Abstract
DNA methyltransferase inhibitor, 5-azacitidine (AC) is effective in myelodysplastic syndromes (MDS) and can induce re-expression in cancer. We analyzed the methylation of 25 tumor suppressor genes in AC-treated MDS. Hypermethylation of CDKN2B, FHIT, ESR1, and IGSF4 gene was detected in 9/44 patients. In concordance with the clinical response, a lack of or decreased methylation in 4 patients with hematologic improvements and persistent methylation in 4 others with no response was observed. The mRNA expression of CDKN2B, IGSF4, and ESR1 was significantly reduced in MDS. Our results suggest that methylation changes contribute to disease pathogenesis and may serve as marker to monitor the efficacy of treatments.
Keywords: Demethylation, DNA Methylation, Myelodysplastic Syndromes, DNA Methyltransferase Inhibitors, Azacitidine, MS-MLPA
INTRODUCTION
Myelodysplastic syndrome (MDS) is a clonal stem cell disorder characterized by ineffective hematopoiesis in one or more of the lineages in the bone marrow and an increased tendency towards acute leukemia transformation (1). MDS patients were treated with supportive care measures only; high-dose chemotherapy programs modeled after acute myelogenous leukemia (AML) therapy or allogenic stem cell transplantation (1, 2). Recently, 5-azacitidine (AC) and 5-aza-2-deoxycitidine (DAC) have been used as hypomethylating agents for the treatment of MDS.
The aberrant methylation of CpG islands or CpG sites, in or near the promoter region of many genes was found to be associated with the transcriptional inactivation of important tumor suppressor genes (TSG) (3-5). These TSG are also often silenced in hematopoietic malignancies by DNA hypermethylation (6). Many of these genes plays roles in multiple fundamental pathways that include cell cycle control (p16INK4a, p15INK4a, Rb1, p27Kip1, p14ARF, and p73), DNA repair (BRCA1 and MGMT), apoptosis inhibition (DAPK, TMS1, and caspase-8), tumor invasion/metastasis (E-cadherin, APC, TIMP3, and VHL), and growth factor responses (ER and EphA3) (7). The DNA methyltransferase (DNMT) inhibitors AC and DAC can induce the functional re-expression of genes silenced by cancer that cause growth arrest and apoptosis in tumor cells (8). The p15 (CDKN2B) of hypermethylation genes may play a causal role in hematologic malignancies such as MDS and AML (3). The methylation of p15 occurs in patients with these disorders and is associated with an increased frequency (or density) and advanced disease (9, 10). In response to the treatment with AC, one study showed remarkable differential demethylation in several myeloid leukemia cell lines and a significant but transient demethylation in MDS patients. Genomic DNA methylation patterns have revealed widespread demethylation in array-based analysis (11).
Recently, AC and DAC are clinically active in MDS (12-14) and other hematologic malignancies. Therapeutic strategies have been developed to effectively inhibit methylation in these and other malignancies such as CDKN2B (15) that can be reactivated after treatment with methylation inhibitors. The aberrant methylation of CpG islands represents an ideal candidate for diagnostic and prognostic cancer markers (16).
We analyzed expression and methylation changes of 25 TSG identified as potential epigenetic markers after Vidaza® (azacitidine; AC) treatment in MDS that further the correlation to clinical response. Based on a complete cell blood count (CBC) and bone marrow findings, our data indicates that methylation changes after AC treatment were correlated with the clinical responses of patients. Our study also confirmed that methylation was associated with a reduced mRNA expression in patients with MDS. Thus, DNA methylation changes can be used as a biomarker in predicting the response to a particular chemotherapeutic agent.
