Abstract
A male patient diagnosed elsewhere as having multiple sclerosis (MS) was recently referred to our MS centre. Despite the presence of scattered T2-hyperintense MS-like lesions on MRI and cerebrospinal fluid (CSF) oligoclonal bands, his MS diagnosis was unpersuasive. Distal symmetric hypotonia, tendon areflexia and distal muscle weakness were present. A mostly demyelinating sensory polyneuropathy was disclosed at electroneurography. Serum IgM band, free monoclonal light chains and increased anti-myelin-associated glycoprotein IgM were detected. At 18 months later, and after three intravenous Ig treatments, a clinical electroneurographic improvement was evident along with the disappearance of some brain MRI lesions, reduction of serum anti-myelin-associated glycoprotein (MAG) IgM level and of the number of CSF oligoclonal bands. Although the cause/effect relation cannot be proven, we hypothesise that not only peripheral but also central demyelination may be related to the presence of anti-MAG antibodies with central nervous system (CNS) patterns on MRI resembling those seen in MS.
BACKGROUND
The coexistence of multiple sclerosis (MS) and anti-myelin-associated glycoprotein (MAG) polyneuropathy (PN) is an exceedingly rare condition. To our knowledge only one case has been reported so far, in one patient experiencing worsening MS after treatment with rituximab for the anti-MAG PN.1
Here, we describe another such unique patient recently referred to our MS Centre. His MS diagnosis was unconvincing and the concomitant anti-MAG PN ignored. His clinical history leads us to significant pathophysiological implications that may be of interest also for the readers.
CASE PRESENTATION
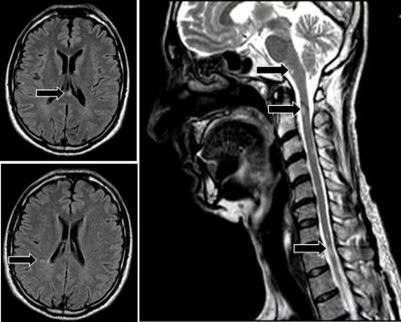
A 44-year-old man was referred to our MS centre for the initiation of a treatment with interferon-β. A MS diagnosis with a 3.5 Kurtzke Expanded Disability Status Scale (EDSS) score2 was given elsewhere based on the history of hands and feet paresthaesia, mild paraparesis, a total of six T2-weighted and fluid-attenuated inversion recovery (FLAIR)-weighted hyperintense MS-like white matter lesions without contrast enhancement (indicated with black arrows on fig 1) on brain and spinal MRI. Cerebrospinal fluid (CSF) protein level and cell count were normal while 10 oligoclonal bands (OCB) were detected.
Figure 1.
Fluid-attenuated inversion recovery (FLAIR) T2-weighted hyperintense multiple sclerosis (MS)-like white matter lesions on brain MRI; no contrast enhancement was present on corresponding T1-weighted images (not shown). Multiple T2-weighted hyperintense signals on spinal MRI are also shown and indicated with black arrows.
However, on neurological examination the patient showed diffuse tendon areflexia with distal hypotonia, mild gait ataxia with positive Romberg test, decreased vibratory and pinprick sensation on lower limbs. Bilateral foot drop (steppage gait) was evident. According to the modified Medical Research Council Scale, extensor digitorum, extensor hallucis and tibialis anterior muscle strengths were of grade 4 on the right side and grade 3 on the left side. A left Babinski response was present.
Paresthaesia on hands and feet started 9 years previously, followed by a very slowly increasing gait ataxia and footdrop. The clinical picture was substantially more stable during the last 4 years. The rest of his medical, social and working history was negligible.
INVESTIGATIONS
A subsequent electrophysiological study showed a prevalently demyelinating sensory motor (sensory greatly prevailing) PN, increased terminal motor latency in the peroneal nerves and decreased to abolished sensitive potential amplitudes of sural nerves. No conduction blocks were detected. Visual evoked potentials were normal.
Repeated serum protein electrophoreses (average IgM 840 mg/dl, range 40–230) and immunofixation revealed an IgM band and free monoclonal λ light chains allowing a haematological diagnosis of IgM monoclonal gammopathy of undetermined origin (MGUS). High level of anti-MAG IgM antibodies were detected in serum (3000 UA, normal values <1000). Other anti-neuron antibodies, including anti-Hu, were normal.
Routine tests, including thyroid hormones, glucose test, vitamin B12 and chest x rays were normal. Additional tests requested for the differential diagnosis (see list below), such as tumoural and hepatitis C markers, bone x ray, lactic acid, inflammatory, autoimmune and infectious serology, were also negative or within the normal range. Family history was negligible.
DIFFERENTIAL DIAGNOSIS
Concomitant central and peripheral demyelination.
