Abstract
The development of normal and abnormal glandular structures in the prostate is controlled at the endocrine and paracrine levels by reciprocal interactions between epithelium and stroma. To study these processes, it is useful to have an efficient method of tissue acquisition for reproducible isolation of cells from defined histologies. Here we assessed the utility of a standardized system for acquisition and growth of prostatic cells from different regions of the prostate with different pathologies, and we compared the abilities of stromal cells from normal peripheral zone, benign prostatic hyper-plasia (BPH-S), and cancer to induce the growth of a human prostatic epithelial cell line (BPH-1) in vivo. Using the tissue recombination method, we showed that grafting stromal cells (from any histology) alone or BPH-1 epithelial cells alone produced no visible grafts. Recombining stromal cells from normal peripheral zone with BPH-1 cells also produced no visible grafts (n = 15). Recombining BPH-S with BPH-1 cells generated small, well-organized, and sharply demarcated grafts approximately 3–4 mm in diameter (n = 9), demonstrating a moderate inductive ability of BPH-S. Recombining stromal cells from cancer with BPH-1 cells generated highly disorganized grafts that completely surrounded the host kidney and invaded into adjacent renal tissue, demonstrating induction of an aggressive phenotype. We conclude that acquisition of tissue from toluidine blue dye-stained specimens is an efficient method to generate high-quality epithelial and/or stromal cultures. Stromal cells derived by this method from areas of BPH and cancer induce epithelial cell growth in vivo, which mimics the natural history of these diseases.
PRIMARY CULTURE OF prostatic cells from surgical specimens is a valuable in vitro model that can offer insight into normal prostatic growth regulation and the changes that occur during disease. However, complex endocrine and/or paracrine interactions between epithelium and stroma that may be important for prostatic disease such as prostate cancer or benign prostatic hyperplasia (BPH) are not easily studied in vitro.
Gerald R. Cunha (1–6) has developed tissue reconstitution methods for urogenital-derived tissues that allow for evaluating the roles of epithelial-stromal interactions in glandular morphogenesis and tumorigenesis. These landmark experiments over the last several decades have driven our understanding of the complexity of phenomena that contribute to the genetic and epigenetic control of tissue structure and function. Of particular note is the development by Cunha and colleagues (7) of a model of human prostate cancer development that relies on epithelial-stromal interactions. This model system uses the human prostatic epithelial cell line (BPH-1), which was originally derived from benign prostatic tissue after Simian virus 40 large T antigen immortalization of primary cells. These cells represent an initiated prostatic cell. Alone they are not tumorigenic when xenografted sc or under the renal capsule of nude mice. However, when the cells are recombined with tumor-associated prostatic stromal cells and grafted under the renal capsules of nude mice, the BPH-1 cells form invasive tumors. In contrast, normal prostatic stromal cells do not stimulate BPH-1 tumorigenesis. These elegant experiments highlight the intimate contribution of the stromal microenvironment to prostate tumorigenesis. BPH is a nonmalignant disease of the prostate that may be the result of disruption of normal adult epithelial-stromal signaling. The effects of BPH-derived prostatic stromal cells on the growth of BPH-1 grafts have not been previously investigated.
A major limitation to use of the prostate recombination model described above is the availability of stromal cells from histologically distinct regions of the prostate with well-defined pathology. Peehl and colleagues (8, 9) developed the best-characterized culture system for the isolation and maintenance of human prostatic cells. In their method, a small sample of tissue is dissected from the prostatectomy specimen under aseptic conditions. The area of the prostate from which the specimen is removed is inked and fixed for subsequent pathological evaluation.
The advantage of the system described by Peehl et al. (10) is the ability to isolate cells from the various regions of the prostate (peripheral zone, central zone, or transition zone) and specific histological phenotypes (normal, BPH, or cancer).
The major disadvantage of the methods developed by Peehl and colleagues is that the initial identification of tumor in the fresh tissue relies on palpation of the prostate to identify nodules. The pathology of the sample is not known until considerable cell culture work has been performed to grow and bank the cells. In the era of prostate-specific antigen screening, palpable nodules of sufficient size are increasingly rare in many clinical practices. In addition, the exact margins of the tumor are not readily visible by gross examination. Fischer et al. (11) described a method of tissue acquisition in which cross-sections of the prostate are stained with toluidine blue dye. Toluidine blue dye stains nuclei dark blue and allows for the immediate visual examination of glandular morphology using a dissecting microscope. In that report they described the utility of this system for epithelial cell acquisition. However, the utility of this system for stromal cell acquisition and studying epithelial-stromal interactions has not been explored.