MATERIALS AND METHODS
Patient samples
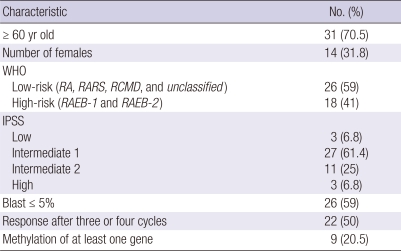
A total of 44 MDS patients (30 men and 14 women; age range, 15-82 yr), including 13 with refractory anemia (RA), one case of RA with ringed sideroblasts (RARS), 9 cases of refractory cytopenia with multilineage dysplasia (RCMD), one case of refractory cytopenia with multilineage dysplasia and ringed sideroblasts (RCMD-RS), 9 cases of RA with excess blasts-1 (RAEB-1), 9 cases of RA with excess blasts-2 (RAEB-2) and 2 cases of MDS-unclassified (MDS-U) (WHO classification), were examined before (as part of the initial diagnostic process) and during treatment with AC. The AC dose for all patients was 75 mg/m2/d for 7 days and repeated on a 28-day cycle. The response was assessed prior to each cycle of treatment. Therapy was continued for three cycles after complete remission (CR) or until progressive disease or toxicity in patients with partial remission (PR) or hematologic improvement (HI). Blood was obtained before or at the beginning of the first cycle (as before treatment) and at the beginning of the 4th, 5th, and 6th cycles (in turn as after 3, 4, and 5 cycle's treatment). The characteristics of the patients are summarized in Table 1. Samples were obtained from patients who were admitted between 2004 and 2008 at Chonnam National University Hwasun Hospital (Jeonnam, Korea). Peripheral blood (PB)-MNCs from 3 healthy donors served as non-malignant controls.
Table 1.
Characteristics of the study group (44 patients)
WHO, World Health Organization; RA, refractory anemia; RARS, RA with ringed sideroblasts; RCMD, refractory cytopenia with multilineage dysplasia; RAEB, RA with excess blasts; IPSS, International Prognostic Scoring System.
DNA methylation and fragment analyses
Genomic DNA was extracted from PB using a QIAamp DNA Blood Mini kit (Qiagen, Valencia, CA, USA) according to the manufacturer's protocol. An ME001 probe mix kit was used for methylation-specific multiplex ligation-dependent probe amplification (MS-MLPA), according to the manufacturer's instructions (MRC-Holland, Amsterdam, Netherlands). The kit contains 25 sequences corresponding to TSG that are frequently silenced by methylation in different tumors but are unmethylated in the blood-derived DNA of healthy individuals. MS-MLPA starts with sample DNA denaturation and the hybridization of the MLPA probes their specific DNA targets. Methylated hybrids of the sample DNA are prevented from being digested by HhaI, and are then amplified by PCR to generate a signal on an ABI 3100 sequencer (Applied Biosystems, Foster City, CA, USA). The data shown are the mean of triplicate reactions.
Data normalization and analysis were performed using the built-in MLPA application in GeneMarker ver. 1.5 (Soft-Genetics). To determine the methylation status, the normalized height-ratio data for each ligated sample were compared with the height-ratio data for the same sample after digestion with HhaI. Aberrant methylation was scored when the calculated methylation was > 10% (hypermethylation). Percentages below this level were regarded as background.
RNA isolation and quantitative real-time RT-PCR
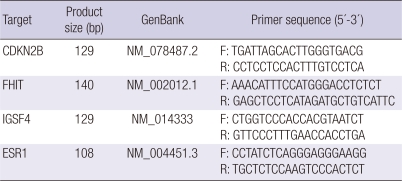
RNA was extracted using the Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. First-strand cDNA was synthesized from RNA using random primers and a Moloney murine leukemia virus (MMLV) reverse transcriptase, according to the manufacturer's protocol. For the quantification of CDKN2B, FHIT, IGSF4, and ESR1 transcripts, real-time PCR was performed using a 72-well Rotor-Gene RG-3000 (Corbett Research, Sydney, Australia) with a 10 µL final reaction mixture containing 250 nM in each primer, 1 × SYBR premix Ex Taq (Takara, Tokyo, Japan), and 2 µL of cDNA. The primer sequences and annealing temperatures used to amplify each of the genes tested are listed in Table 2. The reaction mixture was preheated to 95℃ for 10 sec, followed by 45 cycles of 95℃ for 10 sec, 60℃ for 20 sec, and 72℃ for 20 sec. As a reference transcript for each expression level, the housekeeping gene β2-microglobin was amplified. All reactions included negative controls where the reverse transcriptase was omitted.
