cells persists throughout the first 18 months of life
In order to determine the evolution of the neonatal immune response to viral infections, we isolated mononuclear cells from cord blood as well as peripheral blood from 2-, 6- and 18-month-old children and compared their ability to produce IFN-α in response to CpG 2216 to that of adult controls. The clinical characteristics of the cohort are shown in Tables 1 and 2. We found that the response of neonatal mononuclear cells to CpG 2216 was impaired compared to that of adult PBMC (Fig. 1). Indeed, IFN-α production was significantly lower in CBMC than in adult PBMC (6·35 pg/ml ± 1·37 versus 118·93 pg/ml ± 30·89; P ≤ 0·001), and increased at 2 months (44·44 pg/ml ± 7·77) and 6 months (43·27 pg/ml ± 12·32) while remaining statistically lower than adult levels (P ≤ 0·05 and P ≤ 0·01, respectively). IFN-α levels at 18 months were still less than half the adult levels (58·33 pg/ml ± 23·77), but the difference did not reach statistical significance (P = 0·1068), due probably to the smaller number of patients in this group. No correlation was found between IFN-α levels and clinical data regarding the delivery, feeding, early signs of allergy or vaccinations of the children at the time of blood collection (data not shown). In order to determine whether IFN-α was the only CpG-induced response to be affected, we also measured the production of IL-10 or IL-12p70 in the culture supernatants and found no age-related modulation of the response, although most of our values were at the limit of detection of assay both in infants and adult controls (data not shown).
Table 1.
Clinical data of the cohort
| n (%) | Months | |
|---|---|---|
| Sex | ||
| Male | 39 (45%) | |
| Female | 47 (55%) | |
| Delivery | ||
| Midwifery | 72 (84%) | |
| Caesarean | 14 (16%) | |
| Feeding | ||
| Unknown | 6 (7%) | |
| Breast milk | 61 (71%) | |
| Mean duration ± s.d. | 6·2 ± 3·9 | |
| Median duration (range) | 6 (1–18) | |
| Powder milk | 19 (22%) | |
| Cumulative allergic status at 18 months | ||
| Unknown | 16 (19%) | |
| No allergy | 49 (58%) | |
| Allergy | 20 (23%) |
s.d., standard deviation.
Table 2.
Delay between blood test and vaccination
| n (%) | Days | |
|---|---|---|
| 2 months (n = 43/43) | ||
| Not yet vaccinated | 24 (51%) | |
| Already vaccinated | 23 (49%) | |
| Mean ± s.d. | 12 ± 9 | |
| Median (range) | 9 (2–39) | |
| 6 months (n = 35/39) | ||
| Mean ± s.d. | 53 ± 19 | |
| Median (range) | 56 (14–74) | |
| 18 months (n = 12/14) | ||
| Mean ± s.d. | 81 ± 51 | |
| Median (range) | 81 (14–186) |
s.d., standard deviation.
Fig. 1.
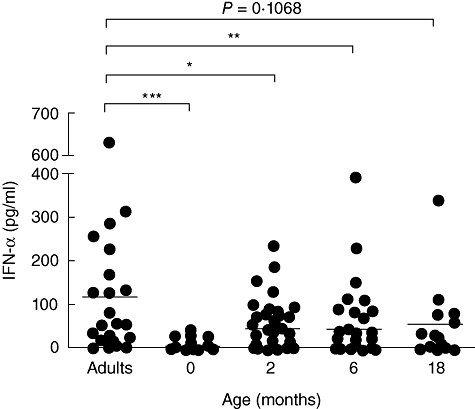
Interferon (IFN)-α production in peripheral blood mononuclear cells from adults and neonates. Cord blood mononuclear cells (n = 47) or peripheral blood mononuclear cells from adults (n = 23), 2-month (n = 47), 6-month (n = 39) or 18-month (n = 14)-old children were cultured in a 48-well plate (106 cells/ml) in the presence of 20 µg/ml of either unmethylated cytosine-phosphate-guanosine-rich oligodeoxynucleotides (CpG-ODN) (CpG 2216) or non-stimulatory CpG-ODN (CpG-CTRL). After 24 h of stimulation, IFN-α production was evaluated in the culture supernatants using enzyme-linked immunosorbent assay. The results, obtained by subtracting the control values from the stimulated values, are expressed in pg/ml. *P-values ≤ 0·05 were considered significant; **P-values ≤ 0·01 were considered very significant; ***P-values ≤ 0·001 were considered highly significant.
