Abstract
Purpose
Physical activity may influence ovarian cancer risk and outcomes through effects on ovulation, inflammatory markers and other processes. We examined associations between self-reported physical activity and ovarian cancer risk and survival in a population-based, case-control study in North Carolina.
Methods
The analyses involved 638 epithelial ovarian cancer cases and 683 controls recruited between 1999-2008. Logistic regression analyses were used to assess ovarian cancer risk in relation to reported average physical activity at various time periods. Kaplan-Meier analyses and proportional hazards modeling were used to assess associations between physical activity and survival among ovarian cancer cases.
Results
Modestly reduced risks for ovarian cancer were observed in some categories of physical activity, but there were no consistent patterns of greater reductions in risk with higher activity levels. Physical activity prior to diagnosis was not significantly related to ovarian cancer survival overall, but survival was better for women who reported >2 hours of activity/week as compared to those reporting <1 hour/week among women who were non-obese (multivariable hazard ratio=0.69, 95% CI 0.47 – 1.00)
Conclusions
Our data provide weak evidence in support of beneficial effects of physical activity on ovarian cancer risk and survival, but results should be interpreted cautiously because of the lack of a clear dose response relation with higher levels of exercise and the likely misclassification of self-reported activity.
Introduction
Physical activity has been examined as a risk factor for many types of cancer, with consistent findings of protective associations for certain types of cancer, including colon and postmenopausal breast cancer.(1-3). The literature on physical activity and ovarian cancer risk is more limited, and the evidence for a protective association is less clear than for other types of cancer. Although the majority of the studies in the literature have not reported statistically significant inverse associations between physical activity and epithelial ovarian cancer risk (4-19), a meta-analysis of twelve cohort and case-control studies reported a pooled estimate of an approximate 20% reduction in ovarian cancer risk when comparing the highest to the lowest categories of recreational physical activity.(19) Proposed mechanisms for the protective effects of physical activity on ovarian cancer include its effects on ovulation, sex hormone levels, metabolic pathways, inflammatory markers and the immune system.(19-22)
Physical activity has been associated with improved survival in breast and colorectal cancer (23-27), but there are few reports on the effect of physical activity and ovarian cancer outcomes. To our knowledge, only one published paper has described associations between physical activity and ovarian cancer survival. The investigators reported no significant associations with either recent or early life physical activity.(28) Other cross-sectional analyses on the effects of physical activity in women with an ovarian cancer diagnosis suggest that cases who engage in physical activity have a better quality of life, less fatigue, less peripheral neuropathy and improved psychosocial functioning.(29,30) Whether these benefits of physical activity translate into improved survival has not yet been studied in ovarian cancer patients.
In this report we use data from a population-based case-control study to evaluate the relationship between self-reported physical activity at different periods of life on both ovarian cancer risk and survival. Because physical activity is a modifiable risk factor, if it is shown to be associated with reduced risk for or improved survival from ovarian cancer, it could represent an intervention to reduce morbidity and mortality from this highly lethal disease.
Methods
The study population was subjects from the North Carolina Ovarian Cancer Study, a population-based, case-control study conducted between 1999 and 2008. Cases were identified through the North Carolina Central Cancer Registry using rapid case ascertainment. Eligible cases had epithelial ovarian cancer, were aged 20 to 74 years at diagnosis, had no prior history of ovarian cancer and resided in the 48-county study area. They had to be mentally competent to give informed consent and complete an interview in English. All cases underwent standardized pathology review to confirm the diagnosis. Among 1671 eligible cases, 1107 (66.2%) agreed to be in the study. Reasons for non-participation were death (n=67, 4.0%), debilitating illness (n=44, 2.6%), physician refusal (n=79, 4.7%), patient refusal (n=194, 11.6%) and inability to contact (n=180, 10.8%).
Control women from the same 48-county region were identified using random digit dialing and were frequency matched to cases by age (five-year categories) and race (Black/non-Black). As with the cases, controls had to be English-speaking and mentally competent to complete the interview. Controls were required to have at least one intact ovary and no history of ovarian cancer. Screening for eligibility could not be completed for 14% of phone numbers. Among women who passed the eligibility screening (n=2378), 73% agreed to be sent additional study information. Among these 1736 potential controls, 1053 agreed to be in the study (60.7%). Non-participation was due to refusal (n=474, 27.3%), inability to contact (n=207, 11.9%) and death (n=2, 0.1%). The protocol was approved by the Duke University Medical Center Institutional Review Board and the human subjects committees at the Central Cancer Registry and each hospital where cases were identified.
In-person interviews were conducted by nurses who obtained written informed consent, administered a questionnaire, drew a blood sample and performed anthropometric measurements (height, weight, and waist and hip circumferences). The risk factor information collected included family history of cancer, reproductive history, hormone use and lifestyle characteristics such as smoking and alcohol consumption. Physical activity was ascertained by asking each subject to report the number of hours per week participating in aerobic activities, defined as activities that raised one's heart rate or made one sweat. The amount of activity was reported for the teenage years and each age decade from the 20s to the 70s. For the present analyses, categories of physical activity were ≤1 hour per week, >1 to ≤2 hours per week, >2 to ≤3 hours per week and >3 hours per week. Physical activity questions used for this analysis were added to the survey midway through the study, and these data were available for approximately two-thirds of the study subjects (638 cases and 683 controls). The results in this paper focus on the average aerobic activity over a woman's lifetime (starting at age 20), her average activity in the previous 10 years and her activity at the point one year prior to diagnosis or interview.
For the survival analyses, information on date of death among the ovarian cancer cases was obtained from the Social Security Death Index and from the North Carolina Central Cancer Registry. The last search for deaths was performed in May 2009, which would identify deaths that occurred through January 2009, because of the lag time for deaths to appear in the index. Any woman for whom there was no report of a death from either source was presumed alive as of that date.
