Abstract
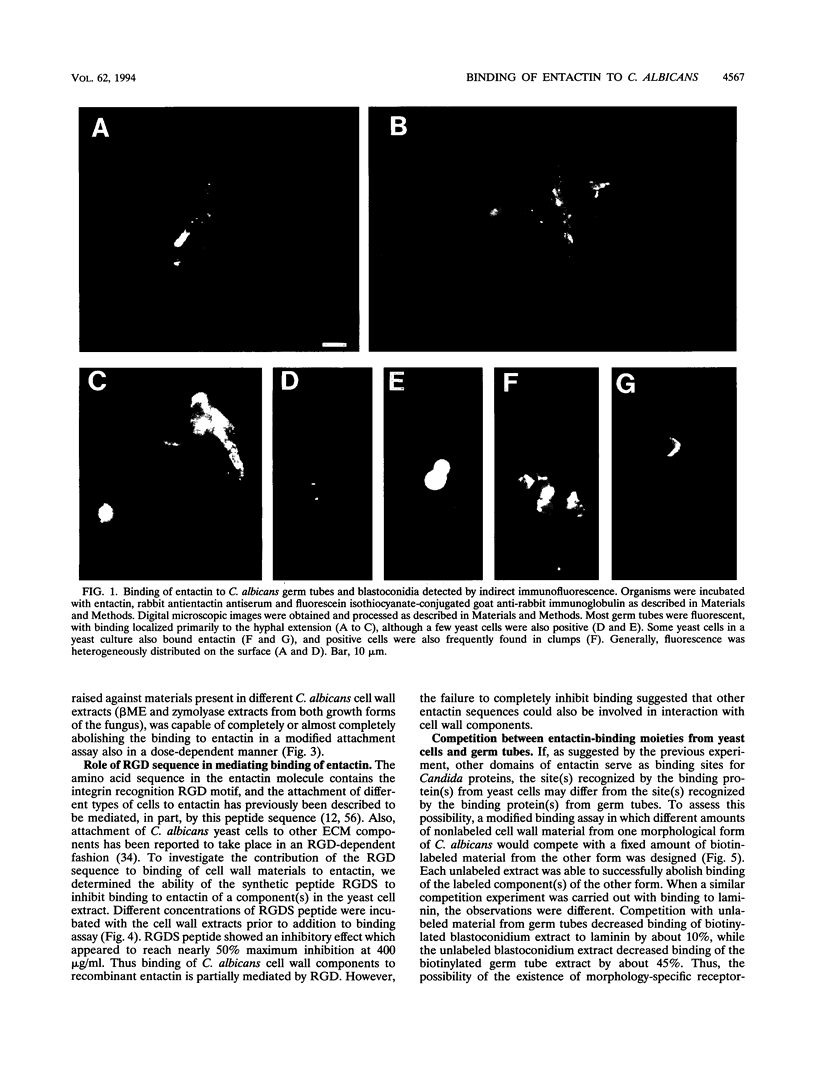
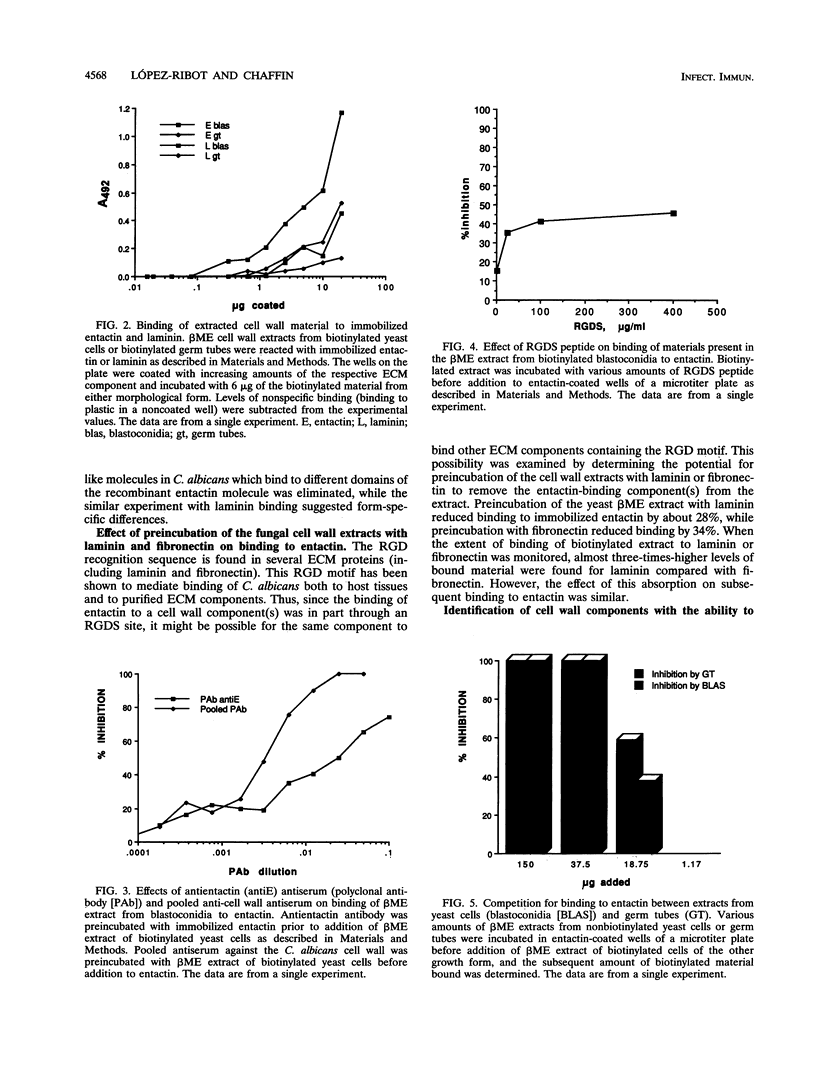
We have investigated the interaction between Candida albicans and entactin, a recently characterized glycoprotein present in the extracellular matrix, especially in the basement membrane. Organisms of both the yeast and the hyphal morphologies of the fungus had the ability to bind recombinant entactin, as detected by an indirect immunofluorescence assay. Material present in the 2-mercaptoethanol cell wall extracts from both C. albicans growth forms was capable of binding to immobilized recombinant entactin in a dose-dependent manner. Binding to entactin was approximately twice that observed for laminin. Binding of an extract component(s) to entactin was partially inhibited by an Arg-Gly-Asp-Ser peptide. A polyclonal antientactin antiserum, as well as a pooled antiserum preparation raised against components present in different C. albicans cell wall extracts, completely or almost completely abolished binding. The existence of morphology-specific receptor-like molecules which bind to different domains of the entactin molecule was ruled out in a competition binding assay. The entactin-binding material(s) in the cell wall also displayed some ability to bind laminin and fibronectin, since preadsorption in the presence of these extracellular matrix components resulted in reduction of binding to entactin. Moieties with a molecular mass of approximately 25, 44, and 65 kDa present in the 2-mercaptoethanol cell wall extracts from both blastoconidia and germ tubes were detected in a ligand affinity blotting experiment as having the ability to bind entactin. Interactions between C. albicans and entactin could be important in mediating adhesion of the fungus to the host tissues and may play a role in the establishment of the disseminated form of the disease.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alaei S., Larcher C., Ebenbichler C., Prodinger W. M., Janatova J., Dierich M. P. Isolation and biochemical characterization of the iC3b receptor of Candida albicans. Infect Immun. 1993 Apr;61(4):1395–1399. doi: 10.1128/iai.61.4.1395-1399.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouali A., Robert R., Tronchin G., Senet J. M. Characterization of binding of human fibrinogen to the surface of germ-tubes and mycelium of candida albicans. J Gen Microbiol. 1987 Mar;133(3):545–551. doi: 10.1099/00221287-133-3-545. [DOI] [PubMed] [Google Scholar]
- Bouchara J. P., Tronchin G., Annaix V., Robert R., Senet J. M. Laminin receptors on Candida albicans germ tubes. Infect Immun. 1990 Jan;58(1):48–54. doi: 10.1128/iai.58.1.48-54.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brassart D., Woltz A., Golliard M., Neeser J. R. In vitro inhibition of adhesion of Candida albicans clinical isolates to human buccal epithelial cells by Fuc alpha 1----2Gal beta-bearing complex carbohydrates. Infect Immun. 1991 May;59(5):1605–1613. doi: 10.1128/iai.59.5.1605-1613.