Materials and Methods
Sample acquisition
All prostatectomies were performed by urologists in the urology department at Wake Forest University School of Medicine. All patients gave informed consent for use of surgical specimens for research. Use of surgical specimens for tissue culture was approved by the Institutional Review Board of Wake Forest University School of Medicine. The procedure used for sample acquisition was an adaptation of the procedure described by Fischer et al. (11) (Fig. 1A). A 3- to 5-mm cross-section was cut in the horizontal plane of the prostate. The sample was then stained with sterile 0.5% aqueous toluidine blue. Inspection of glandular structure was performed using an SMZ 1500 stereo dissecting scope (Nikon, Tokyo, Japan) connected to a Nikon DXM1200 digital camera. A sterile cork borer (no. 1, 2, or 3, depending on the size of the tumor) was used to remove a plug from the area of interest. Each hole in a prostate cross-section was inked with a different color dye. These areas were incorporated into the routine processing of the prostatectomy for pathological analyses. The plug that was removed for culture was processed by our normal primary culture technique (see below) except that a longitudinal slice was removed for histologic examination. This slice was fixed, embedded in paraffin, and stained with hematoxylin and eosin.
Fig. 1.
Acquisition and culture of human prostate specimens. A, Flow chart and schematic of sample acquisition. Posterior view of prostate with attached seminal vesicles showing removal of a complete cross-section. Open circles in cross-section indicate hypothetical locations of plug removal after inspection of toluidine staining under a high-power dissecting scope. Arrows from circles indicate the flow of plugs for tissue culture. Arrow below indicates flow of the remainder of prostate specimen. Diagonal arrow indicates the flow of the longitudinal slice for pathological evaluation. TZ, Transition zone. B, Schematic of processing of samples removed for growth of epithelial and/or stromal cells as described above.
Histochemistry and pathology
All histology including fixation, embedding, sectioning, staining, and cytokeratin 903 immunohistochemistry of prostatectomy samples and longitudinal sections of plugs was performed by the pathology department's clinical laboratory using routine procedures. Blocks that incorporated the stained hole for each sample taken were made. In addition, blocks containing the longitudinal slice of plugs were prepared. Samples were classified as cancer if the hole and longitudinal section from which it was derived was surrounded by cancer glands in 90% of the area. Cork-borer samples were classified as mixed if the hole (and longitudinal section) from which it was derived was surrounded by a mixture of cancer and benign glands, in which the proportion of cancer glands was less than 90% of the total. Samples were classified as benign if there were no cancer glands surrounding the hole or in the longitudinal section.
Isolation and maintenance of prostatic cells
Prostatic epithelial cells were isolated using minor modifications of the protocol of Peehl (12, 13) as previously published by us (14, 15). Stromal cells were isolated by methods previously described (16). A schematic of these procedures is depicted in Fig. 1B. Nomenclature for epithelial cell strains is WFU followed by the strain number (in serial order of acquisition) and then the histology of origin (PZ, peripheral zone; BPH; CA, cancer) (e.g. WFU9CA). Stromal cell strains have an additional S at the end (e.g. WFU9CA-S).
Prostatic tissue recombinants and renal grafting
All use of animals was in accordance with policies and guidelines from the Animal Care and Use Committee of Wake Forest University School of Medicine. BPH-1 cells were obtained from Dr. Cunha. Stromal cells were cultured from fresh prostatic tissue as described above. To prepare tissue recombinants, semiconfluent stromal cells (PZ-S, BPH-S, or CA-S) were mixed with BPH-1 cells at a ratio of 250,000 stromal cells to 100,000 BPH-1 cells in a sterile microcentrifuge tube in 1 ml HEPES-buffered saline. The cell mixture was pelleted and resuspended in 50 μl of rat-tail collagen (pretitrated to pH 7.4) and inoculated in the center of a well of a multiwell plate. After collagen polymerization the button was overlaid with normal growth medium (RPMI 1640 plus 5% fetal bovine serum). Sixteen to 24 h later the tissue recombinant button was grafted under the renal capsule of a nude mouse using published protocols from Dr. Cunha's laboratory (1–6).