Comparisons of risk factor characteristics between cases and controls or between cases that survived and those who died were evaluated with chi-square tests, t-tests and logistic regression. Unconditional logistic regression models were used to calculate odds ratios (ORs) and 95 percent confidence intervals (CIs) associated with various measures of physical activity, controlling for the matching variables of age and race and other potential confounders including established ovarian cancer risk factors (oral contraceptive use, months of pregnancy, family history of ovarian cancer, menopausal status) and factors related to physical activity (body mass index (BMI) and smoking status). Kaplan-Meier plots were used to compare the survival experience of the ovarian cancer cases by levels of physical activity. Cox proportional hazards models were used to calculate hazards ratios (HRs) and 95% CIs for various levels of physical activity controlling for potential confounding variables. The proportional hazards assumption was tested using Martingale residuals and the assumption was not violated for any of the covariates included in the models.
Results
The analyses were restricted to cases with invasive epithelial ovarian cancer and those cases and controls who provided information on physical activity (638 cases and 683 controls). The distribution of levels of physical activity among cases and controls by selected characteristics is presented in Table 1. We observed statistically significant differences in reported levels of physical activity by BMI in both cases and controls (p=0.009 and 0.003, respectively), by age in cases (p=0.006) and by oral contraceptive use in controls (p=0.018). As reported previously (31), case-control comparisons in our study population showed the expected inverse associations with pregnancy and oral contraceptive use and increased risk with a family history of ovarian cancer in a first degree relative. There were no significant associations with smoking history or BMI (data not shown).
Table 1.
Reported physical activity levels at one year before diagnosis or interview for cases and controls by levels of potential confounding variables, the North Carolina Ovarian Cancer Study
| Cases | Controls | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Weekly hours of aerobic activity | Weekly hours of aerobic activity | |||||||||
| N | ≤1 hr | >1 to ≤2 | >2 to <3 | >3 | N | ≤1 hr | >1 to ≤2 | >2 to <3 | >3 | |
| Age (years) | ||||||||||
| <50 | 152 | 42.8% | 27.6% | 20.4% | 9.2% | 189 | 45.0% | 26.5% | 14.8% | 13.8% |
| 50 – 59 | 206 | 44.7% | 24.3% | 13.6% | 17.5% | 244 | 43.0% | 29.9% | 13.9% | 13.1% |
| 60 – 69 | 202 | 56.9% | 20.8% | 9.4% | 12.9% | 180 | 42.8% | 27.8% | 12.2% | 17.2% |
| 70 - 74 | 78 | 61.5% | 16.7% | 11.5% | 10.3% | 70 | 51.4% | 24.3% | 12.9% | 11.4% |
| Race | ||||||||||
| African American | 80 | 47.5% | 30.0% | 15.0% | 7.5% | 134 | 49.3% | 24.6% | 15.7% | 10.4% |
| All others | 558 | 50.5% | 22.0% | 13.4% | 14.0% | 549 | 43.2% | 28.6% | 13.1% | 15.1% |
| Pregnancy (months) | ||||||||||
| None | 106 | 50.9% | 25.5% | 13.2% | 10.4% | 60 | 46.7% | 23.3% | 16.7% | 13.3% |
| 1 to 8 | 33 | 48.5% | 24.2% | 18.2% | 9.1% | 34 | 44.1% | 26.5% | 17.6% | 11.8% |
| 9 to 18 | 235 | 45.1% | 24.3% | 15.3% | 15.3% | 255 | 38.8% | 32.2% | 16.9% | 12.2% |
| >18 | 260 | 55.0% | 21.2% | 11.9% | 11.9% | 30 | 48.5% | 25.5% | 10.3% | 15.8% |
| Oral contraceptives | ||||||||||
| None | 214 | 58.4% | 18.7% | 10.7% | 12.1% | 172 | 53.5% | 23.3% | 11.6% | 11.6% |
| <1 year | 92 | 42.4% | 26.1% | 12.0% | 19.6% | 77 | 37.7% | 31.2% | 19.5% | 11.7% |
| 1 to <5 years | 134 | 49.3% | 23.1% | 14.2% | 13.4% | 192 | 43.2% | 25.5% | 18.2% | 13.0% |
| ≥5 years | 161 | 46.0% | 28.0% | 16.1% | 9.9% | 214 | 43.0% | 30.4% | 8.4% | 18.2% |
| Menopausal status | ||||||||||
| Post | 456 | 53.1% | 22.4% | 11.4% | 13.2% | 457 | 44.4% | 28.0% | 13.1% | 14.4% |
| Pre | 174 | 43.7% | 25.9% | 18.4% | 12.1% | 220 | 43.6% | 28.2% | 14.1% | 14.1% |
| Family history of ovarian cancer in 1st degree relative |
||||||||||
| No | 595 | 50.1% | 23.4% | 13.8% | 12.8% | 663 | 43.9% | 27.9% | 14.0% | 14.2% |
| Yes | 42 | 50.0% | 19.0% | 11.9% | 19.0% | 18 | 55.6% | 27.8% | 0.0% | 16.7% |
| Body mass index | ||||||||||
| <25 | 246 | 39.8% | 26.4% | 16.7% | 17.1% | 245 | 35.1% | 31.0% | 14.7% | 19.2% |
| 25 to <30 | 171 | 54.4% | 21.1% | 11.1% | 13.5% | 198 | 42.4% | 28.3% | 17.7% | 11.6% |
| 30 to <35 | 105 | 54.3% | 23.8% | 10.5% | 11.4% | 114 | 52.6% | 23.7% | 11.4% | 12.3% |
| ≥35 | 90 | 63.3% | 17.8% | 13.3% | 5.6% | 105 | 56.2% | 27.6% | 7.6% | 8.6% |
| Smoking status | ||||||||||
| Current | 110 | 51.8% | 20.9% | 18.2% | 9.1% | 129 | 49.6% | 28.7% | 9.3% | 12.4% |
| Former | 209 | 52.6% | 20.6% | 11.0% | 15.8% | 205 | 42.9% | 24.4% | 18.0% | 14.6% |
| Never | 319 | 48.0% | 25.4% | 13.8% | 12.9% | 349 | 43.3% | 29.5% | 12.6% | 14.6% |
| Stage | ||||||||||
| I | 157 | 48.4% | 25.5% | 14.6% | 11.5% | |||||
| II | 46 | 54.3% | 15.2% | 17.4% | 13.0% | |||||
