Abstract
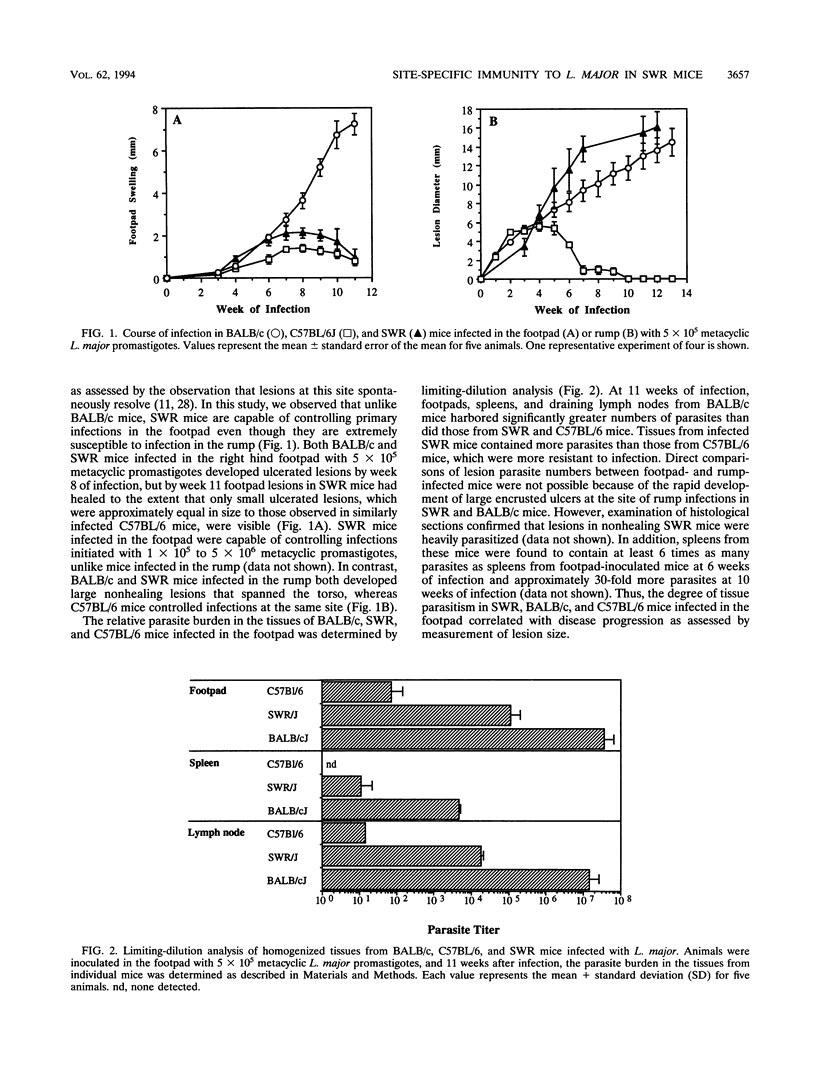
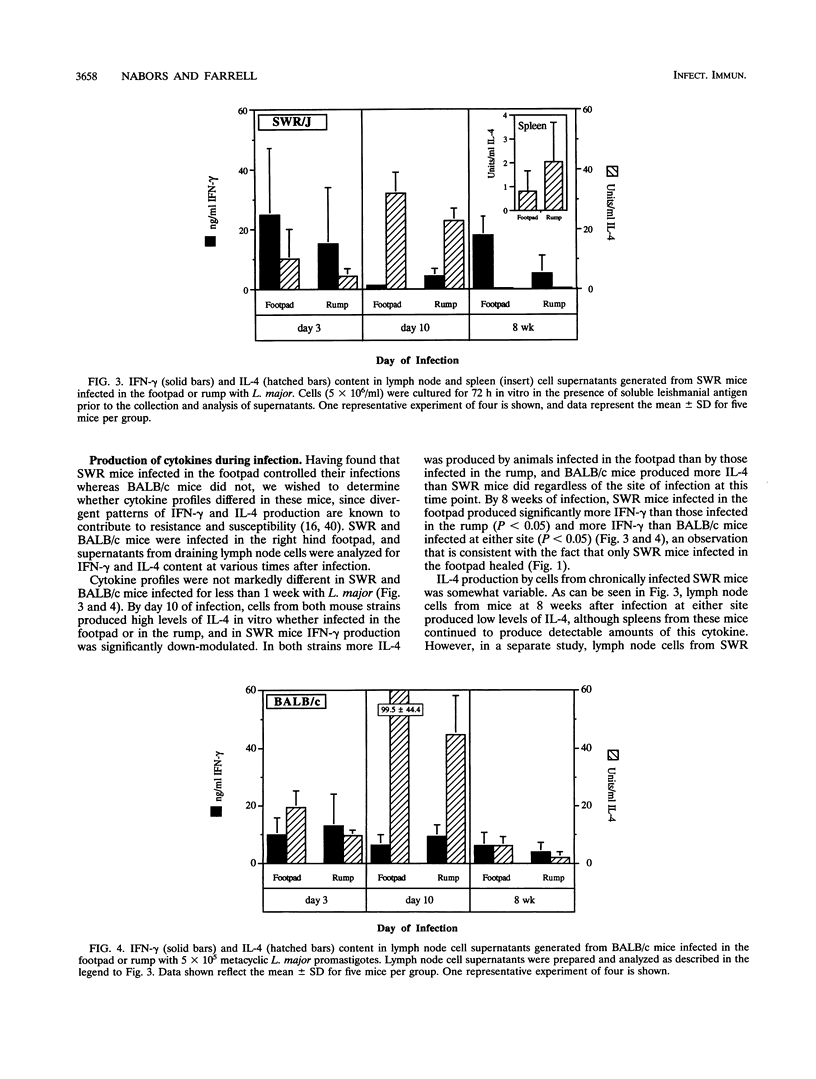
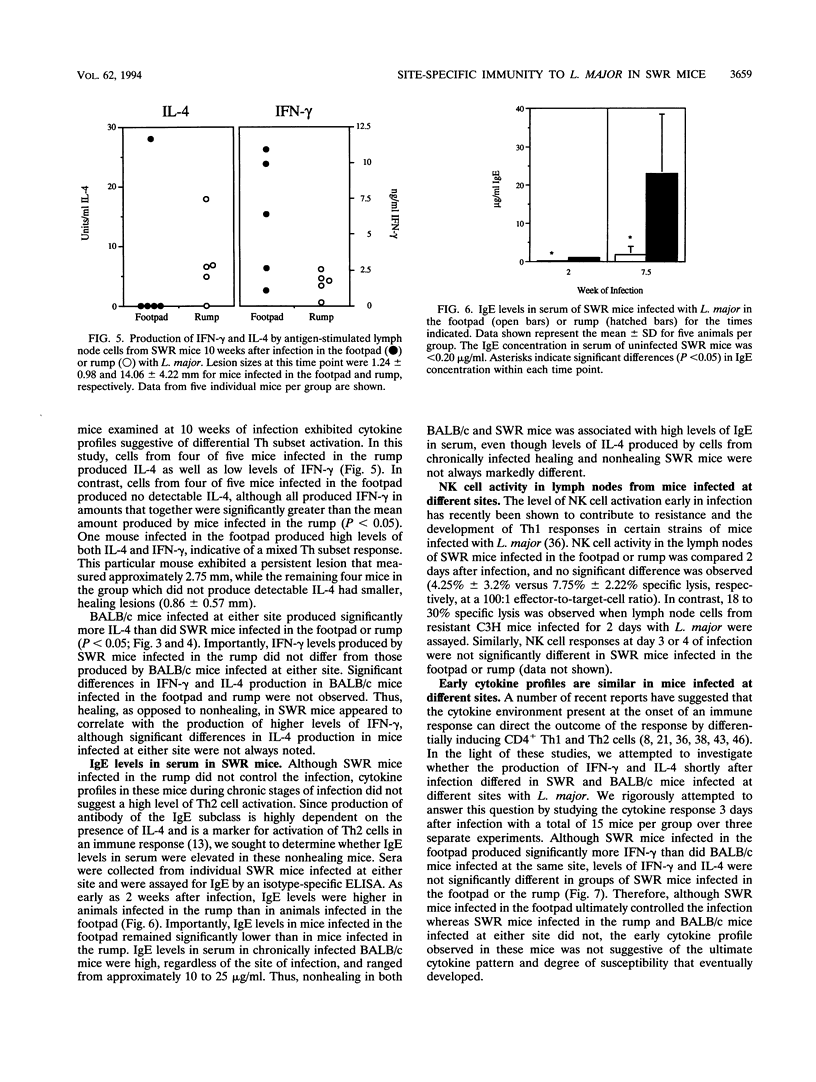
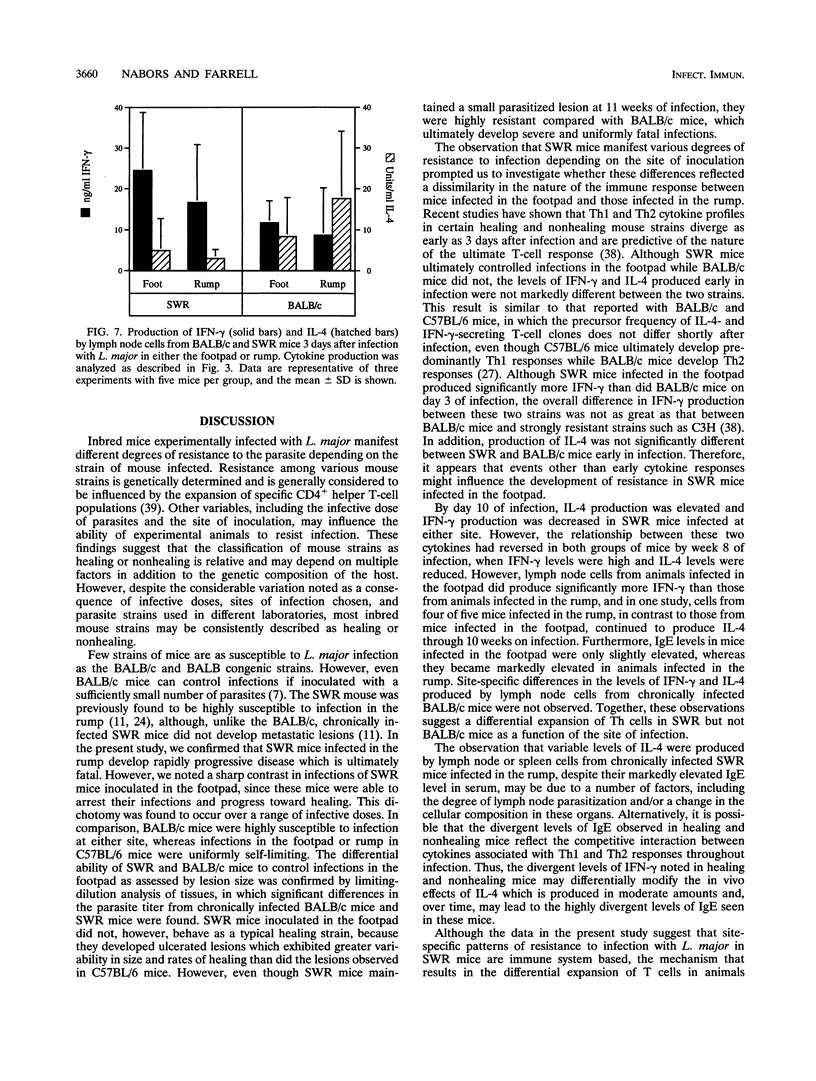
Inbred strains of mice usually develop either of two divergent patterns of infection in response to Leishmania major. Resistant mice, which develop self-limiting infections, respond immunologically with the activation of gamma interferon-secreting Th1 helper T cells, while nonhealing infections in susceptible mice are characterized by the proliferation of interleukin-4-secreting Th2 cells. Development of these divergent responses is dependent primarily on the strain of mouse infected, although factors such as the infective dose, species, and strain of parasite can also influence the degree of resistance. In this study, we show that a single mouse strain, SWR, can develop totally divergent patterns of infection depending on the site of parasite inoculation. Both SWR mice and highly susceptible BALB/c mice developed progressive, ultimately fatal disease when inoculated in the dorsal skin over the base of the tail. However, SWR mice infected in the hind footpad developed far less severe infections, which were for the most part controlled, whereas BALB/c mice infected in this site developed severe, nonhealing lesions. Production of gamma interferon and interleukin-4 and measurement of immunoglobulin E levels in serum were used to assess the degree of Th1 and Th2 cell activation in infected mice. Cytokine profiles early in infection had characteristics of a mixed Th1-Th2 response and were similar in SWR mice infected at either site. These early cytokine responses were not predictive of the ultimate disease outcome, since lymph node cells from healing mice eventually produced higher levels of gamma interferon than did those from nonhealing mice, and healing mice had lower levels of immunoglobulin E in serum, suggesting a functional bias toward Th1 cell activity in these animals. The differential ability of SWR mice to heal infections at different cutaneous sites provides a new model for the study of resistance to cutaneous leishmaniasis. Unlike traditional models of infection in which resistant and susceptible strains of mice are compared, this model allows for the study of factors that contribute to healing and nonhealing infections in a genetically identical strain of mouse.