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderone R. A., Braun P. C. Adherence and receptor relationships of Candida albicans. Microbiol Rev. 1991 Mar;55(1):1–20. doi: 10.1128/mr.55.1.1-20.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderone R. A., Linehan L., Wadsworth E., Sandberg A. L. Identification of C3d receptors on Candida albicans. Infect Immun. 1988 Jan;56(1):252–258. doi: 10.1128/iai.56.1.252-258.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderone R. A. Recognition between Candida albicans and host cells. Trends Microbiol. 1993 May;1(2):55–58. doi: 10.1016/0966-842x(93)90033-n. [DOI] [PubMed] [Google Scholar]
- Casanova M., Chaffin W. L. Cell wall glycoproteins of Candida albicans as released by different methods. J Gen Microbiol. 1991 May;137(5):1045–1051. doi: 10.1099/00221287-137-5-1045. [DOI] [PubMed] [Google Scholar]
- Casanova M., Gil M. L., Cardeñoso L., Martinez J. P., Sentandreu R. Identification of wall-specific antigens synthesized during germ tube formation by Candida albicans. Infect Immun. 1989 Jan;57(1):262–271. doi: 10.1128/iai.57.1.262-271.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casanova M., Lopez-Ribot J. L., Martinez J. P., Sentandreu R. Characterization of cell wall proteins from yeast and mycelial cells of Candida albicans by labelling with biotin: comparison with other techniques. Infect Immun. 1992 Nov;60(11):4898–4906. doi: 10.1128/iai.60.11.4898-4906.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casanova M., Lopez-Ribot J. L., Monteagudo C., Llombart-Bosch A., Sentandreu R., Martinez J. P. Identification of a 58-kilodalton cell surface fibrinogen-binding mannoprotein from Candida albicans. Infect Immun. 1992 Oct;60(10):4221–4229. doi: 10.1128/iai.60.10.4221-4229.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaffin W. L., Collins B., Marx J. N., Cole G. T., Morrow K. J., Jr Characterization of mutant strains of Candida albicans deficient in expression of a surface determinant. Infect Immun. 1993 Aug;61(8):3449–3458. doi: 10.1128/iai.61.8.3449-3458.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakravarti S., Tam M. F., Chung A. E. The basement membrane glycoprotein entactin promotes cell attachment and binds calcium ions. J Biol Chem. 1990 Jun 25;265(18):10597–10603. [PubMed] [Google Scholar]
- Chung A. E., Dong L. J., Wu C., Durkin M. E. Biological functions of entactin. Kidney Int. 1993 Jan;43(1):13–19. doi: 10.1038/ki.1993.4. [DOI] [PubMed] [Google Scholar]
- Critchley I. A., Douglas L. J. Isolation and partial characterization of an adhesin from Candida albicans. J Gen Microbiol. 1987 Mar;133(3):629–636. doi: 10.1099/00221287-133-3-629. [DOI] [PubMed] [Google Scholar]
- Cutler J. E. Putative virulence factors of Candida albicans. Annu Rev Microbiol. 1991;45:187–218. doi: 10.1146/annurev.mi.45.100191.001155. [DOI] [PubMed] [Google Scholar]
- Dedhar S., Jewell K., Rojiani M., Gray V. The receptor for the basement membrane glycoprotein entactin is the integrin alpha 3/beta 1. J Biol Chem. 1992 Sep 15;267(26):18908–18914. [PubMed] [Google Scholar]