| III | 412 | 50.5% | 23.8% | 12.6% | 13.1% | |||||
| IV | 17 | 52.9% | 5.9% | 11.8% | 29.4% | |||||
| Grade | ||||||||||
| Well-differentiated | 77 | 51.9% | 28.6% | 14.3% | 5.2% | |||||
| Moderately differentiated | 166 | 52.4% | 22.3% | 12.7% | 12.7% | |||||
| Poorly differentiated | 371 | 49.9% | 22.1% | 12.9% | 15.1% | |||||
| Histology | ||||||||||
| Serous | 358 | 48.9% | 23.2% | 14.5% | 13.4% | |||||
| Mucinous | 33 | 51.5% | 24.2% | 15.2% | 9.1% | |||||
| Endometrioid | 101 | 58.4% | 19.8% | 9.9% | 11.9% | |||||
| Clear cell | 70 | 42.9% | 27.1% | 15.7% | 14.3% | |||||
| Other | 76 | 51.3% | 22.4% | 11.8% | 14.5% | |||||
Associations between categories of physical activity and ovarian cancer risk are presented in Table 2. We examined associations with reported activity one year prior to diagnosis or interview, average activity in the ten years before diagnosis or interview and average activity during adulthood. As compared to women reporting less than one hour of aerobic activity per week, women reporting 1 to <2 hours per week of activity in the year before or in the 10 years before diagnosis/interview had a statistically significant reduced risk for ovarian cancer. Most of the ORs associated with higher levels of activity tended to be less than one, however the associations were not statistically significant and there were no consistent trends of greater reductions in risk with more hours of reported activity per week. Adjustment for BMI and other ovarian cancer risk factors did not result in important changes in the ORs as compared to those adjusted only for age and race. Excluding the term for BMI from the multivariable models had minimal effects on the OR estimates.
Table 2.
Odds ratios (ORs) and 95% confidence intervals (CIs) for ovarian cancer by reported level of physical activity in different time periods before diagnosis or interview, the North Carolina Ovarian Cancer Study.
| Cases | Controls | Age and race adjusted |
Multivariable* adjusted with BMI |
Multivariable** adjusted without BMI |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | % | N | % | OR | 95% CI | OR | 95% CI | OR | 95% | |
| Aerobic activity at one year before diagnosis or interview |
||||||||||
| ≤1 hour per week | 320 | (50.2) | 303 | (44.4) | 1.00 | Referent | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 147 | (23.0) | 190 | (27.8) | 0.74 | (0.57 – 0.97) | 0.74 | (0.55 – 0.99) | 0.79 | (0.60 – 1.04) |
| >2 to ≤3 hours per week | 87 | (13.6) | 93 | (13.6) | 0.92 | (0.66 – 1.28) | 0.84 | (0.58 – 1.22) | 0.87 | (0.61 – 1.24) |
| >3 hours per week | 84 | (13.2) | 97 | (14.2) | 0.80 | (0.57 – 1.11) | 0.74 | (0.51 – 1.07) | 0.76 | (0.53 – 1.09) |
| Average aerobic activity in 10 years before diagnosis or interview |
||||||||||
| ≤1 hour per week | 223 | (35.0) | 209 | (30.6) | 1.00 | Referent | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 177 | (27.7) | 235 | (34.4) | 0.72 | (0.55 – 0.94) | 0.71 | (0.53 – 0.96) | 0.72 | (0.54 – 0.96) |
| >2 to ≤3 hours per week | 128 | (20.1) | 121 | (17.7) | 1.02 | (0.74 – 1.40) | 0.92 | (0.65 – 1.29) | 0.99 | (0.71 – 1.38) |
| >3 hours per week | 110 | (17.2) | 118 | (17.3) | 0.87 | (0.63 – 1.20) | 0.80 | (0.56 – 1.15) | 0.80 | (0.57 – 1.13) |
| Average aerobic activity during adulthood (age ≥20 years) |
||||||||||
| ≤1 hour per week | 75 | (11.8) | 72 | (10.5) | 1.00 | Referent | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 219 | (34.3) | 211 | (30.9) | 0.97 | (0.66 – 1.41) | 1.05 | (0.69 – 1.59) | 0.99 | (0.67 – 1.48) |
| >2 to ≤3 hours per week | 180 | (28.2) | 238 | (34.8) | 0.71 | (0.48 – 1.03) | 0.74 | (0.48 – 1.13) | 0.60 | (0.46 – 1.03) |
| >3 hours per week | 164 | (25.7) | 162 | (23.7 | 0.95 | (0.64 – 1.40) | 0.94 | (0.61 – 1.45) | 0.90 | (0.60 – 1.37) |
Adjusted for age, race, body mass index one year before diagnosis, menopausal status, smoking status, duration of oral contraceptive use, months of pregnancy and history of ovarian cancer in a first degree relative.
Adjusted for all variables above except body mass index.
Duration of follow-up for the ovarian cancer cases ranged from 3 to 111 months, with a median follow-up time of 38.5 months. There were 238 deaths among the 638 women with ovarian cancer. A comparison of characteristics of ovarian cancer cases who survived and those who died is presented in Table 3. The mean age at diagnosis among the women who died was two years older than the women who were still alive at the last follow-up. As expected, higher stage and less differentiated grade were strongly predictive of mortality. Survival was better for women who had cancers with mucinous, endometrioid or clear cell histologies as compared to those with serous or other histologies. Survival was not significantly associated with oral contraceptive use, family history of ovarian cancer, BMI or smoking status. There also were no statistically significant associations with selected co-morbid conditions including diabetes, thyroid conditions, depression and prior cancer diagnoses.
Table 3.