Results and Discussion
Toluidine blue dye tissue acquisition
We adopted a prospective technique for acquiring cell populations with defined pathological origin. Our procedure is an adaptation of the procedure described by Fischer et al. (11). Figure 2 shows typical images from a toluidine blue-stained cross-section. The sample is scanned at low magnification (×10) to identify the areas of potential benign tissue (Fig. 2A). These areas are then compared with areas of potential tumor tissue (Fig. 2D). The prostate specimen depicted in Fig. 2 contains a transition zone tumor (Fig. 2D). This is evident by comparing the size of the adjacent benign glands with putative tumor glands at a given magnification. Usually we use ×30 magnification for this comparison (Fig. 2, B and E). Magnification at ×100 is used for further validation (Fig. 2, C and F). Also shown in Fig. 2D is the presence of a stromal nodule. These are often clearly delineated from surrounding tissue by their paucity of glandular structures and bulging nature. Samples from these nodules can be removed and grown for BPH-derived stromal and epithelial cells.
Fig. 2.

Toluidine blue-stained cross-sections of fresh prostatectomy specimens. Typical images of toluidine blue-stained prostate cross-section (n = 10 cross-sections). All photomicrographs are from the same cross-section prepared as described in Materials and Methods under Sample acquisition. An area of benign-appearing glands is depicted in A–C. An area of apparent cancerous-appearing glands is depicted in D–F. The arrows indicate the area used for further magnification. A and D, magnification, ×10; B and E, magnification, ×30; C and F, magnification, ×100. Compare gland size, closeness, and regularity between B and E or between C and F.
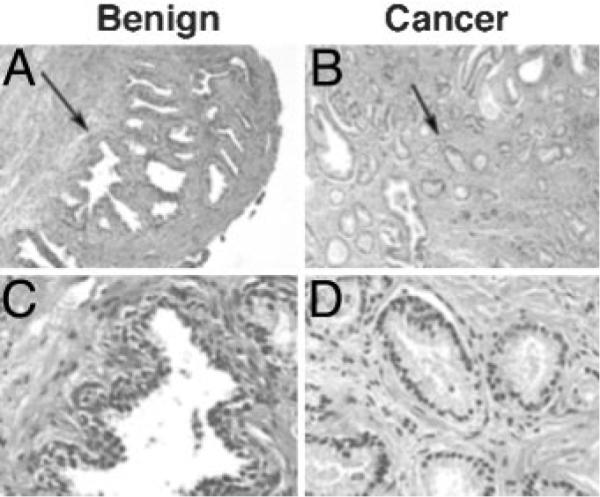
Once we are confident of the pathology of the area, we obtain a plug using a sterile cork borer as described in Materials and Methods. Each hole in a prostate cross-section is inked with a different color dye. Figure 3 shows sections from the pathology blocks containing the inked holes in the prostate sample depicted in Fig. 2. The hole remaining after the plug was removed from the predicted benign sample is depicted in Fig. 3A. The adjacent glandular architecture is indicative of a benign sample. To confirm this, we probed an adjacent section for the expression of basal cytokeratins (Fig. 3, B and C), a marker of benign glandular structure. The results confirm our prospective analysis of the sample that this region contains only benign glands with an intact basal layer. The hole remaining after the plug was removed for the predicted cancer specimen was inked orange and is visible in Fig. 3D. The adjacent glandular architecture indicates a cancer sample. There is a complete lack of basal cells in these adjacent glands shown by a lack of basal-specific cytokeratin expression (Fig. 3, E and F) and the structure of the glands as a single cell layer (Fig. 3F).
Fig. 3.

Histopathology of prostate surrounding plugs removed for culture. Photomicrographs of areas of benign (A–C) and cancer (D–F) prostatic tissue obtained from the same prostate cross-section depicted in Fig. 2 and adjacent to plugs used for culture. Samples for culture were acquired by the toluidine blue dye method, the hole was inked (note green and orange ink in holes), and the adjacent tissue was fixed and embedded in paraffin by routine histological techniques. Sections from the blocks were prepared and stained with hematoxylin and eosin (A and D, magnification, ×40). Adjacent sections were incubated with an antibody against basal-specific high-molecular-weight cytokeratins (Cytokeratin 903) and peroxidase stained (B and E, magnification, ×100; C and F, magnification, ×400). Arrows in A and D indicate areas used for higher-magnification photomicrographs.
