Abstract
Protamine has been widely used as a pharmaceutical product and natural food preservative. However, few studies have been conducted to assess the beneficial function of dietary protamine. This study examined the effects of dietary salmon protamine on serum and liver lipid levels and the expression levels of genes encoding proteins involved in lipid homeostasis in the liver of rats. Groups of male Wistar rats were fed AIN93G diet containing 2% or 5% protamine. After 4 weeks of feeding these diets, markedly decreased serum and liver cholesterol (CHOL) and triacylglycerol levels were noted. Increased activity of liver carnitine palmitoyltransferase-2 and acyl-CoA oxidase, which are key enzymes of fatty acid β-oxidation in the mitochondria and peroxisomes, was found in rats fed on protamine. Furthermore, rats fed protamine showed enhanced fecal excretion of CHOL and bile acid and increased liver mRNA expression levels of ATP-binding cassette (ABC) G5 and ABCG8, which form heterodimers and play a major role in the secretion of CHOL into bile. The decrease in triacylglycerol levels in protamine-fed rats was due to the enhancement of liver β-oxidation. Furthermore, rats fed protamine exhibited decreased CHOL levels through the suppression of CHOL and bile acid absorption and the enhancement of CHOL secretion into bile. These results suggest that dietary protamine has beneficial effects that may aid in the prevention of lifestyle-related diseases such as hyperlipidemia and atherosclerosis.
Keywords: Protamine, lipid metabolism, β-oxidation, cholesterol, triacylglycerol
Introduction
In Western industrialized countries, the incidence of lifestyle-related diseases, such as atherosclerosis, hypertension, obesity, and hyperlipidemia, has been increasing in the last few decades [1]. Dietary therapy is important and could be considered as the first choice treatment or at least as important as medical treatment. Epidemiological evidence from Greenland Inuit and Japanese fishing villages suggests that eating fish and marine animals can prevent coronary heart disease (CHD) [2,3]. Dietary studies from many investigators have similarly shown that regular fish intake affects several humoral and cellular factors involved in CHD and may prevent atherosclerosis, thrombosis, and sudden cardiac death [4]. Some researchers have indicated that the beneficial effects of fish are attributable to their n-3 polyunsaturated fatty acids (PUFA), such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) [5]. Many studies have reported that EPA and DHA have beneficial effects on the prevention of hyperlipidemia and atherosclerosis by decreasing the serum triacylglycerol (TG) content [6]. However, it is not possible to explain these health functions of fish-based foods only in terms of EPA and DHA. In fact, general dietary habits include not only fish oil but also the fish itself, which provide nutrients such as proteins as well as fat [7]. On the other hand, some studies have indicated that fish meat protein also influences blood lipid contents and stimulates fibrinolysis in experimental animals [8,9]. Furthermore, we have previously reported the effects of dietary fish protein on serum and liver lipid concentrations in rats [10].
In the past, the potential health functions of fish tissues, except for muscle, have attracted little attention. For example, for the testes and ovaries, which are considered as edible parts, there is only information concerning the high levels of cholesterol (CHOL) and nucleic acids. Therefore, we focused on the effect of protamine from salmon milt on lipid metabolism. Protamine from salmon milt, also called salmine, has a low molecular weight of around 4,000-5,000, which plays a role in protecting DNA from being damaged. As a pharmaceutical product, protamine acts as an antidote to heparin, maintains antihyperglycemic effects together with insulin, and also functions to restrict fat absorption in the intestine [11]. In addition, protamine has also been widely used as a natural food preservative, because protamine has a strong antibacterial effect [12]. Previous studies showed that protamine strongly inhibited the hydrolysis of trioleoylglycerol emulsified with phosphatidylcholine [13] and protamine reduce weight gain and body fat accumulation through the inhibition of dietary fat absorption [14]. However, few studies have evaluated the beneficial effect of protamine besides the inhibition of fat absorption. To examine the effect of dietary protamine on lipid metabolism, we evaluated serum and liver lipid contents, lipid metabolizing enzyme activities in the liver, the excretion of fecal lipid, and the expression mRNA levels of CHOL-related enzymes in rats.
Materials and Methods
Materials
Salmon protamine was provided by Asama Chemical Co., Ltd. (Tokyo, Japan). AIN-93G vitamin mix, AIN-93G mineral mix, α-cornstarch, β-cornstarch, cellulose, sucrose, and casein were purchased from Oriental Yeast Co., Ltd (Tokyo, Japan). L-Cystine, choline bitartrate, and soybean oil were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). All other chemicals used were of reagent grade.