Selected characteristics of ovarian cancer cases by survival status, the North Carolina Ovarian Cancer Study
| Deceased (N=238) |
Alive (N=400) |
|||||
|---|---|---|---|---|---|---|
| Mean | (SD) | Mean | (SD) | p-value | ||
| Age at diagnosis (years) | 58.3 | (10.2) | 56.3 | (10.5) | 0.018 | |
| BMI (kg/m2) | 27.9 | (6.7) | 28.1 | (7.0) | 0.677 | |
| Mean time to death/duration of follow- up (months) |
32.9 | (17.9) | 51.5 | (24.4) | <0.001 | |
| N | (%) | N | (%) | HR* | 95% CI | |
|
| ||||||
| Stage | ||||||
| I | 19 | (8.1) | 138 | (34.9) | 1.00 | Referent |
| II | 9 | (3.8) | 37 | (9.3) | 1.28 | (0.62 – 2.61) |
| III | 198 | (83.9) | 214 | (54.0) | 5.29 | (3.62 – 7.72) |
| IV | 10 | (4.2) | 7 | (1.8) | 6.83 | (3.83 – 12.18) |
| Missing | 2 | 4 | ||||
| Grade | ||||||
| Well-differentiated | 10 | (4.3) | 67 | (17.5) | 1.00 | Referent |
| Moderately differentiated | 65 | (28.0) | 101 | (26.4) | 2.54 | (1.66 – 3.89) |
| Poorly differentiated/undifferentiated | 157 | (67.7) | 214 | (56.0) | 2.49 | (1.65 – 3.74) |
| Missing | 6 | 18 | ||||
| Histology | ||||||
| Serous | 156 | (65.5) | 202 | (50.5) | 1.00 | Referent |
| Mucinous | 5 | (2.1) | 28 | ( 7.0) | 0.27 | (0.13 – 0.57) |
| Endometrioid | 22 | (9.2) | 79 | (19.8) | 0.42 | (0.29 – 0.60) |
| Clear cell | 20 | (8.4) | 50 | (12.5) | 0.52 | (0.34 – 0.79) |
| Other | 35 | (14.7) | 41 | (10.3) | 0.93 | (0.69 – 1.26) |
| Months of pregnancy | ||||||
| Never pregnant | 32 | (13.5) | 74 | (18.6) | 1.00 | Referent |
| 1 – 8 | 7 | (3.0) | 26 | (6.5) | 0.88 | (0.48 – 1.59) |
| 9 – 18 | 90 | (38.0) | 145 | (36.5) | 1.23 | (0.87 – 1.73) |
| >18 | 108 | (45.6) | 152 | (38.3) | 1.25 | (0.89 – 1.75) |
| Missing | 1 | 3 | ||||
| Oral contraceptive use (years) | ||||||
| Never | 84 | (35.4) | 130 | (32.5) | 1.00 | Referent |
| <1 year | 33 | (13.9) | 59 | (14.8) | 1.01 | (0.72 – 1.42) |
| 1 to <5 years | 51 | (21.5) | 83 | (20.8) | 1.05 | (0.79 – 1.40) |
| ≥5 years | 59 | (24.9) | 102 | (25.5) | 0.99 | (0.75 – 1.31) |
| Unknown duration | 10 | (4.2) | 26 | (6.5) | ||
| Missing | 1 | 0 | ||||
| Menopausal status | ||||||
| Post-menopausal | 175 | (74.2) | 281 | (71.3) | 1.00 | Referent |
| Pre-menopausal | 61 | (25.8) | 113 | (28.7) | 1.44 | (1.01 – 2.07) |
| Missing | 2 | 6 | ||||
| Family history of ovarian cancer in 1st degree relative |
||||||
| No | 220 | (92.8) | 375 | (93.8) | 1.00 | Referent |
| Yes | 17 | (7.2) | 25 | ( 6.3) | 1.20 | (0.79 – 1.82) |
| Missing | 1 | 0 | ||||
| Body mass index (kg/m2) one year before diagnosis or interview |
||||||
| <25 | 95 | (42.2) | 151 | (39.0) | 1.00 | Referent |
| 25 - <30 | 60 | (26.7) | 111 | (28.7) | 1.03 | (0.79 – 1.35) |
| 30 - <35 | 37 | (16.4) | 68 | (17.6) | 1.14 | (0.84 – 1.54) |
| ≥35 | 33 | (14.7) | 57 | (14.7) | 1.07 | (0.76 – 1.50) |
| Missing | 13 | 13 | ||||
| Smoking status | ||||||
| Current smoker | 41 | (17.2) | 69 | (17.3) | 1.00 | Referent |
| Former smoker | 75 | (31.5) | 134 | (33.5) | 0.93 | (0.66 – 1.30) |
| Never smoker | 122 | (51.3) | 197 | (49.3) | 1.06 | (0.78 – 1.42) |
| Missing | ||||||
| Selected co-morbid conditions | ||||||
| Insulin-dependent diabetes | ||||||
| No | 232 | (97.5) | 390 | (97.5) | 1.00 | Referent |
| Yes | 6 | (2.5) | 10 | ( 2.5) | 1.62 | (0.87 – 3.01) |
| Non-insulin dependent diabetes | ||||||
| No | 214 | (89.9) | 375 | (93.8) | 1.00 | Referent |
| Yes | 24 | (10.1) | 25 | (6.3) | 1.35 | (0.96 – 1.91) |
| Hyperthyroidism | ||||||
| No | 228 | (95.8) | 386 | (96.5) | 1.00 | Referent |
| Yes | 10 | (4.2) | 14 | (3.5) | 1.02 | (0.57 – 1.83) |
| Hypothyroidism | ||||||
| No | 210 | (88.6) | 340 | (85.0) | 1.00 | Referent |
| Yes | 27 | (11.4) | 60 | (15.0) | 1.02 | (0.57 – 1.83) |
| Missing | 1 | 0 | ||||
| Depression requiring treatment | ||||||
| No | 181 | (76.1) | 292 | (73.0) | 1.00 | Referent |
| Yes | 57 | (23.9) | 108 | (27.0) | 0.99 | (0.78 – 1.27) |
| Prior history of cancer | ||||||
| No | 202 | (84.9) | 324 | (81.2) | 1.00 | Referent |
