Abstract
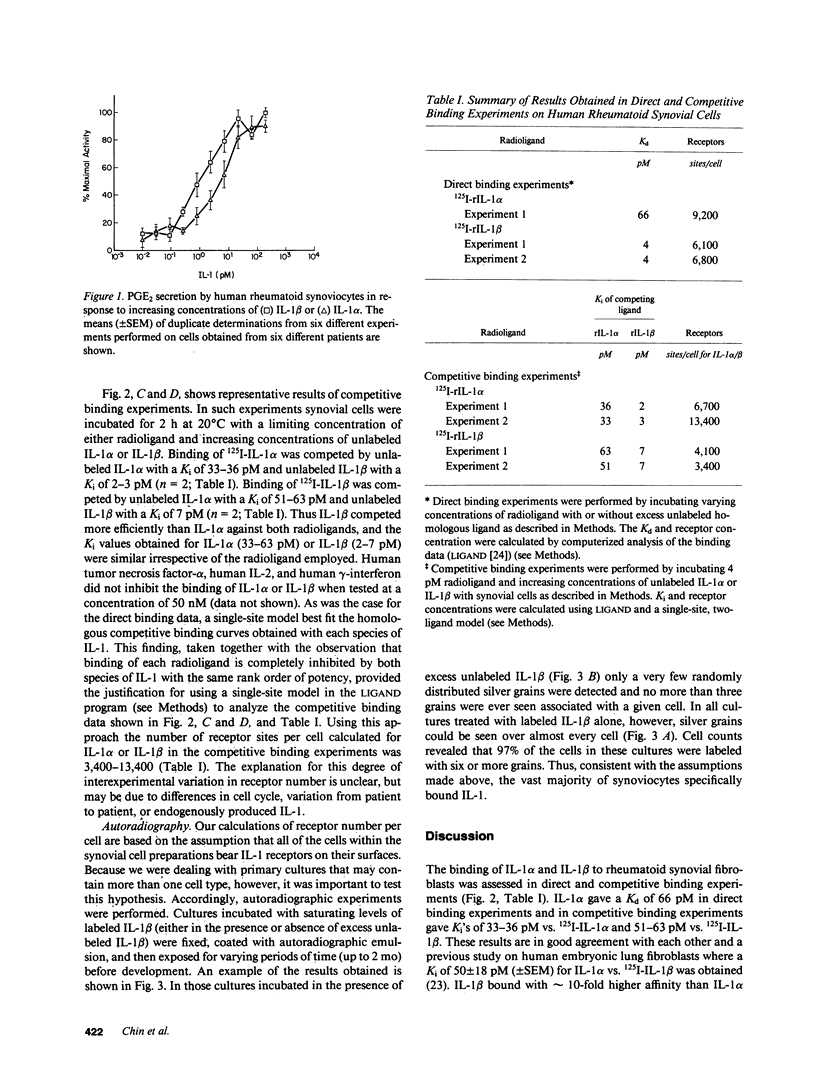
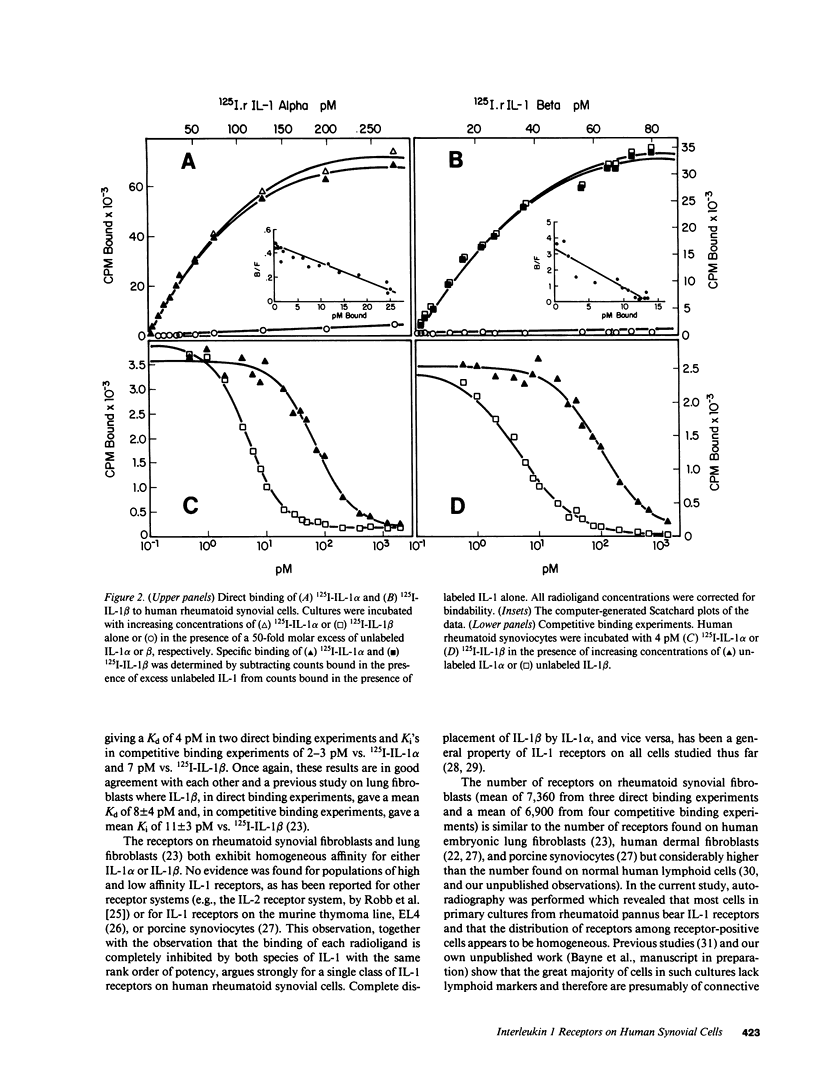
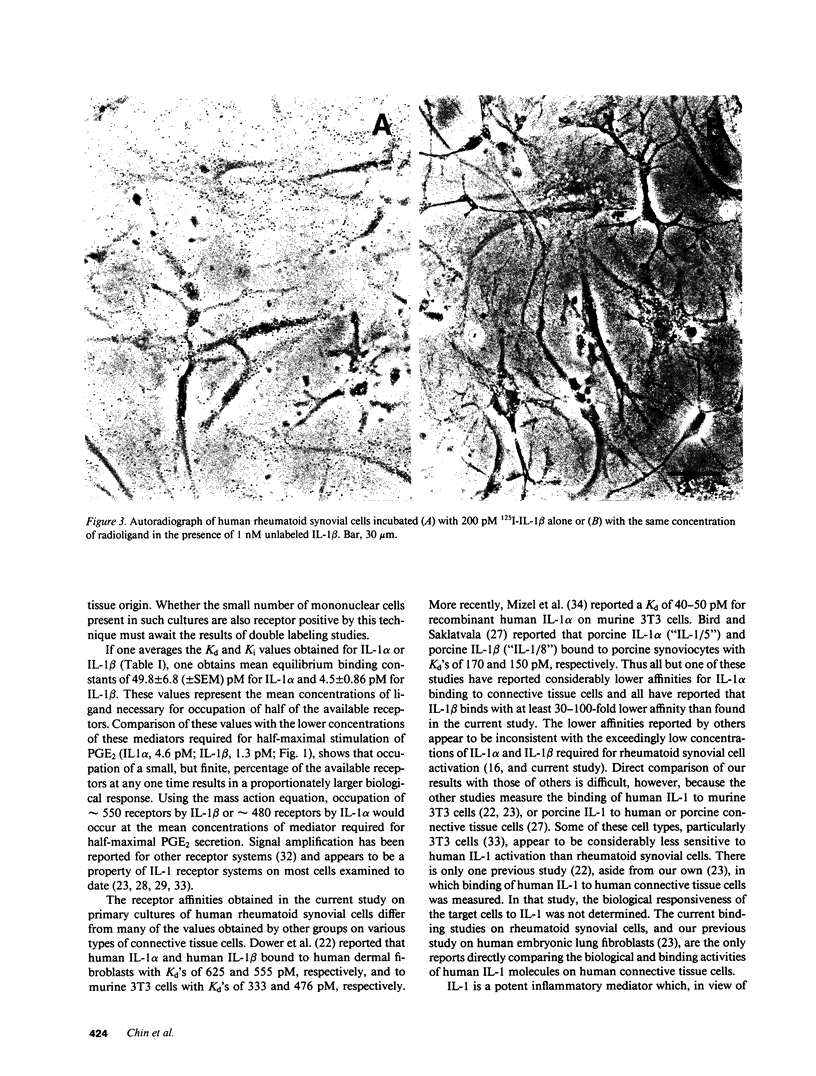
In this report the binding of recombinant human interleukins 1 alpha and 1 beta (rIL-1 alpha and rIL-1 beta) to primary cultures of human rheumatoid synovial cells is measured and compared to the concentrations of these mediators required for stimulation of PGE2 production by these same cells. The average concentration of IL-1 alpha required for half-maximal stimulation of PGE2 was 4.6 +/- 1.5 pM (+/- SEM) (n = 6), whereas for IL-1 beta half-maximal stimulation was observed at a concentration of 1.3 +/- 0.24 pM (n = 6). Both direct and competitive binding experiments were performed. In direct binding experiments, IL-1 alpha bound with a Kd of 66 pM (n = 1), while IL-1 beta bound with a Kd of 4 pM (n = 2). In competitive binding experiments, IL-1 alpha inhibited binding of 125I-IL-1 alpha with a Ki of 33-36 pM (n = 2) and binding of 125I-IL-1 beta with a Ki of 51-63 pM (n = 2). IL-1 beta inhibited binding of 125I-IL-1 alpha with a Ki of 2-3 pM (n = 2) and binding of 125I-IL-1 beta with a Ki of 7 pM (n = 2). The binding data were best fit by a model specifying a single class of receptors with homogeneous affinity for either IL-1 alpha or IL-1 beta and with an abundance of 3,000-14,000 sites per cell. Autoradiography showed that the vast majority of the synoviocytes within the cultures possessed IL-1 receptors. Comparison of biological response curves with the binding curves indicates that the observed receptors exhibit sufficiently high affinity to mediate the response of human synoviocytes to low picomolar concentrations of IL-1 alpha and IL-1 beta.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker D. G., Dayer J. M., Roelke M., Schumacher H. R., Krane S. M. Rheumatoid synovial cell morphologic changes induced by a mononuclear cell factor in culture. Arthritis Rheum. 1983 Jan;26(1):8–14. doi: 10.1002/art.1780260102. [DOI] [PubMed] [Google Scholar]
- Bird T. A., Saklatvala J. Identification of a common class of high affinity receptors for both types of porcine interleukin-1 on connective tissue cells. Nature. 1986 Nov 20;324(6094):263–266. doi: 10.1038/324263a0. [DOI] [PubMed] [Google Scholar]