Table 2.
Primers used for quantitative real-time PCR
Statistical analysis
For the mRNA data, 95% confidence intervals (CIs) were calculated to assess the significance of the 2-ΔΔCT (17). Differences in continuous variables were analyzed by the Mann-Whitney U-test for distributions between two groups. All statistical analyses were performed using SPSS software (ver.12.0). For all analyses, the P values were two-tailed; a P values < 0.05 was considered statistically significant.
Ethics statement
This study was approved by the institutional review board of the Chonnam National University Hwasun Hospital in Hwasun, Korea (IRB number: CNUHHIRB 2009-22). At the time of PB collection, all case and control subjects provided informed consent to participate in this study.
RESULTS
Methylation status in MDS patients and methylation reversal after 3-5 courses of AC treatment corresponded to the clinical response
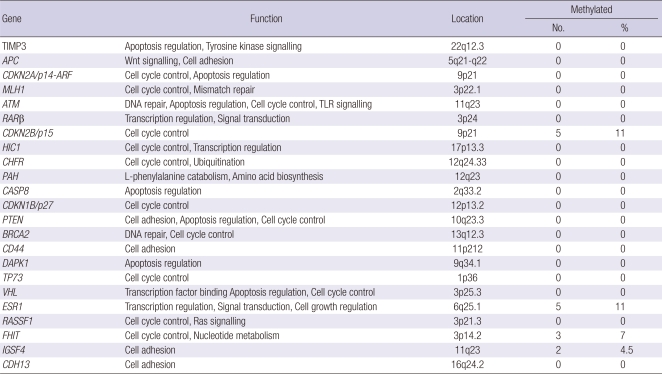
Alterations in DNA methylation are common in a variety of tumors, as well as during development (18). All MDS patients at the time of diagnosis were screened for gene methylation; only those samples containing at least one methylated gene, were analyzed further by MS-MLPA following 3-5 cycles of AC. The PB in this study is similar to that taken previously (10, 11, 20, 24). Hypermethylation of at least one gene was detected in 9/44 cases (20.5%) that included CDKN2B, FHIT, IGSF4, and ESR1 in MDS (Table 3), but not in any of the control samples. Our results indicate increased methylation among high-risk patients (5/18, 27.8%) compared with low-risk patients (4/26, 15.4%) (P < 0.05). Either CDKN2B or ESR1 was methylated in 5 patients (5/44, 11.4%), whereas FHIT and IGSF4 was methylated in 3 patients (3/44, 6.8%) and 2 patients (2/44, 4.5%) respectively.
Table 3.
Methylation frequencies of tumor suppressor genes in 44 AC-treated MDS patients
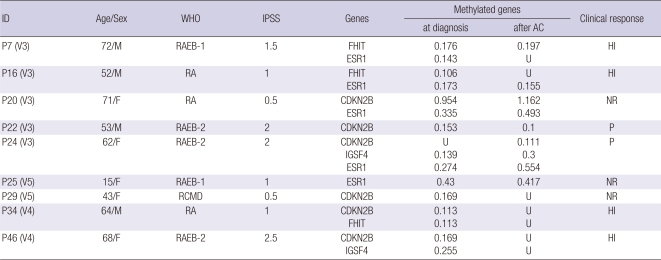
We examined the methylation status of 25 TSG after 3-5 courses of AC treatment in 9 MDS patients with methylation at the time of diagnosis, and assessed the subsequent change in methylation in association with the correlative clinical response. At the time that blood was obtained after treatment, the clinical response was evaluated respectively. Increased or reduced methylation level of at least 10% is considered reinforcement or reduction respectively. Our data revealed an agreement between demethylation and the clinical response in 8 of 9 cases (Table 4). Patients (P20, P25, P22, and P24) had the persistent methylation that correlated to either no clinical response or tends to leukemia. Specially, P24 (RAEB-2 subtype) progressed to leukemia, showed increased methylation level at ESR1 and IGSF4 as well as de novo methylation of CDKN2B. The pretreated methylated genes were reduced or loss for 3 patients (P16, P34, and P46) who exhibited an increase in hematologic improvement (HI). In patients P34 and P46, no methylation was observed after treatment. Patient P16 exhibited ESR1 and FHIT methylation and then remained only ESR1. In the case of patient P7, the results indicated a loss of ESR1 and remained FHIT methylation with no accompanying clinical response after 3 courses (the patient refused the fourth course of AC); however, hematologic improvement was observed in this patient after follow-up. It is possible that the therapeutic effect may be delayed with a response occurring several weeks after treatment (19). Although patient P29 (RCMD subtype) had no clinical response, the methylation at CDKN2B disappeared after 5 courses of AC.