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barral-Netto M., Barral A., Brownell C. E., Skeiky Y. A., Ellingsworth L. R., Twardzik D. R., Reed S. G. Transforming growth factor-beta in leishmanial infection: a parasite escape mechanism. Science. 1992 Jul 24;257(5069):545–548. doi: 10.1126/science.1636092. [DOI] [PubMed] [Google Scholar]
- Behforouz N. C., Wenger C. D., Mathison B. A. Prophylactic treatment of BALB/c mice with cyclosporine A and its analog B-5-49 enhances resistance to Leishmania major. J Immunol. 1986 Apr 15;136(8):3067–3075. [PubMed] [Google Scholar]
- Belosevic M., Finbloom D. S., Van Der Meide P. H., Slayter M. V., Nacy C. A. Administration of monoclonal anti-IFN-gamma antibodies in vivo abrogates natural resistance of C3H/HeN mice to infection with Leishmania major. J Immunol. 1989 Jul 1;143(1):266–274. [PubMed] [Google Scholar]
- Berman J. D., Neva F. A. Effect of temperature on multiplication of Leishmania amastigotes within human monocyte-derived macrophages in vitro. Am J Trop Med Hyg. 1981 Mar;30(2):318–321. doi: 10.4269/ajtmh.1981.30.318. [DOI] [PubMed] [Google Scholar]
- Biegel D., Topper G., Rabinovitch M. Leishmania mexicana: temperature sensitivity of isolated amastigotes and of amastigotes infecting macrophages in culture. Exp Parasitol. 1983 Dec;56(3):289–297. doi: 10.1016/0014-4894(83)90074-7. [DOI] [PubMed] [Google Scholar]
- Bretscher P. A., Wei G., Menon J. N., Bielefeldt-Ohmann H. Establishment of stable, cell-mediated immunity that makes "susceptible" mice resistant to Leishmania major. Science. 1992 Jul 24;257(5069):539–542. doi: 10.1126/science.1636090. [DOI] [PubMed] [Google Scholar]
- Chatelain R., Varkila K., Coffman R. L. IL-4 induces a Th2 response in Leishmania major-infected mice. J Immunol. 1992 Feb 15;148(4):1182–1187. [PubMed] [Google Scholar]
