Abstract
Ischaemic tissue damage represents the ultimate form of tissue pathophysiology due to cardiovascular disease, which is the leading cause of morbidity and mortality across the globe. A significant amount of basic research and clinical investigation has been focused on identifying cellular and molecular pathways to alleviate tissue damage and dysfunction due to ischaemia and subsequent reperfusion. Over many years, the gaseous molecule nitric oxide (NO) has emerged as an important regulator of cardiovascular health as well as protector against tissue ischaemia and reperfusion injury. However, clinical translation of NO therapy for these pathophysiological conditions has not been realized for various reasons. Work from our laboratory and several others suggests that a new form of NO-associated therapy may be possible through the use of nitrite anion (sodium nitrite), a prodrug which can be reduced to NO in ischaemic tissues. In this manner, nitrite anion serves as a highly selective NO donor in ischaemic tissues without substantially altering otherwise normal tissue. This surprising and novel discovery has reinvigorated hopes for effectively restoring NO bioavailability in vulnerable tissues while continuing to reveal the complexity of NO biology and metabolism within the cardiovascular system. However, some concerns may exist regarding the effect of nitrite on carcinogenesis. This review highlights the emergence of nitrite anion as a selective NO prodrug for ischaemic tissue disorders and discusses the potential therapeutic utility of this agent for peripheral vascular disease.
Keywords: Angiogenesis, Arteriogenesis, Nitric oxide, Peripheral arterial disease
1. Introduction
Cardiovascular disease (CVD) is now entrenched as the foremost burdensome pathophysiological condition throughout the globe. Knowledge and treatment of CVD have been refined over the past several decades resulting in significant new advances for therapeutic interventions. Although lipid-lowering drugs and anti-thrombotic agents have revolutionized how CVD is treated, these and other agents do not completely rectify all pathological cellular responses during disease, indicating the need for different approaches to CVD management. In particular, the gaseous signalling molecule nitric oxide (NO) has been well characterized as a critical mediator of cardiovascular health that is often dysfunctional or lost during various disease states. Restoration of NO production and bioavailability has long been a desire for both researchers and clinicians, so that one day pharmacological agents could replete NO levels in diseased cardiovascular tissue. Targeted, efficacious NO therapy has been difficult to achieve due to many issues such as the volatile nature of NO itself and its chemical congers, the inability to achieve selective tissue delivery which can lead to undesirable side effects, and the controversial nature of NO biological functions in either beneficial or harmful ways, which could result in undesirable side effects. Fortunately, there may still be hope for targeted, effective, and minimally damaging NO-based therapy. Over the past several years, numerous laboratories have discovered that the nitrite anion serves as a prodrug for NO formation through a one-electron reduction reaction that occurs in various ways, thus providing a novel way to deliver NO bioequivalents that are beneficial for CVD.
2. NO biology and cardiovascular pathophysiology
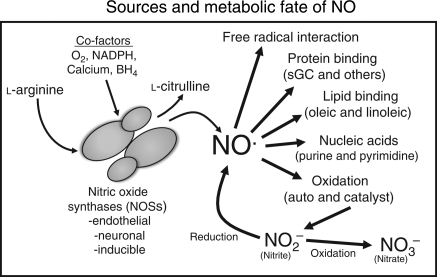
In order to appreciate the potential therapeutic usefulness of nitrite anion, it is important to understand NO metabolism and function in cardiovascular health and disease. Seminal studies from different laboratories identified the gaseous free radical NO to be the elusive EDRF (endothelial-derived relaxing factor) which is central in regulating vascular tone and was the basis of the 1998 Nobel Prize in Physiology or Medicine.1,2 NO is important for a diverse array of molecular, cellular, and physiological responses including regulation of vascular inflammation and platelet adhesion, governing vascular growth and maturation, and modulation of cellular respiration, survival, and proliferation.1,3 NO reactivity in biological systems was initially described by reactions with metallo (haem) proteins and other free radical species, the classical example being NO-dependent activation of the haem protein-soluble guanylate cyclase (sGC) leading to activation of signalling cascades stimulating vasodilation.1,2,4 Yet, numerous studies have shown NO interacts with many other biological targets including proteins, lipids, and nucleic acids (Figure 1).5–8 NO also serves as an effective redox modulator through its direct ability to act as an antioxidant or by induction of antioxidant defences.9–11 Enzymatic processes largely accomplish NO generation, which results in both paracrine and endocrine effects of NO. Enzymatic NO formation occurs through sequential hydroxylation of l-arginine to N-hydroxy-l-arginine and then subsequent oxidation to l-citrulline and NO by different NO synthases (NOSs), namely neuronal NOS (NOS1), inducible NOS (NOS2), and endothelial NOS (NOS3). NOS enzyme function is regulated by a combination of variables including substrate (l-arginine) and co-factor (e.g. O2, NADPH, BH4, calcium–calmodulin, and other binding proteins) bioavailability along with various post-translational modifications of the enzyme such as phosphorylation, myristoylation, and S-nitrosylation.12,13 Importantly, tissue ischaemia results in perturbation of substrate and cofactor bioavailability leading to diminished NOS-dependent NO formation.
Figure 1.
Sources and metabolic fate of nitric oxide. Diagram illustrating substrate and cofactors required for nitric oxide formation along with end target reaction molecules and salvage pathways.
Studies over the past 20 years have revealed that NO influences a multitude of intracellular responses including signal transduction, gene expression, and cell viability and proliferation through various mechanisms. It has been reported that these responses are significantly influenced by local amounts of NO that are generated or bioavailable.14 For example, low (nM) concentrations of NO have been reported to increase signal activation of ERK and Akt/PKB as well as HIF-1α which contribute to cell survival; whereas, higher (µM) concentrations may elicit nitrosative stress through protein modification that can lead to cell damage or death.10,15 In this way, NO has been reported to be an important cytoprotective agent against cardiovascular ischaemia and reperfusion injury through multiple mechanisms, including limitation of inflammation, thrombosis, prevention of apoptosis, and changes in intracellular redox status.16,17 NO is also an important mediator of physiological and therapeutic angiogenesis.18–20 The importance of NO in regulating angiogenic activity is attributed to the fact that VEGF-dependent angiogenesis involves signalling responses leading to NO production.21,22 Subsequent NO production can then activate other signalling pathways including sGC, PI3K, AKT, ERK 1/2, PKC, tyrosine kinases, and transcription factor activation to augment endothelial cell proliferation and survival.23–26 Genetic deficiency of endothelial nitric oxide synthase (eNOS), the predominant enzyme responsible for producing NO in the cardiovascular system, blunts angiogenesis, increases ischaemia-reperfusion injury, and enhances vascular inflammatory responses signifying that eNOS-derived NO is central in regulating cardiovascular homeostatsis, remodelling, and growth.27–32 Studies have also revealed that discrete intracellular localization of eNOS and subsequent NO generation play important roles in modulating various signalling and cellular responses.33–37 Together, these reports clearly delineate the importance and complexity of NO effects in cardiovascular tissues and its generation by eNOS.
A consistent feature of CVD is dysfunction of NO-mediated signalling processes and physiological responses. Indeed, loss of NO bioavailability has been reported in patients and animal models of CVD.38 This can result from defective NOS enzyme function and/or diversion of NO away from ‘normal’ targets to reactions with reactive oxygen species (e.g. superoxide anion radical) leading to the formation of secondary reactive nitrogen species (e.g. peroxynitrite) which can exacerbate tissue pathology.10,39 Restoring NO bioavailability during CVD represents an important therapeutic goal, which has not been realized largely due to the inability of site-specific delivery and undesired side effects of previous NO-donor compounds.
3. Nitrite biology and conversion to NO
Early studies examining physiological effects of nitrite found that high micromolar concentrations of nitrite could stimulate vasodilation; however, the fact that normal plasma concentrations of nitrite are much lower (nM) lead to the idea that nitrite was not involved in regulating vascular tone.40 Moreover, physiological concentrations of nitrite were not found to stimulate vasodilation in humans reinforcing the idea that endogenous physiological levels of nitrite represent an inert end product of NO oxidation and reflect eNOS activity.41 This idea has changed over the past several years as many investigators have demonstrated that near physiological plasma concentrations (high nM to low µM) of nitrite anion elicit vasodilation in hypoxic tissues.42–47 These findings have led to the appreciation that nitrite is not simply an inert metabolic by-product of NO bioavailability, but rather represents a novel and effective endogenous prodrug for generation of NO during various tissue pathologies. Importantly, nitrite reduction to NO has been observed across many animal species including mice, rats, dogs, non-human primates, and humans indicating that this response represents a critically important salvage pathway in which to sustain some amount of NO production during states of duress (i.e. ischaemia).42,43,45–47 Together, these recent findings reveal a novel nitrite/NO metabolic system designed to maintain the capability of NO production under multiple conditions.