Primary progressive MS and anti-MAG polyneuropathy?
Primary progressive MS and chronic inflammatory demyelinating polyneuropathy (CIDP)?
Myeloma, Waldestrom, paraneoplastic (anti-Hu)?
Inflammatory: immune disease (Sjögren, Lambert–Eaton myasthenic syndrome (LES), vasculitis, sarcoidosis)?
Infectious (hepatitis C virus, syphilis, HIV)?
Endocrine or metabolic?
Hereditary leukodystrophies, heredoataxia, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL)?
TREATMENT
Treatment with high-dose intravenous methylprednisolone (1 g/day for 5 days) proved to be clinically ineffective after 2 weeks.
By avoiding the possible MS-detrimental rituximab option,1 after signing an informed consent and with a diagnosis of anti-MAG-associated PN, the patient was treated with intravenous Ig (0.4 g/kg/day) for 5 days. Symptoms began to subside 3 weeks later. After 3 months of sustained benefit, symptoms slowly relapsed and, for this reason, he underwent a second intravenous Ig treatment. The therapy was again safe, and symptoms start to subside after 3 weeks. A third intravenous Ig course was administered after 4 months of clinical comfort followed by initial symptoms of re-exacerbation.
OUTCOME AND FOLLOW-UP
At 18 months after our first examination and 2 months after the third intravenous Ig course, a clear symmetrical sensory motor improvement was clinically detectable. Gait ataxia was reduced and the Romberg test became negative (the patient now sways but does not fall when the eyes are closed). Extensor digitorum, extensor hallucis and tibialis anterior muscle strengths were of grade 5 on the right side and grade 4 on the left side. Steppage gait disappeared. Paresthaesia disappeared on hands, and was reduced on feet. Babinski response persisted.
Although sensory motor conduction velocities kept stably reduced, electroneurography demonstrated a shortening of the distal motor latency in the peroneal nerves (>30%) and a threefold increase of the sensitive potential amplitudes of the left sural nerve. Interestingly, two T2-weighted hyperintense brain MRI lesions disappeared while spinal MRI was unchanged.
CSF OCB number was reduced (from 10 to 4), CSF anti-MAG antibodies were undetectable and serum anti-MAG antibody level was within the normal range.
DISCUSSION
With some exceptions,3,4 demyelinating polyneuropathies associated with IgM paraproteinaemia and anti-MAG antibodies have clinical, electrophysiological as well as pathological homogeneity.5,6 Anti-MAG PN is a predominantly large fibre sensory neuropathy, with sensory ataxia as major hallmark.7 Weakness, when present, is predominantly distal and develops later in the course, sometimes years from sensory symptoms.8 The clinical course is typically slowly or very slowly progressive. Studies on nerve conduction show a pattern of demyelination with or without concomitant axonal damage, a lower frequency of conduction blocks and an increase in distal motor latencies as compared to other forms of chronic inflammatory demyelinating PN (CIDP).5
MAG is a common glycoprotein in central and peripheral nervous systems. Despite variations in their affinity do occur, antibodies to MAG bind PNS and CNS myelin.9 Monoclonal antibodies to MAG injected into mammalian optic nerve produce a demyelination associated with ultrastructural patterns observed pathologically in MS.10
While clinical and electroneurographic patterns, together with their transient improvement after the immunomodulatory treatment,11 allow a diagnosis of anti-MAG PN, several concerns pertain the underlying CNS disease mimicking MS.
The presence of a neurological deterioration, of a pyramidal sign (Babinski) of non-punctuate demyelinative lesions at brain and spinal MRI (see fig 1) and of OCB at the CSF examination, all would lead the patient to meet existing criteria for a diagnosis of MS and, with the lack of clear onset and relapses given, a primary progressive form ought to be considered.12,13 However, some controversial points also need to be taken into great account: the normal latency of the visual evoked potentials,12 the lack of a persistent clinical progression, the very limited “central” signs (Babinski) and the very mild neurological deterioration 9 years after disease onset make this MS diagnosis at least uncertain.13 Finally, and at a postdiagnostic level, the very unusual reduction of the OCB number may also be antithetic with a diagnosis of MS since OCB are known to represent a stable abnormality in MS.14
Our observations should not be interpreted as if we think the initial diagnosis of MS was dramatically incorrect and/or the MRI abnormalities of an incidental kind. Dissecting MS from MS-like diseases is sometime tough going and not infrequently patients treated for MS have a MS-mimicking disease instead.15 Our observation should be interpreted on the basis of the current literature9,10 which offers the possibility to hypothesise that the underlying anti-MAG IgM paraproteinaemia has a full potential to induce a central demyelination with MRI features typical of MS.