- Childs G. E., Lightner L. K., McKinney L., Groves M. G., Price E. E., Hendricks L. D. Inbred mice as model hosts for cutaneous leishmaniasis. I. Resistance and susceptibility to infection with Leishmania braziliensis, L. mexicana, and L. aethiopica. Ann Trop Med Parasitol. 1984 Feb;78(1):25–34. doi: 10.1080/00034983.1984.11811769. [DOI] [PubMed] [Google Scholar]
- Curry R. C., Kiener P. A., Spitalny G. L. A sensitive immunochemical assay for biologically active MuIFN-gamma. J Immunol Methods. 1987 Nov 23;104(1-2):137–142. doi: 10.1016/0022-1759(87)90497-2. [DOI] [PubMed] [Google Scholar]
- DeTolla L. J., Scott P. A., Farrell J. P. Single gene control of resistance to cutaneous leishmaniasis in mice. Immunogenetics. 1981;14(1-2):29–39. doi: 10.1007/BF00344297. [DOI] [PubMed] [Google Scholar]
- Ding A., Nathan C. F., Graycar J., Derynck R., Stuehr D. J., Srimal S. Macrophage deactivating factor and transforming growth factors-beta 1 -beta 2 and -beta 3 inhibit induction of macrophage nitrogen oxide synthesis by IFN-gamma. J Immunol. 1990 Aug 1;145(3):940–944. [PubMed] [Google Scholar]
- Finkelman F. D., Holmes J., Katona I. M., Urban J. F., Jr, Beckmann M. P., Park L. S., Schooley K. A., Coffman R. L., Mosmann T. R., Paul W. E. Lymphokine control of in vivo immunoglobulin isotype selection. Annu Rev Immunol. 1990;8:303–333. doi: 10.1146/annurev.iy.08.040190.001511. [DOI] [PubMed] [Google Scholar]
- Gajewski T. F., Joyce J., Fitch F. W. Antiproliferative effect of IFN-gamma in immune regulation. III. Differential selection of TH1 and TH2 murine helper T lymphocyte clones using recombinant IL-2 and recombinant IFN-gamma. J Immunol. 1989 Jul 1;143(1):15–22. [PubMed] [Google Scholar]
- Heinzel F. P., Rerko R. M., Hatam F., Locksley R. M. IL-2 is necessary for the progression of leishmaniasis in susceptible murine hosts. J Immunol. 1993 May 1;150(9):3924–3931. [PubMed] [Google Scholar]
- Heinzel F. P., Sadick M. D., Holaday B. J., Coffman R. L., Locksley R. M. Reciprocal expression of interferon gamma or interleukin 4 during the resolution or progression of murine leishmaniasis. Evidence for expansion of distinct helper T cell subsets. J Exp Med. 1989 Jan 1;169(1):59–72. doi: 10.1084/jem.169.