Red blood cell intracellular nitrite anion may serve as a reservoir buffer for NO equivalents which can be reduced back to NO through various mechanisms during tissue hypoxia or ischaemia. Nitrite anion reduction to NO was initially appreciated by the ability of deoxyhaemoglobin to reduce nitrite back to bioavailable NO equivalents.48 While it is now clear that deoxyhaemoglobin serves as a nitrite reductase, several mechanistic details by which this occurs still remain unclear and are discussed in greater detail by Patel et al. in this issue.49 Given the complexities of NO-haem reactions, hematocrit levels, and relative oxygen tensions within the vasculature, it is difficult to predict the precise amount of NO generated within the red cell that can diffuse and become available for other tissues.14,50–52 It is equally logical to expect that nitrite reduction to NO may occur more directly within specific tissue compartments responsible for regulating various physiological responses as reports have shown that tissue lysates from these organs can mediate nitrite reduction to NO under anoxic or hypoxic conditions.53,54 Currently, it is unknown whether all cells can universally reduce nitrite to NO or if there is cell-type specificity, which reduction mechanism is most critical, or under what conditions this could occur.
Several different reduction mechanisms are involved in converting nitrite to NO in a tissue or cell compartment-specific manner. We previously reported nitrite accumulation may preferentially occur within ischaemic tissue, facilitating nitrite reduction in discrete tissue compartments that require it.55 Recently Vitturi et al. have identified that nitrite entry into red cells involves a deoxygenation sensitive process that could involve enhanced nitrite import, decreased export, or both.56 However, specific mechanisms of nitrite uptake and transport by tissues remain poorly understood and require further study. Several different molecular pathways necessary for nitrite reduction are available within ischaemic tissues. Deoxymyoglobin can serve as a nitrite reductase leading to NO production during tissue ischaemia, playing an important role in preserving NO bioavailability during significant deprivation of oxygen.45,57,58 This process is important for nitrite-mediated cytoprotection during ischaemia that could involve different protective mechanisms.45,58 Tissue ischaemia also increases molybdenum hydroxylase enzyme expression or activity which could also be important for nitrite reduction.59,60 Xanthine oxidase (XO) reduction of nitrite to NO has been reported and may be an important mechanism for non-enzymatic NO generation under hypoxic conditions.61 The Zweier laboratory has shown that XO activity mediates nitrite reduction to NO tissue lysates from the liver and heart which is inversely related to oxygen tension.62,63 Feelisch et al.64 has also shown that nitrite is readily converted into NO in hypoxic tissue within discrete intracellular compartments, with the majority of reduction occurring in microsomes, possibly by XO. Lastly, Webb et al.65 reported deoxygenated red blood cells can also reduce nitrite through an XO-dependent process, with increasing acidosis and hypoxia influencing this response. Studies have also implicated aldehyde oxidase (AO) activity in reducing nitrite to NO in tissue lysates and intact vessels.66,67 However, the importance of AO over XO or tissue-specific responses between these processes remains poorly understood and requires further investigation. It will be difficult to determine the true importance of XO-dependent nitrite reduction for ischaemic cytoprotection in vivo, as XO inhibitors that block nitrite reduction also blunt ischaemia-reperfusion injury due to inhibition of superoxide production.68,69
Tissue ischaemia also results in decreased cellular pH which could result in acidic disproportionation of nitrite to NO independent of protein enzymatic activity.70 Aamand et al.71 recently demonstrated that carbonic anhydrase robustly increases nitrite-dependent NO formation which is potentiated by low tissue pH that occurs during ischaemia. Mitochondrial cytochrome c is also capable of reducing nitrite through haem iron pentacoordination under anoxic and acidic conditions.72 Mitochondrial nitrite reduction to NO can be further influenced by cytochrome c oxidase and the availability of an electron donor such as NADPH.73 Lastly, Vanin et al.74 has reported that recombinant eNOS protein can reduce nitrite to NO under hypoxic conditions which is similar to work from Webb et al.65 reporting that the NOS inhibitor l-NAME can attenuate nitrite reduction in blood vessels. Together, these reports demonstrate nitrite reduction to NO is highly complex and likely occurs through several different mechanisms thereby establishing redundant pathways with which to maintain NO bioavailability.
3.1. Biological effects of nitrite anion
With numerous ways of converting nitrite to NO, it is not surprising that there are multiple cellular responses to nitrite therapy (e.g. sodium nitrite). The preponderance of studies have shown that beneficial effects of near physiological plasma concentrations (high nanomolar) of sodium nitrite therapy emanate from the reduction of nitrite back to NO as the NO scavengers carboxy-PTIO (2-phenyl-4,4,5,5,-tetramethylimidazoline-1-oxyl-3-oxide) or cell free haemoglobin blunt many of the beneficial effects of nitrite therapy.43,45,55,75 However, the biological effects of high, supraphysiological plasma concentrations of nitrite (high micromolar) may directly alter cellular responses through events such as protein nitration that mediate cell injury.76,77 This dichotomous response may be related to the observation that low-dose sodium nitrite therapies are more efficacious than higher doses.45,55 Nonetheless, in several disease models and at a range of concentrations, sodium nitrite therapy has been shown to be beneficial for experimental CVD and ischaemic tissue dysfunction. Likely, this is due to several nitrite reducing mechanisms including deoxyhaemoglobin, deoxymyoglobin, acidosis, elevated XO expression or function, and altered mitochondrial respiration found in diseased tissue.
Specific biological effects of nitrite can vary and will undoubtedly reflect the response of the tissue or cell type involved as well as the dose administered. The identities of protective signalling pathways for nitrite therapy remain largely unknown but likely involve pathways affected by bona fide NO. Low-dose sodium nitrite therapy during acute ischaemia–reperfusion (I/R) injury of either the heart or liver can rapidly elevate intracellular NO metabolites such S-nitrosothiols followed by a later increase in N-nitrosamines and iron-nitrosyl proteins.45 These observations are consistent with NO-dependent changes in intracellular signalling cascades and protein functions as previously described.9,15 Conversely, Bryan et al.78 reported high micromolar doses of sodium nitrite increase cGMP levels, and HSP70 and haem oxygenase-1 (HO-1) expression in tissues through a carboxy-PTIO-independent manner implicating direct effects of nitrite anion itself. Low-dose sodium nitrite therapy also up-regulates HO-1 expression which appears to be an important mediator of nitrite-dependent cytoprotection.45 Nitrite conversion to NO can inhibit mitochondrial respiration and prevent uncoupling of electron transport which could attenuate apoptosis signalling.58 Lastly, sodium nitrite therapy may also facilitate ischaemic preconditioning through its effects on the mitochondria.79 These studies demonstrate that the effect of nitrite on cellular signalling and metabolic responses is complex and that additional studies in this area are needed to better understand specific cytoprotective mechanisms involved in nitrite therapy during tissue ischaemia.
3.2. The nitrite/NO endocrine system
Recent work from ours and other laboratories suggests that nitrite anion can act in an endocrine manner through organ-specific up-regulation of NO synthesis that increases systemic plasma nitrite levels which benefits distal tissues during ischaemic stress. Due to the short half-life of NO in blood and other biological compartments, it was thought that NO production is only capable of local site-specific effects. Therefore, to address the functional and metabolic relationship between NO and nitrite, Elrod et al.80 performed an elegant series of experiments demonstrating organ-specific eNOS-derived NO production is able to affect functional responses of distant organs. In this study, specific cardiac myocyte eNOS overexpression was shown to significantly increase plasma, cardiac, and hepatic tissue nitrite, nitrate, and nitrosothiol levels. Surprisingly, these mice were observed to have significant protection against acute hepatic ischaemia/reperfusion injury in an NO-dependent manner indicating that in this model cardiac myocyte-derived NO or its bioequivalent (e.g. nitrite) could be transported through the circulation to increase distal organ NO bioavailability. Likewise, we recently demonstrated that the anti-platelet agent dipyridamole significantly increases plasma nitrite levels which augments ischaemia-induced arteriogenesis and angiogenesis during chronic ischaemia of the hindlimb.81 Using eNOS deficient mice, we discovered that dipyridamole therapy significantly increased eNOS activity in non-ischaemic skeletal muscle tissue leading to increased NO production and plasma nitrite levels which subsequently enhanced ischaemic muscle tissue nitrite levels and vascular growth in an NO-dependent manner.81 These studies, coupled with those describing nitrite reduction back to NO within tissues, clearly demonstrate that nitrite anion serves as a stable NO equivalent that is reciprocally influenced by NOS-derived NO to establish an endocrine system to maintain adequate NO bioavailability. However, the field is only beginning to appreciate the importance and nature of the nitrite/NO endocrine system as little is known about specific-mechanisms of nitrite transport and retention in tissues and how increasing tissue nitrite levels affects endogenous enzymatic sources of NO generation.55,82
Dipyridamole has been previously reported to have deleterious effects when administered orally to patients with pre-existing atherosclerosis. Keltz et al.83 found that dipyridamole could induce myocardial ischaemia when administered orally in approximately 5% of patients reviewed, who had previous cardiovascular complications, through a possible ‘steal phenomenon'.84 The amount of dipyridamole used in this study was 100 mg and myocardial infarction was observed shortly after oral administration. However, chronic dosing of dipyridamole, 75 mg twice a day over 5 days, did not elicit myocardial ischaemia following high-dose dipyridamole stress tests in at-risk patients highlighting the complexity surrounding the effects of this drug.85 Importantly, extended release formulation (200 mg twice daily) of dipyridamole combined with aspirin therapy has proven successful in clinical trials testing the efficacy of dipyridamole for secondary stroke prevention with no increased incidence in myocardial ischaemia after repeated dosing of dipyridamole.84 In aggregate, these studies suggest that chronic dipyridamole therapy may be clinically useful despite previous negative reports. Additional experimental and clinical studies are needed to better determine when and if dipyridamole therapy may be useful for patients with peripheral vascular disease.