| Yes | 36 | (15.1) | 75 | (18.8) | 1.03 | (0.77 – 1.38) |
| Missing | 0 | 1 | ||||
ORs adjusted for age and race
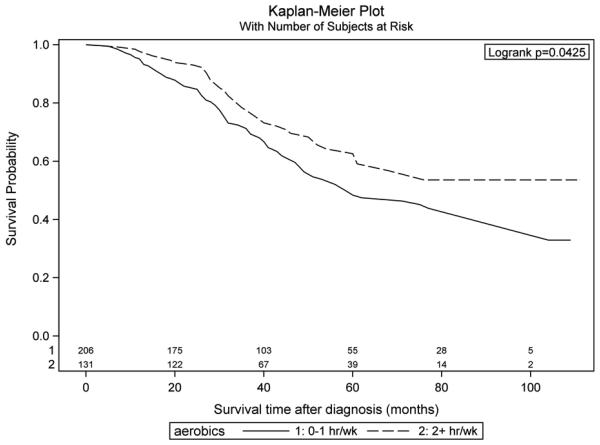
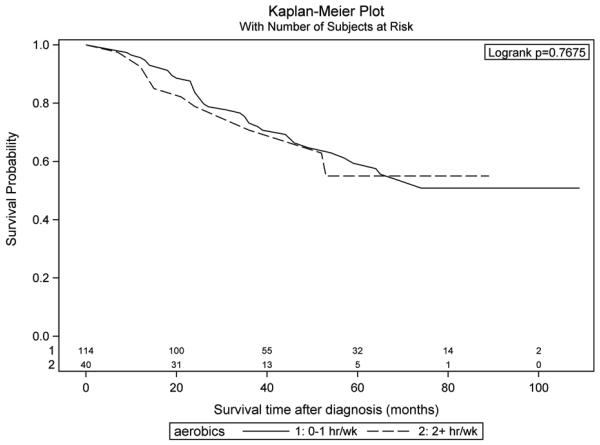
In Table 4, we present hazard ratios by level of reported physical activity at one year before diagnosis or interview, average activity in the 10 years before diagnosis or interview and average activity during adulthood. Overall, we found no statistically significant associations between physical activity and survival for any of these time periods. In further analyses, we examined the associations between physical activity at one year before diagnosis stratifying by BMI. In Figures 1 and 2, we depict Kaplan Meier survival curves comparing women with <1 hour/week of activity at one year before diagnosis to those with >2 hours/week, stratifying by reported BMI one year before diagnosis. Among women with a BMI<30, those with <1 hour/week of reported physical activity had significantly poorer survival than those reporting >2 hours/week of activity (Figure 1, p=0.043 by log-rank test), whereas among women with a BM≥30, there was no association between physical activity and ovarian cancer survival (Figure 2, p=0.77). The significant association among women with a BMI<30 that we observed in the unadjusted Kaplan Meier analysis also was observed in multivariable analyses controlling for age, race, menopausal status, smoking status, first degree family history of ovarian cancer, TNM stage and grade. The hazard ratio associated with >2 hours/week of aerobic activity was 0.69 (95% CI 0.47 – 1.00) compared to those with <1 hour/week of activity among the women with a BMI<30.
Table 4.
Hazard ratios (HRs) and 95% confidence intervals (CIs) for overall survival by reported level of physical activity.
| Cases | Controls | Age- and race- adjusted |
Multivariable- adjusted* |
|||||
|---|---|---|---|---|---|---|---|---|
| N | (%) | N | (%) | HR | 95% CI | HR | 95% CI | |
| Aerobic activity one year before diagnosis or interview |
||||||||
| ≤1 hour per week | 320 | (50.2) | 303 | (44.4) | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 147 | (23.0) | 190 | (27.8) | 0.90 | (0.65 – 1.24) | 0.94 | (0.67 – 1.32) |
| >2 to ≤3 hours per week | 87 | (13.6) | 93 | (13.6) | 0.79 | (0.52 – 1.20) | 0.74 | (0.46 – 1.18) |
| >3 hours per week | 84 | (13.2) | 97 | (14.2) | 0.83 | (0.55 – 1.26) | 0 . 9 0 | (0.58 – 1.40) |
| Average aerobic activity in 10 years before diagnosis or interview |
||||||||
| ≤1 hour per week | 223 | (35.0) | 209 | (30.6) | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 177 | (27.7) | 235 | (34.4) | 1.21 | (0.89 – 1.65) | 1.24 | (0.89 – 1.72) |
| >2 to ≤3 hours per week | 128 | (20.1) | 121 | (17.7) | 1.00 | (0.69 – 1.45) | 0.90 | (0.60 – 1.36) |
| >3 hours per week | 110 | (17.2) | 118 | (17.3) | 0.86 | (0.58 – 1.28) | 0.90 | (0.59 – 1.37) |
| Average aerobic activity during adulthood (age ≥20 years) |
||||||||
| ≤1 hour per week | 75 | (11.8) | 72 | (10.5) | 1.00 | Referent | 1.00 | Referent |
| >1 to ≤2 hours per week | 219 | (34.3) | 211 | (30.9) | 1.01 | (0.67 – 1.52) | 1.04 | (0.67 – 1.62) |
| >2 to ≤3 hours per week | 180 | (28.2) | 238 | (34.8) | 1.04 | (0.68 – 1.59) | 1.11 | (0.70 – 1.75) |
| >3 hours per week | 164 | (25.7) | 162 | (23.7) | 0.93 | (0.60 – 1.46) | 0.89 | (0.54 – 1.45) |
Adjusted for age, race, body mass index one year before diagnosis, menopausal status, smoking status, duration of oral contraceptive use, months of pregnancy and history of ovarian cancer in a first degree relative
Figure 1.
Survival among ovarian cancer cases by level of reported aerobic physical activity at one year before diagnosis, cases with body mass index (BMI) <30kg/m2.
Figure 2.
Survival among ovarian cancer cases by level of reported aerobic physical activity at one year before diagnosis, cases with body mass index (BMI) ≥30kg/m2.