Animal care and experimental diets
Five-week-old male Wistar rats obtained from Shimizu Laboratory Supplies Co., Ltd (Kyoto, Japan) were housed in plastic cages in an air-conditioned room (temperature, 20-22℃; humidity, 55-65%; lights on, 08:00-20:00 h). After acclimation for 3 days with a diet prepared according to that recommended by the American Institute of Nutrition (AIN-93G) [15], rats were divided randomly into three groups and given free access to water and the experimental diet. Table 1 shows the composition of the experimental diets prepared according to AIN-93G. Experimental diet consumption and body weight were recorded every two days. Feces were collected from each group every 24 hours for seven days prior to sacrifice. After treatment with the experimental diets for 4 weeks, rats were weighed and sacrificed under Nembutal (Dainippon Sumitomo Pharma Co., Ltd, Osaka, Japan) anesthesia. Rats were not fasted before being sacrificed because food deprivation prior to sacrifice leads to a significant down-regulation of the genes involved in fatty acid synthesis and CHOL metabolism [16]. Blood was collected from the descending aorta, and serum was prepared by centrifugation at 1,500 × g for 15 min and then stored at -80℃ until analysis. Liver and abdominal white adipose tissue (WAT) were rapidly removed in their entirety and were weighed, rinsed, frozen in liquid nitrogen, and kept at -80℃. Aliquots of the liver were taken for mRNA expression analysis and stored in RNA-Later Storage Solution (Sigma Chemical Co., St. Louis, MO, USA). The experimental protocol was reviewed and approved by the Animal Ethics Committee of Kansai Medical University and followed the "Guide for the Care and Use of Experimental Animals" of the Prime Minister's Office of Japan.
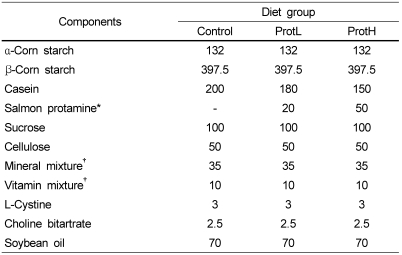
Table 1.
Composition of the experimental diets (g/kg)
Diets were prepared based on the AIN-93G composition.
*Protamine purity of salmon protamine was 83.5%.
†AIN-93G formula
Analysis of experimental diets and salmon protamine amino acid composition
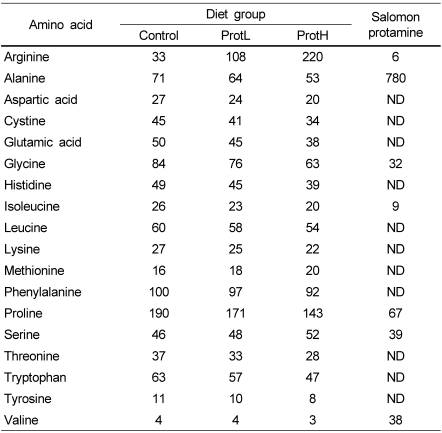
The experimental diets and salmon protamine amino acid composition was analyzed by high-performance liquid chromatography as follows. The samples were hydrolyzed using 6 M HCl. The hydrolyzed samples were subjected to HPLC, in which the samples were automatically derivatized using о-phthalaldehyde and separated on a reversed-phase column (GL Sciences Inertsil ODS-3, 5 µm, 250 × 4.6 mm) at 40℃ with 25 mM sodium phosphate buffer (pH 7.0) and acetonitrile using a gradient program at a flow rate of 1.0 mL/min. The eluate was monitored by fluorescence detection with an excitation wavelength of 340 nm and emission wavelength of 450 nm. The identification and quantitation of each amino acid were carried out using commercially available authentic standard mixtures and a CR-6A Chromatopac integrator (Shimadzu, Kyoto, Japan). Table 2 summarizes the amino acid composition of each diet and protamine.
Table 2.
Amino acid contents of the experimental diets and salmon protamine (g/kg protein)
ND, Not detected; ProtL, diet containing 2% protamine; ProtH, diet containing 5% protamine
Analysis of serum and liver lipids
Serum total lipid (TL), phospholipids (PL), TG, CHOL, high density lipoprotein-cholesterol (HDL-C), and low density lipoprotein-cholesterol (LDL-C) contents were analyzed using an Olympus AU5431 automatic analyzer.
The very low density lipoprotein (VLDL) fraction was isolated from the fresh serum by ultracentrifugation (Himac CS-100, Hitachi Koki Co., Ltd., Tokyo, Japan) at a density of < 1.006 g/ml after adjustment with NaCl, and then the chylomicron fraction was removed because the rats were not fasted before sacrifice [17]. Serum VLDL TG and CHOL contents were analyzed using enzymatic kits obtained from Wako Pure Chemical Industries, Ltd. (Osaka, Japan).
Liver lipids were extracted using a mixture of chloroform: methanol:water (1:1:0.9, v/v). The liver TG content was determined as follows; liver lipids were dissolved in an equal volume of dimethylsulfoxide and then used to determine the TG content using an enzymatic assay kit (Triglyceride-E-Test Wako, Wako Pure Chemical Industries, Ltd., Osaka, Japan). The liver CHOL content was analyzed using a SE-30 column (Shinwa Chemical Industries LTD., Kyoto, Japan) with a GC-14B gas chromatograph and 5α-cholestane as an internal standard. The liver PL content was measured by phosphorus analysis of liver lipids, and the liver protein content was determined by the method of Lowry et al. [18] using bovine serum albumin as a standard.