- Durkin M. E., Chakravarti S., Bartos B. B., Liu S. H., Friedman R. L., Chung A. E. Amino acid sequence and domain structure of entactin. Homology with epidermal growth factor precursor and low density lipoprotein receptor. J Cell Biol. 1988 Dec;107(6 Pt 2):2749–2756. doi: 10.1083/jcb.107.6.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilmore B. J., Retsinas E. M., Lorenz J. S., Hostetter M. K. An iC3b receptor on Candida albicans: structure, function, and correlates for pathogenicity. J Infect Dis. 1988 Jan;157(1):38–46. doi: 10.1093/infdis/157.1.38. [DOI] [PubMed] [Google Scholar]
- Gustafson K. S., Vercellotti G. M., Bendel C. M., Hostetter M. K. Molecular mimicry in Candida albicans. Role of an integrin analogue in adhesion of the yeast to human endothelium. J Clin Invest. 1991 Jun;87(6):1896–1902. doi: 10.1172/JCI115214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Y., van Rooijen N., Cutler J. E. Binding of Candida albicans yeast cells to mouse popliteal lymph node tissue is mediated by macrophages. Infect Immun. 1993 Aug;61(8):3244–3249. doi: 10.1128/iai.61.8.3244-3249.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawkes R. Identification of concanavalin A-binding proteins after sodium dodecyl sulfate--gel electrophoresis and protein blotting. Anal Biochem. 1982 Jun;123(1):143–146. doi: 10.1016/0003-2697(82)90634-0. [DOI] [PubMed] [Google Scholar]
- Hazen K. C., Hazen B. W. Hydrophobic surface protein masking by the opportunistic fungal pathogen Candida albicans. Infect Immun. 1992 Apr;60(4):1499–1508. doi: 10.1128/iai.60.4.1499-1508.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemler M. E. Adhesive protein receptors on hematopoietic cells. Immunol Today. 1988 Apr;9(4):109–113. doi: 10.1016/0167-5699(88)91280-7. [DOI] [PubMed] [Google Scholar]
- Kalo A., Segal E., Sahar E., Dayan D. Interaction of Candida albicans with genital mucosal surfaces: involvement of fibronectin in adherence. J Infect Dis. 1988 Jun;157(6):1253–1256. doi: 10.1093/infdis/157.6.1253. [DOI] [PubMed] [Google Scholar]
- Kanbe T., Han Y., Redgrave B., Riesselman M. H., Cutler J. E. Evidence that mannans of Candida albicans are responsible for adherence of yeast forms to spleen and lymph node tissue. Infect Immun. 1993 Jun;61(6):2578–2584. doi: 10.1128/iai.61.6.2578-2584.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanbe T., Jutila M. A., Cutler J. E. Evidence that Candida albicans binds via a unique adhesion system on phagocytic cells in the marginal zone of the mouse spleen. Infect Immun. 1992 May;60(5):1972–1978. doi: 10.1128/iai.60.5.1972-1978.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanbe T., Li R. K., Wadsworth E., Calderone R. A., Cutler J. E. Evidence for expression of the C3d receptor of Candida albicans in vitro and in vivo obtained by immunofluorescence and immunoelectron microscopy. Infect Immun. 1991 May;59(5):1832–1838. doi: 10.1128/iai.59.5.1832-1838.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz A., Fish A. J., Kleppel M. M., Hagen S. G., Michael A. F., Butkowski R. J. Renal entactin (nidogen): isolation, characterization and tissue distribution. Kidney Int. 1991 Oct;40(4):643–652. doi: 10.1038/ki.1991.256. [DOI] [PubMed] [Google Scholar]