4. Therapeutic potential of inorganic nitrite for peripheral vascular disease
With the discovery of nitrite reduction back to NO under permissive conditions, inorganic nitrite therapy may prove very useful for various CVD states. Historically, sodium nitrite was briefly used at the turn of the twentieth century for severe hypertension, as it was able to effectively reduce blood pressure within several hours after administration. However, due to side effects such as methemoglobinaemia, headache, syncope, and its lower efficacy on blood pressure reduction compared with amyl nitrite, the use of sodium nitrite fell out of preference against other anti-hypertensive therapeutics and is now primarily used as an antidote to cyanide poisoning.86,87 Methemoglobin formation is one of the more problematic side effects of sodium nitrite with levels of 20–30&per; causing respiratory-related complications in otherwise healthy patients.88 Another complication of sodium nitrite was appreciated in the 1970s suggesting that consumption of large quantities of sodium nitrite resulted in gastric nitrosamine formation which was thought to contribute to carcinogenesis leading to strict government regulations for human nitrite consumption.89–91 Closer investigation of this hypothesis by the US Department of Health and Human Services National Toxicology Program found that the risk of sodium nitrite-mediated carcinogenesis is equivocal to nonexistent at large doses (100–220 mg/kg a day) which are a log order higher than doses shown to confer protection against ischaemic tissue damage (i.e. 100–200 µg/kg).92 Moreover, a recent report goes so far as to suggest that orally administered ‘sub-toxic doses’ of nitrite may facilitate NO-mediated apoptosis within a tumour.93 The principal idea of this hypothesis is that well-vascularized tumours never reach a critical NO concentration to induce apoptosis due to rapid loss of NO equivalents down a gradient formed between tumour cells and the vascular system.93 As such, increasing circulating levels of bioavailable NO equivalents might diminish this gradient loss thus augmenting tumour cell NO accumulation and ultimately apoptosis.93 While this and other hypotheses remain currently untested, it is clear that substantial precautions must be taken to address and monitor the effect of nitrite therapy on tumourigenesis events during clinical trials and future therapeutic regimens.
4.1. Peripheral arterial disease
Peripheral arterial disease (PAD) is broadly defined as an occlusive vascular disorder of the upper or lower extremities typically due to pathological events such as atherosclerosis, vasculitis, or thrombosis abnormalities. While the overall incidence of CVD has largely stabilized, the incidence of PAD is clearly increasing.94 PAD has historically been diagnosed in elderly populations; however, there has been a rise in disease incidence in younger patients (40 years old or less) due to the epidemics of diabetes and obesity.95,96 Hence, PAD incidence will continue to grow due to increases in the elderly population, diabetes, and obesity in the USA and Europe, as well as in other regions with increasing prevalence of diabetes and smoking (e.g. Asia). The 2005 ACC/AHA guidelines on PAD estimated the following rates of averse limb and cardiovascular outcomes at 5 years with 70–80&per; having stable claudication, 10–20&per; with progressive claudication, and 1–2% evolving to critical limb ischaemia (CLI) in patients initially diagnosed with noncritical claudication.97 Importantly, PAD patients are at significantly greater risk for myocardial infarction or stroke associated with an increased mortality rate.98 Such alarming statistics of PAD incidence, morbidity, and mortality highlight the urgent need for better treatment of this condition.
Several different therapeutic approaches may be employed for the management of PAD including supervised exercise therapy and pharmacological agents such as anti-platelet agents (clopidogrel), pentoxyphylline, and cilostazol, and lipid-lowering agents (statins) which all may provide some degree of benefit, but often have different durations of efficacy amongst PAD patients.99,100 Generally, anti-platelet agents are used in conjunction with aspirin for the treatment of PAD. However, some anti-platelet agents such as pentoxiphylline have limited to no ability to reduce PAD-associated claudication.101 Surgical revascularization and percutaneous intraluminal stenting techniques can also improve blood flow to the extremities affected by PAD.102 Unfortunately, a sizeable portion of patients with PAD, especially those with CLI, often respond poorly to these treatments because of diffuse atherosclerotic disease, poor distal conduits, or co-morbid conditions. Therapeutic angiogenesis still remains an attractive treatment approach for PAD patients and those in which surgical intervention provides a poor prognosis (e.g. CLI). Several different therapeutic angiogenesis agents have been identified including growth factors, transcription factors, and signalling molecules which substantially augment chronic ischaemia-induced angiogenesis in animal models.103 However, to date, successful gene or cell therapy approaches in animal models have proved disappointing in clinical trials for PAD therapy.104,105 Several possible factors could explain these results including suboptimal clinical trial designs, differences in vascular pathophysiology between rodents vs. man, as well as the fact that many experimental agents are preclinically evaluated in healthy animals with immediate therapeutic intervention.
Why then might nitrite anion therapy succeed where so many others failed? The first answer to this question is straightforward and centres on the fact that the majority of previous therapeutic angiogenesis approaches would have required functional cofactors or normal tissue responses to exert their effects. For example, VEGF-based therapy largely relies on eNOS/NO production to mediate its effects, yet eNOS dysfunction and oxidative stress is rampant in PAD patients. Likewise, other approaches such as l-arginine supplementation, eNOS gene therapy, or transcription factor therapies depend on ‘normal’ metabolic factors and responses such as availability of NADPH, BH4, ATP, and proper protein translation and signalling responses which may be greatly diminished or altered in ischaemic tissue. In diseased tissue settings, a more logical approach would be to cooperatively use or amplify tissue rescue responses such as nitrite reduction back to NO through a multitude of different ways. In this way, nitrite therapy may help ‘right the ship’ such that other approaches could even be employed to further stimulate vascular growth and tissue healing. The second answer to this question is also the fact that NO plays a central role in maintaining vascular health and critically orchestrates endothelial cell growth and vascular remodelling responses. This is exemplified by the fact that loss of NO bioavailability or function is a common denominator in major vascular disorders including hypertension, atherosclerosis, arteriosclerosis, PAD, and numerous vasculidities.38,106 Lastly, it is still not as widely appreciated among research investigators and clinicians that plasma nitrite levels accurately reflect the degree of eNOS function and amount of NO bioavailability which inversely correlates with the number of CVD risk factors compared with other indicators of vascular health such as hs-C-reactive protein.107,108
Recent evidence is also emerging that plasma nitrite levels may also reflect physiological differences in PAD patients with or without diabetes mellitus. Allen et al.109 recently published an interesting study looking at the effects exercise on plasma nitrite levels in different groups of PAD patients. The patients studied were categorized into one of four different groups: RF group had more than two traditional cardiovascular risk factors but no diagnosed CVD, DM group had type-2 diabetes but no diagnosed CVD, PAD group had intermittent claudication for more than 3 months; and DM + PAD. Brachial artery dilation per cent change during exercise was found to be highest in the RF group and lowest in the PAD and DM + PAD groups. These results correlated with plasma nitrite levels which showed increases in RF nitrite levels and a significant decrease in nitrite levels in PAD and DM + PAD groups. Interestingly the DM, PAD, and DM + PAD groups had higher levels of plasma nitrate than the RF group possibly suggesting that ongoing vascular disease could decrease plasma nitrite levels due to further oxidation to nitrate thereby decreasing the amount of readily convertible bioequivalents NO in these patients. This observation is corroborated by previous reports examining NO metabolites in PAD patients which reported increased nitrite/nitrate plasma levels using the Griess method of detection.110,111 It is important to clarify that these studies did not distinguish absolute plasma levels of nitrite vs. nitrate thereby resulting in a misleading conclusion that NO production is increased in these patients when it is possible that the plasma nitrite/nitrate balance shifts towards nitrate which does not readily confer ischaemic tissue cytoprotection like nitrite.45,55 It is clear that additional studies are needed to better understand the nitrite/nitrate relationship in PAD and their association with changes in redox status.
