Abstract
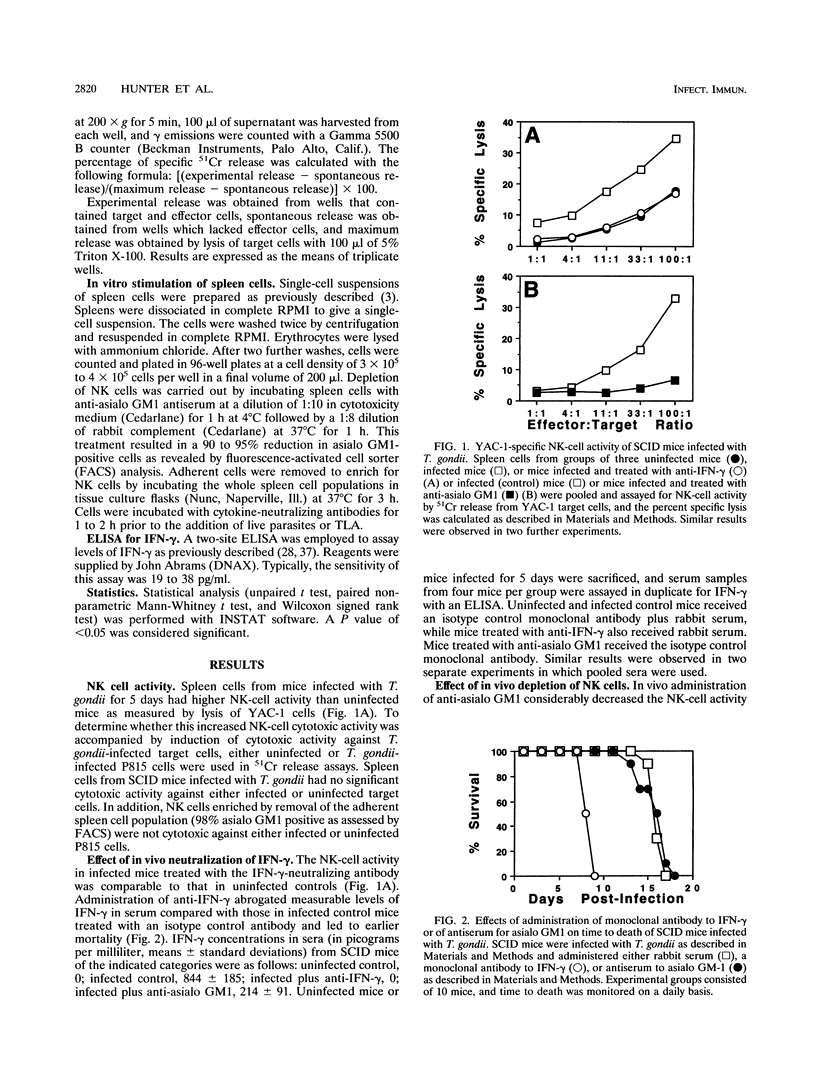
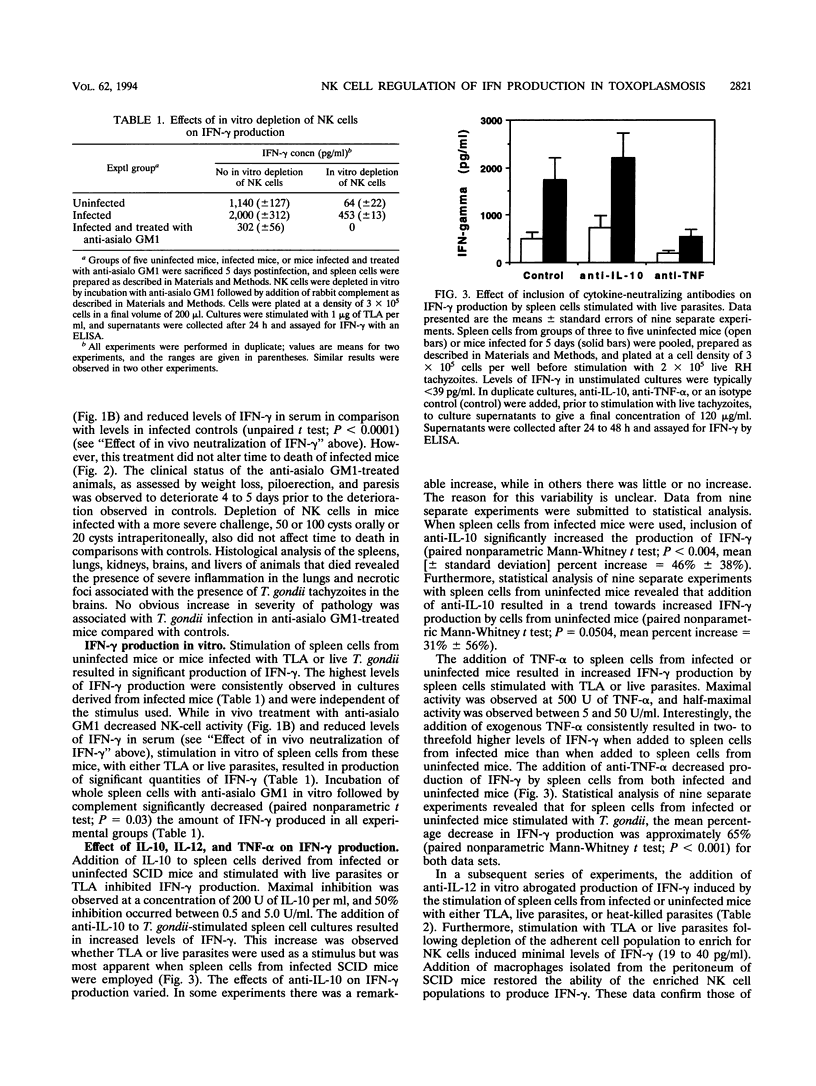
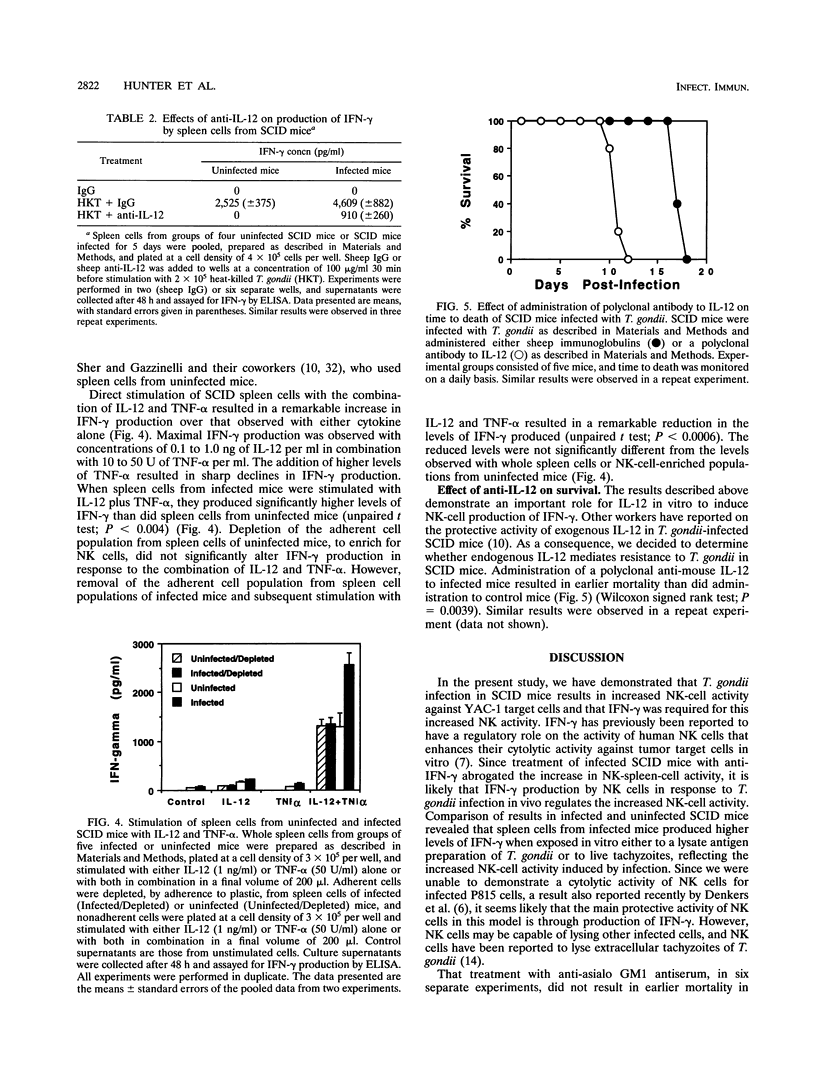
Previous studies of mice have implicated natural killer (NK) cells as mediators of protective activity against Toxoplasma gondii through their production of gamma interferon (IFN-gamma). In the present study, we have compared NK-cell activity in infected and uninfected SCID mice. Our data reveal that infection results in increased levels of IFN-gamma in serum and elevated NK-cell activity but that these NK cells were not cytotoxic for T. gondii-infected P815 cells. Treatment with anti-IFN-gamma antibody abrogated the increase in NK-cell activity and resulted in earlier mortality of infected mice. In vivo treatment with anti-asialo GM1 antiserum reduced NK cell activity and levels of IFN-gamma in serum but did not alter time to death. Spleen cells from infected mice produced higher levels of IFN-gamma than those from uninfected mice when stimulated in vitro with live T. gondii or parasite antigen preparations. Further analysis revealed that interleukin 10 (IL-10) inhibited, whereas tumor necrosis factor alpha (TNF-alpha) and IL-12 enhanced, IFN-gamma production by spleen cells from infected or uninfected mice. The combination of IL-12 and TNF-alpha induced higher levels of IFN-gamma from whole spleen cells of infected mice than from those of uninfected mice. Depletion of the adherent cell population from the spleen cells of infected mice led to a significant reduction in the levels of IFN-gamma produced after stimulation with IL-12 plus TNF-alpha. Similar results did not occur with cells from uninfected mice. These data indicate that other cytokines produced by the adherent cell population from infected mice may be involved in maximal production of IFN-gamma by NK cells stimulated with IL-12 and TNF-alpha. To assess the importance of endogenous IL-12, a polyclonal anti-IL-12 was administered to infected SCID mice. This treatment led to earlier mortality, indicating that endogenous IL-12 mediates resistance to T. gondii.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bancroft G. J., Schreiber R. D., Bosma G. C., Bosma M. J., Unanue E. R. A T cell-independent mechanism of macrophage activation by interferon-gamma. J Immunol. 1987 Aug 15;139(4):1104–1107. [PubMed] [Google Scholar]
- Bancroft G. J., Schreiber R. D., Unanue E. R. Natural immunity: a T-cell-independent pathway of macrophage activation, defined in the scid mouse. Immunol Rev. 1991 Dec;124:5–24. doi: 10.1111/j.1600-065x.1991.tb00613.x. [DOI] [PubMed] [Google Scholar]