The removed plug is processed by our normal primary culture technique except that a longitudinal slice is removed for histology before dissociation of the tissue. The growth of the derived cultures does not appear to be compromised by the toluidine staining technique (data not shown).
The longitudinal slice removed from the plug is fixed, embedded in paraffin, and stained with hematoxylin and eosin. Figure 4 shows the sections from the longitudinal slices from the plugs removed from the samples depicted in Fig. 2. The results demonstrate our ability to prospectively predict the location of benign or cancer glands in fresh specimens (Fig. 4, A and B).
Fig. 4.
Histopathology of longitudinal section of plugs removed for culture. Photomicrographs of a longitudinal slice from a plug of benign (A and D) and cancer (B and C) prostatic tissue obtained from the same prostate cross-section depicted in Figs. 2 and 3. A longitudinal slice from the acquired plug was removed, and the tissue was fixed and embedded in paraffin by routine histological techniques. Sections from the blocks were prepared and stained with hematoxylin and eosin (A and B, magnification, ×100; C and D, magnification, ×400). Arrows in A and B indicate areas used for higher-magnification photomicrographs.
We sampled 10 prostatectomy specimens between July 2002 and December 2002 using the toluidine blue staining method to observe glandular architecture. Of 27 samples collected from these specimens, 14 samples appeared to be cancer and 13 appeared to be benign judging from the toluidine blue dye-staining pattern. Twelve of 14 putative cancer samples (86%) were confirmed cancer by pathologic examination of adjacent tissue and the longitudinal plug. The remaining two putative cancer samples were determined to be of mixed origin. All 13 putative benign samples (100%) were confirmed benign samples by pathologic examination. Of the confirmed benign and cancer-derived samples, eight sets are matched to the same prostate. This system allows for efficient acquisition of patient-matched normal and diseased prostatic tissue.
BPH-S and CA-S induce prostate epithelial growth in vivo
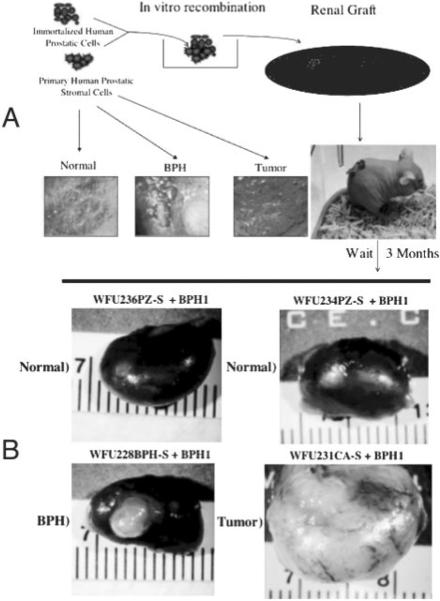
To validate our histological findings in a functional assay and test the utility of this acquisition for studying epithelialstromal interactions, we xenografted prostate tissue recombinants under the renal capsules of nude mice. Figure 5A shows a schematic of the protocol. Using this protocol, we established 15 grafts from PZ-S/BPH-1 recombinants, nine grafts from BPH-S/BPH-1 recombinants, and seven grafts from CA-S/BPH-1 recombinants. Each animal was grafted with two or three PZ-S/BPH-1 recombinant buttons on the left kidney, and the contralateral kidney was grafted with two grafts of either BPH-S/BPH-1 or CA-S/BPH-1 recombinants. We also established two grafts each of PZ-S, BPH-S, CA-S, or BPH-1 cells alone as controls. No growth of any of these control grafts was observed (data not shown). Figure 5B shows representative results from grafts with prostate tissue recombinants. All PZ-S/BPH-1 recombinants produced grafts that contained little or no growth (1 mm or less). The top left and right images in Fig. 5B are typical of results from BPH-1 recombinants with the two different PZ-S strains (WFU236PZ-S and WFU234PZ-S). Note the flattened opaque collagen disks visible at the site of the grafts. These results are consistent with other observations that show that stromal cells from the normal peripheral zone have little or no inductive ability.
Fig. 5.
BPH-S and CA-S cells induce prostate epithelial growth. A, Schematic of prostate tissue recombination protocol. Isolated prostatic stromal cells of defined histological origin (representative toluidine-stained sections are depicted) are combined with BPH-1 cells in vitro in a collagen gel matrix. The recombinant button is grafted under the renal capsule of a nude mouse and allowed to grow for 3 months. B, Representative photomicrographs of dissected kidneys grafted with prostate tissue recombinants. The stromal strain used for each graft is indicated above the corresponding photomicrograph.