To confirm the results and ensure that the differences seen were not due to different patient groups, we focused upon the IFN-α production in 24 children for whom blood samples were available at 0 and 2 months. In those samples, the IFN-α production in response to CpG 2216 was decreased in three of 24 individuals or only maintained in six of 24 individuals at the age of 2 months compared to birth. However, using the non-parametric Wilcoxon's paired t-test, we confirmed a very significant increase of the IFN-α production after 2 months (P ≤ 0·01) (Fig. 2).
Fig. 2.
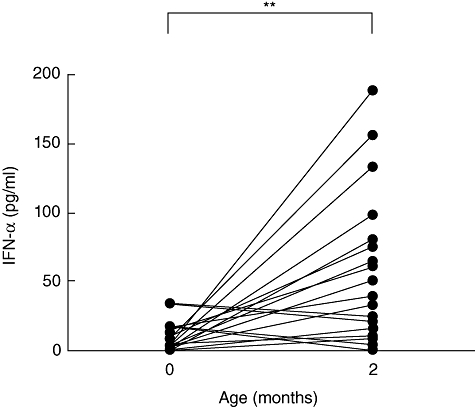
Production of interferon (IFN)-α in children for whom blood was available at 0 and 2 months of age. Cord blood mononuclear cells (n = 24) and related 2-month-old (n = 24) peripheral blood mononuclear cells were cultured in a 48-well plate (106 cells/ml) in the presence of 20 µg/ml of either unmethylated cytosine-phosphate-guanosine-rich oligodeoxynucleotides (CpG-ODN) (CpG 2216) or non-stimulatory CpG-ODN (CpG-CTRL). After 24 h of stimulation, IFN-α production was evaluated in the culture supernatants using enzyme-linked immunosorbent assay. The results, obtained by subtracting the control values from the stimulated values, are expressed in pg/ml. **P-values ≤ 0·01 were considered very significant.
Together, these data demonstrated an impairment of the neonatal IFN-α response to viral products, which persisted from birth up to 18 months of age, despite a significant increase already evident in the first 2 months of life.
IL-10 production remains impaired significantly during the first 18 months of life
In order to determine the evolution of other critical immune responses in neonates, we studied IL-10 production by mononuclear cells in response to LPS stimulation (Fig. 3). We found that neonatal mononuclear cells produced significantly lower levels of IL-10 following LPS stimulation than adult PBMC (124·93 pg/ml ± 16·55 versus 804·41 pg/ml ± 141·57, P ≤ 0·001). In addition, we noted a progressive increase in IL-10 production with time (178·79 pg/ml ± 28·47 at 2 months, 284·97 pg/ml ± 27·24 at 6 months and 212·07 pg/ml ± 127·1 at 18 months), although IL-10 levels remained significantly lower than in adults throughout the first 18 months of life (P ≤ 0·001). Together, these data demonstrated a deficiency in the IL-10 response of neonates, which was not corrected completely during the first 18 months of life.
Fig. 3.
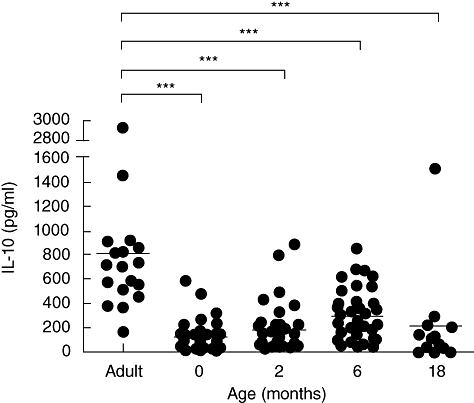
Interleukin (IL)-10 production in peripheral blood mononuclear cells from adults and neonates. Cord blood mononuclear cells (n = 46) or peripheral blood mononuclear cells from adults (n = 18), 2-month (n = 42), 6-month (n = 50) or 18-month (n = 10)-old children were cultured in a 48-well plate (106 cells/ml) in the presence or absence of 1 µg/ml of lipopolysaccharide. After 24 h of stimulation, IL-10 production was evaluated in the culture supernatants using a 6-plex bead array. The results, obtained by subtracting the control values from the stimulated values, are expressed in pg/ml. ***P-values ≤ 0·001 were considered as highly significant.
Discussion
In this study, we have demonstrated that the immune response of neonates is impaired compared to adults throughout the first 18 months of life. Indeed, both the IFN-α response elicited by the TLR-9 ligand, CpG 2216, and the IL-10 response elicited by LPS were reduced significantly in CBMCs when compared to adult PBMCs. Interestingly, both responses had already increased significantly by 2 months of age and continued to increase over the 18-month period, but remained lower than adult levels.
