Abstract
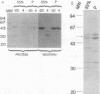
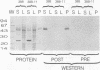
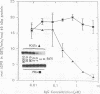
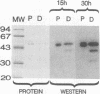
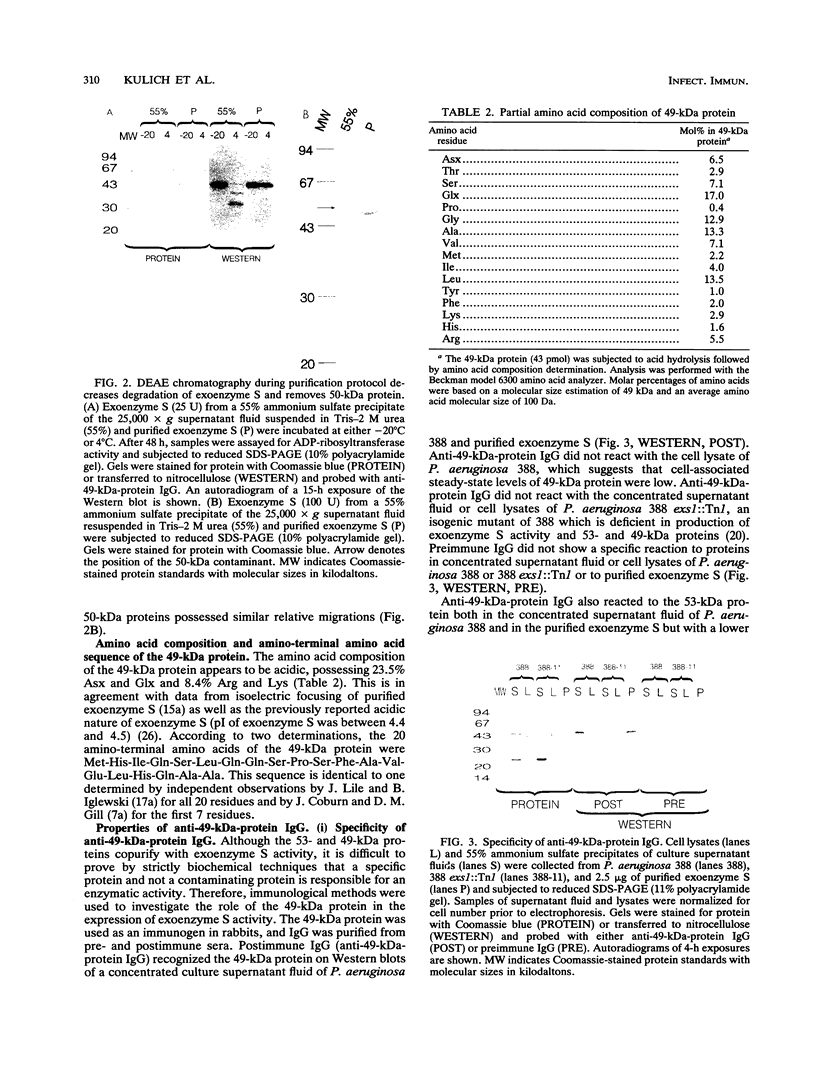
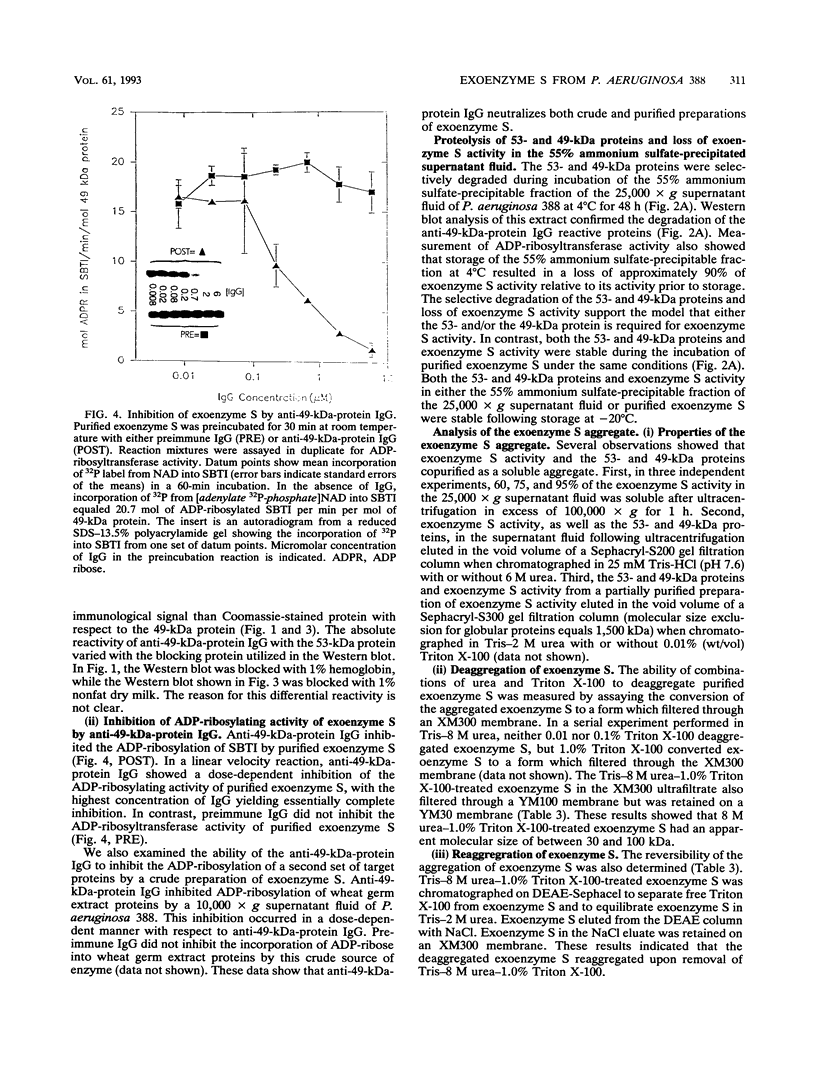
Exoenzyme S was purified > 1,500-fold from the culture supernatant fluid of Pseudomonas aeruginosa 388 at high yield without utilization of solvents or detergents. Two proteins, with apparent molecular sizes of 53 and 49 kDa, cofractionated with exoenzyme S activity. Rabbit anti-49-kDa-protein immunoglobulin G was prepared by using sodium dodecyl sulfate-polyacrylamide gel electrophoresis-purified 49-kDa protein as immunogen. Anti-49-kDa-protein IgG inhibited the ADP-ribosyltransferase activity of purified exoenzyme S in a dose-dependent manner, which indicated a role for the 49-kDa protein in the ADP-ribosylation reaction. Analysis by ultrafiltration showed that exoenzyme S activity and the 53- and 49-kDa proteins cofractionated and that exoenzyme S was apparently > 300 kDa in size. Urea (8 M) and 1.0% Triton X-100 reversibly decreased the apparent molecular sizes of exoenzyme S activity and the 53- and 49-kDa proteins to between 30 and 100 kDa.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barbieri J. T., Moloney B. K., Mende-Mueller L. M. Expression and secretion of the S-1 subunit and C180 peptide of pertussis toxin in Escherichia coli. J Bacteriol. 1989 Aug;171(8):4362–4369. doi: 10.1128/jb.171.8.4362-4369.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodey G. P., Bolivar R., Fainstein V., Jadeja L. Infections caused by Pseudomonas aeruginosa. Rev Infect Dis. 1983 Mar-Apr;5(2):279–313. doi: 10.1093/clinids/5.2.279. [DOI] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Chung D. W., Collier R. J. Enzymatically active peptide from the adenosine diphosphate-ribosylating toxin of Pseudomonas aeruginosa. Infect Immun. 1977 Jun;16(3):832–841. doi: 10.1128/iai.16.3.832-841.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coburn J., Dillon S. T., Iglewski B. H., Gill D. M. Exoenzyme S of Pseudomonas aeruginosa ADP-ribosylates the intermediate filament protein vimentin. Infect Immun. 1989 Mar;57(3):996–998. doi: 10.1128/iai.57.3.996-998.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coburn J., Kane A. V., Feig L., Gill D. M. Pseudomonas aeruginosa exoenzyme S requires a eukaryotic protein for ADP-ribosyltransferase activity. J Biol Chem. 1991 Apr 5;266(10):6438–6446. [PubMed] [Google Scholar]