In contrast, all BPH-1 recombinants with BPH-S cells produced 3- to 4-mm-diameter grafts that were sharply circumscribed (the lower left image of Fig. 5B shows a representative graft). Both BPH-S strains (WFU225BPH-S and WFU228BPH-S) showed a similar inductive ability. Histological examination of these grafts revealed densely packed, well-organized tubular epithelium with minimal stroma, sharply demarcated from surrounding renal tissue (Fig. 6, A and B). To our knowledge, the use of BPH-S in this recombination system has not been previously tested.
Fig. 6.
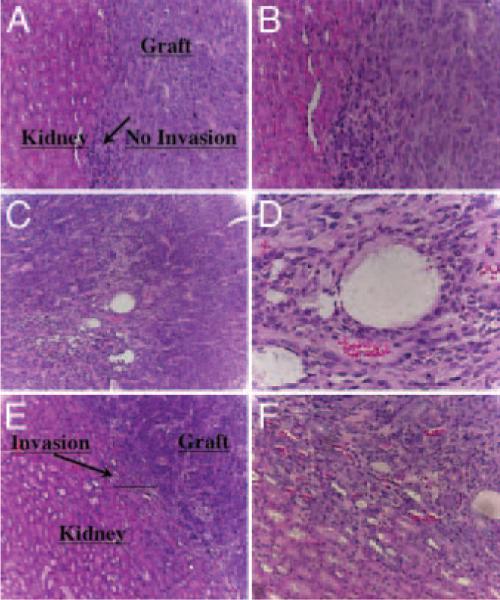
Histology of BHP-S/BPH-1 and CA-S/BPH-1 recombinants. Kidneys were dissected from animals, fixed in 10% formalin, and embedded in paraffin, and 5-μM sections through the grafts were made. Digital images show hematoxylin-eosin stained sections. A (magnification, ×10) and B (magnification, ×20), Graft-kidney interface of a BPH-S/BPH-1 recombinant showing sharp demarcation between the interfaces. Note the organization of the graft into well-ordered solid tubular structures. C (magnification, ×10) and D (magnification, ×40), CA-S/BPH-1 grafts showing a more disorganized structure with high vascularity. E (magnification, ×10) and F (magnification, ×20), Graft-kidney interface of a CA-S/BPH-1 recombinant showing infiltration of the graft and associated neovasculature into the adjacent renal tissue.
McNeal (17) suggested that BPH is a disease of the prostatic stroma. His observations have led to the hypothesis that BPH is a result of the reawakening of the embryonic inductive abilities of the prostatic stroma. The inductive abilities of the BPH stroma are thought to be the normal embryonic signals that induce normal epithelial structures. Our results are suggestive of this. BPH-1 cells are a transformed cell line that cannot differentiate into ductal structures with differentiated lumenal and basal cells. However, the sharp demarcation between the graft and kidney as well as the well-ordered appearance of the solid tubes reflects a relatively benign structure. Some in vitro data that show disruptions of the IGF signaling axis in BPH-S support McNeal's hypothesis (18, 19). However, our studies are the first evidence to support a functional inductive capacity of BPH-S in prostatic tissue recombinants. The simplified method of acquiring prostatic stromal cells described here, combined with the prostate tissue recombination model, should facilitate dissecting the signaling pathways leading to BPH.
Stunningly, BPH-1 recombinants with CA-S cells produced much more robust growth than the other recombinants tested. The lower right image in Fig. 5B shows an extreme example of this aggressive growth induced by the CA-S cell strain WFU231CA-S (note that all images in Fig. 5B are of the same magnification). A second CA-S strain (WFU233CA-S) also produced aggressive-looking grafts, but these grafts did not completely encapsulate the kidney (data not shown). Histological assessment of the graft reveals a moderately differentiated, highly vascularized tumor (Fig. 6, C and D) with invasion into surrounding renal tissue (Fig. 6, E and F). Our data confirm the findings reported by Olumi et al. (7) that demonstrate the ability of CA-S to produce aggressive prostatic tumors when recombined with BPH-1 cells. It also appears that the phenotype from the CA-S induction is more aggressive that that from the BPH-S induction. The CA-S induces more ragged margins with invasion into the kidney, whereas the BPH-S induces more sharply circumscribed margins with no evidence of invasion. These differences in the phenotypes between these two recombinant models suggest fundamental differences in the nature of the inducers. Further investigation of these models will be facilitated by the systems outlined here.