Discussion
Our results provide weak evidence in support of a reduced risk of ovarian cancer among women who engaged in aerobic physical activity as compared to those who reported less than one hour of activity per week. Although most of the ORs associated with higher levels of physical activity were less than one, statistically significant associations were identified only for women reporting one to two hours of weekly activity in either the year before or the ten years before diagnosis. There was no clear dose response relation between the amount of physical activity and risk for ovarian cancer. The magnitude of the ORs for ovarian cancer risk in the present study, while not statistically significant, are consistent with a meta-analysis conducted by Olsen et al. (19) in which they reported a pooled relative risk of 0.81 (95% CI , 0.72 – 0.92) when comparing the highest to lowest categories of recreational physical activity. It is noteworthy that among the individual studies included in the meta-analysis, the majority showed modest inverse associations with physical activity that were not statistically significant. In additional studies published after the meta-analysis, a case-control study (4) reported a significant inverse relationship among women with the highest frequency and intensity of physical activity whereas a cohort study (32) reported no association with either vigorous or moderate physical activity.
The lack of statistically significant associations between physical activity and ovarian cancer risk in most studies may reflect that the risk reduction associated with higher levels of physical activity is modest and some of the studies may have had inadequate power to detect an association. It also may be a reflection of the difficulty in measuring physical activity in relation to cancer risk, where the most relevant exposure period may be many years before the actual diagnosis of cancer. An individual's physical activity is likely to vary across seasons of the year and over one's lifespan. Many epidemiologic studies, including the North Carolina Ovarian Cancer Study, asked study participants to give an estimate of average physical activity in various periods of life, a task that is challenging and fraught with error. In addition, the questions used to assess physical activity vary in detail across studies, with various surveys obtaining more or less detail about the type of activity, intensity and frequency.
The effect of the misclassification of physical activity would depend in part on whether it differed by case-control status. It is impossible to determine with certainty whether there is differential misclassification between cases and controls, however one approach is to compare findings from case-control studies to those from cohort studies, where report of physical activity precedes the diagnosis and would not be subject to recall bias related to case status. In the meta-analysis by Olsen, et al.(19), the pooled estimate from the case-control studies (OR= 0.79, 95% CI 0.70 – 0.85) was very similar to the pooled estimate from the cohort studies (OR=0.81, 95% CI 0.72 – 0.92). The similarity of these estimates argues against the ORs in case-control studies being biased due to differential misclassification of physical activity. Instead, it suggests that the inevitable misclassification of physical activity is likely to be non-differential by case-control status. In the situation of a dichotomous variable, non-differential misclassification usually results in an underestimate of the true association however misclassification of a multilevel variable like physical activity would not necessarily result in a bias towards the null. Therefore it cannot be concluded with certainty that misclassification of physical activity will result in an attenuation of its true association with ovarian cancer.
A limitation of our study is its low response rates, particularly among control women. The lack of information from non-participants precludes us from determining how non-response may have affected our findings, however our study population does show the expected associations with well-established risk factors for ovarian cancer. In addition, when we compared selected characteristics of the controls to comparable statistics reported by the North Carolina Behavioral Risk Factor Surveillance Study, we found similar prevalences of factors such as smoking, hysterectomy status and overweight, suggesting that the controls were a fairly representative sample of the North Carolina population despite the sub-optimal response rates.
Our North Carolina study population differs from other studies of ovarian cancer in its racial composition, with African American women comprising approximately 10% of the population. We did find that African American women reported levels of physical activity that were somewhat lower, although not statistically significant different from white women. Our analyses controlled for race as a potential confounding variable.
Our analyses of physical activity among ovarian cancer cases found evidence of improved survival for women who engaged in greater than two hours of activity per week as compared to those who reported less than one hour per week, but only among women who were not obese (BMI<30kg/m2). In the only other published study of physical activity and ovarian cancer survival, a population-based, prospective study conducted in Sweden, there was no overall association between physical activity during childhood, young adulthood or recent years and survival after ovarian cancer, but there was a statistically significant survival advantage among women with Stage I or II disease who were in the highest category of activity at ages 18 to 30.(28)
Although the literature on physical activity and ovarian cancer survival is very limited, studies of other cancer types that have evaluated physical activity both before and after diagnosis suggest that higher levels of physical activity may improve outcomes in people diagnosed with cancer. (23-26,33) Higher levels of self-reported physical activity and measures of cardiorespiratory fitness have been associated with improved survival from breast, colorectal and lung cancer.(23-26,33) Further studies among women with ovarian cancer, including intervention studies designed to improve fitness after diagnosis, are warranted.
An obvious limitation of both our analysis and the Swedish survival study is that the physical activity data were collected shortly after the time of the ovarian cancer diagnosis and no information was obtained about changes in physical activity that occurred after diagnosis. One might presume that the women who were most physically active before being diagnosed with cancer would be more likely to continue to exercise, but individual variation is expected. Some women who were physically active before their cancer diagnosis may have been unable to continue to exercise at the same level afterwards, whereas the cancer diagnosis could have served as an impetus to improve lifestyle habits among some women who were inactive before their diagnosis. It is also likely that the ability to exercise after diagnosis may be related to stage of disease, and women with more advanced disease and a poorer prognosis may be less likely to engage in physical activity. The uncertainty about how pre-diagnostic exercise patterns relate to post-diagnosis physical activity underscores the limitation of having reports of physical activity only around the time of diagnosis.
Our survival analyses also are limited by the incomplete information on co-morbid conditions, which could be associated with both physical activity and risk for death. Our analyses were based on deaths from all causes, of which the vast majority would be expected to be ovarian cancer-related, however there is the possibility of confounding by co-morbid conditions. A prospective study that ascertains changes in physical activity in the months and years following an ovarian cancer diagnosis and has more specific information on other conditions that contribute to risk for mortality could provide more definitive evidence on the effect of physical activity on ovarian cancer survival.