Our laboratory has reported that sodium nitrite anion therapy serves as a highly potent and selective NO donor which stimulates ischaemic vasodilation, cytoprotection, and robust angiogenic activity in the permanent femoral artery ligation model of hindlimb ischaemia.55 In this study, we found that sodium nitrite therapy quickly restored ischaemic hindlimb reperfusion and blood vessel growth over a few days in a NO-dependent manner; whereas, sodium nitrate therapy was unable to significantly augment these parameters. We also observed that tissue nitrite levels were significantly elevated at early time points compared with the non-ischaemic limb, and that tissue nitroso-thiol and nitroso-haem were elevated later as levels of nitrite decreased. These observations suggest that nitrite anion could be differentially taken up and metabolized in ischaemic vs. non-ischaemic tissue. Interestingly, we also found that nitrite therapy diminished eNOS protein expression over time, reinforcing the notion of a nitrite/NO endocrine system and corroborating a similar observation by Lang et al.82 One limitation of our study was the fact that sodium nitrite therapy was begun immediately after induction of permanent ischaemia, as it could be argued that immediate initiation of nitrite therapy might quickly augment collateral vessel perfusion and limit tissue damage.112 However, unpublished findings from our lab demonstrate that even when sodium nitrite therapy is began 5 days post-hindlimb ligation it still elicits rapid restoration of ischaemic limb blood flow. Together, these findings demonstrate that nitrite therapy augments ischaemic tissue reperfusion using treatment regimens that more closely reflect clinical situations.
We have also begun to identify specific molecular mediators involved in nitrite-dependent ischaemic tissue revascularization and repair using whole genome expression profile analysis in the permanent hindlimb ischaemia model.113 In this study, we examined genome-wide expression differences in ischaemic gastrocnemius muscle tissue from PBS control and sodium nitrite-treated animals. Using Ingenuity network analysis, we identified a broad spectrum of changes in gene expression, many of which have not been previously associated with ischaemic healing responses or nitrite/NO metabolism pathways. Nitrite anion therapy elicited a strong and predominant decrease in gene expression with a clear effect on innate and acquired immune response genes while selectively increasing vascular and muscle gene expression at day 3. Conversely, genome expression analysis at day 7 revealed that nitrite therapy predominantly up-regulated gene expression with significant increases of tissue repair and extracellular matrix genes demonstrating that nitrite therapy alters tissue responses in a multifactoral manner over time (i.e. affecting more than one target) thereby maximally augmenting tissue reperfusion and healing.
Several interesting genes were identified in our gene array study that confirms nitrite therapy affects multiple tissue types within the ischaemic limb. Nitrite therapy quickly increased vascular endothelial cadherin (VE-cadherin/cadherin 5) expression which is a key regulator of endothelial cell angiogenic activation.114,115 Interestingly, nitrite therapy significantly increased muscle gene expression involving cellular metabolism, repair, and differentiation. Myosin heavy and light chain gene expression were significantly elevated at day 3 along with glycogen synthase 1 indicating that nitrite augments ischaemic myocyte function. Nitrite therapy also increased myogenin expression by day 3 post-ligation suggestive of increased myoblast or satellite cell differentiation.116,117 Lastly, nitrite therapy significantly decreased expression of innate and acquired immune response genes including CXCL6, CXCL12, C-type lectin domain family adhesion proteins, allograft inflammatory factor 1, and formyl peptide receptor 2 among several others. It appears that nitrite therapy simultaneous confers anti-inflammatory and pro-cardiovascular/muscle gene expression early on which is all likely to be important for effective resolution of chronic tissue ischaemia. However, future studies focusing on the roles of these molecules during nitrite therapy are needed in order to better understand specific mechanisms of nitrite-mediated cytoprotection and tissue repair.
4.2. Atherosclerotic vascular disease
Atherosclerosis is the primary pathophysiological mediator of peripheral vascular disease, involving both inflammatory responses and endothelial cell dysfunction. NO production decreases due to several factors (e.g. oxidative stress, dysfunctional NOS activity, increased arginase expression, and others) as peripheral vascular disease progressively develops.118,119 This decrease in NO bioavailability directly facilitates increased leucocyte adhesion, platelet aggregation, and adherence, and results in dysregulation of vascular tone.120–122 These inflammatory factors coupled with a reduction in NO synthesis clearly contribute to initiation and propagation of atherosclerosis, suggesting that nitrite therapy might be a useful modality for atherosclerotic vascular disease.110,123,124 Recent work from Stokes et al.125 suggests that this could be true. This study reported that oral nitrite therapy in the drinking water significantly attenuated hypercholesterolaemia-mediated leucocyte recruitment to the vascular wall and also restored endothelial cell-dependent vasorelaxation responses. These initial observations suggest that the nitrite therapy approach to increase NO bioavailability could possibly be beneficial during atherosclerotic inflammatory vessel disease. However, no studies have examined whether nitrite therapy affects atherogenesis development and vascular disease in experimental models.
5. Conclusion
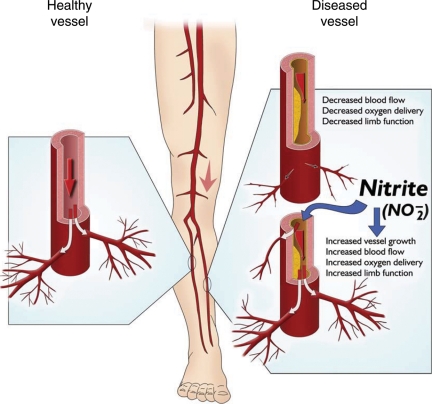
Nitrite anion is now well known to act as an important buffer for NO bioavailability and has been shown to enhance multiple vascular responses necessary for reperfusion of chronically ischaemic tissue (Figure 2). The field has made significant advances in understanding the mechanisms of nitrite reduction to NO and what pathophysiological conditions may benefit from nitrite therapy. However, many questions still remain such as: does nitrite reduction occur within specific vascular cells, and if so, by what mechanism? Is nitrite therapy protective in experimental peripheral vascular disease models of animals with ongoing disease such as diabetes or atherosclerosis? What molecular and signalling pathways does nitrite therapy activate to confer protection against chronic tissue ischaemia? While these and many other questions remain, it is clear that clinical studies may be warranted to evaluate whether nitrite therapy holds promise for the treatment of peripheral vascular diseases.
Figure 2.
Illustration of a physiologically healthy vessel compared with a diseased vessel and the effect nitrite therapy has on diseased vessels. The healthy vessel on the left is capable of adequately regulating blood flow to perfuse distal tissues. When this same vessel becomes dysfunctional due to atherosclerosis, vascular responsiveness is lost concomitant with luminal stenosis leading to reduced blood flow and oxygen delivery in distal tissues. Nitrite is able to augment collateral vessel perfusion and increase blood vessel growth thereby increasing blood flow to ischaemic tissues.
Funding
This work was supported by NIH grants HL80482, HL94021, and DK43785.
Conflict of interest: C.G.K. is a participant on a pending US patent filed on November 15, 2007 (patent no. 61/003150) regarding the use of nitrite salts in chronic ischaemia. C.G.K. has a commercial interest in Theravasc, Inc.