Development of adenocarcinoma is usually thought of in the context of the somatic mutation theory in which the epithelium is the primary target of mutagenesis (20). The results reported here support a growing body of work from a number of different epithelial tumor systems demonstrating that the tumor microenvironment and specifically, epithelial-mesenchymal interactions are critically important for tumorigenesis (reviewed by Ingber in Ref. 21). Additionally, recent evidence from rodent mammary carcinogenesis demonstrates that the stroma is a direct target of carcinogenic insult that may supersede genetic insult to the epithelium (22). These studies argue that more focused attention should be directed toward the nature of cancer-associated stroma. The system reported here will provide a means to study the specific roles of the stroma and its interaction with epithelium in tumorigenesis.
In summary, we have validated a method of determining prospectively the glandular architecture (malignant or benign) of fresh prostatic samples collected for use in tissue culture. The system reduces the waste of collecting and culturing specimens with undesirable attributes (mixed benign and malignant origin). We can acquire defined cancer samples from a majority of prostatectomy specimens. This will increase the availability of primary cancer strains for prostate cancer research. Stromal nodules can also be easily identified, which should contribute to the availability of BPH-derived stromal lines. Using this system, we have demonstrated fundamental differences in the inductive capabilities of stromal cells derived from normal or diseased prostatic tissue. The model systems described here should facilitate the investigation of epithelial stromal interactions in the etiology of prostatic diseases.
Acknowledgments
The authors thank Jerry Cunha for providing the BPH-1 cell line and most importantly for his advice and training with the prostate tissue recombination method. This study would not have been possible without his openness and willingness to provide training. We also thank Greg Kucera (Tumor Tissue Procurement Core, Wake Forest University School of Medicine) for assistance in acquiring prostate samples.
This work was supported by grants from the National Institutes of Health (DK52623, CA101023) and the Kulynych Interdisciplinary Cancer Research Fund (to S.D.C.). W.W.B. was supported by a training grant from the National Cancer Institute (T32 CA079448).
Abbreviations
- BPH
Benign prostatic hyperplasia
- BPH-1
human prostatic epithelial cell line
- CA
cancer
- PZ
peripheral zone
Footnotes
Endocrinology is published monthly by The Endocrine Society (http://www.endo-society.org), the foremost professional society serving the endocrine community.
References
- 1.Cunha G. Epithelio-mesenchymal interaction in primordial gland structures which become responsive to androgenic stimulation. Anat Rec. 1972;172:179–196. doi: 10.1002/ar.1091720206. [DOI] [PubMed] [Google Scholar]
- 2.Cunha GR, Chung LW. Stromal-epithelial interactions—I. Induction of prostatic phenotype in urothelium of testicular feminized (Tfm/y) mice. J Steroid Biochem. 1981;14:1317–1324. doi: 10.1016/0022-4731(81)90338-1. [DOI] [PubMed] [Google Scholar]
- 3.Cunha GR, Fujii H, Neubauer BL, Shannon JM, Sawyer L, Reese BA. Epithelial-mesenchymal interactions in prostatic development. I. morphological observations of prostatic induction by urogenital sinus mesenchyme in epithelium of the adult rodent urinary bladder. J Cell Biol. 1983;96:1662–1670. doi: 10.1083/jcb.96.6.1662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cunha GR. Androgenic effects upon prostatic epithelium are mediated via trophic influences from stroma. Prog Clin Biol Res. 1984;145:81–102. [PubMed] [Google Scholar]
- 5.Cunha G, Donjacour A, Cooke P, Mee S, Bigsby RM, Higgins SJ, Sugimura Y. The endocrinology and developmental biology of the prostate. Endocr Rev. 1987;8:338–362. doi: 10.1210/edrv-8-3-338. [DOI] [PubMed] [Google Scholar]