Table 4.
Methylation reversal in AC-treated MDS patients and their clinical responses
V (No.), number of cycles of AC treatment; U, Unmethylated; HI, Hematologic improvement; NR, no response; P, progression to AML.
Methylation status of CDKN2B and FHIT using pyrosequencing in MDS patients with methylation at the time of diagnosis was confirmed in the association shown between DNA methylation reversal and a clinical response (data not shown). The methylation status changed following 3-5 courses of AC, a demethylation agent, (correlated with the clinical response observed), based on recommended criteria.
Reduced gene expression in the MDS patients versus controls
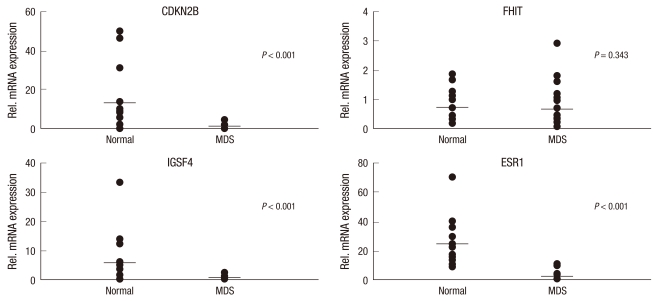
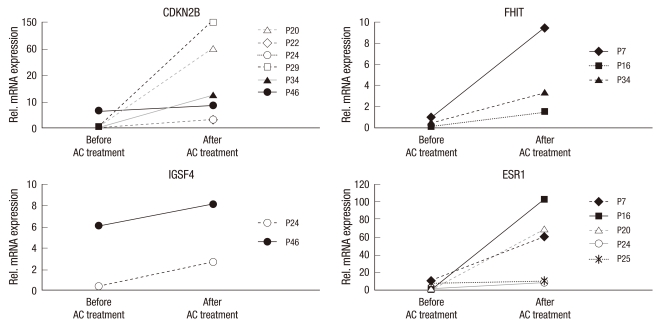
The transcriptional silencing of TSG associated with DNA methylation is a common epigenetic event in hematologic malignancies. To verify the contribution of DNA methylation to the inactivation of transcription, we compared mRNA expression levels in the MDS and control group. The mean level of mRNA expression for CDKN2B, FHIT, IGSF4, and ESR1 was higher in the control group (16 PB samples) than in the MDS group (30 PB samples) (Fig. 1). The difference for CDKN2B, IGSF4, and ESR1 mRNA expression was statistically significant (P < 0.05) whereas the difference for FHIT was not (P = 0.343). Additionally, most of the individuals for whom CDKN2B, FHIT, IGSF4, and ESR1 were initial methylated showed reduced mRNA compared with the median value. Despite the lack of a clinical response in the 9 samples (in terms of their mRNA levels after 3 5 courses of AC), we also found evidence for the reactivation of gene expression in response to the demethylation agent (Fig. 2).
Fig. 1.
Quantitative analysis of mRNA expression of CDKN2B, FHIT, IGSF4, and ESR1 in 30 MDS patients and 16 normal controls were analyzed at diagnosis. MDS group was significantly lower level in CDKN2B, IGSF4, and ESR1 (P < 0.05). The horizontal bars indicate the median value for each sample group.
Fig. 2.