References
- 1.Ignarro LJ. Nitric oxide as a unique signaling molecule in the vascular system: a historical overview. J Physiol Pharmacol. 2002;53:503–514. [PubMed] [Google Scholar]
- 2.Moncada S, Higgs EA. The discovery of nitric oxide and its role in vascular biology. Br J Clin Pharmacol. 2006;147(Suppl 1:):S193–201. doi: 10.1038/sj.bjp.0706458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Napoli R, Guardasole V, Zarra E, Matarazzo M, D'Anna C, Sacca F, et al. Vascular smooth muscle cell dysfunction in patients with migraine. Neurology. 2009;72:2111–2114. doi: 10.1212/WNL.0b013e3181aa53ce. [DOI] [PubMed] [Google Scholar]
- 4.Cooper CE. Nitric oxide and iron proteins. Biochim Biophys Acta. 1999;1411:290–309. doi: 10.1016/s0005-2728(99)00021-3. [DOI] [PubMed] [Google Scholar]
- 5.Freeman BA, Baker PR, Schopfer FJ, Woodcock SR, Napolitano A, d'Ischia M. Nitro-fatty acid formation and signaling. J Biol Chem. 2008;283:15515–15519. doi: 10.1074/jbc.R800004200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martinez MC, Andriantsitohaina R. Reactive nitrogen species: molecular mechanisms and potential significance in health and disease. Antioxid Redox Signal. 2009;11:669–702. doi: 10.1089/ars.2007.1993. [DOI] [PubMed] [Google Scholar]
- 7.Stamler JS, Hausladen A. Oxidative modifications in nitrosative stress. Nat Struct Biol. 1998;5:247–249. doi: 10.1038/nsb0498-247. [DOI] [PubMed] [Google Scholar]
- 8.Sun J, Steenbergen C, Murphy E. S-nitrosylation NO-related redox signaling to protect against oxidative stress. Antioxid Redox Signal. 2006;8:1693–1705. doi: 10.1089/ars.2006.8.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Thomas DD, Miranda KM, Colton CA, Citrin D, Espey MG, Wink DA. Heme proteins and nitric oxide (NO): the neglected, eloquent chemistry in NO redox signaling and regulation. Antioxid Redox Signal. 2003;5:307–317. doi: 10.1089/152308603322110887. [DOI] [PubMed] [Google Scholar]
- 10.Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev. 2007;87:315–424. doi: 10.1152/physrev.00029.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Taylor CT, Moncada S. Nitric oxide, cytochrome C oxidase, and the cellular response to hypoxia. Arterioscler Thromb Vasc Biol. 2010;30:643–647. doi: 10.1161/ATVBAHA.108.181628. [DOI] [PubMed] [Google Scholar]
- 12.Fulton D, Gratton JP, Sessa WC. Post-translational control of endothelial nitric oxide synthase: why isn't calcium/calmodulin enough? J Pharmacol Exp Ther. 2001;299:818–824. [PubMed] [Google Scholar]
- 13.Kone BC, Kuncewicz T, Zhang W, Yu ZY. Protein interactions with nitric oxide synthases: controlling the right time, the right place, and the right amount of nitric oxide. Am J Physiol. 2003;285:F178–190. doi: 10.1152/ajprenal.00048.2003. [DOI] [PubMed] [Google Scholar]
- 14.Lancaster JR., Jr. A tutorial on the diffusibility and reactivity of free nitric oxide. Nitric Oxide. 1997;1:18–30. doi: 10.1006/niox.1996.0112. [DOI] [PubMed] [Google Scholar]
- 15.Thomas DD, Ridnour LA, Isenberg JS, Flores-Santana W, Switzer CH, Donzelli S, et al. The chemical biology of nitric oxide: implications in cellular signaling. Free Radic Biol Med. 2008;45:18–31. doi: 10.1016/j.freeradbiomed.2008.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Laroux FS, Lefer DJ, Kawachi S, Scalia R, Cockrell AS, Gray L, et al. Role of nitric oxide in the regulation of acute and chronic inflammation. Antioxid Redox Signal. 2000;2:391–396. doi: 10.1089/15230860050192161. [DOI] [PubMed] [Google Scholar]
- 17.Lefer DJ. Myocardial protective actions of nitric oxide donors after myocardial ischemia and reperfusion. New Horiz (Baltimore, MD) 1995;3:105–112. [PubMed] [Google Scholar]
- 18.Morbidelli L, Donnini S, Ziche M. Role of nitric oxide in the modulation of angiogenesis. Curr Pharm Des. 2003;9:521–530. doi: 10.2174/1381612033391405. [DOI] [PubMed] [Google Scholar]
- 19.Cooke JP, Losordo DW. Nitric oxide and angiogenesis. Circulation. 2002;105:2133–2135. doi: 10.1161/01.cir.0000014928.45119.73. [DOI] [PubMed] [Google Scholar]
- 20.Luque Contreras D, Vargas Robles H, Romo E, Rios A, Escalante B. The role of nitric oxide in the post-ischemic revascularization process. Pharmacol Ther. 2006;112:553–563. doi: 10.1016/j.pharmthera.2006.05.003. [DOI] [PubMed] [Google Scholar]
- 21.Papapetropoulos A, Garcia-Cardena G, Madri JA, Sessa WC. Nitric oxide production contributes to the angiogenic properties of vascular endothelial growth factor in human endothelial cells. J Clin Invest. 1997;100:3131–3139. doi: 10.1172/JCI119868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ziche M, Morbidelli L, Choudhuri R, Zhang HT, Donnini S, Granger HJ, et al. Nitric oxide synthase lies downstream from vascular endothelial growth factor-induced but not basic fibroblast growth factor-induced angiogenesis. J Clin Invest. 1997;99:2625–2634. doi: 10.1172/JCI119451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kawasaki K, Smith RS, Jr., Hsieh CM, Sun J, Chao J, Liao JK. Activation of the phosphatidylinositol 3-kinase/protein kinase Akt pathway mediates nitric oxide-induced endothelial cell migration and angiogenesis. Mol Cell Biol. 2003;23:5726–5737. doi: 10.1128/MCB.23.16.5726-5737.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jones MK, Tsugawa K, Tarnawski AS, Baatar D. Dual actions of nitric oxide on angiogenesis: possible roles of PKC, ERK, and AP-1. Biochem Biophys Res Commun. 2004;318:520–528. doi: 10.1016/j.bbrc.2004.04.055. [DOI] [PubMed] [Google Scholar]
- 25.Morbidelli L, Pyriochou A, Filippi S, Vasileiadis I, Roussos C, Zhou Z, et al. The soluble guanylyl cyclase inhibitor NS-2028 reduces vascular endothelial growth factor-induced angiogenesis and permeability. Am J Physiol Regul Integr Comp Physiol. 2008;298:R824–832. doi: 10.1152/ajpregu.00222.2009. [DOI] [PubMed] [Google Scholar]
- 26.Murohara T, Asahara T. Nitric oxide and angiogenesis in cardiovascular disease. Antioxid Redox Signal. 2002;4:825–831. doi: 10.1089/152308602760598981. [DOI] [PubMed] [Google Scholar]
- 27.Duda DG, Fukumura D, Jain RK. Role of eNOS in neovascularization: NO for endothelial progenitor cells. Trends Mol Med. 2004;10:143–145. doi: 10.1016/j.molmed.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 28.Fukumura D, Gohongi T, Kadambi A, Izumi Y, Ang J, Yun CO, et al. Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factor-induced angiogenesis and vascular permeability. Proc Natl Acad Sci USA. 2001;98:2604–2609. doi: 10.1073/pnas.041359198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Murohara T, Asahara T, Silver M, Bauters C, Masuda H, Kalka C, et al. Nitric oxide synthase modulates angiogenesis in response to tissue ischemia. J Clin Invest. 1998;101:2567–2578. doi: 10.1172/JCI1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yu J, deMuinck ED, Zhuang Z, Drinane M, Kauser K, Rubanyi GM, et al. Endothelial nitric oxide synthase is critical for ischemic remodeling, mural cell recruitment, and blood flow reserve. Proc Natl Acad Sci USA. 2005;102:10999–11004. doi: 10.1073/pnas.0501444102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jones SP, Girod WG, Palazzo AJ, Granger DN, Grisham MB, Jourd'Heuil D, et al. Myocardial ischemia-reperfusion injury is exacerbated in absence of endothelial cell nitric oxide synthase. Am J Physiol. 1999;276:H1567–1573. doi: 10.1152/ajpheart.1999.276.5.H1567. [DOI] [PubMed] [Google Scholar]
- 32.Jones SP, Greer JJ, Kakkar AK, Ware PD, Turnage RH, Hicks M, et al. Endothelial nitric oxide synthase overexpression attenuates myocardial reperfusion injury. Am J Physiol Heart Circ Physiol. 2004;286:H276–282. doi: 10.1152/ajpheart.00129.2003. [DOI] [PubMed] [Google Scholar]