- Cameron P. M., Limjuco G. A., Chin J., Silberstein L., Schmidt J. A. Purification to homogeneity and amino acid sequence analysis of two anionic species of human interleukin 1. J Exp Med. 1986 Jul 1;164(1):237–250. doi: 10.1084/jem.164.1.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cameron P., Limjuco G., Rodkey J., Bennett C., Schmidt J. A. Amino acid sequence analysis of human interleukin 1 (IL-1). Evidence for biochemically distinct forms of IL-1. J Exp Med. 1985 Sep 1;162(3):790–801. doi: 10.1084/jem.162.3.790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin J., Cameron P. M., Rupp E., Schmidt J. A. Identification of a high-affinity receptor for native human interleukin 1 beta and interleukin 1 alpha on normal human lung fibroblasts. J Exp Med. 1987 Jan 1;165(1):70–86. doi: 10.1084/jem.165.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clemmensen I., Hølund B., Andersen R. B. Fibrin and fibronectin in rheumatoid synovial membrane and rheumatoid synovial fluid. Arthritis Rheum. 1983 Apr;26(4):479–485. doi: 10.1002/art.1780260405. [DOI] [PubMed] [Google Scholar]
- Dayer J. M., Passwell J. H., Schneeberger E. E., Krane S. M. Interactions among rheumatoid synovial cells and monocyte-macrophages: production of collagenase-stimulating factor by human monocytes exposed to concanavalin A or immunoglobulin Fc fragments. J Immunol. 1980 Apr;124(4):1712–1720. [PubMed] [Google Scholar]
- Dewhirst F. E., Stashenko P. P., Mole J. E., Tsurumachi T. Purification and partial sequence of human osteoclast-activating factor: identity with interleukin 1 beta. J Immunol. 1985 Oct;135(4):2562–2568. [PubMed] [Google Scholar]
- Dower S. K., Call S. M., Gillis S., Urdal D. L. Similarity between the interleukin 1 receptors on a murine T-lymphoma cell line and on a murine fibroblast cell line. Proc Natl Acad Sci U S A. 1986 Feb;83(4):1060–1064. doi: 10.1073/pnas.83.4.1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dower S. K., Kronheim S. R., Hopp T. P., Cantrell M., Deeley M., Gillis S., Henney C. S., Urdal D. L. The cell surface receptors for interleukin-1 alpha and interleukin-1 beta are identical. Nature. 1986 Nov 20;324(6094):266–268. doi: 10.1038/324266a0. [DOI] [PubMed] [Google Scholar]