Preparation of the liver and enzyme activity assay
Liver tissue was homogenized in 10 volumes of a 0.25 M sucrose solution containing 1 mM EDTA-2Na in 3 mM Tris-HCl buffer (pH 7.4). The homogenate was centrifuged at 500 × g for 10 min at 4℃. The supernatant was recentrifuged at 9,000 × g for 10 min at 4℃ to sediment the mitochondria, and the remaining supernatant was collected. Furthermore, the supernatant was ultracentrifuged at 105,000 × g for 60 min at 4℃ to give a remaining supernatant and a microsome fraction.
Acyl-CoA oxidase (ACO, EC 1.3.3.6) activity was assayed in the 500 × g supernatant fraction of the liver homogenate [19]. Carnitine palmitoyltransferase-2 (CPT2, EC 2.3.1.21) activity in the mitochondrial fraction was assayed as described by Markwell et al. [20]. The activities of fatty acid synthase (FAS) [21], acetyl-CoA carboxylase (ACC, EC 6.4.1.2) [22], and glucose-6-phosphate dehydrogenase (G6PDH, EC 1.1.1.49) [23] in the 105,000 × g supernatant fraction and microsomal phosphatidate phosphatase (PAP, EC 3.1.3.4) [24] in the microsome fraction were assayed spectrophotometrically. The protein content of various fractions was determined according to the method of Lowry et al. [18].
Analysis of the fecal lipids and nitrogen
Total bile acid content in the feces was determined as µmoles 3α-hydroxysteroid based on the molar extinction coefficient of NADH at 340 nm, and fecal CHOL content was determined by gas-liquid chromatography using a SE-30 column with a GC-14B gas chromatograph (Shimadzu, Kyoto, Japan) and 5α-cholestane as an internal standard. Fecal fatty acid content was determined by the method of van de Kamer et al. [25]. Fecal protein content was determined by the Kjeldahl method. The apparent digestibility of protein was calculated as Apparent digestibility (%) = (Protein intake - Fecal protein)/Protein intake × 100.
Analysis of liver mRNA expression
Total RNA was extracted from the liver using an RNeasy Mini Kit (Qiagen, Tokyo, Japan) in accordance with the manufacturer's protocol. Then, cDNA was synthesized from total RNA using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems Japan Ltd., Tokyo, Japan). Real-time quantitative polymerase chain reaction (PCR) analysis was performed using an automated sequence detection system (ABI Prism 7000; Applied Biosystems Japan Ltd., Tokyo, Japan). 3-Hydroxy-3-methylglutaryl-CoA reductase (HMGR), cholesterol 7α-hydroxylase (CYP7A1), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA expression levels were determined using TaqMan Gene Expression Assays (Applied Biosystems Japan Ltd., Tokyo, Japan). PCR Primers (HMGR: Rn00565598_m1; CYP7A1: Rn00564065_m1; GAPDH: Rn99999916_s1) were purchased from Applied Biosystems (Japan Ltd., Tokyo, Japan). In addition, low density lipoprotein receptor (LDLR), scavenger receptor class B type 1 (SR-B1), ATP-binding cassette (ABC) A1 (ABCA1), ABCG5, ABCG8, and GAPDH mRNA expression levels were determined using SYBR Green PCR Master Mix (Applied Biosystems, Tokyo, Japan). PCR primers (Table 3) for LDL-R, SR-B1, ABCA1, ABCG5, ABCG8, and GAPDH were designed using Primer Express 3.0 software (Applied Biosystems, Tokyo, Japan). The PCR cycling conditions were 50℃ for 2 min and 95℃ for 10 min, followed by 40 cycles of 95℃ for 15 sec and 60℃ for 1 min. The expression level of the housekeeping gene GAPDH served as an internal control and was used for normalization.
Table 3.
Sequences of the specific primers used for reverse transcriptase polymerase chain reaction analysis
GAPDH, glyceraldehyde-3-phosphate dehydrogenase; LDL-R, low density lipoprotein receptor; SR-B1, scavenger receptor class B type 1; ABCA1, ATP-binding cassette A1; ABCG5, ATP-binding cassette G5; ABCG8, ATP-binding cassette G8; F, forward; R, reverse
Statistical analysis
Data are presented as means ± SD of seven rats. Statistical differences between multiple groups were determined by analysis of variance (ANOVA). Statistical comparisons were made using the Tukey-Kramer test. Difference were considered significant at P < 0.05.
Results
Growth parameters and organ weights
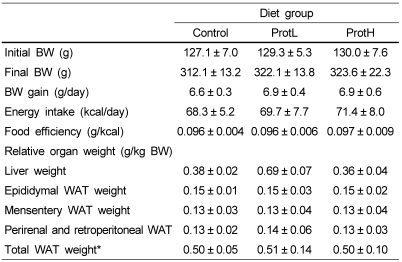
Table 4 shows body weight, body weight gain, food intake, food efficiency, relative liver, and various WAT weights. There were no significant differences in body weight, body weight gain, food intake, or food efficiency among the groups. Dietary protamine did not affect the growth of rats. In addition, relative liver and various WAT weights did not show any differences among the groups.
Table 4.
Initial body weight, final body weight, body weight gain, energy intake, energy efficiency ratio, relative liver weight, and various WAT weights of rats fed the experimental diets for 4 weeks
Data are means ± SD (n = 7). Values not sharing a common letter are significantly different at P < 0.05. Data were analyzed by Tukey-Kramer test. Rats were fed the experimental diets for 4 weeks. Food consumption and body weight were recorded every two days. The liver and WAT weights were obtained after sacrifice.