- 33.Boo YC, Kim HJ, Song H, Fulton D, Sessa W, Jo H. Coordinated regulation of endothelial nitric oxide synthase activity by phosphorylation and subcellular localization. Free Radic Biol Med. 2006;41:144–153. doi: 10.1016/j.freeradbiomed.2006.03.024. [DOI] [PubMed] [Google Scholar]
- 34.Fulton D, Babbitt R, Zoellner S, Fontana J, Acevedo L, McCabe TJ, et al. Targeting of endothelial nitric-oxide synthase to the cytoplasmic face of the Golgi complex or plasma membrane regulates Akt- vs. calcium-dependent mechanisms for nitric oxide release. J Biol Chem. 2004;279:30349–30357. doi: 10.1074/jbc.M402155200. [DOI] [PubMed] [Google Scholar]
- 35.Iwakiri Y, Satoh A, Chatterjee S, Toomre DK, Chalouni CM, Fulton D, et al. Nitric oxide synthase generates nitric oxide locally to regulate compartmentalized protein S-nitrosylation and protein trafficking. Proc Natl Acad Sci USA. 2006;103:19777–19782. doi: 10.1073/pnas.0605907103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jagnandan D, Sessa WC, Fulton D. Intracellular location regulates calcium-calmodulin-dependent activation of organelle-restricted eNOS. Am J Physiol Cell Physiol. 2005;289:C1024–1033. doi: 10.1152/ajpcell.00162.2005. [DOI] [PubMed] [Google Scholar]
- 37.Zhang Q, Church JE, Jagnandan D, Catravas JD, Sessa WC, Fulton D. Functional relevance of Golgi- and plasma membrane-localized endothelial NO synthase in reconstituted endothelial cells. Arterioscler Thromb Vasc Biol. 2006;26:1015–1021. doi: 10.1161/01.ATV.0000216044.49494.c4. [DOI] [PubMed] [Google Scholar]
- 38.Napoli C, Ignarro LJ. Nitric oxide and pathogenic mechanisms involved in the development of vascular diseases. Arch Pharm Res. 2009;32:1103–1108. doi: 10.1007/s12272-009-1801-1. [DOI] [PubMed] [Google Scholar]
- 39.Liu VW, Huang PL. Cardiovascular roles of nitric oxide: a review of insights from nitric oxide synthase gene disrupted mice. Cardiovasc Res. 2008;77:19–29. doi: 10.1016/j.cardiores.2007.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Furchgott RF. Studies on relaxation of rabbit aorta by sodium nitrite: the basis for the proposal that the acid-activatable inhibitory factor from retractor penis is inorganic nitrite and the endothelium-derived relaxing factor is nitric oxide. In: Vanhoutte PM, editor. Vasodilation: Vascular Smooth Muscle, Peptides, and Endothelium. New York: Raven Press; 1988. pp. p401–414. [Google Scholar]
- 41.Lauer T, Preik M, Rassaf T, Strauer BE, Deussen A, Feelisch M, et al. Plasma nitrite rather than nitrate reflects regional endothelial nitric oxide synthase activity but lacks intrinsic vasodilator action. Proc Natl Acad Sci USA. 2001;98:12814–12819. doi: 10.1073/pnas.221381098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cosby K, Partovi KS, Crawford JH, Patel RP, Reiter CD, Martyr S, et al. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat Med. 2003;9:1498–1505. doi: 10.1038/nm954. [DOI] [PubMed] [Google Scholar]
- 43.Crawford JH, Isbell TS, Huang Z, Shiva S, Chacko BK, Schechter AN, et al. Hypoxia, red blood cells, and nitrite regulate NO-dependent hypoxic vasodilation. Blood. 2006;107:566–574. doi: 10.1182/blood-2005-07-2668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gladwin MT. Evidence mounts that nitrite contributes to hypoxic vasodilation in the human circulation. Circulation. 2008;117:594–597. doi: 10.1161/CIRCULATIONAHA.107.753897. [DOI] [PubMed] [Google Scholar]
- 45.Duranski MR, Greer JJ, Dejam A, Jaganmohan S, Hogg N, Langston W, et al. Cytoprotective effects of nitrite during in vivo ischemia-reperfusion of the heart and liver. J Clin Invest. 2005;115:1232–1240. doi: 10.1172/JCI22493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hunter CJ, Dejam A, Blood AB, Shields H, Kim-Shapiro DB, Machado RF, et al. Inhaled nebulized nitrite is a hypoxia-sensitive NO-dependent selective pulmonary vasodilator. Nat Med. 2004;10:1122–1127. doi: 10.1038/nm1109. [DOI] [PubMed] [Google Scholar]
- 47.Dejam A, Hunter CJ, Tremonti C, Pluta RM, Hon YY, Grimes G, et al. Nitrite infusion in humans and nonhuman primates: endocrine effects, pharmacokinetics, and tolerance formation. Circulation. 2007;116:1821–1831. doi: 10.1161/CIRCULATIONAHA.107.712133. [DOI] [PubMed] [Google Scholar]
- 48.Doyle MP, Pickering RA, DeWeert TM, Hoekstra JW, Pater D. Kinetics and mechanism of the oxidation of human deoxyhemoglobin by nitrites. J Biol Chem. 1981;256:12393–12398. [PubMed] [Google Scholar]
- 49.Patel RP, Hogg N, Kim-Shapiro DB. The potential role of the red blood cell in nitrite-dependent regulation of blood flow. Cardiovasc Res. 2011;89:507–515. doi: 10.1093/cvr/cvq323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chakraborty S, Balakotaiah V, Bidani A. Diffusing capacity reexamined: relative roles of diffusion and chemical reaction in red cell uptake of O2, CO, CO2, and NO. J Appl Physiol. 2004;97:2284–2302. doi: 10.1152/japplphysiol.00469.2004. [DOI] [PubMed] [Google Scholar]
- 51.Huang KT, Huang Z, Kim-Shapiro DB. Nitric oxide red blood cell membrane permeability at high and low oxygen tension. Nitric Oxide. 2007;16:209–216. doi: 10.1016/j.niox.2006.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kim-Shapiro DB, Schechter AN, Gladwin MT. Unraveling the reactions of nitric oxide, nitrite, and hemoglobin in physiology and therapeutics. Arterioscler Thromb Vasc Biol. 2006;26:697–705. doi: 10.1161/01.ATV.0000204350.44226.9a. [DOI] [PubMed] [Google Scholar]
- 53.Alzawahra WF, Talukder MA, Liu X, Samouilov A, Zweier JL. Heme proteins mediate the conversion of nitrite to nitric oxide in the vascular wall. Am J Physiol Heart Circ Physiol. 2008;295:H499–508. doi: 10.1152/ajpheart.00374.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zweier JL, Li H, Samouilov A, Liu X. Mechanisms of nitrite reduction to nitric oxide in the heart and vessel wall. Nitric Oxide. 2010;22:83–90. doi: 10.1016/j.niox.2009.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kumar D, Branch BG, Pattillo CB, Hood J, Thoma S, Simpson S, et al. Chronic sodium nitrite therapy augments ischemia-induced angiogenesis and arteriogenesis. Proc Natl Acad Sci USA. 2008;105:7540–7545. doi: 10.1073/pnas.0711480105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vitturi DA, Teng X, Toledo JC, Matalon S, Lancaster JR, Jr., Patel RP. Regulation of nitrite transport in red blood cells by hemoglobin oxygen fractional saturation. Am J Physiol Heart Circ Physiol. 2009;296:H1398–1407. doi: 10.1152/ajpheart.01303.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rassaf T, Flogel U, Drexhage C, Hendgen-Cotta U, Kelm M, Schrader J. Nitrite reductase function of deoxymyoglobin: oxygen sensor and regulator of cardiac energetics and function. Circ Res. 2007;100:1749–1754. doi: 10.1161/CIRCRESAHA.107.152488. [DOI] [PubMed] [Google Scholar]
- 58.Shiva S, Huang Z, Grubina R, Sun J, Ringwood LA, MacArthur PH, et al. Deoxymyoglobin is a nitrite reductase that generates nitric oxide and regulates mitochondrial respiration. Circ Res. 2007;100:654–661. doi: 10.1161/01.RES.0000260171.52224.6b. [DOI] [PubMed] [Google Scholar]
- 59.Garattini E, Mendel R, Romao MJ, Wright R, Terao M. Mammalian molybdo-flavoenzymes, an expanding family of proteins: structure, genetics, regulation, function and pathophysiology. Biochem J. 2003;372:15–32. doi: 10.1042/BJ20030121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Grisham MB, Granger DN. Neutrophil-mediated mucosal injury. Role of reactive oxygen metabolites. Dig Dis Sci. 1988;33:6S–15S. doi: 10.1007/BF01538126. [DOI] [PubMed] [Google Scholar]
- 61.Millar TM, Stevens CR, Benjamin N, Eisenthal R, Harrison R, Blake DR. Xanthine oxidoreductase catalyses the reduction of nitrates and nitrite to nitric oxide under hypoxic conditions. FEBS Lett. 1998;427:225–228. doi: 10.1016/s0014-5793(98)00430-x. [DOI] [PubMed] [Google Scholar]