Demethylation due to AC treatment in the methylated samples was associated with mRNA re-expression in the MDS group after 3-5 treatment cycles. Data shown are representative of three independent experiments. P means patient number.
DISCUSSION
Many genes important for most of the carcinogenic pathways are hypermethylated in MDS (7). DNMT inhibitors, that inhibit DNA methylation in various studies (15, 20, 21), represent a major advance in treatment (13, 14); however, it is still unclear whether methylation reversal corresponds to a clinical response. The present study examined genome-specific methylation changes in MDS patients who received conventional AC treatment using MS-MLPA. Our results indicate a higher degree of methylation among high-risk patients (5/18, 27.8%) versus low-risk patients (4/26, 15.4%; P < 0.05), similar to previous reports (9, 22). Interestingly, a correlation was detected between clinical responses based on conventional hematologic improvement and quantitative gene methylation dynamics that followed 3-5 courses of AC treatment.
Many studies have shown that the demethylation and reactivation of some genes correlate with drug responses to DAC and AC in primary cells such as MDS patients (15, 21), and AML (20). In addition, the large-scale demethylating activity of AC was shown in a subset of MDS patients (11). These results are similar to the result that responding patients with a partial recovery (or complete recovery) showed a reduction in or the absence of methylated genes and that patients with an AML transformation (or no response) showed the maintenance to reinforcement of methylation status after treatment. However, Raj et al. (23) showed an AC reduced CDKN2B methylation uncorrelated with the response. Currently, no association between the clinical response and baseline methylation or methylation reversal in the bone marrow or purified CD34+ cells was reported (24).
The authors focused on molecular changes during cycle 1 to ensure the study of proximal changes in the diseased cells. However, our present findings indicate that important epigenetic alterations may occur in later cycles. The poor responses of the patients with strong methylation (such as P20, P24, and P25) suggest the need for tailored therapy (alternative doses and combination strategies) to optimize their responses. It was previously shown that methylation reversal during cycle 1 predicted the clinical response in MDS/AML patients treated with AC and phenylbutyrate (25). A limited response in cytopenia was detected in patient P29; however, a lack of CDKN2B hypermethylation and reactivation of mRNA expression was detected earlier during AC treatment. This indicated that the demethylating agent became activated and that other unknown mechanisms may have prevented hematologic improvements (i.e., a demethylation-independent response). In their review, Silverman et al. (26) presented a mechanism for a hematologic response, with an AC blocking leukemia inhibitory factor, oncostatin M, interleukin (IL)-6, and IL-11; alternatively; AC may act as a biological response modifier to restore normal hematopoiesis. It is possible that the methylation/demethylation of genes other than CDKN2B is critical to this response (7). Taken together, these data indicate that methylation reversal can be used as a molecular marker following AC treatment. Consistent with this, gene methylation increased during disease progression with little to no reversal occurring after AC treatments in conjunction with the lack of a clinical response or leukemic transformation may be indicative of a poor prognosis.
The most widespread epigenetic abnormality in tumorigenesis is the silencing of a gene transcription associated with a rise in DNA methylation in normally unmethylated gene promoter regions. In this study, we found that the mRNA expression of several methylated genes, including CDKN2B, IGSF4, and ESR1, in the MDS group was reduced at the time of the initial diagnosis of the patient. In contrast, the mRNA expression of FHIT in MDS was not significantly altered when compared with the controls. The results indicate that CDKN2B, IGSF4, and ESR1 expression is involved in the pathogenesis of MDS at an early stage while FHIT expression appears to be important at a later phase in the development of MDS (27). Additionally, because the expression level decreased in association with methylation at the time of diagnosis (while the expression level increased in response to treatment with AC) the reduction in transcription caused by methylation might be involved in the pathogenesis and disease progression of MDS.
When compared with the incidences reported by others (29), e.g. for p15 (15), and for FHIT (27), low methylation incidences in this study were observed. This may be due to the underestimation for the methylation of TSG such as a limited number of CpGs in MS-MLPA (28), detection in non-purification of malignant cells and low frequency (41%) patients with a blast more than 5%. We also omitted clinical responders without baseline methylation that seemed to mask potential associations between the reversal of methylation and a clinical response. Thus, other means of controlling the response of patients (such as histone code or apoptosis) should be considered.