Results obtained in mice and in humans suggested that neonatal T cell-dependent responses are different in early life [5], although mature responses to vaccines or infectious pathogens were also demonstrated during the postnatal period (reviewed in [30]). Indeed, cord blood T cells and mononuclear cells respond normally when stimulated by allogeneic adult DC, suggesting that immature APC functions could be a reason for the defective immune response observed in neonates. In fact, neonatal monocyte-derived DC were shown to produce lower concentrations of the Th1-promoting cytokine, IL-12p70, in response to Bordetella pertusis toxin or CD40 ligation [9,10]. Moreover, various studies report that neonatal myeloid or plasmacytoid DC response to TLR-3 and TLR-4 [10–12,14] or to TLR-9 ligands [7,8] were impaired. The neonatal defect could not be attributed to lower expression of the corresponding TLR or to fewer cell counts in cord blood-derived cells [8,10,31–33]. So far, these observations are still controversial, as Gold et al. [34] found substantial IFN-α levels in neonates compared to adult levels in response to CpG-ODN, although IFN-α levels were significantly lower but not abolished completely in neonatal whole blood or purified pDC. This could be explained partially by the fact that the B cell response after TLR-9 triggering could also influence the pDC and Th1/Th2 T cell responses [35]. In this study, using freshly isolated CBMC or PBMC from healthy newborns or adults, we confirmed that IFN-α production in response to TLR-9 ligand was diminished significantly in neonates, and we demonstrated that this response remained lower than the adult response up to 18 months of age. These results are in line with the report by Nguyen et al. [36], showing that the type I IFN-dependent chemokines interferon gamma-induced protein (IP)-10 and monokine induced by interferon-γ (MIG) levels were lower than the adult levels even at 1 year of age. It is interesting to note that clinical parameters such as delivery, feeding, allergy or vaccination status at the time of blood collection had no influence on the data.
Several studies seem to demonstrate higher IL-10 responses in cord blood compared to adult samples [12,36,37], but it has to be noted that these studies stimulated whole blood samples from cord blood or before 1 month of age and could also reflect the impact of factors present in the blood itself. Indeed, Belderbos et al. have demonstrated that adult PBMC produced higher levels of IL-10 in response to LPS when stimulated in the presence of neonatal plasma than when stimulated in the presence of adult plasma [37]. In this study, we used isolated mononuclear cells to investigate the immune status of the cell population itself. We found that, in contrast to the results obtained with whole blood stimulation, isolated neonatal cord blood mononuclear cells showed a deficient production of IL-10 in response to LPS after 2 months which persisted up to 18 months of age. In addition to the variation in the test samples themselves, the discrepancy between our results and previous reports could be explained by differences in kinetics and in the concentrations of LPS used as well as the stimulation protocol, as we used LPS alone, but some other studies have used LPS in the presence of IFN-γ[37]. Taken together, our results confirm the immaturity of the neonatal immune system by demonstrating a deficiency in both IFN-α and IL-10 responses, which persisted throughout the first 18 months of life, despite a significant amelioration with time. This deficient immune response could be relevant to the understanding of the susceptibility of newborns to infections, and to the use of CpG-ODN as vaccine adjuvant in order to boost immunity of newborns. In addition, this study brings new insight into the ability of the neonate to establish immune tolerance and homeostasis.
Acknowledgments
This study was supported by a grant from the government of the Walloon Region (FIRST 051/6261 – CRISTALL Study), Belgium, Partenariat Public Privé between Université Catholique de Louvain and GSK Biologicals. The research programme on allergy in neonates is supported by the Fondation St Luc pour la recherche médicale. Olivier Vosters was supported by the Fonds Spécial de Recherche, Université Catholique de Louvain. Catherine Lombard is supported by a Post Doctoral grant from the Fonds National de La Recherche Scientifique. Gwenaelle Sana is supported by the FRIA (Fonds pour la Recherche dans l'Industrie et l' Agriculture). Françoise Smets is supported by Fonds National de La Recherche Scientifique. The authors are thankful to the nurses, mothers and children involved in this study. The authors wish to thank Stéphanie Evrard for her excellent technical assistance.
Disclosure
Authors have no disclosures to declare.