- Coburn J. Pseudomonas aeruginosa exoenzyme S. Curr Top Microbiol Immunol. 1992;175:133–143. doi: 10.1007/978-3-642-76966-5_7. [DOI] [PubMed] [Google Scholar]
- Coburn J., Wyatt R. T., Iglewski B. H., Gill D. M. Several GTP-binding proteins, including p21c-H-ras, are preferred substrates of Pseudomonas aeruginosa exoenzyme S. J Biol Chem. 1989 May 25;264(15):9004–9008. [PubMed] [Google Scholar]
- Cover T. L., Blaser M. J. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J Biol Chem. 1992 May 25;267(15):10570–10575. [PubMed] [Google Scholar]
- Gill D. M., Coburn J. ADP-ribosylation by cholera toxin: functional analysis of a cellular system that stimulates the enzymic activity of cholera toxin fragment A1. Biochemistry. 1987 Oct 6;26(20):6364–6371. doi: 10.1021/bi00394a009. [DOI] [PubMed] [Google Scholar]
- Iglewski B. H., Sadoff J., Bjorn M. J., Maxwell E. S. Pseudomonas aeruginosa exoenzyme S: an adenosine diphosphate ribosyltransferase distinct from toxin A. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3211–3215. doi: 10.1073/pnas.75.7.3211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn R. A., Gilman A. G. The protein cofactor necessary for ADP-ribosylation of Gs by cholera toxin is itself a GTP binding protein. J Biol Chem. 1986 Jun 15;261(17):7906–7911. [PubMed] [Google Scholar]
- Knudsen K. A. Proteins transferred to nitrocellulose for use as immunogens. Anal Biochem. 1985 Jun;147(2):285–288. doi: 10.1016/0003-2697(85)90273-8. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leunk R. D., Johnson P. T., David B. C., Kraft W. G., Morgan D. R. Cytotoxic activity in broth-culture filtrates of Campylobacter pylori. J Med Microbiol. 1988 Jun;26(2):93–99. doi: 10.1099/00222615-26-2-93. [DOI] [PubMed] [Google Scholar]
- Nicas T. I., Bradley J., Lochner J. E., Iglewski B. H. The role of exoenzyme S in infections with Pseudomonas aeruginosa. J Infect Dis. 1985 Oct;152(4):716–721. doi: 10.1093/infdis/152.4.716. [DOI] [PubMed] [Google Scholar]