BW, body weight; WAT, white adipose tissue
*Total WAT = epididymal WAT + mesentery WAT + perirenal and retroperitoneal WAT
Serum and liver lipid parameters
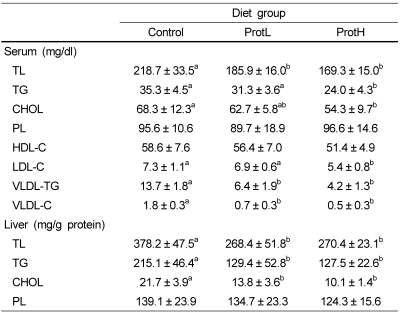
Table 5 shows the serum lipid and liver lipid contents. The serum TL, VLDL-TG, and VLDL-C contents in the low protamine (2%; ProtL) and high protamine (5%; ProtH) groups were significantly lower than in the control group, and there was no difference between the ProtL and ProtH groups. The serum TG, CHOL, and LDL-C contents were significantly lower the ProtH group compared with the control group. The serum HDL-C content in rats fed protamine tended to be lower than in rats fed casein, but not to a significant degree. No differences were found in serum PL among the groups.
Table 5.
Lipid content in the serum and liver of rats fed the experimental diets for 4 weeks
Data are means ± SD (n = 7). Values not sharing a common letter are significantly different at P < 0.05. Data were analyzed by Tukey-Kramer test. The serum was obtained from blood in the portal vein. The serum lipid contents were measured using an Olympus AU5431 automatic analyzer.
TL, total lipid; TG, triacylglycerol; CHOL, cholesterol; PL, phospholipid; HDL-C, high density lipoprotein-cholesterol; LDL-C, low density lipoprotein-cholesterol; VLDL-TG, very low density lipoprotein-triacylglycerol; VLDL-C, very low density lipoprotein-cholesterol
The liver TL, TG, and CHOL contents in rats fed protamine were markedly lower than in rats fed casein. Furthermore, there was no difference between the ProtL and ProtH groups in the liver TL, TG, and CHOL contents. No differences were found in liver PL among the groups.
Liver enzyme activity
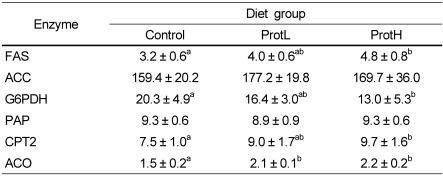
We assayed the activities of fatty acid β-oxidation and fatty acid synthesis related enzymes to investigate the regulatory effect of dietary protamine on fatty acid metabolism in the liver (Table 6). The activity of CPT2, a key enzyme of fatty acid β-oxidation in mitochondria, was significantly higher in the ProtH group than in the control group. ACO activity, a key enzyme of fatty acid oxidation in peroxisomes, was significantly higher in protamine fed rats than in the control group, and FAS activity, a key enzyme in fatty acid synthesis, was significantly higher in the ProtH group than in the control group. In contrast, there was no difference among the groups in the activity of ACC, another key enzyme in fatty acid synthesis. The activity of G6PDH, a key enzyme in the production of cellular NADPH, which is required for the biosynthesis of fatty acids and CHOL, was significantly lower in the ProtH group compared with the control group. The activity of PAP, a rate limiting enzyme for TG synthesis in the liver, showed no differences among the groups.
Table 6.
Activities of enzymes related to the fatty acid metabolic pathways in the liver of Wistar rats (nmol/min mg protein)
Data are means ± SD (n = 7). Values not sharing a common letter are significantly different at P < 0.05. Data were analyzed by Tukey-Kramer test. The activities of enzymes related to the fatty acid metabolism were analyzed by absorption spectrometer.
FAS, fatty acid synthase; ACC, acetyl-CoA carboxylase; G6PDH, glucose-6-phosphate dehydrogenase; PAP, phosphatidate phosphatase; CPT2, carnitine palmitoyl transferase-2; ACO, acyl-CoA oxidase
Fecal lipids and protein digestibility
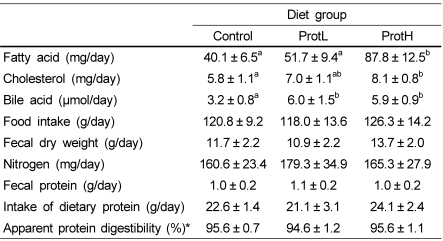
We determined the fecal lipid and protein contents to examine the effect of dietary protamine on fecal lipid excretion and apparent protein digestibility (Table 7). The fecal fatty acid content, which is an index of fat absorption in the small intestine, was significantly higher in the ProtH group than in the control or ProtL groups. The fecal CHOL content was significantly higher in the ProtH group than in the control group, and those in the ProtL group tended to be higher than in the control group. The fecal bile acid content was significantly higher in rats fed protamine than in rats fed casein. Fecal dry weight, nitrogen content, and apparent protein digestibility were not different among the groups.
Table 7.