References
- 1.Wilson CB, Lewis DB. Basis and implications of selectively diminished cytokine production in neonatal susceptibility to infection. Rev Infect Dis. 1990;12(Suppl. 4):S410–20. doi: 10.1093/clinids/12.supplement_4.s410. [DOI] [PubMed] [Google Scholar]
- 2.Siegrist CA. Vaccination in the neonatal period and early infancy. Int Rev Immunol. 2000;19:195–219. doi: 10.3109/08830180009088505. [DOI] [PubMed] [Google Scholar]
- 3.Adkins B, Leclerc C, Marshall-Clarke S. Neonatal adaptive immunity comes of age. Nat Rev Immunol. 2004;4:553–64. doi: 10.1038/nri1394. [DOI] [PubMed] [Google Scholar]
- 4.Levy O. Innate immunity of the human newborn: distinct cytokine responses to LPS and other Toll-like receptor agonists. J Endotoxin Res. 2005;11:113–16. doi: 10.1179/096805105X37376. [DOI] [PubMed] [Google Scholar]
- 5.Adkins B. T-cell function in newborn mice and humans. Immunol Today. 1999;20:330–5. doi: 10.1016/s0167-5699(99)01473-5. [DOI] [PubMed] [Google Scholar]
- 6.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–52. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 7.Danis B, George TC, Goriely S, et al. Interferon regulatory factor 7-mediated responses are defective in cord blood plasmacytoid dendritic cells. Eur J Immunol. 2008;38:507–17. doi: 10.1002/eji.200737760. [DOI] [PubMed] [Google Scholar]
- 8.De Wit D, Olislagers V, Goriely S, et al. Blood plasmacytoid dendritic cell responses to CpG oligodeoxynucleotides are impaired in human newborns. Blood. 2004;103:1030–2. doi: 10.1182/blood-2003-04-1216. [DOI] [PubMed] [Google Scholar]
- 9.Tonon S, Goriely S, Aksoy E, et al. Bordetella pertussis toxin induces the release of inflammatory cytokines and dendritic cell activation in whole blood: impaired responses in human newborns. Eur J Immunol. 2002;32:3118–25. doi: 10.1002/1521-4141(200211)32:11<3118::AID-IMMU3118>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 10.Goriely S, Vincart B, Stordeur P, et al. Deficient IL-12(p35) gene expression by dendritic cells derived from neonatal monocytes. J Immunol. 2001;166:2141–6. doi: 10.4049/jimmunol.166.3.2141. [DOI] [PubMed] [Google Scholar]
- 11.Goriely S, Van Lint C, Dadkhah R, et al. A defect in nucleosome remodeling prevents IL-12(p35) gene transcription in neonatal dendritic cells. J Exp Med. 2004;199:1011–16. doi: 10.1084/jem.20031272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.De Wit D, Tonon S, Olislagers V, et al. Impaired responses to Toll-like receptor 4 and Toll-like receptor 3 ligands in human cord blood. J Autoimmun. 2003;21:277–81. doi: 10.1016/j.jaut.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 13.Aksoy E, Albarani V, Nguyen M, et al. Interferon regulatory factor 3-dependent responses to lipopolysaccharide are selectively blunted in cord blood cells. Blood. 2007;109:2887–93. doi: 10.1182/blood-2006-06-027862. [DOI] [PubMed] [Google Scholar]
- 14.Upham JW, Lee PT, Holt BJ, et al. Development of interleukin-12-producing capacity throughout childhood. Infect Immun. 2002;70:6583–8. doi: 10.1128/IAI.70.12.6583-6588.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kawai T, Akira S. Innate immune recognition of viral infection. Nat Immunol. 2006;7:131–7. doi: 10.1038/ni1303. [DOI] [PubMed] [Google Scholar]
- 16.Fitzgerald-Bocarsly P, Dai J, Singh S. Plasmacytoid dendritic cells and type I IFN: 50 years of convergent history. Cytokine Growth Factor Rev. 2008;19:3–19. doi: 10.1016/j.cytogfr.2007.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang X, Deriaud E, Jiao X, Braun D, Leclerc C, Lo-Man R. Type I interferons protect neonates from acute inflammation through interleukin 10-producing B cells. J Exp Med. 2007;204:1107–18. doi: 10.1084/jem.20062013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu YJ. IPC: professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. Annu Rev Immunol. 2005;23:275–306. doi: 10.1146/annurev.immunol.23.021704.115633. [DOI] [PubMed] [Google Scholar]
- 19.Farkas L, Kvale EO, Johansen FE, Jahnsen FL, Lund-Johansen F. Plasmacytoid dendritic cells activate allergen-specific TH2 memory cells: modulation by CpG oligodeoxynucleotides. J Allergy Clin Immunol. 2004;114:436–43. doi: 10.1016/j.jaci.2004.04.035. [DOI] [PubMed] [Google Scholar]