Overall, our findings are suggestive of modest benefits of physical activity in relation to both ovarian cancer risk and survival. While the interpretation of our results must be tempered by considering the inevitable misclassification of physical activity in retrospective epidemiologic studies, there are biological reasons that support the plausibility of exercise reducing cancer risk and improving survival.(20)
The factors most consistently associated with ovarian cancer risk, oral contraceptive use and greater number of pregnancies, are hypothesized to reduce risk by reducing the number of ovulatory cycles.(34) It is well-established that physical activity affects menstrual patterns. Oligomenorrhea or amenorrhea is commonly reported in competitive athletes, and increases in cycle length have been reported among women engaging in less vigorous activity who continue to cycle regularly.(35,36) Although relatively few women in our study reported activity levels that would likely result in amenorrhea, more subtle effects of moderate physical activity on menstrual cycle length could result in fewer lifetime ovulatory cycles and reduce ovarian cancer risk.
Effects on inflammatory processes and immune response also could account for reduced risk of or better prognosis after ovarian cancer in more physically active women. Epithelial inflammation has been proposed as a common pathway linking several established risk factors for ovarian cancer including ovulation, endometriosis and talc exposure.(37) In some studies, physical activity has been associated with reduced levels of inflammatory markers such as C-reactive protein and tumor necrosis factor (TNF)-α.(20) Similarly, markers of immune function have been reported to be enhanced among those engaging in moderate levels of physical activity. (20)
The data from the present study add to the body of literature on physical activity ovarian cancer risk and survival. While some of the findings are weakly supportive of inverse associations with both risk and survival, our study was limited by the many challenges of assessing physical activity in epidemiologic studies, including retrospective assessment of activity over a lifetime, inaccurate recall and lack of objective measures of physical fitness. The literature on the effects of physical activity on ovarian cancer risk and survival is suggestive, but not conclusive, that there are beneficial effects. Although it is not clear what it is about exercise that will specifically benefit ovarian cancer patients, the general health benefits that accompany physical activity suggest that recommendations to engage in exercise training to the extent possible would be reasonable and prudent for ovarian cancer patients.
Acknowledgment
This work was supported by National Cancer Institute Grant R01-CA76016.
List of Abbreviations
- BMI
body mass index
- CI
confidence interval
- HR
hazard ratio
- OR
odds ratio
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Tardon A, Lee WJ, Delgado-Rodriguez M, et al. Leisure-time physical activity and lung cancer: a meta-analysis. Cancer Causes Control. 2005;16:389–97. doi: 10.1007/s10552-004-5026-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wolin KY, Yan Y, Colditz GA, et al. Physical activity and colon cancer prevention: a metaanalysis. Br J Cancer. 2009;100:611–6. doi: 10.1038/sj.bjc.6604917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Monninkhof EM, Elias SG, Vlems FA, et al. Physical activity and breast cancer: a systematic review. Epidemiology. 2007;18:137–57. doi: 10.1097/01.ede.0000251167.75581.98. [DOI] [PubMed] [Google Scholar]
- 4.Rossing MA, Cushing-Haugen KL, Wicklund KG, et al. Recreational physical activity and risk of epithelial ovarian cancer. Cancer Causes Control. 2010;10:485–91. doi: 10.1007/s10552-009-9479-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schnohr P, Gronbaek M, Petersen L, et al. Physical activity in leisure-time and risk of cancer: 14-year follow-up of 28,000 Danish men and women. Scand J Public Health. 2005;33:244–9. doi: 10.1080/14034940510005752. [DOI] [PubMed] [Google Scholar]
- 6.Weiderpass E, Margolis KL, Sandin S, et al. Prospective study of physical activity in different periods of life and the risk of ovarian cancer. Int J Cancer. 2006;118:3153–60. doi: 10.1002/ijc.21800. [DOI] [PubMed] [Google Scholar]
- 7.Lahmann PH, Friedenreich C, Schulz M, et al. Physical activity and ovarian cancer risk: the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiol Biomarkers Prev. 2009;18:351–4. doi: 10.1158/1055-9965.EPI-08-0958. [DOI] [PubMed] [Google Scholar]
- 8.Zhang M, Lee AH, Binns CW. Physical activity and epithelial ovarian cancer risk: a case-control study in China. Int J Cancer. 2003;105:838–43. doi: 10.1002/ijc.11165. [DOI] [PubMed] [Google Scholar]
- 9.Hannan LM, Leitzmann MF, Lacey JV, Jr., et al. Physical activity and risk of ovarian cancer: a prospective cohort study in the United States. Cancer Epidemiol Biomarkers Prev. 2004;13:765–70. [PubMed] [Google Scholar]
- 10.Mink PJ, Folsom AR, Sellers TA, et al. Physical activity, waist-to-hip ratio, and other risk factors for ovarian cancer: a follow-up study of older women. Epidemiology. 1996;7:38–45. doi: 10.1097/00001648-199601000-00008. [DOI] [PubMed] [Google Scholar]
- 11.Bertone ER, Willett WC, Rosner BA, et al. Prospective study of recreational physical activity and ovarian cancer. J Natl Cancer Inst. 2001;93:942–8. doi: 10.1093/jnci/93.12.942. [DOI] [PubMed] [Google Scholar]
- 12.Bertone ER, Newcomb PA, Willett WC, et al. Recreational physical activity and ovarian cancer in a population-based case-control study. Int J Cancer. 2002;99:431–6. doi: 10.1002/ijc.10365. [DOI] [PubMed] [Google Scholar]