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinzel F. P., Sadick M. D., Mutha S. S., Locksley R. M. Production of interferon gamma, interleukin 2, interleukin 4, and interleukin 10 by CD4+ lymphocytes in vivo during healing and progressive murine leishmaniasis. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7011–7015. doi: 10.1073/pnas.88.16.7011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard J. G., Hale C., Liew F. Y. Immunological regulation of experimental cutaneous leishmaniasis. IV. Prophylactic effect of sublethal irradiation as a result of abrogation of suppressor T cell generation in mice genetically susceptible to Leishmania tropica. J Exp Med. 1981 Mar 1;153(3):557–568. doi: 10.1084/jem.153.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard J. G. Immunological regulation and control of experimental leishmaniasis. Int Rev Exp Pathol. 1986;28:79–116. [PubMed] [Google Scholar]
- Hsieh C. S., Heimberger A. B., Gold J. S., O'Garra A., Murphy K. M. Differential regulation of T helper phenotype development by interleukins 4 and 10 in an alpha beta T-cell-receptor transgenic system. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):6065–6069. doi: 10.1073/pnas.89.13.6065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirkpatrick C. E., Nolan T. J., Farrell J. P. Rate of Leishmania-induced skin-lesion development in rodents depends on the site of inoculation. Parasitology. 1987 Jun;94(Pt 3):451–465. doi: 10.1017/s0031182000055803. [DOI] [PubMed] [Google Scholar]
- Leal L. M., Moss D. W., Kuhn R., Müller W., Liew F. Y. Interleukin-4 transgenic mice of resistant background are susceptible to Leishmania major infection. Eur J Immunol. 1993 Feb;23(2):566–569. doi: 10.1002/eji.1830230241. [DOI] [PubMed] [Google Scholar]
- Liew F. Y., Hale C., Howard J. G. Prophylactic immunization against experimental leishmaniasis. IV. Subcutaneous immunization prevents the induction of protective immunity against fatal Leishmania major infection. J Immunol. 1985 Sep;135(3):2095–2101. [PubMed] [Google Scholar]