- Nicas T. I., Frank D. W., Stenzel P., Lile J. D., Iglewski B. H. Role of exoenzyme S in chronic Pseudomonas aeruginosa lung infections. Eur J Clin Microbiol. 1985 Apr;4(2):175–179. doi: 10.1007/BF02013593. [DOI] [PubMed] [Google Scholar]
- Nicas T. I., Iglewski B. H. Contribution of exoenzyme S to the virulence of Pseudomonas aeruginosa. Antibiot Chemother (1971) 1985;36:40–48. doi: 10.1159/000410470. [DOI] [PubMed] [Google Scholar]
- Nicas T. I., Iglewski B. H. Isolation and characterization of transposon-induced mutants of Pseudomonas aeruginosa deficient in production of exoenzyme S. Infect Immun. 1984 Aug;45(2):470–474. doi: 10.1128/iai.45.2.470-474.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicas T. I., Iglewski B. H. The contribution of exoproducts to virulence of Pseudomonas aeruginosa. Can J Microbiol. 1985 Apr;31(4):387–392. doi: 10.1139/m85-074. [DOI] [PubMed] [Google Scholar]
- Roth J. Methods for assessing immunologic and biologic properties of iodinated peptide hormones. Methods Enzymol. 1975;37:223–233. doi: 10.1016/s0076-6879(75)37018-3. [DOI] [PubMed] [Google Scholar]
- Sokol P. A., Dennis J. J., MacDougall P. C., Sexton M., Woods D. E. Cloning and expression of the Pseudomonas aeruginosa exoenzyme S toxin gene. Microb Pathog. 1990 Apr;8(4):243–257. doi: 10.1016/0882-4010(90)90051-q. [DOI] [PubMed] [Google Scholar]
- Stevenson G. T., Dorrington K. J. The recombination of dimers of immunoglobulin peptide chains. Biochem J. 1970 Aug;118(5):703–712. doi: 10.1042/bj1180703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Woods D. E., Hwang W. S., Shahrabadi M. S., Que J. U. Alteration of pulmonary structure by Pseudomonas aeruginosa exoenzyme S. J Med Microbiol. 1988 Jun;26(2):133–141. doi: 10.1099/00222615-26-2-133. [DOI] [PubMed] [Google Scholar]
- Woods D. E., Que J. U. Purification of Pseudomonas aeruginosa exoenzyme S. Infect Immun. 1987 Mar;55(3):579–586. doi: 10.1128/iai.55.3.579-586.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods D. E., Sokol P. A. Use of transposon mutants to assess the role of exoenzyme S in chronic pulmonary disease due to Pseudomonas aeruginosa. Eur J Clin Microbiol. 1985 Apr;4(2):163–169. doi: 10.1007/BF02013591. [DOI] [PubMed] [Google Scholar]