- 13.Biesma RG, Schouten LJ, Dirx MJ, et al. Physical activity and risk of ovarian cancer: results from the Netherlands Cohort Study (The Netherlands) Cancer Causes Control. 2006;17:109–15. doi: 10.1007/s10552-005-0422-3. [DOI] [PubMed] [Google Scholar]
- 14.Riman T, Dickman PW, Nilsson S, et al. Some life-style factors and the risk of invasive epithelial ovarian cancer in Swedish women. Eur J Epidemiol. 2004;19:1011–9. doi: 10.1007/s10654-004-1633-8. [DOI] [PubMed] [Google Scholar]
- 15.Anderson JP, Ross JA, Folsom AR. Anthropometric variables, physical activity, and incidence of ovarian cancer: The Iowa Women's Health Study. Cancer. 2004;100:1515–21. doi: 10.1002/cncr.20146. [DOI] [PubMed] [Google Scholar]
- 16.Pan SY, Ugnat AM, Mao Y. Physical activity and the risk of ovarian cancer: a case-control study in Canada. Int J Cancer. 2005;117:300–7. doi: 10.1002/ijc.21157. [DOI] [PubMed] [Google Scholar]
- 17.Patel AV, Rodriguez C, Pavluck AL, et al. Recreational physical activity and sedentary behavior in relation to ovarian cancer risk in a large cohort of US women. Am J Epidemiol. 2006;163:709–16. doi: 10.1093/aje/kwj098. [DOI] [PubMed] [Google Scholar]
- 18.Cottreau CM, Ness RB, Kriska AM. Physical activity and reduced risk of ovarian cancer. Obstet Gynecol. 2000;96:609–14. doi: 10.1016/s0029-7844(00)00972-8. [DOI] [PubMed] [Google Scholar]
- 19.Olsen CM, Bain CJ, Jordan SJ, et al. Recreational physical activity and epithelial ovarian cancer: a case-control study, systematic review, and meta-analysis. Cancer Epidemiol Biomarkers Prev. 2007;16:2321–30. doi: 10.1158/1055-9965.EPI-07-0566. [DOI] [PubMed] [Google Scholar]
- 20.McTiernan A. Mechanisms linking physical activity with cancer. Nat Rev Cancer. 2008;8:205–11. doi: 10.1038/nrc2325. [DOI] [PubMed] [Google Scholar]
- 21.Fair AM, Montgomery K. Energy balance, physical activity, and cancer risk. Methods Mol Biol. 2009;472:57–88. doi: 10.1007/978-1-60327-492-0_3. [DOI] [PubMed] [Google Scholar]
- 22.Shephard RJ, Shek PN. Associations between physical activity and susceptibility to cancer: possible mechanisms. Sports Med. 1998;26:293–315. doi: 10.2165/00007256-199826050-00002. [DOI] [PubMed] [Google Scholar]
- 23.Irwin ML, Smith AW, McTiernan A, et al. Influence of pre- and postdiagnosis physical activity on mortality in breast cancer survivors: the health, eating, activity, and lifestyle study. J Clin Oncol. 2008;26:3958–64. doi: 10.1200/JCO.2007.15.9822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Holmes MD, Chen WY, Feskanich D, et al. Physical activity and survival after breast cancer diagnosis. JAMA. 2005;293:2479–86. doi: 10.1001/jama.293.20.2479. [DOI] [PubMed] [Google Scholar]
- 25.Holick CN, Newcomb PA, Trentham-Dietz A, et al. Physical activity and survival after diagnosis of invasive breast cancer. Cancer Epidemiol Biomarkers Prev. 2008;17:379–86. doi: 10.1158/1055-9965.EPI-07-0771. [DOI] [PubMed] [Google Scholar]
- 26.Meyerhardt JA, Giovannucci EL, Holmes MD, et al. Physical activity and survival after colorectal cancer diagnosis. J Clin Oncol. 2006;24:3527–34. doi: 10.1200/JCO.2006.06.0855. [DOI] [PubMed] [Google Scholar]
- 27.Meyerhardt JA, Heseltine D, Niedzwiecki D, et al. Impact of physical activity on cancer recurrence and survival in patients with stage III colon cancer: findings from CALGB 89803. J Clin Oncol. 2006;24:3535–41. doi: 10.1200/JCO.2006.06.0863. [DOI] [PubMed] [Google Scholar]
- 28.Yang L, Klint A, Lambe M, et al. Predictors of ovarian cancer survival: a population-based prospective study in Sweden. Int J Cancer. 2008;123:672–9. doi: 10.1002/ijc.23429. [DOI] [PubMed] [Google Scholar]
- 29.Stevinson C, Faught W, Steed H, et al. Associations between physical activity and quality of life in ovarian cancer survivors. Gynecol Oncol. 2007;106:244–50. doi: 10.1016/j.ygyno.2007.03.033. [DOI] [PubMed] [Google Scholar]
- 30.Stevinson C, Steed H, Faught W, et al. Physical activity in ovarian cancer survivors: associations with fatigue, sleep, and psychosocial functioning. Int J Gynecol Cancer. 2009;19:73–8. doi: 10.1111/IGC.0b013e31819902ec. [DOI] [PubMed] [Google Scholar]
- 31.Moorman PG, Palmieri RT, Akushevich L, et al. Ovarian cancer risk factors in African-American and White women. Am J Epidemiol. 2009;170:598–606. doi: 10.1093/aje/kwp176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Leitzmann MF, Koebnick C, Moore SC, et al. Prospective study of physical activity and the risk of ovarian cancer. Cancer Causes Control. 2009;20:765–73. doi: 10.1007/s10552-008-9291-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kohman LWD, Herndon JE, et al. Association between cardiorespiratory fitness and overall survival in operable lung cancer patients: ancillary analysis of protocol 9238. J Clin Oncol. 2009;27 Abstr 7518. [Google Scholar]
- 34.Fleming JS, Beaugie CR, Haviv I, et al. Incessant ovulation, inflammation and epithelial ovarian carcinogenesis: revisiting old hypotheses. Mol Cell Endocrinol. 2006;247:4–21. doi: 10.1016/j.mce.2005.09.014. [DOI] [PubMed] [Google Scholar]
- 35.Henley K, Vaitukaitis JL. Exercise-induced menstrual dysfunction. Annu Rev Med. 1988;39:443–51. doi: 10.1146/annurev.me.39.020188.002303. [DOI] [PubMed] [Google Scholar]
- 36.Sternfeld B, Jacobs MK, Quesenberry CP, Jr., et al. Physical activity and menstrual cycle characteristics in two prospective cohorts. Am J Epidemiol. 2002;156:402–9. doi: 10.1093/aje/kwf060. [DOI] [PubMed] [Google Scholar]
- 37.Ness RB, Cottreau C. Possible role of ovarian epithelial inflammation in ovarian cancer. J Natl Cancer Inst. 1999;91:1459–67. doi: 10.1093/jnci/91.17.1459. [DOI] [PubMed] [Google Scholar]