- Dower S. K., Kronheim S. R., March C. J., Conlon P. J., Hopp T. P., Gillis S., Urdal D. L. Detection and characterization of high affinity plasma membrane receptors for human interleukin 1. J Exp Med. 1985 Aug 1;162(2):501–515. doi: 10.1084/jem.162.2.501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durum S. K., Schmidt J. A., Oppenheim J. J. Interleukin 1: an immunological perspective. Annu Rev Immunol. 1985;3:263–287. doi: 10.1146/annurev.iy.03.040185.001403. [DOI] [PubMed] [Google Scholar]
- Gowen M., Mundy G. R. Actions of recombinant interleukin 1, interleukin 2, and interferon-gamma on bone resorption in vitro. J Immunol. 1986 Apr 1;136(7):2478–2482. [PubMed] [Google Scholar]
- Gowen M., Wood D. D., Ihrie E. J., McGuire M. K., Russell R. G. An interleukin 1 like factor stimulates bone resorption in vitro. Nature. 1983 Nov 24;306(5941):378–380. doi: 10.1038/306378a0. [DOI] [PubMed] [Google Scholar]
- Hølund B., Clemmensen I., Wanning M. Sequential appearance of fibronectin and collagen fibres in experimental arthritis in rabbits. Histochemistry. 1984;80(1):39–44. doi: 10.1007/BF00492769. [DOI] [PubMed] [Google Scholar]
- Kilian P. L., Kaffka K. L., Stern A. S., Woehle D., Benjamin W. R., Dechiara T. M., Gubler U., Farrar J. J., Mizel S. B., Lomedico P. T. Interleukin 1 alpha and interleukin 1 beta bind to the same receptor on T cells. J Immunol. 1986 Jun 15;136(12):4509–4514. [PubMed] [Google Scholar]
- Krane S. M., Dayer J. M., Simon L. S., Byrne M. S. Mononuclear cell-conditioned medium containing mononuclear cell factor (MCF), homologous with interleukin 1, stimulates collagen and fibronectin synthesis by adherent rheumatoid synovial cells: effects of prostaglandin E2 and indomethacin. Coll Relat Res. 1985 Mar;5(2):99–117. doi: 10.1016/s0174-173x(85)80033-9. [DOI] [PubMed] [Google Scholar]