- Brinkmann V., Sharma S. D., Remington J. S. Different regulation of the L3T4-T cell subset by B cells in different mouse strains bearing the H-2k haplotype. J Immunol. 1986 Nov 1;137(9):2991–2997. [PubMed] [Google Scholar]
- Chan S. H., Perussia B., Gupta J. W., Kobayashi M., Pospísil M., Young H. A., Wolf S. F., Young D., Clark S. C., Trinchieri G. Induction of interferon gamma production by natural killer cell stimulatory factor: characterization of the responder cells and synergy with other inducers. J Exp Med. 1991 Apr 1;173(4):869–879. doi: 10.1084/jem.173.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherwinski H. M., Schumacher J. H., Brown K. D., Mosmann T. R. Two types of mouse helper T cell clone. III. Further differences in lymphokine synthesis between Th1 and Th2 clones revealed by RNA hybridization, functionally monospecific bioassays, and monoclonal antibodies. J Exp Med. 1987 Nov 1;166(5):1229–1244. doi: 10.1084/jem.166.5.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Djeu J. Y., Stocks N., Zoon K., Stanton G. J., Timonen T., Herberman R. B. Positive self regulation of cytotoxicity in human natural killer cells by production of interferon upon exposure to influenza and herpes viruses. J Exp Med. 1982 Oct 1;156(4):1222–1234. doi: 10.1084/jem.156.4.1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiorentino D. F., Zlotnik A., Mosmann T. R., Howard M., O'Garra A. IL-10 inhibits cytokine production by activated macrophages. J Immunol. 1991 Dec 1;147(11):3815–3822. [PubMed] [Google Scholar]
- Gazzinelli R. T., Hakim F. T., Hieny S., Shearer G. M., Sher A. Synergistic role of CD4+ and CD8+ T lymphocytes in IFN-gamma production and protective immunity induced by an attenuated Toxoplasma gondii vaccine. J Immunol. 1991 Jan 1;146(1):286–292. [PubMed] [Google Scholar]
- Hakim F. T., Gazzinelli R. T., Denkers E., Hieny S., Shearer G. M., Sher A. CD8+ T cells from mice vaccinated against Toxoplasma gondii are cytotoxic for parasite-infected or antigen-pulsed host cells. J Immunol. 1991 Oct 1;147(7):2310–2316. [PubMed] [Google Scholar]
- Hauser W. E., Jr, Sharma S. D., Remington J. S. Augmentation of NK cell activity by soluble and particulate fractions of Toxoplasma gondii. J Immunol. 1983 Jul;131(1):458–463. [PubMed] [Google Scholar]