Fecal fatty acid, cholesterol, bile acid, nitrogen contents, and protein digestibility in rats fed the experimental diets for 4 weeks
Data are means ± SD (n = 7). Values not sharing a common letter are significantly different at P < 0.05.
Feces were collected from each group every 24 hours for seven days prior to sacrifice.
*Apparent protein digestibility (%) = (intake of dietary protein - fecal protein) / intake of dietary protein × 100
mRNA expression levels
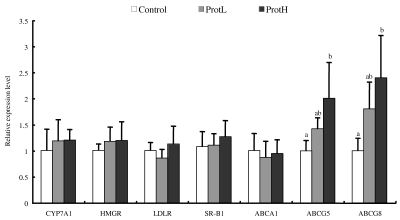
We determined the mRNA expression levels of enzymes and receptors related to CHOL metabolism in the liver tissue to evaluate the effect of dietary protamine using real-time quantitative PCR (Fig. 1). The expression levels of CYP7A1, HMGR, LDLR, SR-B1, and ABCA1 were not different among the groups. In contrast, the expression levels of ABCG5 and ABCG8, which play major roles in the secretion of CHOL into bile, were higher in the ProtH group than in the control group, while those in the ProtL group tended to be higher than in the control group.
Fig. 1.
The expression levels of enzymes related to cholesterol metabolism in the liver of Wistar rat fed diets of the control (□), ProtL (▒), and ProtH (■) diets. Data are means ± SD (n = 7). Relative values are presented as the ratio of each mRNA to GAPDH mRNA. Values not sharing a common letter are significantly different at P < 0.05. Data were analyzed by Tukey-Kramer test. The expression levels of enzymes related to cholesterol metabolism were analyzed by the real-time quantitative polymerase chain reaction method. CYP7A1, cholesterol 7α-hydroxylase; HMGR, 3-hydroxy-3methylglutaryl-CoA reductase; LDL-R, low density lipoprotein receptor; SR-B1, scavenger receptor class B type 1; ABCA1, ATP-binding cassette A1; ABCG5, ATP-binding cassette G5; ABCG8, ATP-binding cassette G8; GAPDH, glyceraldehyde-3-phosphate dehydrogenase
Discussion
Dietary therapy can be considered as the first choice treatment or at least as important as medical treatment. Dietary protein has been shown to affect lipid metabolism in humans and animals [26-28]. For example, it is known that soy protein relative to casein exerted hypocholesterolemic and hypotriglyceridemic effects in experimental animals [26-28]. In addition to this effect of plant protein, fish protein also seemed to influence blood lipid contents and stimulated fibrinolysis in experimental animals [8-10]. Protamine from salmon is rich in the basic amino acid arginine, but there have been few studies on the beneficial health effects of protamine. Therefore, this study examined the effect of protamine from salmon on lipid metabolism in serum and the liver compared with the effect of casein.
In the present study, we demonstrated that dietary protamine significantly lowered the serum and liver TG contents compared with dietary casein. The reasons for the decrease in serum and liver TG contents were thought to be related to the absorption of lipid from the small intestine [13,29], and we analyzed the effect of dietary protamine on fat absorption by determining the total amount of fatty acids in feces. The fecal fatty acids content was higher in the ProtH group than in the control group. Tsujita et al. [13] suggested that a basic protein or protamine strongly inhibited the hydrolysis of trioleoylglycerol emulsified with phosphatidylcholine. Moreover, the basic peptide polylysine is a candidate agent for the inhibition of intestinal lipid absorption while resisting proteolysis [29]. Thus, we hypothesized that the inhibition of fat absorption by basic peptides produced during the digestion of protamine, which contains large amounts of arginine corresponding to about 68 mol% of its total amino acids. A previous study suggested that protamine reduced the plasma TG concentration through the inhibition of dietary fat absorption [14]. However, in the present study, daily lipid (TG) intake was about 8.5 g, and daily fatty acids intake was about 8.0 g when converting TG into fatty acids. The daily excretion of fecal fatty acids was 40.1 ± 6.5 mg and 87.8 ± 12.5 mg for the control and ProtH groups, respectively. Consequently, the apparent lipid digestibility was about 99.5% and 99.0% for the control and ProtH groups, respectively. Therefore, we speculated that the dietary protamine increased fecal fatty acid excretion but did not extremely decrease the serum and liver TG contents.
Another reason considered for the decrease in serum and liver TG contents was the alteration of enzyme activity related to TG metabolism in the liver, and we examined the effect of dietary protamine on enzyme activity related to fatty acid β-oxidation and fatty acid synthesis. As shown in Table 6, CPT2 and ACO activities, which are key enzymes of fatty acid β-oxidation in mitochondria and peroxisomes, in the liver were higher in rats fed protamine than in rats fed casein. FAS activity, a rate limiting enzyme in fatty acid synthesis, was higher in rats fed protamine than in rats fed casein. Moreover, PAP activity, a key enzyme in the regulation of de novo TG synthesis, was not different among the groups. Therefore, we hypothesized that the decrease in liver TG content with the protamine-containing diets were attributed to the enhancement of fatty acid β-oxidation and not to the alteration of de novo TG synthesis. The increased FAS activity due to dietary protamine may be prevented by the enhancement of fatty acid β-oxidation.