Indeed, our case significantly differs from the one reported in the literature. The patient described by Benedetti and colleagues1 had a clear relapsing/remitting followed by a secondary progressive, steroid-responsive MS course. Additionally, MS onset and diagnosis preceded by at least 18 years the onset of symptoms and signs due to the anti-MAG PN. By contrast, our case clearly lacks a definite temporal separation between central and peripheral symptoms and signs.
In conclusion, although the direct cause/effect relation between anti-MAG and MS-like MRI lesions cannot be proven, having assessed most plausible differential diagnoses, our case may be the first description of a concomitant central demyelinating disease in pathogenic link with the presence of anti-MAG IgM antibodies during the course of a benign IgM paraproteinaemia with polyneuropathy.
LEARNING POINTS
Clinical examination is the most important tool in multiple sclerosis (MS) diagnosis and the concept of “no better explanation” has to be taken into great account.12
Even the primary progressive form of MS has its own diagnostic criteria that ought to be fulfilled.13
In our short-run case (16 months), intravenous Ig prove to be safe and beneficial in the treatment of polyneuropathy with anti-myelin-associated glycoprotein (MAG) monoclonal IgM paraproteinaemia.
Footnotes
Competing interests: None.
Patient consent: Patient/guardian consent was obtained for publication.
REFERENCES
- 1.Benedetti L, Franciotta D, Vigo T, et al. Relapses after treatment with rituximab in a patient with multiple sclerosis and anti myelin-associated glycoprotein polyneuropathy. Arch Neurol 2007; 64: 1531–3 [DOI] [PubMed] [Google Scholar]
- 2.Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an Expanded Disability Status Scale (EDSS). Neurology 1983; 33: 1444–52 [DOI] [PubMed] [Google Scholar]
- 3.Isoardo G, Migliaretti G, Ciaramitaro P, et al. Differential diagnosis of chronic dysimmune demyelinating polyneuropathies with and without anti-MAG antibodies. Muscle Nerve 2005; 31: 52–8 [DOI] [PubMed] [Google Scholar]
- 4.Ellie E, Vital A, Steck A, et al. Neuropathy associated with benign anti-myelin-associated glycoprotein IgM gammopathy: clinical, immunological neurophysiological, pathological findings and response to treatment in 33 cases. J Neurol 1996; 243: 34–43 [DOI] [PubMed] [Google Scholar]
- 5.Cocito D, Durelli L, Isoardo G. Different clinical, electrophysiological and immunological features of CIDP associated with paraproteinaemia. Acta Neurol Scand 2003; 108: 274–80 [DOI] [PubMed] [Google Scholar]
- 6.Notermans NC, Franssen H, Eurelings M, et al. Diagnostic criteria for demyelinating polyneuropathy associated with monoclonal gammopathy. Muscle Nerve 2000; 23: 73–9 [DOI] [PubMed] [Google Scholar]
- 7.Ropper AH, Gorson KC. Neuropathy associated with paraproteinemia. New Engl J Med 1998; 338: 1601–7 [DOI] [PubMed] [Google Scholar]
- 8.Smith IS. The natural history of chronic demyelinating neuropathy associated with benign IgM paraproteinemia. Brain 1994; 117: 949–57 [DOI] [PubMed] [Google Scholar]
- 9.Fluri F, Ferracin F, Erne B, et al. Microheterogeneity of anti-myelin-associated glycoprotein antibodies. J Neurol Sci 2003; 207: 43–9 [DOI] [PubMed] [Google Scholar]
- 10.Sergott RC, Brown MJ, Lisak RP, et al. Antibody to myelin-associated glycoprotein produces central nervous system demyelination. Neurology 1988; 38: 422–6 [DOI] [PubMed] [Google Scholar]
- 11.Lunn MPT, Nobile-Orazio E. Immunotherapy for IgM antimyelin-associated glycoprotein paraprotein-associated peripheral neuropathies. Cochrane Database Syst Rev 2003; 1: CD002827. [DOI] [PubMed] [Google Scholar]
- 12.Polman CH, Reingold SC, Edan G, et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 2005; 58: 840–6 [DOI] [PubMed] [Google Scholar]
- 13.Jenkins TM, Khaleeli Z, Thompson AJ. Diagnosis and management of primary progressive multiple sclerosis. Minerva Med 2008; 99: 141–55 [PubMed] [Google Scholar]
- 14.Link H, Huang YM. Oligoclonal bands in multiple sclerosis cerebrospinal fluid: an update on methodology and clinical usefulness. J Neuroimmunol 2006; 180: 17–28 [DOI] [PubMed] [Google Scholar]
- 15.Poser CM. Misdiagnosis of multiple sclerosis and beta-interferon. Lancet 1997; 349: 1916. [DOI] [PubMed] [Google Scholar]