- Hauser W. E., Jr, Sharma S. D., Remington J. S. Natural killer cells induced by acute and chronic toxoplasma infection. Cell Immunol. 1982 May 15;69(2):330–346. doi: 10.1016/0008-8749(82)90076-4. [DOI] [PubMed] [Google Scholar]
- Hauser W. E., Jr, Tsai V. Acute toxoplasma infection of mice induces spleen NK cells that are cytotoxic for T. gondii in vitro. J Immunol. 1986 Jan;136(1):313–319. [PubMed] [Google Scholar]
- Heinzel F. P., Schoenhaut D. S., Rerko R. M., Rosser L. E., Gately M. K. Recombinant interleukin 12 cures mice infected with Leishmania major. J Exp Med. 1993 May 1;177(5):1505–1509. doi: 10.1084/jem.177.5.1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard M., O'Garra A., Ishida H., de Waal Malefyt R., de Vries J. Biological properties of interleukin 10. J Clin Immunol. 1992 Jul;12(4):239–247. doi: 10.1007/BF00918147. [DOI] [PubMed] [Google Scholar]
- Hsieh C. S., Macatonia S. E., Tripp C. S., Wolf S. F., O'Garra A., Murphy K. M. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science. 1993 Apr 23;260(5107):547–549. doi: 10.1126/science.8097338. [DOI] [PubMed] [Google Scholar]
- Hsu D. H., Moore K. W., Spits H. Differential effects of IL-4 and IL-10 on IL-2-induced IFN-gamma synthesis and lymphokine-activated killer activity. Int Immunol. 1992 May;4(5):563–569. doi: 10.1093/intimm/4.5.563. [DOI] [PubMed] [Google Scholar]
- Hunter C. A., Abrams J. S., Beaman M. H., Remington J. S. Cytokine mRNA in the central nervous system of SCID mice infected with Toxoplasma gondii: importance of T-cell-independent regulation of resistance to T. gondii. Infect Immun. 1993 Oct;61(10):4038–4044. doi: 10.1128/iai.61.10.4038-4044.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huskinson-Mark J., Araujo F. G., Remington J. S. Evaluation of the effect of drugs on the cyst form of Toxoplasma gondii. J Infect Dis. 1991 Jul;164(1):170–171. doi: 10.1093/infdis/164.1.170. [DOI] [PubMed] [Google Scholar]
- Johnson L. L. SCID mouse models of acute and relapsing chronic Toxoplasma gondii infections. Infect Immun. 1992 Sep;60(9):3719–3724. doi: 10.1128/iai.60.9.3719-3724.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi M., Fitz L., Ryan M., Hewick R. M., Clark S. C., Chan S., Loudon R., Sherman F., Perussia B., Trinchieri G. Identification and purification of natural killer cell stimulatory factor (NKSF), a cytokine with multiple biologic effects on human lymphocytes. J Exp Med. 1989 Sep 1;170(3):827–845. doi: 10.1084/jem.170.3.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langermans J. A., Van der Hulst M. E., Nibbering P. H., Hiemstra P. S., Fransen L., Van Furth R. IFN-gamma-induced L-arginine-dependent toxoplasmastatic activity in murine peritoneal macrophages is mediated by endogenous tumor necrosis factor-alpha. J Immunol. 1992 Jan 15;148(2):568–574. [PubMed] [Google Scholar]