In this study, the serum VLDL-TG and VLDL-C contents in rats fed protamine were significantly lower than in rats fed casein. VLDL, which is a TG-rich lipoprotein, is assembled and stored in the liver by microsomal triglyceride transfer protein and apolipoprotein B. In addition, VLDL hydrolyzes TG to fatty acid and monoacylglycerol by the action of lipoprotein lipase in a number of peripheral tissues, including adipose tissue, skeletal and cardiac muscle, and the mammary gland [30]. Further studies are necessary to clarify the effect of dietary protamine on lipoprotein metabolism of VLDL hydrolysis, assembly, and secretion in peripheral tissue and the liver.
Another interesting result was the effect of protamine on CHOL metabolism. The serum CHOL and LDL-C contents were significantly decreased in the ProtH group compared with the control group. In addition, the liver CHOL contents in rats fed protamine were significantly lower than in rats fed casein (Table 5). Nagata et al. [28] found that the degree of serum CHOL lowering depended on the extent fecal steroid excretion. In the present study, the fecal excretion of CHOL and bile acid were higher in rats fed protamine than in rats fed casein (Table 7). Higaki et al. [27] suggested that feeding soy protein stimulated the fecal excretion of bile acid and demonstrated that the increase in fecal bile acid was accompanied by an increase in fecal nitrogen excretion. The fecal excretion of bile acid suggested that hydrophobic peptides were produced by intestinal digestion of protein bound bile acid by hydrophobic interaction [31]. Moreover, Kayashita et al. [32] reported that a dietary buckwheat protein product had lower plasma CHOL content and enhanced excretion of fecal CHOL and nitrogen compared with rats fed casein, and there was a significant correlation between fecal CHOL and nitrogen. Thus, dietary proteins such as soy and buckwheat protein increased the fecal excretion of steroids concomitant with increased fecal nitrogen due to its low digestibility. However, dietary protamine increased the fecal excretion of CHOL and bile acid, although there was no influence on the fecal excretion of nitrogen. This result indicated the possible effect of dietary protamine on the fecal excretion of CHOL and bile acid by a new mechanism. The mechanism of fecal CHOL and bile acid excretion in rats fed protamine is not clear at present, and further experiments will be necessary to clarify this mechanism and the effects on the binding capacities of bile acid and the micellar solubility of CHOL and bile acid.
It is well known that serum and liver CHOL levels are maintained by a balance between CHOL biosynthesis and catabolism in the liver, uptake into the liver, and secretion and excretion from the liver. The decreased liver CHOL content found in rats fed protamine could be expected to arise from a decrease in CHOL synthesis, an increase in CHOL catabolism to bile acid, an increase in CHOL efflux, or a decrease in CHOL uptake. Therefore, we evaluate the mRNA expression levels of enzymes and receptors related to CHOL metabolism in the liver using quantitative real-time PCR. CYP7A1, a rate limiting enzyme of bile acid synthesis, HMGR, a rate limiting enzyme of CHOL synthesis, and LDLR and SR-B1, uptake serum lipoproteins, showed differences in rats fed protamine compared with in rats fed casein (Fig. 1). The CHOL lowering effect of dietary protamine was not attributable to the decrease in CHOL biosynthesis, catabolism, or uptake.
Much attention has been focused on the finding that control of the delivery and disposal of CHOL are regulated by membrane transporters of the ATP-binding cassette (ABC) superfamily [33]. We analyzed the liver mRNA expression levels of ABCA1, a transporter involved in the production of HDL, ABCG5 and ABCG8, which form heterodimers and play a major role in the secretion of CHOL into bile. The expression levels of ABCA1 were not different among the groups. Surprisingly, dietary protamine increased the expression levels of ABCG5 and ABCG8 in the liver. This results in decreased liver CHOL through the enhancement of the secretion of CHOL into bile. In addition, the increased expression levels of ABCG5 and ABCG8 also influenced the excretion of fecal CHOL. Hence, we suspected that the hypocholesterolemic effect of dietary protamine was attributable to the enhanced excretion of fecal CHOL and bile acid and the secretion of CHOL into bile.
These results showed that dietary protamine enhanced β-oxidation activities and the expression levels of ABCG5 and ABCG8 in the liver. We predicted that the alteration in lipid metabolism in rats fed on the protamine diet were possibly due to arginine, a large amount of which is present in protamine. Dietary L-arginine produces nitric oxide (NO) with NO synthase in virtually all mammalian cells and plays an important role in lipid metabolism [34]. Future studies are necessary to test this hypothesis.
This study showed that dietary salmon protamine, compared with dietary casein, had a TG lowering effect in serum and the liver, which at least in part arose by the enhancement of β-oxidation in the liver. The decrease in serum CHOL, LDL-C, and liver CHOL contents in rats fed protamine were suggested to be related to the enhancement of fecal excretion of CHOL and bile acid and the increased liver mRNA expression levels of ABCG5 and ABCG. In view of the fecal nitrogen results (Table 7), we suspected that the enhanced fecal excretion of CHOL and bile acid were possibly by a different mechanism from the effect of dietary soy protein. The observed reductions in serum TG, CHOL, and LDL-C and liver TG and CHOL in rats fed protamine suggested possible beneficial effects on the development and prevention lifestyle-related diseases, such as hyperlipidemia and atherosclerosis.