- 62.Li H, Samouilov A, Liu X, Zweier JL. Characterization of the magnitude and kinetics of xanthine oxidase-catalyzed nitrite reduction. Evaluation of its role in nitric oxide generation in anoxic tissues. J Biol Chem. 2001;276:24482–24489. doi: 10.1074/jbc.M011648200. [DOI] [PubMed] [Google Scholar]
- 63.Li H, Samouilov A, Liu X, Zweier JL. Characterization of the effects of oxygen on xanthine oxidase-mediated nitric oxide formation. J Biol Chem. 2004;279:16939–16946. doi: 10.1074/jbc.M314336200. [DOI] [PubMed] [Google Scholar]
- 64.Feelisch M, Fernandez BO, Bryan NS, Garcia-Saura MF, Bauer S, Whitlock DR, et al. Tissue processing of nitrite in hypoxia: an intricate interplay of nitric oxide-generating and -scavenging systems. J Biol Chem. 2008;283:33927–33934. doi: 10.1074/jbc.M806654200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Webb AJ, Milsom AB, Rathod KS, Chu WL, Qureshi S, Lovell MJ, et al. Mechanisms underlying erythrocyte and endothelial nitrite reduction to nitric oxide in hypoxia: role for xanthine oxidoreductase and endothelial nitric oxide synthase. Circ Res. 2008;103:957–964. doi: 10.1161/CIRCRESAHA.108.175810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li H, Kundu TK, Zweier JL. Characterization of the magnitude and mechanism of aldehyde oxidase-mediated nitric oxide production from nitrite. J Biol Chem. 2009;284:33850–33858. doi: 10.1074/jbc.M109.019125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pinder AG, Pittaway E, Morris K, James PE. Nitrite directly vasodilates hypoxic vasculature via nitric oxide-dependent and -independent pathways. Br J Pharmacol. 2009;157:1523–1530. doi: 10.1111/j.1476-5381.2009.00340.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Grisham MB, Hernandez LA, Granger DN. Xanthine oxidase and neutrophil infiltration in intestinal ischemia. Am J Physiol. 1986;251:G567–574. doi: 10.1152/ajpgi.1986.251.4.G567. [DOI] [PubMed] [Google Scholar]
- 69.Mitsuoka H, Schmid-Schonbein GW. Mechanisms for blockade of in vivo activator production in the ischemic intestine and multi-organ failure. Shock (Augusta, Ga) 2000;14:522–527. doi: 10.1097/00024382-200014050-00005. [DOI] [PubMed] [Google Scholar]
- 70.Samouilov A, Kuppusamy P, Zweier JL. Evaluation of the magnitude and rate of nitric oxide production from nitrite in biological systems. Arch Biochem Biophys. 1998;357:1–7. doi: 10.1006/abbi.1998.0785. [DOI] [PubMed] [Google Scholar]
- 71.Aamand R, Dalsgaard T, Jensen FB, Simonsen U, Roepstorff A, Fago A. Generation of nitric oxide from nitrite by carbonic anhydrase: a possible link between metabolic activity and vasodilation. Am J Physiol Heart Circ Physiol. 2009;297:H2068–2074. doi: 10.1152/ajpheart.00525.2009. [DOI] [PubMed] [Google Scholar]
- 72.Basu S, Azarova NA, Font MD, King SB, Hogg N, Gladwin MT, et al. Nitrite reductase activity of cytochrome c. J Biol Chem. 2008;283:32590–32597. doi: 10.1074/jbc.M806934200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Castello PR, David PS, McClure T, Crook Z, Poyton RO. Mitochondrial cytochrome oxidase produces nitric oxide under hypoxic conditions: implications for oxygen sensing and hypoxic signaling in eukaryotes. Cell Metab. 2006;3:277–287. doi: 10.1016/j.cmet.2006.02.011. [DOI] [PubMed] [Google Scholar]
- 74.Vanin AF, Bevers LM, Slama-Schwok A, van Faassen EE. Nitric oxide synthase reduces nitrite to NO under anoxia. Cell Mol Life Sci. 2007;64:96–103. doi: 10.1007/s00018-006-6374-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Dezfulian C, Raat N, Shiva S, Gladwin MT. Role of the anion nitrite in ischemia-reperfusion cytoprotection and therapeutics. Cardiovasc Res. 2007;75:327–338. doi: 10.1016/j.cardiores.2007.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hazen SL, Zhang R, Shen Z, Wu W, Podrez EA, MacPherson JC, et al. Formation of nitric oxide-derived oxidants by myeloperoxidase in monocytes: pathways for monocyte-mediated protein nitration and lipid peroxidation in vivo. Circ Res. 1999;85:950–958. doi: 10.1161/01.res.85.10.950. [DOI] [PubMed] [Google Scholar]
- 77.Podrez EA, Schmitt D, Hoff HF, Hazen SL. Myeloperoxidase-generated reactive nitrogen species convert LDL into an atherogenic form in vitro. J Clin Invest. 1999;103:1547–1560. doi: 10.1172/JCI5549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bryan NS, Fernandez BO, Bauer SM, Garcia-Saura MF, Milsom AB, Rassaf T, et al. Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat Chem Biol. 2005;1:290–297. doi: 10.1038/nchembio734. [DOI] [PubMed] [Google Scholar]
- 79.Shiva S, Sack MN, Greer JJ, Duranski M, Ringwood LA, Burwell L, et al. Nitrite augments tolerance to ischemia/reperfusion injury via the modulation of mitochondrial electron transfer. J Exp Med. 2007;204:2089–2102. doi: 10.1084/jem.20070198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Elrod JW, Calvert JW, Gundewar S, Bryan NS, Lefer DJ. Nitric oxide promotes distant organ protection: evidence for an endocrine role of nitric oxide. Proc Natl Acad Sci USA. 2008;105:11430–11435. doi: 10.1073/pnas.0800700105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Venkatesh PK, Pattillo CB, Branch B, Hood J, Thoma S, Illum S, et al. Dipyridamole enhances ischaemia-induced arteriogenesis through an endocrine nitrite/nitric oxide-dependent pathway. Cardiovasc Res. 2010;85:661–670. doi: 10.1093/cvr/cvq002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lang JD, Jr., Teng X, Chumley P, Crawford JH, Isbell TS, Chacko BK, et al. Inhaled NO accelerates restoration of liver function in adults following orthotopic liver transplantation. J Clin Invest. 2007;117:2583–2591. doi: 10.1172/JCI31892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Keltz TN, Innerfield M, Gitler B, Cooper JA. Dipyridamole-induced myocardial ischemia. JAMA. 1987;257:1515–1516. doi: 10.1001/jama.257.11.1515. [DOI] [PubMed] [Google Scholar]
- 84.Crown N, Mysak T. Safety of fixed-dose aspirin-extended-release dipyridamole in patients with ischemic heart disease. Am J Health Syst Pharm. 2010;67:728–733. doi: 10.2146/ajhp080645. [DOI] [PubMed] [Google Scholar]
- 85.Guideri F, Capecchi PL, Acampa M, Cuomo A, Lazzerini PE, De Giorgi L, et al. Oral low-dose dipyridamole protects from intravenous high-dose dipyridamole-induced ischemia. A stress echocardiographic study. Int J Cardiol. 2002;83:209–215;. doi: 10.1016/s0167-5273(02)00061-x. discussion 215–206. [DOI] [PubMed] [Google Scholar]
- 86.Hunault CC, van Velzen AG, Sips AJ, Schothorst RC, Meulenbelt J. Bioavailability of sodium nitrite from an aqueous solution in healthy adults. Toxicol Lett. 2009;190:48–53. doi: 10.1016/j.toxlet.2009.06.865. [DOI] [PubMed] [Google Scholar]
- 87.Baskin SI, Horowitz AM, Nealley EW. The antidotal action of sodium nitrite and sodium thiosulfate against cyanide poisoning. J Clin Pharmacol. 1992;32:368–375. doi: 10.1002/j.1552-4604.1992.tb03849.x. [DOI] [PubMed] [Google Scholar]
- 88.Umbreit J. Methemoglobin—it's not just blue: a concise review. Am J Hematol. 2007;82:134–144. doi: 10.1002/ajh.20738. [DOI] [PubMed] [Google Scholar]
- 89.Lijinsky W. Significance of in vivo formation of N-nitroso compounds. Oncology. 1980;37:223–226. doi: 10.1159/000225440. [DOI] [PubMed] [Google Scholar]
- 90.Mirvish SS. The etiology of gastric cancer. Intragastric nitrosamide formation and other theories. J Natl Cancer Inst. 1983;71:629–647. [PubMed] [Google Scholar]
- 91.Odashima S. Overview: N-nitroso compounds as carcinogens for experimental animals and man. Oncology. 1980;37:282–286. doi: 10.1159/000225453. [DOI] [PubMed] [Google Scholar]
- 92.U.S. National Toxicology Program. Toxicology and carcinogenesis studies of sodium nitrite (CAS NO. 7632-00-0) in F344/N rats and B6C3F1 mice (drinking water studies) Natl Toxicol Program Tech Rep Ser. 2001;495:7–273. [PubMed] [Google Scholar]