- 20.Akbar AN, Lord JM, Salmon M. IFN-alpha and IFN-beta: a link between immune memory and chronic inflammation. Immunol Today. 2000;21:337–42. doi: 10.1016/s0167-5699(00)01652-2. [DOI] [PubMed] [Google Scholar]
- 21.Asselin-Paturel C, Boonstra A, Dalod M, et al. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nat Immunol. 2001;2:1144–50. doi: 10.1038/ni736. [DOI] [PubMed] [Google Scholar]
- 22.Bauer S, Kirschning CJ, Hacker H, et al. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci USA. 2001;98:9237–42. doi: 10.1073/pnas.161293498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kadowaki N, Ho S, Antonenko S, et al. Subsets of human dendritic cell precursors express different Toll-like receptors and respond to different microbial antigens. J Exp Med. 2001;194:863–9. doi: 10.1084/jem.194.6.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Siegal FP, Kadowaki N, Shodell M, et al. The nature of the principal type 1 interferon-producing cells in human blood. Science. 1999;284:1835–7. doi: 10.1126/science.284.5421.1835. [DOI] [PubMed] [Google Scholar]
- 25.Bowie AG. Translational mini-review series on Toll-like receptors: recent advances in understanding the role of Toll-like receptors in anti-viral immunity. Clin Exp Immunol. 2007;147:217–26. doi: 10.1111/j.1365-2249.2006.03301.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Klinman DM. Immunotherapeutic uses of CpG oligodeoxynucleotides. Nat Rev Immunol. 2004;4:249–58. doi: 10.1038/nri1329. [DOI] [PubMed] [Google Scholar]
- 27.Goldman M. Translational mini-review series on Toll-like receptors: Toll-like receptor ligands as novel pharmaceuticals for allergic disorders. Clin Exp Immunol. 2007;147:208–16. doi: 10.1111/j.1365-2249.2006.03296.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mosser DM, Zhang X. Interleukin-10: new perspectives on an old cytokine. Immunol Rev. 2008;226:205–18. doi: 10.1111/j.1600-065X.2008.00706.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Commins S, Steinke JW, Borish L. The extended IL-10 superfamily: IL-10, IL-19, IL-20, IL-22, IL-24, IL-26, IL-28, and IL-29. J Allergy Clin Immunol. 2008;121:1108–11. doi: 10.1016/j.jaci.2008.02.026. [DOI] [PubMed] [Google Scholar]
- 30.Marchant A, Goldman M. T cell-mediated immune responses in human newborns: ready to learn? Clin Exp Immunol. 2005;141:10–18. doi: 10.1111/j.1365-2249.2005.02799.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Holt PG, Upham JW, Sly PD. Contemporaneous maturation of immunologic and respiratory functions during early childhood: implications for development of asthma prevention strategies. J Allergy Clin Immunol. 2005;116:16–24. doi: 10.1016/j.jaci.2005.04.017. quiz 5. [DOI] [PubMed] [Google Scholar]
- 32.Teig N, Moses D, Gieseler S, Schauer U. Age-related changes in human blood dendritic cell subpopulations. Scand J Immunol. 2002;55:453–7. doi: 10.1046/j.1365-3083.2002.01068.x. [DOI] [PubMed] [Google Scholar]
- 33.de Vries E, de Bruin-Versteeg S, Comans-Bitter WM, et al. Longitudinal survey of lymphocyte subpopulations in the first year of life. Pediatr Res. 2000;47:528–37. doi: 10.1203/00006450-200004000-00019. [DOI] [PubMed] [Google Scholar]
- 34.Gold MC, Donnelly E, Cook MS, Leclair CM, Lewinsohn DA. Purified neonatal plasmacytoid dendritic cells overcome intrinsic maturation defect with TLR agonist stimulation. Pediatr Res. 2006;60:34–7. doi: 10.1203/01.pdr.0000220352.13547.f4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun CM, Deriaud E, Leclerc C, Lo-Man R. Upon TLR9 signaling, CD5+ B cells control the IL-12-dependent Th1-priming capacity of neonatal DCs. Immunity. 2005;22:467–77. doi: 10.1016/j.immuni.2005.02.008. [DOI] [PubMed] [Google Scholar]
- 36.Nguyen M, Leuridan E, Zhang T, et al. Acquisition of adult-like TLR4 and TLR9 responses during the first year of life. PLoS ONE. 2010;5:e10407. doi: 10.1371/journal.pone.0010407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Belderbos ME, van Bleek GM, Levy O, et al. Skewed pattern of Toll-like receptor 4-mediated cytokine production in human neonatal blood: low LPS-induced IL-12p70 and high IL-10 persist throughout the first month of life. Clin Immunol. 2009;133:228–37. doi: 10.1016/j.clim.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]