- Milon G., Titus R. G., Cerottini J. C., Marchal G., Louis J. A. Higher frequency of Leishmania major-specific L3T4+ T cells in susceptible BALB/c as compared with resistant CBA mice. J Immunol. 1986 Feb 15;136(4):1467–1471. [PubMed] [Google Scholar]
- Moriearty P. L., Grimaldi Júnior G. Host factors influencing outcome of Leishmania mexicana mexicana infection in mice. Mem Inst Oswaldo Cruz. 1983 Jan-Mar;78(1):49–59. doi: 10.1590/s0074-02761983000100006. [DOI] [PubMed] [Google Scholar]
- Morris L., Troutt A. B., Handman E., Kelso A. Changes in the precursor frequencies of IL-4 and IFN-gamma secreting CD4+ cells correlate with resolution of lesions in murine cutaneous leishmaniasis. J Immunol. 1992 Oct 15;149(8):2715–2721. [PubMed] [Google Scholar]
- Nacy C. A., Fortier A. H., Meltzer M. S., Buchmeier N. A., Schreiber R. D. Macrophage activation to kill Leishmania major: activation of macrophages for intracellular destruction of amastigotes can be induced by both recombinant interferon-gamma and non-interferon lymphokines. J Immunol. 1985 Nov;135(5):3505–3511. [PubMed] [Google Scholar]
- Nacy C. A., Fortier A. H., Pappas M. G., Henry R. R. Susceptibility of inbred mice to Leishmania tropica infection: correlation of susceptibility with in vitro defective macrophage microbicidal activities. Cell Immunol. 1983 Apr 15;77(2):298–307. doi: 10.1016/0008-8749(83)90030-8. [DOI] [PubMed] [Google Scholar]
- Passwell J. H., Shor R., Shoham J. The enhancing effect of interferon-beta and -gamma on the killing of Leishmania tropica major in human mononuclear phagocytes in vitro. J Immunol. 1986 Apr 15;136(8):3062–3066. [PubMed] [Google Scholar]
- Poulter L. W., Pandolph C. R. Mechanisms of immunity to leishmaniasis. IV. Significance of lymphatic drainage from the site of infection. Clin Exp Immunol. 1982 May;48(2):396–402. [PMC free article] [PubMed] [Google Scholar]
- Preston P. M., Dumonde D. C. Experimental cutaneous leishmaniasis. V. Protective immunity in subclinical and self-healing infection in the mouse. Clin Exp Immunol. 1976 Jan;23(1):126–138. [PMC free article] [PubMed] [Google Scholar]
- Sacks D. L., Barral A., Neva F. A. Thermosensitivity patterns of Old vs. New World cutaneous strains of Leishmania growing within mouse peritoneal macrophages in vitro. Am J Trop Med Hyg. 1983 Mar;32(2):300–304. doi: 10.4269/ajtmh.1983.32.300. [DOI] [PubMed] [Google Scholar]