In conclusion, changes in methylation caused by AC treatment may contribute to the clinical response of patients, as shown by the improvement of specific hematologic factors in our study. Demethylation agent-induced treatment is associated with hypomethylation and the reestablishment of a gene-specific expression. The sustained response observed in patients with AC requires validation in a larger cohort. Additional studies that include the identification of relevant TSG for drug-induced demethylation are needed to confirm the results.
Footnotes
The study was supported by a grant from the Korea Health 21 R&D Project, Ministry of Health and Welfare, Republic of Korea (A01-0385-A70604-07M7-00000A).
AUTHOR SUMMARY
DNA Methylation Changes Following 5-azacitidine Treatment in Patients with Myelodysplastic Syndrome
Huong Thi Thanh Tran, Hee Nam Kim, Il-Kwon Lee, Yeo-Kyeoung Kim, Jae-Sook Ahn, Deok-Hwan Yang, Je-Jung Lee, and Hyeoung-Joon Kim
DNA methyltransferase inhibitor, 5-azacitidine (AC), which is effective in myelodysplastic syndromes (MDS), can induce re-expression in cancer. We analyzed methylation of 25 tumor suppressor genes in AC-treated MDS. Hypermethylation of CDKN2B, FHIT, ESR1, and IGSF4 gene was detected in 9/44 patients. In concordance with clinical response, a lack or decreased methylation in 4 patients with hematologic improvements and persistent methylation in 4 others with no response was observed. mRNA expression of CDKN2B, IGSF4, and ESR1 was significantly reduced in MDS. Our results suggest that methylation changes contribute to disease pathogenesis and may serve as a marker to monitor efficacy of AC treatment.
References
- 1.Corey SJ, Minden MD, Barber DL, Kantarjian H, Wang JC, Schimmer AD. Myelodysplastic syndromes: the complexity of stem-cell diseases. Nat Rev Cancer. 2007;7:118–129. doi: 10.1038/nrc2047. [DOI] [PubMed] [Google Scholar]
- 2.Atallah E, Garcia-Manero G. Treatment strategies in myelodysplastic syndromes. Cancer Invest. 2008;26:208–216. doi: 10.1080/07357900701788122. [DOI] [PubMed] [Google Scholar]
- 3.Baylin SB. DNA methylation and gene silencing in cancer. Nat Clin Pract Oncol. 2005;2(Suppl 1):S4–S11. doi: 10.1038/ncponc0354. [DOI] [PubMed] [Google Scholar]
- 4.Hong SJ, Kang MI, Oh JH, Jung YC, Kim YH, Kim SJ, Choi SH, Seo EJ, Choi SW, Rhyu MG. DNA methylation and expression patterns of key tissue-specific genes in adult stem cells and stomach tissues. J Korean Med Sci. 2009;24:918–929. doi: 10.3346/jkms.2009.24.5.918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Park SH, Jung KC, Ro JY, Kang GH, Khang SK. 5' CpG island methylation of p16 is associated with absence of p16 expression in glioblastomas. J Korean Med Sci. 2000;15:555–559. doi: 10.3346/jkms.2000.15.5.555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Krug U, Ganser A, Koeffler HP. Tumor suppressor genes in normal and malignant hematopoiesis. Oncogene. 2002;21:3475–3495. doi: 10.1038/sj.onc.1205322. [DOI] [PubMed] [Google Scholar]