- Parker S. J., Roberts C. W., Alexander J. CD8+ T cells are the major lymphocyte subpopulation involved in the protective immune response to Toxoplasma gondii in mice. Clin Exp Immunol. 1991 May;84(2):207–212. doi: 10.1111/j.1365-2249.1991.tb08150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perussia B., Chan S. H., D'Andrea A., Tsuji K., Santoli D., Pospisil M., Young D., Wolf S. F., Trinchieri G. Natural killer (NK) cell stimulatory factor or IL-12 has differential effects on the proliferation of TCR-alpha beta+, TCR-gamma delta+ T lymphocytes, and NK cells. J Immunol. 1992 Dec 1;149(11):3495–3502. [PubMed] [Google Scholar]
- Romagnani S. Induction of TH1 and TH2 responses: a key role for the 'natural' immune response? Immunol Today. 1992 Oct;13(10):379–381. doi: 10.1016/0167-5699(92)90083-J. [DOI] [PubMed] [Google Scholar]
- Sander B., Höidén I., Andersson U., Möller E., Abrams J. S. Similar frequencies and kinetics of cytokine producing cells in murine peripheral blood and spleen. Cytokine detection by immunoassay and intracellular immunostaining. J Immunol Methods. 1993 Dec 3;166(2):201–214. doi: 10.1016/0022-1759(93)90361-a. [DOI] [PubMed] [Google Scholar]
- Schlüter D., Deckert-Schlüter M., Schwendemann G., Brunner H., Hof H. Expression of major histocompatibility complex class II antigens and levels of interferon-gamma, tumour necrosis factor, and interleukin-6 in cerebrospinal fluid and serum in Toxoplasma gondii-infected SCID and immunocompetent C.B-17 mice. Immunology. 1993 Mar;78(3):430–435. [PMC free article] [PubMed] [Google Scholar]
- Sharma S. D., Mullenax J., Araujo F. G., Erlich H. A., Remington J. S. Western Blot analysis of the antigens of Toxoplasma gondii recognized by human IgM and IgG antibodies. J Immunol. 1983 Aug;131(2):977–983. [PubMed] [Google Scholar]
- Sharma S. D., Verhoef J., Remington J. S. Enhancement of human natural killer cell activity by subcellular components of Toxoplasma gondii. Cell Immunol. 1984 Jul;86(2):317–326. doi: 10.1016/0008-8749(84)90386-1. [DOI] [PubMed] [Google Scholar]