- Sacks D. L., Hieny S., Sher A. Identification of cell surface carbohydrate and antigenic changes between noninfective and infective developmental stages of Leishmania major promastigotes. J Immunol. 1985 Jul;135(1):564–569. [PubMed] [Google Scholar]
- Sadick M. D., Heinzel F. P., Holaday B. J., Pu R. T., Dawkins R. S., Locksley R. M. Cure of murine leishmaniasis with anti-interleukin 4 monoclonal antibody. Evidence for a T cell-dependent, interferon gamma-independent mechanism. J Exp Med. 1990 Jan 1;171(1):115–127. doi: 10.1084/jem.171.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scharton T. M., Scott P. Natural killer cells are a source of interferon gamma that drives differentiation of CD4+ T cell subsets and induces early resistance to Leishmania major in mice. J Exp Med. 1993 Aug 1;178(2):567–577. doi: 10.1084/jem.178.2.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott P. A., Farrell J. P. Experimental cutaneous leishmaniasis: disseminated leishmaniasis in genetically susceptible and resistant mice. Am J Trop Med Hyg. 1982 Mar;31(2):230–238. doi: 10.4269/ajtmh.1982.31.230. [DOI] [PubMed] [Google Scholar]
- Scott P. IFN-gamma modulates the early development of Th1 and Th2 responses in a murine model of cutaneous leishmaniasis. J Immunol. 1991 Nov 1;147(9):3149–3155. [PubMed] [Google Scholar]
- Scott P. Impaired macrophage leishmanicidal activity at cutaneous temperature. Parasite Immunol. 1985 May;7(3):277–288. doi: 10.1111/j.1365-3024.1985.tb00076.x. [DOI] [PubMed] [Google Scholar]
- Scott P., Kaufmann S. H. The role of T-cell subsets and cytokines in the regulation of infection. Immunol Today. 1991 Oct;12(10):346–348. doi: 10.1016/0167-5699(91)90063-Y. [DOI] [PubMed] [Google Scholar]