- 7.Galm O, Herman JG, Baylin SB. The fundamental role of epigenetics in hematopoietic malignancies. Blood Rev. 2006;20:1–13. doi: 10.1016/j.blre.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 8.Berg T, Guo Y, Abdelkarim M, Fliegauf M, Lübbert M. Reversal of p15/INK4b hypermethylation in AML1/ETO-positive and -negative myeloid leukemia cell lines. Leuk Res. 2007;31:497–506. doi: 10.1016/j.leukres.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 9.Tien HF, Tang JH, Tsay W, Liu MC, Lee FY, Wang CH, Chen YC, Shen MC. Methylation of the p15(INK4B) gene in myelodysplastic syndrome: it can be detected early at diagnosis or during disease progression and is highly associated with leukaemic transformation. Br J Haematol. 2001;112:148–154. doi: 10.1046/j.1365-2141.2001.02496.x. [DOI] [PubMed] [Google Scholar]
- 10.Uchida T, Kinoshita T, Nagai H, Nakahara Y, Saito H, Hotta T, Murate T. Hypermethylation of the p15INK4B gene in myelodysplastic syndromes. Blood. 1997;90:1403–1409. [PubMed] [Google Scholar]
- 11.Stresemann C, Bokelmann I, Mahlknecht U, Lyko F. Azacytidine causes complex DNA methylation responses in myeloid leukemia. Mol Cancer Ther. 2008;7:2998–3005. doi: 10.1158/1535-7163.MCT-08-0411. [DOI] [PubMed] [Google Scholar]
- 12.Silverman LR, McKenzie DR, Peterson BL, Holland JF, Backstrom JT, Beach CL, Larson RA Cancer and Leukemia Group B. Further analysis of trials with azacitidine in patients with myelodysplastic syndrome: studies 8421, 8921, and 9221 by the Cancer and Leukemia Group B. J Clin Oncol. 2006;24:3895–3903. doi: 10.1200/JCO.2005.05.4346. [DOI] [PubMed] [Google Scholar]
- 13.Silverman LR, Demakos EP, Peterson BL, Kornblith AB, Holland JC, Odchimar-Reissig R, Stone RM, Nelson D, Powell BL, DeCastro CM, Ellerton J, Larson RA, Schiffer CA, Holland JF. Randomized controlled trial of azacitidine in patients with the myelodysplastic syndrome: a study of the cancer and leukemia group B. J Clin Oncol. 2002;20:2429–2440. doi: 10.1200/JCO.2002.04.117. [DOI] [PubMed] [Google Scholar]
- 14.Kantarjian H, Issa JP, Rosenfeld CS, Bennett JM, Albitar M, DiPersio J, Klimek V, Slack J, de Castro C, Ravandi F, Helmer R, 3rd, Shen L, Nimer SD, Leavitt R, Raza A, Saba H. Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer. 2006;106:1794–1803. doi: 10.1002/cncr.21792. [DOI] [PubMed] [Google Scholar]
- 15.Daskalakis M, Nguyen TT, Nguyen C, Guldberg P, Köhler G, Wijermans P, Jones PA, Lübbert M. Demethylation of a hypermethylated P15/INK4B gene in patients with myelodysplastic syndrome by 5-Aza-2'-deoxycytidine (decitabine) treatment. Blood. 2002;100:2957–2964. doi: 10.1182/blood.V100.8.2957. [DOI] [PubMed] [Google Scholar]
- 16.Baylin SB, Herman JG. DNA hypermethylation in tumorigenesis: epigenetics joins genetics. Trends Genet. 2000;16:168–174. doi: 10.1016/s0168-9525(99)01971-x. [DOI] [PubMed] [Google Scholar]
- 17.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 18.Das PM, Singal R. DNA methylation and cancer. J Clin Oncol. 2004;22:4632–4642. doi: 10.1200/JCO.2004.07.151. [DOI] [PubMed] [Google Scholar]
- 19.Issa JP, Garcia-Manero G, Giles FJ, Mannari R, Thomas D, Faderl S, Bayar E, Lyons J, Rosenfeld CS, Cortes J, Kantarjian HM. Phase 1 study of low-dose prolonged exposure schedules of the hypomethylating agent 5-aza- 2'-deoxycytidine (decitabine) in hematopoietic malignancies. Blood. 2004;103:1635–1640. doi: 10.1182/blood-2003-03-0687. [DOI] [PubMed] [Google Scholar]