- 6.Day K, McCabe M, Zhao X, Wang Y, Davis JN, Phillips J, Von Geldern M, Ried T, KuKuruga MA, Cunha GR, Hayward SW, Day ML. Rescue of embryonic epithelium reveals that the homozygous deletion of the retinoblastoma gene confers growth factor independence and immortality but does not influence epithelial differentiation of tissue morphogenesis. J Biol Chem. 2002;277:44475–44484. doi: 10.1074/jbc.M205361200. [DOI] [PubMed] [Google Scholar]
- 7.Olumi A, Grossfeld G, Hayward S, Carroll P, Tlsty T, Cunha G. Carcinma-associated fibroblasts direct tumor progression of initiated human prostate epithelium. Cancer Res. 1999;59:5002–5011. doi: 10.1186/bcr138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Peehl D. Are primary cultures realistic models of prostate cancer? J Cell Biochem. 2004;91:185–195. doi: 10.1002/jcb.10691. [DOI] [PubMed] [Google Scholar]
- 9.Peehl DM, Sellers RG. Basic FGF, EGF, and PDGF modify TGFβ-induction of smooth muscle cell phenotype in human prostatic stromal cells. Prostate. 1998;35:125–134. doi: 10.1002/(sici)1097-0045(19980501)35:2<125::aid-pros6>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 10.Peehl D, Wong S, Bazinet M, Stamey T. In vitro studies of human prostatic epithelial cells: attempts to identify distinguishing features of malignant cells. Growth Factors. 1989;1:237–250. doi: 10.3109/08977198908998000. [DOI] [PubMed] [Google Scholar]
- 11.Fischer A, Phillips A, Taysavang P, McKenney J, Amin A. Method for procuring specific populations of viable human prostate cells for research. Lab Invest. 2001;81:501–507. doi: 10.1038/labinvest.3780258. [DOI] [PubMed] [Google Scholar]
- 12.Peehl DM. Serial culture of adult human prostatic epithelial cells. J Tissue Cult Tech. 1985;9:53–60. [Google Scholar]
- 13.Peehl DM. Culture of human prostatic epithelial cells. In: Freshney IA, editor. Culture of epithelial cells. Wiley Liss Inc.; New York: 1992. pp. 159–180. [Google Scholar]
- 14.Barreto A, Schwartz G, Woodruff R, Cramer S. 25-Hydroxyvitamin D3, the prohormone of 1,25-dihydroxyvitamin D3, inhibits the proliferation of primary prostatic epithelial cells. Cancer Epidemiol Biomarkers Prev. 2000;9:265–270. [PubMed] [Google Scholar]
- 15.Rao A, Woodruff R, Wade W, Kute T, Cramer S. Synergistic Inhibition of prostatic epithelial cell growth by genistein and vitamin D. J Nutr. 2002;132:3191–3194. doi: 10.1093/jn/131.10.3191. [DOI] [PubMed] [Google Scholar]
- 16.Peehl DM, Sellers RG. Induction of smooth muscle cell phenotype in cultured human prostatic stromal cells. Exp Cell Res. 1997;232:208–215. doi: 10.1006/excr.1997.3525. [DOI] [PubMed] [Google Scholar]
- 17.McNeal J. Pathology of benign prostatic hyperplasia. Insight into etiology. Urol Clin North Am. 1990;17:477–486. [PubMed] [Google Scholar]
- 18.Cohen P, Peehl DM, Rosenfeld R. Insulin-like growth factor 1 in relation to prostate cancer and benign prostatic hyperplasia. Br J Cancer. 1998;78:554–556. doi: 10.1038/bjc.1998.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dong G, Rajah R, Vu T, Hoffman AR, Rosenfeld RG, Roberts CT, Jr, Peehl DM, Cohen P. Decreased expression of Wilms' tumor gene WT-1 and elevated expression of insulin growth factor-II (IGF-II) and type 1 IGF receptor genes in prostatic stromal cells from patients with benign prostatic hyperplasia. J Clin Endocrinol Metab. 1997;82:2198–2203. doi: 10.1210/jcem.82.7.4067. [DOI] [PubMed] [Google Scholar]
- 20.Hanahan D, Wienberg R. The hallmarks of cancer. Cell. 2000;100:57–70. doi: 10.1016/s0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 21.Ingber D. Cancer as a disease of epithelial-mesenchymal interactions and extracellular matrix regulation. Differentiation. 2002;70:547–560. doi: 10.1046/j.1432-0436.2002.700908.x. [DOI] [PubMed] [Google Scholar]
- 22.Maffini M, Soto A, Calabro J, Ucci A, Sonnenschein C. The stroma as a crucial target in rat mammary gland carcinogenesis. J Cell Sci. 2004;117:1495–1502. doi: 10.1242/jcs.01000. [DOI] [PubMed] [Google Scholar]