- Scott P., Natovitz P., Coffman R. L., Pearce E., Sher A. Immunoregulation of cutaneous leishmaniasis. T cell lines that transfer protective immunity or exacerbation belong to different T helper subsets and respond to distinct parasite antigens. J Exp Med. 1988 Nov 1;168(5):1675–1684. doi: 10.1084/jem.168.5.1675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott P., Pearce E., Natovitz P., Sher A. Vaccination against cutaneous leishmaniasis in a murine model. I. Induction of protective immunity with a soluble extract of promastigotes. J Immunol. 1987 Jul 1;139(1):221–227. [PubMed] [Google Scholar]
- Seder R. A., Paul W. E., Davis M. M., Fazekas de St Groth B. The presence of interleukin 4 during in vitro priming determines the lymphokine-producing potential of CD4+ T cells from T cell receptor transgenic mice. J Exp Med. 1992 Oct 1;176(4):1091–1098. doi: 10.1084/jem.176.4.1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher A., Gazzinelli R. T., Oswald I. P., Clerici M., Kullberg M., Pearce E. J., Berzofsky J. A., Mosmann T. R., James S. L., Morse H. C., 3rd Role of T-cell derived cytokines in the downregulation of immune responses in parasitic and retroviral infection. Immunol Rev. 1992 Jun;127:183–204. doi: 10.1111/j.1600-065x.1992.tb01414.x. [DOI] [PubMed] [Google Scholar]
- Solbach W., Forberg K., Kammerer E., Bogdan C., Röllinghoff M. Suppressive effect of cyclosporin A on the development of Leishmania tropica-induced lesions in genetically susceptible BALB/c mice. J Immunol. 1986 Jul 15;137(2):702–707. [PubMed] [Google Scholar]
- Swain S. L., Weinberg A. D., English M., Huston G. IL-4 directs the development of Th2-like helper effectors. J Immunol. 1990 Dec 1;145(11):3796–3806. [PubMed] [Google Scholar]
- Titus R. G., Ceredig R., Cerottini J. C., Louis J. A. Therapeutic effect of anti-L3T4 monoclonal antibody GK1.5 on cutaneous leishmaniasis in genetically-susceptible BALB/c mice. J Immunol. 1985 Sep;135(3):2108–2114. [PubMed] [Google Scholar]
- Tsunawaki S., Sporn M., Ding A., Nathan C. Deactivation of macrophages by transforming growth factor-beta. Nature. 1988 Jul 21;334(6179):260–262. doi: 10.1038/334260a0. [DOI] [PubMed] [Google Scholar]
- Wilson H. R., Dieckmann B. S., Childs G. E. Leishmania braziliensis and Leishmania mexicana: experimental cutaneous infections in golden hamsters. Exp Parasitol. 1979 Apr;47(2):270–283. doi: 10.1016/0014-4894(79)90079-1. [DOI] [PubMed] [Google Scholar]