References
- 1.Formiguera X, Cantón A. Obesity: epidemiology and clinical aspects. Best Pract Res Clin Gastroenterol. 2004;18:1125–1146. doi: 10.1016/j.bpg.2004.06.030. [DOI] [PubMed] [Google Scholar]
- 2.Dyerberg J, Bang HO, Hjorne N. Fatty acid composition of the plasma lipids in Greenland Eskimos. Am J Clin Nutr. 1975;28:958–966. doi: 10.1093/ajcn/28.9.958. [DOI] [PubMed] [Google Scholar]
- 3.Iso H, Kobayashi M, Ishihara J, Sasaki S, Okada K, Kita Y, Kokubo Y, Tsugane S JPHC Study Group. Intake of fish and n3 fatty acids and risk of coronary heart disease among Japanese: the Japan Public Health Center-Based (JPHC) Study Cohort I. Circulation. 2006;113:195–202. doi: 10.1161/CIRCULATIONAHA.105.581355. [DOI] [PubMed] [Google Scholar]
- 4.He K, Song Y, Daviglus ML, Liu K, Van Horn L, Dyer AR, Greenland P. Accumulated evidence on fish consumption and coronary heart disease mortality: a meta-analysis of cohort studies. Circulation. 2004;109:2705–2711. doi: 10.1161/01.CIR.0000132503.19410.6B. [DOI] [PubMed] [Google Scholar]
- 5.Simopoulos AP. Omega-3 fatty acids in inflammation and autoimmune diseases. J Am Coll Nutr. 2002;21:495–505. doi: 10.1080/07315724.2002.10719248. [DOI] [PubMed] [Google Scholar]
- 6.Simopoulos AP. Omega-3 fatty acids in health and disease and in growth and development. Am J Clin Nutr. 1991;54:438–463. doi: 10.1093/ajcn/54.3.438. [DOI] [PubMed] [Google Scholar]
- 7.Dahl L, Bjørkkjaer T, Graff IE, Malde MK, Klementsen B. Fish--more than just omega 3. Tidsskr Nor Laegeforen. 2006;126:309–311. [PubMed] [Google Scholar]
- 8.Zhang X, Beynen AC. Influence of dietary fish proteins on plasma and liver cholesterol concentrations in rats. Br J Nutr. 1993;69:767–777. doi: 10.1079/bjn19930077. [DOI] [PubMed] [Google Scholar]
- 9.Murata M, Sano Y, Bannai S, Ishihara K, Matsushima R, Uchida M. Fish protein stimulated the fibrinolysis in rats. Ann Nutr Metab. 2004;48:348–356. doi: 10.1159/000081971. [DOI] [PubMed] [Google Scholar]
- 10.Hosomi R, Fukunaga K, Arai H, Nishiyama T, Yoshida M. Effects of dietary fish protein on serum and liver lipid concentrations in rats and the expression of hepatic genes involved in lipid metabolism. J Agric Food Chem. 2009;57:9256–9262. doi: 10.1021/jf901954r. [DOI] [PubMed] [Google Scholar]
- 11.Jaques LB. Protamine--antagonist to heparin. Can Med Assoc J. 1973;108:1291–1293. 1295–1297. [PMC free article] [PubMed] [Google Scholar]
- 12.Aspedon A, Groisman EA. The antibacterial action of protamine: evidence for disruption of cytoplasmic membrane energization in Salmonella typhimurium. Microbiology. 1996;142:3389–3397. doi: 10.1099/13500872-142-12-3389. [DOI] [PubMed] [Google Scholar]
- 13.Tsujita T, Matsuura Y, Okuda H. Studies on the inhibition of pancreatic and carboxylester lipases by protamine. J Lipid Res. 1996;37:1481–1487. [PubMed] [Google Scholar]
- 14.Duarte-Vázquez MA, García-Padilla S, Olvera-Ochoa L, González-Romero KE, Acosta-Iñiguez J, De la Cruz-Cordero R, Rosado JL. Effect of protamine in obesity induced by high-fat diets in rats. Int J Obes (Lond) 2009;33:687–692. doi: 10.1038/ijo.2009.78. [DOI] [PubMed] [Google Scholar]
- 15.Reeves PG, Nielsen FH, Fahey GC., Jr AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993;123:1939–1951. doi: 10.1093/jn/123.11.1939. [DOI] [PubMed] [Google Scholar]
- 16.Horton JD, Bashmakov Y, Shimomura I, Shimano H. Regulation of sterol regulatory element binding proteins in livers of fasted and refed mice. Proc Natl Acad Sci U S A. 1998;95:5987–5992. doi: 10.1073/pnas.95.11.5987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hatch FT. Practical methods for plasma lipoprotein analysis. Adv Lipid Res. 1968;6:1–68. [PubMed] [Google Scholar]
- 18.Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 19.Ide T, Watanabe M, Sugano M, Yamamoto I. Activities of liver mitochondrial and peroxisomal fatty acid oxidation enzymes in rats fed trans fat. Lipids. 1987;22:6–10. doi: 10.1007/BF02534867. [DOI] [PubMed] [Google Scholar]