- Klotz S. A. Adherence of Candida albicans to components of the subendothelial extracellular matrix. FEMS Microbiol Lett. 1990 Mar 15;56(3):249–254. doi: 10.1016/s0378-1097(05)80049-7. [DOI] [PubMed] [Google Scholar]
- Klotz S. A., Maca R. D. Endothelial cell contraction increases Candida adherence to exposed extracellular matrix. Infect Immun. 1988 Sep;56(9):2495–2498. doi: 10.1128/iai.56.9.2495-2498.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klotz S. A., Smith R. L. A fibronectin receptor on Candida albicans mediates adherence of the fungus to extracellular matrix. J Infect Dis. 1991 Mar;163(3):604–610. doi: 10.1093/infdis/163.3.604. [DOI] [PubMed] [Google Scholar]
- Klotz S. A., Smith R. L., Stewart B. W. Effect of an arginine-glycine-aspartic acid-containing peptide on hematogenous candidal infections in rabbits. Antimicrob Agents Chemother. 1992 Jan;36(1):132–136. doi: 10.1128/aac.36.1.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lee K. L., Buckley H. R., Campbell C. C. An amino acid liquid synthetic medium for the development of mycelial and yeast forms of Candida Albicans. Sabouraudia. 1975 Jul;13(2):148–153. doi: 10.1080/00362177585190271. [DOI] [PubMed] [Google Scholar]
- Li R. K., Cutler J. E. Chemical definition of an epitope/adhesin molecule on Candida albicans. J Biol Chem. 1993 Aug 25;268(24):18293–18299. [PubMed] [Google Scholar]
- Lopez-Ribot J. L., Casanova M., Martinez J. P., Sentandreu R. Characterization of cell wall proteins of yeast and hydrophobic mycelial cells of Candida albicans. Infect Immun. 1991 Jul;59(7):2324–2332. doi: 10.1128/iai.59.7.2324-2332.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Ribot J. L., Casanova M., Monteagudo C., Sepúlveda P., Martínez J. P. Evidence for the presence of a high-affinity laminin receptor-like molecule on the surface of Candida albicans yeast cells. Infect Immun. 1994 Feb;62(2):742–746. doi: 10.1128/iai.62.2.742-746.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann K., Deutzmann R., Aumailley M., Timpl R., Raimondi L., Yamada Y., Pan T. C., Conway D., Chu M. L. Amino acid sequence of mouse nidogen, a multidomain basement membrane protein with binding activity for laminin, collagen IV and cells. EMBO J. 1989 Jan;8(1):65–72. doi: 10.1002/j.1460-2075.1989.tb03349.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcantonio E. E., Hynes R. O. Antibodies to the conserved cytoplasmic domain of the integrin beta 1 subunit react with proteins in vertebrates, invertebrates, and fungi. J Cell Biol. 1988 May;106(5):1765–1772. doi: 10.1083/jcb.106.5.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez J. P., López-Ribot J. L., Chaffin W. L. Heterogeneous surface distribution of the fibrinogen-binding protein on Candida albicans. Infect Immun. 1994 Feb;62(2):709–712. doi: 10.1128/iai.62.2.709-712.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E. Fibronectin and its receptors. Annu Rev Biochem. 1988;57:375–413. doi: 10.1146/annurev.bi.57.070188.002111. [DOI] [PubMed] [Google Scholar]