- Lowenthal J. W., MacDonald H. R. Binding and internalization of interleukin 1 by T cells. Direct evidence for high- and low-affinity classes of interleukin 1 receptor. J Exp Med. 1986 Oct 1;164(4):1060–1074. doi: 10.1084/jem.164.4.1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- March C. J., Mosley B., Larsen A., Cerretti D. P., Braedt G., Price V., Gillis S., Henney C. S., Kronheim S. R., Grabstein K. Cloning, sequence and expression of two distinct human interleukin-1 complementary DNAs. Nature. 1985 Jun 20;315(6021):641–647. doi: 10.1038/315641a0. [DOI] [PubMed] [Google Scholar]
- Matsushima K., Akahoshi T., Yamada M., Furutani Y., Oppenheim J. J. Properties of a specific interleukin 1 (IL 1) receptor on human Epstein Barr virus-transformed B lymphocytes: identity of the receptor for IL 1-alpha and IL 1-beta. J Immunol. 1986 Jun 15;136(12):4496–4502. [PubMed] [Google Scholar]
- McCroskery P. A., Arai S., Amento E. P., Krane S. M. Stimulation of procollagenase synthesis in human rheumatoid synovial fibroblasts by mononuclear cell factor/interleukin 1. FEBS Lett. 1985 Oct 21;191(1):7–12. doi: 10.1016/0014-5793(85)80983-2. [DOI] [PubMed] [Google Scholar]
- McGuire-Goldring M. B., Meats J. E., Wood D. D., Ihrie E. J., Ebsworth N. M., Russell R. G. In vitro activation of human chondrocytes and synoviocytes by a human interleukin-1-like factor. Arthritis Rheum. 1984 Jun;27(6):654–662. doi: 10.1002/art.1780270608. [DOI] [PubMed] [Google Scholar]
- Mizel S. B., Dayer J. M., Krane S. M., Mergenhagen S. E. Stimulation of rheumatoid synovial cell collagenase and prostaglandin production by partially purified lymphocyte-activating factor (interleukin 1). Proc Natl Acad Sci U S A. 1981 Apr;78(4):2474–2477. doi: 10.1073/pnas.78.4.2474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizel S. B., Kilian P. L., Lewis J. C., Paganelli K. A., Chizzonite R. A. The interleukin 1 receptor. Dynamics of interleukin 1 binding and internalization in T cells and fibroblasts. J Immunol. 1987 May 1;138(9):2906–2912. [PubMed] [Google Scholar]