- 93.Heller A. Electrochemistry and nitric oxide mass transport in cancer: why ingestion of sodium nitrite could be effective in treating vascularized tumors. Phys Chem Chem Phys. 2010;12:9972–9975. doi: 10.1039/c004520a. [DOI] [PubMed] [Google Scholar]
- 94.Stehouwer CD, Clement D, Davidson C, Diehm C, Elte JW, Lambert M, et al. Peripheral arterial disease: a growing problem for the internist. Eur J Intern Med. 2009;20:132–138. doi: 10.1016/j.ejim.2008.09.013. [DOI] [PubMed] [Google Scholar]
- 95.Dinh T, Scovell S, Veves A. Peripheral arterial disease and diabetes: a clinical update. Int J Low Extrem Wounds. 2009;8:75–81. doi: 10.1177/1534734609336768. [DOI] [PubMed] [Google Scholar]
- 96.Huysman E, Mathieu C. Diabetes and peripheral vascular disease. Acta Chir Belg. 2009;109:587–594. doi: 10.1080/00015458.2009.11680493. [DOI] [PubMed] [Google Scholar]
- 97.Mohler E, 3rd, Giri J. Management of peripheral arterial disease patients: comparing the ACC/AHA and TASC-II guidelines. Curr Med Res Opin. 2008;24:2509–2522. doi: 10.1185/03007990802274379. [DOI] [PubMed] [Google Scholar]
- 98.Welten GM, Schouten O, Chonchol M, Hoeks SE, Bax JJ, Van Domburg RT, et al. Prognosis of patients with peripheral arterial disease. J Cardiovasc Surg. 2009;50:109–121. [PubMed] [Google Scholar]
- 99.Aronow H, Hiatt WR. The burden of peripheral artery disease and the role of antiplatelet therapy. Postgrad Med. 2009;121:123–135. doi: 10.3810/pgm.2009.07.2038. [DOI] [PubMed] [Google Scholar]
- 100.Dobesh PP, Stacy ZA, Persson EL. Pharmacologic therapy for intermittent claudication. Pharmacotherapy. 2009;29:526–553. doi: 10.1592/phco.29.5.526. [DOI] [PubMed] [Google Scholar]
- 101.White C. Clinical practice. Intermittent claudication. N Engl J Med. 2007;356:1241–1250. doi: 10.1056/NEJMcp064483. [DOI] [PubMed] [Google Scholar]
- 102.Singh KP, Patel MR, Kandzari DE, Zidar JP. Peripheral arterial disease: an overview of endovascular therapies and contemporary treatment strategies. Rev Cardiovasc Med. 2006;7:55–68. [PubMed] [Google Scholar]
- 103.Emanueli C, Madeddu P. Therapeutic angiogenesis: translating experimental concepts to medically relevant goals. Vascul Pharmacol. 2006;45:334–339. doi: 10.1016/j.vph.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 104.De Haro J, Acin F, Lopez-Quintana A, Florez A, Martinez-Aguilar E, Varela C. Meta-analysis of randomized, controlled clinical trials in angiogenesis: gene and cell therapy in peripheral arterial disease. Heart Vessels. 2009;24:321–328. doi: 10.1007/s00380-008-1140-z. [DOI] [PubMed] [Google Scholar]
- 105.Gupta R, Tongers J, Losordo DW. Human studies of angiogenic gene therapy. Circ Res. 2009;105:724–736. doi: 10.1161/CIRCRESAHA.109.200386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kevil CG, Patel RP. Preserving vessel function during ischemic disease: new possibilities of inorganic nitrite therapy. Expert Rev Cardiovasc Ther. 2008;6:1175–1179. doi: 10.1586/14779072.6.9.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kleinbongard P, Dejam A, Lauer T, Jax T, Kerber S, Gharini P, et al. Plasma nitrite concentrations reflect the degree of endothelial dysfunction in humans. Free Radic Biol Med. 2006;40:295–302. doi: 10.1016/j.freeradbiomed.2005.08.025. [DOI] [PubMed] [Google Scholar]
- 108.Kleinbongard P, Dejam A, Lauer T, Rassaf T, Schindler A, Picker O, et al. Plasma nitrite reflects constitutive nitric oxide synthase activity in mammals. Free Radic Biol Med. 2003;35:790–796. doi: 10.1016/s0891-5849(03)00406-4. [DOI] [PubMed] [Google Scholar]
- 109.Allen JD, Miller EM, Schwark E, Robbins JL, Duscha BD, Annex BH. Plasma nitrite response and arterial reactivity differentiate vascular health and performance. Nitric Oxide. 2009;20:231–237. doi: 10.1016/j.niox.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.de Haro Miralles J, Martinez-Aguilar E, Florez A, Varela C, Bleda S, Acin F. Nitric oxide: link between endothelial dysfunction and inflammation in patients with peripheral arterial disease of the lower limbs. Interact Cardiovasc Thorac Surg. 2009;9:107–112. doi: 10.1510/icvts.2008.196428. [DOI] [PubMed] [Google Scholar]
- 111.Ferlito S, Gallina M, Catassi S, Bisicchia A, Di Salvo MM. Nitrite plasma levels in normolipemic and hypercholesterolemic patients with peripheral occlusive arteriopathy. Panminerva Med. 1999;41:307–309. [PubMed] [Google Scholar]
- 112.Mazzone M, Carmeliet P. Drug discovery: a lifeline for suffocating tissues. Nature. 2008;453:1194–1195. doi: 10.1038/4531194a. [DOI] [PubMed] [Google Scholar]
- 113.Pattillo CB, Fang K, Pardue S, Kevil CG. Genome expression profiling and network analysis of nitrite therapy during chronic ischemia: possible mechanisms and interesting molecules. Nitric Oxide. 2010;22:168–179. doi: 10.1016/j.niox.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Vestweber D. VE-cadherin: the major endothelial adhesion molecule controlling cellular junctions and blood vessel formation. Arterioscler Thromb Vasc Biol. 2008;28:223–232. doi: 10.1161/ATVBAHA.107.158014. [DOI] [PubMed] [Google Scholar]
- 115.Wallez Y, Vilgrain I, Huber P. Angiogenesis: the VE-cadherin switch. Trends Cardiovasc Med. 2006;16:55–59. doi: 10.1016/j.tcm.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 116.Berkes CA, Tapscott SJ. MyoD and the transcriptional control of myogenesis. Semin Cell Dev Biol. 2005;16:585–595. doi: 10.1016/j.semcdb.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 117.Yablonka-Reuveni Z, Day K, Vine A, Shefer G. Defining the transcriptional signature of skeletal muscle stem cells. J Anim Sci. 2008;86:E207–216. doi: 10.2527/jas.2007-0473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Huang PL. eNOS, metabolic syndrome and cardiovascular disease. Trends Endocrinol Metab. 2009;20:295–302. doi: 10.1016/j.tem.2009.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Victor VM, Rocha M, Sola E, Banuls C, Garcia-Malpartida K, Hernandez-Mijares A. Oxidative stress, endothelial dysfunction and atherosclerosis. Curr Pharm Des. 2009;15:2988–3002. doi: 10.2174/138161209789058093. [DOI] [PubMed] [Google Scholar]
- 120.Boger RH, Bode-Boger SM, Thiele W, Junker W, Alexander K, Frolich JC. Biochemical evidence for impaired nitric oxide synthesis in patients with peripheral arterial occlusive disease. Circulation. 1997;95:2068–2074. doi: 10.1161/01.cir.95.8.2068. [DOI] [PubMed] [Google Scholar]
- 121.Kubes P, Suzuki M, Granger DN. Nitric oxide: an endogenous modulator of leukocyte adhesion. Proc Natl Acad Sci USA. 1991;88:4651–4655. doi: 10.1073/pnas.88.11.4651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Lefer AM, Ma XL. Decreased basal nitric oxide release in hypercholesterolemia increases neutrophil adherence to rabbit coronary artery endothelium. Arterioscler Thromb. 1993;13:771–776. doi: 10.1161/01.atv.13.6.771. [DOI] [PubMed] [Google Scholar]
- 123.De Haro J, Acin F, Lopez-Quintana A, Medina FJ, Martinez-Aguilar E, Florez A, et al. Direct association between C-reactive protein serum levels and endothelial dysfunction in patients with claudication. Eur J Vasc Endovasc Surg. 2008;35:480–486. doi: 10.1016/j.ejvs.2007.10.016. [DOI] [PubMed] [Google Scholar]
- 124.De Haro J, Acin F, Medina FJ, Lopez-Quintana A, March JR. Relationship between the plasma concentration of C-reactive protein and severity of peripheral arterial disease. Clin Med. 2009;3:1–7. doi: 10.4137/cmc.s1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Stokes KY, Dugas TR, Tang Y, Garg H, Guidry E, Bryan NS. Dietary nitrite prevents hypercholesterolemic microvascular inflammation and reverses endothelial dysfunction. Am J Physiol Heart Circ Physiol. 2009;296:H1281–1288. doi: 10.1152/ajpheart.01291.2008. [DOI] [PubMed] [Google Scholar]