- Sasaki M., Kleinman H. K., Huber H., Deutzmann R., Yamada Y. Laminin, a multidomain protein. The A chain has a unique globular domain and homology with the basement membrane proteoglycan and the laminin B chains. J Biol Chem. 1988 Nov 15;263(32):16536–16544. [PubMed] [Google Scholar]
- Senior R. M., Gresham H. D., Griffin G. L., Brown E. J., Chung A. E. Entactin stimulates neutrophil adhesion and chemotaxis through interactions between its Arg-Gly-Asp (RGD) domain and the leukocyte response integrin. J Clin Invest. 1992 Dec;90(6):2251–2257. doi: 10.1172/JCI116111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skerl K. G., Calderone R. A., Segal E., Sreevalsan T., Scheld W. M. In vitro binding of Candida albicans yeast cells to human fibronectin. Can J Microbiol. 1984 Feb;30(2):221–227. doi: 10.1139/m84-033. [DOI] [PubMed] [Google Scholar]
- Tosh F. D., Douglas L. J. Characterization of a fucoside-binding adhesin of Candida albicans. Infect Immun. 1992 Nov;60(11):4734–4739. doi: 10.1128/iai.60.11.4734-4739.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tronchin G., Bouchara J. P., Robert R. Dynamic changes of the cell wall surface of Candida albicans associated with germination and adherence. Eur J Cell Biol. 1989 Dec;50(2):285–290. [PubMed] [Google Scholar]
- Tronchin G., Bouchara J. P., Robert R., Senet J. M. Adherence of Candida albicans germ tubes to plastic: ultrastructural and molecular studies of fibrillar adhesins. Infect Immun. 1988 Aug;56(8):1987–1993. doi: 10.1128/iai.56.8.1987-1993.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsao T., Hsieh J. C., Durkin M. E., Wu C. Y., Chakravarti S., Dong L. J., Lewis M., Chung A. E. Characterization of the basement membrane glycoprotein entactin synthesized in a baculovirus expression system. J Biol Chem. 1990 Mar 25;265(9):5188–5191. [PubMed] [Google Scholar]
- Wayner E. A., Carter W. G. Identification of multiple cell adhesion receptors for collagen and fibronectin in human fibrosarcoma cells possessing unique alpha and common beta subunits. J Cell Biol. 1987 Oct;105(4):1873–1884. doi: 10.1083/jcb.105.4.1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C., Chung A. E. Potential role of entactin in hemostasis. Specific interaction of entactin with fibrinogen A alpha and B beta chains. J Biol Chem. 1991 Oct 5;266(28):18802–18807. [PubMed] [Google Scholar]
- Wu C., Reing J., Chung A. E. Entactin forms a complex with fibronectin and co-localizes in the extracellular matrix of the embryonal carcinoma-derived 4CQ cell line. Biochem Biophys Res Commun. 1991 Aug 15;178(3):1219–1225. doi: 10.1016/0006-291x(91)91023-6. [DOI] [PubMed] [Google Scholar]
- Yelian F. D., Edgeworth N. A., Dong L. J., Chung A. E., Armant D. R. Recombinant entactin promotes mouse primary trophoblast cell adhesion and migration through the Arg-Gly-Asp (RGD) recognition sequence. J Cell Biol. 1993 May;121(4):923–929. doi: 10.1083/jcb.121.4.923. [DOI] [PMC free article] [PubMed] [Google Scholar]