- 20.Yang AS, Doshi KD, Choi SW, Mason JB, Mannari RK, Gharybian V, Luna R, Rashid A, Shen L, Estecio MR, Kantarjian HM, Garcia-Manero G, Issa JP. DNA methylation changes after 5-aza-2'-deoxycytidine therapy in patients with leukemia. Cancer Res. 2006;66:5495–5503. doi: 10.1158/0008-5472.CAN-05-2385. [DOI] [PubMed] [Google Scholar]
- 21.Mund C, Hackanson B, Stresemann C, Lübbert M, Lyko F. Characterization of DNA demethylation effects induced by 5-Aza-2'-deoxycytidine in patients with myelodysplastic syndrome. Cancer Res. 2005;65:7086–7090. doi: 10.1158/0008-5472.CAN-05-0695. [DOI] [PubMed] [Google Scholar]
- 22.Uchida T, Kinoshita T, Hotta T, Murate T. High-risk myelodysplastic syndromes and hypermethylation of the p15Ink4B gene. Leuk Lymphoma. 1998;32:9–18. doi: 10.3109/10428199809059242. [DOI] [PubMed] [Google Scholar]
- 23.Raj K, John A, Ho A, Chronis C, Khan S, Samuel J, Pomplun S, Thomas NS, Mufti GJ. CDKN2B methylation status and isolated chromosome 7 abnormalities predict responses to treatment with 5-azacytidine. Leukemia. 2007;21:1937–1944. doi: 10.1038/sj.leu.2404796. [DOI] [PubMed] [Google Scholar]
- 24.Fandy TE, Herman JG, Kerns P, Jiemjit A, Sugar EA, Choi SH, Yang AS, Aucott T, Dauses T, Odchimar-Reissig R, Licht J, McConnell MJ, Nasrallah C, Kim MK, Zhang W, Sun Y, Murgo A, Espinoza-Delgado I, Oteiza K, Owoeye I, Silverman LR, Gore SD, Carraway HE. Early epigenetic changes and DNA damage do not predict clinical response in an overlapping schedule of 5-azacytidine and entinostat in patients with myeloid malignancies. Blood. 2009;114:2764–2773. doi: 10.1182/blood-2009-02-203547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gore SD, Baylin S, Sugar E, Carraway H, Miller CB, Carducci M, Grever M, Galm O, Dauses T, Karp JE, Rudek MA, Zhao M, Smith BD, Manning J, Jiemjit A, Dover G, Mays A, Zwiebel J, Murgo A, Weng LJ, Herman JG. Combined DNA methyltransferase and histone deacetylase inhibition in the treatment of myeloid neoplasms. Cancer Res. 2006;66:6361–6369. doi: 10.1158/0008-5472.CAN-06-0080. [DOI] [PubMed] [Google Scholar]
- 26.Silverman LR. DNA methyltransferase inhibitors in myelodysplastic syndrome. Best Pract Res Clin Haematol. 2004;17:585–594. doi: 10.1016/j.beha.2004.08.010. [DOI] [PubMed] [Google Scholar]
- 27.Iwai M, Kiyoi H, Ozeki K, Kinoshita T, Emi N, Ohno R, Naoe T. Expression and methylation status of the FHIT gene in acute myeloid leukemia and myelodysplastic syndrome. Leukemia. 2005;19:1367–1375. doi: 10.1038/sj.leu.2403805. [DOI] [PubMed] [Google Scholar]
- 28.Nygren AO, Ameziane N, Duarte HM, Vijzelaar RN, Waisfisz Q, Hess CJ, Schouten JP, Errami A. Methylation-specific MLPA (MS-MLPA): simultaneous detection of CpG methylation and copy number changes of up to 40 sequences. Nucleic Acids Res. 2005;33:e128. doi: 10.1093/nar/gni127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hess CJ, Errami A, Berkhof J, Denkers F, Ossenkoppele GJ, Nygren AO, Schuurhuis GJ, Waisfisz Q. Concurrent methylation of promoters from tumor associated genes predicts outcome in acute myeloid leukemia. Leuk Lymphoma. 2008;49:1132–1141. doi: 10.1080/10428190802035990. [DOI] [PubMed] [Google Scholar]