- 20.Markwell MA, McGroarty EJ, Bieber LL, Tolbert NE. The subcellular distribution of carnitine acyltransferases in mammalian liver and kidney. A new peroxisomal enzyme. J Biol Chem. 1973;248:3426–3432. [PubMed] [Google Scholar]
- 21.Kelley DS, Nelson GJ, Hunt JE. Effect of prior nutritional status on the activity of lipogenic enzymes in primary monolayer cultures of rat hepatocytes. Biochem J. 1986;235:87–90. doi: 10.1042/bj2350087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tanabe T, Nakanishi S, Hashimoto T, Ogiwara H, Nikawa J, Numa S. Lipids Part C. Methods Enzymol. New York: Academic Press; 1981. Acetyl-CoA carboxylase from rat liver : EC 6.4.1.2 Acetyl-CoA: carbon-dioxide ligase (ADP-forming) pp. 5–16. [DOI] [PubMed] [Google Scholar]
- 23.Kelley DS, Kletzien RF. Ethanol modulation of the hormonal and nutritional regulation of glucose 6-phosphate dehydrogenase activity in primary cultures of rat hepatocytes. Biochem J. 1984;217:543–549. doi: 10.1042/bj2170543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ichihara K, Norikura S, Fujii S. Microsomal phosphatidate phosphatase in maturing safflower seeds. Plant Physiol. 1989;90:413–419. doi: 10.1104/pp.90.2.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van de Kamer JH, ten Bokkel Huinink H, Weyers HA. Rapid method for the determination of fat in feces. J Biol Chem. 1949;177:347–355. [PubMed] [Google Scholar]
- 26.Moriyama T, Kishimoto K, Ngai K, Urade R, Ogawa T, Uthumi S, Maruyama N, Maebuchi M. Soybean beta-conglycinin diet suppresses serum triglyceride levels in normal and genetically obese mice by induction of beta-oxidation, downregulation of fatty acid synthase, and inhibition of triglyceride absorption. Biosci Biotechnol Biochem. 2004;68:352–359. doi: 10.1271/bbb.68.352. [DOI] [PubMed] [Google Scholar]
- 27.Higaki N, Sato K, Suda H, Suzuka T, Komori T, Saeki T, Nakamura Y, Ohtsuki K, Iwami K, Kanamoto R. Evidence for the existence of a soybean resistant protein that captures bile acid and stimulates its fecal excretion. Biosci Biotechnol Biochem. 2006;70:2844–2852. doi: 10.1271/bbb.60237. [DOI] [PubMed] [Google Scholar]
- 28.Nagata Y, Tanaka K, Sugano M. Serum and liver cholesterol levels of rats and mice fed soy-bean protein or casein. J Nutr Sci Vitaminol (Tokyo) 1981;27:583–593. doi: 10.3177/jnsv.27.583. [DOI] [PubMed] [Google Scholar]
- 29.Tsujita T, Sumiyoshi M, Takaku T, Momsen WE, Lowe ME, Brockman HL. Inhibition of lipases by epsilon-polylysine. J Lipid Res. 2003;44:2278–2286. doi: 10.1194/jlr.M300151-JLR200. [DOI] [PubMed] [Google Scholar]
- 30.Fielding BA, Frayn KN. Lipoprotein lipase and the disposition of dietary fatty acids. Br J Nutr. 1998;80:495–502. doi: 10.1017/s0007114598001585. [DOI] [PubMed] [Google Scholar]
- 31.Iwami K, Kitagawa M, Ibuki F. Effect of dietary proteins and/or their digestive products on intestinal taurocholate absorption. J Nutr Sci Vitaminol (Tokyo) 1990;36:S141–S146. doi: 10.3177/jnsv.36.supplementii_s141. [DOI] [PubMed] [Google Scholar]
- 32.Kayashita J, Shimaoka I, Nakajoh M, Yamazaki M, Kato N. Consumption of buckwheat protein lowers plasma cholesterol and raises fecal neutral sterols in cholesterol-Fed rats because of its low digestibility. J Nutr. 1997;127:1395–1400. doi: 10.1093/jn/127.7.1395. [DOI] [PubMed] [Google Scholar]
- 33.Tamehiro N, Shigemoto-Mogami Y, Kakeya T, Okuhira K, Suzuki K, Sato R, Nagao T, Nishimaki-Mogami T. Sterol regulatory element-binding protein-2- and liver X receptor-driven dual promoter regulation of hepatic ABC transporter A1 gene expression: mechanism underlying the unique response to cellular cholesterol status. J Biol Chem. 2007;282:21090–21099. doi: 10.1074/jbc.M701228200. [DOI] [PubMed] [Google Scholar]
- 34.García-Villafranca J, Guillén A, Castro J. Involvement of nitric oxide/cyclic GMP signaling pathway in the regulation of fatty acid metabolism in rat hepatocytes. Biochem Pharmacol. 2003;65:807–812. doi: 10.1016/s0006-2952(02)01623-4. [DOI] [PubMed] [Google Scholar]