- Pettipher E. R., Higgs G. A., Henderson B. Interleukin 1 induces leukocyte infiltration and cartilage proteoglycan degradation in the synovial joint. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8749–8753. doi: 10.1073/pnas.83.22.8749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robb R. J., Greene W. C., Rusk C. M. Low and high affinity cellular receptors for interleukin 2. Implications for the level of Tac antigen. J Exp Med. 1984 Oct 1;160(4):1126–1146. doi: 10.1084/jem.160.4.1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rupp E. A., Cameron P. M., Ranawat C. S., Schmidt J. A., Bayne E. K. Specific bioactivities of monocyte-derived interleukin 1 alpha and interleukin 1 beta are similar to each other on cultured murine thymocytes and on cultured human connective tissue cells. J Clin Invest. 1986 Sep;78(3):836–839. doi: 10.1172/JCI112649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saklatvala J., Pilsworth L. M., Sarsfield S. J., Gavrilovic J., Heath J. K. Pig catabolin is a form of interleukin 1. Cartilage and bone resorb, fibroblasts make prostaglandin and collagenase, and thymocyte proliferation is augmented in response to one protein. Biochem J. 1984 Dec 1;224(2):461–466. doi: 10.1042/bj2240461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott D. L., Wainwright A. C., Walton K. W., Williamson N. Significance of fibronectin in rheumatoid arthritis and osteoarthrosis. Ann Rheum Dis. 1981 Apr;40(2):142–153. doi: 10.1136/ard.40.2.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiozawa S., Ziff M. Immunoelectron microscopic demonstration of fibronectin in rheumatoid pannus and at the cartilage-pannus junction. Ann Rheum Dis. 1983 Jun;42(3):254–263. doi: 10.1136/ard.42.3.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sklar L. A., Hyslop P. A., Oades Z. G., Omann G. M., Jesaitis A. J., Painter R. G., Cochrane C. G. Signal transduction and ligand-receptor dynamics in the human neutrophil. Transient responses and occupancy-response relations at the formyl peptide receptor. J Biol Chem. 1985 Sep 25;260(21):11461–11467. [PubMed] [Google Scholar]
- Wood D. D., Bayne E. K., Goldring M. B., Gowen M., Hamerman D., Humes J. L., Ihrie E. J., Lipsky P. E., Staruch M. J. The four biochemically distinct species of human interleukin 1 all exhibit similar biologic activities. J Immunol. 1985 Feb;134(2):895–903. [PubMed] [Google Scholar]