- Sher A., Oswald I. P., Hieny S., Gazzinelli R. T. Toxoplasma gondii induces a T-independent IFN-gamma response in natural killer cells that requires both adherent accessory cells and tumor necrosis factor-alpha. J Immunol. 1993 May 1;150(9):3982–3989. [PubMed] [Google Scholar]
- Sibley L. D., Adams L. B., Fukutomi Y., Krahenbuhl J. L. Tumor necrosis factor-alpha triggers antitoxoplasmal activity of IFN-gamma primed macrophages. J Immunol. 1991 Oct 1;147(7):2340–2345. [PubMed] [Google Scholar]
- Starnes H. F., Jr, Pearce M. K., Tewari A., Yim J. H., Zou J. C., Abrams J. S. Anti-IL-6 monoclonal antibodies protect against lethal Escherichia coli infection and lethal tumor necrosis factor-alpha challenge in mice. J Immunol. 1990 Dec 15;145(12):4185–4191. [PubMed] [Google Scholar]
- Subauste C. S., Dawson L., Remington J. S. Human lymphokine-activated killer cells are cytotoxic against cells infected with Toxoplasma gondii. J Exp Med. 1992 Dec 1;176(6):1511–1519. doi: 10.1084/jem.176.6.1511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subauste C. S., Koniaris A. H., Remington J. S. Murine CD8+ cytotoxic T lymphocytes lyse Toxoplasma gondii-infected cells. J Immunol. 1991 Dec 1;147(11):3955–3959. [PubMed] [Google Scholar]
- Suzuki Y., Orellana M. A., Wong S. Y., Conley F. K., Remington J. S. Susceptibility to chronic infection with Toxoplasma gondii does not correlate with susceptibility to acute infection in mice. Infect Immun. 1993 Jun;61(6):2284–2288. doi: 10.1128/iai.61.6.2284-2288.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki Y., Remington J. S. Dual regulation of resistance against Toxoplasma gondii infection by Lyt-2+ and Lyt-1+, L3T4+ T cells in mice. J Immunol. 1988 Jun 1;140(11):3943–3946. [PubMed] [Google Scholar]
- Tripp C. S., Wolf S. F., Unanue E. R. Interleukin 12 and tumor necrosis factor alpha are costimulators of interferon gamma production by natural killer cells in severe combined immunodeficiency mice with listeriosis, and interleukin 10 is a physiologic antagonist. Proc Natl Acad Sci U S A. 1993 Apr 15;90(8):3725–3729. doi: 10.1073/pnas.90.8.3725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry J. C., Schreiber R. D., Unanue E. R. Regulation of gamma interferon production by natural killer cells in scid mice: roles of tumor necrosis factor and bacterial stimuli. Infect Immun. 1991 May;59(5):1709–1715. doi: 10.1128/iai.59.5.1709-1715.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong S. Y., Remington J. S. Biology of Toxoplasma gondii. AIDS. 1993 Mar;7(3):299–316. doi: 10.1097/00002030-199303000-00001. [DOI] [PubMed] [Google Scholar]



